Abstract
Introduction:
Studies indicate that ∼7% of pregnant individuals in North America consume cannabis in pregnancy. Pre-clinical studies have established that maternal exposure to Δ9-tetrahydrocannabinol (THC; major psychoactive component in cannabis) leads to fetal growth restriction and impaired cardiac function in offspring. However, the effects of maternal exposure to cannabidiol (CBD; major non-euphoric constituent) on cardiac outcomes in offspring remain unknown. Therefore, our objective is to investigate the functional and underlying molecular impacts in the hearts of offspring exposed to CBD in pregnancy.
Methods:
Pregnant Wistar rats were exposed to either 3 or 30 mg/kg CBD or vehicle control i.p. daily from gestational day 6 to term. Echocardiography was used to assess cardiac function in male and female offspring at postnatal day (PND) 21. Furthermore, quantitative polymerase chain reaction (qPCR), immunoblotting, and bulk RNA-sequencing (RNA-seq) were performed on PND21 offspring hearts.
Results:
Despite no differences in the heart-to-body weight ratio, both doses of CBD led to reduced cardiac function exclusively in male offspring at 3 weeks of age. Underlying this, significant alterations in the expression of the endocannabinoid system (ECS; e.g., decreased cannabinoid receptor 2) were observed. In addition, bulk RNA-seq data demonstrated transcriptional pathways significantly enriched in mitochondrial function/metabolism as well as development.
Conclusion:
Collectively, we demonstrated for the first time that gestational exposure to CBD, a constituent perceived as safe, leads to early sex-specific postnatal cardiac deficits and alterations in the cardiac ECS in offspring.
Introduction
With the increased legalization across North America, studies suggest that the self-reported prevalence of cannabis use in pregnancy is ∼7%, with daily or near-daily use at 3.4%. 1 Depending on the population studied, sample size, definition of cannabis use, and the methods of detecting cannabis, prevalence can range anywhere between 2% and 35%.1–4 Studies indicate that consumption of cannabis products may be due to managing preexisting conditions such as depression and chronic pain as well as pregnancy-related symptoms such as nausea. 5 Furthermore, a large-scale integrative review indicates that the uncertainty regarding adverse perinatal consequences was a strong driver for continued use in pregnancy. 6 Overall, this is of concern given some clinical studies report that maternal cannabis use leads to low birthweight outcomes,7,8 which is associated with an increased risk for cardiovascular disease. 9
Δ 9 -tetrahydrocannabinol (THC), the major psychoactive constituent, and cannabidiol (CBD) the major non-euphoric constituent, are the two most abundant and widely known constituents in cannabis. THC and CBD are highly lipophilic and can cross the placental barrier into fetal circulation where they can impact placental function and/or exert direct effects on the developing fetus. 10 To date, we and others have demonstrated that prenatal THC exposure in rodents leads to placental insufficiency and fetal growth restriction.11–14 Furthermore, this results in abnormal fetal heart development, postnatal cardiac remodeling, and impaired cardiac function in 3-week old offspring.15,16 However, the impact that prenatal CBD has on fetal development and postnatal heart function remains unknown. This is a crucial knowledge gap given that CBD products have skyrocketed over the last few years and are perceived and marketed as a safe and therapeutic constituent. 17
A recent Canadian study indicates that up to one in four North Americans report using CBD products 18 and another study reporting that a majority (62%) of those who consume CBD use it to treat medical conditions such as pain and anxiety, 19 both of which are common symptoms of pregnancy. Indeed, preliminary data suggest that 20–30% of women would take CBD products to treat nausea, anxiety, and pain during pregnancy. 20 To date, there are limited pre-clinical studies investigating the safety of prenatal CBD exposure on offspring. Among the studies that do, only the reproductive,21,22 neurobehavioral,23–25 hepatic, 26 and pancreatic outcomes 27 in offspring have been explored. Given that maternal THC exposure impacts heart function in offspring,15,16 and that Δ 9 -THC and CBD may share some common signaling pathways, 28 there is great impetus to study the effects CBD has on fetal development and postnatal cardiac outcomes.
Phytocannabinoids can interact with the canonical endocannabinoid system (ECS), most well-studied through the cannabinoid receptor 1 (CB1) and 2 (CB2). This ECS also includes endogenously produced endocannabinoids, namely anandamide (AEA) and 2-arachidonyl glycerol (2-AG) and their respective enzymes responsible for its turnover. This system is known to be expressed in peripheral tissue, including the heart (CB1 and CB2 found in the rat and human myocardium29,30 as well as cardiomyocytes 31 ). In the heart, it seems that the regulation of the ECS is not involved in cardiovascular function under normal physiological conditions, however, cardiac pathologies are associated with an altered ECS, as reviewed.32–36 This altered ECS is thought to be a cardioprotective response with much of the protective role seemingly attributed to the activation of CB2 in the heart.35,36
Conversely, current knowledge on the cardiac ECS suggests that increases in CB1 activation have cardiodeleterious effects, while antagonism has cardioprotective effects, as previously reviewed.32,35,36 Despite the importance of endocannabinoids in cardiovascular disease, little is known regarding the effects of the ECS on the heart after gestational cannabinoid exposure. Interestingly, CBD has been demonstrated to modulate the ECS.37–39 For example, CBD has been reported to be a negative allosteric modulator of the CB1 receptor 38 and to inhibit fatty acid amide hydrolase (FAAH, a major enzyme that degrades the endocannabinoid AEA) in mouse brain, 39 which may increase AEA. However, it remains elusive if this occurs during critical windows of heart development.
This opens the possibility for CBD to “rewire” the ECS in development. It has been previously postulated that cannabinoid exposure in utero may be a “first hit” to the ECS, jeopardizing normal ECS development, which may increase susceptibility to future stressors and cardiovascular disease. 40 Therefore, the goal of this study is to investigate the effects of prenatal CBD exposure on the postnatal cardiac function and to determine if this is associated with alterations in the ECS in the heart.
Methods
CBD animal model
All animal procedures were conducted in accordance with the guidelines and standards of the Canadian Council on Animal Care. Animal Use Protocol (AUP No. 2019-126) was approved and was monitored by the Western University Animal Care Committee. All investigators understood and followed the ethical principles outlined by Grundy 41 and the methods were performed in accordance with Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines. Time-pregnant Wistar rat dams were purchased from Charles River (La Salle) and were maintained at 22°C on a light–dark cycle (12:12 light:dark) with access to food and water ad libitum. Dams arrived at the animal facility at gestational day (GD) 3 and were left to acclimatize for 3 days. Throughout gestation, maternal food intake and body weight were monitored to study the maternal weight gain and nutrition, as previously published. 11 Length of gestation was also measured to account for pre-term labor.
At birth, litters were counted and culled to 8 for uniform postnatal nutrition. Of the culled pups, hearts were weighed and flash frozen for RNA analysis. At postnatal day (PND) 21, male and female offspring were sacrificed with an overdose of pentobarbital i.p. followed by decapitation. Heart tissues were dissected, weighed, and flash frozen in liquid nitrogen for further protein and RNA analysis.
Drug preparation and exposure
CBD was received in powder form with 99.9% purity (Canopy) and was mixed with cremophor to the experimental concentrations. Similar to our previously published prenatal cannabinoid exposure paradigms,11,15,42–45 pregnant dams were randomly assigned to a treatment group and administered 3 mg/kg (n=8), 30 mg/kg of CBD (n=9), or vehicle control (1:18 cremophor: saline, n=8) daily via i.p. injection from GD6 to GD22 (birth). This window of exposure was selected because it has been demonstrated that THC exposure before GD5.5 adversely impacts implantation in rodents. 46
Although, to our knowledge, no studies examine the effects of CBD on implantation and spontaneous abortion in rodents before GD5.5, in vitro studies suggest that it may impact implantation. 47 The low dose of 3 mg/kg i.p. CBD has been previously reported to result in a serum concentration of ∼9 ng/mL. 48 Chronic exposure (14 days) to 30 mg/kg CBD per day has also been demonstrated to be safe and lead to stable serum concentrations of 180–220 ng/mL in vivo. 49 While humans studies on maternal CBD plasma concentrations are limited, we chose these doses of CBD to reflect the wide range of CBD reported in human umbilical cord tissue (10–332 ng/mL) in fetuses exposed to cannabis in pregnancy. 50 With respect to nonpregnant people, CBD plasma levels also vary greatly between individuals and depend on dose and route of exposure.
A small study demonstrated that individuals smoking a single cigarette led to an average peak blood plasma level of CBD, 110 ng/mL (range: 42–191 ng/mL), and in another study, 20 mg [2H]CBD via i.v. injection led to CBD plasma levels peaking at 686 ng/mL (356–962 ng/mL)—as reviewed. 51 Moreover, our dose range of CBD has an estimated human equivalent dose range of ∼0.48–4.8 mg/kg using scaling factors for body surface area. 52
Echocardiographic assessment of cardiac function
The Vevo2100 Ultrasound Imaging System was used to assess cardiac function. A two-dimensional echocardiographic footage was captured blinded in PND21 vehicle and CBD-exposed offspring. Parasternal short axial and long axial views were captured using a 40 MHz linear transducer, as previously published. 15 Throughout the echocardiography, animals were sedated using isoflurane (0.5–1.5%) through a nose cone. Heart rate was measured using electrode probes on the extremities and body temperature was maintained with a heating dock and monitored using a rectal probe. Real-time images obtained in the short axial view were used to measure parameters such as left ventricular posterior wall (LVPW) and anterior wall (LVAW) thickness as well as left ventricular interior diameter (LVID) at systole and diastole. Estimates were made for cardiac function parameters (e.g., stroke volume, ejection fraction and cardiac output) using left ventricular traces in systole and diastole. 53
RNA extractions and real-time quantitative reverse transcription polymerase chain reaction
Total RNA was isolated from rat heart tissue using TRIzol (Invitrogen) and chloroform (Sigma-Aldrich) extraction. RNA was diluted to 2 μg for reverse-transcriptase with a High-Capacity cDNA (complementary DNA) Reverse Transcriptase Kit (No. 4368814; Applied Biosystems) to make cDNA. Primer sets were designed using the NCBI Primer-BLAST tool (Table 1). SsoFast Eva green supermix (Bio-Rad) and Bio-Rad CFX384 Real-Time System were used with cyclic conditions set at 95°C for 3 min, followed by 43 cycles of 95°C for 15 sec, 58°C for 30 sec, and 72°C for 30 sec. All primer sets used were tested for one distinct melt peak and determined to fall within 90–110% primer efficiency. Relative transcript (messenger RNA [mRNA]) abundance obtained for all target genes of interest was normalized to the geometric means of 18S and β-actin.
Primer Sequences for Quantitative Real-Time Polymerase Chain Reaction
CB1, cannabinoid receptor 1; CB2, cannabinoid receptor 2; MGL, monoacylglycerol lipase.
The ΔCt values for each primer were calibrated to experimental samples with the lowest transcript abundance (highest Ct value). Relative transcript abundance was then calculated for each primer set as determined by the formula 2ΔΔCt, where ΔΔCt was the normalized value.
Protein extraction and Western blot
Total protein from rat heart tissue was extracted by homogenization and sonication in RIPA buffer solution with protease and phosphatase inhibitors. Total protein samples were quantified using a BCA protein assay kit (No. 23225; Thermo Scientific) and diluted to 20 μg/well of protein. A pooled sample was prepared with contribution from every sample across treatment groups. Protein samples were separated via gel electrophoresis using 4–12% Bis-Tris gradient gels (Invitrogen) and transferred onto polyvinylidene difluoride membranes. Ponceau S (0.1%) staining was applied to the membranes and imaged to ensure equal loading and proper transfer of proteins. Membranes were blocked with 5% bovine serum albumin (BSA) for 1 h at room temperature and then probed overnight in 4° with a primary antibody for CB2 (ab3561; Abcam) or monoacylglycerol lipase (MGL; sc-398942; Santa Cruz), diluted to 1:2000 and 1:1000 in 5% BSA, respectively.
Goat anti-rabbit (7074P2; Cell Signaling Technology) or horse anti-mouse (7076S; Cell Signaling Technology) IgG HRP-linked secondary antibody was diluted to 1:10,000 in 5% BSA and rotated at room temperature for 1 h. Immunoreactive bands were detected using the Super Signal West Dura Chemiluminescent Substrate (Thermo Fisher Scientific) and then imaged on ChemiDoc Imager (Bio-Rad). For quantification, relative band density was normalized to total protein using Ponceau S staining and quantified using the Image Lab software 6.0.1 (Bio-Rad). When two blots were used for quantitative measurements, a pooled sample ratio between the two blots was used to normalize densitometry values.
RNA-sequencing and analysis
Previously collected and frozen hearts from 3-week-old male vehicle (n=7), CBD 3 mg/kg (n=6)-, and CBD 30 mg/kg (n=5)-exposed offspring were sent to Genome Quebec (Montreal, Canada) for total TRIzol-phenol RNA extraction, library preparation, and RNA-sequencing (RNA-seq). Quality of RNA was assessed using the RNA integrity number (RIN), with all samples having an RIN score ≥7.0. Paired end reads (25 million, 100 bp) were sequenced on the Illumina NovaSeq platform (NovaSeq 6000).
All reads were aligned and annotated with the latest ENSEMBL rat genome release available (mRatBN7.2) using the R package Rsubread with default settings. Raw counts were generated using the Rsubread sub package featureCounts. To correct for batch effects, the R package ComBat_seq was used. 54 The treatment group of the hearts was included as a factor for the model. Lowly expressed genes were then filtered out using a counts per million (CPM) cutoff of 0.5 in at least five or more samples. To convert ENSEMBL gene IDs to gene symbols, the gprofiler2 R interface for g:Profiler was used. Normalization and differential expression analysis were done using the edgeR R package. 55 Counts were normalized for both library size and library composition using the trimmed means of M-values (TMM) method.
Normalized counts were then fit to a genewise negative binomial generalized linear model, and a quasilikelihood F test used for differential expression analysis of contrasts of interest. To account for multiple testing, p-values were adjusted using Benjamini & Hochberg false discovery rate (FDR) correction. An FDR cutoff of <0.05 was used to determine significance. Gene set enrichment analysis (GSEA) using the Gene Ontology (GO) database was performed using clusterProfiler. To generate a ranked metric for the GSEA, the −log10 transformed unadjusted p-values for all genes in the analysis (14,959 genes) were multiplied by the sign of their respective fold change and arranged in descending order. Significance for the GSEA was determined with an FDR cutoff of <0.01.
Statistical analysis
All maternal, neonatal, echocardiographic, quantitative polymerase chain reaction (qPCR), and immunoblotting data are represented as mean±standard error of the mean. We sought to determine whether either dose of CBD led to changes relative to the vehicle control, therefore one-way analysis of variance (ANOVA) with post hoc Dunnett's multiple comparisons test was used. Values of p<0.05 were considered statistically significant. Statistical outliers were determined using Grubb's test. All aforementioned statistical tests were completed with GraphPad 8 Prism software. For statistical analysis used in RNA-seq, see above in the “RNA-Sequencing and Analysis” section. Throughout all experiments, to avoid litter bias, offspring were randomly selected from separate litters (i.e., n=1 represents pups from a single pregnant rat).
Results
CBD exposure in rats does not affect maternal weight gain and food intake
Similar to previous studies with maternal THC exposure,11,12,56 maternal exposure to a daily dose of 3 or 30 mg/kg i.p. injections from GD6 to GD22 did not alter maternal food intake or weight gain (Table 2). Moreover, both doses of CBD did not affect litter sizes and nearly all pups were born at-term, and therefore, there were no significant differences in gestational length (Table 2).
Maternal Outcome Measurements
All values are expressed as mean±SEM, N=8–9 moms/group. PND21 survival measurements include all males and females. One-way ANOVA post hoc Dunnett's was used for statistical analysis.
ANOVA, analysis of variance; CBD, cannabidiol; GD, gestational day; PND, postnatal day; SEM, standard error of the mean; VEH, vehicle.
CBD exposure in rats does not affect neonatal and postnatal weight measurements
Given previous studies demonstrated that maternal THC exposure leads to fetal growth deficits 11 and smaller hearts at birth, 15 we measured these parameters in the current study but found that neither doses of prenatal CBD altered birthweight or heart-to-body weight ratios at birth (Table 3). Similarly, both sexes at PND21 did not exhibit significant changes in body weight or heart-to-body weight ratios in either dose relative to vehicle control (Table 3).
Offspring Postnatal Weight Measurements
All values are expressed as mean±SEM, N=7
PND21 male but not female offspring exposed to a daily dose of 3 or 30 mg/kg CBD in gestation exhibit reduced cardiac function
To determine if maternal CBD exposure impacts postnatal left ventricular function in offspring, echocardiography was performed on males and females at PND21. Male offspring exposed to 3 and 30 mg/kg CBD in gestation demonstrated a significant (p<0.05) reduction of ∼27% and ∼30% in stroke volume, respectively (Fig. 1b). In addition, male offspring exposed to either dose of CBD exhibited a significant (p<0.01 for CBD3; p<0.05 for CBD30) decrease of >30% in cardiac output (Fig. 1c). Furthermore, male offspring exposed to 30 mg/kg CBD displayed a significant (p<0.01) 10% reduction in ejection fraction (Fig. 1d). Furthermore, there were no immediate signs of neonatal mortality up to PND21 in both doses, nor were there any changes in LVID (Fig. 1f, g), wall thickness, LV mass, or LV volume at systole and diastole via echocardiography (Supplementary Table S1).
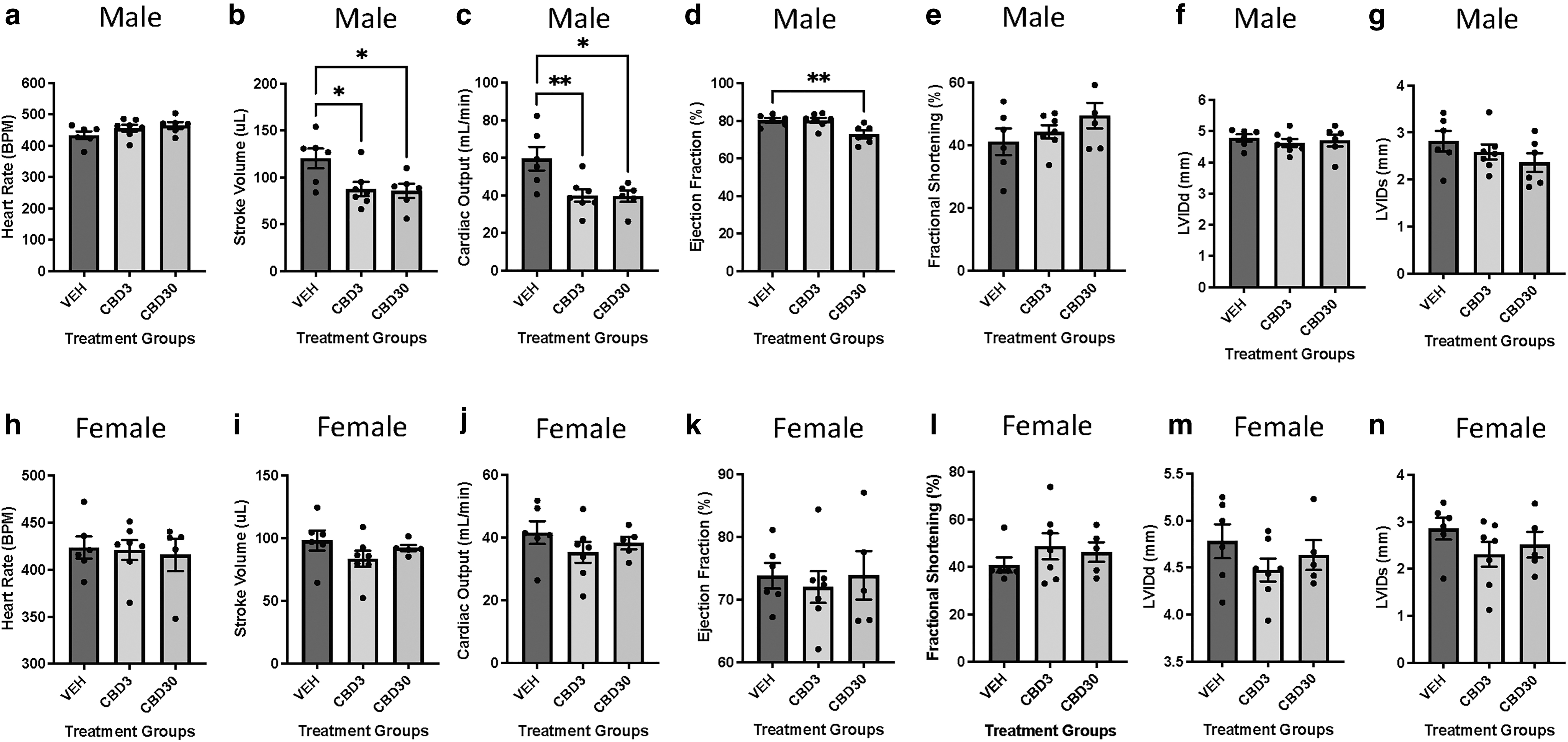
Echocardiographic assessment indicates that 3 and 30 mg/kg/day CBD during gestation leads to decreased cardiac function in males but not female offspring at PND21. Male
Unlike males, the cardiac function in female CBD-exposed offspring remained unaffected relative to their vehicle control groups at PND21 (Fig. 1h–n and Supplementary Table S1).
The ECS was altered in PND21 male offspring hearts exposed to CBD in gestation
Given that the ECS is known to be dysregulated in cardiovascular disease state,34–36 we next sought to determine whether the observed cardiac deficits in CBD-exposed offspring males were associated with sex-specific alteration in the ECS. To investigate whether males and females exhibited changes in the cardiac ECS, we measured the steady-state mRNA transcript abundance of the major endocannabinoid receptors as well as degradative and synthesizing endocannabinoid enzymes. This included CB1 and CB2 as well as synthesizing (NAPE-PLD, N-acyl phosphatidylethanolamine phospholipase D and DAGL, diacylglycerol lipase) and degrading enzymes (FAAH and MGL). qPCR data indicated that there was a significant (p<0.05) decrease in the CB2 receptor mRNA expression (Fig. 2b) in male offspring hearts exposed to 30 mg/kg CBD in gestation. Consistent with this trend, immunoblots also demonstrated a significant (p<0.05) decrease in protein expression of CB2 in these CBD-exposed male offspring hearts (Fig. 2m).
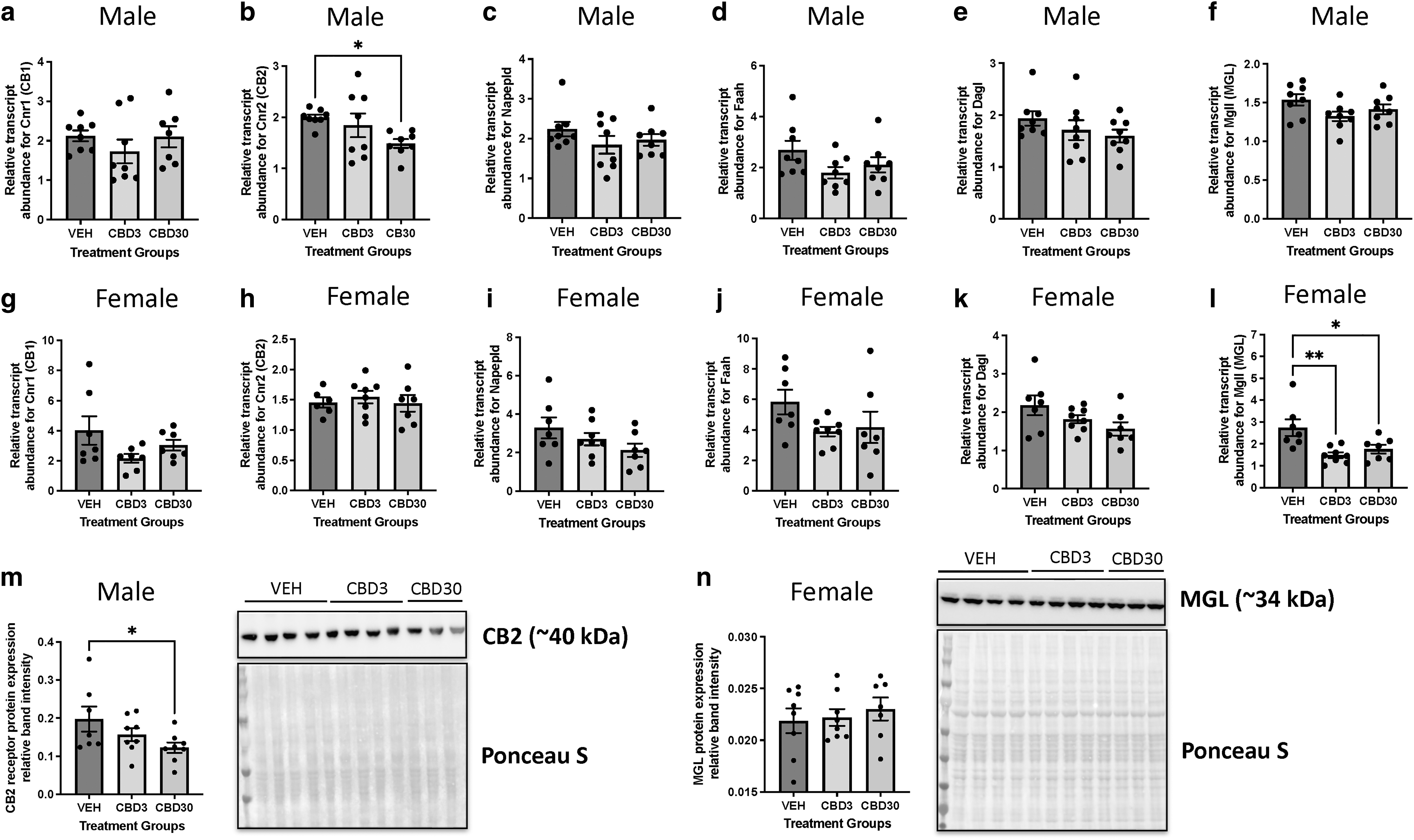
Gestational exposure to CBD leads to sex-specific alterations in cardiac endocannabinoid system targets in offspring at PND21. RT-qPCR measurements for steady-state mRNA levels of
Although CBD-exposed females did not demonstrate alterations in the expression of cannabinoid receptors, females exposed to both doses of CBD exhibited significant (p<0.01 for CBD3; p<0.05 for CBD30) reductions in cardiac Mgll (an enzyme that degrades primarily 2-AG) transcript abundance (Fig. 2l). However, immunoblotting revealed that there were no significant changes in the protein expression in MGL in female hearts exposed to CBD (Fig. 2n).
Alteration in the ECS mRNA expression was observed as early as PND1 in male offspring hearts exposed to CBD in gestation
Given that male PND21 hearts demonstrated decreases in CB2 expression, despite the cessation of CBD exposure at birth, we next sought to determine whether these changes occurred earlier in development. Interestingly, there was a significant (p<0.05) reduction in cardiac CB2 receptor transcript abundance in the CBD30-treated group at birth (PND1) (Fig. 3b), similar to PND21 offspring. In addition, there was a significant (p<0.05) transcript reduction in endocannabinoid synthesizing enzyme, Daglα (an enzyme that synthesizes 2-AG), in groups exposed to both doses (Fig. 3e), which may have effects on 2-AG synthesis.
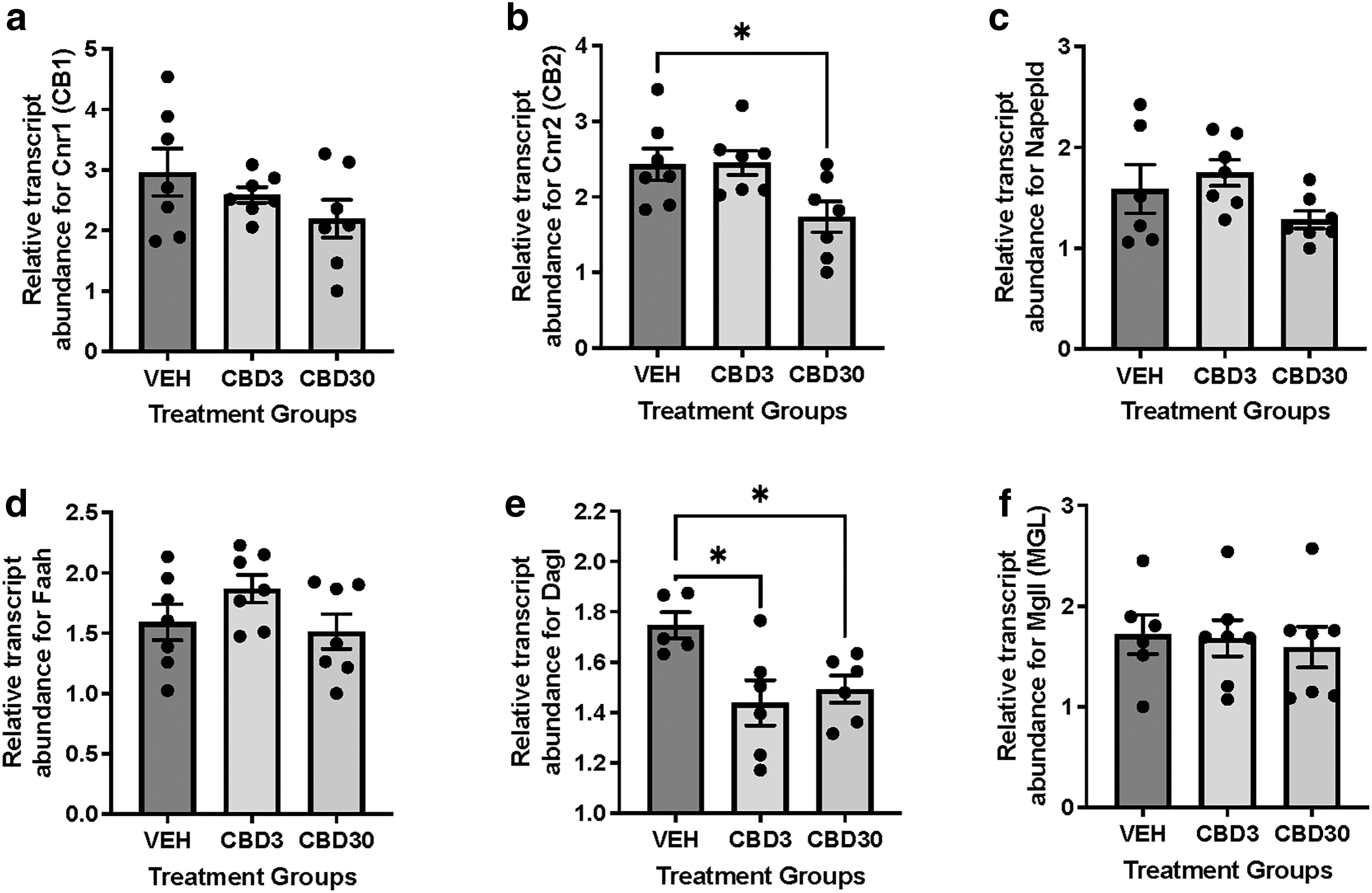
Gestational exposure to CBD decreases transcript levels of cannabinoid receptor 2 and diacylglycerol lipase in male offspring hearts at birth. RT-qPCR measurements for steady-state mRNA levels of
Transcriptomic changes in PND21 male offspring hearts exposed to 3 and 30 mg/kg CBD during gestation
Given that an altered ECS (e.g., decreased CB2) can impact cellular and molecular pathways underlying cardiac function, we next sought to examine the postnatal transcriptome-wide changes using bulk RNA-seq in exposed male offspring hearts to further delineate the underlying mechanisms involved. Since changes in heart function and ECS were primarily observed in males, we focused on male hearts for bulk RNA-seq. Applying a p-value cutoff (p<0.05) and a fold change cutoff of two-fold, we identified 6 upregulated and 39 downregulated differentially expressed genes (DEGs) in offspring hearts exposed to 3 mg/kg CBD, as well as 7 upregulated and 73 downregulated DEGs in offspring hearts exposed to 30 mg/kg CBD (Supplementary Fig. S1b, c for volcano plots and Supplementary Tables S2 and S3 for gene lists). When an FDR cutoff (p<0.05) was applied to the DEG analysis, no DEG was identified.
This may be due to the variability we observed between treatment groups (Supplementary Fig. S1a) attributed to the heterogeneity of heart tissue. Given this, we performed GSEA (which accounts for all genes irrespective of meeting our cutoffs during our DEG analysis) using GO terms with an FDR cutoff of 0.01, to assess transcriptome wide pathways. We identified 54 downregulated and 30 upregulated GO terms for the CBD3 group as well as 42 downregulated and 34 upregulated GO terms for the CBD30 group (Supplementary Tables S4 and S5 for GSEA list). Interestingly, offspring hearts exposed to both doses of CBD demonstrated similar upregulated pathways related to mitochondria, its organization, and metabolic processes (i.e., “mitochondrial ATP synthesis coupled electron transport,” “mitochondrial membrane organization,” and “oxidative phosphorylation”) (Fig. 4a, c).
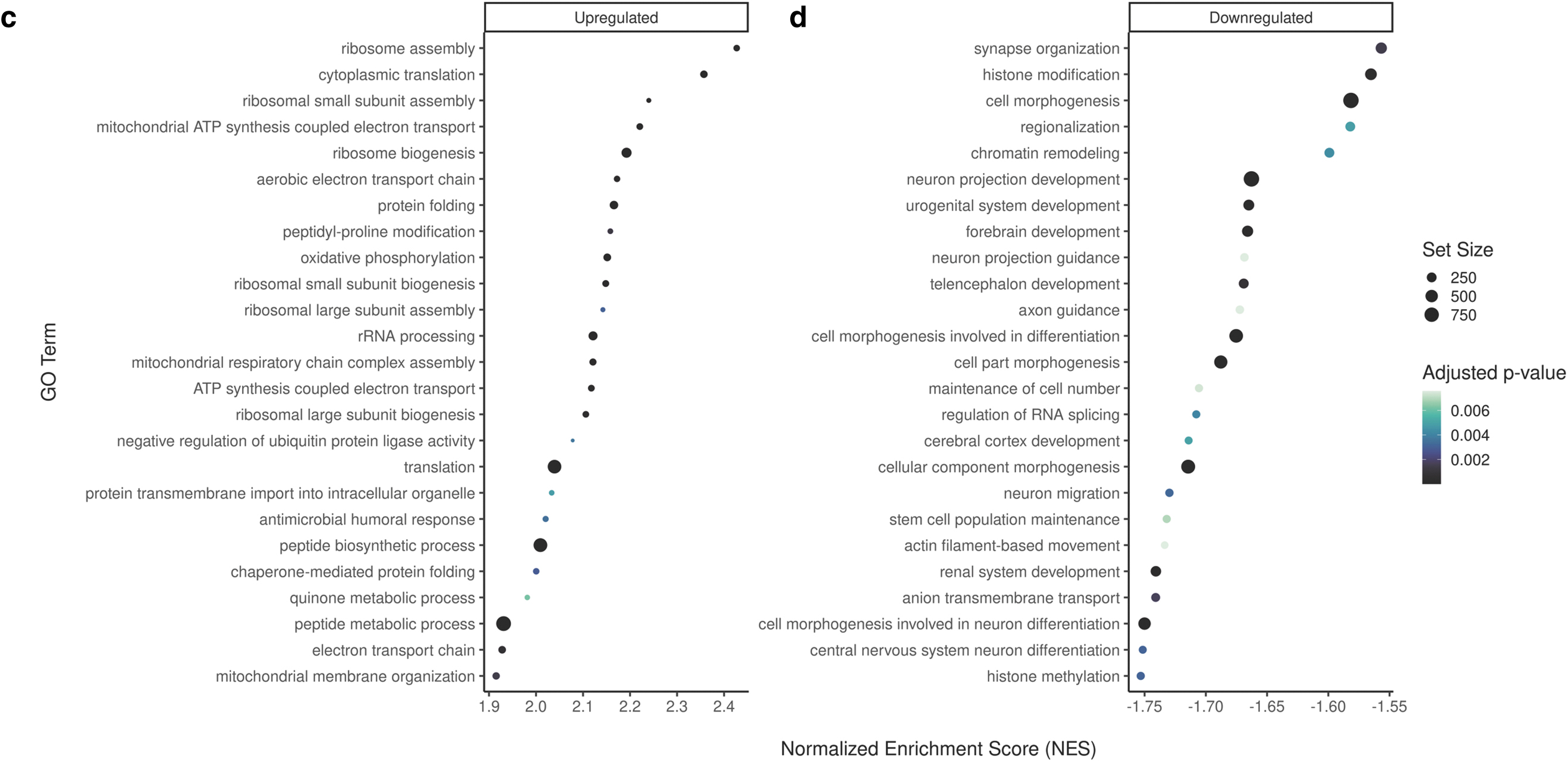
Bubble plot of the top GO terms from gene-set enrichment analysis in male offspring hearts exposed to 3 or 30 mg/kg CBD during gestation. The top 25 upregulated GO terms in offspring exposed to
For the downregulated pathways, offspring hearts exposed to both doses of CBD also exhibited similar changes in various developmental processes (i.e., “cell morphogenesis involved in differentiation,” “developmental growth,” and “animal organ morphogenesis”) (Fig. 4d and Supplementary Tables S4 and S5). Notably, both doses led to multiple downregulated pathways related to neuron development (Fig. 4b, d). The CBD 30 mg/kg-exposed male offspring specifically demonstrated downregulation in “heart development” (Supplementary Table S5).
Discussion
Despite the increasing prevalence of cannabis use in pregnancy 1 and rising popularity of CBD, the impact of CBD during pregnancy on fetal development and postnatal cardiac outcomes remains unknown. In this present study, we provide novel evidence that gestational exposure to CBD in rats leads to adverse cardiac outcomes in 3-week-old male but not female offspring.
It has been previously reported in both pre-clinical and clinical studies that either cannabis or THC alone leads to deficits in fetal growth.7,8,11,12,57 We and others have previously established that maternal exposure to THC in rats does not alter maternal outcome measurements (i.e., food intake, weight gain, and gestation length) but leads to low birthweight as well as a significantly reduced heart-to-body weight ratio at birth. 11 In vivo studies also demonstrated that maternal THC exposure leads to decreased cardiac function and cardiac remodeling in offspring.15,16 Such effects on the heart are thought to be, at least in part, due to fetal growth restriction 15 given that low birthweight increases the risk of cardiovascular disease later in life.9,58–60 Similar to THC, gestational CBD exposure does not alter maternal outcomes or litter size, which eliminates confounders such as altered maternal nutrition and fetal demise.
However, our current study differs from THC-exposed offspring, given that CBD-exposed animals did not exhibit changes in neonatal weight measurements (i.e., body weight and heart-to-body weight ratio) at birth. By PND21, males but not female CBD-exposed offspring demonstrated significant decreases in heart function with the higher dose demonstrating a reduction in additional parameters for contractile function (i.e., ejection fraction). The observed comprised heart function was not associated with mortality. We and others have similarly reported impairment of heart function in THC-exposed offspring at PND21.15,16 These studies demonstrated a thicker left ventricular wall in THC-exposed hearts, which is not apparent in the CBD offspring. Despite this, these current results pose concerns that prenatal exposure to CBD leads to reduced systolic function at such an early age in life.
We anticipate that our observed cardiac deficits may progress with age similar to other models of perinatal insults (i.e., drug exposure, hypoxia, and maternal diet).61–63 Furthermore, the reduced cardiac function could also suggest that these animals may have heightened susceptibility to future stressors (e.g., high-fat diet and ischemia/reperfusion).
Prior studies have associated a dysregulated ECS with various cardiovascular pathologies.32,35,36,64 Given we postulated that prenatal CBD could impact the ECS, we examined whether there were any changes in the ECS associated with the observed adverse cardiac outcomes in males. Females exhibited a decrease in transcript abundance in Mgl, however, no changes were observed after protein analysis. Interestingly, specifically in males, we found a reduction in both the mRNA and protein levels of CB2 in PND21 offspring hearts. This prompted us to retroactively examine the hearts at birth, which revealed that there were indeed similar decreases in mRNA levels of CB2. Daglα transcript abundance was also decreased; however, it is unknown if this impacts 2-AG synthesis. Collectively, these data suggest that the changes in the ECS, in particular CB2, likely began in utero, supporting the notion that prenatal CBD may be able to directly “rewire” the ECS during development. 40
As previously mentioned, CB2 plays a cardioprotective role in the pathophysiology of cardiovascular disease. In fact, rodent studies with CB2-deficient mice demonstrated cardiac maladaptation and a decrease in left ventricular function upon insult (repetitive ischemia/reperfusion and pressure overload).65,66 Moreover, these animals had an altered regulation of contractile proteins and a decreased antioxidative capacity (weaker upregulation of antioxidative enzymes). 65 Given this, we postulate that the downregulation of CB2 observed in our CBD30-exposed male hearts may underpin, at least in part, the more pronounced cardiac impairments relative to the control group. Moreover, since it has been shown that CB2 deficiency leads to exacerbated cardiac dysfunction in adults, this further supports the idea that the hearts of these animals may be more susceptible to a maladaptive response to future stressors and advancing age.
Based on the observed cardiac and endocannabinoid outcomes, we further examined transcriptome-wide changes in these CBD-exposed hearts. Specifically, we wanted to define alterations in various molecular processes associated with the functional cardiac deficits in these offspring. The most notable findings from our RNA-seq data were the upregulation of pathways involving mitochondrial metabolism (ETC, ATP synthesis, OXPHOS), which was consistent between males exposed to both doses of CBD in utero. Admittedly, the directionality we observed in these pathways was unexpected given that various models of different cardiovascular diseases often report decreases rather than increases in metabolic pathways involving oxidative phosphorylation and ATP synthesis. For example, in heart failure, there is typically a decrease in oxidative capacity, which leads to energy deficits (as reviewed in Lopaschuk et al. 67 ).
However, there are a few considerations worth noting. First, as the offspring are still young, these upregulated transcriptomic pathways could be early compensatory/adaptive mechanisms due to changes in energy demands in the etiology of a compromised heart.67,68 Second, increases in mitochondrial respiration and ATP production are directly associated with reactive oxygen species production due to increased activity in the ETC, 68 which may negatively impact cardiac function. If there are indeed increases in reactive oxygen species, more studies will be required to determine whether cells are equipped with the proportionate antioxidative capacity (i.e., SOD1 and SOD2). Third, it is unknown whether upregulation of mitochondrial pathways is related to an abundance of mitochondrial mRNA due to increased mitochondrial fission. Previous studies with placental cells demonstrated that THC increases mitochondrial fission markers but also found more lower quality mitochondria. 69 This may be of great interest considering that abnormal mitochondrial fission contributes to disease development and ultimately heart failure.70,71
With respect to downregulated GO terms, both doses of CBD in gestation led to a few pathways involved in development (i.e., “animal organ morphogenesis”). The higher dose-exposed offspring hearts also had a decrease in the “heart development” pathway. These transcriptome changes could suggest that prenatal exposure to CBD adversely impacts normal developmental trajectories. It is tempting to speculate that this, in part, culminates into the cardiac deficits observed. Furthermore, it is noteworthy that frequent hits on GO terms involving axon and dendrite growth and development (i.e., “cell morphogenesis involved in neuron differentiation,” “neuron migration,” “neuron projection morphogenesis”) were also observed. Although the effects of prenatal CBD on cardiac neural tissue are unknown, one study demonstrated that offspring exposed to maternal CBD (20 mg/kg/day) led to similar changes in GO terms from differential methylated loci in brain tissue. 23 One possibility for this observation in our current study may relate to the cardiac autonomic nervous system, which can influence atrioventricular node conduction as well as contractility, ultimately effecting hemodynamics (i.e., stroke volume, cardiac output, and heart rate), 72 however, more studies are needed to explore this.
Our current study investigates the effects of CBD exposure during gestation and therefore may be limited in its scope with respect to other exposure windows such as prepregnancy and lactation. Although future studies should examine earlier windows of exposure to CBD, it is important to consider that exposure to other phytocannabinoids (e.g., THC) before E5.5 does adversely impact implantation in the rodents. 46 In addition, we acknowledge that i.p. injection is not a route of administration of cannabinoids in humans. The focus of the experimental design was to achieve a serum concentration similar to that of human users. Moreover, we have previously demonstrated that this prenatal cannabinoid model42,43 reciprocates the metabolic outcomes observed in clinical studies. 8
Another limitation of the study is that bulk RNA-seq is limited in its ability to account for different cell populations and may lead to variability between samples, which could hinder the detection of DEGs. Lastly, in our RNA-seq analysis, we only examined male hearts due to the observed changes in heart function and cardiac ECS specifically in males. As such, we were unable to tease out the potential transcriptome changes in females that may underlie the sex-specific effects.
Our study has a few future directions. First, future studies could examine whether a secondary insult (e.g., high-fat diet or ischemia/reperfusion) in adulthood leads to maladaptation and exacerbates cardiac deficits in exposed offspring. Second, while the current study does not elucidate why CBD-exposed females do not exhibit changes in cardiac function, future studies are warranted to address whether changes in sex hormone signaling (i.e., receptor mediated) or epigenetic mechanisms (i.e., imprinting) are involved. Long-term studies are warranted to address whether these cardiac deficits remain sex-specific after reproductive senescence in females. Third, further studies could address how different routes of cannabis exposure impact development and postnatal cardiac outcomes.
In summary, the current study has demonstrated that gestational exposure to 3 and 30 mg/kg CBD leads to cardiac deficits exclusively in 3-week-old male offspring. Furthermore, this may be attributed to early but persistent changes in the ECS during perinatal life resulting in decreased cardioprotection, and/or alterations in transcriptome pathways involved in mitochondrial function/metabolism. Considering that these offspring are both young and exhibit compromised cardiac function, there is great impetus to examine these animals later in life. Ultimately, these observed effects of maternal exposure to CBD, a constituent perceived and marketed as safe, may pose concern regarding the cardiovascular outcomes in offspring.
Data Availability
The raw and processed RNA-seq data used for the current study are accessible through NCBI Gene Expression Omnibus accession series GSE242079.
Footnotes
Acknowledgments
We thank Drs. Qingping Feng and Sharon Lu for their assistance with the Vevo2100 Ultrasound Imaging System.
Authors' Contributions
K.L., D.R.C.N., and D.B.H. were involved in the design, execution, and interpretation of experiments. K.L., M.N., and M.H.S. were involved with implementing the animal model. S.V. performed the bulk RNA-seq analysis and created the corresponding figures. S.R.L. contributed to the design of the animal experiments and in the preparation and dosing of vehicle and CBD in vivo. K.L. wrote the first draft of the article; all authors edited drafts of the article.
Author Disclosure Statement
The authors declare no competing interests.
Funding Information
This work was supported by the Canadian Institutes of Health Project Grant (R4228A28) to D.B.H. and D.R.C.N. and a Canadian Heart and Stroke Foundation Grant-in-Aid (G-19-0026343) to D.B.H. K.L. was the recipient of the Ontario Graduate Scholarship and Canada Graduate Scholarship (Doctoral) from the Canadian Institutes of Health Research.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
