Abstract
Introduction:
Treatment studies in FMR1 knockout rodent models have found that minocycline and lovastatin each improve synaptic, neurological, and behavioral functioning, and open-label chronic dosing studies in human patients with fragile X syndrome (FXS) have demonstrated modest clinical improvements. Findings from blinded studies are mixed, and there is a limited understanding of electrophysiological target engagement that would facilitate cross-species translational studies. Smaller-scale, acute (e.g., single-dose) drug studies may speed treatment identification by detecting subtle electrophysiological and behavioral changes.
Materials and Methods:
Twenty-nine participants with FXS (31% female) ages 15–45 years completed a randomized, double-blind, crossover study in which they received a single oral dose of 40 mg of lovastatin, 270 mg of minocycline, or placebo, with a 2-week washout period between dosing visits. Participants completed a comprehensive neuropsychological battery and three EEG paradigms (resting state; auditory chirp; auditory habituation) before and 4 hours after dosing.
Results:
No serious adverse events were reported, and both drugs were well-tolerated. Compared with placebo, there were no overall treatment effects for any outcomes, including EEG, but several modest drug responses varied as a function of sex and age. Lovastatin treatment was associated with improved spatial awareness in older participants and females compared with minocycline and placebo.
Discussion:
We show that single-dose drug studies are highly feasible in FXS and that patients with FXS can complete a range of EEG and behavioral tasks, many of which have been shown to be reliable and may therefore be sensitive to subtle drug target engagement.
Conclusions:
Acute single doses of lovastatin or minocycline did not lead to changes in electrophysiological or performance-based measures. This may be due to the limited effects of these drugs in human patients or limited acute effects relative to chronic dosing. However, the study design was further validated for use in neurodevelopmental populations.
Introduction
Fragile X syndrome (FXS) is the most common inherited form of intellectual disability and monogenetic cause of autism spectrum disorder (Hunter et al., 2014). Despite its prevalence and impact, there are no FDA-approved treatments for FXS, and promising drugs studied in animal models of FXS have failed to translate into approved treatments in humans (Berry-Kravis et al., 2018). Current pharmacological strategies focus on symptom management (e.g., SSRIs for anxiety) (Erickson et al., 2017).
Since 2016, our NIH U54 Fragile X Center has advocated for the benefits of parallel acute, single-dose drug studies in FMR1 knockout (KO) mice and human patients with FXS. As demonstrated in our recently published study of baclofen, single-dose designs utilizing our established cross-species neurophysiological biomarkers allow for the detection of small electrophysiological changes (e.g., parallel reductions in gamma power) indicative of potential mechanisms (Jonak et al., 2022; Lovelace et al., 2020; Norris et al., 2022; Pedapati et al., 2022; Smith et al., 2021; Wang et al., 2017). This allows for efficient testing of hypothesized neurophysiological mechanisms and serves as a rapid screening tool for potential therapeutic drugs before expanding to more time- and cost-intensive chronic dosing studies. Further, the single-dose model has shown promise in other neuropsychiatric conditions, including schizophrenia (Lahti et al., 2005; Light et al., 2017), hyperactivity (Di Martino et al., 2004), and suicidal ideation (Wilkinson et al., 2018). Last, our family advisory council, comprised of caregivers and self-advocate stakeholders, has identified the single-dose design as strongly acceptable and feasible for their families.
Thus, the present study used a single-dose design to study the acute effects of minocycline and lovastatin, two medications that have shown promise for the treatment of FXS across preclinical and human studies. Minocycline is an FDA-approved tetracycline antibiotic with neuroactive properties as an antagonist of matrix metalloproteinase-9 (MMP-9), a dendritic protein that helps regulate the extracellular environment and supports dendritic spine development (Wang et al., 2008). Low levels of fragile X messenger ribonucleoprotein (FMRP) are associated with high levels of MMP-9, and minocycline lowers MMP-9 (Bilousova et al., 2009; Dziembowska et al., 2013; Ethell and Ethell, 2007). Empirical studies of FMR1 KO mice have shown that minocycline treatment is associated with cellular (Bilousova et al., 2009), electrophysiological (Dansie et al., 2013; Lovelace et al., 2020), and behavioral benefits (Bilousova et al., 2009; Dansie et al., 2013; Rotschafer et al., 2012). Specifically, previous findings indicate that minocycline increases gamma-band phase locking to auditory chirp stimuli in FMR1 KO mice, but it does not reduce resting-state gamma power beyond the effect seen in placebo (Lovelace et al., 2020). Lovastatin is an HMG-CoA reductase inhibitor that is FDA-approved for the treatment of hyperlipidemia. In models of FXS, lovastatin may correct excess protein synthesis by reducing excessive extracellular signal-regulated kinase (ERK1/2) activity (Asiminas et al., 2019; Hou et al., 2006; Muscas et al., 2019; Osterweil et al., 2013). Like minocycline, empirical studies of lovastatin in FMR1 KO mice have shown normalized electrophysiology (Muscas et al., 2019; Osterweil et al., 2013) and cognitive benefits (Asiminas et al., 2019; Yau et al., 2016).
Human clinical trials of minocycline and lovastatin in FXS have identified modest positive effects on the Clinical Global Impressions-Improvement (CGI-I) scale in some patients with FXS, but insights into the modifiers of treatment response (e.g., age, sex, FMRP expression) are limited (Çaku et al., 2014; Champigny et al., 2021; Leigh et al., 2013; Paribello et al., 2010; Thurman et al., 2020). Given that there are very few blinded, placebo-controlled trials of either drug in FXS (one blinded study for each drug) (Leigh et al., 2013; Thurman et al., 2020), additional blinded studies are needed to assess the reproducibility of previous positive results from open-label studies. Furthermore, insufficient backtranslation of common clinical outcomes from these previous studies like the CGI-I into the FMR1 KO model hinders cross-species studies that facilitate further treatment refinement (e.g., dosing, optimal developmental timing). There is limited knowledge of the impact of either drug on biomarkers that translate across animal models and humans, such as resting-state and sensory-evoked EEG outcomes. Our group and others have argued for the translational utility of these EEG measures as key biomarkers for treatment development in FXS (Ethridge et al., 2019; Jonak et al., 2022; Kenny et al., 2022; Schneider et al., 2013). In FXS, EEG outcomes translate well across animal and human studies (Jonak et al., 2022; Pedapati et al., 2022) and are reliable (Liu et al., 2023), clinically relevant (Ethridge et al., 2019; Pedapati et al., 2022; Schmitt et al., 2022; Wang et al., 2017), and sensitive to single-dose treatment effects (Jonak et al., 2022). EEG outcomes also may help overcome the challenges of placebo effects in past human clinical trials in FXS (Erickson et al., 2017; Luu et al., 2020). Blinded treatment studies of minocycline or lovastatin in humans that build on EEG findings from treatment studies in rodents are sparse but a key next step in assessing the clinical value of these drugs.
To address these knowledge gaps, we used an extensive clinical and neurophysiological battery to test the effects of a single dose of either minocycline or lovastatin compared with placebo in adolescents and adults with FXS. We hypothesized that, compared with placebo, both minocycline and lovastatin treatments would be associated with increased auditory chirp synchronization. We also conducted exploratory analyses of several behavioral outcomes validated for use in FXS clinical trials.
Materials and Methods
Study design and dosing information
This was a randomized, double-blind, placebo-controlled, single-dose, crossover study of minocycline and lovastatin conducted at Cincinnati Children’s Hospital Medical Center. The study protocol was reviewed and approved by the local Institutional Review Board and registered at clinicaltrials.gov (NCT02998151), and the trial was overseen by an independent data safety monitor. Potential participants were eligible if they were: (1) 15–55 years of age, and (2) had a diagnosis of FXS, defined as having the full FMR1 mutation (>200 CGG repeats), confirmed via past testing results made available in a participant’s medical record or via Southern Blot and/or PCR conducted in collaboration with the Molecular Diagnostic Laboratory at Rush University. Potential participants were excluded if they had: (1) a history of intolerance to lovastatin or minocycline; (2) taken any investigational drug within 3 months; (3) a history of substance abuse or dependence within 6 months; (4) a significant psychiatric or central nervous system neurological disease unrelated to FXS; (5) uncontrolled seizures and/or were currently taking anticonvulsants, barbiturates, lithium, or benzodiazepines; (6) changed any psychiatric medications within 4 weeks of testing; (7) for biological females of childbearing potential, a positive urine pregnancy test; and (8) a creatinine clearance <50 mL/min. Parents, caregivers, or other legally authorized representatives provided written consent for participants younger than 18 years of age or for participants who were unable to provide consent due to limited decision-making capacity stemming from their intellectual disability. Participants provided their written consent when possible and otherwise provided verbal assent when possible.
All participants completed a screening visit to characterize baseline physical and psychiatric functioning. Days between the baseline visit and first dosing visit ranged from 1 to 56 days (mean = 21 days; standard deviation = 16 days). An investigational pharmacist responsible for dispensing the medication generated a randomization schedule and was the only individual unblinded to treatment assignment/order. Either lovastatin (40 mg), minocycline (270 mg extended release [ER]—ER selected due to its tolerability), or placebo were orally administered at each dosing visit following completion of predose clinical assessments. Postdose measures were completed four hours after dosing (Table 1). A 2- or 4-week washout period separated each dosing visit (Fig. 1). This washout period varied because the present study was a part of a larger single-dose study that also included a fourth, previously published, baclofen treatment condition (Jonak et al., 2022); for example, if a participant received baclofen treatment at their second dosing visit, there were four weeks separating their first and third visit. Crossover effect differences between a 2- or 4-week washout period were expected to be minimal given the acute/single-dose design and relatively rapid washout period of each drug. One day and 7 days after each dosing visit, participants completed a phone call with a study coordinator to provide information about side effects or other adverse events.
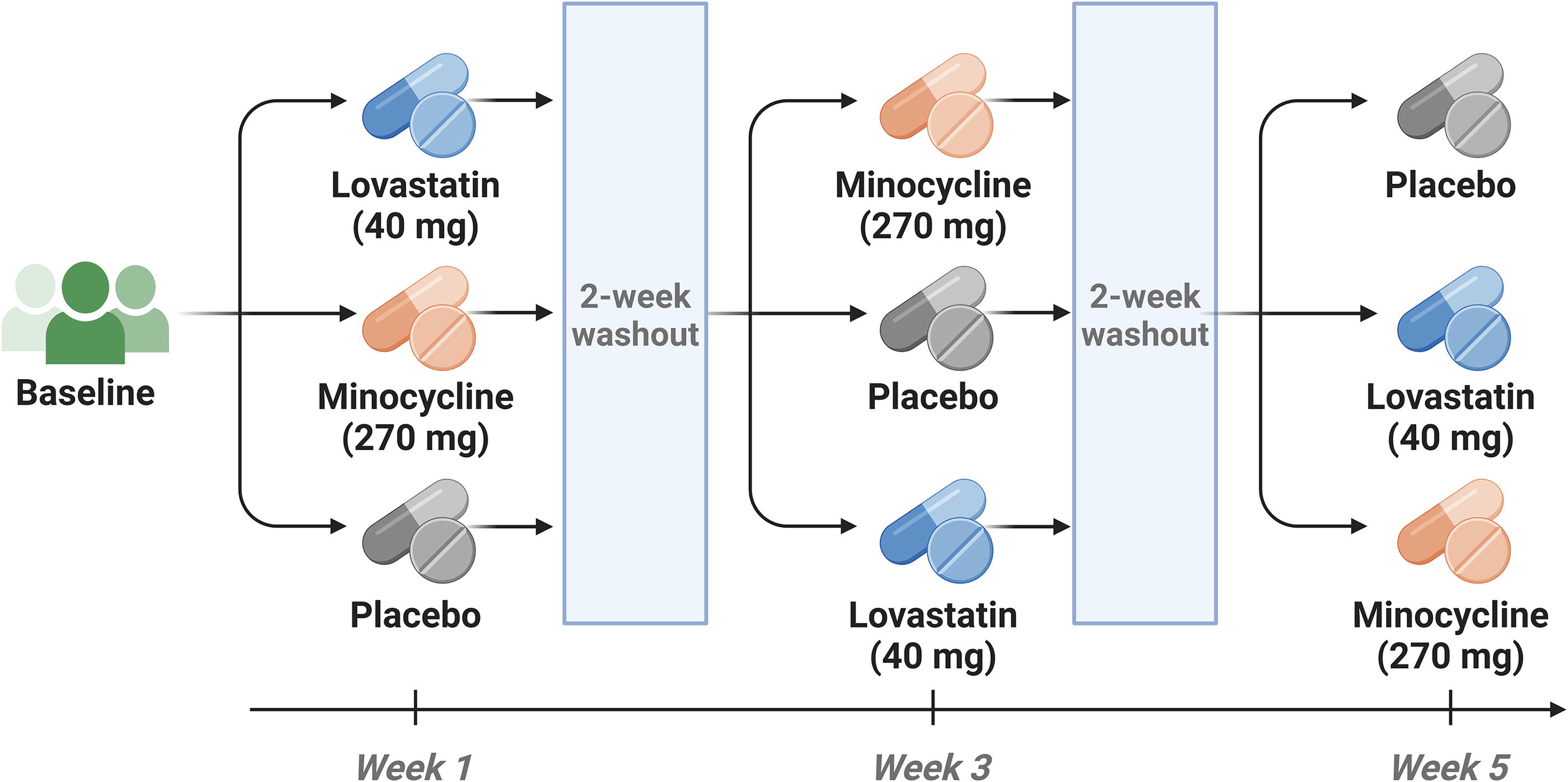
Study timeline and dosing schedule.
Schedule of Measures
Procedures
EEG
EEG recordings were obtained using our previously reported behavioral methods (Liu et al., 2023). EEG recordings were collected with a Phillips/EGI Net Amps 400 system (Eugene, Oregon, USA) using a 128-channel Hydrocel saline-based electrode net sampled at 1000 Hz.
Five minutes of resting-state EEG data were collected while participants sat with their eyes open in a quiet room and watched a silent video of their choosing. Resting-state EEG raw data were filtered using methods similar to our previously reported parameters (Pedapati et al., 2022), and full preprocessing details are reported in Supplementary Materials. Following source localization, relative band power was calculated for the following bands of interest: delta (2–3.5 Hz), theta (4–7.5 Hz), alpha (8–12.5 Hz), beta (13–29.5 Hz), low gamma (30–55 Hz), high gamma (65–80 Hz). To minimize the number of comparisons and to align analyses with our previously published single-dose baclofen trial (Jonak et al., 2022), the Desikan-Killiany atlas was used to generate average power for left and right frontal/prefrontal, temporal, parietal, and occipital lobes for each band (i.e., averaged across all regions of interest subsumed by that lobe) (Supplementary Figure S1, reproduced with permission from Pedapati et al., 2022).
During the “chirp” task, participants listened to a 2-second chirp-modulated tone designed to induce synchronized brain oscillations. Chirp stimuli and EEG data processing are identical to our previously reported methods (Jonak et al., 2022). Chirp outcomes included intertrial phase coherence (ITPC) to the chirp stimulus centered at 40 Hz, ITPC to the chirp stimulus centered at 80 Hz, and average single-trial power (not baseline-corrected) in the low-gamma (30–60 Hz) and high-gamma (61–100 Hz) bands.
During the auditory habituation task, participants listened to four white noise bursts separated by 500 ms interstimulus intervals designed to assess the habituation of the neural response to repeated stimuli. Auditory habituation stimuli and EEG data processing are identical to our previously reported methods (Ethridge et al., 2019). For event-related potential (ERP) analyses (N1, P2), habituation was characterized as the change in ERP amplitude relative to the initial stimulus. Auditory habituation outcomes included habituation of N1/P2 response and average single-trial power in the low-gamma (30–60 Hz) and high-gamma (61–100 Hz) bands.
Eye-Tracking
Our eye-tracking paradigms included two tasks: a face-viewing task and a social preference task. Procedural and preprocessing details for both tasks are identical to those previously reported by our group (Hong et al., 2019; Reisinger et al., 2019; Shaffer et al., 2017).
During the face-viewing task, participants viewed a set of adult human faces from the NimStim Face Stimulus Set (Tottenham et al., 2009), while their visual fixations were tracked using an infrared camera (Tobii Technology, Sweden). Face-viewing outcomes were averaged across all possible faces (i.e., male, female, emotional valence). Outcomes of interest were percent time viewing the face, percent time viewing the eyes, and percent time viewing the mouth (all calculated from the total time viewing the screen).
During the social preference task, participants viewed two simultaneous, side-by-side videos (one depicting socially salient information and the other depicting geometric/non-social information) while the location of their gaze was recorded in real time. Outcomes of interest were percent time viewing the social video and percent time viewing the geometric video (both calculated from the total time viewing the screen).
Test of Attentional Performance for Children
The Test of Attentional Performance for Children (KiTAP) is a computer-based measure of executive functioning and includes measures of processing speed, attention, inhibitory control, and cognitive flexibility. The KiTAP has been validated for use in adults with FXS (Knox et al., 2012). Participants completed four KiTAP subtests: Alertness (processing speed), Distractibility (attention), Go/No-Go (response inhibition), and Flexibility (cognitive flexibility). Prior to each subtest, participants received verbal instructions and completed a practice task to ensure comprehension.
Four KiTAP outcomes with the strongest evidence for reliability, validity, and relation to neurophysiology in FXS were selected based on findings from previous studies of FXS (Budimirovic et al., 2017; Schmitt et al., 2022): Alertness median reaction time, the number of Distractibility commission errors, the number of Go/No-Go commission errors, and Go/No-Go median reaction time.
Aberrant Behavior Checklist–Community
The Aberrant Behavior Checklist–Community (ABC) is a caregiver report capturing the most common areas of caregiver concern in FXS (Sansone et al., 2012): irritability/aggression, lethargy, stereotypy, hyperactivity, inappropriate speech, and social avoidance. The ABC was completed during the baseline visit to quantify baseline levels of caregiver-reported symptom severity.
The Repeatable Battery for the Assessment of Neuropsychological Status
Participants only completed the List Learning subtest of the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS), a brief measure of verbal working memory. During this task, participants are asked to repeat as many words as they can from a list of words read to them. Raw scores were analyzed for the RBANS (i.e., total number of words remembered across all five trials).
Woodcock-Johnson III Tests of Cognitive Abilities
Participants completed the Woodcock-Johnson III Tests of Cognitive Abilities (WJ-III) Spatial Relations and Auditory Attention subtests during their baseline visit and postdose to assess visuospatial and auditory attention skills. Comparison of postdose performance to the baseline visit minimized practice effects that emerged during same-day testing for the WJ-III. The Spatial Relations subtest measures visual feature detection, mental rotation, and matching. The Auditory Attention subtest measures selective auditory attention and requires individuals to identify words they hear amid increasingly loud background noise. Age-normed standard scores were analyzed for the WJ-III.
Statistical analyses
Linear mixed effects models with repeated measures were used to compare outcomes across all three drug conditions. This method allowed for the simultaneous modeling of drug and sequence (i.e., drug order) effects while avoiding the listwise removal of participants with missing data (i.e., participants who did not complete all three conditions: N = 2 for placebo and N = 2 for minocycline). The subject was the random effect in all models. Predictors for all models included drug, phase (baseline or predose, and postdose), and visit number/order. The interaction between drug and visit was included to test for crossover. Age and sex were included as covariates in all models. Age- and sex-dependent (Age × Drug; Sex × Drug) drug effects were also assessed for all outcomes. Resting-state EEG models also included Frequency band × Drug (delta, theta, alpha, beta, low gamma, high gamma) and Region of Interest (ROI) × Drug interaction terms, as well as accompanying age- and sex-associated interactions (e.g., Frequency band × Drug × Sex). Similarly, habituation EEG models of N1 and P2 amplitude also included Repetition × Drug (initial, second, third, or fourth stimulus) as well as accompanying age- and sex-associated interactions (e.g., Repetition × Drug × Sex). The models were designed using baseline measurements in a crossover design. This approach involved assigning a dummy treatment value for baseline measurements (baseline or predose, depending on the specific outcome) for each visit (Jones and Kenward, 2015). p-Values <0.05 were considered significant. No adjustments were made for multiple comparisons because this was a pilot study.
We calculated the test–retest reliability of our EEG measures using our previously reported methods (Liu et al., 2023). In short, we calculated a same-day intraclass correlation coefficient (ICC) using a two-way simple random effects model (using a single measure, absolute agreement), or ICC(A,1), within each drug condition.
Except ICC analyses, all statistical analyses were conducted using SAS® version 9.4 (SAS Institute Inc., Cary, NC). ICC analyses were conducted using the irr R package (Gamer et al., 2022). All data were visualized using the ggplot2 R package (Wickham, 2016). R analyses used R version 4.3.1 (R Core Team, 2023).
Results
Participant demographics
Twenty-nine individuals participated in the study, with 26 completing all three treatment arms (Fig. 2). The trial was terminated early due to the onset of the COVID-19 pandemic. Altogether, 27 participants completed the placebo condition, 29 completed the lovastatin condition, and 27 completed the minocycline condition. Reasons for early withdrawal from the study included: difficulties with transportation to the study site (N = 1), combined difficulties with transportation to the study site and irritability/difficulty tolerating the length of testing procedures (N = 1), and inability to be contacted (i.e., lost to follow-up; N = 1). Participants who completed each condition did not differ on age, sex, IQ, or mosaic status (all p > 0.98). Demographics and baseline clinical characteristics are available in Table 2. Baseline laboratory data are available in Supplementary Table S1 (Supplementary Data S1).
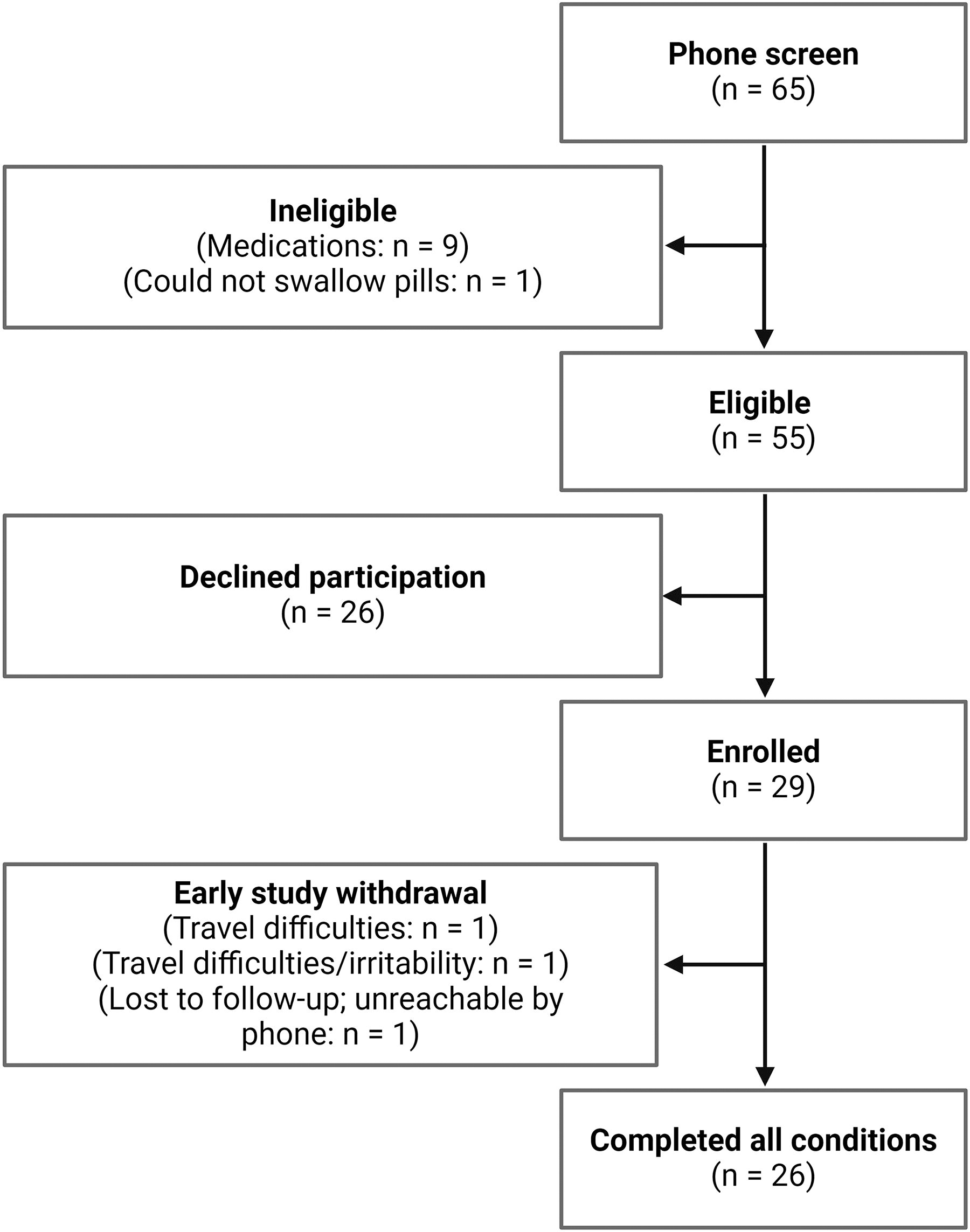
Participant screening and enrollment.
Demographic Information and Baseline Characteristics
IQ, intelligence quotient; M, mean; N, sample size; SD, standard deviation.
Five participants (17.2%) did not use any concomitant medication during the study period, while all remaining participants used at least one concomitant medication, as is typical in FXS (Dominick et al., 2021) (Supplementary Table S2 in Supplementary Data S1).
Safety
All treatments were well tolerated with no drug-associated increased risks noted (see Supplementary Materials for full details).
Electrophysiological target engagement
No treatment order (i.e., crossover/sequence) effects were observed for any EEG outcome.
Resting-state EEG
In females, global resting-state theta band power was increased on lovastatin compared with placebo (t 6214.53 = 2.52, p = 0.012). This pattern in females was driven by a large reduction in theta power postdose in the placebo condition and a very small increase in theta power postdose in the lovastatin condition. There were no other treatment effects for resting-state EEG power (Fig. 3). Consistent with previous literature in FXS (Pedapati et al., 2022; Smith et al., 2021; Van der Molen and Van der Molen, 2013; Wang et al., 2017), females showed greater alpha power and lower theta power than males regardless of treatment condition or brain region (Fig. 4). Females also had lower delta power than males regardless of treatment condition or brain region.
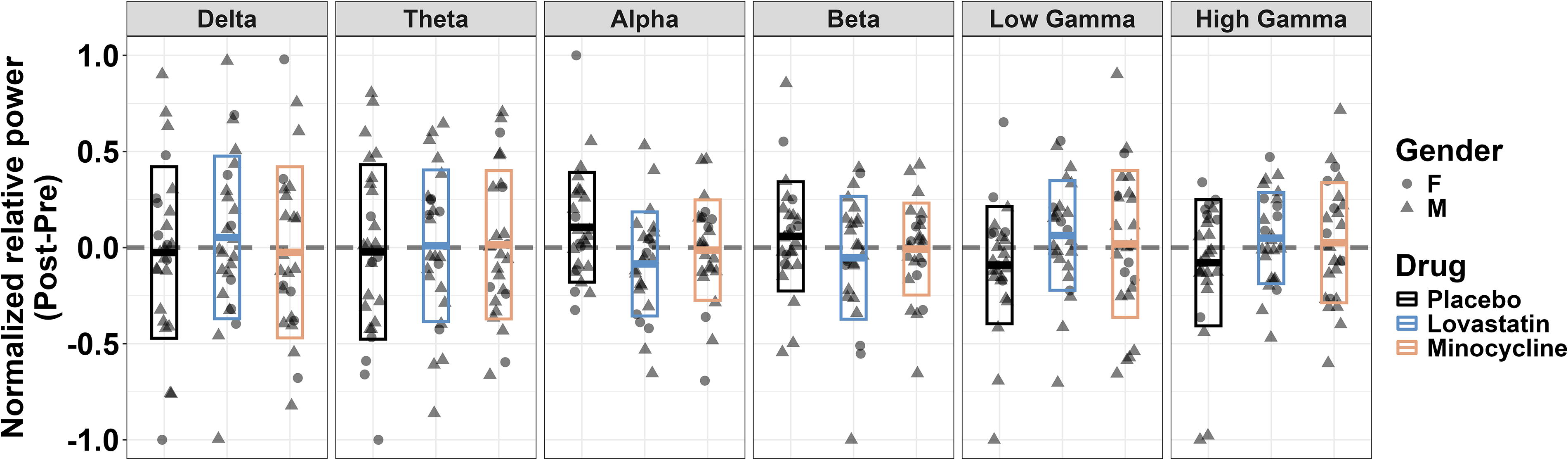
Null effects of treatment across all resting-state EEG frequency bands.
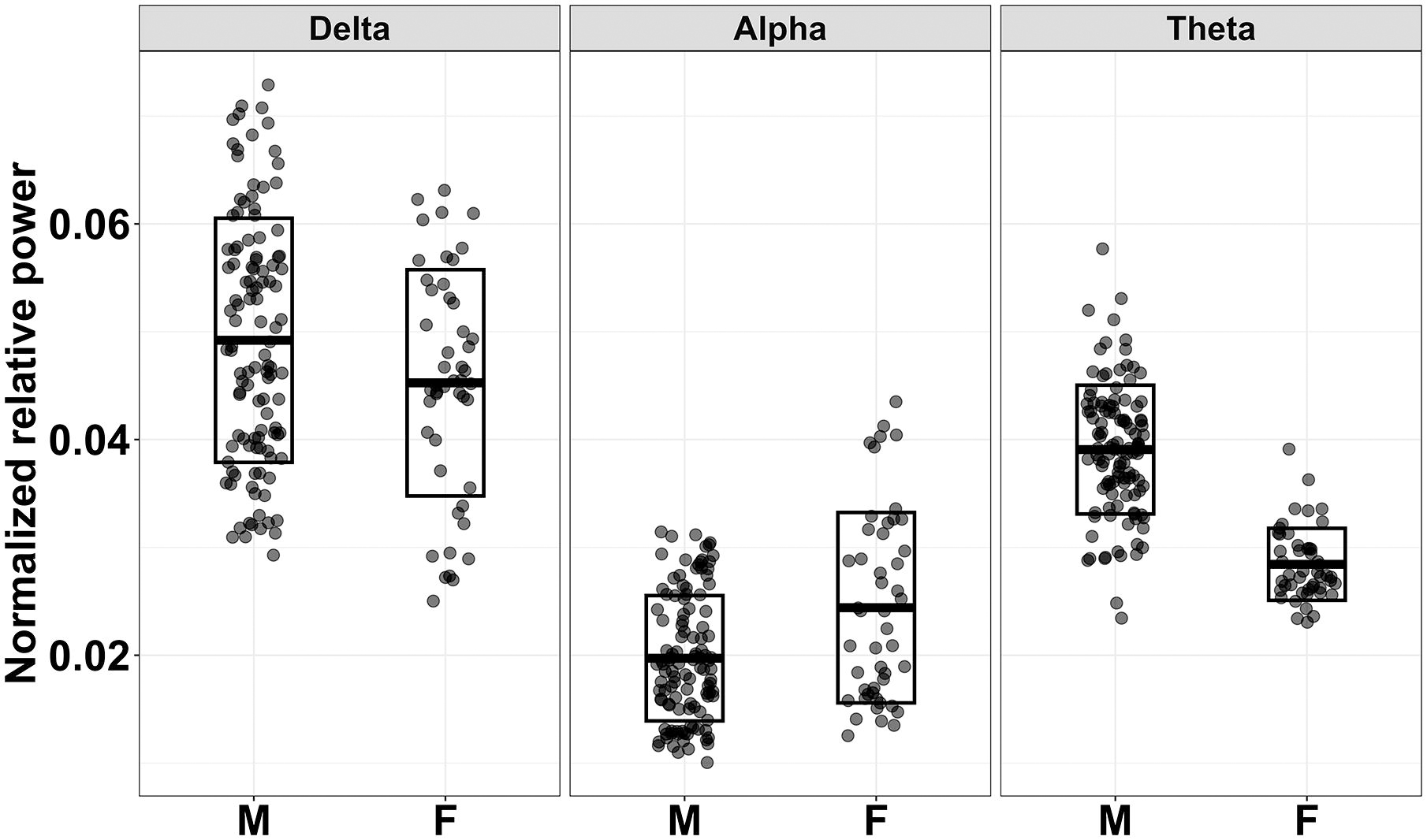
Sex differences in resting EEG power, averaged across brain regions/ROIs. Females with FXS showed greater alpha power, lower theta power, and lower delta power than males with FXS regardless of treatment condition or brain region. All comparisons of males and females for the depicted bands were significant at p < 0.05. FXS, fragile X syndrome; ROI, region of interest.
Auditory chirp EEG
There were no effects of treatment for any auditory chirp outcome (ps > 0.20). Neither sex nor age moderated treatment effects for auditory chirp EEG outcomes (ps > 0.12).
Auditory habituation EEG
There were no effects of treatment for any auditory habituation outcome (ps > 0.87). There was a modest age-dependent drug effect for habituation of P2 amplitude (Drug × Repetition × Age: F (16,407.443) = 1.819, p = 0.027). Although post hoc analyses (i.e., associations between age and P2 amplitude) were not significant (ps > 0.21), this effect was driven by older participants showing slightly elevated P2 amplitudes across stimulus repetitions following placebo, whereas P2 amplitudes were similar for all participants pre- and post- minocycline and lovastatin treatments.
Exploratory behavioral analyses
Eye-Tracking
There were no overall treatment effects for any eye-tracking outcome (ps > 0.308). Moderating effects of sex on treatment are reported in the Supplementary Materials and should be interpreted with caution due to the small number of female participants (N = 5) available with valid eye-tracking data.
KiTAP
There were no overall treatment effects for any KiTAP outcome (ps > 0.19). However, following placebo, older participants made a greater number of errors on the KiTAP Distraction (Drug × Age: F (4,104.713) = 8.064, p < 0.001) and Go/No-Go subtests (drug × age: F (4,98.247) = 2.821, p = 0.029), whereas performance did not decline in any participants following minocycline and lovastatin treatment.
RBANS
There was no overall treatment effect for the number of words remembered on the RBANS List Learning subtest (F (2,81.659) = 0.121, p = 0.886). There also were no moderating effects of sex or age on treatment.
WJ-III
There was no overall treatment effect for either WJ-III subtest (p’s > 0.073). However, both age (F (2,43.419) = 4.530, p = 0.016) and sex (F (2, 42.220) = 5.539, p = 0.007) moderated treatment response on Spatial Awareness. Specifically, lovastatin was associated with improved spatial awareness in older participants compared with minocycline and placebo (Supplementary Figure S2). Lovastatin was also associated with improved spatial awareness in female participants relative to males and relative to females in the other treatment conditions.
Test–retest reliability of EEG
ICC values for all three drug conditions (for resting-state, low and high gamma were averaged together to allow direct comparison to our previous study), and all EEG tasks are reported in Table 3. For resting-state power, same-day test–retest reliability (ICC values) for all three conditions and all frequency bands except gamma were similar between the current and our previous analysis in FXS (Liu et al., 2023). For resting-state gamma power, our ICC value for placebo (ICC = 0.693) was slightly lower than previously reported (ICC = 0.814). There was even greater within-subject same-day variability in gamma power for the lovastatin (ICC = 0.560) and minocycline (ICC = 0.577) conditions compared with our placebo condition and the ICC value previously reported.
Comparison of Same-Day (4-Hour Test–Retest) EEG Intraclass Correlation Coefficients
ICCs for N1, P2, and ERP onset only were calculated for responses to the initial stimulus.
ERP, event-related potential; ICC, intraclass correlation coefficients; ITC, intertrial coherence.
Discussion
We conducted a randomized, double-blind, placebo-controlled, crossover study of single-dose minocycline and lovastatin in patients with FXS to assess acute electrophysiological target engagement. Our secondary goal was to assess subtle behavioral and cognitive response occurring in response to treatment and to examine possible moderators of treatment response. Although we did not find any overall treatment effects for primary EEG or secondary behavioral outcomes, several small behavioral treatment effects varied as a function of age and sex, and all drugs were well-tolerated without significant adverse events.
Single-dose clinical trial models for FXS
First, consistent with our previously reported study of single-dose baclofen (Jonak et al., 2022), we demonstrate the strong feasibility of this single-dose drug challenge study design. Twenty-six out of 29 participants completed all three treatment arms. All participants were able to successfully complete our EEG tasks, which we attribute to our previously reported extensive behavioral supports and EEG technicians with expertise in testing neurodevelopmental populations (Liu et al., 2023).
Given (1) the abundance of preclinical and open-label multiple-dose studies describing the benefits of lovastatin and minocycline in FXS (Asiminas et al., 2019; Bilousova et al., 2009; Çaku et al., 2014; Champigny et al., 2021; Dansie et al., 2013; Lovelace et al., 2020; Osterweil et al., 2013), and (2) our findings of acute EEG effects of single-dose baclofen (Jonak et al., 2022), we were cautiously optimistic at study onset regarding the potential for treatment-dependent changes in EEG signal following acute dosing of lovastatin or minocycline. Our null findings may indicate that behavioral and EEG effects of minocycline and lovastatin from preclinical studies of FXS do not translate to human patients. Almost all studies of each drug in humans with FXS have been open-label trials using caregiver- and clinician-rated endpoints, which are more susceptible to placebo effects (Erickson et al., 2017; Luu et al., 2020). The only randomized, placebo-controlled, double-blind trial of lovastatin in humans with FXS found null drug effects (Thurman et al., 2020). Similarly, the only randomized, placebo-controlled, double-blind trial of minocycline in human patients found modest treatment effects on the CGI (about 0.5 points improvement on minocycline) but null effects for all other a priori outcomes (ABC, Vineland, Expressive Vocabulary, Visual Analog Scale) (Leigh et al., 2013). Thus, given the minimal behavioral effects of either drug seen in these double-blind studies, our null findings using EEG- and performance-based measures replicate and extend these findings in humans with FXS. However, given the unique single-dose design of our study, we also reiterate three other possible explanations for these null group-level treatment effects (Erickson et al., 2017).
First, a single dose of lovastatin or minocycline may be insufficient to drive electrophysiologic or behavioral change. Much of the preclinical evidence points toward treatment-dependent molecular benefits operating on relatively longer timescales that may require chronic dosing or extended observation windows (e.g., reductions in excess protein synthesis; changes in dendritic spine morphology). Although the extent to which these mechanisms translate to humans is not fully understood, these specific drugs may need multiple doses to drive molecular and electrophysiological change and facilitate behavioral improvements. Second, single-dose effects for lovastatin and minocycline may only occur in younger cohorts during a sensitive period of enhanced neural plasticity. Third, effective treatment for FXS may require the simultaneous use of multiple drugs or a combination of drugs and behavioral therapy. Consistent with this, the largest treatment effects for lovastatin and minocycline seen in humans to date were observed in chronic dosing studies combining both treatments (Champigny et al., 2021).
Ultimately, our findings indicate that single-dose studies are feasible in FXS. Clinical trial designs, however, must be thoughtfully paired with drugs based on their hypothesized mechanisms. We are currently engaged in a follow-up study of several different drugs (baclofen, memantine, roflumilast) that we believe could be more sensitive to acute neurophysiological effects (NCT05418049).
Reliability of EEG outcomes
As shown in Table 3, our findings are also in keeping with the previously demonstrated good test–retest reliability of our resting-state EEG outcomes (Liu et al., 2023), except for lower reliability observed in the gamma band. Relative to the gamma-band power ICC we previously reported (ICC = 0.814), our current estimates of same-day reliability were lower across all three drug conditions (placebo = 0.693, lovastatin = 0.560, minocycline = 0.577). Liu’s estimates included participants from this and other FXS drug studies and used mildly different EEG preprocessing parameters than the current study. Combined with the small amount of average gamma-band power, these changes may amplify small differences in gamma (especially the high-gamma range) and reduce test–retest reliability. Of note, gamma-band power ICCs for both of our active drug conditions were lower than our placebo condition, indicating greater individual-level variability following treatment compared with placebo. This may provide tentative evidence for individual drug responders, but we did not identify cohesive features of this “subgroup” despite testing null sex and age effects.
Limited effects of single-dose minocycline
Previously reported positive effects of minocycline have been more consistent relative to those of lovastatin in human trials of FXS. Paribello and colleagues (2010) conducted the first open-label add-on treatment trial of minocycline in FXS, finding positive effects associated with minocycline on the CGI, Visual Analog Scale, and the irritability subscale of the ABC (Paribello et al., 2010). A separate randomized, double-blind, placebo-controlled crossover study of minocycline found modest improvements in the CGI only (Leigh et al., 2013). The only EEG study of minocycline in humans with FXS found increased habituation to auditory stimuli after minocycline treatment compared with placebo in a subset (n = 12) of the same cohort (Schneider et al., 2013). Notably, our group previously found that resting-state EEG power is similarly affected across minocycline and vehicle treatment in FMR1 KO mice (Lovelace et al., 2020).
Contrasting these chronic dosing studies, we found no clear positive electrophysiological or cognitive/behavioral effects following a single dose of minocycline. This is surprising because the capacity for both chronic (Morin-Parent et al., 2024) and acute (Lang et al., 2013) minocycline dosing to reduce neural hyperexcitability in FXS is supported by preclinical and translational evidence. For example, acute inhibitory effects on human cortical excitability have been demonstrated following a single oral dose of minocycline (Lang et al., 2013). Lang and colleagues found longer cortical silent periods (i.e., silent electromyography [EMG] immediately following transcranial magnetic stimulation [TMS] of motor cortex) following minocycline treatment. The neuroprotective effects of minocycline (He et al., 2021; Henry et al., 2008; Tikka et al., 2001), including anti-inflammatory and antiapoptotic properties that may drive hypothesized electrophysiologic change, have been extensively documented in studies of neurodegenerative conditions (Gordon et al., 2004; Kuang et al., 2009). Antineuroinflammatory properties in particular are relatively fast acting (Elewa et al., 2006), further underscoring the potential utility of acute minocycline dosing in translational studies assessing neurophysiologic target engagement. Thus, our null findings for acute minocycline treatment provide indirect evidence that the primary mechanism driving behavioral and electrophysiological change reported in previous chronic dosing studies may be longer acting (e.g., downstream maturation of dendritic morphology facilitated by reduced MMP-9) and may not be antineuroinflammatory.
Single-dose lovastatin responders
This was the first single-dose study of lovastatin in patients with neurodevelopmental disabilities, including FXS. Open-label trials of lovastatin have demonstrated treatment-associated benefits in humans with FXS (Çaku et al., 2014; Champigny et al., 2021; Morin-Parent et al., 2024), but the only extant blinded trial did not find any evidence for a positive drug-specific effect (Thurman et al., 2020). The Phase II open-label LovaMiX trial first assigned participants to either lovastatin or minocycline before later combining both drugs, finding that lovastatin monotherapy reduced the severity of scores on several subscales of the Social Responsiveness Scale and the number of errors on the KiTAP Distractibility subtest (Champigny et al., 2021). Follow-up ancillary analyses of the LovaMiX TMS and EMG paradigms suggested the combined treatment reduced cortical excitability (Morin-Parent et al., 2024). A more recent blinded placebo-controlled trial combined chronic lovastatin dosing (10–40 mg for 20 weeks) with an open-label telehealth language intervention (Thurman et al., 2020). Language improved in both the lovastatin and placebo groups, presumably due to the concurrent language intervention. To our knowledge, there have been no EEG studies of lovastatin treatment in humans with FXS.
We identified marginal sex- and age-associated interactions: lovastatin was associated with improved spatial awareness in female participants over ∼30 years of age. None of the previous trials of lovastatin in FXS have used visuospatial skills as an outcome measure, but we opted to include it in our design due to the strong test–retest reliability previously demonstrated in FXS (Berry-Kravis et al., 2008). Mechanistically, it is not clear why the behavioral benefits of lovastatin may be selective to visuospatial skills. Female-specific effects raise the question of a moderating effect of FMRP expression (driven by X-inactivation in females) on lovastatin response, but information on FMRP expression was not available for all participants in this sample. It also is not clear why older patients in our sample showed more positive effects on visuospatial skills following lovastatin treatment. Nonsignificant associations between age and visuospatial function in our sample cast doubt that this finding is driven by older individuals having “more room to improve” in response to treatment. Ultimately, we argue that our findings of limited treatment-specific behavioral change following lovastatin treatment, outside of several marginal interactions, are most consistent with Thurman and colleagues’ 2020 trial of chronic lovastatin dosing (the only previous blinded trial of lovastatin in FXS).
Limitations
There are several limitations to our study. First, data loss from eye-tracking tasks was high (about 25%), which may have led to underpowered moderator analyses for our eye-tracking analyses (i.e., N = 5 females). Second, we were not adequately powered to fully assess the moderating impact of other potentially relevant biological factors (e.g., mosaic status) that drive interindividual variability in FXS. An alternative approach for similar trials may be to recruit a more homogeneous group (e.g., fully methylated, full mutation males). Last, the limited racial/ethnic diversity of our sample restricts the generalizability of findings. This reflects broader systemic issues hindering access to genetic testing and other clinical services for FXS families from marginalized racial and ethnic backgrounds (Crawford et al., 2002; Kidd et al., 2017). Limited social–demographic diversity is relevant to clinical trials beyond generalizability because social–environmental factors (tightly linked to demographic factors) in part explain the wide range of clinical outcomes in FXS (McKinney et al., 2024). Along with the field at large, we are engaged in concerted efforts to increase the diversity of our research participants.
Conclusion
We conducted the first acute dosing study of lovastatin and minocycline in patients with FXS. Although we demonstrated strong feasibility and test–retest reliability of a single-dose design in FXS, we found minimal evidence for group-wide treatment effects across both electrophysiologic and behavioral outcomes following a single dose of either drug. However, we found that several modest lovastatin effects varied as a function of biological sex and age, though these small effects require replication.
Clinical Significance
A single dose of lovastatin or minocycline is likely insufficient to drive electrophysiological target engagement in FXS as a whole. Studies aiming to establish target engagement across species, identify subgroups of treatment responders, and clarify optimal dosing for these specific drugs should consider multiple-dose, blinded designs.
Footnotes
Acknowledgments
Much of this work, including the primary grant funding for this study (U54HD082008), was conceptualized and led by the late Dr. John Sweeney, who served as a dedicated mentor to the entire study team and is sorely missed. The authors wish to thank the participating patients and families seen in the Cincinnati Fragile X Research and Treatment Center, without whom this work would not be possible. The authors are grateful for the genetic testing services provided by the Molecular Diagnostic Laboratory at Rush University (Principal Investigator: Dr. Elizabeth Berry-Kravis). The authors also wish to thank Drs. Amy Lang, Mary Keenan-Pfeiffer, and Bridget Murphy, who provided helpful feedback on an earlier draft of this article.
Authors’ Contributions
As noted above, one of our esteemed coauthors, J.S., recently passed away. Prior to his passing, J.S. significantly contributed to the conception of the present study. The authors include him as a coauthor to acknowledge his contributions and honor his commitment to advancing our understanding of fragile X syndrome. The remaining authors assume responsibility for the final content of the article. C.E. (along with J.S. as described above) conceived, designed, and executed the study. L.S. assisted with data collection, data interpretation, and article edits. H.R., D.C., N.F., M.H., and R.S. assisted with data collection and subject retention. L.A.D., W.S.M., and S.D. preprocessed and reviewed resting-state EEG data. L.E., J.N., and L.A.D. preprocessed and reviewed auditory chirp EEG data. P.H. and W.S.M. conducted the statistical analyses. D.R. and M.H. designed and executed the eye-tracking tasks. E.P., K.D., and R.S. assisted with study execution and data interpretation.
Disclosures
The authors have no conflicts of interest to disclose.
Supplementary Material
Supplementary Data S1
Supplementary Figure S1
Supplementary Figure S2
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
