Abstract
Objective:
Neuropsychiatric disorders are common in children with bowel and bladder dysfunction (BBD), a syndrome associated with urinary frequency, urgency, holding, incontinence, and constipation. We evaluated BBD symptom severity in children and youth attending a tertiary care obsessive-compulsive disorder (OCD) clinic.
Methods
: Consecutive patients attending initial OCD assessments between 2016 and 2020 were invited to participate in a registry study. Diagnosis of OCD and comorbidities was established by structured clinical interview. OCD severity and impact were assessed with the Children's Yale-Brown Obsessive-Compulsive Scale (CY-BOCS) and the Child Obsessive Compulsive Impact Scale (COIS-R; self-report), respectively. BBD symptoms were quantified with the Vancouver Symptom Score (VSS), a validated self-report measure.
Results:
One hundred twelve participants completed the VSS (mean age 13.5 ± 3.3, range 7–20). Based on a cutoff score of 11 corresponding to pediatric urologist-diagnosed BBD, 30.4% of participants screened positive, including more females than males (39.3% vs. 21.4%; p = 0.04). Daytime urinary incontinence was present in a greater proportion of participants with OCD forbidden thoughts (34.8% vs. 8.2%, p = 0.002), major depressive disorder (MDD; 38.5% vs. 6.8%, p = 0.001), and somatization disorder (60% vs. 9%, p = 0.001) compared with those without. A regression model including CY-BOCS, COIS-R, psychiatric comorbidities, medications, age, and gender explained 52.2% of the variance in VSS; COIS-R, tic disorder, and MDD were significant predictors.
Conclusion:
BBD symptoms are common and associated with high OCD-related impairment and psychiatric comorbidities. Standardized assessment may facilitate identification of BBD symptoms in this population and is critical to mitigating long-term physical and mental health impacts. Further studies are required to assess the relationship between BBD and OCD treatment outcomes.
Introduction
Bowel and bladder dysfunction (BBD) describes a constellation of lower urinary tract symptoms (LUTS) accompanied by constipation or encopresis. In children, BBD may manifest as day- or night-time incontinence, urinary frequency, urgency, or holding (Yang et al. 2018). Symptoms often persist into adulthood; long-term sequelae include urinary retention and persistent urinary incontinence (Santos et al. 2017). Social stigma related to incontinence is associated with low self-esteem, isolation, poor school performance, and behavioral changes (Hooman et al. 2011; Wolfe-Christensen et al. 2016; Iscan and Ozkayin 2020).
Multiple etiological factors likely contribute to BBD but remain incompletely understood (Franco 2011b). Some voiding behaviors, in particular delay or inhibition of micturition/defecation, may contribute to nonphysiological contraction of the pelvic floor during elimination (Palmer 2010). Maturational delays in nervous system development and bladder sphincter coordination can result in uninhibited detrusor contractions combined with voluntary contraction of the external urethral sphincter, leading to increased bladder pressure (Franco 2007). Factors modulating spinal reflexes and autonomic activity may also contribute (Roy and Green 2019).
Abnormalities in brain regions involved in mapping body sensations (right insula/somatosensory cortex), emotional processing (anterior cingulate cortex/limbic cortex and prefrontal cortex), and decision-making (orbitofrontal cortex) play a role in both continence and psychiatric symptoms (Franco 2011a). 20%–30% of patients with primary nocturnal enuresis may have an underlying neuropsychiatric disorder (von Gontard and Equit 2015; MacNeily 2016), and BBD is frequently diagnosed in children with attention-deficit/hyperactivity disorder (ADHD) (Duel et al. 2003) and autism spectrum disorder (Logan et al. 2014; Niemczyk et al. 2018).
There are more limited data to inform the diagnosis and management of BBD in children with obsessive-compulsive disorder (OCD). Children with OCD may present with urination compulsions (Jiwanmall and Kattula 2016; Rahmat 2020) leading to urinary frequency, or with “phantom incontinence,” a sensation of wetness despite no objective signs based on child/parent report (Arlen et al. 2014). In our clinical experience, bathroom avoidance together with urinary holding, fluid restriction, or saving/storage of urine are common.
Straining to override a tight pelvic floor in an attempt to ensure complete elimination can lead to complications such as varicoceles, epididymitis, and retrograde ejaculation in males. Overwiping can lead to local irritation and, in females, dysuria. Urinary frequency and incontinence have also been described as features of pediatric acute-onset neuropsychiatric syndrome (PANS) and pediatric acute-onset neuropsychiatric disorder associated with streptococcal infection (PANDAS) (Chang et al. 2015). However, it has not become standard practice to assess OCD patients for BBD.
Several questionnaires have been developed for identifying children likely to have urologist-diagnosed BBD. The Vancouver Symptom Score (VSS) was initially validated in 62 children attending a Pediatric Urology clinic at our institution and fits a four-factor model comprising urinary incontinence, urgency, obstructive symptoms, and constipation/fecal soiling (Afshar et al. 2009). It has been further validated in American (Drzewiecki et al. 2012) and Dutch (‘t Hoen et al. 2016) cohorts. The goal of this exploratory study was to use this assessment tool to determine the prevalence of BBD symptoms in children and youth with OCD attending a tertiary care subspecialty clinic, and to evaluate the relationship between BBD symptoms and phenotype, including OCD severity.
Methods
Study cohort
Consecutive patients attending an initial assessment at the BC Children's Hospital Provincial OCD Program between August 2016 and January 2020 were invited to participate in a clinical registry study. Diagnoses of OCD and psychiatric comorbidities were established through a clinical psychologist-administered structured interview and confirmed by a child and adolescent psychiatrist. Baseline assessments were conducted with participants and at least one caregiver.
Parent- and child-report measures were completed online through research electronic data capture (REDCap; Vanderbilt University, Nashville, TN) (Harris et al. 2009). Consenting participants meeting criteria for OCD described by the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV; American Psychiatric Association, 2000) with onset under age 18 who had completed the VSS were included. This study was approved by the University of British Columbia Children's and Women's Research Ethics Board.
Measures
OCD severity was assessed using the Children's Yale-Brown Obsessive-Compulsive Scale (CY-BOCS) (Scahill et al. 1997). Presence of specific symptom dimensions was determined as described previously using the CY-BOCS Symptom Checklist to score each of four factors: forbidden thoughts, symmetry, cleaning, and hoarding (Bloch et al. 2008). Psychiatric disorders were assessed using either the Anxiety Disorders Interview Schedule for DSM-IV: Parent Version (Silverman and Nelles 1988) (2011–2016) or an internally developed semistructured interview (2017–2019). Items were added to address diagnostic criteria for PANS and PANDAS (Swedo et al. 1998; Chang et al. 2015).
The Child Obsessive Compulsive Impact Scale (COIS-R) was used to assess self-reported OCD impact (Piacentini et al. 2007). Urinary and bowel symptoms were evaluated using the VSS (Afshar et al. 2009), a 13-question 52-point questionnaire that queries the quantity and frequency of daytime urinary incontinence, frequency, urgency, holding maneuvers, dysuria, nocturia, enuresis, constipation, and encopresis (Supplementary Fig. S1). Each item is scored on a 5-point Likert scale. A cutoff score ≥11 was used to define a positive screen for BBD (Afshar et al. 2009). Subscores for symptom clusters were generated by adding items together as specified in Table 2 for daytime incontinence, irritative, and obstructive LUTS.
Median Scores for Individual Vancouver Symptom Score Items and Subgroups
Note that a median and IQR of 0 (0–0) does not imply that all respondents rated the item as 0; the second column reports the proportion of respondents endorsing a score of at least 1 on each item.
Greater than median of 0 for controls and equivalent to that of cases in initial VSS scale validation study(20).
Greater than median of 3 for controls (6 for cases) in initial scale validation study.
Greater than median of 6 for controls (14 for cases) in initial scale validation study.
IQR, interquartile range; LUTS, lower urinary tract symptoms; VSS, Vancouver Symptom Score.
Analysis
Statistical analyses were conducted using SPSS (Version 24; IBM Corporation). Differences between means were analyzed by one-way analysis of variance (ANOVA), two-sided Student's t-test, or Mann–Whitney U test (for individual ordinal items or subscores). Pearson's chi-squared test was used to compare frequencies for nominal variables with two or more categories. Associations between continuous variables were evaluated with Pearson or Spearman correlations as specified. Individual VSS item scores are reported as a median and range given the ordinal nature of the data. Initial analyses evaluated differences between individuals screening positive versus negative for BBD.
We conducted further analysis using total VSS score as a continuous outcome to avoid information lost due to arbitrary dichotomization (Fedorov et al. 2009). Multiple linear regression was used to determine predictors of VSS score using stepwise modeling with sequential addition of participant characteristics (CY-BOCS, COIS-R, medications used by ≥10 participants, age/gender, and comorbidities affecting ≥5 participants with a potential association with BBD). Predictors were retained in the models regardless of the statistical significance of their beta estimates. Change statistics were evaluated for each step and the significance of the final model was determined by ANOVA.
Multicollinearity was assessed by examining variance inflation factors (all <3 for included predictors). Internal consistency of the VSS was assessed with Cronbach's alpha. Exploratory factor analysis was performed using principal axis factoring and Varimax rotation with Kaiser normalization. Correction for multiple comparisons was applied using a Benjamini–Hochberg procedure with false discovery rate of 0.05. Proportions of participants are presented as percentages based on the number of responses for the corresponding individual item. Continuous variables are reported as mean ± SD or median (interquartile range [IQR]) as indicated.
Results
Of 130 participants with OCD consenting to registry participation, 112 (86%) completed the VSS. There was no significant difference between participants who completed the VSS compared with those who did not with respect to age (13.5 ± 3.3 years vs. 13.8 ± 2.8 years, p = 0.154), gender (50% vs. 47.4% female, p = 0.832), or CY-BOCS score (23.5 ± 7.0 vs. 25.4 ± 7.2, p = 0.234). Demographic information for the 112 participants completing the VSS is shown in Table 1. CY-BOCS scores reflected moderate-to-severe symptoms in the group overall, and psychiatric comorbidities were prevalent. 30.4% of participants screened positive for BBD, including more females than males (39.3% vs. 21.4%; p = 0.04).
Participant Characteristics
Average age of onset based on parent report of first symptom. Average age at diagnosis was 9.6 ± 5.2 (range 0–18) years.
Fewer than five participants each self-identified as West Asian (3.6%), Indigenous (2.7%), Black (2.7%), Latin American (2.7%), or other race/ethnicity including Arab, Filipino, and Korean (all <2%).
Current or lifetime diagnosis of comorbidity. Anxiety disorders include generalized anxiety disorder, panic disorder, separation anxiety, social anxiety, and specific phobias. Three participants met criteria for PANS and two for PANDAS. One met criteria for intellectual disability, 1 for bulimia nervosa (out of 39 screened for eating disorders), and 4 for psychosis. Medical history based on parent report with potential bowel/bladder implications included Crohn's disease (one participant), diabetes (two participants), and previous head injury (two participants).
CY-BOCS, Children's Yale-Brown Obsessive-Compulsive Scale; OCD, obsessive-compulsive disorder; PANDAS, pediatric acute-onset neuropsychiatric disorder associated with streptococcal infection; PANS, pediatric acute-onset neuropsychiatric syndrome.
The distribution of VSS scores is shown in Figure 1. Mean score was 9.0 ± 5.4 (median 8, IQR 5–12), and was greater in females than males (9.8 ± 5.5 vs. 8.2 ± 5.2, p = 0.0117). Total VSS score was higher in participants with OCD forbidden thoughts (10.2 ± 5.5 vs. 7.3 ± 4.8, p = 0.008), symmetry-related obsessions/compulsions (9.7 ± 5.6 vs. 6.6 ± 3.9, p = 0.004), comorbid ADHD (10.4 ± 5.2 vs. 7.4 ± 4.5, p = 0.004), or somatization disorder (corresponding to a DSM-IV diagnosis of somatoform disorder; 19.0 ± 5.7 vs. 8.1 ± 4.4, p < 0.0005) compared with those without, but did not differ between groups based on other symptom dimensions or comorbidities when corrected for multiple comparisons.
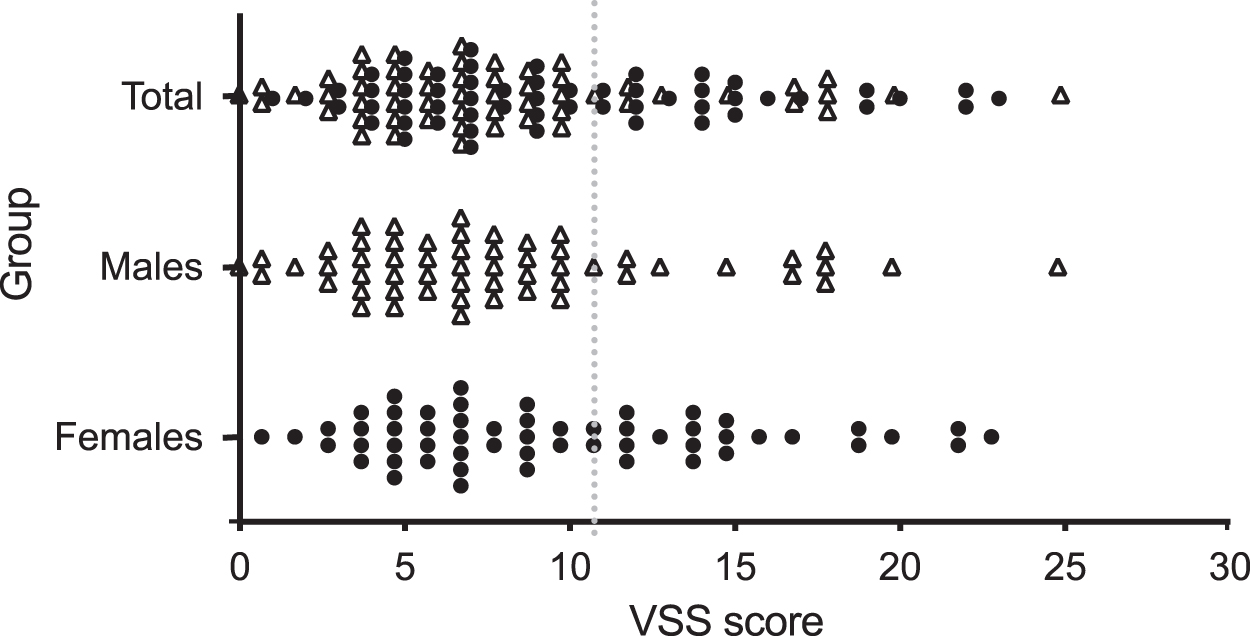
Total distribution of VSS scores. VSS, Vancouver Symptom Score.
Table 2 shows median scores for individual BBD items. More females than males reported waking to void most nights (n = 12/56 or 21.8% vs. n = 2/56 or 3.6%, p = 0.004; item 8). Night-time awakening was also more common among participants with certain comorbidities compared with those without: ADHD (n = 8/41 or 19.5% vs. n = 3/53 or 5.7%, p = 0.038), major depressive disorder (MDD) (n = 4/13 or 30.8% vs. n = 6/71 or 8.3%, p = 0.012), and somatization disorder (n = 2/5 or 40.0% vs. n = 7/77 or 9.1%, p = 0.032) but not anxiety disorders (14.6% vs. 10.0%, p = 0.489), pervasive developmental disorder (n = 0/8 or 0% vs. n = 7/68 or 10.3%, p = 0.341), or tic disorders (n = 9/74 or 12.5% vs. n = 2/21 or 9.5%, p = 0.739).
There was a significant difference in the proportion of participants with any daytime urinary incontinence compared with those without reporting holding of urine more than half the time (n = 5/13 or 38.5% vs. n = 9/97 or 9.3%, p = 0.003; item 5), consistent with the known association between holding and incontinence. Although the proportion of respondents reporting frequent holding in item 5 did not differ based on participant characteristics, urinary infrequency (as reported in item 3, defined as urinating 1–4 times per day) was more common among participants with moderate-to-severe compared with more mild OCD symptoms (n = 61/92 or 66.3% vs. n = 5/14 or 35.7%, p = 0.028).
Of participants with above-threshold VSS scores, only 17.6% reported problems with bladder control or bedwetting on medical history at intake; nevertheless, this is greater than the 3.8% of participants screening negative for BBD (p = 0.013). Total VSS score was greater in participants describing OCD-related bathroom interference as “very much” on the COIS-R (mean VSS score 8.0 ± 5.2) compared with “not at all” (12.2 ± 6.3) on a 5-point Likert scale (p = 0.033).
Total COIS-R score was higher in those with VSS ≥11 compared with those without, as was the prevalence of MDD and somatization disorder (Table 3). Individual OCD symptom dimension severity scores did not vary with BBD status. CY-BOCS score was higher in females with likely BBD compared with those without (26.3 ± 5.3 vs. 22.1 ± 7.3, d = 0.66, p = 0.028) but did not differ among males (21.8 ± 6.9 vs. 23.6 ± 7.4 d = −0.25, p = 0.453).
Participant Characteristics Stratified by Bowel And Bladder Dysfunction Screen Status Based on Vancouver Symptom Score ≥11
Significant differences with correction for multiple comparisons are denoted by *.
Anxiety disorder refers to any of generalized anxiety disorder, separation anxiety disorder, specific phobia, social phobia, or panic disorder. Diagnosis was based on standardized interview except for tic disorders, for which missing data necessitated use of parent-report.
ADHD, attention-deficit/hyperactivity disorder; COIS-SR, Child Obsessive Compulsive Impact Scale revised; OCD, obsessive-compulsive disorder; OR, odds ratio; VSS, Vancouver Symptom Score.
Predictors of total VSS score were assessed using multiple regression (Table 4). CY-BOCS alone did not explain any variance in VSS score (0.000%, p = 0.995). Addition of COIS-R, OCD symptom dimensions, and comorbidities to the model increased explained variance by 23.8% (F-change p < 0.0005), 6.8% (p = 0.273), 6.8% (p = 0.273), and 18.9% (p = 0.003), respectively. The final model including medications, age, and gender explained 53.8% of the total variance. COIS-R (β = 0.483, p = 0.001) and presence of comorbid MDD (β = 0.312, p = 0.020) or tic disorder (β = 0.316, p = 0.008) significantly contributed to the model. When parent- rather than self-reported COIS-R was included, this measure was not a significant predictor (β = 0.262, p = 0.069).
Multiple Linear Regression of Participant Characteristics Associated with Vancouver Symptom Score
n = 61 following listwise deletion of missing data.
Variables explaining a significant proportion of the variance (p < 0.05, not corrected for multiple comparisons) are denoted by *.
ADHD, attention-deficit/hyperactivity disorder; COIS-SR, Child Obsessive Compulsive Impact Scale revised; CY-BOCS, Children's Yale-Brown Obsessive-Compulsive Scale; OCD, obsessive-compulsive disorder.
Exploratory factor analysis was performed to assess empirically derived symptom clusters (Supplementary Table S1). Cronbach's alpha was 0.646, consistent with significant heterogeneity reported previously (Afshar et al. 2009). Five principal components explained 66.8% of the total variance. Common variance explained by the five factors after extraction was 49.4%. Supplementary Table S1 shows the loadings of variables on individual factors. Factors 1–3 corresponded to those described previously (Afshar et al. 2009): irritative symptoms (items 4, 5, 6, and 8, explaining 24% of the total variance); obstructive symptoms (items 9 and 10, explaining 14% of variance); and daytime urinary incontinence (items 1 and 2, explaining 12% of variance).
Although they did not load to a single factor in this analysis, stool symptoms (items 11, 12, and 13) were also combined to generate a subscore as described previously (Afshar et al. 2009). Subscores are reported in Table 2. Daytime urinary incontinence was present in 13% of participants (n = 14), with higher mean subscores in participants with MDD (1.5 ± 2.1 vs. 0.2 ± 0.6, p < 0.001), somatization disorder (2.8 ± 2.8 vs. 0.2 ± 0.7, p < 0.001), and OCD forbidden thoughts (0.7 ± 1.4 vs. 0.0 ± 0.0, p = 0.003), but not other comorbidities or symptom dimensions compared with those without.
Only three participants endorsed encopresis and four participants reported bed-wetting between three nights per month and five nights per week. Supplementary Table S2 shows correlations between participant characteristics defined by continuous variables and VSS subscores. COIS-R score correlated with irritative and obstructive LUTS, and forbidden thoughts with daytime incontinence. CY-BOCS was not associated with any VSS subscore in the overall sample; it correlated weakly with daytime incontinence in females (rs = 0.370, p = 0.006, n = 54) but not in males (rs = 0.056, p = 0.685, n = 54).
Discussion
To our knowledge, this study is the first to employ a standardized assessment tool to quantify BBD symptoms in children and youth with OCD. Our main findings are as follows: (1) BBD is common in this population and may not be captured by general screening questions about bowel or bladder “control”; (2) children with OCD and VSS scores ≥11 are more likely to have comorbid depression or somatization, to report greater OCD-related impairment, and (among females only) to have higher CY-BOCS scores compared with those who do not; (3) predictors of VSS score include self-reported OCD-related impairment, comorbid tic disorder, and MDD; and (4) specific BBD symptoms, including irritative/obstructive LUTS and daytime urinary incontinence, are associated with OCD-related impairment and forbidden thoughts, respectively.
Use of common medications, including serotonin reuptake inhibitors, antipsychotics, and stimulants, were not associated with BBD severity.
The mean VSS score of 9.0 ± 5.4 (median 8) in our cohort was greater than that of control groups in nearly all previous related studies, including 50 children from an otolaryngology clinic (mean score 6.9 ± 3.7, median 6) (Afshar et al. 2009), 133 pediatric urology outpatients without BBD (mean score 7.77 ± 0.42) (Drzewiecki et al. 2012), 60 primary school children age 8–12 (median score 6) (‘t Hoen et al. 2016), and 120 children age 11–17 from primary care and endocrinology clinics, with diabetes (median 7.5) and without (median 6.0) (Kelly et al. 2018). One study reported a mean score of 9.3 ± 7.8 (median 8) in 63 primary care patients age 8–11 attending yearly well-child visits (Ching et al. 2015); however, more than half of participants had chronic emotional/behavioral or physical conditions.
The greater proportion of females than males with BBD symptoms in this study is consistent with previous reports (Drzewiecki et al. 2012). In the absence of a control group, we cannot draw definitive conclusions about BBD prevalence in OCD compared with other groups; however, a positive screen in at least a third of children points to a symptom cluster that may be impairing, missed without standardized assessment, and amenable to treatment.
That participants endorsing significant OCD-related problems with using the bathroom had higher VSS scores than those without suggests that at least some portion of BBD symptoms are likely to be OCD related. The median score for frequency of night-time awakenings to urinate was higher in this study than in previously reported controls (Afshar et al. 2009), perhaps consistent with differences in the perisleep arousal system in pediatric OCD (Jaspers-Fayer et al. 2018). Moreover, urinary holding was more common in participants with moderate-to-severe OCD compared with those with mild symptoms, pointing to the potential role of scheduled voiding as management for both BBD and as a component of exposure–response therapy for OCD.
Factors contributing to avoidance of school or public bathrooms in OCD may include obsessions related to contamination or exposure. Bullying also contributes to school bathroom avoidance (Kistner 2009) and is experienced at higher rates by children with OCD (Storch et al. 2006) and BBD (Ching et al. 2015). It is also possible that primary BBD symptoms trigger or exacerbate OCD obsessions, resulting in a cycle that is challenging to interrupt.
CY-BOCS scores were higher in females who screened positive for BBD compared with those who did not, and were weakly correlated with daytime urinary incontinence. These findings are consistent with the reported association between OCD severity and urinary incontinence among 104 adult inpatients (Drummond et al. 2012). Studies in adults have also identified associations between scores on the Obsessive-Compulsive Inventory and overactive bladder (Hsiao et al. 2014; Ahn et al. 2016). We found that forbidden thoughts were associated with higher VSS scores and with daytime incontinence.
Patients with forbidden thoughts may be more likely to alter their bowel or bladder habits in association with obsessions related to urine, stool, sexual activity, genitals, or other “private” bodily functions. Further study in larger samples is needed to understand how characteristics of patients with OCD—including sex, gender, and symptom content—relate to specific BBD symptoms.
Factors unrelated to specific OCD symptoms but associated with distress may also contribute to BBD severity. Consistent with this hypothesis, variance in total VSS score was predicted by COIS-R, a measure of OCD-specific functional impairment (Piacentini et al. 2007), but not by CY-BOCS. A relationship with functional impairment is consistent with well-characterized associations between BBD and non-OCD psychiatric comorbidities, including MDD (Marciano et al. 2016), stressful life events (Joinson et al. 2016), adverse childhood experiences (Logan et al. 2014), and bullying victimization and perpetration (Ching et al. 2015).
This relationship could also reflect a tendency of those reporting more severe impairment in one domain to attend to symptoms in another due to negative cognitive bias or perfectionism. That self- but not parent-rated COIS-R was a significant predictor of VSS suggests that aspects of OCD-related impairment may not be observable to the family.
This study included only three participants with a diagnosis of PANS and did not detect significant differences in VSS scores based on this diagnosis, although power was limited. Our results emphasize the prevalence of BBD symptoms in general pediatric OCD and their range of severity. Frankovich et al. (2015) described “urinary issues”—including polyuria and enuresis—in 45% of 47 consecutive clinic patients meeting PANS criteria.
Similarly, a study comparing cohorts of patients with PANDAS found a prevalence of urinary complaints between 42% and 81% (Swedo et al. 2015). It is unclear how frequency, urgency, and enuresis have been defined in previous studies and whether they are specific to PANS/PANDAS, OCD symptoms or severity, associated distress, comorbidities, or a combination of these factors. It is difficult to compare our data in the absence of common symptom/frequency definitions. Use of standardized assessments may allow more consistent reporting and comparison across studies.
As suggested by our data, the presence of psychiatric comorbidities—including tic disorder, somatic symptom disorder, MDD, and ADHD—may also affect the severity of BBD in children with OCD. The association with tic disorders is consistent with previously reported delays in normative milestones for bladder and bowel function in Tourette syndrome (Comings and Comings 1987). The presence of other non-OCD comorbidities such as depression also impacts BBD-related quality of life (Vrijens et al. 2015; Lai et al. 2016). Severity of somatization and depression in adults has also been suggested to correlate with LUTS severity, and to respond to treatment for LUTS (Cho et al. 2015). How OCD and psychiatric comorbidities in children affect the response to behavioral treatments for BBD remains to be determined.
Limitations of this study include low power to detect differences in VSS scores based on patient characteristics and a sample size that prohibits assessment of interaction factors in linear regression modeling or predictors of low-frequency symptoms such as incontinence. Because a total cutoff score of 11 has been used previously to categorize patients with likely BBD, we have reported patient-related characteristics based on this stratification, but validation in children with OCD has not been performed.
It is reassuring that previous findings including a higher proportion of females than males with above-threshold scores were replicated in this population. Further study will be required to determine whether BBD prevalence is higher in OCD compared with the general pediatric population, and to determine responsiveness to treatment for both OCD and BBD.
Conclusions
Approximately one third of children and youth attending a tertiary OCD clinic screened positive for BBD, suggesting that bowel and bladder symptoms are not restricted to those with a diagnosis of PANS/PANDAS in whom they have been previously described as pathognomonic. BBD was associated with high OCD-related impairment and psychiatric comorbidities. Standardized assessment may facilitate identification of BBD symptoms in this population and is critical to mitigating long-term physical and mental health impacts in children and youth with OCD. Further studies are required to assess the relationship between BBD and OCD treatment outcomes.
Clinical Significance
This study has several implications for clinical practice. First, general screening questions may not capture the spectrum or severity of problematic bladder symptoms experienced by patients with OCD. Given the potential associated functional impairment, such symptoms should be asked about specifically or a standardized assessment tool employed in psychiatric treatment settings. Second, identification of patients with OCD and other psychiatric comorbidities who may benefit from behavioral treatment for BBD is critical to avoiding long-term physical and mental health sequelae, including the potential for interaction with OCD-related symptoms.
Further studies are required to understand the relationships among specific BBD and OCD symptoms, and to assess responsiveness to treatments for both BBD and OCD. Finally, the association between psychiatric disorders and BBD symptoms identified suggests that thoughtful use of screening tools and appropriate referral to mental health services in the pediatric urology setting may be warranted.
Footnotes
Acknowledgments
We thank BC Children's Hospital Provincial OCD Program staff, Antony Au, Boyee Lin, and Cynthia Lu for their contributions to the OCD registry data collection and maintenance. We also thank Maryam Noparast, Pediatric Urology Research Associate (Division of Pediatric Urology, BC Children's Hospital) for review of the electronic version of the VSS implemented in our registry.
Disclosures
No competing financial interests exist.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
