Abstract
Introduction:
While available for decades, the use of bupropion has increased in recent years. To provide an updated review on the use of bupropion, this article aimed to describe bupropion prescription details, potential indication, and treatment duration in children, young adults, and adults starting bupropion treatment.
Methods:
Individuals aged 6–64 newly initiating bupropion hydrochloride treatment were identified from commercial claims data (MarketScan, 1/1/2016–12/31/2022). New bupropion use was defined as at least 1 year without any prior bupropion dispensed prescription. Potential indications for bupropion treatment were identified from inpatient/outpatient records (ICD-10-CM diagnoses) in the 30 days prior to bupropion initiation. All analyses were stratified by age: children (6–17 years), young adults (18–29 years), and adults (30–64 years) and treatment duration up to 1 year was estimated with Kaplan–Meier estimation.
Results:
The study sample included 39,833 children, 177,710 young adults, and 548,557 adults newly initiating bupropion treatment. Bupropion extended-release 24-hour 150 mg was the most common (62%) formulation and dose at initiation. Depression was the most prevalent potential indication (children = 57%, young adults = 47%, adults = 36%) and attention-deficit/hyperactivity disorder (ADHD) was the next most common potential indication in children (25%) and young adults (12%); tobacco cessation and weight loss also identified as potential indications. Twenty-two percent of bupropion initiators were on concurrent selective serotonin reuptake inhibitor treatment. In children, suicidal ideation (16.3%), poisoning (5.9%), and anorexia or bulimia nervosa (2.2%) were relatively common diagnoses prior to bupropion initiation. Overall, 39%–45% remained on bupropion treatment for at least 6 months, with variation by potential indication.
Conclusion:
The antidepressant bupropion is prescribed to children, young adults, and adults for a variety of indications in the United States, with depression and ADHD the most common indications in children. As the prescribing of bupropion becomes more widespread, additional safety and effectiveness data will be necessary to inform prescribing decisions, particularly in populations with unknown efficacy.
Introduction
The antidepressant bupropion was approved by the US Food and Drug Administration (FDA) for depression in 1985. Bupropion was subsequently withdrawn from the market due to seizure risk and reintroduced in 1989 with lower maximum dose recommendations (Huecker et al., 2024). Currently, bupropion is FDA approved in adults for major depressive disorder, seasonal affective disorder, and as an aid to smoking cessation treatment and is also prescribed off-label for antidepressant-induced sexual dysfunction, attention-deficit/hyperactivity disorder (ADHD), weight loss in obese or overweight persons, depression associated with bipolar disorder (Hirsch and Birnbaum, 2024; Huecker et al., 2024; Taylor et al., 2013; Verbeeck et al., 2017), and as an adjunctive medication for partially effective selective serotonin reuptake inhibitor (SSRI) treatment of depression (Lee et al., 2021; Santarsieri and Schwartz, 2015). There is also evidence supporting the use of bupropion for prevention of seasonal affective disorder recurrence (Gartlehner et al., 2019) and major depressive disorder with anxiety (Papakostas et al., 2008).
While available for decades, the use of bupropion has increased in recent years. In 2004, there were an estimated 17.9 million bupropion prescriptions in the United States, increasing to 29.1 million in 2022 (Mikulic M, 2024). Bupropion was the third most commonly prescribed antidepressant in Medicaid in 2021 and totaled approximately $140 million dollars in spending in 2018 (Elmarasi and Fuehrlein, 2024). Almost 3% (2.7%) of reproductive-aged women (15–44 years) used bupropion in 2013, the second most commonly used antidepressant agent in this population (Dawson et al., 2016). In adolescents and young adults (12–25 years), there was a 66% increase in bupropion prescriptions from 2016 to 2022, becoming the fourth most commonly dispensed antidepressant in this young age group by 2022 (Chua et al., 2024).
Several factors contribute to clinical antidepressant selection, including efficacy, indication(s), guidelines, prior response to other antidepressants, and preferences for anticipated side-effects (American Psychiatric Association, 2010; Bayes and Parker, 2019). The rising use of bupropion is likely a function of its expanding on-label and off-label indications along with its distinct side effect profile compared with other antidepressants, particularly SSRIs, the most commonly prescribed antidepressant class (Clark et al., 2023; Kennedy et al., 2016; UpToDate, 2024). Bupropion, a norepinephrine and dopamine reuptake inhibitor, is one of a few antidepressants that does not cause sexual dysfunction (American Psychiatric Association, 2010; NAMI, 2016; Thase et al., 2005), a fairly common side effect of SSRIs, and bupropion is associated with a lower risk of weight gain compared with other antidepressants in adults (American Psychiatric Association, 2010; Patel et al., 2016; Petimar et al., 2024; UpToDate, 2024). For child and adolescent depression, bupropion is not a recommended treatment (Birmaher and Brent, 2007; MacQueen et al., 2016; Walter et al., 2023) given the lack of randomized controlled trials to assess safety and efficacy in young people.
Treatment with bupropion comes with safety concerns, particularly seizures, overdose, and high-dose toxicity (American Psychiatric Association, 2010; Finkelstein et al., 2013; Huecker et al., 2024; Snehal et al., 2023; Spiller et al., 2010). In the setting of overdose, seizures may occur and cardiovascular side effects are common, with potentially significant cardiac toxicity (Minns AB et al., 2024). Bupropion was ranked as one of the antidepressants posing the greatest risks to users in terms of poisoning severity (Hunter et al., 2022; White et al., 2008). In adolescents with suicide attempts by poisoning, bupropion was associated with higher rates of seizures and hospital admissions compared with TCAs and SSRIs (Overberg et al., 2019; Sheridan et al., 2018). Further, as with all antidepressants, bupropion has an FDA Boxed Warning for drug-induced suicidal thoughts and behaviors in children and young adults (Huecker et al., 2024) and therefore requires careful monitoring in patients with depression. Given safety concerns, bupropion is contraindicated in patients with anorexia or bulimia nervosa or with seizure disorders, including patients with factors predisposing to seizures (American Psychiatric Association, 2010; Huecker et al., 2024; Lee et al., 2021; Santarsieri and Schwartz, 2015). Certain formulations and higher doses of bupropion may also require greater caution (Huecker et al., 2024).
Given bupropion’s common and increasing use, multiple indications, alternative side-effect profile, and potential safety concerns, an updated description of the population starting bupropion treatment is needed. These details can inform future targets for safety investigations and whether efforts are needed to improve bupropion prescribing. Therefore, we sought to describe children, young adults, and adults initiating bupropion treatment from 2017 to 2022, including a characterization of the initial dispensed bupropion prescription, potential indication, patient characteristics, and treatment duration.
Methods
Datasource and study population
We used data from the MarketScan commercial claims database from 1/1/2016 to 12/31/2022, covering privately insured employees and dependents across the United States (Truven, 2024). MarketScan contains patient-level details on insurance enrollment and claims for inpatient services, outpatient services, and outpatient dispensed prescriptions. Prescription files include details on dispensed medication type, days supplied, and National Drug Code classification. Service visits contain dates, diagnostic and procedure codes, and clinician details.
Our sample included privately insured individuals aged 6–64 years who were newly initiating bupropion treatment. We defined new bupropion use as starting the medication after at least 1 year without any prior bupropion prescription dispensed. Continuous insurance enrollment coverage was required in the year before bupropion initiation.
Bupropion
Bupropion was ascertained with records of dispensed prescriptions for bupropion hydrochloride. Details on the initial bupropion prescription were collected including formulation (e.g., extended release), days’ supply, and strength. We did not include combination products (e.g., naltrexone-bupropion; dextromethorphan-bupropion).
Potential indication
We estimated the “potential indication” for bupropion treatment based on inpatient or outpatient ICD-10-CM diagnoses received on or within the 30 days prior to the day of bupropion initiation. Based on approved and off-label indications for bupropion, we included diagnoses for depression (including seasonal affective disorder), tobacco dependence and smoking cessation counseling, sexual dysfunction (proxy for use for antidepressant-induced sexual dysfunction), ADHD, bipolar disorder, and overweight or obesity (proxy for weight loss) (Supplementary Table S1). In addition to describing all diagnoses of potential indications, we used a hierarchical, mutually exclusive classification to summarize the potential indication (listed in decreasing hierarchical order, with approved indications first): (1) depression/seasonal affective disorder (SAD), (2) tobacco dependence, (3) sexual dysfunction, (4) ADHD, (5) bipolar disorder, and (6) obesity/overweight. For example, under the hierarchical classification, an individual with a recent depression and ADHD diagnosis would only be classified under depression.
Patient characteristics
The primary stratification variable was age group. We identified concurrent antidepressant treatment as a marker for use of bupropion as a possible adjunctive to SSRI or other antidepressant treatment. This was defined as individuals with an SSRI or other antidepressant prescription in the 15 days prior to or in the 15 days post bupropion initiation. Additional covariates were used to describe the study population and included sex, psychiatric and nonpsychiatric diagnoses, other dispensed prescription medications, psychotherapy services, recent (30 days) contact with a mental health clinician, and healthcare utilization (inpatient admissions, ED visits, outpatient visits). Patient characteristics were identified from inpatient and outpatient records, prescription records, and enrollment files dating back 12 months before treatment initiation.
Treatment duration
Length of bupropion treatment was estimated as the number of days between bupropion initiation and discontinuation based on prescription records. Discontinuation was defined as no prescription fill after the end of the prior prescription’s days’ supply, allowing a 30-day grace period. Treatment duration up to 1 year was evaluated.
Analysis
All analyses were stratified by age group: children (6–17 years), young adults (18–29 years), and adults (30–64 years), based on potential differences in prescribing practices, including formulation and indication, and on variation in risks. For example, young adults have elevated rates of substance use and overdose compared with other age groups, which relates to toxicity concerns for bupropion. We described the initial bupropion prescription, potential indication(s), potential use as antidepressant adjunctive, and patient characteristics of bupropion initiators. For the primary analysis, when considering potential indication, we allowed for multiple diagnoses and in a secondary analysis we considered potential indication under the hierarchical, mutually exclusive classification. We estimated treatment duration with Kaplan–Meier analysis, censoring at insurance disenrollment, treatment discontinuation, 1 year after treatment initiation, or end of available data. We reported the proportion and 95% confidence intervals of those remaining on bupropion treatment at 6 months and at 1 year by potential indication and by common mental health diagnoses. For secondary analyses, we stratified potential indications by sex and we compared potential indications of initiators in 2017 versus 2022 to determine if the population initiating bupropion treatment shifted during this 5-year time period. We additionally examined the potential indication in 2020 to determine if changes occurred during the beginning of the COVID-19 pandemic. The study was approved by the Rutgers University Institutional Review Board.
Results
Bupropion new users
We identified 39,833 children (6–17 years), 177,710 young adults (18–29 years), and 548,557 adults (30–64 years) newly initiating bupropion treatment from 2017 to 2022. The majority of initial dispensed prescriptions across age groups were for 30 days (79%–88%). Extended-release 24-hour 150 mg was the most common formulation and dose (62%) dispensed, followed by 12-hour sustained release 150 mg (16%), with variation by age (Table 1). In the youngest children (6–12 years), extended-release 150 mg was the most common (39%), followed by immediate release 75 mg (28%), and sustained release 100 mg (24%).
Characteristics of the Initial Bupropion Prescription Dispensed to Children, Young Adults, and Adults, 2017–2022
Children aged 6–12 years (n = 3480) vs. 13–17 years: extended release (40.1% vs. 64.8%), sustained-release (27.6% vs. 21.7%), immediate release (31.6% vs. 12.9%).
Young adults aged 18–24 years (n = 114,567) vs. 25–29 years: extended release (71.6% vs. 70.8%), sustained-release (19.5% vs. 20.8%), immediate release (8.7% vs. 8.1%).
Includes those filling multiple prescriptions on the date of initiation and missing/not classified formulation or strength.
<0.1% with multiple/other sustained-release or immediate-release dose.
The most common potential indication for all age groups was depression, with 57% of children, 47% of young adults, and 36% of adults having a recent depression diagnosis (Table 2). ADHD was the next most common potential indication in children (25%) and young adults (12%) vs. overweight or obesity in adults (16%). The younger children (6–12 years) were more likely to have ADHD as the potential indication (46%) than children aged 13–17 years (23%). A third (33%) of children, 26% of young adults, and 20% of adults were on concurrent SSRI treatment, with an additional 7%, 8%, and 10%, respectively, on concurrent non-SSRI antidepressant treatment.
Potential Bupropion Indication and Adjunct Antidepressant Treatment with Bupropion Treatment in Children, Young Adults, and Adults
Defined as antidepressant prescription dispensed in the 15 days prior to or 15 days post the initial bupropion dispensing.
Children aged 6–12 years vs. 13–17 years, potential indication (any diagnosis): depression (32.7% vs. 59.0%), ADHD (46.0% vs. 23.2%), bipolar disorder (2.1% vs. 3.8%), overweight/obese (5.0% vs. 4.6%).
Young adults aged 18–24 years vs. 25–29 years, potential indication (any diagnosis): depression (48.3% vs. 43.6%), smoking cessation (3.9% vs. 5.8%), ADHD (13.0% vs. 10.6%), bipolar disorder (4.9% vs. 4.0%), overweight/obese (7.5% vs. 12.6%).
ADHD, attention-deficit/hyperactivity disorder; SNRI, serotonin and norepinephrine reuptake inhibitors; SSRI, selective serotonin reuptake inhibitors.
Patient characteristics
The majority of bupropion initiators had at least one mental health diagnosis in the prior year (93% children, 86% young adults, 74% adults), with depression and anxiety as the most common diagnoses (Table 3). ADHD, adjustment disorders, and insomnia were also prevalent psychiatric diagnoses across age groups.
Selected Patient Characteristics of Children, Young Adults, and Adults Initiating Bupropion Treatment
NSAID, nonsteroidal anti-inflammatory drugs; SD, standard deviation; SNRI, serotonin and norepinephrine reuptake inhibitors; SSRI, selective serotonin reuptake inhibitors.
Over half of bupropion initiators received at least one other antidepressant in the year before starting bupropion (Table 2); among bupropion initiators with a depression diagnosis, prior antidepressant use was more prevalent (children 77%; young adults 68%; adults 69%). Psychotherapy claims in the prior year were present in 63% of children, 37% of young adults, and 21% of adults. In children, stimulants (26%), antipsychotics (17%), and hydroxyzine (16%) were also commonly dispensed before bupropion initiation. In adults (30–64 years), benzodiazepines (25%), opioids (31%), and NSAIDs (28%) were commonly dispensed before bupropion initiation.
Prior to bupropion dispensing, 0.7%–1.2% of all patients had a baseline epilepsy diagnosis and 6.3%–9.7% had a substance use disorder diagnosis (2.1%–4.5% alcohol use disorder diagnosis). In children, suicidal ideation (16.3%), self-harm (6.2%), poisoning events (5.9%), and anorexia nervosa or bulimia nervosa (2.2%) were relatively common diagnoses prior to bupropion initiation. These preexisting diagnoses were less common in young adults (4.8%, 1.4%, 2.6%, 1.2%) and adults (1.1%, 0.3%, 1.5%, 0.4%).
Treatment duration
Overall, 45% of children, 39% of young adults, and 45% of adults remained on bupropion treatment for at least 6 months, and approximately one-fourth (25%, 21%, and 28%, respectively) remained on treatment for at least 1 year (Supplementary Table S2; Supplementary Fig. S1). Treatment length varied by potential indication, with the shortest treatment duration for smoking cessation and the longest continued in bupropion users with depression (Figure 1).
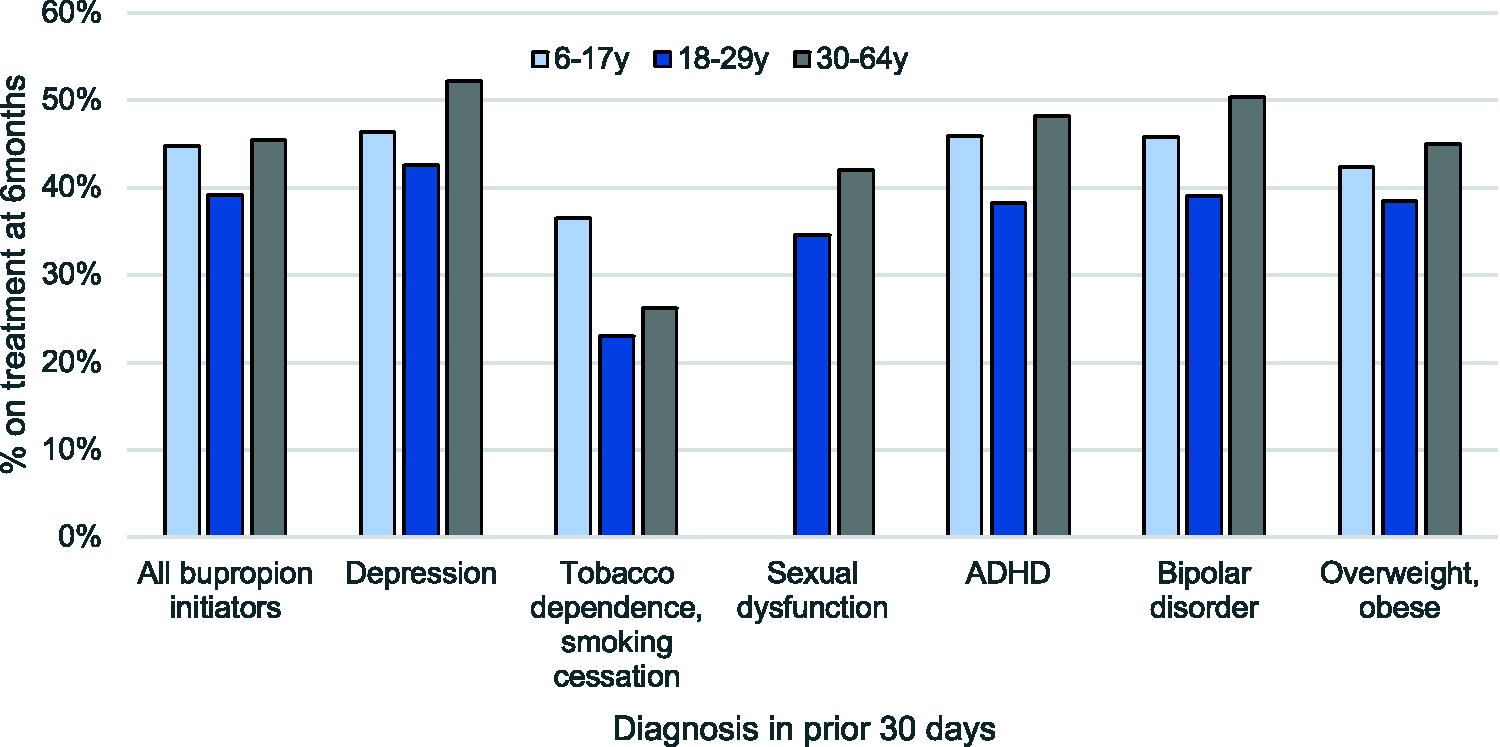
Percent of bupropion new users continuing treatment for at least 6 months by age and potential indication.
Additional analyses
Potential indication varied by sex, with generally more males having a potential indication for smoking cessation, sexual dysfunction, or ADHD than female bupropion initiators (Supplementary Table S3). In children, depression was a more common indication in female than male initiators (61% vs. 50%). From 2017 to 2022, there were increases in ADHD as a potential indication in children (24.9%–27.1%), young adults (10.5%–15.9%), and adults (4.0%–7.4%, Supplementary Table S4). There were slight reductions in smoking cessation in young adults and adults from 2017 to 2022, and a higher proportion of bupropion initiators had depression as a potential indication in 2020 compared with 2017 and 2022. The proportion with concurrent SSRI treatment was similar in 2017 and 2022 across age groups.
Discussion
The antidepressant bupropion is prescribed to privately insured children, young adults, and adults in the United States for a variety of indications, with the majority receiving a diagnosis of depression and having prior antidepressant use. While bupropion offers an alternative side effect profile compared with other antidepressants, there are safety concerns with bupropion, and certain populations may be elevated risk. We provide an updated description of individuals starting bupropion treatment; these details can assist in targeting bupropion safety and effectiveness investigations as rigorous, age-specific data remains necessary to guide prescribing decisions.
While available for decades, the indications for bupropion have expanded in recent years, including in children. We observed that bupropion is used for a variety of potential indications, with depression the most common indication in our sample and highest in children (57%). The frequent use of other antidepressants prior to bupropion initiation, particularly in those with depression, indicates that bupropion is typically not the first-line pharmaceutical option for depression. In adults, American Psychiatric Association guidelines include bupropion as a potential first-line pharmacotherapy option for adult depression (American Psychiatric Association, 2010). However, in pediatric depression guidelines, bupropion is not typically recommended or even mentioned (Cheung et al., 2018; MacQueen et al., 2016; Walter et al., 2023), in part related to limited data on bupropion efficacy and safety for pediatric depression (Clark et al., 2023). Bupropion is also used as an adjunctive treatment for depression, with 44% of surveyed psychiatrists selecting bupropion augmentation as the preferred intervention for partial SSRI response in adults (Goldberg et al., 2015). The high prevalence of both prior and concurrent antidepressant treatment in our sample of bupropion initiators may reflect clinician attempts to improve treatment effectiveness or address side-effects.
A quarter of children starting bupropion treatment had a recent ADHD diagnosis, a proportion that grew from 2017 to 2022. Bupropion may be viewed as a safer alternative to stimulant medications, which are scheduled controlled substances with a risk of misuse and abuse. While bupropion is not FDA approved for ADHD, meta-analyses found bupropion efficacious compared with placebo for adults and, in most measures, for children (Cortese et al., 2018; Ng, 2017; Verbeeck et al., 2017). Still, some of the evidence supporting bupropion for treating ADHD comes from poorly conducted trials, small sample sizes, and does not include long-term assessments (Clark et al., 2023; Verbeeck et al., 2017). In our sample, 39% of children, 22% of young adults, and 11% of adults had either a stimulant prescription or ADHD diagnosis in the prior year, indicating that a substantial proportion of bupropion users have ADHD, whether or not ADHD is the primary indication. This carries potential implications for polypharmacy, particularly in children. In the United States, stimulant use has increased in recent years, particularly following the COVID-19 pandemic (Chai et al., 2024). This increasing use contributed to stimulant shortages which began towards the end of our study period (U.S. Food and Drug Administration, 2023), these shortages may have impacted the use of bupropion for ADHD.
Diagnoses of prior seizures, anorexia and bulimia nervosa, self-harm, suicidal ideation, and poisonings in individuals starting bupropion treatment raise potential concerns. While infrequent in our sample, prior seizures are a contraindication for bupropion treatment given the risk of seizures at higher doses, which is further elevated for individuals with certain comorbidities and on concurrent medications (NAMI, 2016). Anorexia nervosa and bulimia nervosa are also contraindicated, with concerns over the increased risk of seizures and association with weight loss (American Psychiatric Association, 2010; Petimar et al., 2024). Substance use may increase seizure risk, with abrupt discontinuation of alcohol or sedatives a labeled contraindication. While we are unable to evaluate abrupt discontinuation, we did observe that 3.9% of bupropion new users (2.1% in children) had a diagnosed alcohol use disorder and 0.6% (0.7% in children) had a sedative, hypnotic, anxiolytic use disorder diagnosis. There are additional clinical concerns when prescribing bupropion that were not evaluated in this publication, including in individuals with risk factors for seizures, such as head injuries, stroke, and brain infections (Schachter SC, 2024) and concerns over potential drug–drug interactions (Reese et al., 2008; Sager et al., 2021). In certain clinical situations, caution is warranted in bupropion prescribing; the limited evidence on bupropion effectiveness and safety in younger populations underscores the need for further research.
The concern over bupropion treatment in individuals with prior poisonings or suicidal behaviors is related to the elevated risks of a subsequent overdose or self-harm event (Daly et al., 2020; Goldman-Mellor et al., 2020; Olfson et al., 2018, 2020) along with the toxicity of bupropion in the case of overdose (Huecker et al., 2024; Snehal et al., 2023). In adolescents with a suspected suicide reported to Poison Control, bupropion was more likely to be associated with death (0.2% vs. 0%), cardiopulmonary resuscitation (0.5% vs. 0.01%), and intubation (4.9% vs. 0.3%) compared with overdoses involving SSRIs (Overberg et al., 2019). In pediatric single-psychotropic substance ingestions, bupropion was associated with worsened clinical outcomes including treatment on a critical care unit (27%; SSRIs, 9%) and intubation/ventilation (4% vs. SSRIs, <1%) (Hunter et al., 2022). Importantly, any risk of antidepressant treatment should be weighed against the risk of no treatment (Fornaro et al., 2019; Shain and Committee On Adolescence, 2016) and it is imperative that individuals with depression and suicidal behaviors continue to receive necessary mental health care. Some individuals, particularly youth, need careful monitoring while on bupropion treatment.
Two-thirds of initial dispensed bupropion prescriptions were extended-release formulations. There is limited research on potential risk by formulation. In a 2005–2007 examination of patients hospitalized for high-dosage ingestion of bupropion extended-release, seizure occurred in 32% of patients at an average of 7.3 hours after ingestion (Starr et al., 2009). Comparatively, the incidence of seizures, and time to seizure, was lower in studies of cases that comprised a majority of sustained or immediate release formulations (Minns AB et al., 2024; Shepherd et al., 2004; Spiller et al., 1994). As such, there are recommendations for patients with a suspected overdose of extended-release bupropion to be monitored for at least 24 hours given the possibility of delayed seizures (Huecker et al., 2024; Starr et al., 2009). In a recent examination of bupropion overdoses presenting to a hospital (n = 437), 28% had a seizure and 2% (n = 8) had a seizure more than 8 hours after hospital arrival (Offerman et al., 2020). Late seizure events were associated with symptoms of tachycardia or altered mental status prior to the seizure (Offerman et al., 2020). Nevertheless, we are unaware of longitudinal data comparing risks across bupropion formulations, information critical to inform bupropion-related prescribing and care.
The literature on bupropion’s alternative side effect profile related to weight gain and antidepressant-induced sexual dysfunction compared with other antidepressants could improve treatment initiation and adherence for certain patients. For example, in adults with MDD, bupropion and SSRIs were effective, but bupropion had lower rates of sexual-dysfunction side effects (Thase et al., 2005). Further, a 2024 study found bupropion treatment was associated with the lowest risk of weight gain of all antidepressants considered (Petimar et al., 2024), with higher bupropion adherence at 6 months (41% vs. 28%–36% in other antidepressants) but not by 12 months (17% vs. 16%–21%) (Petimar et al., 2024).
Given potential benefits of bupropion for certain patients, it will be important to examine safety concerns, especially for populations and indications lacking effectiveness data. We know from prior literature that seizures are a safety concern with bupropion and that, among those with an antidepressant overdose, those involving bupropion tend to be more severe (Finkelstein et al., 2013; Hunter et al., 2022; Overberg et al., 2019; White et al., 2008). However, we do not have quality data on the actual risk of an overdose, seizure, or other adverse outcome in individuals starting bupropion treatment, nor do we know which individuals are at the highest risk. This information is needed for patients, caregivers, and clinicians to make informed treatment decisions.
Our analyses are based on dispensed prescription records for bupropion. We cannot determine the timing of the prescription and if medications were consumed, and prescriptions filled outside of insurance would be missed. Our sample included privately insured individuals aged 6–64 years; patterns of bupropion use may differ in publicly insured and uninsured individuals and in individuals 65+ years of age. Further, future research could examine prescribing variation across finer age stratification than the three age groups we examined. To capture initiation for smoking cessation, we used an expanded definition and included codes for tobacco dependence and smoking cessation counseling. As diagnoses are not directly linked with prescriptions, we do not know the precise indication for bupropion initiation. Our potential indication variable is limited as a fourth of bupropion initiators in our sample did not have a potential indication identified from baseline diagnoses. We believe this is related to the fact that some indications lack direct diagnostic codes (e.g., obesity and overweight ICD-10-CM codes to proxy for weight loss; sexual dysfunction codes used broadly for antidepressant-induced sexual dysfunction), indications and diagnoses are not always recorded in antidepressant users (Mojtabai and Olfson, 2011; Zito et al., 2020), and bupropion may have been prescribed for conditions outside our potential indications. Finally, we cannot discern the clinical contexts of individual prescribing decisions.
As the prescribing of bupropion becomes more widespread, it is vital to understand how and to whom the medication is being prescribed. Additional safety and effectiveness data is needed to further inform the prescribing of bupropion to children, young adults, and adults. Concerns are particularly heightened given the toxicity of bupropion and the use in individuals with prior suicidal behaviors or in populations without known efficacy.
Footnotes
Disclaimer
The funding agencies had no role in the conduct of the study; design, management, analysis, or interpretation of the data; and preparation, review, or approval of the article.
Disclosures
H.S. reports consulting fees from The Pew Charitable Trusts and the American Society of Addiction Medicine. G.B., M.O., D.H., E.S., and D.C. report no conflicts of interest.
Supplementary Material
Supplementary Figure S1
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
