Abstract
Objective:
Cariprazine is a dopamine D3-preferring D3/D2 and serotonin 5-HT1A receptor partial agonist approved to treat adults with schizophrenia and manic/mixed or depressive episodes associated with bipolar I disorder. This sequential-cohort, dose-escalation study was the first to evaluate the pharmacokinetic, safety, and tolerability profile of cariprazine and its two major active metabolites, desmethyl-cariprazine (DCAR) and didesmethyl-cariprazine (DDCAR), in pediatric patients with schizophrenia or bipolar I disorder.
Methods:
This phase I open-label study enrolled patients with schizophrenia (13–17 years of age) or bipolar I disorder (10–17 years of age). Patients met the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria for schizophrenia or bipolar I disorder and had Positive and Negative Syndrome Scale (PANSS) total scores ≥70 or Young Mania Rating Scale (YMRS) total scores ≥20. Patients were assigned to one of four treatment groups to receive 6 weeks of cariprazine treatment through slow titration to 1.5, 3, or 4.5 mg/d or fast titration to 4.5 mg/d. Pharmacokinetics, adverse events (AEs), and various safety parameters were analyzed. Efficacy was evaluated as an exploratory outcome.
Results:
A total of 50 participants were enrolled. Based on mean trough levels, steady state appeared to be reached within 1–2 weeks for cariprazine and DCAR and within 4–5 weeks for DDCAR. Systemic exposure of cariprazine, DCAR, and DDCAR generally increased approximately in proportion to the increases in dose from 1.5 to 4.5 mg/d. The most frequent treatment-related, treatment-emergent AEs included sedation, parkinsonism, tremor, dystonia, and blurred vision. Improvements from baseline on the PANSS and YMRS were observed throughout treatment.
Conclusion:
In this first investigation of cariprazine in a pediatric population with schizophrenia or bipolar disorder, pharmacokinetic parameters were consistent with those observed in adults. Cariprazine appeared to be safe and tolerable in children and adolescents.
Introduction
Schizophrenia and bipolar disorder are chronic and complex psychiatric disorders, which can begin during childhood or adolescence. Early-onset schizophrenia, which refers to schizophrenia with onset during childhood, is rare but has been associated with greater cognitive impairments and impulsivity traits than adult-onset schizophrenia (Kao and Liu 2010). Similarly, younger onset bipolar disorder, which typically develops during adolescence or early adulthood, is associated with poorer functional outcomes and greater illness severity than adult-onset bipolar disorder (Post et al. 2010; Baldessarini et al. 2012). Pharmacotherapy is the mainstay of managing these disorders in children and adolescents (McClellan et al. 2007, 2013), and early treatment is critical to minimize morbidity and preserve cognitive functioning (Karson et al. 2016). However, these disorders are often unrecognized or misdiagnosed in children, leading to a potential delay in appropriate treatment that can negatively affect adult outcomes (Kranzler and Cohen 2013). Furthermore, once a diagnosis is received, children and adolescents may receive inappropriate medications or no medications at all. For example, in a study of 412 children between the ages of 6 and 17 years with bipolar I disorder, approximately one-third received no pharmacotherapy; of those who did receive medication, 71% received nonrecommended treatment, most commonly antidepressant monotherapy (Dusetzina et al. 2011).
Atypical antipsychotics are considered first-line medications for treating schizophrenia and a first-line option for treating bipolar disorder in children and adolescents (McClellan et al. 2007, 2013). However, these medications can cause adverse events (AEs), including extrapyramidal symptoms (EPS), weight gain, sedation, and elevated prolactin levels. Children are often more sensitive to these side effects, particularly sedation, compared with adults (Kranzler and Cohen 2013; Liu et al. 2019). Therefore, there is a need for efficacious treatment options with manageable side effects to treat schizophrenia and bipolar disorder in the pediatric population.
Cariprazine is an oral, dopamine D3-preferring D3/D2 and serotonin 5-HT1A receptor partial agonist approved for the treatment of adults with schizophrenia (1.5–6 mg/d) and treatment of manic/mixed (3–6 mg/d) or depressive (1.5–3 mg/d) episodes associated with bipolar I disorder. In randomized, double-blind, placebo-controlled trials, cariprazine demonstrated efficacy versus placebo in adults with schizophrenia (Durgam et al. 2014, 2015a; Kane et al. 2015) and in adults with manic/mixed episodes (Calabrese et al. 2015; Durgam et al. 2015b; Sachs et al. 2015) or depressive episodes (Durgam et al. 2016; Earley et al. 2019, 2020b) associated with bipolar I disorder. Cariprazine displays a unique pharmacokinetic profile with two active metabolites: desmethyl-cariprazine (DCAR) and didesmethyl-cariprazine (DDCAR). DDCAR is the most prominent active moiety, representing 64% of total cariprazine exposure at steady state (Periclou et al. 2021). In adults, the effective half-life (T1/2) of cariprazine is about 1 week for all active moieties (Nakamura et al. 2016), and population pharmacokinetic data show that cariprazine and DCAR concentrations decrease by >90% within 1 week following the last dose and that DDCAR concentrations decrease by about 90% within 4 weeks (Periclou et al. 2021).
This is the first study of the pharmacokinetic parameters of cariprazine in pediatric patients. The goals of the study were to assess the pharmacokinetic profile of cariprazine, DCAR, and DDCAR using slow and fast titration schedules and to evaluate the safety and tolerability of cariprazine in a pediatric population with schizophrenia or bipolar I disorder. In addition, the efficacy of cariprazine was examined as an exploratory outcome.
Materials and Methods
Study participants
This study was conducted at three investigative sites in the United States from February 15, 2017, to October 9, 2019 in conformance with the International Council for Harmonisation E6 guideline for Good Clinical Practice and the principles of the Declaration of Helsinki. Investigators from each study site obtained approval of the study protocol from a properly constituted institutional review board prior to study initiation. At the first visit, the study was discussed with each participant and with the participant's parent or legally authorized representative, and written informed consent was obtained. Participants were eligible if they were between the ages of 13–17 years with schizophrenia or 10–17 years with bipolar I disorder, as defined in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria (American Psychiatric Publishing 2013) and confirmed by the Kiddie Schedule for Affective Disorders and Schizophrenia-Present and Lifetime (K-SADS-PL).
For patients with schizophrenia, the Positive and Negative Syndrome Scale (PANSS) (Kay et al. 1987) total scores were ≥70; for patients with bipolar I disorder, the Young Mania Rating Scale (YMRS) (Young et al. 1978) total scores were ≥20. Participants who displayed suicidal or homicidal attempt or intent in the 6 months before screening were excluded.
Study design
In this multicenter, open-label, sequential-cohort, dose-escalation study, the pharmacokinetic profile of cariprazine was assessed in participants with schizophrenia or bipolar I disorder. The multiple-dose study enrolled participants with schizophrenia or bipolar I disorder between the ages of 13–17 years in Part A, and those with bipolar I disorder between the ages of 10–12 years were enrolled in Part B. Consistent with most pharmacokinetic studies, participants and caregivers were compensated for their time and effort.
Patients were screened for 28 days before dosing, and previous antipsychotics or mood stabilizers were discontinued during a washout period of 4–7 days before dosing. The study included a 42-day treatment period and a 42-day post-treatment pharmacokinetic follow-up. Participants were required to be inpatient during the initiation of study treatment (days 1–7). After 7 days, the participant could remain an inpatient or continue treatment as an outpatient, based on clinical judgment. Participants were required to be inpatient from day 41 until collection of the 24-hour blood sample on day 43. After the last dose of cariprazine on day 42, patients could resume their previous medications for schizophrenia or bipolar I disorder.
Participants were assigned to one of eight cohorts and received cariprazine up to 1.5, 3, or 4.5 mg/d (predicted for pediatric participants via simulations based on adult population models) through slow titration in Treatments A, B, or C or fast titration in Treatment D (Fig. 1).
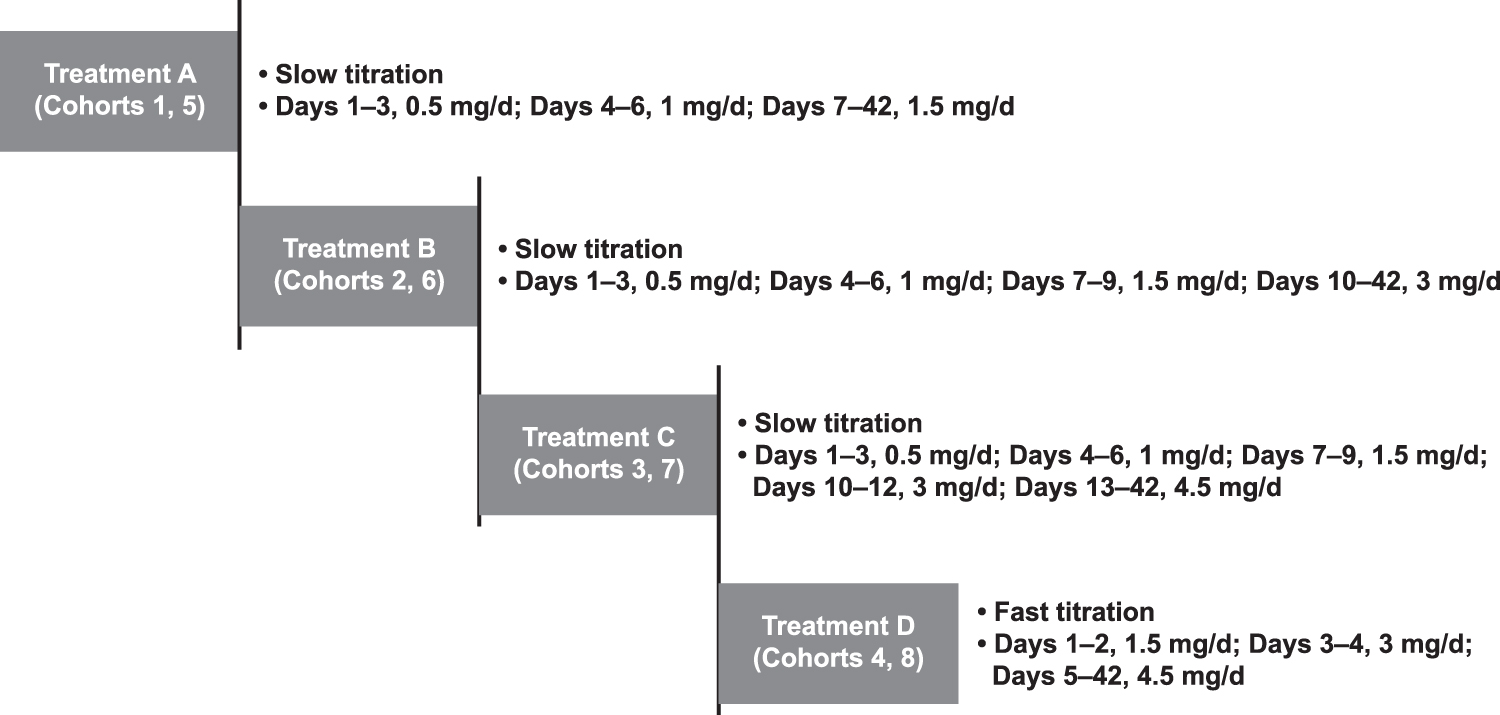
Patient treatment schedule.
Sample collection
Blood samples were obtained and processed for measurement of cariprazine, DCAR, and DDCAR on day 1 at 0 hour (predose), and at 2, 3, 4, 6, 8, 24 hours postdose; on days 7, 14, 21, 28, 35, 39, 40, and 41 predose; and on day 42 predose and at 2, 3, 4, 6, 8, 24, 48, 168, 336, 672, and 1008 hours postdose. Plasma concentrations of cariprazine, DCAR, and DDCAR were analyzed using a validated liquid chromatography with tandem mass spectrometry method. The lower limits of quantification were 0.02 ng/mL for cariprazine and DCAR, and 0.05 ng/mL for DDCAR.
Assessments
The pharmacokinetic parameters of cariprazine and its metabolites measured after the first and last doses included the following: maximum plasma drug concentration (Cmax), time corresponding to maximal plasma concentration (Tmax), minimum plasma drug concentration during a dosing interval at steady state (Cmin,ss), area under the plasma concentration versus time curve from time 0 to the end of the dosing interval, τ, (AUC0−τ), and terminal elimination half-life (T1/2). In addition, the apparent clearance (CL/F) and apparent volume of distribution during the terminal phase (Vz/F) were determined for cariprazine.
At each visit, AEs, ocular examinations, clinical laboratory evaluations, vital signs, and 12-lead ECG parameters were assessed. In addition, the Columbia-Suicide Severity Rating Scale (C-SSRS) (Posner et al. 2011) and the Children's Depression Rating Scale-Revised (CDRS-R) (Poznanski and Mokros 1996) were administered to assess suicidality and depression. To assess EPS, the Abnormal Involuntary Movement Scale (AIMS) (Guy 1976), the Barnes Akathisia Rating Scale (BARS) (Barnes 1989), and the Simpson-Angus Scale (SAS) (Hawley et al. 2003) were assessed at weekly intervals until day 56 and then every 2 weeks until day 84.
To evaluate the efficacy of cariprazine as an exploratory outcome, change from baseline to the end of treatment on the PANSS and YMRS total scores was assessed in participants with schizophrenia or bipolar I disorder, respectively. The Clinical Global Impressions-Severity (CGI-S) (Guy 1976) was also assessed at each follow-up visit to assess global symptoms throughout the study.
Data analysis
The principal parameters describing the pharmacokinetics of cariprazine, DCAR, and DDCAR were derived from plasma concentrations through noncompartmental analysis. Safety parameters were summarized by treatment group, and all data were analyzed descriptively.
Results
Patient characteristics
A total of 50 pediatric patients with schizophrenia or bipolar I disorder were enrolled and received at least one dose of cariprazine; overall, 48 patients (96%) completed both the 42-day treatment and follow-up periods. Only five participants had schizophrenia, while all other participants had bipolar I disorder. Patient demographics and baseline characteristics were relatively similar between treatment groups (Table 1).
Demographics and Baseline Characteristics of the Safety Population
The combined safety population includes all patients who took at least one dose of cariprazine; treatment groups were as follows: Group A, slow titration to 1.5 mg/day; Group B, slow titration to 3 mg/day; Group C, slow titration to 4.5 mg/day; Group D, fast titration to 4.5 mg/day.
n = 2; b n = 3; c n = 11; d n = 12; e n = 9.
BMI, body mass index; PANSS, Positive and Negative Syndrome Scale; SD, standard deviation; YMRS, Young Mania Rating Scale.
Pharmacokinetics
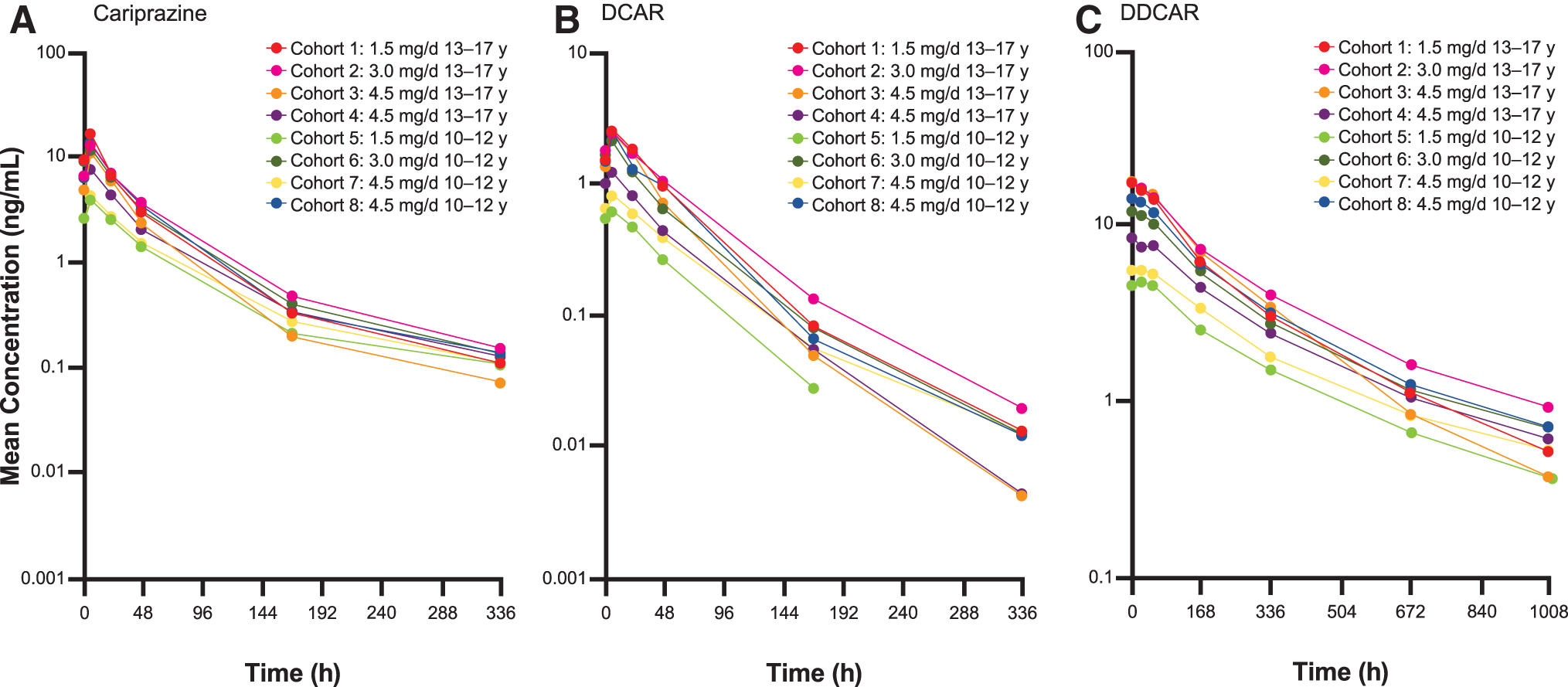
Following first dose, mean plasma concentrations of cariprazine increased more than dose proportionally from 0.5 to 1.5 mg/d and were higher for participants 10–12 years of age than for participants 13–17 years of age. Measurements of DCAR plasma concentration were generally low, and DDCAR levels were undetectable in most participants after the initial dose; therefore, pharmacokinetic parameters could not be determined on day 1 for these analytes. Based on mean trough levels between days 7 and 42, steady state appeared to be reached within 1–2 weeks for cariprazine and DCAR and within 4–5 weeks for DDCAR. Mean steady-state area under the curve (AUC) values in Cohorts 1–8 increased in proportion to dose increases from 1.5 to 4.5 mg/d for all three analytes. Dose-normalized steady-state Cmax and AUC values for all three analytes were similar between the age groups.
Following multiple-dose administration of 1.5–4.5 mg/d, the rank of exposure based on AUC was DDCAR > cariprazine > DCAR. The CL/F for cariprazine ranged from 14.46 to 21.18 L/h and was similar across cohorts. The pharmacokinetic parameters of cariprazine, DCAR, and DDCAR after the last dose are shown in Tables 2 –4, respectively.
Pharmacokinetic Parameters of Cariprazine After Last Dose
n = 5; b n = 4.
AUC0−τ, area under the plasma concentration versus time curve from time 0 to the end of the dosing interval; Cavg, average plasma drug concentration; CL/F, apparent clearance; Cmax, maximum plasma drug concentration; Cmin, minimum plasma drug concentration during a dosing interval; SD, standard deviation; T1/2, half-life; Tmax, time corresponding to maximal plasma concentration.
Pharmacokinetic Parameters of Desmethyl-Cariprazine After Last Dose
n = 5.
AUC0−τ, area under the plasma concentration versus time curve from time 0 to the end of the dosing interval; Cavg, average plasma drug concentration; Cmax, maximum plasma drug concentration; Cmin, minimum plasma drug concentration during a dosing interval; SD, standard deviation; T1/2, half-life; Tmax, time corresponding to maximal plasma concentration.
Pharmacokinetic Parameters of Didesmethyl-Cariprazine After Last Dose
n = 5.
AUC0−τ, area under the plasma concentration versus time curve from time 0 to the end of the dosing interval; Cavg, average plasma drug concentration; Cmax, maximum plasma drug concentration; Cmin, minimum plasma drug concentration during a dosing interval; SD, standard deviation; T1/2, half-life; Tmax, time corresponding to maximal plasma concentration.
Following the last dose (discontinuation of treatment), mean plasma concentrations of cariprazine and DCAR declined by ∼40%–50% within 1 day and >90% within 1 week. Mean plasma concentrations of DDCAR declined by ∼50% within 1 week and 90% within 4 weeks after the last dose (Fig. 2).
Safety and tolerability
There were no deaths, serious AEs (SAEs), or discontinuations due to AEs experienced by participants throughout treatment or follow-up. Based on C-SSRS assessments, no suicidal ideation or behavior was reported during the 42-day dosing period or during the 42-day follow-up period. Furthermore, there appeared to be no worsening of depression symptoms based on CDRS-R total scores, with a mean (standard deviation; SD) change from baseline to end of the treatment period of −3.7 (2.6). The most common treatment-emergent AEs (TEAEs) in ≥2 participants overall were sedation (18%), blurred vision (10%), parkinsonism (8%), dystonia (8%), and tremor (6%) (Table 5). Across all 4 treatment groups in Part A and Part B combined, the majority of TEAEs were mild (85%), and none were considered severe throughout the treatment period. Four patients who were receiving the fast titration (Treatment D) exhibited parkinsonism; all cases were mild in severity and resolved following treatment with benztropine.
Safety Profile
Italicized TEAEs indicate treatment related to either Part A or Part B.
From baseline to end-of-treatment period.
SD, standard deviation; TEAE, treatment-emergent adverse event.
In Part A, there were no reported cases of treatment-emergent akathisia or parkinsonism based on the BARS and SAS criteria, respectively. In Part B, a single instance of treatment-emergent parkinsonism based on SAS criteria was reported on day 7 for one participant receiving Treatment D, and a single instance of akathisia based on BARS criteria (moderate; related to study treatment) was reported at an unscheduled assessment on day 15 for one participant receiving Treatment D. The AIMS total score was zero for all participants in Parts A and B at baseline and at all subsequent study visits.
There were no clinically meaningful imbalances in the nature, number, or severity of the reported TEAEs between age groups, except that the reported incidence of increased weight was higher for participants aged 10–12 years in Part B (38.5%) versus participants aged 13–17 years in Part A (12.5%). In Part A, mean (SD) weight changes from baseline through the end of the 42-day cariprazine treatment period ranged from +0.8 (2.0) to +2.9 (1.9) kg across treatment groups. In Part B, mean (SD) weight changes ranged from +1.1 (1.2) to +3.9 (1.8) kg across treatment groups. A total of 11 patients had a weight increase of ≥7% from baseline to the end of the treatment period.
At the end of the study, z-scores reflecting change in baseline weight for age and gender were within 2 SDs, suggesting that changes over the course of the study were within age- and gender-appropriate values for all patients. Safety observations were generally similar between titration schedules, except for transient potentially clinically significant (PCS) decreases in diastolic blood pressure (i.e., observed value ≤50 mm Hg and decrease ≥15 mm Hg from baseline) observed in two participants in Part A receiving the faster titration (Treatment D) on the first day of dosing (−17 and −28 mm Hg, respectively).
These decreases were accompanied by systolic blood pressure decreases of 21 and 6 mm Hg, respectively, and pulse increases of 18 and 12 beats per minute (bpm), respectively. Neither systolic blood pressure nor pulse changes met the PCS criteria (i.e., observed value ≤80 mm Hg and decrease ≥20 mm Hg from baseline and observed value ≥130 bpm and increase ≥15 bpm from baseline) in these two patients on day 1. The PCS diastolic blood pressure decreases resolved within 24 hours in both patients. In Part B, five patients experienced transient PCS increases in systolic blood pressure during the treatment period, ranging from 22 to 30 mm Hg with no observed temporal pattern. No presyncopal or syncopal events were reported throughout the study. Compared with patients receiving the slower titration schedules, patients receiving the fast titration schedule reported higher rates of parkinsonism and agitation.
Mean changes in laboratory tests were unremarkable. Metabolic (glucose, total cholesterol, and triglycerides) and white blood cell function parameters (white blood cell and neutrophil counts) were generally within normal limits throughout the study. Prolactin levels were within normal range at baseline for both study parts (Part A: 10 ng/mL at baseline; Part B: 6.9 ng/mL at baseline) and generally remained stable and within the normal range throughout treatment and follow-up. Mean (SD) changes in prolactin levels from baseline to end-of-treatment period were +2.7 (9.7) ng/mL (range: −23.0 to +19.6 ng/mL) in Part A and +0.3 (7.0) ng/mL (range: −15.9 to +11.6 ng/mL) in Part B. No participant had a postbaseline ECG change in QTc >60 milliseconds or a Fredericia QTc interval of >500 milliseconds during pharmacokinetic profiling.
Exploratory efficacy
Following treatment with cariprazine, the mean change in PANSS total score from baseline to end of cariprazine treatment on day 42 was −11.6 for patients with schizophrenia (Fig. 3A); and the mean change in YMRS total score from baseline to end of treatment on day 42 was −8.9 for patients with bipolar I disorder (Fig. 3B). Similar mean changes were observed to the end of the follow-up period (mean change from baseline to end of follow-up period on day 84 was −11.8 for PANSS total score and −9.8 for YMRS total scores).
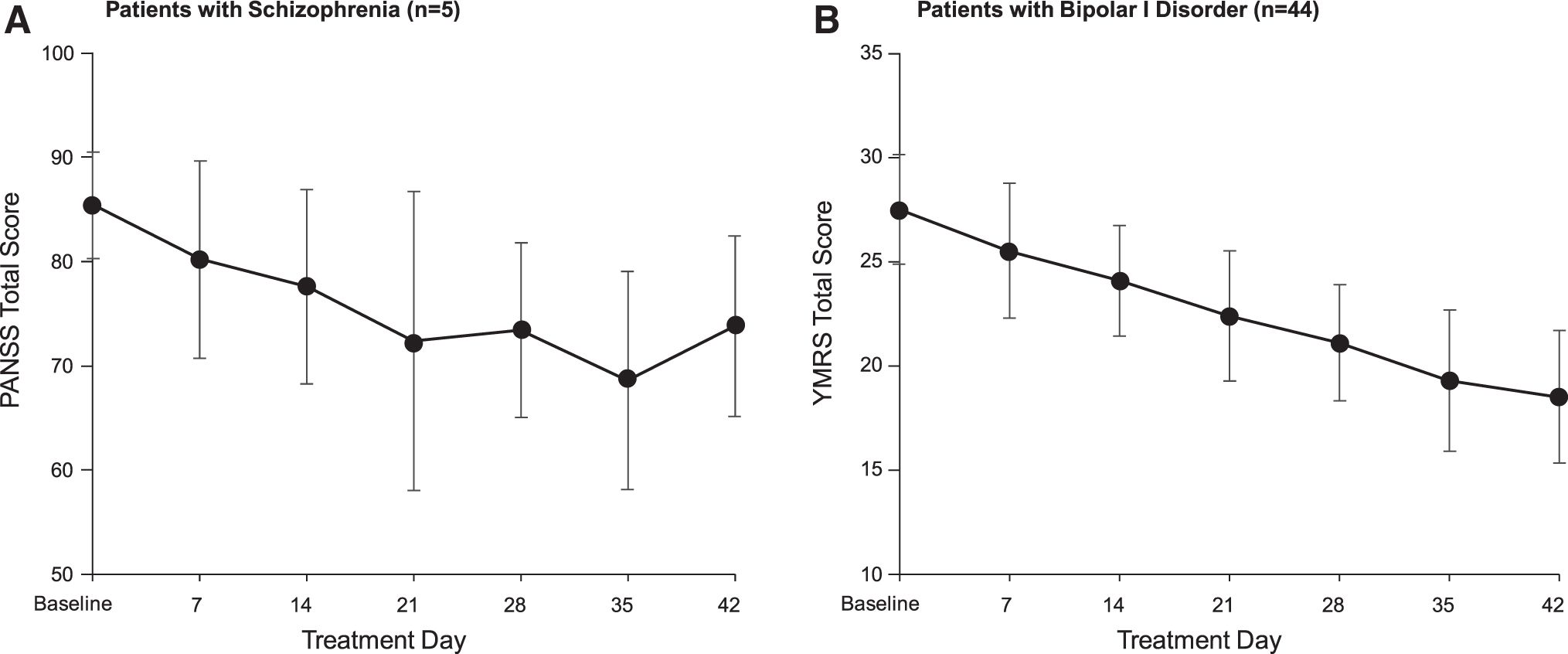
Change from baseline to end of treatment (day 42) on
Of note, following the last dose of cariprazine on day 42, patients were allowed to resume their previous medications for schizophrenia or bipolar I disorder. Improvements were also seen on the CGI-S following treatment (mean change from baseline to end of treatment on day 42 was −0.6) and were maintained to the end of the follow-up period (mean change from baseline to end of follow-up period on day 84 was −0.9).
Discussion
In this first investigation of cariprazine in children and adolescents (10–17 years), cariprazine displayed a pharmacokinetic profile consistent with that of the adult population. Following multiple-dose administration, the rank of exposure based on AUC was DDCAR > cariprazine > DCAR, and the plasma concentration time-profiles in this pediatric population were similar to previous pharmacokinetic results in adults (Periclou et al. 2021). Systemic exposure of cariprazine and its active metabolites generally increased approximately in proportion to the increase in dose from 1.5 to 4.5 mg/d for all three analytes. The time to reach steady state appeared to be about 1–2 weeks for cariprazine and DCAR and about 4–5 weeks for DDCAR. The clearance of cariprazine was similar across doses and age groups. During the 42-day dosing period, gradual titration of cariprazine to doses of 1.5, 3, and 4.5 mg/d and fast titration to 4.5 mg/d were safely tolerated in pediatric patients with schizophrenia or bipolar I disorder.
Cariprazine appeared to be safe and well-tolerated overall in a pediatric population. The safety observations were generally consistent with the known safety profile of cariprazine in adults. There were no deaths, SAEs, or discontinuations due to AEs and no suicidal ideation or behavior reported throughout the study. Across all cohorts, TEAEs that were determined to be related to study treatment in ≥4 participants overall were sedation (18%; 9/50) and parkinsonism (8%; 4/50). Akathisia and EPS are common concerns with dopamine antagonists and partial agonists, and cariprazine has been associated with higher rates of these AEs versus placebo in adult studies (Earley et al. 2017a, 2020a; Barabassy et al. 2021). In this study of children and adolescents, akathisia was uncommon and was only reported in one patient in Part A and 1 in Part B.
There were four reported incidences of parkinsonism, all of which were mild in severity and resolved following treatment with benztropine. Other AEs that are common with dopamine D2 antagonists include metabolic changes and elevations in prolactin levels (Henderson and Doraiswamy 2008). These can be particularly concerning in children and adolescents due to the risk of obesity and diabetes from weight gain (Pramyothin and Khaodhiar 2015) and delayed puberty due to altered estrogen and testosterone levels from hyperprolactinemia (Matalliotakis et al. 2019). Across all treatment groups, three patients in Part A (12.5%) and eight patients in Part B (30.8%) experienced weight gain ≥7%. Although age- and gender-adjusted weight (z-scores) reflected appropriate growth in this otherwise healthy population of pediatric participants, weight gain in the pediatric population, particularly in patients 10–12 years old, warrants further investigation.
There appeared to be no significant changes in prolactin levels among age and treatment groups, which is consistent with adult findings (Barabassy et al. 2021). Since D2 receptor antagonism is associated with hyperprolactinemia (Haddad and Wieck 2004), the mechanism of action of cariprazine as a D3-preferring D3/D2 receptor partial agonist may contribute to its favorable effects on metabolic changes and prolactin levels. In this study, cariprazine appeared to be well-tolerated overall and displayed a safety profile consistent with known adult safety data.
In general, there were no clinically meaningful imbalances in the nature, number, or severity of reported TEAEs between the two age groups and titration schedules. One exception was that there was a greater incidence of weight gain ≥7% from baseline in participants aged 10–12 years in Part B versus participants aged 13–17 years in Part A. This could be due to faster growth rates in younger children (i.e., 10–12 years) compared with older children (i.e., 13–17 years), but is nonetheless a noteworthy finding. In adults, cariprazine is associated with mean changes in body weight of +0.8 kg at 6 weeks for 1.5–3 mg/d and +1.0 kg for 4.5–6 mg/d in patients with schizophrenia and +0.5 kg for cariprazine 3–6 mg/d in adults with bipolar mania (Vraylar 2019). It is possible that the favorable weight profile of cariprazine in adults does not translate to the pediatric population, particularly younger children aged 10–12 years, although it is difficult to draw definitive conclusions given the small sample size of this study and lack of placebo comparator. Future studies should monitor weight gain associated with cariprazine use in the pediatric population.
When comparing the titration rates, transient but PCS decreases in diastolic blood pressure were observed during the fast titration following the first dose. However, no AEs were reported on the day of the decreases, which resolved within 24 hours and no presyncopal or syncopal events were reported during the study. Patients receiving the faster titration (Treatment D) also reported higher rates of parkinsonism and agitation than those receiving the slower titration (Treatments A, B, and C). Previous adult data suggest that lower doses and slower titrations of cariprazine may be better tolerated than fast titrations. In an analysis of phase 2 and phase 3 bipolar mania studies (Earley et al. 2017a) that used a rapid titration schedule, rates of akathisia, restlessness, and EPS were higher than in bipolar depression studies (Earley et al. 2019, 2020b) that used a slower titration schedule. Similar trends have been noted in schizophrenia trials (Earley et al. 2017b), suggesting that lower doses and slower titrations reduce the risk of these AEs. Results from a negative symptom study of patients with schizophrenia (Nemeth et al. 2017) showed lower rates of akathisia (8%) with slower titration of cariprazine compared with results from a safety analysis (Barabassy et al. 2021) that used a faster titration schedule with rates of akathisia as high as 18% in the 6 mg/d group. The findings of this study suggest that certain AEs may be more common with the rapid titration schedule and should warrant caution in the pediatric population. A slower cariprazine titration may offer greater tolerability than a rapid titration, although comparisons between the titration rates should be made with caution due to the small size of the current study.
Exploratory efficacy results showed improvements in psychiatric symptoms from baseline to the end of cariprazine treatment based on the PANSS, YMRS, and CGI-S total scores. In addition, observed symptom improvements were maintained throughout the follow-up period despite cariprazine treatment discontinuation. During the follow-up period, some participants restarted their previous medications; however, sustained improvement with cariprazine may also be partially attributed to its unique pharmacology as shown in an analysis by Correll et al. (2019) that reported delayed incidence of schizophrenia relapse in adults who discontinued cariprazine compared with adults who discontinued other schizophrenia medications in similarly designed relapse prevention studies. The authors hypothesized that these results were due to the relatively long half-life of DDCAR; furthermore, plasma concentrations indicated that DDCAR may continue to occupy D2 and D3 receptors for 4 weeks following the last dose. Since the half-life of DDCAR in pediatric patients appears to be similar to that in adults, it is possible that sustained improvement in psychiatric symptoms may also occur in the pediatric population. Further studies examining the long-term efficacy of cariprazine in children and adolescents are needed to corroborate these findings.
The United States Food and Drug Administration (FDA) has recently concluded that pharmacokinetic data and analyses can be used as a basis for extrapolating efficacy from adult to pediatric patients by selecting doses in pediatric patients that achieve similar drug exposure as in adults (Kalaria et al. 2020). For example, this is applicable to atypical antipsychotics that share a similar mechanism of action (e.g., D2-receptor antagonism or partial agonism, 5-HT1A partial agonism, and/or 5-HT2A antagonism) and are approved for the treatment of schizophrenia and/or bipolar disorder in adults. Therefore, the results from this pediatric pharmacokinetic study are important, and these preliminary, exploratory efficacy data might be helpful. Nonetheless, additional long-term study is also needed to assess the safety and tolerability of long-term treatment of pediatric patients with cariprazine.
The results of this study should be interpreted within its limitations. Since only patients 10–17 years old with bipolar I disorder and 13–17 years old with schizophrenia were enrolled, this limits the generalizability to other indications and age ranges. In addition, although the study was conducted at multiple sites, many patients were enrolled from a single clinical site and were predominantly African American, which may further limit the generalizability to other patient populations. Also, the open-label nature of the study limits definitive efficacy conclusions due to the lack of a comparator drug. The small number of participants with schizophrenia precluded meaningful comparisons of PANSS total scores by cariprazine dosing schedules.
Larger, randomized, double-blind trials are warranted to establish optimal dosing regimens and confirm these safety findings in the pediatric population. Currently, an open-label study (NCT04578756) is ongoing to assess the long-term safety and tolerability of cariprazine in participants between the ages of 10 and 17 years with schizophrenia or bipolar I disorder.
Conclusions
In conclusion, the pharmacokinetic profile of cariprazine in pediatric patients was consistent with that observed in adults. Cariprazine appeared to be safe and well-tolerated in a pediatric population aged 10–17 years. The psychiatric symptom improvement provides support for future investigation of cariprazine use in children and adolescents with schizophrenia or bipolar I disorder.
Clinical Significance
This study aimed to evaluate the pharmacokinetics, safety, and tolerability of cariprazine in pediatric patients with schizophrenia (aged 13–17 years) or bipolar I disorder (aged 10–17 years) and to examine efficacy in the pediatric population as an exploratory outcome. Cariprazine displayed a similar pharmacokinetic profile to that observed in adults and appeared to be safe and well-tolerated among pediatric participants. Symptom improvement from baseline was observed throughout the treatment period. These findings support the need for further investigation into cariprazine use for children and adolescents with schizophrenia or bipolar I disorder.
Footnotes
Acknowledgments
The authors would like to the thank Antonia Periclou, PhD, for her contributions to this analysis. Writing and editorial assistance were provided by Caroline Warren, PharmD, of Prescott Medical Communications Group (Chicago, IL), which was funded by AbbVie. All authors met the ICMJE authorship criteria. Neither honoraria nor payments were made for authorship.
Authors' Contributions
All authors contributed to the interpretation of data and provided critical reviews of all the article drafts. All authors approved the article for submission to this journal.
Disclosures
T.R. is an employee and shareholder of AbbVie. R.R. was an investigator in this study and received financial support from AbbVie, which funded the study. P.P.Y. is an employee and shareholder of AbbVie. W.R.E. was an employee of AbbVie at the time of the study and is a shareholder of AbbVie, AstraZeneca, and Eli Lilly. A.L.H. is an employee and shareholder of AbbVie.
