Abstract
Introduction:
Accurate assessment of treatment outcomes in patients with Tourette syndrome (TS) is essential for evidence-based clinical care. This report determined the minimal clinically important difference (MCID) on the Yale Global Tic Severity Scale (YGTSS) Total Tic Score (YGTSS-TTS) and YGTSS Impairment Scale (YGTSS-I), using the Clinical Global Impression of TS Severity (CGI-TS-S) and Improvement (CGI-TS-I) as anchors, in pediatric patients with TS receiving pharmacotherapy.
Materials and Methods:
Analyses used data from two clinical trials of ecopipam (a randomized controlled trial and its open-label extension). Receiver operating characteristic (ROC) analysis determined the percentage reduction in YGTSS scores that distinguished patients with improvement from those with no change or worsening on the CGI-TS-S and CGI-TS-I. Spearman’s correlation, empirical cumulative distribution function, and probability distribution function analyses examined relationships between YGTSS-TTS and CGI-TS-S or CGI-TS-I.
Results:
Overall, 133 patients (75.2% male; mean [SD] age, 12.7 [2.8]) were included; 63.2% had improvement on the CGI-TS-S, and 78.2% showed improvement on the CGI-TS-I. Percentage reduction in YGTSS scores that distinguished improvement from no change or worsening on the CGI-TS-S and CGI-TS-I ranged from 18.6%–33.3% (area under the ROC curve range, 0.71–0.81). Improvement on the YGTSS-TTS was correlated with posttreatment CGI-TS-S (r = −0.65; p < 0.001) and CGI-TS-I (r = −0.61; p < 0.001) scores. The MCID for YGTSS-TTS was achieved by 67% and 62% of patients with improvement on the CGI-TS-S and CGI-TS-I, respectively.
Conclusions:
This analysis is the first to determine the MCID for YGTSS in a pediatric population with TS receiving pharmacotherapy. Whether using CGI-TS-S or CGI-TS-I as the anchor, a 25% reduction in YGTSS scores was a generally appropriate minimum threshold to define clinically meaningful improvement in this population. Findings offer an objective threshold for classifying clinically meaningful improvement in children and adolescents receiving pharmacotherapy for TS in clinical practice.
Introduction
Tourette syndrome (TS) is a chronic neurodevelopmental disorder that affects ∼0.5% of the population and is characterized by motor and vocal tics of varying frequency, severity, and complexity that begin in childhood or adolescence and persist for ≥1 year (Johnson et al., 2023; Scharf et al., 2015). Although exhibiting a periodic waxing and waning pattern, tics typically peak in severity during early adolescence (Ueda and Black, 2021). In addition to tics, patients with TS often experience comorbidities, such as anxiety, attention-deficit/hyperactivity disorder, and obsessive-compulsive disorder (Hirschtritt et al., 2015). Tics and co-occurring symptoms lead children with TS to experience substantial distress, functional impairment, and diminished quality of life, highlighting the need to identify effective interventions to provide symptom relief (Cavanna et al., 2013; Evans et al., 2016).
Large meta-analyses of randomized controlled trials (RCTs) have demonstrated the efficacy of evidence-based behavioral and pharmacologic interventions for reducing tic severity and impairment (Farhat et al., 2023; McGuire et al., 2014). Objective monitoring of treatment response in RCTs is essential to evaluate the benefit of an evidence-based intervention, which then informs treatment planning and clinical decisions. However, standardized assessments that are suitable to determine clinical benefit in the research setting may be less useful than individualized assessments in clinical practice. Determining objective benchmarks of symptom improvement for evidence-based rating scales can help implement objective monitoring of an individual patient’s response to TS intervention in clinical practice.
Several evidence-based assessments for TS are applied in clinical research and practice (Martino et al., 2017; McGuire et al., 2012). The Yale Global Tic Severity Scale (YGTSS) is considered the gold standard for measuring tic severity and can assess dimensional symptom improvement during treatment (Leckman et al., 1989; McGuire, et al., 2012, 2021). The Clinical Global Impression of TS Severity (CGI-TS-S) is a health care provider (HCP) rating of severity relative to the entire TS patient population, and the CGI of TS Improvement (CGI-TS-I) is an HCP rating of within-patient change since treatment initiation (Guy, 1976; Leckman, et al., 1989); both scales can take into account all available information (e.g., subjective reporting from patient and family members and direct clinical observation) in addition to tics (Jeon et al., 2013). These three measures provide comprehensive and complementary information about the symptoms and severity of TS. Whereas YGTSS characterizes the presence and severity of tics using a standardized approach, the CGI-TS-S and CGI-TS-I enable individualized assessment that can capture particularly distressing tics and whether the treatment has been sufficient to transition a patient from one severity category to another (e.g., from “moderate” to “mild”) (McGuire, et al., 2021).
The U.S. Food and Drug Administration upholds the importance of determining clinically meaningful within-patient change for pharmacotherapies (US Food and Drug Administration, 2018). The minimal clinically important difference (MCID) for an outcome of interest can be determined with an anchor-based approach (Hudgens et al., 2024; Ondo et al., 2016), such as using the CGI-TS-S and CGI-TS-I as external criteria (i.e., anchors) to identify patients with perceptible improvement in TS severity and determine the corresponding change in YGTSS scores for those patients. Two prior investigations included the YGTSS and CGI of Improvement (CGI-I [not specific to tic disorders]) to establish benchmarks of clinically meaningful improvement (Jeon, et al., 2013; Storch et al., 2011). Storch et al. (2011) used signal detection analyses in 108 patients aged 5–17 years with TS to identify that a ≥35% reduction in the YGTSS Total Tic Score (YGTSS-TTS) detected a treatment response on the CGI-I but, given low sensitivity, suggested that a 25% improvement might be more useful clinically. Notably, this report included heterogeneous study designs (e.g., RCTs, open-label studies) and interventions (e.g., pharmacotherapy, psychotherapy, and naturalistic care) (Storch, et al., 2011). Jeon et al., (2013) used three analytic methodologies in 232 children and adults from RCTs of behavioral therapy to identify that a ≥25% reduction in the YGTSS-TTS optimally predicted a treatment response on the CGI-I. Despite its statistical rigor, this report only included patients who received behavioral therapy, and its findings may not be generalizable to pharmacotherapy (Jeon, et al., 2013).
Two important issues remain. First, it is critical to determine the optimal reduction in the YGTSS that reflects clinically meaningful improvement from pharmacotherapy (i.e., what percentage reduction in the YGTSS corresponds with improvement in the CGI-TS-S and CGI-TS-I in patients receiving pharmacotherapy?). Second, it is important to identify the percentage reduction on the YGTSS that corresponds with movement from one severity category to another on the CGI-TS-S. Ecopipam is a first-in-class selective dopamine D1 receptor antagonist under investigation as a potential treatment for TS. Treatment with ecopipam (1.8 mg/kg/day for 12 weeks) significantly reduced YGTSS-TTS and CGI-TS-S scores from baseline with significance versus placebo (both, p < 0.05) in a phase 2b RCT (Gilbert et al., 2023a), and sustained benefit was demonstrated during the 12-month open-label extension (OLE) (both, p < 0.0001 vs. baseline) (Gilbert et al., 2023b). The aim of this analysis was to determine MCIDs for the YGTSS-TTS and YGTSS-I using the CGI-TS-S and CGI-TS-I as anchors in children and adolescents with TS who participated in these two ecopipam trials.
Materials and Methods
Study design and participants
Post hoc analyses were performed using data from the phase 2b, randomized, double-blind, placebo-controlled trial of ecopipam in children and adolescents with TS (ClinicalTrials.gov identifier NCT0400799) and its OLE (NCT04114539). The trials were conducted in accordance with the International Council on Harmonisation of Good Clinical Practice guidelines and the ethical principles of the Declaration of Helsinki. Detailed methods have been reported previously (Gilbert, et al., 2023a; Gilbert, et al., 2023b). Briefly, patients were eligible for the phase 2b trial if they: (1) were aged 6 to <18 years at screening, (2) had a diagnosis of TS according to the Diagnostic and Statistical Manual for Mental Disorders–5th Edition (DSM-V) criteria at baseline, and (3) had a YGTSS-TTS score ≥20 at screening and baseline visits (Gilbert, et al., 2023a). Exclusion criteria included: (1) any unstable primary mood disorder according to DSM-V criteria at screening, (2) history of schizophrenia, bipolar disorder, or other psychotic disorder, (3) history of other neurological conditions associated with abnormal movements, or (4) a significant suicide risk. Medications prescribed for the treatment of anxiety, depression, or attention-deficit/hyperactivity disorder were permitted if the dosage was stable for ≥4 weeks before screening (Gilbert, et al., 2023b).
Measures
Yale Global Tic Severity Scale
The YGTSS serves as the primary outcome measure in many clinical trials for TS (Gilbert, et al., 2023a; Piacentini et al., 2010; Wilhelm et al., 2012). The YGTSS-TTS assesses the severity of motor and vocal tics in the past week across five dimensions: number, frequency, intensity, complexity, and interference with behavior, speech, action, or communication (Leckman, et al., 1989; McGuire, et al., 2012). These items are summed to produce the YGTSS-TTS, which ranges from 0 to 50, with higher scores indicating greater severity (Leckman, et al., 1989; McGuire, et al., 2012). The YGTSS also has an impairment scale (YGTSS-I) that collectively assesses impairment in self-esteem, family life, social acceptance, and overall functioning caused by motor and vocal tics (score range, 0–50 [severe impairment]) (Leckman, et al., 1989; McGuire, et al., 2012). The YGTSS-TTS and YGTSS-I have shown good reliability, validity, and treatment sensitivity (Haas et al., 2021; Jeon, et al., 2013; Leckman, et al., 1989; McGuire et al., 2018, 2021).
Clinical Global Impression of Tourette Syndrome Severity and Improvement
The CGI-TS-S and CGI-TS-I are HCP-rated tic disorder–specific adaptations of the CGI-S and CGI-I scales and assess TS severity and TS global improvement, respectively (Guy, 1976; Leckman et al., 1988). The CGI-TS-S consists of a single item rated on a 7-point scale that ranges from 1 (“normal”) to 7 (“extremely ill”), and the CGI-TS-I is a single item rated on a 7-point scale that ranges from 1 (“very much improved”) to 7 (“very much worse”) (Guy, 1976).
Procedures
The study protocols, including the informed consent and assent forms, received written institutional review board approval (Advarra, Inc., Columbia, MD; IRB00000971). For patients aged <18 years, parents or legal guardians provided written informed consent, and patients provided written assent before any study procedures were initiated. In the OLE study, patients aged 18 years provided written informed consent. Individuals were randomly assigned 1:1 to receive either ecopipam (target oral dose, 1.8 mg/kg/day) or placebo for 12 weeks (4-week titration phase and 8-week treatment phase), followed by dose tapering by 25 mg/day until fully discontinued. Participants who completed the phase 2b trial were eligible to enroll in the OLE (Gilbert, et al., 2023b), in which ecopipam was titrated over 4 weeks to achieve a target oral dose of 1.8 mg/kg/day for a total treatment period of 12 months. The YGTSS and CGI-TS-S were assessed during study visits at baseline and Weeks 4, 6, 8, and 12 of the phase 2b trial, and at OLE baseline and Months 1, 3, 6, 9, and 12 of the OLE. The CGI-TS-I was assessed at the same timepoints with the exception of baseline.
Statistical analyses
Post hoc analyses were performed with pooled data from the phase 2b and OLE trials using the modified intent-to-treat population from the phase 2b trial (received ≥1 dose of study drug and completed ≥1 postbaseline assessment). For patients randomly assigned to receive ecopipam in the phase 2b trial, data from baseline and Week 12 (or closest available visit [≥4 weeks after baseline]) were used. For patients randomly assigned to receive a placebo in the phase 2b trial who enrolled and received ecopipam in the OLE study, data from the OLE baseline and Month 3 (or closest available visit [≥4 weeks after OLE baseline]) were used. Statistical analyses were performed using SAS 9.4 or higher (SAS Institute, Cary, NC).
An anchor-based approach determined the MCID for the YGTSS-TTS and YGTSS-I using the CGI-TS-S and CGI-TS-I as anchors. Specifically, the MCID was defined as the mean percent reduction in YGTSS score among patients with ≥1-point improvement from baseline on the CGI-TS-S and among those with ratings of “improved” or better (i.e., scores ≤3) on the CGI-TS-I 12 weeks after first exposure to ecopipam. Receiver operating characteristic (ROC) analyses determined the cutoffs for percentage improvement in YGTSS scores that distinguished participants with improvement from those with no change or worsening on the anchor measures. The area under the ROC curve (AUC) evaluated the overall classification performance of each of the ROC models (i.e., the ability to distinguish between improvement and no change or worsening on the anchor measures). ROC analysis was performed using logistic regression. Spearman’s correlation coefficients were calculated for percentage improvement in YGTSS-TTS and anchor measure scores at 12 weeks (or the closest available timepoint) after first exposure to ecopipam. Probability density function (PDF) curves for the distribution of YGTSS scores among participants with improvement versus no change or worsening on the anchor measures were estimated using kernel density methods. Smoothed empirical cumulative distribution function curves were created by numerically integrating PDF curves using the trapezoidal rule.
Results
A total of 133 patients with TS were included in the analyses. Participants were predominantly male (75.2%), 12–17 years of age (64.7%), and had marked baseline tic severity on the YGTSS-TTS (mean [SD] score, 33.0 [7.3]), YGTSS-I (31.1 [9.5]), and CGI-TS-S (4.7 [1.0]; Supplementary Table S1). Of 133 patients, 84 (63.2%) exhibited improvement on the CGI-TS-S, with 49 (36.8%) displaying no change or worsening. On the CGI-TS-I, 104 patients (78.2%) exhibited improvement, whereas only 29 (21.8%) showed no change or worsening.
Determining clinically meaningful change in YGTSS-TTS
Two discrimination criteria were applied in ROC analyses to determine the MCID for YGTSS-TTS: (1) the percentage reduction in YGTSS-TTS minimizing the difference between sensitivity and specificity, and (2) Youden’s J. Using the first criterion, the percentage reduction in YGTSS-TTS that optimally distinguished between improvement versus no change or worsening on the CGI-TS-S was 21.1% (AUC, 0.81; sensitivity ≈ specificity, 0.71), and the cutoff with the CGI-TS-I as anchor was 18.6% (AUC, 0.78; sensitivity, 0.71 ≈ specificity, 0.72) (Fig. 1; Table 1). Using Youden’s J criterion, the cutoff for percentage reduction in YGTSS-TTS score that distinguished between improvement versus no change or worsening on the CGI-TS-S was 25.0%, and on the CGI-TS-I was 22.9% (Table 1).
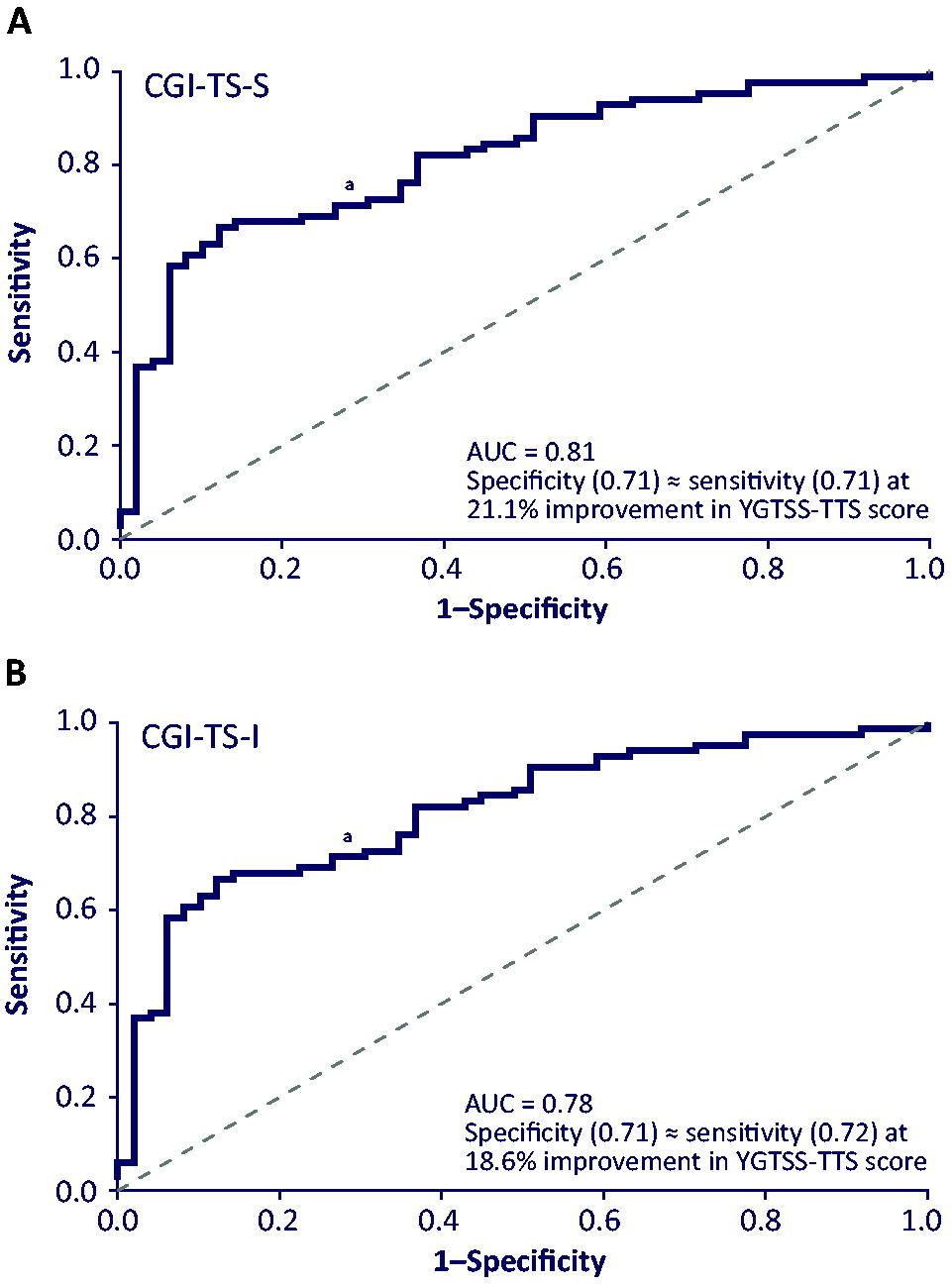
ROC curves: YGTSS-TTS improvement threshold for the optimal distinction between improvement and no change or worsening on the
ROC Analysis Results for YGTSS-TTS
Analyses were based on one data point per patient using the timepoint closest to 12 weeks after the first exposure to ecopipam (≥4 weeks after baseline).
AUC, area under the curve; CGI-TS-I, Clinical Global Impression of Tourette Syndrome Improvement; CGI-TS-S, Clinical Global Impression of Tourette Syndrome Severity; ROC, receiver operating characteristic; YGTSS-TTS, Yale Global Tic Severity Scale Total Tic Score.
Figure 2 displays the relationship between percentage improvement in YGTSS-TTS and posttreatment CGI-TS-S (Fig. 2A) and CGI-TS-I scores (Fig. 2B). There was a large negative correlation between YGTSS-TTS improvement and posttreatment CGI-TS-S score (r = −0.65; p < 0.001) and between YGTSS-TTS improvement and posttreatment CGI-TS-I score (r = −0.61; p < 0.001). Stated differently, a greater percentage improvement in YGTSS-TTS score was associated with lower TS severity and greater TS global improvement following 12 weeks of ecopipam treatment (Fig. 3; Supplementary Fig. S1). Considering the Youden’s J analysis, a ≥25.0% improvement in YGTSS-TTS was seen in 67% of patients with improvement on the CGI-TS-S and 12% of patients with no change or worsening, and a ≥22.9% improvement in YGTSS-TTS score was seen in 62% of patients with improvement on the CGI-TS-I and 10% of those with no change or worsening.
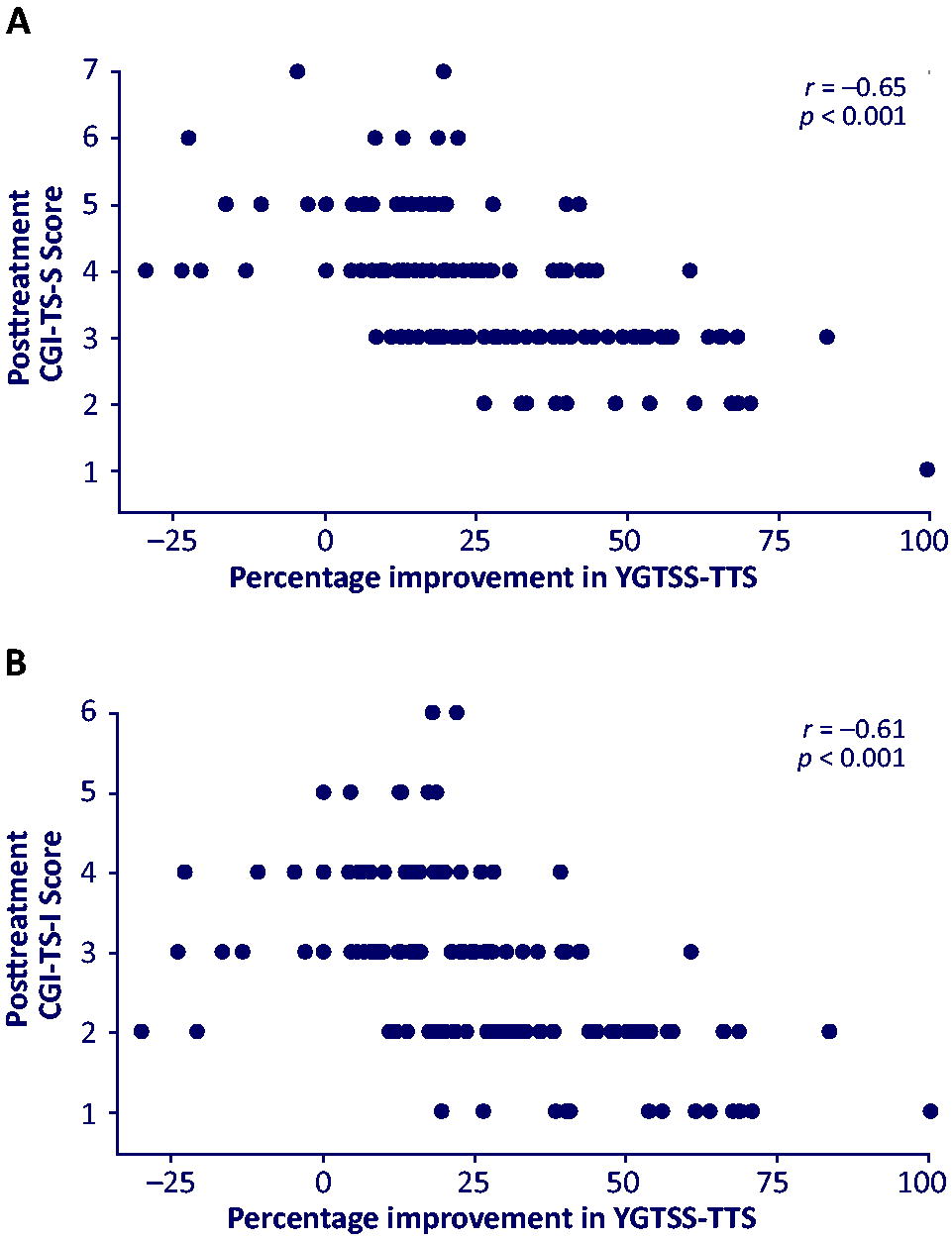
Correlation between YGTSS-TTS improvement and posttreatment
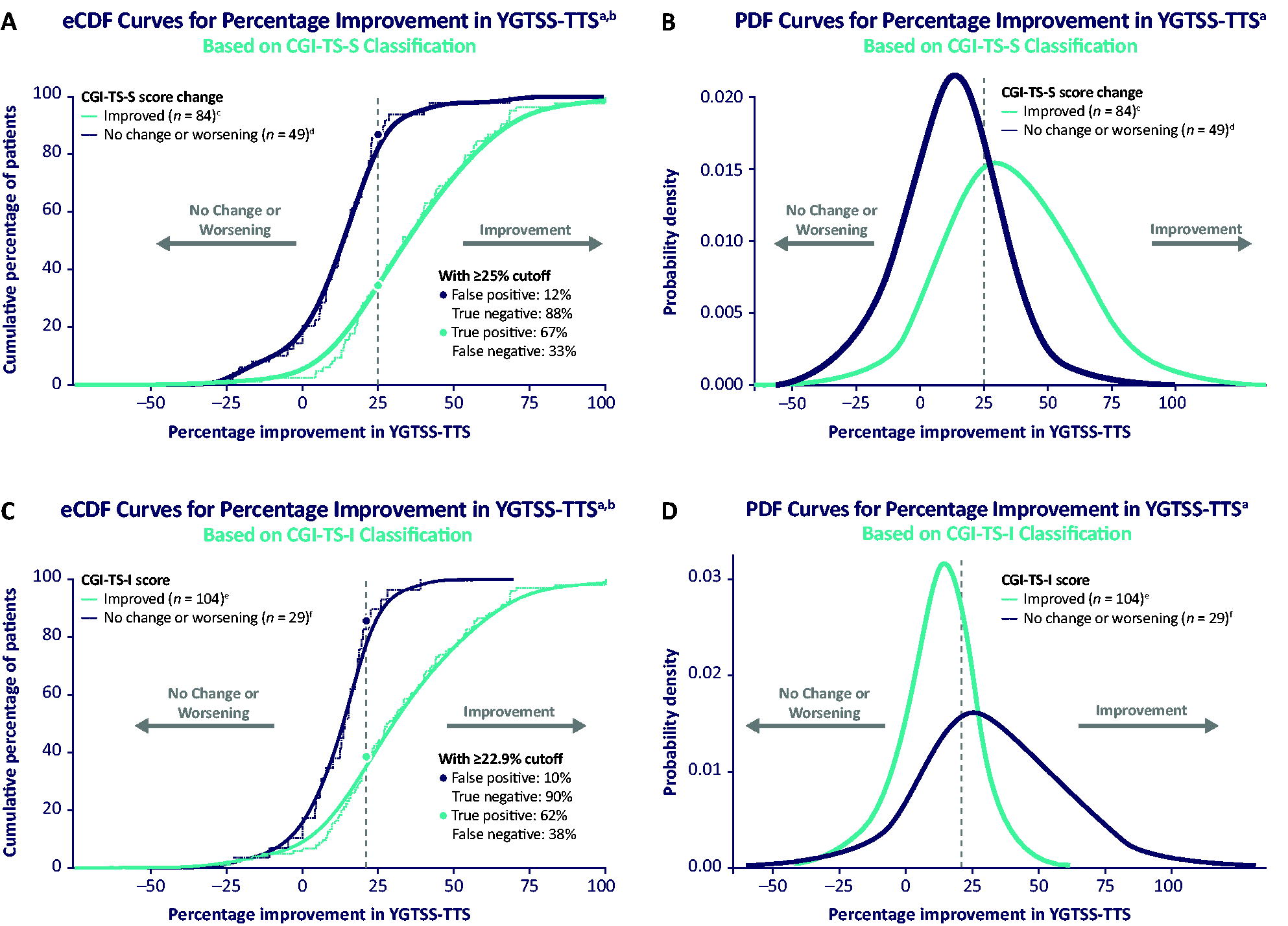
Greater percentage improvement in YGTSS-TTS among patients with improvement on the
Determining clinically meaningful change in YGTSS-I score
Using Youden’s J, the cutoff for percentage improvement in YGTSS-I that distinguished between improvement versus no change or worsening on the CGI-TS-S was 25.0% (AUC, 0.78), and between those on the CGI-TS-I was 33.3% (AUC, 0.71) (Table 2).
ROC Analysis Results for YGTSS-I
Analyses were based on one data point per patient using the timepoint closest to 12 weeks after first exposure to ecopipam (≥4 weeks after baseline).
Youden’s J cutoff method.
AUC, area under the curve; CGI-TS-I, Clinical Global Impression of Tourette Syndrome Improvement; CGI-TS-S, Clinical Global Impression of Tourette Syndrome Severity; ROC, receiver operating characteristic; YGTSS-I, Yale Global Tic Severity Scale Impairment.
Discussion
Given the recognized importance of establishing benchmarks of clinical improvement that can inform research and clinical practice (US Food and Drug Administration, 2018), this report determined the MCID for the YGTSS among children and adolescents with TS receiving pharmacotherapy. The YGTSS is recognized as the gold standard evidence-based assessment for evaluating tic severity and tic-related impairment, has shown good psychometric properties, and is the most commonly used tic assessment measure worldwide (Haas, et al., 2021; Martino, et al., 2017; McGuire, et al., 2018). Findings from the current analysis suggest that a ≥25% reduction in the YGTSS-TTS and YGTSS-I generally represents clinically meaningful improvement whether using the CGI-TS-S or CGI-TS-I as the anchor. The area under the ROC curves ranged from 0.71–0.81, indicating strong discrimination between improvement and no change or worsening on anchor measures (Mandrekar, 2010). Results were comparable regardless of the scale, subscale, or cutoff method used. Although this benchmark for clinical improvement is lower than a prior report in a participant sample with various treatment modalities (Storch, et al., 2011), the current findings are consistent with the benchmark for treatment response on the YGTSS-TTS determined among patients with TS who received behavioral therapy (Jeon, et al., 2013).
From a clinical perspective, these findings define a metric of clinical improvement that providers treating patients with TS can use to monitor treatment response to pharmacotherapy in everyday practice using the YGTSS. Importantly, the YGTSS MCID is consistent across behavioral and pharmacologic treatment modalities. Current practice parameters and treatment guidelines provide treatment recommendation guidance based on broad categories of tic severity (e.g., “mild,” “moderate,” and “severe”) that are informed by HCP experience and expertise (McGuire, et al., 2021; Murphy et al., 2013; Pringsheim et al., 2019). While previous work has used the YGTSS and CGI-TS-S to develop guidelines for tic severity categories and guide intervention selection (McGuire, et al., 2021), the current findings address a critical research-to-practice gap to facilitate objective monitoring of response to pharmacotherapy in alignment with U.S. Food and Drug Administration recommendations (US Food and Drug Administration, 2018). Indeed, the MCID for the YGTSS may be particularly helpful in guiding treatment planning for less experienced HCPs, who may have initial difficulty discerning between “moderate” and “severe” tics (McGuire, et al., 2021). As well, the determination of YGTSS MCID enables the comparison of treatment outcomes across RCTs and may facilitate translation from research to clinical practice. Notably, when applying the YGTSS MCID to determine treatment response for a child with very high or very low tic severity, some consideration is warranted. For instance, a child with a very high tic severity score (e.g., YGTSS-TTS of 40) would need to achieve a 10-point reduction to meet the ≥25% improvement threshold, whereas a child with moderate tic severity (e.g., YGTSS-TTS of 20) would only need to achieve a 5-point reduction. Thus, the utilization of multiple TS assessments (e.g., YGTSS, CGI-TS-S, and CGI-TS-I) can still prove beneficial.
Despite the numerous strengths of the current analysis (e.g., RCT design, use of multiple evidence-based tic screening instruments, and diagnostic criteria), several limitations exist. As is common in RCTs of patients with TS (Coffey et al., 2021; Piacentini, et al., 2010; Sallee et al., 2017; Wilhelm, et al., 2012), there was limited racial diversity and Hispanic ethnicity representation in the current sample. Thus, the MCID for YGTSS should be further investigated in under-represented racial and ethnic populations. Second, the sample included some assessments performed by investigators who were unmasked to treatment condition (i.e., in participants randomly assigned to receive placebo in the RCT whose later OLE data were used in analyses), which may have increased the possibility of bias in ratings. Third, analyses were underpowered to group data by age, sex, comorbidities, and/or tic characteristics. Although the YGTSS should not perform differently based on these factors, further examination of the YGTSS MCID across these characteristics could be clinically informative.
Conclusions
In summary, findings from the current analysis suggest that a 25% reduction is an appropriate minimum threshold for defining clinically meaningful improvement in YGTSS-TTS and YGTSS-I in a pediatric population with TS receiving pharmacotherapy. While these findings address a critical research-to-practice gap, further analyses are needed to determine the change in YGTSS scores that predict perceptible improvement on patient- and caregiver-rated scales (e.g., Gilles de la Tourette Syndrome Quality of Life Scale for Children and Adolescents, Caregiver Global Impression of Change) and to establish clinically meaningful improvement in YGTSS scores in adults with TS receiving pharmacotherapy.
Clinical Significance
To help clinicians objectively identify improvement in individuals with TS, this report determined the MCID for the YGTSS-TTS and YGTSS-I in children and adolescents who completed two clinical trials of ecopipam—a selective dopamine D1 receptor antagonist in development for TS. Whether using CGI-TS-S or CGI-TS-I as the anchor, a reduction in the YGTSS score of at least 25% represented an appropriate threshold to define clinically meaningful improvement. This objective benchmark can be used to classify clinically meaningful improvement and guide treatment planning in pediatric patients receiving pharmacotherapy for TS.
Footnotes
Acknowledgments
Editorial and medical writing assistance was provided under the direction of the authors by Mary Beth Moncrief, PhD, and Rachel Haake, PhD, Synchrony Medical Communications, LLC, West Chester, PA. Funding for this assistance was provided by Emalex Biosciences, Inc., Chicago, IL.
Authors’ Contributions
J.F.M. and G.B.K.: Conceptualization, methodology, writing—original draft, writing—review and editing. R.M.B.: Methodology, formal analysis, writing—review and editing. S.D.A.: Writing—review and editing. F.E.M.: Conceptualization, writing—review and editing. S.P.W.: Writing—review and editing. T.M.C.: Writing—review and editing. D.L.G.: Data curation, writing—review and editing.
Data Sharing
The data that support the findings of this analysis are available upon reasonable written request.
Author Disclosure Statement
J.F.M. and R.M.B. report being consultants for Emalex Biosciences, Inc. G.B.K., S.D.A., and F.E.M. are employees of, and have a personal equity interest in, Emalex Biosciences, Inc. S.P.W. and T.M.C. are employees of Paragon Biosciences, which has a controlling equity interest in Emalex Biosciences, Inc.; additionally, they have a personal equity interest in Emalex Biosciences, Inc. D.L.G. reports being a clinical trial site investigator for Emalex Biosciences, Inc. and PTC Therapeutics.
Supplementary Material
Supplementary Figure S1
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
