Abstract
Background:
Cannabinoids have been suggested to alleviate frequently experienced symptoms of reduced mental well-being such as anxiety and depression. Mental well-being is an important subdomain of health-related quality of life (HRQoL). Reducing symptoms and maintaining HRQoL are particularly important in malignant primary brain tumor patients, as treatment options are often noncurative and prognosis remains poor. These patients frequently report unprescribed cannabinoid use, presumably for symptom relieve. As studies on brain tumor patients specifically are lacking, we performed a meta-analysis of the current evidence on cannabinoid efficacy on HRQoL and mental well-being in oncological and neurological patients.
Methods:
We performed a systematic PubMed, PsychINFO, Embase, and Web of Science search according to PRISMA guidelines on August 2 and 3, 2021. We included randomized controlled trials (RCTs) that assessed the effects of tetrahydrocannabinol (THC) or cannabidiol (CBD) on general HRQoL and mental well-being. Pooled effect sizes were calculated using Hedges g. Risk of bias of included studies was assessed using Cochrane's Risk of Bias tool.
Results:
We included 17 studies: 4 in oncology and 13 in central nervous system (CNS) disease. Meta-analysis showed no effect of cannabinoids on general HRQoL (g=−0.02 confidence interval [95% CI −0.11 to 0.06]; p=0.57) or mental well-being (g=−0.02 [95% CI −0.16 to 0.13]; p=0.81).
Conclusions:
RCTs in patients with cancer or CNS disease showed no effect of cannabinoids on HRQoL or mental well-being. However, studies were clinically heterogeneous and since many glioma patients currently frequently use cannabinoids, future studies are necessary to evaluate its value in this specific population.
Introduction
Cannabinoids have been reported to reduce anxiety and depressive symptoms in various populations. 1 Anxiety and depression are symptoms that patients with a primary brain tumor often experience. Malignant brain tumors limit life expectancy and health-related quality of life (HRQoL). They are mostly incurable; therefore, a main focus of treatment is to preserve HRQoL. HRQoL is a patient-reported outcome (PRO) reflecting the well-being of a patient and the extent of disease interference in daily activities.2–4 It consists of multiple dimensions, of which an important one is mental well-being. Mental well-being can be referred to as both emotional and psychological functioning and the absence of mental health complaints such as depression and anxiety.5,6
After their diagnosis, malignant primary brain tumor patients report that they value quality of life over a prolonged survival. 7 Nonetheless, quality of life, and specifically mental well-being, is often compromised by frequently experienced symptoms such as anxiety (>50%) or depression (>40%).8,9 A small survey study in Florida showed that a third of these patients use cannabinoids to relieve their symptoms, a finding corroborated by an informal assessment at our hospital. 10 Consequently, many brain tumor patients have questions about the possible benefits of cannabinoids. Unfortunately, clinicians cannot address these questions or prescribe cannabinoids without evidence. Therefore, a complete overview of the possible beneficial effects of cannabinoids on HRQoL in brain tumor patients is needed.
The cannabis plant contains many cannabinoids, Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD) being the most prevalent. 11 THC has psychoactive effects, whereas CBD is associated with anxiolytic and anticonvulsant properties 12 Both THC and CBD interact with the cannabinoid CB1 and CB2 receptors. 13 CBD is not only an antagonist of the CB1 receptor but also inhibits endocannabinoid degradation, thereby increasing its availability to the CB1 receptor. 12 In vitro and in vivo studies have shown that CBD also interacts with the serotonin-1A receptor, a key target involved in treatment of anxiety and depression.12,14
Moreover, cannabinoids could reduce pain. 15 For medical use, both plant-derived and synthetic forms of cannabinoids are available, containing various CBD:THC ratios. Availability and legal permissions for use are rising, but differ per country, with medical marijuana currently permitted in >30 countries for a variety of indications, ranging from refractory epilepsy to spasticity. 16 In the Netherlands, CBD is freely available, whereas THC use is permitted in small amounts. Medical cannabinoids can be prescribed for specified indications, although this is not common practice yet.
Only two studies have investigated the effects of cannabinoids on HRQoL in brain tumor patients: an open-label study investigated effect of THC on chemotherapy-induced nausea and vomiting in 32 primary brain tumor patients. HRQoL was a secondary outcome measure and did not improve. 17 Another study investigated the tolerability of two different ratios of CBD:THC (1:1 and 1:4) and its effects on HRQoL in 88 patients with high-grade gliomas (primary malignant brain tumors). They found that the functional and physical domain improved more in the 1:1 group than with the CBD:THC 1:4 ratio. Total HRQoL did not differ between different CBD:THC ratios. 18 In addition, a survey study among glioma patients showed that a third of patients used cannabinoids, and symptom relief was reported in 83%. 10 However, these study designs are unsuitable for drawing any firm conclusion on efficacy.
Randomized controlled trials (RCTs) into the effects of cannabinoids on HRQoL in brain tumor patients are lacking. As a derivate, we performed a meta-analysis in two populations that share characteristics with brain tumor patients: patients with chronic central nervous system (CNS) disease and oncological patients. We assessed both general HRQoL and its subdomain mental well-being as reduced mental well-being could greatly affect HRQoL. 19 Moreover, pre-clinical and clinical studies show cannabinoids could possibly improve symptoms associated with reduced mental well-being.1,14
Methods
This meta-analysis follows a pre-specified, but unregistered protocol and was performed according to the PRISMA guidelines. 20
Search strategy
The search was carried out on August 2 and 3, 2021 using PubMed, Embase.com, PsychINFO, and Clarivate Analytics/Web of Science Core Collection as sources. Search terms were designed by an experienced librarian and included “cannabinoids,” “cancer,” or “CNS diseases” such as “multiple sclerosis (MS).” Broad search terms were included for mental well-being, focusing on both positive and negative psychological and emotional symptoms and items. See Supplementary Appendix SA1 for the complete search strategy. There were no restrictions applied on publication date or language.
Study inclusions
Studies were eligible for inclusion if patients had any oncological disease or any chronic CNS disease (such as MS or Parkinson's disease [PD]), or a history of an acute event such as stroke or traumatic brain injury with symptoms lasting >3 months. Patients had to be 18 years of age or older. Only prospective RCTs were considered. Both parallel and crossover study designs were allowed. For crossover trials, washout periods had to be at least 1 week. Treatment consisted of cannabinoids in any form (synthetic or plant based), route of administration or dose, given for at least a week to establish a steady-state concentration of active substances. The active component could be THC, CBD, or a combination of both in any composition.
The control group could consist of a placebo or an active control. As this was an exploratory meta-analysis of the efficacy of cannabinoids, we did not assess adverse effects.
Two reviewers (V.B. and J.R.) independently evaluated whether publications were suitable for inclusion. Inconsistencies between reviewers were discussed until consensus was achieved. Titles and abstracts of all entries were screened and articles not describing CNS disease or oncological patients were excluded, as were studies not investigating effects of cannabinoids. Full-text articles were retrieved and assessed. In case of multiple publications from the same study population, the study with the largest sample size was chosen. If sample sizes were similar, the most recent study was included.
Outcome measures and definitions
The outcome measures of this meta-analysis were HRQoL and mental well-being. We have chosen both general HRQoL and its subdomain mental well-being to avoid more general outcomes, as well as more disease-specific health complaints. HRQoL instruments could be used if they assessed HRQoL as a whole, such as the SF-36, or assessed global or general health as part of HRQoL. For mental well-being, all instruments or subscales could be included, which assessed psychological functioning, emotional functioning, mood, anxiety, depression, or mental health. The same study population could be included in both analyses if multiple questionnaires were available.
There were no restrictions on whether HRQoL was a primary or secondary outcome measure in the included studies. Only PROs were included, none completed by a proxy. Baseline and post-intervention assessments had to be available: one-time questionnaires (such as subject global impression of change) were not included. Generic as well as disease-specific HRQoL questionnaires were permitted. For mental well-being, subscales of general HRQoL such as emotional well-being were included, as were anxiety and depression questionnaires as they are inversely correlated with (HR)QoL. 21 If a study included multiple questionnaires to assess the same outcome measure, we included the generic rather than the disease-specific questionnaire. If still multiple questionnaires could be included, we selected the questionnaire with the highest validity.
Data extraction
Characteristics extracted from the studies included the following: publication year; study design (parallel vs. crossover); number of participants; diagnosis of included patients; intervention type, dose, and administration route; duration of the intervention; primary outcome measure and outcome measure of interest; instruments used; and outcome data.
Data from the start and end of each intervention period were extracted. All extracted data were verified by a second reviewer (J.R.). We contacted all corresponding authors by email to request the individual patient data of each study. If applicable, we contacted the sponsor as well. If authors and sponsors were not able or willing to share the individual patient data, we requested the mean change from baseline and its standard deviation (SD).
If we did not receive the requested data, we extracted mean changes from baseline and the corresponding SDs from articles when possible. We also searched trial registration websites for additional public data. If only post-intervention means and SDs were reported, we did not include this study in the meta-analysis as we could not establish the SDs of change. If a median was reported instead of a mean, we excluded this study unless it was explicitly stated that the data were not skewed, in which case, we calculated the mean and SD from the available information. We calculated SDs using confidence intervals (95% CIs) and group size if necessary. If we could not obtain data from the authors or the publication, we excluded the study.
Statistical analysis
Data were analyzed with Rstudio (version 4.0.2). We used the packages “dmetar,” “effsize,” “meta,” “tidyverse,” “dplyr,” and “esc.”22–27 Risk of bias was visualized with the “robvis” package. 28 In studies with multiple intervention groups, such as multiple doses or different forms of cannabinoids, data of intervention groups were pooled and new mean changes and SDs were calculated. 29 We quantified the treatment effect by Hedges' g and its accompanying standard error. 30 For crossover studies, we calculated the Hedges' g using the formula for paired data. Hedges' g corrects for small sample sizes and is calculated by dividing the differences in mean change from baseline by the pooled and weighted SD. A g<0.2 represents a small effect, 0.5 < g<0.8 a moderate effect, and g≥0.8 a large effect.29–31
We used a random-effects model to account for heterogeneity between studies due to differences in disease, intervention, and study duration. 32 We visualized the effect sizes with forest plots. Two-sided p-values <0.05 were considered significant. We tested heterogeneity of study outcomes with I2; <25% was considered negligible and >75% undeniable heterogeneity. 29 We tested for publication bias by using Egger's formula, which tests the degree of funnel plot assymetry. 33
Subgroup analysis
Subgroups were analyzed and compared using a mixed-effects model. Subgroups were defined based on the included population (CNS disease vs. oncological) and the intervention studies (CBD, THC, or combination of both). Heterogeneity between subgroups was assessed using Cochran's Q test. 34
Assessment of bias
Two reviewers (V.B. and J.R.) independently evaluated the risk of bias of the included articles, using the Cochrane Risk of Bias Tool 2.0. 35 Five domains of bias were judged: (1) arising from the randomization process; (2) due to deviations from intended interventions; (3) due to missing outcome data; (4) in measurement of the outcome; and (5) in selection of the reported result. Studies were considered low risk of bias if all domains were judged to be of low risk; if some domains raised some concerns, the study was judged to be of some concern; and when at least one domain was high risk, the study was believed to have a high risk of bias. 35 Inconsistencies between reviewers were discussed with each other until consensus was achieved. 29
Results
Search/inclusions
The literature search yielded 3825 studies; after removing duplicates, 2356 remained. Titles and abstracts were screened (Fig. 1). We excluded 2144 records that did not satisfy pre-defined criteria for study design (such as reviews and protocol articles) or intervention. We read 212 full-text articles and excluded an additional 180 articles based on study population, outcome measurement, study design (such as open-label studies), study duration (mostly single-dose administrations), intervention (no form of THC or CBD), or overlap in study population. Thirty-two records were eligible for inclusion; both assessors (V.B. and J.R.) fully agreed on these inclusions. Fifteen of these studies were excluded due to reporting and provision of data being insufficient for inclusion in the meta-analysis.
Flow chart of study inclusions.
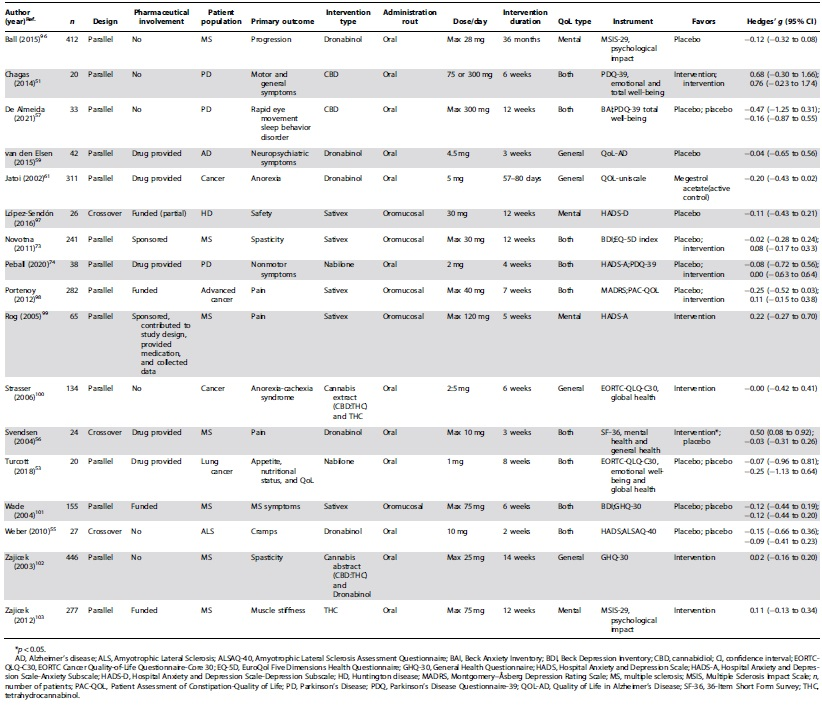
These studies were mostly on MS (47%) and cancer (40%), only one of these had HRQoL as a primary outcome.36–50 Seventeen studies were included: 9 studies could be included in both the analyses for HRQoL and mental well-being, 4 could only be included in the general HRQoL analysis, and the remaining 4 only in the analysis for mental well-being. A variety of instruments was used (Table 1).
Study Characteristics
Most studies included in the meta-analysis investigated MS (41%) or cancer (24%). General HRQoL or mental well-being was the primary end-point in two studies (12%). Pharmaceutical companies were involved in 11 (65%) of the studies: in 5 studies, the drug was provided (29%), and in an additional 6 studies (35%), the pharmaceutical company was the study sponsor.
Individual patient data
Authors from 7 publications (of the 32 eligible studies) shared their raw data upon our request.51–57 These studies concerned MS, PD (twice), Huntington's Disease, lung cancer, Alzheimer's Disease, and Amyotrophic Lateral Sclerosis. One of these studies (14%) received financial support by a pharmaceutical company, compared to 6 (out of 10; 60%) of the studies from which we did not receive individual data. Raw data of only seven studies were considered too few to perform an individual patient data meta-analysis, also because these studies differed in terms of outcome measures and study design.
Interventions
Various drugs were used as interventions (Table 1). Dronabinol was most frequently used (6/17 studies; 35%). This is a synthetic THC analog and administered as a capsule containing 2.5, 5, or 10 mg. 58 Sativex® was used in 5 out of 17 studies (29%). It combines CBD and THC in an oromucosal spray containing 2.5 mg CBD and 2.7 mg THC per spray. 59 Nabilone, a synthetic THC packed in capsules of 1 mg, was used in two of the included studies. 60 Cannabis extract retrieved from the C. sativa plant was applied as well. Two studies investigated monotherapy CBD. Control groups consisted of a placebo in all, but one study, which used megestrol acetate. 61 This is a synthetically derived progesterone that is used to increase appetite in cancer patients.
Meta-analysis
General HRQoL
There was no significant difference between intervention and control groups in general HRQoL based on 12 studies with a total of 1771 patients (g=−0.02 [95% CI −0.11 to 0.06]; p=0.57; Fig. 2). Heterogeneity was negligible (I 2 =0.0%). Egger's test did not indicate the presence of publication bias (p=0.74).
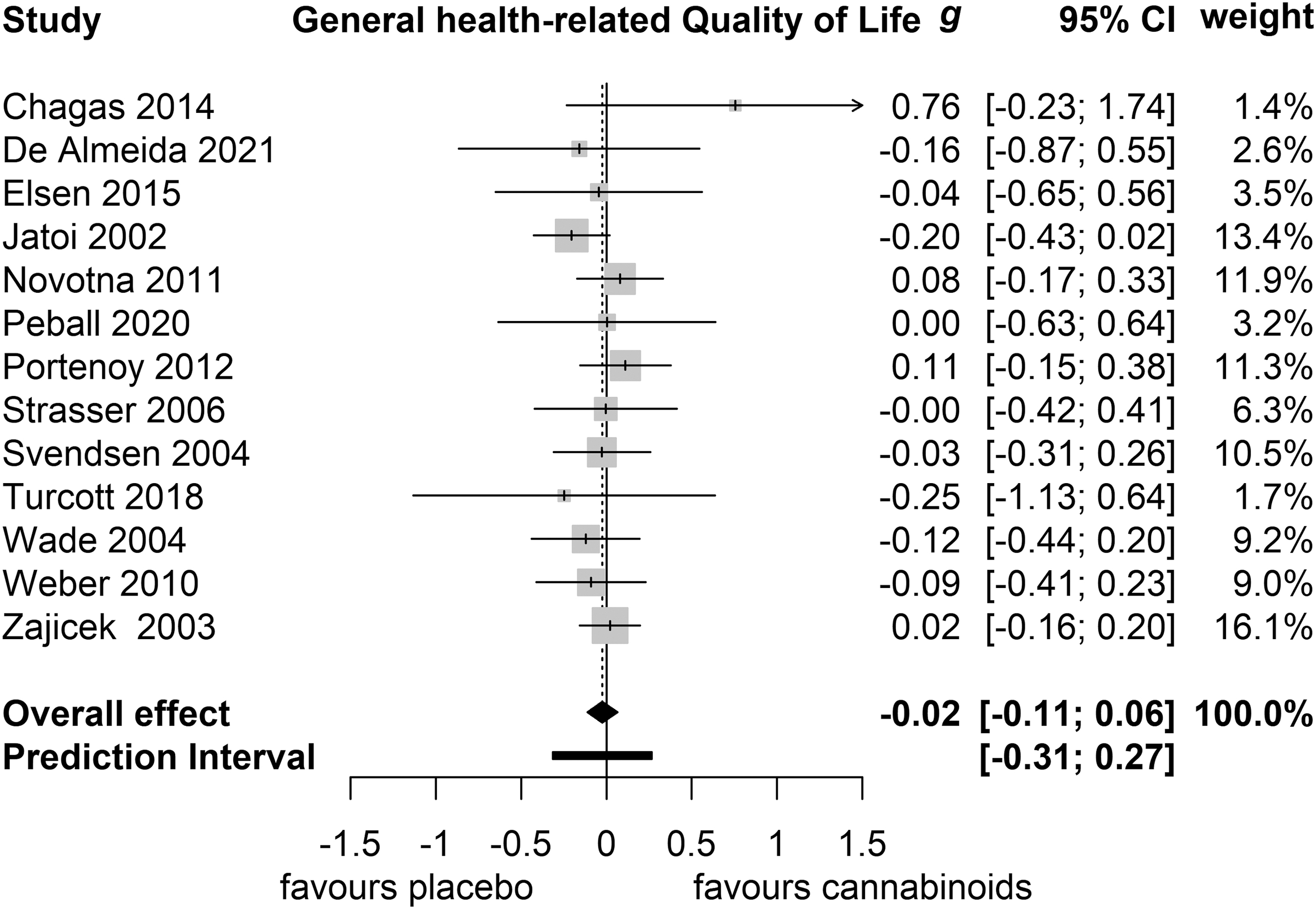
General HRQoL. g, Hedges'g. HRQoL, health-related quality of life.
Mental well-being
Cannabinoids did not significantly affect mental well-being based on 12 studies with a total of 1613 patients (g=−0.02 [95% CI −0.16 to 0.13]; p=0.81; Fig. 3). Heterogeneity was negligible (I 2 =23.7%). One small study did report a beneficiary effect of THC on mental well-being. 56 Egger's test did not indicate the presence of publication bias (p=0.20).
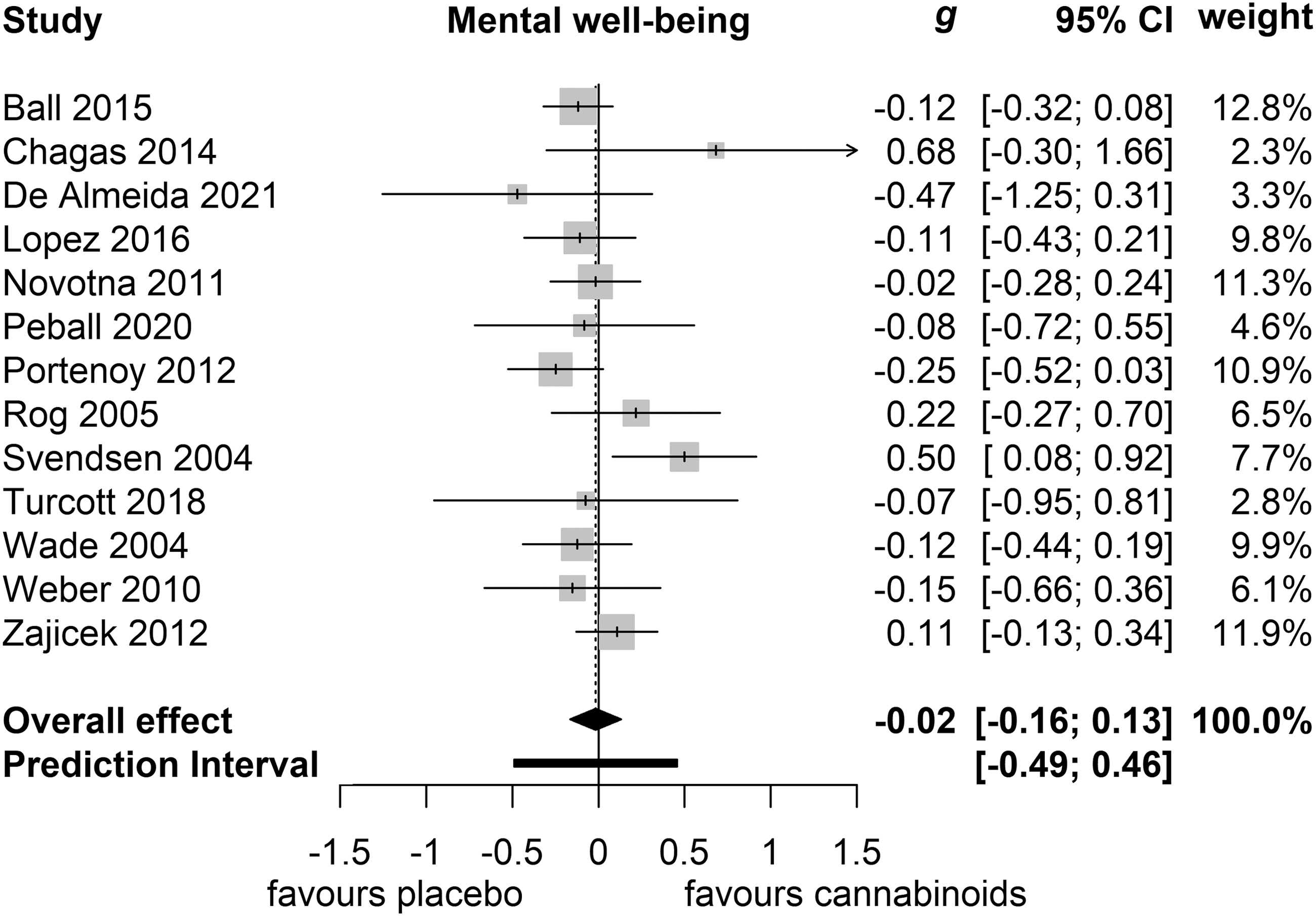
Mental well-being, g, Hedges'g.
Subgroup analyses
No significant difference in outcome was observed between cancer (n=4; 747 patients) and CNS disease (n=8; 1024 patients) populations (Q=0.32; p=0.57; Fig. 4A) in the general HRQoL analysis. In mental well-being, there were too few studies investigating cancer (n=2) to perform subgroup analyses.
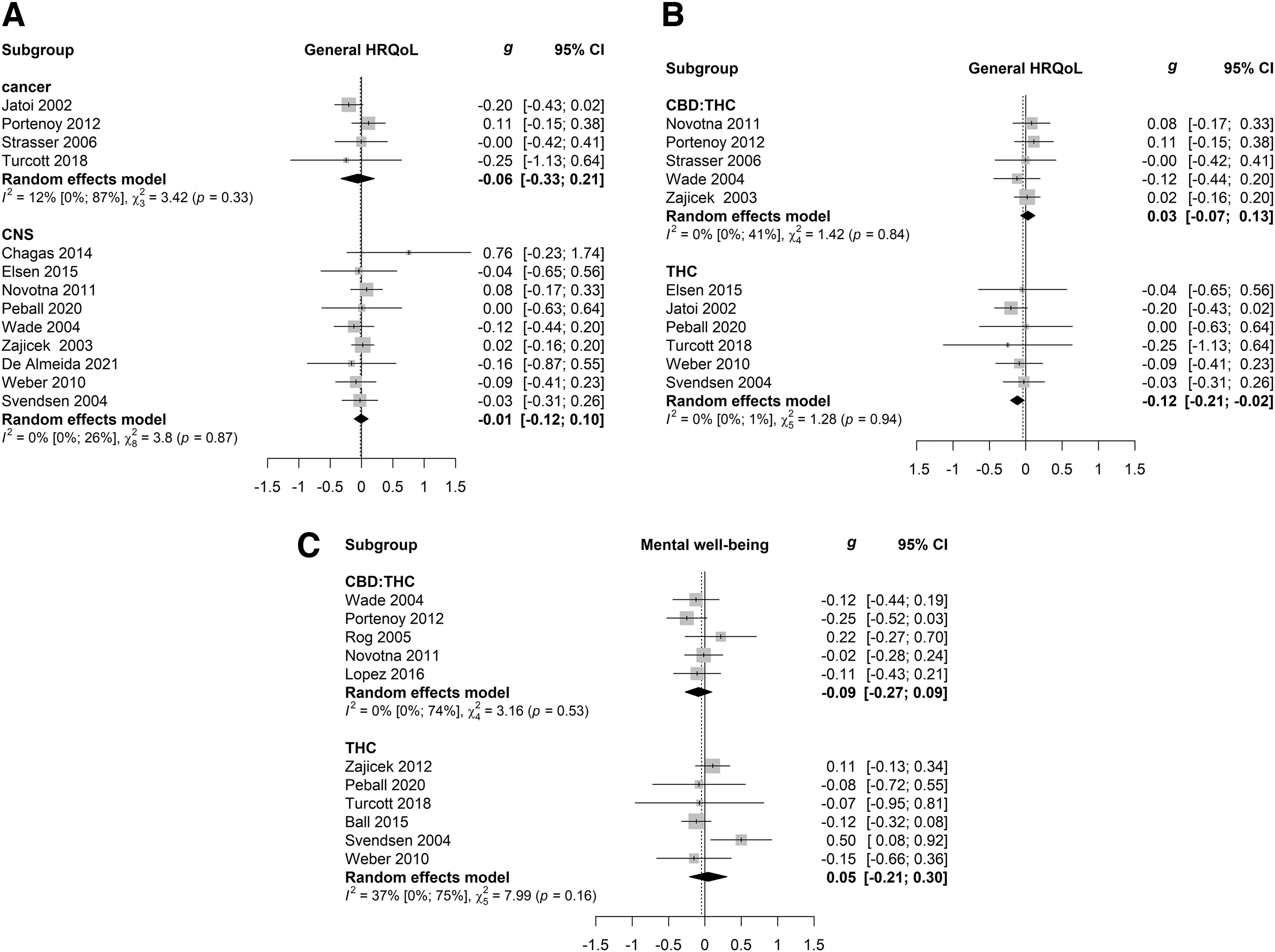
Subgroup analyses.
We also analyzed subgroups based on intervention method. There were only two small studies that investigated CBD separately, so these study were not included in the intervention subgroup analysis in both general HRQoL and mental well-being. The effect of THC combined with CBD (n=5; 1258 patients; g = 0.03) significantly differed from the effect of THC alone (n=6; 462 patients; g=−0.12; Fig. 4B; Q=7.92; p=0.0049) in general HRQoL. However, neither intervention significantly affected HRQoL. There was no significant difference between the effect of CBD:THC (n=5; 769 patients) and THC (n=6; 798 patients) on mental well-being (Q=1.25; p=0.26; Fig. 4C).
Risk of bias
Differences between reviewers were few and mostly arose from one of the reviewers overlooking information such as differences in baseline characteristics. All inconsistencies were easily resolved by mutual agreement. Five studies had a low risk of bias, 2 studies raised some concerns, and 10 studies had a high risk of bias (Fig. 5). This high risk of bias was mainly in the domain of outcome measurement: PROs can be easily influenced by adverse events, especially when adverse events are psychoactive, as is the case with THC. The studies that investigated CBD without THC had indeed a low risk of bias in this domain.
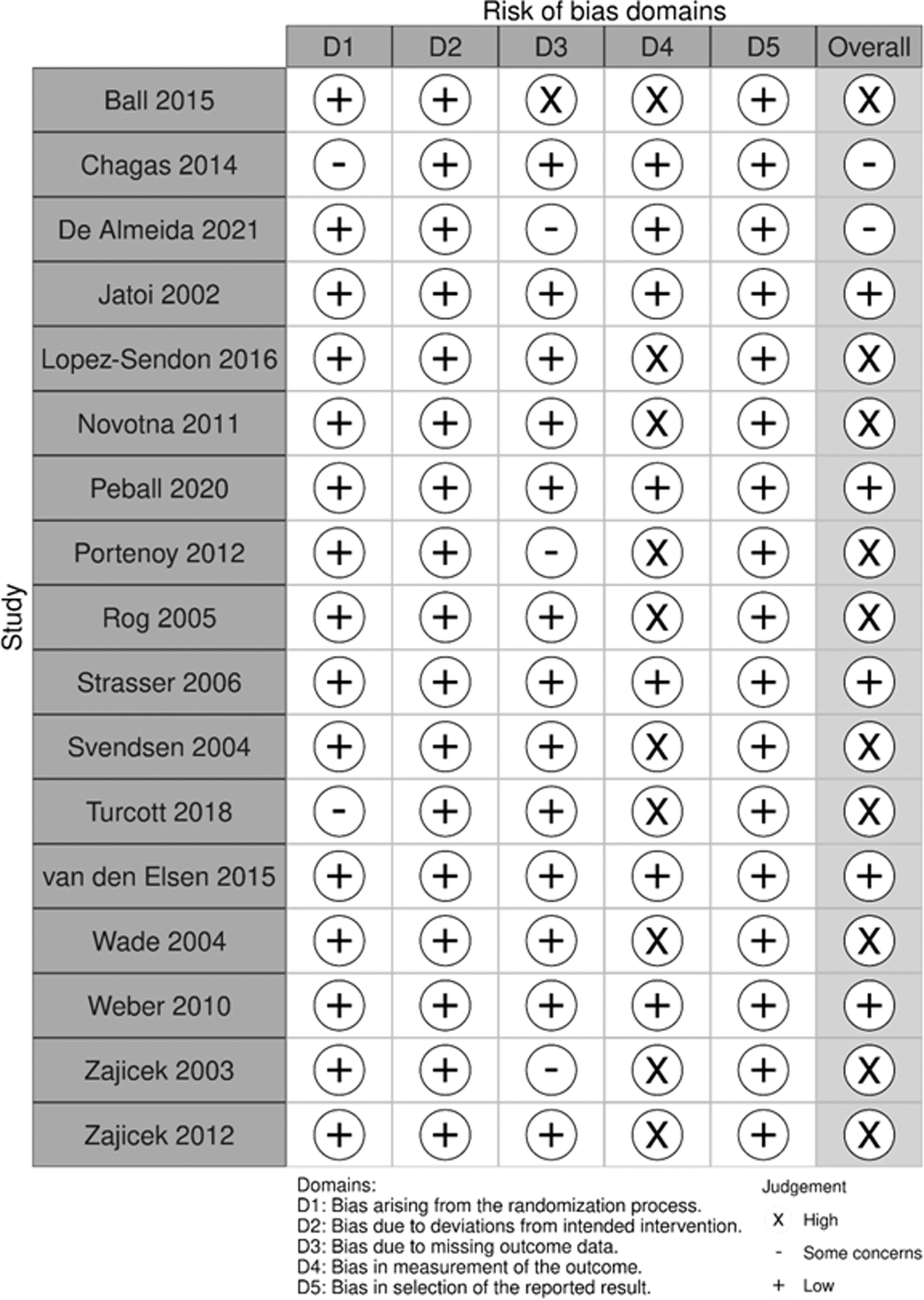
Risk of bias.
Discussion
In this meta-analysis, we aimed to assess the effects of cannabinoids on HRQoL in oncological patients and patients with CNS disease. These studies did not show an effect of cannabinoids on HRQoL. Only one small crossover study in MS patients observed an improvement of THC on mental well-being. 56 The primary outcome measure in this study was pain, which did improve with THC. Possibly, the improved mental well-being was secondary to pain reduction.
Our inability to find an association between cannabinoids and HRQoL improvement in patients with cancer or CNS disease could have several explanations. First, a true effect may not exist—the previously reported subjective reduction of symptoms being entirely due to placebo effects. Moreover, the self-retrieved cannabinoids that patients use and consider beneficial might be different from that in clinical or research setting. 62 Second, a true effect may be present, but not revealed due to suboptimal dosage, route of administration, or composition of cannabinoids. A biphasic effect for THC has been established in both pre-clinical and clinical studies: depending on the dose, THC seems to increase as well as decrease psychological complaints such as anxiety.63–66
In this meta-analysis, doses ranging from 1 mg to max 120 mg were investigated. Contrary to THC, CBD attenuates anxiety in both pre-clinical and clinical studies.67–69 The addition of CBD to THC might even prevent serious adverse effects from THC, such as paranoid psychosis.70,71 An optimal dose of THC or CBD has not been established. Third, a true effect may have been present, but measured with metrics insensitive to changes brought about by cannabinoids. Specific symptom measurements could possibly be more sensitive to cannabinoid effects than general HRQoL assessments. In the studies we retrieved, various general instruments were used, typically covering multiple domains.
The effects of cannabinoids would have to be large and affect multiple domains to show an improvement on these scales. Symptom-specific questionnaires such as the State and Trait anxiety Inventory (STAI) or the BDI are more sensitive to small changes in these subdomains and may better capture relevant changes. In addition, the majority of these studies did not assess HRQoL as a primary end-point; hence, many studies were not powered to detect HRQoL changes. Fourth, a true effect may have been present, but only in a small subgroup of responders, whereas many patients were nonresponders. For example, ∼50% of patients with spasticity have been shown to be a nonresponder to cannabinoids. 72 The wide CIs observed in some of the studies could support such an explanation.
However, two studies included in this meta-analysis did try to separate responders from nonresponders before the start of the main trial. The double-blind, placebo-controlled phase of these studies was preceded by a single-blind phase and an open-label phase, after which the patients who did not show enough improvement on the primary outcome measure were excluded from the second phase.73,74 Even though only the cannabinoid responders were included in the main phase of both studies, no significant effect was found on either general HRQoL or mental well-being. The primary objectives, spasticity and nonmotor symptoms in PD, respectively, did improve.
Our results are corroborated by other RCTs in these populations: of an additional 15 studies that could not be included in the meta-analysis due to insufficient data reporting, only 1 study found cannabinoids improved mental well-being significantly. 43 However, the authors comment that this change was offset by the worsening of another outcome to measure HRQoL, that is, the patient's global impression of change. No other study showed a (trend toward) significant effect on general HRQoL or mental well-being.36–42,44–50 However, a previously published extensive systematic review and meta-analysis on the effects of cannabinoids on mental disorders presented very low-grade evidence that THC:CBD reduces anxiety. 75
Of note, the authors could not include three RCTs due to insufficient data reporting, and hypothesize that including these studies would have resulted in a nonsignificant pooled effect. THC did improve anxiety symptoms in one included open-label study with five patients75,76; monotherapy CBD did not improve anxiety based on two RCTs. 75 In addition, cannabinoids did not improve depressive symptoms.
Cannabinoids are frequently used, presumably to reduce symptoms and consequently potentially improve HRQoL. For example, in Seattle, where cannabis is legal, almost a quarter of cancer patients use cannabis. 77 Cannabinoids are also frequently used by MS patients: a nationwide survey in the United States of >1000 respondents showed that 42% of MS patients recently used cannabis. Of this group, >90% reported the use of cannabis for medical reasons, such as pain or anxiety relief. 78 In three observational studies, some beneficial effects of cannabinoids were reported: a prospective study of 1144 cancer patients showed that after 6 months of medical cannabis use, the percentage of patients with a good HRQoL had increased from <20% to ∼70%. 79
The second study was a survey study in which patients reported an anxiolytic effect of medical cannabis. 80 The third study of >250 patients showed that HRQoL, anxiety, and depression improved after 3 weeks of CBD use. However, these observational studies lack a comparison to control for placebo effects, which are likely to occur. Moreover, a population bias could complicate these results even further as these patients themselves initiated cannabinoid use.
Considerably, heterogeneous patient populations, interventions, and outcome measures were included in this meta-analysis. The subgroup analyses, however, did not indicate differences between active intervention and control group in mental well-being or general HRQoL, except for a difference between the effects of THC and CBD:THC on general HRQoL, but not on mental well-being. CBD:THC did not decrease or increase HRQoL, and THC had only a small, possibly futile negative effect on general HRQoL.
In our efforts to obtain data from individual patients, we had to exclude almost half of the eligible studies due to limited reports and an inability to obtain these data through the authors. Poor reporting on outcome is a well-described phenomenon and might be even more frequent with HRQoL outcomes, corroborating our findings.81–84 Response rates of authors in our study were similar to rates reported by others.85,86 We have the impression that corresponding authors of investigator-initiated studies were more inclined to share their data than authors of industry-sponsored studies, possibly reinforced by the absence of effects on HRQoL or mental well-being. Alternative explanations may be that industry regulations prohibit sharing of these data, or that resources are limited to retrieve and provide such data.
Strengths of this meta-analysis include the application of a rigorous search strategy and study selection without restrictions for publication date, language, or publication type. To limit assumptions and thereby risk of bias, only RCTs were included, and data were not imputed. To further improve the study selection, two raters assessed the suitability of studies for inclusion and determined their risk of bias. All extracted data were verified by a second assessor. Limitations consist of inclusion of studies for analysis, which did not have HRQoL as a primary end-point by design. Also, various metrics were analyzed, which we conceptually compared by analyzing the standardized mean differences. Nevertheless, these metrics may not have been sensitive enough to detect small changes.
Furthermore, we combined intervention groups, which may have resulted in neglecting differences between THC and CBD:THC, or between different dosages of certain interventions. We favored to include all potentially useful information over restriction to a very specific administration and dosage of interventions.
Also, many included studies showed a high risk of bias. The main problem is the frequent occurrence of psychoactive (adverse) effects from THC, but not from the placebo, which may result in unblinding. Especially, in the context of PROs, patient knowledge on active and placebo treatment holds a high risk of bias. One strategy to overcome this problem is the use of CBD instead of THC, as psychoactive effects are lacking.
We did not assess adverse effects as they have been extensively investigated in earlier studies.63,87–89 The reported adverse effects in the included studies were generally mild. However, the literature reports that especially THC can have infrequent, but serious adverse effects, including paranoid psychosis and anxiety, which could affect HRQoL. 90 Adverse effects of high doses of CBD are generally mild and include elevated liver enzymes and sedation. 91
Clinical implications for brain tumor patients
Although patients with a primary brain tumor share some characteristics with patients with cancer or CNS disease, our findings are only indicative: there are of course dissimilarities between these populations. For example, anxiety is more frequently reported by patients with primary malignant brain tumor than in those with other cancer types or MS.9,92–94 This may be related to malignant primary brain tumor prognosis. Moreover, many metrics in this meta-analysis were disease specific, thereby taking into account factors possibly less relevant for glioma, such as rigidity or bladder control. Therefore, the effect of cannabinoids, in particular CBD, on well-being in patients with a primary brain tumor remains elusive. Our findings emphasize the need for well-designed studies to investigate these effects, preferably including disease-specific questionnaires such as the EORTC QLQ-BN20 (quality of life brain tumor module). 95
Conclusion
This meta-analysis of RCTs showed no effect of cannabinoids on HRQoL and mental well-being in patients with cancer or CNS disease based on 17 randomized controlled studies in 1771 and 1613 patients, respectively. However, studies were clinically heterogeneous and only two small studies investigated monotherapy CBD with undecided results. As many primary brain tumor patients currently use cannabinoids, and monotherapy CBD has not been sufficiently investigated, future studies are necessary to evaluate the efficacy of cannabinoids in HRQoL in this specific population.
Footnotes
Authors' Contributions
All authors read and approved the article. Literature search and selection: V.B., J.C.F.K., and J.G.R. Quality appraisal: V.B. and J.G.R. Data analysis: V.B., P.M.v.d.V., and J.G.R. Article draft and revisions: V.B, J.G.R., L.D., M.K., M.C.M.K., P.C.d.W.H., T.W., M.E.v.L., J.M.N., J.L.-S., K.B.S., M.G.O.R., M.W., C.M.O.d.A., and O.A. Data provision: J.L.-S., K.B.S., M.G.O.R., M.W., C.M.O.d.A., and O.A.
Acknowledgment
We would like to thank Yessica Denisse for her critical feedback.
Author Disclosure Statement
Arrieta reports personal fees from Pfizer, grants and personal fees from AstraZeneca, grants and personal fees from Boehringer Ingelheim, personal fees from Lilly, personal fees from Merck, personal fees from Bristol Myers Squibb, and grants and personal fees from Roche, outside the submitted work. The other authors declare no conflict of interests.
Funding Information
This meta-analysis has been funded by the Anita Veldman Foundation (CCA2018-2-17).
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.