Abstract
Objective:
This was an exploratory study to examine the use of atypical antipsychotics in an attention-deficit/hyperactivity disorder (ADHD) clinic.
Method:
A total of 194 patients was examined to compare those receiving atypical or second-generation antipsychotics (atypicals) from those who were not. A sample of 27 children on atypicals received laboratory investigation for indicators of possible metabolic effects.
Results:
In all, 19.1% of the patients in the clinic were receiving atypicals with a mean duration of 313 days; 36 of 37 patients on atypicals had received risperidone, with a mean dose of 0.62 mg. Children receiving atypicals were statistically more likely to have a severe co-morbid disorder, a lower Children's Global Assessment Scale score, a greater total score on the teacher Strengths and Difficulties Questionnaire, and greater difficulty with parent-rated symptoms of being touchy, worried, rages, and explosive outbursts. There were no differences found in measures of functioning, adaptive skills, quality of life, or ADHD symptoms. In the subset of children studied for potential metabolic effects, 68.0% had a waist circumference ≥90th percentile that was independent of weight gain, 18.5% had impaired fasting glucose, 12.5% had elevated blood pressure, 11.1% had elevated triglycerides, and 16.7% met full criteria for metabolic syndrome.
Conclusion:
Clinical implementation of the efficacy studies of risperidone for disruptive behavior disorders has led to a significant change in practice. Almost 1 in 5 patients are now receiving atypical neuroleptics, typically to treat severe co-morbid disorders and symptoms other than ADHD per se. Despite these children receiving low doses, concomitant stimulants, and low body mass index z-scores, a significant proportion of children demonstrated either one or more components or the full criteria for metabolic syndrome.
Introduction
Risperidone is unique in that it is the only atypical antipsychotic that has received Food and Drug Adminstration (FDA) approval for the treatment of schizophrenia in adolescents aged 13–17 and bipolar disorder (BPD) in children and adolescents aged 10–17, and for the treatment of irritability associated with autistic disorder in children and adolescents aged 5–16 years, including symptoms of aggression toward others, deliberate self-injuriousness, temper tantrums, and rapidly changing moods. Double-blind, placebo-controlled trials have demonstrated that risperidone is an effective medication to treat aggression, explosive outbursts, irritability, and extreme agitation in children with disruptive behavior disorders of normal intelligence quotient (IQ) (Findling et al. 2000; Pandina et al. 2006). The practice guideline developed by an international group of experts (the International Consensus Statement on ADHD and DBD) recommended risperidone to treat these symptoms in disruptive behavior disorders (Kutcher et al. 2004).
Several authors (Correll and Carlson 2006) have pointed out that the increase in pediatric prescribing of atypicals is occurring in the absence of long-term safety studies. Although there are now more than 19 double-blind trials of risperidone for various pediatric disorders (psychosis, mood disorders, developmental delay, tics, and autism), most of these studies offer limited safety data because of their short duration and/or small sample size (Jensen et al. 2007). There are longer-term pediatric open-label follow-up studies available that look at safety from the point of view of weight gain, hyperprolactinemia, glucose tolerance, extrapyramidal side effects, and tardive dyskinesia (Ross et al. 2003; Correll et al. 2004; Kane et al. 2004; Martin et al. 2004; Saito et al. 2004; Reyes et al. 2006a; Reyes et al. 2006b; Reyes et al. 2006c).
Although there is evidence of risk of metabolic syndrome from the use of atypicals in adults, there is scant data available on this risk in pediatric patients, and no data are available on the risk of metabolic syndrome in children with disruptive behavior disorders who are thin, currently on stimulants, and treated with low doses. The Metabolic Effects of Antipsychotics in Children is a study in progress that has presented preliminary results suggestive that atypicals do present metabolic risks in children (Nicol et al. 2009). Similarly, in a cross-sectional retrospective study of youths admitted to a short-stay emergency psychiatric inpatient unit, we observed that the prevalence of obesity and glucose intolerance was more than double in youths treated with atypicals compared to atypical-naïve youths (Pangiotopoulos et al. 2009). Calarge et al. (2009) recently demonstrated that in 99 youths treated naturalistically with risperidone for an average of 2.9 years there was a significant increase in body mass index (BMI) z-scores (BMIz), which was not attenuated by concomitant use of stimulants. Overweight children had higher odds of increased waist circumference, triglycerides, low high-density lipoprotein cholesterol (HDL-C), and a higher insulin level. Children whose BMIz increased by 0.5 or more showed greater metabolic changes than children who did not meet this criteria. Although the authors did not report findings for the group as a whole, their data indicate significant risk for metabolic disturbances and increased waist circumference, even in the group where BMIz did not increase by greater than 0.5. Therefore, the study did not determine if there was evidence of normoweight insulin resistance. The Treatment Effectiveness of Early Onset Schizophrenia Spectrum (TEOSS) study reviewed the safety and efficacy of molindone (typical antipsychotic), olanzepine, and risperidone in 168 youths for 8 weeks; metabolic data were available for between 18 and 29 subjects on risperidone. The objective of this study was to determine if second-generation antipsychotics were either more effective or safer than the typical neuroleptic molindone. The olanzapine arm was discontinued early by the data mining safety board due to significant increases in fasting cholesterol, low-density lipoprotein cholesterol (LDL-C), insulin, alanine aminotransferase, and aspartate aminotransferase without evidence of difference in efficacy. No significant metabolic changes were noted for risperidone over the 8 weeks (Sikich et al. 2008). Limitations of this study include the lack of power, lack of a normal control group, a 50% attrition rate, and the short duration of follow-up, which was probably too short to capture metabolic changes.
There are three case reports from Ste. Justine Hospital in Montreal, Quebec, of risperidone-emergent diabetes in normal-weight children. Two of these children had absolute BMI scores less than 17; the doses of risperidone did not exceed 1.5 mg, and in one case the dose was 0.5 mg. Two of the cases normalized with discontinuation of the medication, but one did not (Olivier et al. 2008). These cases were of particular interest in demonstrating the possibility of atypical-induced normoweight diabetes. Recent research in endocrinology has demonstrated that central adiposity (i.e., elevated waist circumference) can be associated with insulin resistance in the face of a normal BMI (Sellers et al. 2008).
This paper has two objectives. First, we evaluated our clinic population to determine how the evidence on efficacy of risperidone for symptoms of aggression or irritability had been implemented in practice by evaluating the prevalence and clinical characteristics of the children who did or did not receive augmentation with an atypical. Second, we evaluated children receiving atypicals for abnormalities suggestive of possible metabolic syndrome to determine if this would be an issue in children who were for the most part thin, on stimulants, and receiving very low doses of atypical medication.
Method
Subjects
The Provincial ADHD Program is unique in that it is the exclusive consultation service for assessment and management of ADHD in the province of British Columbia, servicing a total population of approximately 4 million. The clinic assessment protocol includes a semistructured, 2-hour clinical history by a child psychiatrist, rating scales, and the Schedule for Affective Disorders and Schizophrenia–School Age Version–Epidemiological Version (K-SADS-E) done with parent and child together (except in adolescents) (Kaufman et al. 2000; Rucklidge 2008). All patients receive a five-axes Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) diagnoses (American Psychiatric Association 1994), including the Children's Global Assessment Scale (CGAS) on axis V (Shaffer et al. 1983).
Data for this study were obtained from a clinical database of 196 children between the ages of 6 and 18. There were no specific inclusion or exclusion criteria apart from willingness to participate in the study (92% acceptance) and sufficient knowledge of English to complete rating scales. Of these, 194 subjects had complete data and were used for this analysis. Demographics and presenting characteristics are outlined in Table 1. All subjects had provided informed consent and assent for the use of their data for research purposes, and the study received the approval of the university ethics board. Participants had received treatment as usual both through the clinic and community sources, because patients are seen in consultation in the clinic but followed within their local community. The data entered into the database were collected prospectively over the last 4 years. The study sample was divided into three groups: those receiving atypicals with or without stimulants (n = 37), those on stimulant or nonstimulant treatment for ADHD (n = 134), and those who were on no medication (n = 23). Laboratory testing for indicators of metabolic syndrome was available for 27 children who were receiving atypicals.
The standard deviation (SD) or number of cases is in parentheses where appropriate. Percentages may not add up to 100% due to rounding.
Age range was 6–18.
Median values from the Hollingshead Four Factor Index.
6 means “technicians, semi-professionals, small business owners.”
7 means “smaller business owners, farm owners, managers, minor professionals.”
Abbreviations: ADHD = attention-deficit/hyperactivity disorder.
Measures
The clinical assessment includes a battery of rating scales designed to obtain information from parents, teachers, and youth on several different dimensions. ADHD symptoms are assessed by using the 26 item MTA-Swanson, Nolan and Pelham IV (SNAP-IV) scale (Bussing et al. 2008), which rates the 9 items of attention, 9 items of hyperactive impulsive symptoms, and 8 items of oppositional defiant disorder (ODD) on a 4-point Likert scale from “not at all” to “very much.” Broad-spectrum psychopathology as well as ADHD and prosocial skills are assessed with the Strengths and Difficulties Questionnaire (SDQ). The SDQ has the advantage in that it is well-validated, short, and normed on more than 10,000 subjects, with parent, teacher, youth, and follow-up report forms and with well constructed age and gender norms (Goodman et al. 2003). These measures were completed by parents and teachers. Additional parent-rated symptoms were included on the same 4-point Likert scale as the SNAP, which were known to be indications for atypical use including: Touchy, Angry, Physical fights, Verbal fights, Worry, Violent thoughts, Stormy, Mood swings, Self injury, Trouble sleeping, Rages, Explosive, and Reactive to Stress.
Adverse events were measured systematically at all visits and whenever medication was discontinued using the Pediatric Adverse Event Rating Scale (PAERS), developed by John March for the Child and Adolescent Psychiatric Trials Network (March et al. 2007). All adverse events were rated for severity, functional impairment, and likelihood of being related to a particular drug by clinician interview with the parent and child. The likelihood of the event being related to a particular drug is rated by the clinician based on temporal correlations with onset or discontinuation of drug use, past history, and known medication side effect profiles.
Parent ratings of adaptive skills and quality of life were also included to obtain data on developmental skills and quality of life. The Adaptive Behavior Assessment System (Wei et al. 2008) assesses adaptive skills in the areas of Communication, Functional Academics, Self-Direction, Leisure, Social, Community Use, Home Living, Health and Safety, and Self-Care. The measure has been extensively normed on a large, ethnically and regionally diverse population. The Child Health Illness Profile (CHIP) (Riley et al. 2004) is a parent-reported health outcome and quality-of-life measure that assesses five domains of health: Satisfaction, Comfort, Resilience, Risk Avoidance, and Achievement. The CHIP is a quality-of-life measure well suited to ADHD, appropriate to children and adolescents, with excellent North American norms.
The laboratory protocol included measurement of height on calibrated stadiometer sensitive to 0.1 cm and measurement of weight on digital scales sensitive to 0.1 kg. Blood pressure (BP) and pulse were measured by calibrated electronic machines and appropriate cuff size. Waist circumference was measured at the top of the iliac crest (equivalent to around the umbilicus in the majority of children) using an anthropometric tape and standardized as follows. The tape measure was snugly positioned on, but not compressing, the skin. Clothing was positioned so the abdomen was exposed, and recordings were taken at the end of a normal expiration and not during breath holding or abdominal muscle contractions. The laboratory protocol recommended by our program for use of atypicals in children (as of 2009) included: Electrocardiogram (EKG) and fasting lab work (12 hours after last food intake) that included glucose, insulin, cholesterol, HDL-C, LDL-C, triglycerides, liver function tests (aspartate aminotransferase [AST], alanine aminotransferase [ALT], γ-glutamyl transpeptidase [GGT], amylase), thyroid-stimulating hormone (TSH), prolactin, hemoglobin, platelets, and white blood count.
The criteria for abnormal values were defined as follows. BMI was standardized for age and sex and converted to a z-score. Overweight was defined as BMI ≥85th percentile and obese as BMI ≥95th percentile for age and sex (Centers for Disease Control and Prevention 2007). Waist circumference (Fernandez et al. 2004) and blood pressure (Falkner 2004; Falkner and Daniels 2004) were compared to published pediatric standards. A pediatric definition of metabolic syndrome (Cook 2003) was used as modified from that of the Third National Cholesterol Education Program (NCEP III), comprising any three of the following features: Triglycerides ≥1.24 mmol/L; HDL-C ≤1.03 mmol/L; waist circumference ≥90th percentile for age and sex; systolic or diastolic BP ≥90th percentile for age, sex, and height; and fasting glucose ≥5.6 mmol/L (modified to reflect the 2007 American Diabetes Association definition of impaired fasting glucose) (American Diabetes Association 2007). To date, laboratory values have been collected on 27 patients.
Analyses
The prevalence of use of atypicals was estimated by dividing the number of patients having ever taken atypical medication by the total sample. The data were examined for dose, duration of medication, and type of medication. The BMIz was compared between the three groups. The groups were then compared on demographic variables, ratings scales, diagnoses, mental status items noted above, and side effects. Differences among the groups were analyzed using chi-squared or one-way analysis of variance (ANOVA) statistics, depending on the type of data involved, with the Tukey least significant difference test being used for post hoc comparisons when ANOVA results were significant. An alpha value of 0.05 was used for all tests. Because the study was exploratory, no Bonferroni correction was performed. Descriptive statistics were used to present the percentage of patients with laboratory abnormalities.
To determine whether there were any differences in children who were on atypicals previously (n = 16) versus currently (n = 21), we replicated our analyses stratifying for this variable. No significant differences were found.
Results
Demographic differences among the subtype groups
The demographic characteristics of the three subgroups are reported in Table 1. No differences were found among the treatment groups in gender, grade, ethnicity, or parental marital status (p > 0.05). The no-medication group was significantly younger than the two medication treatment groups (F[2, 191] = 3.50, p = 0.032; see Table 1).
Prevalence and characteristics of atypical use
Of the 194 children in the study, 37 (19.1%) were on atypicals. Of these 37 patients, 36 had received risperidone (mean dose 0.62 mg, standard deviation [SD] = 0.26). One patient was taking olanzapine, and 2 patients were on quetiapine, 1 of whom had no previous risperidone trial and was taking quetiapine for insomnia and anxiety. The duration of atypical use is based on duplicate chart prescriptions, pharmacy records, and parental recollection, and is approximate within 2 months or treated as missing data. The mean duration was 313 days (SD = 243, range 6–814 days). For those children on atypical medication, all but 1 (97.2%) were also receiving stimulants and/or atomoxetine. Twenty-three (11.9%) children in the total sample were not taking any atypical or ADHD medication, and 134 (69.1%) children in the total sample had received either stimulants, atomoxetine, or both, but had never taken atypicals.
Height, weight, and vitals
There were no statistically significant overall differences in age- and gender-normed z-scores for BMI among the groups. The sample mean BMIz was 0.08 (SD = 1.55), in the middle of the expected range. When BMI z-scores were dichotomized to those who were overweight or obese (i.e., with a BMI over the 85th percentile) and those in the normal range, there was similarly no significant association between medication group and probability of being overweight (χ2 [2, n = 177] = 6.07, p > 0.05), but the study was not sufficiently powered to find these differences due to the small number of cases of obesity within the subgroups. There were significant differences among the groups in pulse rate (F[2, 167] = 4.67, p = 0.011), because both the atypical group (m = 87.19, SD = 14.02) and the ADHD-medication group (m = 82.70, SD = 13.48) had a significantly higher pulse rate than the no-medication group (m = 75.86, SD = 11.75, p < 0.05). There were no group differences in systolic or diastolic BP.
Differences in DSM-IV diagnoses and symptoms
There were no significant group differences on severity of ADHD symptoms as measured by the parent and teacher SNAP or the ADHD subscale of the parent and teacher SDQ. A number of diagnostic categories were present with sufficient frequency in the sample so as to allow comparison among the groups (see Table 2 for diagnostic presentation). No group differences were found among the groups in ADHD subtype, anxiety disorders, or learning disabilities. There was a nonsignificant trend toward differences among the groups in terms of diagnosis of ODD (χ2 [2, n = 194] = 5.93, p = 0.052). A total of 54.1% of the atypical group, 35.1% of the ADHD-medication group, and 26.1% of the no-medication group had a diagnosis of ODD. There was a nonsignificant trend toward group differences on the parent report SNAP ODD scale, (F[2, 163] = 2.99, p = 0.053); the atypical group (m = 1.36, SD = 0.85) scored above the ADHD-medication group (m = 1.02, SD = 0.72), whereas neither group was different from the no-medication group (m = 1.28, SD = 0.83). However, the same difference did reach significance on the teacher report ODD scale, (F[2, 159] = 5.81, p = 0.004); the atypical group (m = 1.03, SD = 0.82) scored above the ADHD-medication group (m = 0.53, SD = 0.71), whereas neither group was different from the no-medication group (m = 0.72, SD = 0.66).
The standard deviation (SD) or number of cases is in parentheses where appropriate. Percentages may not add up to 100% due to rounding.
Abbreviations: ADHD = attention-deficit/hyperactivity disorder; ODD = opposition defiant disorder; PDD = pervasive developmental disorder.
Because the numbers of participants diagnosed with conduct disorder (CD), autism spectrum disorder (ASD), BPD, reactive attachment disorder (RAD), and borderline personality disorder were too low to obtain adequate power, we clustered all of these diagnoses together as possible severe secondary diagnoses that might be considered a predictor for physician prescription of atypical treatment. Children with any of these severe co-morbid diagnoses were significantly more likely (χ2 [2, n = 194] = 28.24, p < 0.001) to receive atypical antipsychotics. Furthermore, 29.7% of the atypical group having at least one diagnosis of this cluster, 4 had ASD, 4 had CD, 4 had BPD, 1 had RAD, and 1 had borderline personality disorder. Of the ADHD-medication group, 3.7% had a diagnosis from within this cluster (2 had ASD, 3 had CD, and 2 had RAD), whereas no child in the no-medication group had any of these diagnoses.
When we evaluated symptoms commonly considered as indication for use of atypicals, we confirmed that the items for Touchy (F[2, 165] = 3.39, p = 0.036), Worry (F[2, 165] = 4.34, p = 0.015), Rages (F[2, 164] = 4.34, p = 0.014), Reactive to Stress (F[2, 165] = 4.24, p = 0.016), and the Explosive item (F[2, 165] = 7.67, p = 0.001) were significant predictors of atypical use. The only significant difference between the groups on SDQ was teacher report of greater severity both in terms of the SDQ total score and increased Conduct problems (F[2,158] = 7.94, p = 0.001), with the atypical group (m = 3.03, SD = 2.38) scoring significantly higher than the ADHD-medication group (m = 1.50, SD = 1.86), whereas neither group was significantly different from the no-medication group (m = 2.36, SD = 2.06). Again, teacher observation of conduct problems would be consistent with the presence of our finding of serious co-morbidity as a predictor of use of atypicals.
There were significant differences in terms of the clinician CGAS rating (F[2, 191] = 4.38, p = 0.014). The atypical group (m = 44, SD = 14) scored significantly lower than the ADHD-medication group (m = 50, SD = 11), but neither group was significantly different from the no-medication group (m = 48, SD = 9). While teachers and clinicians clearly identified these children as having more serious co-morbidity, overall psychopathology, and lower levels of functioning, the parent reports did not identify statistically significant group differences.
Adverse events
Of the many adverse events covered by the PAERS, only five showed differences among the groups. There was a higher rate of decreased appetite in the ADHD-medication group (33.9%) than in the atypical group (17.1%) or the no-medication group (9.1%) and this difference was significant (χ2 [2, n = 184] = 8.15, p = 0.017). There was also a lower rate of motor restlessness in the ADHD-medication group (18.9%, χ2 [2, n = 184] = 7.18, p = 0.033) than in the atypical (34.3%) or no-medication (40.9%) groups. The atypical group reported more difficulties with emotional lability (40.0%, χ2 [2, n = 184] = 8.15, p = 0.017) than the ADHD-medication (15.7%) or no-medication (18.2%) groups. Rebound showed differences among all three groups (χ2 [2, n = 184] = 6.03, p = 0.049), with the highest rates in the atypical group (22.9%), lower rates in the ADHD-medication group (13.4%), and no reported rebound in the no-medication group. Side effects with acne were reported only in the atypical group (11.4%, χ2 (2, n = 184) = 17.41, p < 0.001). No other adverse events significantly discriminated the groups.
Laboratory evaluation of children on atypicals
Of 27 subjects who consented to have laboratory evaluation, 24 were male and 3 were female. The average age was 12.12 years. The mean BMIz of this group was 0.92 (SD = 0.93, range −1.09 to 3.0), and the mean absolute BMI was 21.9 (SD = 5.3, range 16.0–39.9). Six children (25.2%) were overweight and 4 children (16.8%) were obese. The mean period of atypicals for this subset of children was 19.32 months (SD = 17.2). The proportions of children with clinically abnormal lab results on each measure are shown in Fig. 1. For those measures that were part of the metabolic syndrome criteria, 24 children had complete data available. Of these, 17 (68.0%) had a waist circumference ≥90th percentile, 5 (18.5%) had impaired fasting glucose, 3 (11.1%) had elevated triglycerides, 1 (3.7%) had low HDL-C, and 3 (12.5%) had hypertension. Overall, 4 children (16.7%) met criteria for metabolic syndrome, 3 of whom had three risk factors and 1 of whom had four. A further 1 child (4.2%) had two risk factors and 13 children (54.2%) had one risk factor. In addition, 3 children (12.0%) had clinically elevated fasting insulin (greater than 90 pmol/L), 2 of whom were children with metabolic syndrome. Overall, of the 27 subjects evaluated for metabolic disturbances, 70.4% were found to have at least one abnormality that would be considered a cause for clinical concern.
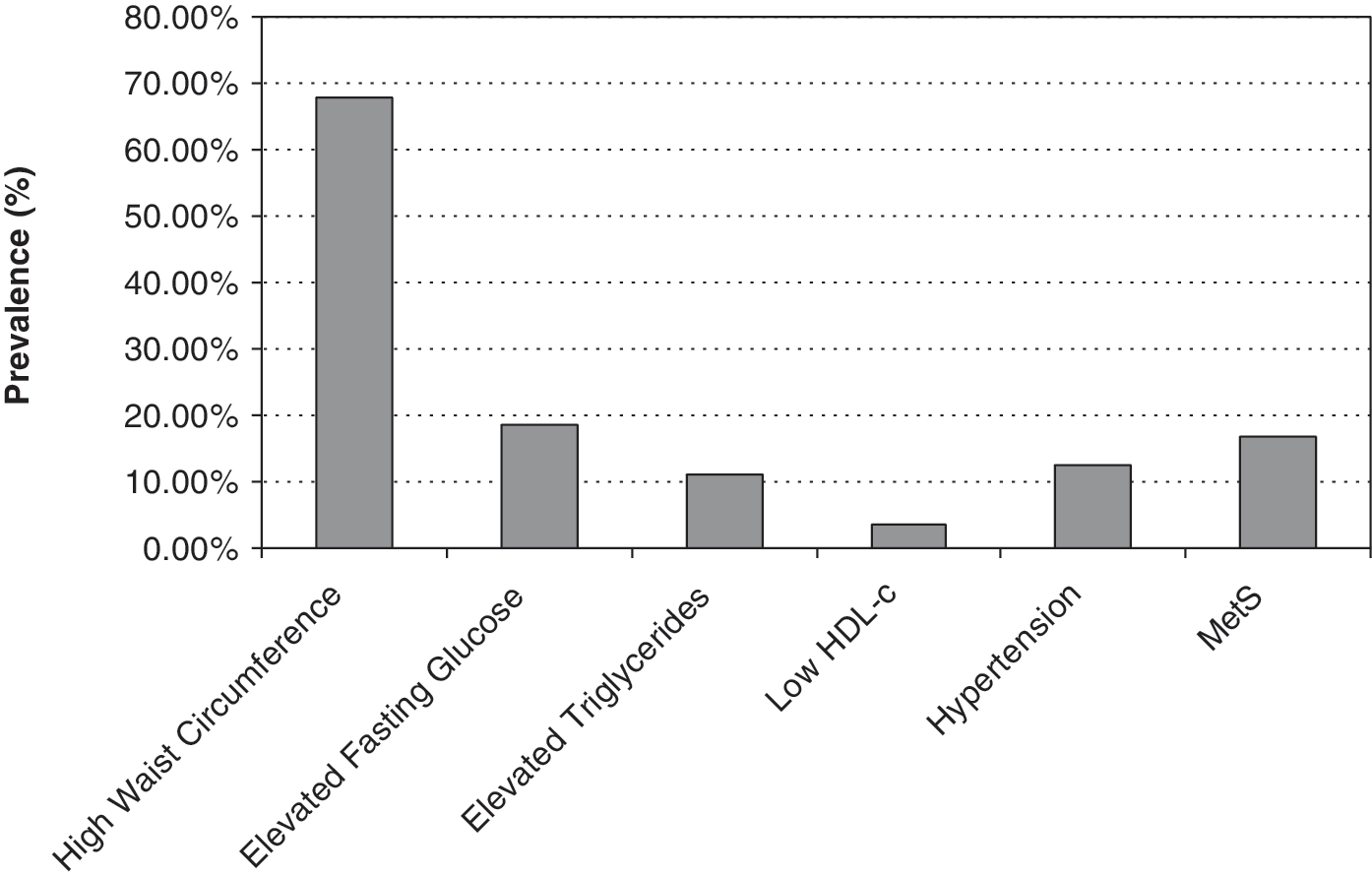
Prevalence of metabolic syndrome and individual components of the syndrome in children with attention-deficit/hyperactivity disorder (ADHD) on atypicals. HDL-c = High-density lipoprotein cholesterol; MetS = metabolic syndrome.
Discussion
Our hypothesis in initiating this clinical review was that a small percentage of ADHD children would be receiving atypical antipsychotics on the basis of the indication of extreme and uncontrollable aggression that was nonresponsive to parent training or other psychosocial treatment routinely offered within our clinic. By contrast, we found that the use of atypicals was not uncommon in our clinical setting, and that the children who were being placed on atypical medication were characterized by various serious and co-morbid conditions and a lower level of functioning. The symptom patterns of these children were consistent with the known response in using atypicals in children with disruptive behavior disorders, but even when evaluated on the atypical medication these children were still more disturbed and dysregulated than the remainder of patients in the ADHD clinic.
The finding on the PAERS that those on atypicals have more rebound than those on stimulants alone may represent a selection bias. Dysregulated and co-morbid children may be more vulnerable to rebound, and this presentation may increase the likelihood of atypical prescription, while it may also be true that concomitant atypical use for this adverse event is not particularly effective. Rebound was defined for the clinicians administering the PAERS as “a worsening of behavior when medication wears off that is atypical for the child's own baseline and reflects anger or irritability that is not characteristic of worsening of ADHD itself.”
We had hypothesized that metabolic abnormalities would not be found because the majority of ADHD children who are on stimulants are thin and the doses used for atypicals in ADHD children are low. Our hypotheses were not validated. Our findings indicated that a substantial fraction of the clinic (19.1%) were receiving atypicals and for substantial periods of time. We did confirm that the doses were low and that atypical medication was being used in addition to stimulants, but neither of these findings was found to prevent metabolic abnormalities. Of the 27 children who received laboratory work-up, 4 (16.7%) met full criteria for metabolic syndrome. Equally significant is the fact that 70.4% of the sample, or 19 out of the 27 children, many of whom were not obese, had at least one abnormal result that would be considered of clinical concern.
The risk for metabolic abnormalities on atypical medication would seem to be relevant to ADHD children both in terms of lipid and glucose metabolism. What we were able to identify and confirm in this paper is the concept of normoweight risk for diabetes. Overall, although there were obese children among those receiving atypicals, the mean BMIz for those on atypicals was not significantly different for those who were on stimulants or no medication. Even children who did not become obese as defined by BMI z-scores often showed abnormally high abdominal girth. Recent research in an Australian Aboriginal pediatric population has demonstrated that central adiposity (i.e., elevated waist circumference) can be associated with insulin resistance in the face of a normal BMI (Sellers et al. 2008).
This study is based on systematic, prospective data collected from a clinical sample. As such, the data reflect treatment as usual and cannot be generalized to making any conclusions as to whether this practice is appropriate or would reflect practice in other settings. Although naturalistic, the data were collected systematically, and many of the results reported are objective even if they are not derived from observers blind to medication status. The N values of the second-generation antipsychatic (SGA) group and treatment-naïve groups are small, and too small to power any subanalyses. Similarly, the N value of the laboratory data is small, although the presence of positive findings of a potentially significant safety concern suggests that if we find these abnormalities in a small sample, they likely would be replicated in a larger study. Although the laboratory data, height, weight, and vitals were collected without blinding to medication status, the standardized nature of these measures makes it highly unlikely that these findings could be explained by investigator bias. Without a control group, our findings represent an association that cannot be considered to be “causal” with respect to atypicals and could theoretically be related to ADHD or other factors. It is possible, for example, that appetite suppression during the day on stimulants and binge appetite increase at night on atypicals lead to a starvation/refeeding pattern that represents the pathway to metabolic stress, rather than the drug itself. Another possibility is that the results reflect the unique attributes of the children selected for atypical treatment, rather than the treatment itself. As this was an exploratory study with multiple analyses, we did not do a Bonferroni correction.
Our findings replicate previous studies (Calarge et al. 2009; Nicol et al. 2009; Panagiotopoulos et al. 2009) in finding elevated risk for metabolic disturbances in children on antipsychotics, although this is the first report that examined children with ADHD, the majority of whom were either thin or not overweight. A strength of this study is that we captured all the children in our clinic who were on atypicals following initiation of the protocol, reducing the chance of selection bias for those who are showing evidence of difficulty. The strength of this early exploratory work is that it indicates that risk for metabolic difficulty may not be limited to patients who have gained weight, especially where there is evidence of increased waist circumference. Our data suggest that further investigation is needed and it cannot be assumed that because a child is thin, receiving low doses of atypicals, or receiving stimulants, that child is not at risk for metabolic problems.
In conclusion, use of low-dose atypical medication in the ADHD population is not uncommon. Atypical antipsychotics are being prescribed for children with severe co-morbidities other than ADHD per se and a lower level of functioning than the rest of the clinic. Atypical antipsychotics used in low doses for disruptive behavior disorders show similar patterns of risk for insulin resistance and changes to lipid profiles in children with ADHD as they do in adults with psychotic disorders. An unanticipated finding of this study has been that it is waist circumference rather than BMI z-score that is most commonly associated with metabolic syndrome. Laboratory evaluation indicated a significant risk for metabolic changes in the pediatric ADHD population, despite low doses, relative absence of obesity, and concurrent use of stimulants. Further research using a randomized controlled design, with safety as the primary objective in this population, is needed to confirm these findings.
Disclosures
Dr. Margaret Weiss is a consultant, has received honoraria and research funds from Shire, Janssen, Purdue, and Eli Lilly. She is a consultant for Takeda, Johnson & Johnson, and Novartis. Dr. Jana Davidson has received honoraria from Purdue. Dr. Constadina Panagiotopoulos, Ms. Lauren Giles, Dr. Chris Gibbins, Mr. Boris Kuzeljevic, and Ms. Rebecca Harrison have no conflicts of interest or financial ties to disclose. Funding to set up the clinic database from which some of this data is drawn was obtained from Janssen, Eli Lily, Purdue, and Shire.
