Abstract
Background:
The endocannabinoid system (ECS) plays a key physiological role in bladder function and it has been suggested as a potential target for relieving lower urinary tract symptoms (LUTSs). Whereas most studies indicate that activating the ECS has some beneficial effects on the bladder, some studies imply the opposite. In this study, we investigated the therapeutic potential of peripheral cannabinoid-1 receptor (CB1R) blockade in a mouse model for LUTSs.
Materials and Methods:
To this end, we used the cyclophosphamide (CYP; 300 mg/kg, intraperitoneal)-induced cystitis model of bladder dysfunction, in which 12-week-old, female C57BL/6 mice were treated with the peripherally restricted CB1R antagonist, JD5037 (3 mg/kg), or vehicle for three consecutive days. Bladder dysfunction was assessed using the noninvasive voiding spot assay (VSA) as well as the bladder-to-body weight (BW) ratio and gene and protein expression levels; ECS tone was assessed at the end of the study.
Results:
Peripheral CB1R blockade significantly ameliorated the severity of CYP-induced cystitis, manifested by reduced urination events measured in the VSA and an increased bladder-to-BW ratio. Moreover, JD5037 normalized CYP-mediated bladder ECS tone imbalance by affecting both the expression of CB1R and the endocannabinoid levels. These effects were associated with the ability of JD5037 to reduce CYP-induced inflammatory response, manifested by a reduction in levels of the proinflammatory cytokine, tumor necrosis factor alpha (TNFα), in the bladder and serum.
Conclusions:
Collectively, our results highlight the therapeutic relevance of peripheral CB1R blockade in ameliorating CYP-induced cystitis; they may further support the preclinical development and clinical use of peripherally restricted CB1R antagonism for treatment of LUTSs.
Introduction
Lower urinary tract symptoms (LUTSs) include increased frequency, urgency, nocturia, incontinence, recurrent urinary tract infections, and even renal dysfunction. 1 LUTSs affect ∼16% of the population aged over 40 years and their prevalence increases with age. 2 LUTSs have a major impact on patients' quality of life due to the tremendous suffering and withdrawal of these individuals from social and professional activities.
The etiology of LUTSs is varied and includes neurogenic conditions (e.g., spinal cord lesions, multiple sclerosis, and diabetic neuropathy), and more commonly, patients present without any defined neurological disorder. 1 In addition, hemorrhagic cystitis, manifested by dysuria and hematuria as a complication of cyclophosphamide (CYP) therapy, is also largely involved in development of LUTSs in oncological patients. 3
CYP, a chemotherapeutic drug used to treat different types of cancers, is metabolized to acrolein by the liver and accumulates in the bladder, resulting in hemorrhagic cystitis and LUTSs, especially in immunocompromised patients. 4 Unfortunately, there is no proven treatment to manage this type of condition, which can severely degrade the patient's quality of life and may possibly lead to renal failure and even death.
The endocannabinoid system (ECS) consists of the two main (most studied) endocannabinoids (eCBs): N-arachidonoylethanolamine (anandamide [AEA]) and 2-arachidonoylglycerol (2-AG), and their synthesizing and degrading enzymes, as well as their G-protein-coupled cannabinoid receptors, CB1R and CB2R. Both cannabinoid receptors have been recently discovered in human and animal bladders, urothelial cells, detrusor muscle, and nerve fibers innervating the bladder.5–8
Most studies show the beneficial effects of ECS activation in ameliorating LUTSs (reviewed in Ref. 9 ). Specifically, it was reported that intravenous (IV) injection of the AEA transport inhibitor, VDM-11, into anesthetized rats, which increases AEA concentrations, increased the micturition interval and threshold pressure. This effect was blocked by a CB1R antagonist (AM251), but not by a CB2R antagonist (AM630). 10 Others have shown that IV injection of a fatty acid amide hydrolase (FAAH) inhibitor into conscious rats also elevates AEA levels and enhances intercontraction intervals, micturition volume, bladder capacity, and threshold pressure. 11
On the other hand, elevated bladder AEA levels can also activate other off-target receptors such as the transient receptor potential cation channel subfamily V member 1 (TRPV1), which may evoke hyperreflexia and hyperalgesia and increase the micturition volume.7,12–14
In recent years, there has been a growing interest in blocking CB1R in peripheral organs for treatment of many pathologies mainly associated with metabolic syndrome (reviewed in Refs.15,16). Moreover, CB1R antagonism was suggested as a potential therapeutic intervention for acute cisplatin-induced renal dysfunction, 17 a disease associated with enhanced inflammation, oxidative/nitrosative stress, and cell death.
In this study, we show (for the first time) that peripheral CB1R antagonism ameliorates CYP-induced cystitis by reducing micturition events, restoring the bladder-to-body weight (BW) ratio, normalizing bladder ECS tone, and reducing inflammation. These results indicate the therapeutic potential of peripherally restricted CB1R antagonism against LUTSs.
Materials and Methods
Animals
The Institutional Animal Care and Use Committee of The Hebrew University (AAALAC accreditation no. 1285; approval no. MD-19-15994-3) approved the experimental protocol used. Animal studies are reported in compliance with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines. 18
CYP-induced cystitis animal model
To establish the CYP-induced cystitis mouse model, 12-week-old, female C57BL/6 mice were divided into three groups: (1) a control group, treated with vehicle (Veh; 1% Tween 80, and 4% dimethyl sulfoxide [DMSO] in saline, intraperitoneal [IP]) for three consecutive days; (2) CYP + Veh group, treated with Veh on the 1st day, CYP (300 mg/kg, IP) + Veh on the 2nd day, and Veh on the 3rd day; and (3) CYP + JD5037 group, treated with JD5037 (3 mg/kg, IP) on the 1st day, CYP + JD5037 on the 2nd day, and JD5037 on the 3rd day (see the scheme in Fig. 3A).
On the 4th day, mice were euthanized by cervical dislocation under anesthesia, the bladder was removed and weighed, and it was either snap-frozen or fixed in 4% buffered formalin. Trunk blood was collected, and serum was separated and stored at −80°C until processed for biochemical evaluation. The number of samples in each experiment was determined according to tissue availability, which was limited due to the small size of a mouse bladder.
Voiding spot assay: noninvasive assessment of bladder dysfunction
On the 3rd day, bladder dysfunction was assessed using a noninvasive voiding spot assay (VSA); each mouse was placed in a single cage with an absorbent filter paper and was allowed to move freely for 4 h; during this time, the micturition events were captured and retained as void spots on the paper.
Normal mice tend to urinate only a few times in the cage corner, whereas CYP-treated mice urinate numerous times all over the filter paper. Urine spots were illuminated with ultraviolet (UV) light using the 2UV Transilluminator (UVP, USA), and the number and area of urine spots were analyzed using ImageJ software (NIH, Bethesda, MD).
Materials
CYP (ENDOXAN) was purchased from Baxter Oncology (Germany). JD5037 was purchased from MedChemExpress (China).
Serum and bladder tumor necrosis factor alpha analyses
Serum and bladder levels of tumor necrosis factor alpha (TNFα) were measured by an ELISA kit (MHSTA50; R&D Systems) according to the manufacturer's protocol.
Histopathological analyses
Paraffin-embedded bladder sections (3 μm) from each group were stained with hematoxylin and eosin. Panoramic bladder images were captured with a Zeiss AxioCam ICc5 color camera mounted on a Zeiss Axio Scope A1 light microscope at ×5 and ×40 magnifications.
Immunohistochemistry
Bladder tissues from Veh- and CYP-injected mice (five animals per group) were fixed in 4% buffered formalin for 48 h and then embedded in paraffin. Sections were deparaffinized and hydrated. Heat-mediated antigen retrieval was performed with 10 mM citrate buffer, pH 6.0 (Thermo Scientific, IL, USA). Endogenous peroxide was inhibited by incubating with a freshly prepared 3% hydrogen peroxide (H2O2) solution in methanol (MeOH).
Unspecific antigens were blocked by incubating sections for 1 h with 2.5% horse serum (VE-S-2000; Vector Laboratories). Next, 3-μm bladder sections were stained for rabbit-anti-CB1R (ACR-001; Alomone), followed by a goat anti-rabbit horseradish peroxidase (HRP) conjugate (ab97085; Abcam). Color was developed after incubation with 3,3′-diaminobenzidine (DAB) substrate (SK-4105, ImmPACT DAB Peroxidase [HRP] Substrate; Vector Laboratories), followed by hematoxylin counterstaining and mounting (VectaMount H-5000; Vector Laboratories).
Stained sections were photographed as described above. Positive areas were quantified using ImageJ software with a minimum of four random images of the detrusor muscle or urothelium per mouse.
Western blotting
Bladder homogenates were prepared in RIPA buffer (25 mM Tris-HCl, pH 7.6; 150 mM NaCl; 1% NP-40; 1% sodium deoxycholate; and 0.1% sodium dodecyl sulfate [SDS]) using the Bullet Blender® and zirconium oxide beads (Next Advance, Inc., NY, USA). Protein concentrations were measured with a Pierce™ BCA Protein Assay Kit (Thermo Scientific). Samples were resolved by SDS-polyacrylamide gel electrophoresis (PAGE) (4–15% acrylamide, 150V) and transferred to polyvinylidene difluoride (PVDF) membranes using the Trans-Blot® Turbo™ Transfer System (Bio-Rad, CA).
Membranes were then incubated for 1 h in 5% milk (in 1× Tris-buffered saline with 0.1% Tween® 20 detergent [TBS-T]) to block unspecific binding and then incubated overnight with rabbit anti-CB1R (#301214; Immunogen) and TRPV1 (#ACC-029; Alomone) antibodies at 4°C. Anti-rabbit HRP-conjugated secondary antibody (#97085; Abcam) was used for 1 h at room temperature, followed by chemiluminescence detection using Clarity™ Western ECL Blotting Substrate (Bio-Rad).
Densitometry was quantified using Bio-Rad CFX Manager software. Quantification was normalized to the anti-β-actin antibody (#ab49900; Abcam).
Real-time PCR
Total bladder messenger RNA (mRNA) was extracted using Bio-Tri RNA lysis buffer (Bio-Lab, Israel), followed by DNase I treatment (Thermo Scientific), and reverse transcribed using the iScript cDNA kit (Bio-Rad). Real-time PCR was performed using the iTaq Universal SYBR Green Supermix (Bio-Rad) and the CFX connect system (Bio-Rad).
The primers used to detect mouse genes are listed in Table 1. Mouse genes were normalized to Ubc.
Mouse Primers Used for Real-Time PCR Analysis
Ccl2 (Mcp1), mast cell proteinase; Cnr, cannabinoid receptor; Col, collagen; Cxcl10 (Ip10), C-X-C motif chemokine ligand 10; Dagl, diacylglycerol lipase; Faah, fatty acid amide hydrolase; Fn, fibronectin; Il, interleukin; Mgll, monoacylglycerol lipase; Napepld, N-acyl phosphatidylethanolamine phospholipase D; Tgf, transforming growth factor; Tnf, tumor necrosis factor; Ubc, ubiquitin C.
Sample preparation and endocannabinoid measurements by liquid chromatography with tandem mass spectrometry
eCBs were extracted, purified, and quantified from bladder lysates. In brief, bladders were homogenized in ice-cold Tris buffer using the Bullet Blender and zirconium oxide beads (Next Advance, Inc.); protein concentration was determined by the bicinchoninic acid (BCA) assay. Samples were then supplemented with an ice-cold extraction buffer [1:1 methanol/Tris buffer + an internal standard (IS)] and chloroform/methanol (2:1), vortexed, and centrifuged.
The lower organic phase was transferred into borosilicate tubes; this step was repeated three times by adding ice-cold chloroform to the samples and transferring the lower organic phase into the same borosilicate tubes. The samples were dried and kept overnight at −80°C, then reconstituted with ice-cold chloroform and acetone, kept at −20°C for 30 min, and then centrifuged to precipitate proteins.
Next, the supernatant was dried and reconstituted in an ice-cold liquid chromatography with tandem mass spectrometry grade methanol and analyzed on an AB Sciex (Framingham, MA) QTRAP® 6500 + mass spectrometer coupled with a Shimadzu (Kyoto, Japan) ultra high-performance liquid chromatography (UHPLC) System. Liquid chromatographic separation was achieved using 5-μL injections of samples into a Kinetex 2.6-μm C18 (100×2.1 mm) column from Phenomenex (Torrance, CA). The autosampler was set at 4°C, and the column was maintained at 40°C during the entire analysis.
The gradient elution mobile phases consisted of 0.1% formic acid in water (phase A) and 0.1% formic acid in acetonitrile (phase B). eCBs were detected in a positive ion mode using electron spray ionization and the multiple reaction monitoring mode of acquisition, using d4-AEA as IS. The collision energy, declustering potential, and collision cell exit potential for the monitored transitions are presented in Table 2.
Multiple Reaction Monitoring Transitions for Endocannabinoid Measurements in Electron Spray Ionization (ESI)+ and ESI−
2-AG, 2-arachidonoylglycerol; AEA, anandamide; CE, collision energy; CXP, collision cell exit potential; DP, declustering potential.
The levels of AEA and 2-AG in the samples were measured against standard curves and normalized to the bladder lysate protein concentration.
Statistics
Values are expressed as the mean±standard error of mean (SEM). Unpaired two-tailed Student's t-test was used to determine differences between the two groups. Results of multiple groups were compared by one-way analysis of variance, followed by one-sided Tukey's test, using GraphPad Prism, v6 for Windows (San Diego, CA). Significance was set at p<0.05.
Results
CYP-induced cystitis exacerbates micturition events and bladder inflammation
To evaluate the severity of CYP-induced cystitis in our mouse model (Fig. 1A), noninvasive VSA was conducted. Significant increases in urine spots (Fig. 1B, C) and the bladder-to-BW ratio (Fig. 1D) were found in the CYP-injected mice. The cystitis was associated with enhanced bladder mRNA expression levels of the inflammatory cytokines, Cxcl10, Ccl2, Il18, Tgfb, and Tnfa (Fig. 1E–I).
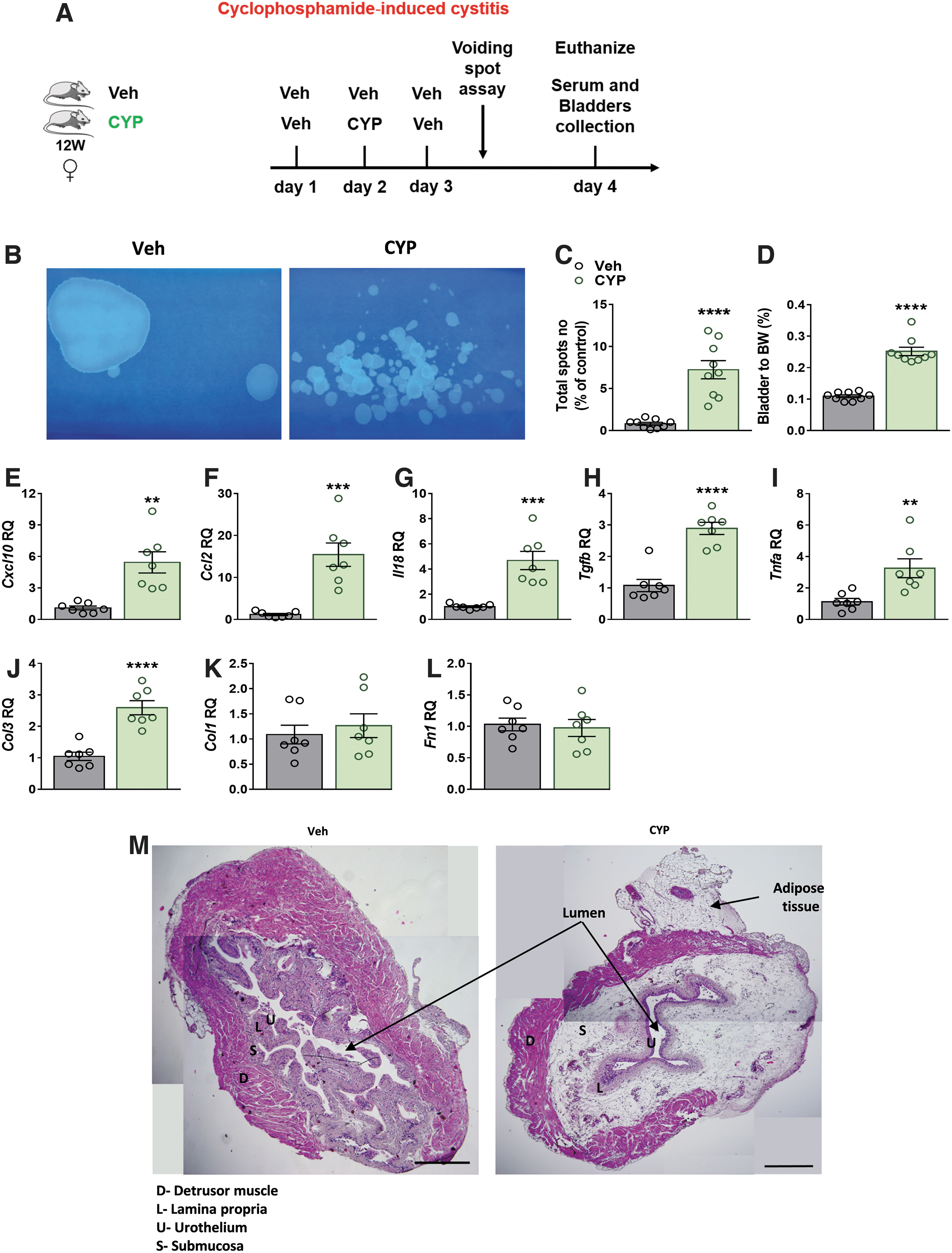
CYP-induced cystitis exacerbates micturition events and bladder inflammation. CYP-induced cystitis—experimental design
In contrast, expression levels of only the fibrotic marker Col3 (Fig. 1J), and not Col1 and Fn1 (Fig. 1K, L), were enhanced following CYP injection, as expected from an acute model. Nevertheless, histological examination of bladders revealed a typical appearance of cystitis with excessive submucosal edema and hemorrhage (Fig. 1M).
CYP-induced cystitis upregulates CB1R and TRPV1 expression
CYP-induced cystitis did not significantly change the mRNA expression levels of Cnr1 or Cnr2 (Fig. 2A, B); however, the protein expression levels of CB1R were markedly elevated (Fig. 2C, D), indicating an elevated ECS tone. CB2R protein levels were not examined due to the lack of a reliable antibody for this receptor. Since the ECS can also activate other off-target receptors, such as the TRPV1, we examined its expression and detected significant upregulation in its protein expression levels (Fig. 2E, F) in the CYP-treated mice.
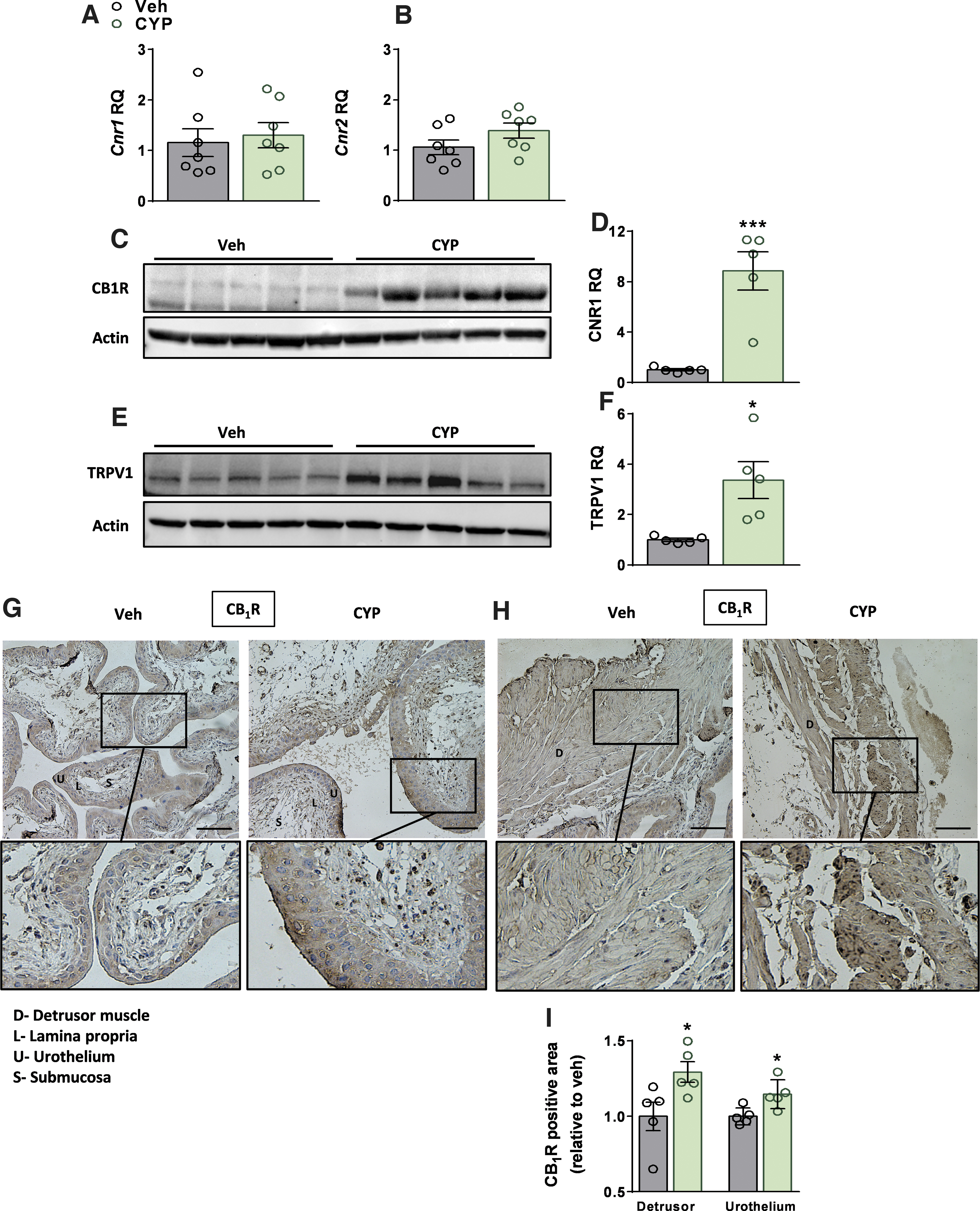
CYP-induced cystitis upregulates CB1R and TRPV1 expression. No significant changes in the bladder mRNA expression levels of Cnr1 or Cnr2 in CYP-injected mice
Elevated CB1R protein expression levels were further validated using immunohistochemistry, revealing significant elevated expression levels in urothelial cells, particularly in the umbrella cells, and the lamina propria, as well as in detrusor muscle cells (Fig. 2G–I).
Peripherally restricted CB1R antagonism attenuates CYP-induced cystitis and inflammation
Treatment with the peripherally restricted CB1R antagonist, JD5037, a day before, in combination with, and a day after CYP injection (Fig. 3A) significantly reduced the elevated urine spots (Fig. 3B, C) and the bladder-to-BW ratio (Fig. 3D) in CYP-injected mice. Histologically, JD5037 reduced the area affected by edema and hemorrhage (Fig. 3E).
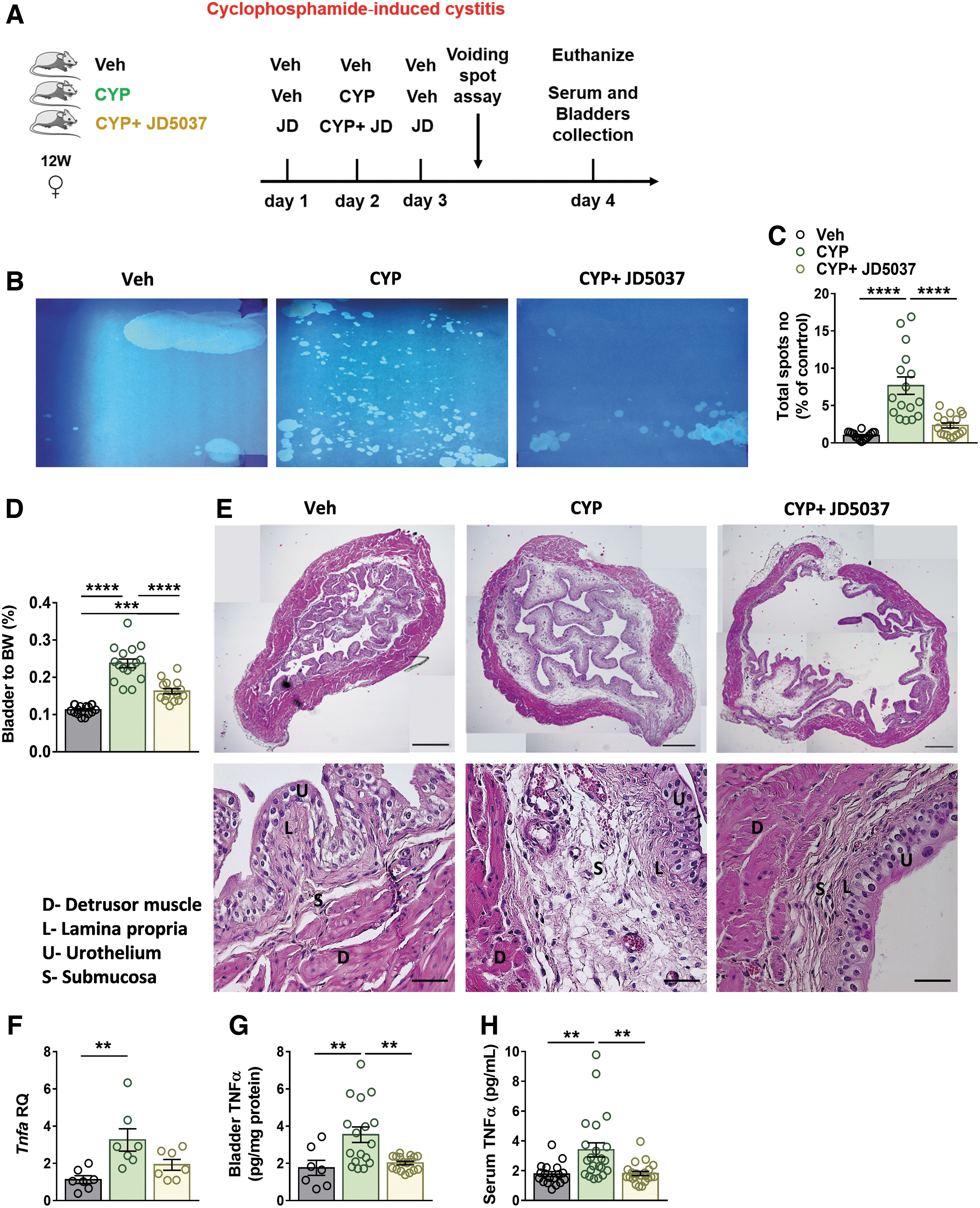
Peripherally restricted CB1R antagonism attenuates CYP-induced cystitis severity and inflammation. A CYP-induced cystitis model with CB1R antagonism—experimental design
Moreover, CB1R antagonism resulted in normalization of elevated mRNA (Fig. 3F) and protein (Fig. 3G) levels of the inflammatory cytokine, TNFα, as well as its circulating levels in the serum (Fig. 3H). Taken together, these results indicate that peripheral CB1R antagonism attenuates cystitis severity.
Peripherally restricted CB1R antagonism normalizes bladder ECS tone
To assess the involvement of the ECS in CYP-induced cystitis and its reversal by JD5037, we assessed changes in expression of the receptors and ligands in this system in our model. Interestingly, the improvements in bladder function by JD5037 were accompanied by downregulation of CB1R (Fig. 4A, B), but not the TRPV1 expression levels (Fig. 4A, C).
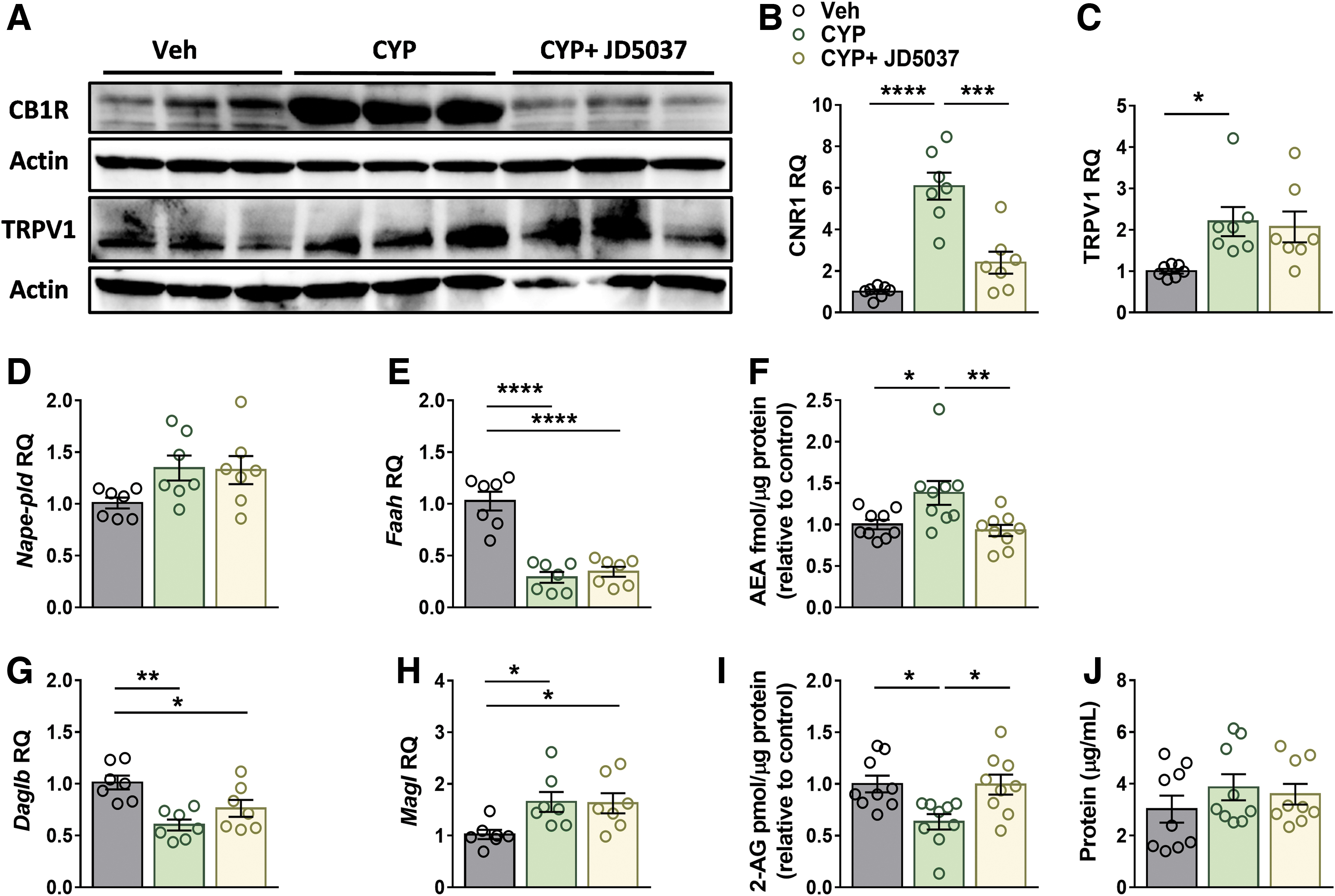
Peripherally restricted CB1R antagonism restores the ECS tone. JD5037 treatment restored bladder protein expression levels of CB1R
We next determined the mRNA levels of degrading and synthesizing enzymes as well as the amount of AEA and 2-AG in the bladder. Whereas CYP-injected mice displayed no significant changes in the expression levels of AEA's synthesizing enzyme, N-acyl phosphatidylethanolamine phospholipase D (Fig. 4D), a significant reduction in the expression of its degrading enzyme, FAAH, (Fig. 4E) was measured.
These changes were found to be in accordance with the actual amount of AEA found in the bladder (Fig. 4F). Interestingly, JD5037 treatment normalized AEA levels in the treated mice (Fig. 4F), although it did not affect the expression of its related enzymes. In contrast to AEA, decreased synthesis and enhanced catabolism of 2-AG, resulting in reduced bladder levels, were found in mice injected with CYP, as manifested by changes in expression levels of 2-AG's synthesizing enzyme, diacylglycerol lipase beta, and its degrading enzyme, monoacylglycerol lipase (Fig. 4G, H), respectively.
Yet, similar to the effect of JD5037 on AEA levels, the treatment also normalized the amount of 2-AG in the bladder (Fig. 4I) without affecting the expression of its related enzymes. Both AEA and 2-AG levels were normalized to the protein levels in each bladder (Fig. 4J), which were not affected by CYP or JD5037 treatment. These findings indicate that peripheral CB1R antagonism can restore bladder ECS tone.
Discussion
The present study reveals, for the first time, the relevance of CB1R antagonism in ameliorating hemorrhagic cystitis. We found that peripheral CB1R antagonism reduces the elevated micturition events, bladder edema, and inflammation as well as restores the bladder ECS tone. Our findings are unique since most publications in this field indicate that activation of CB1R or CB2R is an effective strategy to decrease LUTSs in animal models (reviewed in Ref. 9 ).
These studies mostly refer to the modulatory action of CB1R on the sensory neurons innervating the bladder as well as to the anti-inflammatory effect of CB2R agonism. In contrast, our findings suggest an alternative pathway by which bladder dysfunction can be mitigated through reducing the enhanced activity of the CB1R present on urothelial cells5,8 and the detrusor muscle. 8
The ECS, ubiquitously present in humans and animals, acts both centrally and peripherally to maintain cellular and organ homeostasis. Thus, changes in ECS tone, evidenced by modulation in expression of the cannabinoid receptors, their functional activity (upregulated or downregulated), and the relative number of eCBs, may render the subject susceptible to different diseases. 19
The analgesic effects of CB1R agonists are well established in many chronic disorders. 20 Upregulation of CB1R during cystitis revealed here could be one of the compensatory mechanisms to ameliorate pain and inflammation in acute hemorrhagic cystitis in mice. Indeed, bladder cystitis is characterized by an imbalanced ECS tone, as manifested here by enhanced bladder expression of CB1R and altered AEA and 2-AG levels, as well as changes in their corresponding synthesizing and degrading enzymes.
These findings are in accordance with those reporting elevated AEA levels and bladder inflammation in rats, but not in mouse models for cystitis.14,21,22 However, in contrast to others, they showed no changes in 2-AG levels in a CYP-induced mouse model for cystitis. 22 These discrepancies may be related to differences in the model established, the number of mice used in each experiment, the eCB extraction method, and the normalization method used for calculating eCB levels.
Regarding eCB levels, we used normalization to the bladder's protein content rather than the bladder weight since its increased weight could arise from edema and water absorption, resulting in higher chances for errors in calculating the eCB levels. Nevertheless and surprisingly, we found that peripheral CB1R blockade results in a modulatory effect on the ECS, and normalizes its action under these conditions, both regarding the receptor expression levels and the amount of ligands produced in the bladder.
Although CB1R blockade normalized AEA and 2-AG levels, the mRNA expression levels of their synthesizing and degrading enzymes were not significantly changed; these contradicting results may be explained by changes in their protein expression levels or their activities. Future experiments would need to assess these assumptions experimentally.
In attempting to determine the specific effects of AEA and 2-AG on bladder function, we suggest that the two ligands may play opposing roles. Whereas some studies imply that AEA increases the micturition interval and threshold pressure10,11 and may decrease pain behavior and bladder hypersensitivity,21,22 others have shown that elevated bladder AEA levels may activate pronociceptive TRPV1 channels7,14,23,24 and induce pain.
Our results support the latter observations since we found that CYP elevates AEA levels and TRPV1 expression. In fact, normalization of AEA levels by JD5037 therefore has great importance in ameliorating pain sensation and further suggests that peripheral CB1R blockade may have an indirect analgesic effect in this model; however, this hypothesis should be further tested experimentally with behavioral and pain tests.
Regarding the role of 2-AG in the bladder and its opposite pattern to AEA, reduced inflammation in CB1R antagonist-treated mice may be due to the recovery of 2-AG (preferential a CB2R ligand) levels, and it may support the anti-inflammatory role of CB2R that was well established in cystitis.25,26 Since we have not measured the protein expression of CB2R (due to lack of a valid antibody), we cannot comment further on its role in our settings.
Peripheral CB1R overactivation is strongly related to metabolic inflammation 27 and its peripheral antagonism has great therapeutic potential in reducing it. Specifically, CB1R activation has been associated with increased TNFα secretion in human bladder carcinoma cells, 28 whereas CB1R antagonism was reported to inhibit TNFα production in the inflamed small intestine in rats. 29
Moreover, CB1R genetic deletion reduces TNFα secretion by Kupffer cells, 30 and pharmacological CB1R antagonism or its genetic deletion inhibits TNFα expression in diabetic cardiomyopathy. 31 Moreover, we have previously shown that peripheral CB1R antagonism or its genetic deletion from renal proximal tubule cells normalizes the elevated TNFα levels in both type 1 diabetes and diet-induced obesity mouse models.32,33 In agreement with all of these studies, we show here that peripheral CB1R antagonism also normalizes bladder TNFα levels as well as systemic inflammation.
Although CYP-induced cystitis is not defined as a chronic metabolic disease, there is ample evidence that acute pathologies are also accompanied by an imbalanced ECS tone and enhanced CB1R activity.17,34 In fact, this upregulation may be manifested as a compensatory effect by the damaged organ to restore its function. However, this imbalance may promote unwanted effects such as a destructive inflammatory response.
In conclusion, hemorrhagic cystitis is characterized by bladder dysfunction, inflammation, and altered ECS tone. Peripheral CB1R blockade has the potential to ameliorate these characteristics and to restore normal bladder function. These findings support the rationale for clinical testing of peripheral CB1R blockers that are currently being developed in preclinical settings for the treatment of CYP-induced cystitis in oncologic patients as well as other systemic conditions associated with LUTSs.
Footnotes
Authors' Contributions
L.H., R.L., S.L., and Y.H. conducted the experiments and analyzed the data. A.N. performed the LC-MS/MS analysis. O.N.G. critically reviewed the manuscript. L.H., J.T., and G.H. designed and supervised the experiments and wrote the manuscript.
Author Disclosure Statement
All authors declare that no conflicts of interest exist.
Funding Information
This work was supported by a Mentoring Grant (Hadassah University Medical Center, Jerusalem, Israel) to G.H. and J.T. as well as an Israel Science Foundation (ISF) grant (no. 158/18) to J.T.
