Abstract
First introduced in 1976, 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography (PET) has become an indispensable tool for diagnosis and prognostic evaluation of tumors, heart disease, as well as other conditions, including inflammation and infection. Because 18F-FDG can accurately reflect the glucose metabolism level of organs and tissues, it is known as a “century molecule” and is currently the main agent for PET imaging. The degree of 18F-FDG uptake by cells is related to both the rate of glucose metabolism and glucose transporter expression. These, in turn, are strongly influenced by hypoxia, in which cells meet their energy needs through glycolysis, and 18F-FDG uptake increased due to hypoxia. 18F-FDG uptake is a complex process, and hypoxia may be one of the fundamental driving forces. The correct interpretation of 18F-FDG uptake in PET imaging can help clinics make treatment decisions more accurately and effectively. In this article, we review the application of 18F-FDG PET in tumors, myocardium, and inflammation. We discuss the relationship between 18F-FDG uptake and hypoxia, the possible mechanism of 18F-FDG uptake caused by hypoxia, and the associated clinical implications.
Introduction
18F-fluorodeoxyglucose (18F-FDG) positron emission tomography (PET) imaging is being increasingly utilized in the clinic to follow the metabolic changes in tumor tissue at the molecular level, yielding information on tumor diagnosis and staging and monitoring for recurrence, including metastatic spread and response to therapy. 1,2 In cardiology, 18F-FDG PET myocardial metabolism imaging is considered the gold standard for detecting the myocardial viability. Furthermore, direct myocardial ischemia imaging can overcome some limitations of stress-rest perfusion imaging. 3 Finally, due to the high sensitivity and spatial resolution, 18F-FDG PET is becoming more common in the imaging of inflammation and infectious lesions.
However, some exceptions may occur with clinical imaging; some malignant tumors may have low 18F-FDG uptake and benign lesions with high uptake. 4 –6 In severe multivessel coronary artery disease, myocardial perfusion imaging and resting metabolic imaging may be “normal”. However, direct imaging of exercise-induced myocardial ischemia imaging may reveal the presence of myocardial ischemia. 7 In clinical practice, it is observed that 18F-FDG uptake is low in the acute and high chronic phase of inflammation. 8,9 Plausible explanations to these seemingly contradictory problems are still pending.
Studies have demonstrated that 18F-FDG uptake is not intrinsic to cancer, ischemic myocardium, or inflammation, but hypoxia may be related to it, resulting in altered energy metabolism by increased anaerobic glycolysis. 3,9,10 In 1861, Louis Pasteur found that while yeast cells cultured in anaerobic conditions had reduced growth, their glucose consumption significantly increased, the so-called Pasteur Effect. So, what is the relationship between 18F-FDG uptake and hypoxia and its specific mechanism? Those answers will aid in a correct interpretation of 18F-FDG lesion-uptake in PET imaging, resulting in more accurate and useful clinical treatment decisions.
In this article, we review the application of 18F-FDG PET in cancer, heart, and inflammation and discuss the relationship between 18F-FDG uptake and hypoxia, the possible mechanism of 18F-FDG uptake caused by hypoxia, and their clinical implications.
18F-FDG PET Use in the Imaging of Tumors, Myocardial Ischemia and Inflammation
18F-FDG is the essential imaging agent for PET imaging at present. Like glucose, deoxyglucose and fluorodeoxyglucose are imported into the cell by glucose transporters and phosphorylated by hexokinase. However, in its phosphorylated form, it is not a substrate for the next enzyme in the glycolytic pathway (phosphoglucose isomerase) and thus is not further metabolized. Because of phosphorylation, fluorodeoxyglucose-6-phosphate is membrane-impermeable and thus trapped intracellularly. 11 The accumulation of 18F-FDG allows a PET image to report on the local glucose metabolism in vivo.
The uncontrolled proliferation of tumor cells leads to high glucose consumption, and consequently, intravenous injection of 18F-FDG results in most tumor lesions showing high 18F-FDG uptake. Therefore, 18F-FDG PET imaging is used to detect primary and metastatic lesions at an early stage. Also, 18F-FDG is a promising direct imaging agent for exercise-induced myocardial ischemia. The difference in glucose uptake between normal and ischemic myocardium allows 18F-FDG as a “hot spot” imaging agent for myocardial ischemia imaging in patients with known or suspected coronary artery disease. 3,12 –14 Furthermore, inflammatory lesion glucose metabolism measured by 18F-FDG uptake may result in early diagnosis of the disease, prevention of related complications, and early treatment.
18F-FDG PET Cancer Imaging and Hypoxia
Increased glucose demand regarded as one of the fundamental characteristics of tumors, and this is the rationale for the use of 18F-FDG PET in oncology. However, not all malignant tumors show high levels of 18F-FDG uptake, and conversely, some benign lesions show significant tracer uptake. More and more evidence shows that the accumulation of 18F-FDG in tumors depends on the microenvironment. 15,16 18F-FDG accumulates preferentially in tumor cells with low proliferation and hypoxia, but less so in well perfused, and thus highly proliferative, areas of the tumor. 10,17,18
Hypoxia is a common feature of solid tumors, closely related to radiotherapy and chemotherapy resistance, tumor metastasis, and prognosis. 19,20 Hypoxia arises through the rate of oxygen consumption by cancer exceeding the oxygen delivery rate by the vasculature, which is often irregular, with chaotic blood flow. The unchecked proliferation of tumor cells leads to elevated oxygen demand, as tumor tissue outgrows the existing vasculature. 21,22 Studies have shown that hypoxic conditions play a crucial role in inducing high 18F-FDG accumulation, resulting in anaerobic glycolysis. 23 –25 It is relatively inefficient as a pathway of energy generation, which requires significantly more glucose substrate for equivalent ATP production.18F-FDG uptake is lower in viable and well-perfused tumor areas and therefore fails to map oxygenated tumor cells.
Our previous research proved that 18F-FDG uptake occurred significantly higher in severely hypoxic ascites carcinomas and the hypoxic portion of larger tumors. 10 As in Figure 1, the spatial distribution of 18F-FDG in the A549 peritoneal tumor model precisely matched with tumor hypoxia, visualized by the hypoxia-specific probe pimonidazole. Besides, the clinical data from lung cancers provide indirect evidence that 18F-FDG PET reports on tumor hypoxia (Fig. 2). 26 The tracer signal in the early phase of the 18F-FDG imaging, primarily defined by blood delivery or supply, may assess the tumor perfusion. However, by 60-min after 18F-FDG injection, activity has accumulated in cells via the GLUT-1/hexokinase pathway, which reveals that the tumor volumes associated with low blood perfusion display the greatest concentration of 18F-FDG uptake.
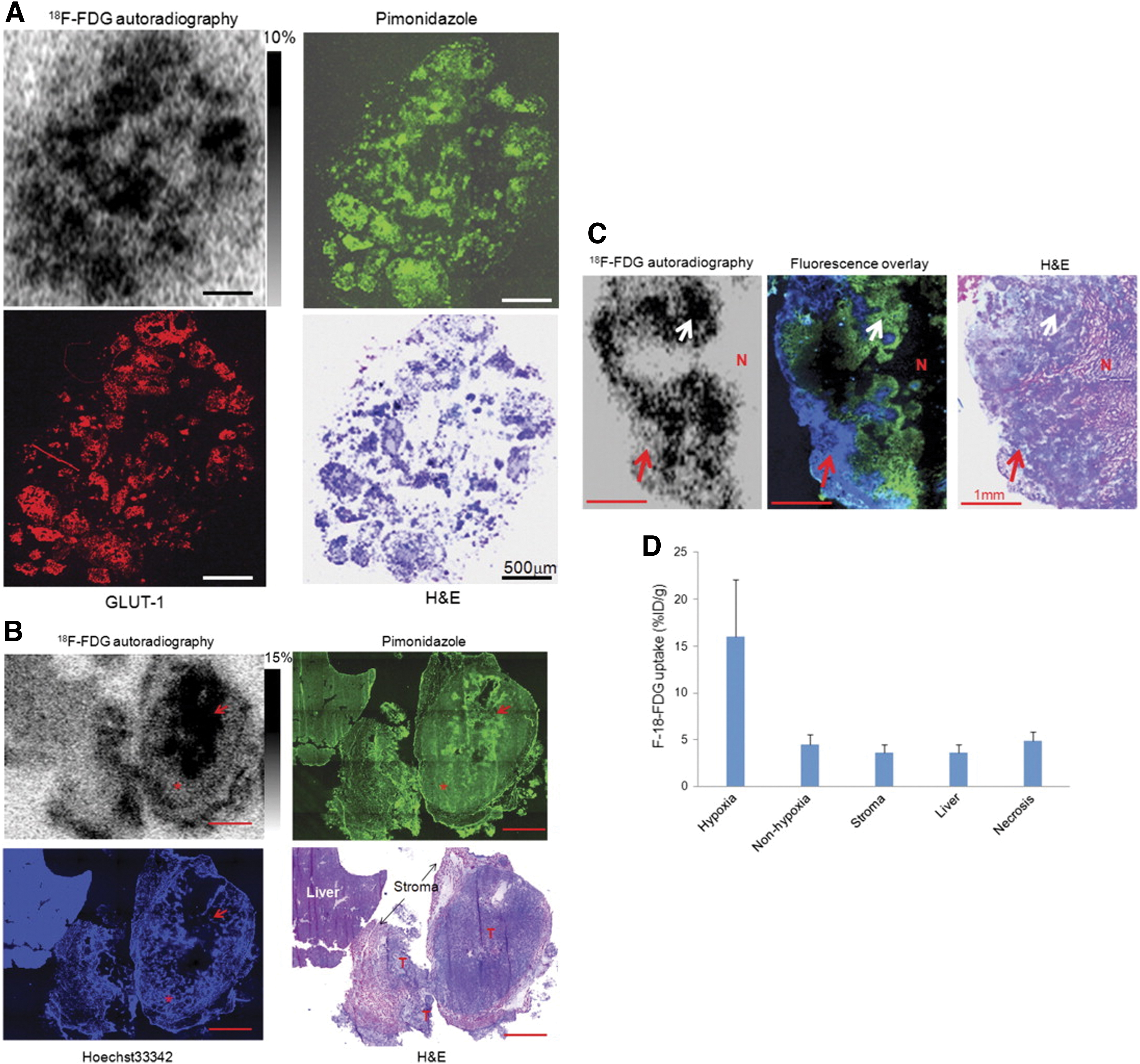
Comparison of 18F-FDG uptake with tumor hypoxia.
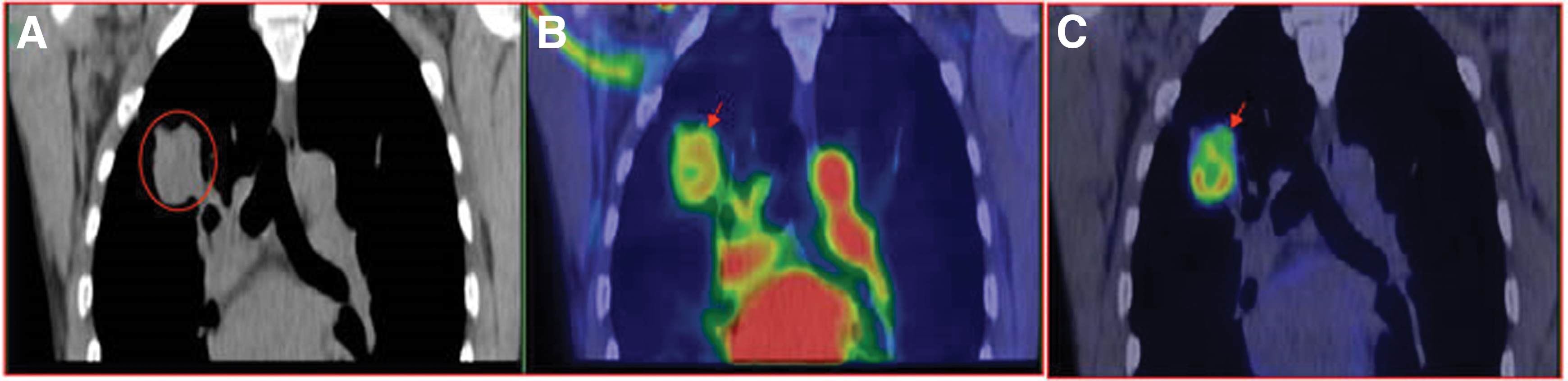
Mismatched intratumoral distribution between early phase perfusion imaging versus 60-min metabolism imaging. A 52-year-old male with pathologically confirmed adenocarcinoma in the right lung, received 6 mCi (222MBq) 18F-FDG bolus injection.
Other studies have reported similar results. Kaira et al. also found a significant correlation between 18F-FDG uptake and hypoxia. 27 In a clinical study of primary and metastatic non-small cell lung cancers, it was reported that low 18F-FDG uptake was associated with well oxygenated tumor regions' high levels of angiogenesis. 17 These findings demonstrated that viable and well-perfused cancer regions in patients were associated with low 18F-FDG uptake, while areas with high 18F-FDG accumulation had low to absent blood perfusion. Therefore, the Pasteur Effect may explain the nature of the 18F-FDG uptake in cancer.
Cell proliferation and hypoxia are usually mutually exclusive. However, the presence of tumor hypoxia results from the extensive increase of tumor cells surpassing angiogenesis. 28 The tumor cells close to the functional vessels should have a higher proliferation rate, while the hypoxic tumor cells located far from the active vessels will propagate slowly. Interestingly, the 18F-FDG uptake of cells with less proliferation in the hypoxic area of the tumor was higher than that in the normoxic tumor zone. 10,29,30 Other studies verified such observations. In a well-differentiated occult sphenoclival tumor, immunohistochemistry showed low tumor cell proliferation but intense focal 18F-FDG uptake, which explained the paradox between tumor differentiation and PET results. 31 In well-differentiated neuroendocrine tumors, 18F-FDG PET uptake correlates with both tumor size and proliferation. 32
A possible explanation is that proliferating tumor cells generate ATP from glucose at least to some extent through efficient oxidative phosphorylation, so less glucose is needed, thus resulting in with low accumulation of 18F-FDG. Also, the necrotic tumor zones were associated with lower 18F-FDG activity. 18 However, there are also different opinions. For example, Türkcan et al. reported that in mouse tumor grafts, the uptake of 18F-FDG in the tumor's core was usually lower than the periphery. Since the tumor core is likely to be highly hypoxic, they conclude that severe hypoxia is not associated with elevated 18F-FDG uptake. 33 We thought a more plausible explanation might be that the tumor center is hypoxic since it is dominated by necrotic tissue, which has no glucose metabolism and no 18F-FDG uptake; The 18F-FDG activity is lower than the periphery of the lesion.
The high accumulation of 18F-FDG does not indicate areas of tumor cells with increased proliferation but tumor cells with a low proliferation rate. Also, the lack of 18F-FDG does not necessarily mean the absence of viable tumor cells.
18F-FDG PET Myocardial Ischemia Imaging and Hypoxia
Direct myocardial ischemia imaging can overcome some of the limitations of stress-rest perfusion imaging currently used and provide a unique opportunity to detect and image an episode of ischemia in the preceding hours, even in the absence of other markers of ongoing myocardial ischemia. 34 Sometimes, even in the presence of severe multivessel coronary artery disease, myocardial perfusion imaging and resting 18F-FDG metabolic imaging may appear normal. However, 18F-FDG can successfully image the exercise-induced direct myocardial ischemia because of the vital link between the tissue 18F-FDG uptake and hypoxia, which has a greater significance for the diagnosis, management of heart disease. The profound metabolic differential between the normal and the ischemic myocardium on exercise permits the use of 18F-FDG for myocardial ischemia imaging.
Several unique characteristics of myocardial metabolism and its myocardial ischemia changes explain why 18F-FDG uptake would respond strongly to exercise-induced myocardial ischemia. Under fasting conditions, the level of free fatty acids is higher. The levels of glucose and insulin are lower, so free fatty acid is the primary source of energy production, and glucose uptake is relatively low. After a carbohydrate-rich meal, both blood sugar and insulin levels are high, so myocardial glucose uptake increases, and glucose accounts for a higher percentage of energy production.
With the occurrence of myocardial ischemia, myocardial cells have undergone profound changes in substrate utilization and metabolism due to hypoxia. Glycolysis becomes the main source of energy production for the sustenance of the ischemic myocardium. However, glycolysis is an inefficient form of energy production. Therefore, myocardial ischemia results in a dramatic and sustained increase in glucose uptake compared with the normal myocardium. However, the normal myocardium would have much lower glucose extraction on exercise under fasting conditions because free fatty acid would be its primary energy production source. Thus, a profound metabolic difference exists between normal and ischemic myocardium on exercise, allowing 18F-FDG to accumulate in ischemic tissue allowing “hot-spot” imaging. 13,14
For example, He et al. found that the areas diagnosed as myocardial ischemia during stress-rest perfusion imaging showed intense 18F-FDG uptake in exercise 18F-FDG ischemia imaging. 3 However, for patients with coronary artery disease who received revascularization of the abnormal myocardium, postexercise 18F-FDG images show no excessive 18F-FDG uptake, in agreement with the notion that the recovery of blood flow providing normal oxygenation will also normalize the18F-FDG PET images. 35
Interestingly, not infrequently, nuclear cardiologists have observed cases with minimal perfusion abnormalities in patients with severe multivessel coronary artery disease, in whom their postexercise myocardial ischemia imaging is showing intense global 18F-FDG uptake. 7 For severe multivessel coronary heart disease, direct myocardial ischemia imaging can show ischemic myocardium more accurately than perfusion imaging. The principal reason of which is the high uptake of 18F-FDG in hypoxic cardiac myocytes with low perfusion. Exercise-induced regional myocardial 18F-FDG uptake is highly specific and sensitive for myocardial ischemia, which may persist 24 h after an episode of myocardial ischemia in some patients. 36
18F-FDG PET Inflammation Imaging and Hypoxia
18F-FDG PET imaging has a high sensitivity for the detection of inflamed or infected tissues, lesions. The 18F-FDG PET imaging may enable us to localize and diagnose inflammatory lesions and estimate the degree of infection to guide clinical treatment decision-making. 37 Specifically, 18F-FDG PET has significant clinical diagnostic value for bacterial or viral pneumonia, endocarditis, arteritis, rheumatoid arthritis, atherosclerosis, tuberculosis, and fever of unknown origin. 38 –40 In the clinical application of 18F-FDG PET imaging, 18F-FDG uptake is usually low in acute inflammation and high in the chronic phase, 8,9 which might be confusing.
The increased uptake of 18F-FDG in inflammatory lesions is due to increased metabolism of inflammatory cells such as macrophages, increased expression of cell-membrane glucose transporter, and increased glucose uptake. 41,42 Of the acute and chronic phase of the inflammation, the vascular reactions dominate the acute phase, and the cellular hyperplasia and necrosis dominate the chronic stage. As the disease progresses, due to increased catabolism at the lesion site, oxygen consumption also increases. Also, disordered local blood circulation and impaired enzyme activity intensify tissue hypoxia. Eventually, tissue metabolism switches to anaerobic glycolysis, with a concomitant increase in 18F-FDG uptake.
Although hypoxia is not apparent in the acute phase of inflammation, it is severe in the chronic phase, so that 18F-FDG uptake could be low in the acute phase of inflammation and high in the chronic phase. In rheumatoid arthritis, Matsui et al. reported tissue hypoxia leading to enhanced 18F-FDG uptake by macrophages and fibroblasts via the glycolytic pathway. 9 Further, studies have shown that intraplaque hypoxia, but not proinflammatory cytokines, potently stimulated glucose uptake in human macrophages and foam cells in atheroma plaques. 43 Future preclinical and clinical studies may need to explore 18F-FDG uptake in inflammation and infection.
Possible Mechanism of Hypoxia-Induced 18F-FDG Uptake
In the tumor, myocardial ischemia, and inflammation imaging, 18F-FDG uptake is positively associated with the degree of hypoxia. The specific mechanism of hypoxia-induced 18F-FDG uptake is still unclear. Nevertheless, a large number of studies have shown that it may be related to metabolic changes, macrophage activation, and alterations in glycolysis-related enzyme activity caused by hypoxia (Fig. 3).
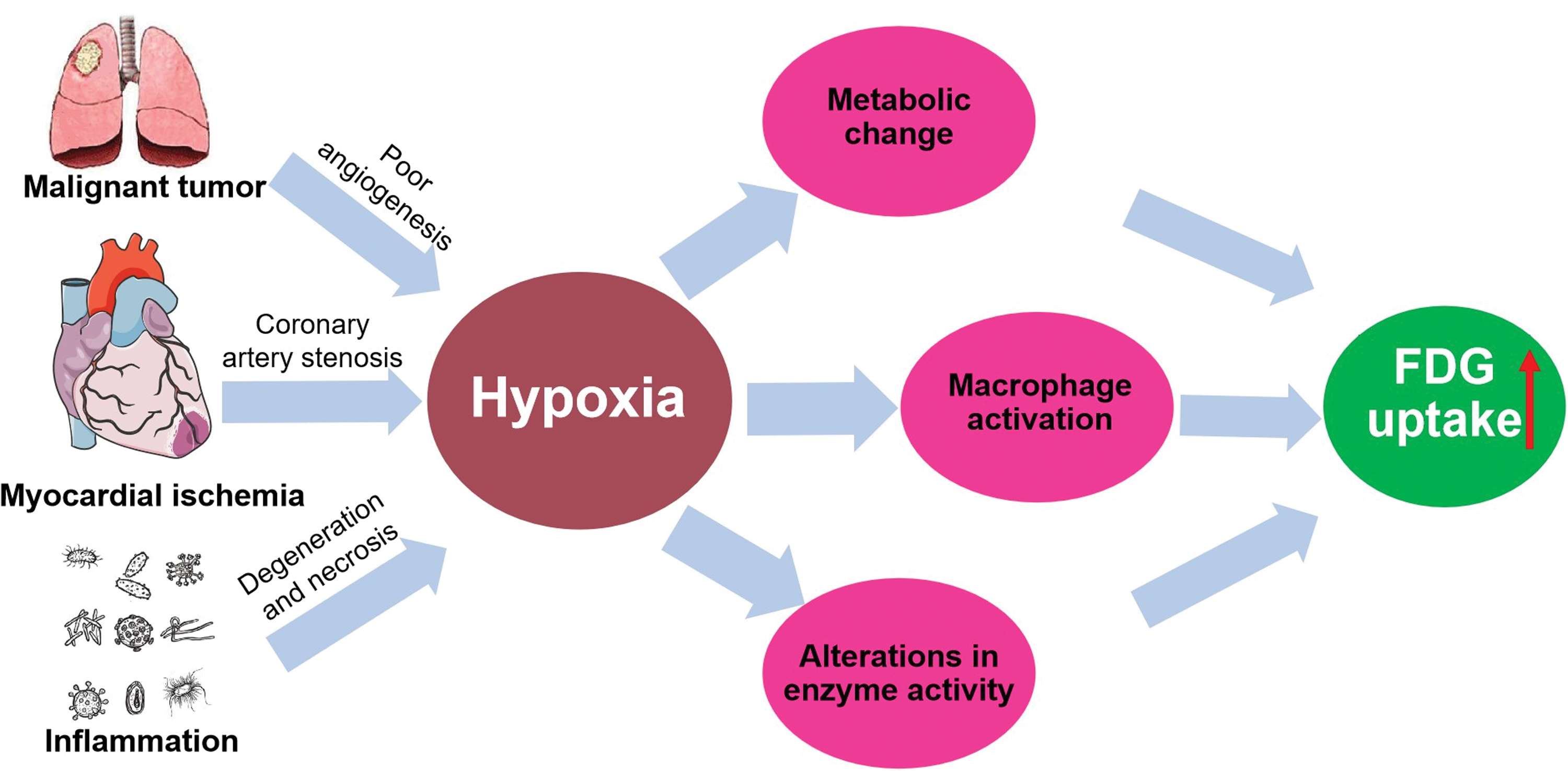
Possible mechanism of hypoxia-induced 18F-FDG uptake. Color images are available online.
Significant consequences of hypoxia are that cells activate an angiogenic program to increase oxygen delivery and adjust their cellular fuel metabolism from mitochondrial respiration to glycolysis. 44 Another result of reduced oxygen availability is the induction of hypoxia-inducible factor (HIF). When activated, HIF enhances glucose uptake and glycolysis by directly inducing the GLUT1 and glycolytic genes transcription. 45 Also, hypoxia leads to the upregulation of hexokinase protein, an essential promoter of glucose uptake and metabolism. 46 Cells must then rely on glycolysis so that their bioenergetics demands are met. Anaerobic glycolysis is an inefficient energy production way, and thus, hypoxic tissue cells need to take up a higher amount of 18F-FDG. Cellular uptake of 18F-FDG is proportional to glycolytic activity and promoted by upregulation of GLUT, increased hexokinase activity, and reduced glucose-6-phosphatase activity.
Malignant tumors may have insufficient angiogenesis resulting in severe hypoxia, making cells switch from aerobic metabolism to anaerobic metabolism. This adaptive response involves the coordinated expression of many HIF regulatory proteins, such as GLUT-1 and various glycolytic enzymes. The overexpression of GLUT-1on the surface of tumor cells helps to transport more glucose into tumor cells to meet the needs of high metabolism and rapid growth. 47 High 18F-FDG uptake and tumor-associated macrophages (TAM) positively correlated with each other in patients with nonsmall cell lung cancer. TAM can significantly enhance tumor hypoxia by increasing peroxisome proliferator-activated receptor gamma coactivator 1-alpha. 48
Also, the ischemic myocardium requires a large amount of glucose to maintain its nutrition, caused by a rapid translocation of specialized glucose transporters, such as GLUT-4 and GLUT-1 from the cytosol to the sarcolemma. 3,12 Lee and his colleagues demonstrated that depletion of macrophages could lead to a significant decrease of 18F-FDG uptake in the heart after acute myocardial infarction in mice. 49
The chronic phase of inflammation is marked by infiltration of inflammatory cells such as macrophages, which can phagocytize pathogens and remove tissue disintegration products. In inflammation, 18F-FDG accumulates mostly in macrophages due to their high glycolytic activity and increased density in inflamed areas. 50 Clinical and preclinical studies have positively associated increased 18F-FDG uptake in atherosclerotic arteries with high macrophage density. 51,52 Hypoxia may affect the metabolic system by increasing activity in the glycolytic and pentose phosphate pathways of infiltrating macrophages in atherosclerotic arteries. Hypoxia is a potent stimulus for increased glucose uptake of macrophages. 53,54
The Correct Interpretation of 18F-FDG PET Imaging and Its Enlightenment to Clinical Practice
Despite that hypoxia may be an essential driving force of 18F-FDG uptake, as a PET imaging agent, uptake of 18F-FDG is not specific to the tumor, ischemic myocardium, or inflammation. In 18F-FDG PET tumor imaging, low or absence of 18F-FDG uptake does not mean that the lesion is a benign tumor and the prognosis is well. Similarly, the high 18F-FDG uptake does not mean that the lesion is a malignant tumor with a poor prognosis.
As mentioned earlier in this text, 18F-FDG tissue uptake is affected positively by the degree of cell hypoxia to some extent, but 18F-FDG is not a specific cancer-avid PET tracer. The reason why PET imaging can successfully use 18F-FDG in cancer detection is that 95% of solid malignant tumors have some degree of hypoxia. 18F-FDG accumulated heavily in anoxic tumor cells with poor proliferation and low blood flow but low uptake in oxygen-rich tumor cells with intense proliferation and high perfusion. 10 Consequently, some untreated malignant lesions may have little or no 18F-FDG uptake. Also, low 18F-FDG accumulation after anticancer therapy does not necessarily mean the total elimination of all viable cancer cells since well-oxygenated or necrotic cancer cells also have low 18F-FDG uptake.
As with 18F-FDG tumor imaging, hypoxic cardiac myocytes with low perfusion showed high 18F-FDG uptake in exercise-induced myocardial ischemia imaging. Direct myocardial ischemia imaging can show ischemic myocardium more accurately than perfusion imaging. Especially in severe multivessel coronary heart disease, direct myocardial ischemia imaging can accurately show the lesions, which provides more accurate imaging evidence for clinical diagnosis and treatment. If there is 18F-FDG uptake in the perfusion defect area, it indicates the existence of viable myocardium. However, 18F-FDG uptake not only indicates myocardial viability but also proves that cardiac myocytes are in a state of ischemia and hypoxia or even on the verge of death, giving the clinic a hint of an urgent need for intervention and treatment.
In 18F-FDG PET inflammation imaging, low 18F-FDG uptake does not necessarily indicate the absence of inflammation, but may imply an acute phase of inflammation known to have no prominent hypoxia. On the contrary, a high degree of 18F-FDG uptake does not indicate the severity or intensity of the inflammation; it merely may imply a chronic phase of inflammation known to be associated with severe hypoxia. The correct interpretation of 18F-FDG uptake help clinicians make accurate and useful treatment decisions resulting in improved patient prognosis outcomes.
Non-18F-FDG PET Hypoxia Imaging Agents
Several specific hypoxia avid PET tracers other than the 18F-FDG metabolic imaging agent, with its tissue uptake enhanced by hypoxia, are available. 18F-Fluoromisonidazole (18F-FMISO) is the most widely used radiolabeled hypoxia imaging tracer. 18F-FMISO is a nitroimidazole compound that enters cells through, and proportional, the blood flow. Redox reactions can occur under the action of xanthine oxidase. In hypoxic cells, the nonoxidized reduced nitro group will permanently firmly combine with some cell components. It is the first hypoxia PET tracer used clinically. It has good reproducibility and can comprehensively evaluate hypoxia, guide treatment, and predict the therapy's efficacy.
More current, PET tracers include, for example, 18F-Fluoroerythronitroimidazole (18F-FETNIM), a novel imaging agent currently under investigation for the in vivo detection of tumor hypoxia, has a lower peripheral metabolic rate, has higher hydrophilicity, and lower neurotoxicity. 55 It is easier to prepare and less costly than 18F-FMISO. The hypoxic PET tracer 18F-FAZA (18F-Fluoroazomycinarabinoside) has a stronger affinity for hypoxic tumor areas and has a faster clearance rate from human tissues. 56 18F-flortanidazole (18F-HX4) is a next-generation 2-nitroimidazole tumor hypoxia imaging tracer, specifically designed to maximize pharmacokinetic and clearance properties. It has the potential to aid hypoxia-targeted treatments. 57 Also, a nitroimidazole group compound, 18F-nitroimidazolpentafluoropropylacetamide (18F-EF5), has considerable membrane permeability and the capability to accumulate in viable hypoxic, although not in apoptotic or necrotic cells. It has more specificity and lower toxicity when taken up by hypoxic cells. 58
Conclusions
18F-FDG is not a specific PET tracer, and its uptake is increased due to hypoxia. In PET imaging of tumors, myocardium, and inflammation, hypoxia may be one of the fundamental driving forces. The possible mechanism of 18F-FDG uptake caused by hypoxia may be related to metabolic changes, macrophage activation, and alterations in glycolysis-related enzyme activity.
Footnotes
Acknowledgment
The authors thank Dr. James Russell, PhD from Memorial Sloan-Kettering Cancer Center (New York, NY) for critically reading the article.
Authors' Contributions
Y.Y. drafted and revised the article. Y.-M.L., Z.-X.H., and A.C.C. reviewed and modified all versions of the article. X.-F.L. supervised, wrote, and modified the article. All coauthors have reviewed and approved the article before submission.
Disclosure Statement
All authors declare that they have no conflict of interest.
Funding Information
The authors' current research is partially supported by an internal grant from Shenzhen People's Hospital (X.-F.L.), Shenzhen Science Technology Project (JCYJ20190806151003583) (X.-F.L.), and grant from China Postdoctoral Science Foundation (2020M673079) (Y.Y.).
