Abstract
Objectives:
We analyzed patient-reported sleep parameters for an extended-release methylphenidate formulation (PRC-063) in adolescents with attention-deficit/hyperactivity disorder.
Methods:
Clinical efficacy and long-term safety/tolerability data from a 4-week, double-blind, placebo-controlled, fixed-dose study (NCT02139111) and a subsequent 6-month, optimized-dose, open-label extension (OLE) study (NCT02168127) were used. In the double-blind study, participants were randomly assigned 1:1:1:1:1 to one of four doses of PRC-063 (25, 45, 70, or 85 mg/day) or placebo. In both the double-blind and OLE studies, sleep outcomes were assessed using the Pittsburgh Sleep Quality Index (PSQI).
Results:
During double-blind treatment, no statistically significant least-squares mean difference in change from baseline between PRC-063 (all doses combined; N = 293) and placebo (N = 74) was found for either global PSQI score (−0.3 vs. −0.5; p = 0.6110) or scores for any of the seven PSQI subscales. Compared with the placebo group, a marginally higher proportion of patients in the PRC-063 group (all doses combined) went from being poor to good sleepers (global PSQI score ≤5; 14.4% vs. 11.3%). In a logistic regression analysis, study treatment was not a predictor of poor sleep (p = 0.5368) at the end of the double-blind study. In the OLE study, there was a trend of improvement in sleep after 1 month of individualized dosing that was maintained through 6 months. Sleep efficiency (time asleep as a proportion of time in bed) showed improvement at the end of the OLE study.
Conclusion:
While individual patients may experience changes in sleep as an adverse event, group data evaluating sleep as an outcome found there were no differences between PRC-063 and placebo in self-reported sleep outcomes on the PSQI.
Introduction
It is well established that sleep problems are prevalent in the adolescent population (Wong et al. 2016; Quante et al. 2019). Deficient sleep, defined as both reduced sleep quantity and quality and timing of sleep that is not aligned with natural circadian rhythms, occurs in adolescents in what has been described as “epidemic” proportions (Owens and Weiss 2017; Becker et al. 2019). The clinical entity of insomnia, defined in DSM-5 as persistent difficulty initiating or maintaining sleep or early morning awakening resulting in some type of daytime impairment (American Psychiatric Association 2013), is prevalent in the adolescent population, especially in association with psychiatric/neurodevelopmental comorbidities. In particular, attention-deficit/hyperactivity disorder (ADHD) is associated with an increase in sleep problems, which can negatively affect daytime functioning (Wajszilber et al. 2018). In addition, stimulants used to treat ADHD have been associated with changes in sleep as an adverse event (AE) in clinical trials, even if group data do not necessarily demonstrate differences in sleep outcomes between drug and placebo (Owens et al. 2016). Thus, the combination of adolescence, ADHD, and potential insomnia secondary to stimulant use puts adolescents with ADHD at very high risk of functionally impairing sleep problems (Craig et al. 2020).
A number of studies have explored the associations between ADHD, poor sleep, and impaired daytime functioning. For example, a recent survey of 7072 Chinese adolescents with ADHD demonstrated significant comorbid insomnia, poor sleep quality, and short sleep duration; furthermore, sleep difficulties were predictive of ADHD symptoms a year later (Liu et al. 2020). A recent cross-sectional study found that almost half of adolescents with ADHD report daytime sleepiness, which was predicted in part by time in bed and bedtime resistance, as well as ADHD-related factors (Langberg et al. 2020). An earlier systematic review of 19 studies using either subjective (self-report) or objective (actigraphy/polysomnography) sleep measures found associations between sleep disturbances and ADHD symptoms in adolescents with ADHD (Lunsford-Avery et al. 2016). Finally, in a study comparing adolescents with and without ADHD, the odds of parent-rated sleep disturbance were six times higher for adolescents with ADHD (Becker et al. 2019).
Two sleep factors in particular have been found to be associated with an increased risk of sleep problems and resultant daytime functional impairment in adolescents with ADHD. First, the tendency toward evening circadian preference predicts both self- and parent-reported sleep problems and daytime sleepiness (Becker et al. 2020). Second, adolescents with ADHD have greater intraindividual sleep variability (night-to-night differences) as compared with their peers, including variability in time in bed, bedtime, morning waking time, sleep duration, sleep-onset latency, sleep quality, and nighttime waking (Langberg et al. 2019).
Stimulants such as methylphenidate and amphetamines are a cornerstone of ADHD pharmacotherapy (Cortese 2020). While long-acting extended-release formulations are intended to improve functioning later in the day, the question remains as to whether this comes at the expense of increased difficulty with sleep or, conversely, is associated with better sleep due to more effective ADHD treatment. Many of the high-risk activities associated with ADHD in adolescents occur late in the day, possibly because of the propensity for the circadian rhythm to be delayed in adolescents with ADHD. A long-acting stimulant might mitigate this risk. On the other hand, recent research has demonstrated that short sleep duration itself is associated with high-risk activities (Owens et al. 2019; Ordway et al. 2020). Studies have found that stimulant treatment in children and adolescents generally does not worsen sleep outcomes, and actually improves sleep in some patients (Gau and Chiang 2009; Becker et al. 2016). However, these studies have not specifically examined the sleep impact of the longest acting stimulants, which relieve symptoms for >12 hours.
Given the possibility that the benefits of long-acting stimulants may occur at the expense of sleep, placebo-controlled studies have specifically examined sleep as both an AE and an outcome. Understanding the effects of stimulants on sleep may now be of increased importance as we have seen the development of both a mixed amphetamine salt (Wigal et al. 2019) and a methylphenidate product (Wigal et al. 2020) in which laboratory classroom studies in adolescents or adult workplace environment studies have demonstrated drug action lasting 16 hours.
PRC-063 (marketed as Adhansia XR® in the United States and as Foquest® in Canada) is a once-daily extended-release methylphenidate hydrochloride formulation which is approved for the treatment of ADHD in patients aged ≥6 years. PRC-063 capsules, which should be taken in the morning or upon awakening, contain identical beads consisting of an immediate-release component (∼20% of the methylphenidate dose) and a controlled-release component (∼80% of the methylphenidate dose) (Adhansia XR 2019). In pharmacokinetic analyses in adolescents aged 13–17 years, an initial peak plasma concentration of methylphenidate occurred at a median of 2 hours post-PRC-063 administration, owing to the rapid release and absorption of the immediate-release component. A second, higher peak plasma concentration at a median of 11 hours resulted from release of the controlled-release component (Adhansia XR 2019). In laboratory classroom and adult workplace environment studies, PRC-063 has demonstrated an onset of clinical effect at 1 hour and a duration of action lasting up to 13 hours in children and 16 hours in adults (Adhansia XR 2019; Childress et al. 2020a, 2020b; Wigal et al. 2020).
The efficacy and long-term safety of PRC-063 in adolescents aged 12–17 years were evaluated in a 4-week, parallel-arm, double-blind, fixed-dose, forced-titration study and a subsequent 6-month open-label extension (OLE) study. The double-blind study found significant improvements versus placebo for ADHD 5 Rating Scale (ADHD-5-RS) scores, as rated by clinicians and parents (Weiss et al. 2021). ADHD-5-RS scores continued to improve during the OLE study when patients were titrated to an optimal PRC-063 dose. The present analyses elaborate on the published finding that there were no statistically significant differences between PRC-063 and placebo in overall sleep quality (Weiss et al. 2021), as measured using the Pittsburgh Sleep Quality Index (PSQI) (Buysse et al. 1989), by looking at outcomes for PSQI subscales and changes in sleep outcomes from baseline to the end of the OLE study.
Methods
Study design
Analyses were conducted based on a 4-week, randomized, double-blind, parallel-arm, fixed-dose study (NCT02139111) and a 6-month OLE study (NCT02168127) of the clinical efficacy and safety of PRC-063 in adolescents with ADHD. Complete details of the methods are provided elsewhere (Weiss et al. 2021). In brief, the studies were conducted in accordance with the Declaration of Helsinki, International Council for Harmonisation (ICH) Good Clinical Practice (GCP), and all applicable national and local laws at 42 sites in Canada and the United States in 2014-2015. Approval from an independent ethics committee or institutional review board, as appropriate, was obtained for each study site. Each potential participant provided written informed assent and their parent/guardian written informed consent before they were screened for entry into the study.
Participants
Eligible participants were males and females (nonpregnant, non-nursing) aged 12-17 years and diagnosed with ADHD (hyperactive/impulsive, inattentive, or combined type; according to DSM-5) who had a total score of ≥24 on the ADHD-5-RS (a clinician-administered scale evaluating 18 symptoms of ADHD on a 4-point scale [0–3], with lower scores indicating less severe ADHD symptoms). Patients who had an allergy to methylphenidate or amphetamines, had a history of serious adverse reactions to methylphenidate, or were known to be nonresponsive to methylphenidate treatment were excluded. Patients were also excluded if they had a psychiatric comorbidity (including functionally impairing sleep problems) that required use of other psychotropic drugs (including prescription sleep medications). Patients were allowed to continue taking melatonin if they had been on a stable dose for ∼4 weeks.
Treatment
In the double-blind study, following a 1-week washout period after screening, participants were randomly assigned at a ratio of 1:1:1:1:1 to receive one of four fixed doses of PRC-063 (25, 45, 70, or 85 mg/day) or matched placebo. Based on a predetermined dose titration schedule, the dose of PRC-063 was increased over 2 weeks until the final dose was achieved. Each participant's daily dose was administered in the morning.
Participants who completed the double-blind study and who continued to fulfill all the eligibility criteria could enter the OLE study. Enrollment of eligible participants continued until half of the sample in the double-blind study had been enrolled in the OLE study. Each participant was treated with PRC-063 at a starting dose chosen by the investigator. Dose optimization was achieved through titration at unscheduled dose-adjustment visits per the investigator's judgment. The maximum dose was 85 mg/day (other permitted doses were 25, 35, 45, 55, and 70 mg/day). Participants who could not tolerate the 25 mg/day dose were withdrawn from the study.
Endpoints
The present analysis is based on endpoints from the double-blind and OLE studies. Endpoints included PSQI scores at baseline, at the end of the 4-week double-blind treatment period, and at monthly visits during the 6-month OLE; and spontaneously reported sleep-related AEs.
The PSQI is a self-reported instrument that was completed by the adolescents. It evaluates seven sleep characteristics over the previous month: overall sleep quality, sleep latency (time to fall asleep), duration of sleep, sleep efficiency (time asleep as a proportion of time in bed), sleep disturbance, requiring medication to sleep, and daytime dysfunction due to sleepiness. Each subscale is scored from 0 to 3 (with a lower score indicating better sleep quality). Summing the scores for all seven subscales gives a global PSQI score of 0–21. Global PSQI score can be categorized as ≤5 = “Good sleep” and >5 = “Poor sleep” (Buysse et al. 1989). Although the PSQI has not been adapted for the adolescent population, its psychometric properties have previously been tested in adolescents (de la Vega et al. 2015; Passos et al. 2017; Raniti et al. 2018). In the double-blind study, PSQI scores were compared for all PRC-063 doses combined versus placebo.
Spontaneously reported AEs were defined as signs, illnesses, or experiences that developed or worsened during the study. AEs were captured from informed consent through safety follow-up scheduled for ∼14 days after the last study visit in the double-blind study (or, for participants who continued to the OLE study, 14 days after the last study visit in the OLE study).
Statistical analyses
All analyses were conducted using SAS version 9.2 or higher (SAS Institute, Cary, NC). Statistical tests were two sided, and p-values ≤0.05 were considered statistically significant. No adjustments were made for multiple comparisons.
For the double-blind study, the sample size was calculated based on the mean difference between PRC-063 (all doses combined) and placebo for ADHD-5-RS total score (the primary endpoint) (Weiss et al. 2021). Per this sample size calculation, the study planned to randomize 360 participants. No sample size was calculated for the OLE study, because enrollment was based on the number of patients completing the double-blind study.
PSQI scores were analyzed using analysis of covariance models that included baseline score as a covariate. The least-squares (LS) mean change from baseline and LS mean difference between PRC-063 and placebo (with the 95% confidence interval) were calculated. Fisher's exact test was used to compare the proportion of good sleepers (global PSQI score ≤5) between PRC-063 and placebo.
Predictors of poor sleep (global PSQI score >5) at the end of the double-blind study were evaluated in a post hoc analysis using a logistic regression model with the following predictor variables: study treatment, sex (male/female), age (continuous), baseline global PSQI score (continuous), prior ADHD stimulant treatment (yes/no), body mass index (BMI, continuous), and ADHD type (hyperactive–impulsive, inattentive, or combined). A type 3 analysis of effects based on the Wald test was performed.
In the double-blind study, the following characteristics of insomnia as an AE were assessed in a further post hoc analysis: duration (only for resolved events), time of onset as a continuous variable (mean, standard deviation, median, minimum, and maximum), and time of onset as a categorical variable (days 1–7, 8–14, 15–21, 22–28, or 29–36 relative to treatment start). MedDRA version 17.0 was used to code AEs.
Results
Patient disposition
Detailed patient disposition is described elsewhere (Weiss et al. 2021). In brief, for the double-blind study, 450 subjects were screened, of whom 367 were randomized to receive either PRC-063 (n = 293) or placebo (n = 74) and were included in the analysis of PSQI scores and sleep-related AEs. One hundred eighty-one eligible participants entered the OLE study, of whom 176 were included in the PSQI-based analysis of sleep (3 participants were enrolled at a site that was excluded from the analysis and 2 participants provided no data).
Demographics and baseline characteristics
As reported previously (Weiss et al. 2021), the following characteristics were similar at baseline in the PRC-063 (all doses combined) and placebo groups in the double-blind study: mean age, sex and race distributions, and mean BMI. The majority of patients in both treatment groups had combined type ADHD: 72.1% for PRC-063 and 71.8% for placebo. Other psychiatric disorders reported in ≥2% of patients in one or both treatment groups were oppositional defiant disorder (9.2% of patients who received PRC-063 and 14.1% of patients who received placebo), insomnia (6.4% and 8.5%), depression (3.9% and 0%), anxiety (2.8% and 2.8%), sleep disorder (1.1% and 2.8%), tic (0.7% and 2.8%), and initial insomnia (2.1% and 0%). Although melatonin was allowed for patients who had taken a stable dose for ∼4 weeks before double-blind study entry, only a small number of patients (5.1% overall; 5.7% in the PRC-063 group and 2.8% in the placebo group) took melatonin during the study. One patient receiving PRC-063 used a combination hypnotic/sedative.
Change in global PSQI score
There was no statistically significant difference between PRC-063 (all doses combined) and placebo for mean global PSQI scores at the end of double-blind treatment (Fig. 1) or mean change in global PSQI score between baseline and the end of double-blind treatment (Supplementary Table S1). At the end of double-blind treatment, the LS mean change from baseline for global PSQI score was similar for PRC-063 (all doses combined) and placebo (−0.3 vs. −0.5; p = 0.6110). For patients enrolled in the OLE study, mean global PSQI score showed an improved trend during the first month of treatment, which was generally maintained throughout the 6 months of treatment (Fig. 1 and Supplementary Table S1).
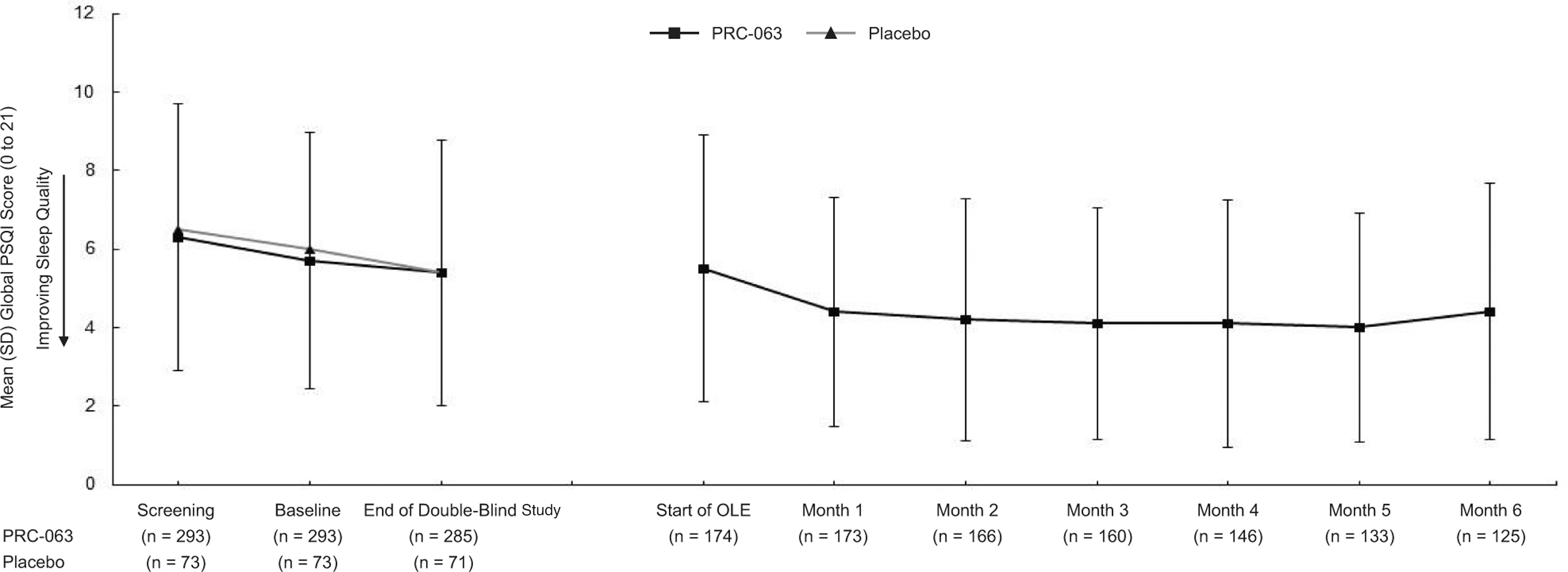
Global PSQI score over time. OLE, open-label extension; PSQI, Pittsburgh Sleep Quality Index; SD, standard deviation.
Changes in PSQI subscale scores
At the end of double-blind treatment, scores for all seven PSQI subscales were similar between patients treated with PRC-063 (all doses combined) and those treated with placebo (Table 1). Mean PSQI scores for all subscales at the end of double-blind treatment were maintained through the end of the OLE study, with the exception of the sleep efficiency subscale, which showed improvement at the end of the OLE study (Table 1).
Pittsburgh Sleep Quality Index Subscale Scores at the End of the Treatment Period: Double-Blind and Open-Label Extension Studies
Change from baseline versus placebo (analysis of covariance with baseline score as a covariate).
CI, confidence interval; LS, least squares; OLE, open-label extension; SD, standard deviation.
Good and poor sleepers by randomized treatment
In the double-blind study, 14.4% of patients treated with PRC-063 went from being poor sleepers (global PSQI score >5) at baseline to good sleepers (global PSQI score ≤5) at the end of treatment, while 11.6% of patients went from being good sleepers at baseline to poor sleepers at the end of treatment. In the placebo group, 11.3% of patients went from being poor sleepers at baseline to good sleepers at the end of treatment, while 5.6% of patients went from being good sleepers at baseline to poor sleepers at the end of treatment (Fig. 2A and Supplementary Table S2). For 74.0% of patients who received PRC-063 and 73.1% of patients who received placebo, sleep status was the same at baseline and the end of double-blind treatment (good at both assessments or poor at both assessments). For patients who entered the OLE study, 21.1% of patients went from being poor sleepers at the end of the double-blind treatment to good sleepers at 6 months, while 8.1% went from being good sleepers to poor sleepers during the same time period (Fig. 2B).
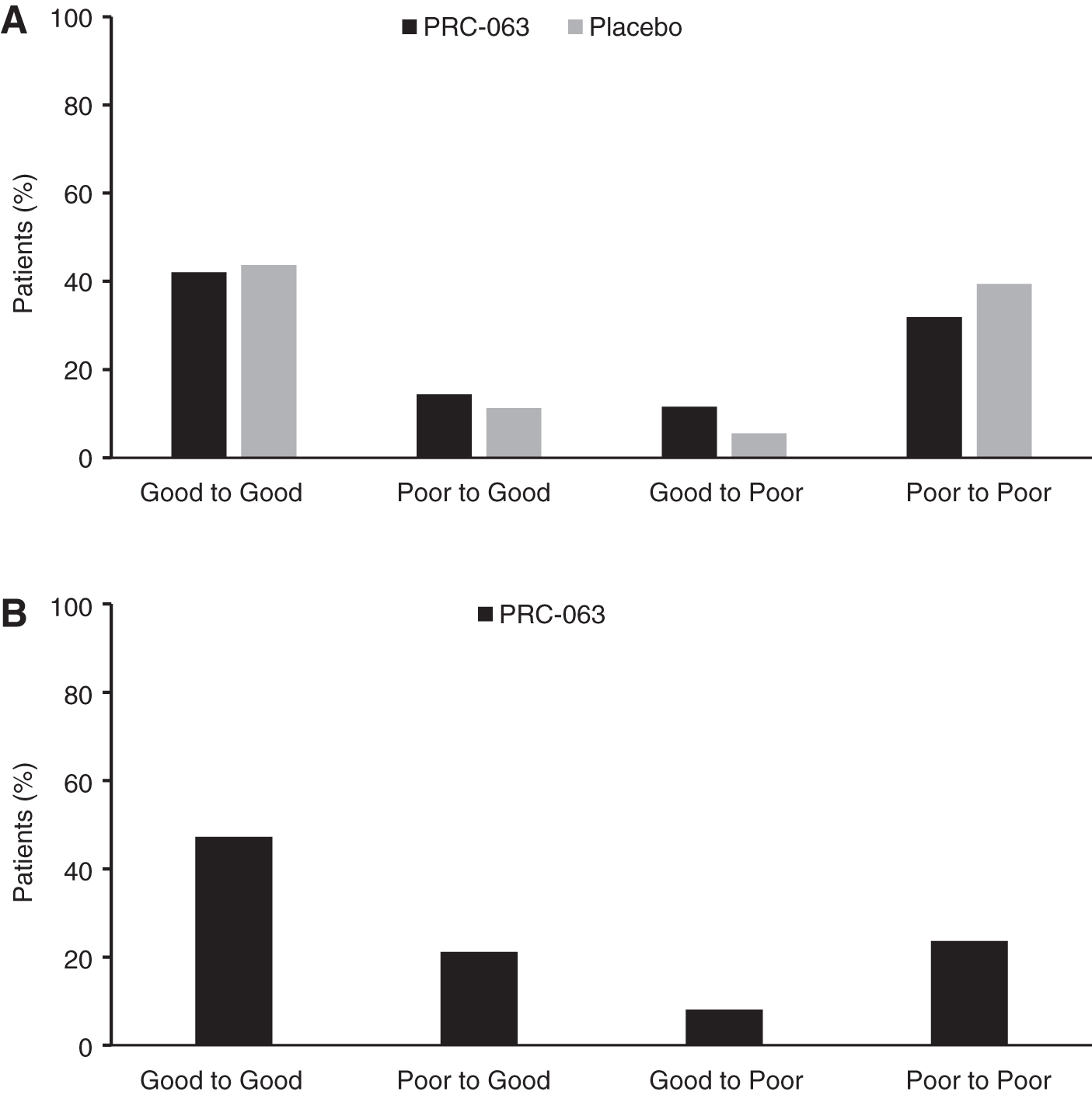
Shift in sleep status during the double-blind study
Predictors of poor sleep at the end of double-blind treatment
In a type 3 logistic regression analysis of effects based on the Wald test, only baseline global PSQI score was a significant predictor of poor sleep at the end of double-blind treatment (odds ratio = 1.587, 95% Wald confidence limit = 1.428–1.763; p < 0.0001) (Supplementary Table S3). Study treatment with PRC-063 was not a significant predictor of poor sleep at the end of double-blind treatment (p = 0.5368). Similarly, sex, age, prior ADHD stimulant treatment, BMI, and ADHD type were not significant predictors of poor sleep.
Incidence of sleep-related AEs
In the double-blind study, PRC-063 (all doses combined) showed higher rates of insomnia and initial insomnia as AEs than placebo (Weiss et al. 2021). In the OLE study, overall rates of insomnia and initial insomnia as AEs were 10.7% and 5.6%, respectively. No events of insomnia led to discontinuation in either study. In the PRC-063 group (all doses combined), the incidence of insomnia AEs (insomnia, initial insomnia, middle insomnia, terminal insomnia) was higher during the first week of double-blind treatment (6.5%) compared with subsequent weeks (Table 2). For patients who were treatment naïve (8.2% vs. 1.4%) and those who had previously received ADHD treatment (3.1% vs. 1.4%), the incidence of insomnia AEs was higher in patients treated with PRC-063 than in patients who received placebo.
Time to Onset of Insomnia and Duration of Insomnia for Insomnia Adverse Events During Double-Blind Treatment
In this analysis, “insomnia” comprises insomnia, initial insomnia, middle insomnia, and terminal insomnia.
ADHD, attention-deficit/hyperactivity disorder; AE, adverse event; SD, standard deviation.
Discussion
This study found that there was no significant difference in any sleep outcomes between adolescents with ADHD randomized to PRC-063 at a daily dose of up to 85 mg and those who received placebo. In the OLE study, treatment with an optimized PRC-063 dose was associated with a trend of improved sleep in the first month, which was then maintained through 6 months. In addition, the proportion of patients who shifted from being poor sleepers (global PSQI score >5) at baseline to good sleepers (global PSQI score ≤5) at the end of double-blind treatment was marginally higher for PRC-063 than for placebo. However, the proportion of patients who shifted from good to poor sleepers was also higher for PRC-063 than for placebo. Finally, the minimal effect of PRC-063 on overall sleep quality was confirmed by a logistic regression analysis, which showed that randomized treatment was not a significant predictor of poor sleep at the end of double-blind treatment.
Our findings mirror previous work in adults demonstrating that while stimulant treatment was not found to impact sleep outcomes versus placebo overall, individual patients may experience either improvement or deterioration in sleep when on active drug (Surman et al. 2019). Our results did not suggest significant differences between PRC-063 and placebo in effects on sleep as an outcome, mirroring findings from previous studies of sleep in children and adolescents during treatment with a 12-hour methylphenidate product (Aptensio XR®) (Owens et al. 2016). By contrast, there appear to be differences between insomnia as an AE with PRC-063 compared with the 16-hour amphetamine formulation SHP465 (Mydayis®): insomnia was a treatment-emergent AE in 38.1% of adolescent patients with ADHD who received SHP465 in a classroom study (Wigal et al. 2019, 2020).
It has been hypothesized that stimulants have a deleterious effect on some aspects of sleep, such as sleep-onset latency, but a beneficial effect on other aspects, such as daytime somnolence. Furthermore, better treatment of ADHD later in the day, with a gradual taper close to bedtime, may be sleep positive for some patients. Total sleep scores may therefore mask particular aspects of sleep impairment. Our results did not suggest differences between PRC-063 and placebo in any sleep outcomes, as measured by the PSQI. Mean PSQI daytime dysfunction due to sleepiness subscale scores were essentially unaffected by PRC-063 treatment in the double-blind and OLE studies. It is possible that stimulants reduce daytime sleepiness by virtue of their alerting effect, while increasing daytime sleepiness as a consequence of their impact on sleep.
A recent meta-analysis of placebo-controlled trials in children and adolescents with ADHD found that methylphenidate was associated with increased risks of various insomnia AEs, including sleep-onset insomnia (Faraone et al. 2019). Although the rate of insomnia as an AE was higher in the 6-month OLE study compared with the 4-week double-blind study (Weiss et al. 2021), the AE profile was within expectations of a methylphenidate product, and patients reported no significant difficulties with falling asleep or staying asleep. None of the patients in either study discontinued due to insomnia AEs. The insomnia AE rates in this study were lower compared with reported rates for 12-hour methylphenidate formulations, SHP465, and other long-acting stimulants (Stein et al. 2012; Coghill et al. 2014; Wigal et al. 2019). Thus, contrary to expectations, the long duration of action of PRC-063 was apparently not associated with a higher rate of sleep-related AEs. One possible explanation for this finding is that occurrence of rebound ADHD symptoms once a 12-hour stimulant has worn off late in the day may be more deleterious to sleep than low residual stimulant levels from a longer acting formulation. In addition, use of a longer acting stimulant may help adolescents to be more efficient in completing tasks such as homework in the evening and may mitigate ADHD-related disruptions, allowing for both an earlier bedtime and better decision making regarding sleep habits.
The studies on which our analyses are based have a number of limitations. Sleep outcomes were based on adolescent self-report, and further study with complementary parent report or actigraphy are needed. Moreover, the randomized, fixed-dose design of the double-blind study means that the assessment of sleep was based on doses that were likely to be either too low (with less impact on sleep) or too high (with more insomnia than would otherwise be expected). In fact, based upon their subsequent optimized open-label PRC-063 dose, 77% of patients in the double-blind study were randomized to a dose that was too high or too low (Weiss et al. 2021), which means that our double-blind study data do not provide information on the level of insomnia for a dose-optimized medication.
It is likely that the OLE sample was affected by selection bias, because it is reasonable to assume that patients who experienced sleep problems during double-blind treatment would be less likely to continue to the open-label study. Furthermore, patients who were known to be nonresponsive to methylphenidate were ineligible to participate in the double-blind and OLE studies. The patient samples in these studies may therefore have been weighted toward responders.
A further limitation is that the studies were not powered to detect differences in subscale scores between treatment groups, so these results should be considered exploratory. The post hoc nature of some other analyses means that they too may have been underpowered. Also, although once-daily PRC-063 treatment may have had a positive impact on rebound, potentially leading to improved sleep-onset latency, we did not measure late afternoon or evening ADHD symptoms to validate this hypothesis. In addition, the possible influence of whether patients participated in the study during periods when they were at school was not investigated. This could have influenced the study outcomes, because sleep patterns in adolescents might be expected to vary depending on their school commitments and school start times. We also did not collect self-report or objective data regarding pubertal status, which is strongly correlated with changes in circadian rhythms and thus with the timing of sleep onset and offset. Finally, a meaningful analysis of duration of insomnia in the OLE study was not possible due to missing data.
Conclusions
Contrary to prior assumptions that stimulant activity late in the day generally causes sleep-onset insomnia, our analyses did not find an increased rate of insomnia AEs with PRC-063 when compared with previous studies of shorter acting extended-release methylphenidate formulations. While individual patients may experience either worsening or improvement in sleep with stimulants, self-reported sleep outcomes on the PSQI were similar for PRC-063 and placebo in the 4-week, fixed-dose, double-blind study. Global PSQI scores subsequently improved, and then stabilized over a 6-month period when patients' PRC-063 dose could be optimized in the OLE study. The improved functional outcomes that may be expected from the extended duration of action of PRC-063, such as an increased capacity for completing homework and a lower likelihood of high-risk activities resulting from poor decision making and impulse control in the evening, do not come at the expense of any worsening of sleep. Measuring sleep as an outcome in its own right, rather than as an AE, leads to a different understanding of the impact of PRC-063 on sleep. Clinicians need to be able to identify specific changes in sleep in both those patients who experience improvement when treated with a stimulant and those who experience sleep difficulty. Further studies should better characterize the population of adolescents with ADHD who are likely to derive sleep benefit or experience insomnia from stimulant treatment.
Clinical Significance
For many years, awareness of sleep-related AEs in some patients treated with stimulants in clinical trials has led health care practitioners to caution patients about the possibility of insomnia when prescribing stimulants for the treatment of ADHD. However, our data for PRC-063 in adolescents with ADHD showed that, while insomnia is an issue for some patients, a substantial number of patients did not have worsened sleep and some showed improvement in sleep, despite the duration of action of the stimulant. Clinical guidance for patients should include systematic assessment of baseline sleep difficulties and re-evaluation post-treatment, as well as assessment of potential improvements in evening activities and bedtime routines. Adolescent patients with ADHD prescribed PRC-063 should be informed that while some patients may experience insomnia, other patients may show improved sleep.
Footnotes
Acknowledgments
Medical writing support was provided by Stephen Gilliver, PhD (Evidera) and Mahesh Paschapur, MPharm (Evidera), and was funded by Purdue in accordance with Good Publication Practice (GPP3) guidelines (
Disclosures
M.D.W. has received consulting fees/honoraria from Tris, Purdue, Adlon, Takeda, Huron, Mundipharma, CBPartners, and Idorsia; support for travel to meetings, article preparation, or other purposes from World Federation of ADHD, Eunethydis, Canadian Attention Deficit Disorder Resource Alliance (CADDRA), Children and Adults with Attention Deficit Disorder (CHADD), American Professional Society for ADHD and Related Disorders (APSARD), Israeli Federation of ADHD, Purdue CA, Purdue US, Rhodes Pharmaceutical, Akili, Takeda, Global Medical Education, and Boston Children's Hospital; payment for lectures from Global Medical Education, Centre for ADHD Awareness Canada (CADDAC), and CADDRA; and royalties from Multi Health Systems and Johns Hopkins University Press. C.S. has received consulting fees from Eisai, McNeil, NLS Pharma, Nutricia, Pfizer, Adlon/Purdue, Rhodes, Shire, Somaxon, Sunovion, Supernus, Takeda, and Teva; research funding from Abbot, Cephalon, Hilda and Preston Davis Foundation, Eli Lilly, Magceutics/Neurocentria, Jazz, Johnson & Johnson/McNeil, Lundbeck, Merck, Nordic Naturals, Shire, and Takeda; payment for lectures from Alcobra, Arbor, McNeil, Janssen, Janssen-Ortho, Novartis, Shire, Reed/MGH Academy (funded by multiple companies), and GME CME (funded by multiple companies); and royalties from Berkeley/Penguin (for “Fast Minds: How to Thrive If You Have ADHD (Or Think You Might)”) and Humana/Springer (for “ADHD in Adults: A Practical Guide to Evaluation and Management”). A.K. has received consulting fees/honoraria and payment for lectures from Elysium and Purdue Pharma. J.O. has received consulting fees from Jazz Pharmaceuticals, Harmony Biosciences, Idorsia, and Clair Labs, and is a member of the Scientific Advisory Board of Sleep Number. E.H. is an employee of Imbrium Therapeutics, a subsidiary of Purdue Pharma, the company that sponsored this research. M.C. is an employee of Purdue Pharma, the company that sponsored this research. G.D. is an employee of Purdue Pharma, the company that sponsored this research.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
