Abstract
Tetrahydrocannabivarin (THCV) is a phytocannabinoid that is becoming popular across the North American cannabis market. THCV has been reported to reduce blood sugar and act as an appetite suppressant in several independent pre-clinical studies, which has earned it the popular nickname of “diet weed,” despite few human studies of these effects. Additionally, THCV is usually and incorrectly categorized as an intoxicating analogue of tetrahydrocannabinol (THC), which causes confusion among both consumers and regulators. In this article, we examine what is known pre-clinically and clinically about THCV, as well as highlight mechanisms of action, in order to clarify the scientific differences between THCV and THC. THCV, although structurally similar to THC, has distinct pharmacological activity and physiological effects at the doses currently reported in the literature. We highlight areas of opportunity for further THCV research in order to determine the full and appropriate potential for unique health, wellness, and therapeutic applications of this compound.
Introduction
Tetrahydrocannabivarins (THCVs) are not tetrahydrocannabinols (THCs) nor are they THC isomers. There is no known biosynthesis pathway in the cannabis plant nor any synthetic chemical reaction that can convert THC to THCV or
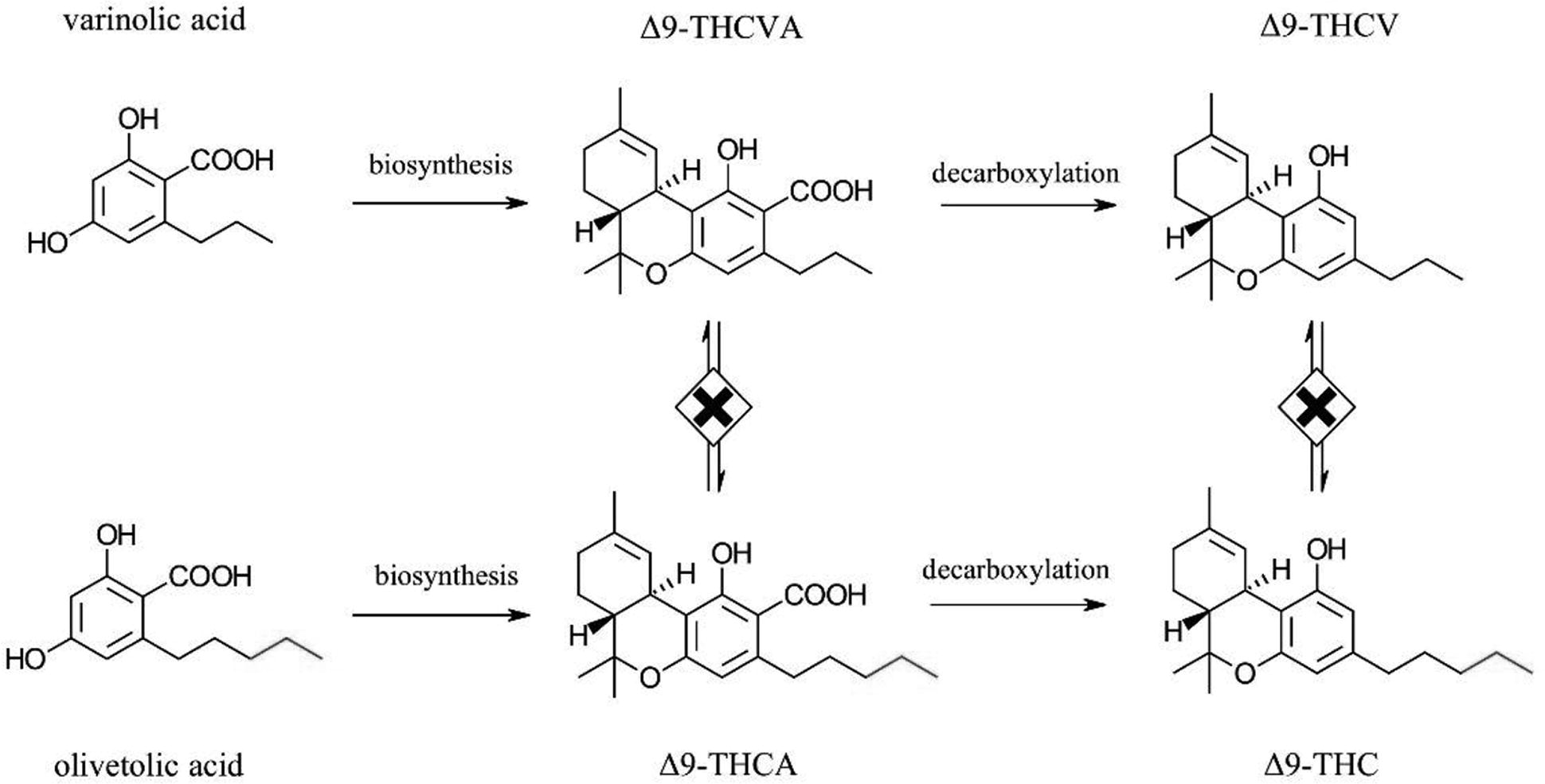
Biosynthesis of △9-THCV and △9-THC from varinolic and olivetolic acids, respectively. THCV, tetrahydrocannabivarin; THC, tetrahydrocannabinols.
One factor possibly contributing to the confusion between THCV and THC is the presence of two common isomers for THCV: Δ9-THCV and Δ8-THCV, similar to the two common isomers for THC, Δ9-THC and Δ8-THC. The Δ9 isomer of THCV, a phytocannabinoid first discovered in
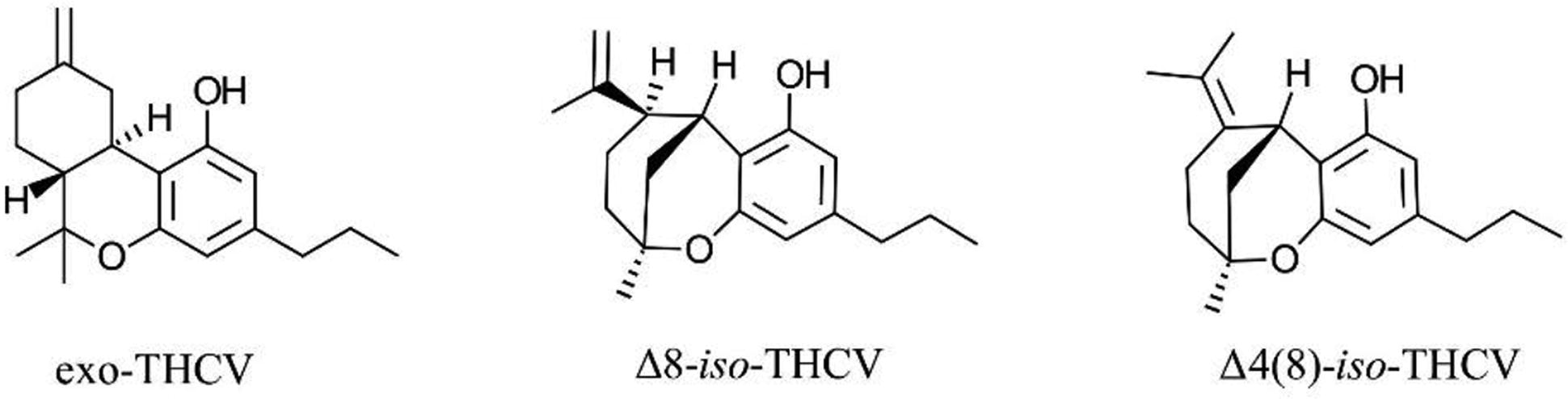
Other THCV isomers that can rise from synthetic or semi-synthetic approaches to Δ8-THCV. THCV, tetrahydrocannabivarin.
Despite the similarity in the isomers for both THCV and THC,
The reason behind the difference in biological properties between THC and THCV is poorly understood. An article by Raïch and coworkers suggests that the difference in the functionality of Δ9-THC and Δ9-THCV at CB1R originates from variation in binding modes that results in qualitatively different effects depending on the signaling pathway engaged upon receptor activation.11,32 However, the functionality of THCV at CB1R appears to be complex, as some studies have highlighted weak CB1R agonist properties for Δ9-THCV. Pertwee suggests that Δ9-THCV behaves mainly as a CB1R antagonist but, at higher doses, Δ9-THCV acts as a CB1R agonist.
7
Indeed, one study has reported Δ9-THCV as a weak partial agonist in the CB1R cAMP inhibition assay.
7
Even weak CB1R agonist activity at high concentrations of Δ9-THCV may lead one to expect intoxicating effects in humans at elevated doses; however, accurately predicting such effects from
A few
We found very limited scientific data in our attempt to compare the activity of THCV and THC at other human receptors. The agonist property of THC (both Δ8 and Δ9) at the CB2 receptor (CB2R) is very well established. 16 THCV is reported to have a weaker affinity toward CB2R than THC (reviewed to some extent here 7 ). However, similar to CB1R, some studies have reported THCV as a potent agonist 17 of CB2R and some as an antagonist. 9 In addition, although both Δ9-THC and Δ9-THCV are reported to affect the serotonergic pathway, particularly through the 5-HT1A receptor,18,19 the absence of side-by-side comparison prevents us from drawing a solid conclusion about functional similarities and differences. Finally, THC and THCV are similarly active at transient receptor potential (TRP) channels such as TRPV1, TRPV2, TRPV3, TRPV4, TRPA1, and TRPM8 (for a review on this topic, see Muller et al. 20 ) with THC being relatively more potent than THCV. However, this similarity is hardly of any significance as almost all phytocannabinoids, intoxicating or not, have some activity at TRP receptors.
Given their differential behavior at CB1R, one area of study has focused on the interaction between THC and THCV. As a competitive antagonist to CB1R agonists, THCV is predicted to act as a competitive antagonist of THC and could inhibit the effects of THC in a dose-dependent fashion in humans and animals. Preliminary scientific data appear to support this hypothesis, at least at specific doses of THCV. Pertwee and coworkers reported that at intravenous doses of 0.1–3 mg, Δ9- and Δ8-THCV attenuated Δ9-THC-induced anti-nociception (tail-flick test) and hypothermia (rectal temperature) in rats, and Δ8-THCV also antagonized Δ9-THC-induced ring immobility.
10
In a recent drug discrimination study,
21
Δ8-THCV (10–100 mg/kg, i.p.) did not alter the discriminative stimulus effects of Δ9-THC in male and female rats trained to discriminate Δ9-THC from vehicle. However, Δ8-THCV showed a weak signal in modulating the Δ9-THC effect by decreasing response rates (response/sec).
21
In humans, 10 mg of oral pure Δ9-THCV administered daily for five days, relative to placebo, inhibited some of the effects of 1 mg intravenous Δ9-THC, such as delayed verbal recall, heart rate, and subjective intensity of effects, although it also increased memory intrusions.
22
The findings of this study may have limited ecological validity, however, as Δ9-THC was administered intravenously rather than
A few human studies of THCV have provided insights into differences in subjective effects relative to THC. There are two human THCV studies that have demonstrated that 10 mg oral Δ9-THCV may be therapeutic in the treatment of obesity
In conclusion, although close in chemical structure, THCV and THC show distinct pharmacological and physiological properties and are produced through distinct biosynthetic pathways. Several published scientific studies have concluded THCV to be an antagonist of CB1R, whereas it is widely accepted that THC is a potent partial agonist for this same receptor. Early human studies indicate that low oral doses (10 mg) of Δ9-THC elicit an intoxicating effect, whereas similar doses of Δ9-THCV do not. Only at much higher oral doses of Δ8-THCV (100 and 200 mg) have any cannabis-like effect been documented, though at a considerably lower magnitude than has been demonstrated with low doses (<10 mg) of Δ9-THC. Although available evidence points to THCV mainly inhibiting the effects of THC, human pharmacokinetic and pharmacodynamic studies of other doses of THCV with oral doses of THC are needed. Based on this perspective of scientific data, we believe that THCV and THC should not be considered in the same category of psychoactive compounds and should not be grouped together from a regulatory standpoint. We also note the potential difference in antagonist potency of Δ9-THCV and Δ8-THCV, suggesting that one of the next steps in THCV research can be devoted to understanding the difference in dose-effect of these isomers in humans.
Footnotes
Author Disclosure Statement
M.H. and M.R. are employees of Nalu Bio. M.H. and E.N.P. are both consultants for Charlotte’s Web and former employees at Canopy Growth Corporation. M.H. is also a former employee of Organigram. M.B.M. is an employee of Charlotte’s Web, Board Member at DeFloria, LLC, and former employee at Canopy Growth Corporation.
Funding Information
No funding was received for this article.
