Abstract
Abstract
N-arachidonoyl dopamine (NADA) is a member of the family of endocannabinoids to which several other N-acyldopamines belong as well. Their activity is mediated through various targets that include cannabinoid receptors or transient receptor potential vanilloid (TRPV)1. Synthesis and degradation of NADA are not yet fully understood. Nonetheless, there is evidence that NADA plays an important role in nociception and inflammation in the central and peripheral nervous system. The TRPV1 receptor, for which NADA is a potent agonist, was shown to be an endogenous transducer of noxious heat. Moreover, it has been demonstrated that NADA exerts protective and antioxidative properties in microglial cell cultures, cortical neurons, and organotypical hippocampal slice cultures. NADA is present in very low concentrations in the brain and is seemingly not involved in activation of the classical pathways. We believe that treatment with exogenous NADA during and after injury might be beneficial. This review summarizes the recent findings on biochemical properties of NADA and other N-acyldopamines and their role in physiological and pathological processes. These findings provide strong evidence that NADA is an effective agent to manage neuroinflammatory diseases or pain and can be useful in designing novel therapeutic strategies.
Introduction
The endocannabinoid (EC) system consists of cannabinoid receptors, mediators, and enzymes responsible for the synthesis and degradation of endogenous ligands, namely ECs. ECs are lipid signaling molecules, which are involved in a diverse range of physiological and pathological processes.1–3 N-acyldopamines consist of a hydrocarbon tail and a polar head group capable of interacting with cell membranes, membrane proteins, or ion channels function. The best examined member of this group is N-arachidonoyl dopamine (NADA) next to endogenous N-oleoyl dopamine (OLDA), N-palmitoyl dopamine (PALDA), and N-stearoyl dopamine (STERDA) and synthetic N-octanoyl dopamine (NOD). 4 The formation and inactivation of N-acyldopamines as well as their significance under physiological and pathological conditions are not fully understood yet. NADA was first synthesized as a pharmacological tool to study the EC system. 5 Later NADA and other N-acyldopamines were identified as endogenous cannabinoids in the mammalian nervous tissue.6,7
Several lines of evidence identified NADA, next to anandamide (AEA), as a member of the endovanilloid family acting as an agonist with similar potency as capsaicin. 6 NADA and OLDA act on transient receptor potential vanilloid (TRPV)1 and play an important role in nociception. It was postulated that endovanilloids such as AEA or NADA participate in the development of neuropathic pain and inflammatory hyperalgesia. 8 Despite similarity in the structures of NADA and AEA, these two ECs vary in their functional activity: some of them will be mentioned later.
This review is divided in four sections. (1) The first section describes distribution and the current status of our understanding on the synthesis, transport, and degradation of N-acyldopamines. (2) The second section summarizes our current knowledge on the pharmacology of N-acyldopamines and theirs receptors, such as cannabinoid (CB), CB-like, and TRP receptors, and coupled signal transduction pathways under physiological and pathological circumstances will be reported. (3) The third section deals with N-acyldopamines mediated modulation of neuropathic pain and inflammatory hyperalgesia. (4) Finally, other effects of N-acyldopamines, protective versus toxic, with actions on immune and those in vascular system are mentioned.
NADA: Chemistry, Distribution, Synthesis, Transport, and Degradation
NADA is an arachidonic acid derivative with a dopamine moiety in its structure (Fig. 1). Using quadrupole time-of-flight analysis, the presence of NADA has been reported in the striatum, hippocampus, cerebellum, thalamus, midbrain, and dorsal root ganglia (DRGs). 6 However, Bradshaw et al. detected NADA exclusively in striatum and hippocampus.9,10 Other N-acyldopamines such as OLDA, PALDA, and STEARDA were found in bovine brain.6,7 A recent study reported NADA at a concentration of 0.74±0.20 pg mg−1 and OLDA at 0.15±0.08 pg mg−1 in the murine striatum. 11 A basal level of 2.6±1.2 pmol g−1 wet tissue weight NADA was found in rat substantia nigra pars compacta. 11 Human plasma and human postmortem brain were devoid of NADA as analyzed by different chromatographical methods.12–14 The synthesis of N-acyldopamines is not yet fully understood. The biosynthesis of NADA has been examined by using both in vivo and in vitro assays. 15 In striatum, a region with high dopamine concentrations, NADA biosynthesis primarily occurred through an enzyme-mediated conjugation of arachidonic acid with dopamine requiring tyrosine hydroxylase (TH).
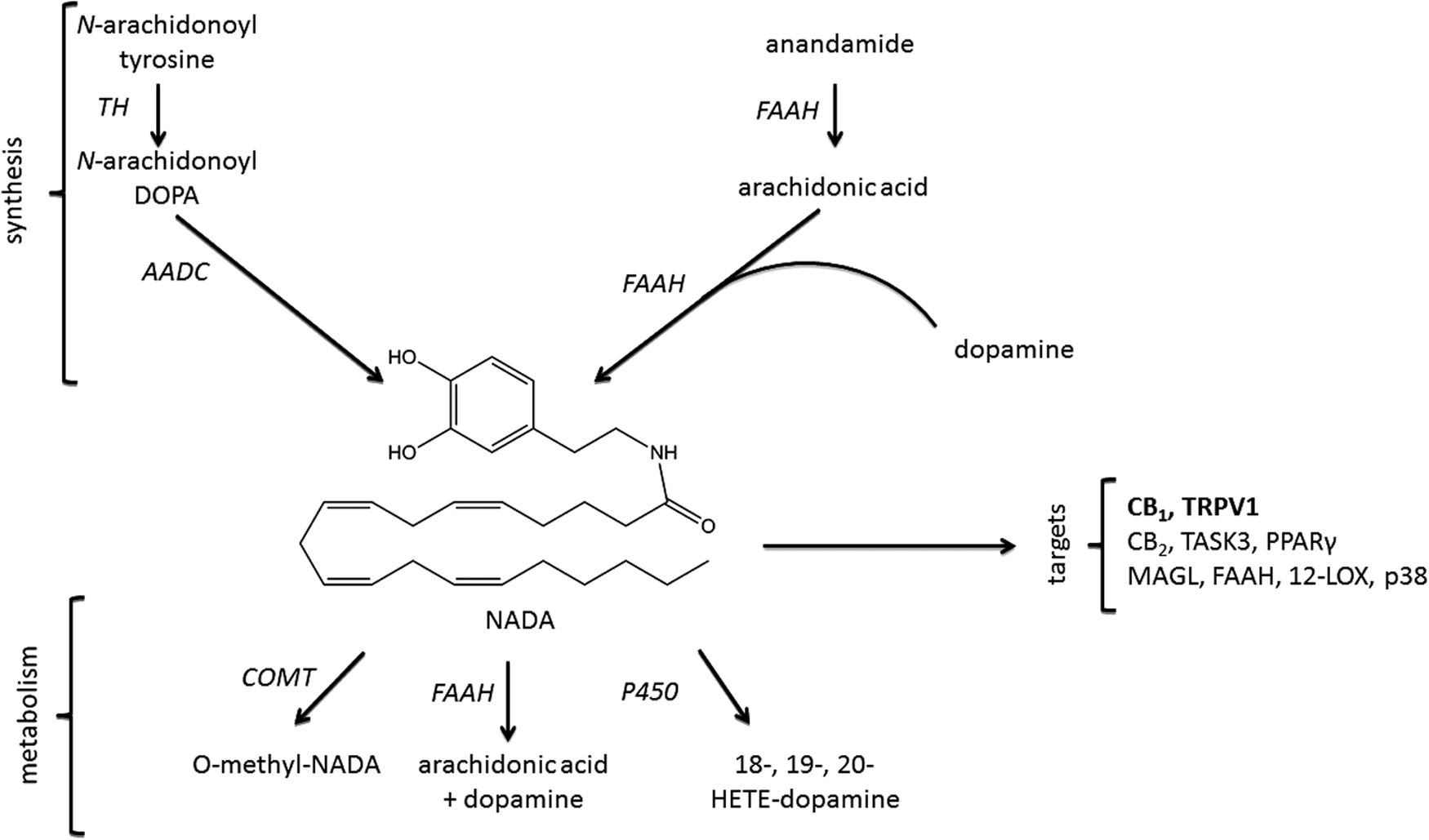
Potential synthesis and degradation pathways of NADA, including its targets. The first proposed pathway involves N-arachidonoyl tyrosine, which can be metabolized by tyrosine hydroxylase (TH) to N-arachidonoyl DOPA and by
NADA synthesis was observed almost exclusively in dopaminergic terminals, indicating that the dopamine level seemed to be the limiting factor. Fatty acid amide hydrolase (FAAH), a membrane-bound enzyme involved in AEA degradation, seemed also to be a rate-limiting enzyme in NADA biosynthesis, as the lack of FAAH led to a decrease of the striatal NADA concentration. 15 Concomitant NADA was shown to be a weak substrate and a competitive inhibitor (IC50=19–100 μM) of FAAH. 5 As a competitive inhibitor, NADA binds to the active site of the enzyme and reduces the proportion of enzyme molecules available for binding the main substrate, namely AEA. In this case, NADA inhibits the inactivation of AEA, suggesting a NADA-dependent potentiation of the AEA effects, or a control mechanism to prevent overloading with NADA.
Nevertheless, incubation of dopamine with arachidonic acid in the presence of FAAH led to the production of detectable amounts of NADA in vitro. 15 Dopamine receptors and FAAH were found in the same brain regions, such as hippocampus, striatum, and parts of cortex.16,17 NADA can, therefore, be produced in brain regions with a meaningful expression of FAAH and TH and released into the extracellular space to act on cells expressing target receptors.
ECs are degraded through a three-step mechanism namely cellular uptake, enzymatic hydrolysis, and re-esterification into membrane phospholipids.18,19 These pathways are also involved in the degradation of NADA (Fig. 1). The cellular uptake of lipids such as AEA occurs through diffusion or/and transporter proteins. 19 So far, there is no direct evidence of a membrane transporter for NADA, although numerous studies assumed its presence.19–23 Pharmacological studies have revealed a rapid uptake of NADA by anandamide membrane transporter (AMT) in C6 glioma cells, leading to inhibition of AMT (IC50=21.5±9.1 μM). 5 At high concentration OLDA (IC50=17.5 μM), PALDA (IC50 > 25 μM), and STERDA (IC50 > 25 μM) inhibit AMT in the [ 14 C] AEA uptake test in RBL-2H3 cells. 7 Regulation of an intracellular transport might have an important protective function. An increase in intracellular NADA concentration can deactivate AMT, preventing the receptor or/and intracellular signal cascades from overstimulation. In C6 glioma cells, NADA was hydrolyzed slower than AEA, probably by FAAH to arachidonic acid and dopamine. As originally postulated and later confirmed, NADA acts as a substrate for catechol-O-methyl-transferase (COMT).6,24 COMT is involved in the inactivation of catecholamines including dopamine and is distributed in the rat cerebral cortex, neostriatum, and cerebellar cortex. 25 This enzyme transforms NADA to O-methyl-NADA, which is less active at TRPV1 than NADA. 6
Like other ECs, NADA was shown to be metabolized through the cytochrome P450 pathway in rat liver microsomes. 26 Furthermore, oxidation of the arachidonoyl moiety played a minor role in endovanilloid inactivation. 6 More immunohistological and colocalization studies are needed to verify the subcellular distribution of FAAH, TH, or COMT, major metabolizing enzymes for NADA (Fig. 1).
Receptors and the Signaling Pathways
CB1 receptor
So far, only NADA and OLDA have been described as agonists of CB1, which is mainly present on neurons5,7 (Table 1). The CB1 receptor is expressed presynaptically on neurons among others in forebrain, hindbrain, and in the spinal cord.27,28 Not only neurons but also glial cells have been reported to express CB1 receptors. 29 The EC signaling plays a crucial modulating role in hippocampal formation, basal ganglia, cerebellum, and neocortex. 30 In the peripheral nervous system, the activity of nerve fibers innervating smooth muscles is modulated by the CB1 receptor. 31 Knockout of the CB1 receptor in mice is not lethal, but significantly leads to affected behavior and learning processes. 32
CB, cannabinoid; EC, endocannabinoid; FAAH, fatty acid amide hydrolase; HEK, human embryonic kidney; PPARγ, peroxisome proliferator-activated receptor-γ; TRPV, transient receptor potential vanilloid.
Natural and recombinant CB1 receptors can be coupled to Gs, Gi/o, and Gq proteins even in the same system.33–36 Several receptors that preferentially couple to Gi/o are able to interact with Gs, particularly when receptors and/or G proteins are expressed at high densities and high concentrations of agonists are present. 37 Signaling downstream of cannabinoid receptors is linked to regulator molecules and intracellular signaling networks that control basic cell functions. The precise characterization signaling effects are different because of strong differences in experimental design. In addition, homo- and heterodimerization of CB1 with other receptors such as dopamine receptors, expression and coupling of CB1 to channels/signaling cascades, or basal activity of those receptors are still a matter of controversial debate. 38
Typical actions mediated by Gi/o are direct inhibition of adenylyl cyclase, ensuing inhibition of protein kinase A, direct modulation of p44/42 mitogen-activated protein kinase (MAPK), activation of G protein coupled inwardly rectifying potassium channels, and inhibition of calcium channels. Modulation of p38 MAPK and c-Jun N-terminal kinases (JNKs) was also observed after Gi coupling. In contrast, Gq was shown to increase the intracellular calcium concentration.33,34,39–42
After activation of CB1 receptor, a transient Ca2+ elevation is evoked in a phospholipase C-dependent manner through either Gi/o or Gq proteins.43,44 Activation of Gs leads to receptor-mediated Ca2+ influx and to continued activation of different phospholipases. 38 In the absence of extracellular calcium, NADA stimulated an intracellular Ca2+ mobilization in undifferentiated N18TG2 neuroblastoma cells. This effect was counteracted by the CB1 antagonist SR141716A and mimicked by CB1 agonist, HU-210. 5 Redmond et al. were unable to reproduce the NADA-mediated elevation of [Ca]i under different assay and cell handling conditions. 45 In the study by Bisogno et al., cells were resuspended in a continuously stirred cuvette, and in Redmonds experiment, monolayer of the cells was plated.5,45
In human breast MCF-7 cancer cells, NADA potently inhibited (IC50=0.25 μM) the proliferation in a CB1-dependent and D2-independent manner. 5 In radioligand binding assay, NADA was shown to bind to hCB1 receptors and displaced both [3H]-CP55940 (Ki=780±240 nM) and [3H]-SR141716A (Ki=230±36 nM) with a similar affinity.5,45 NADA did not modulate p44/42 phosphorylation, adenylate cyclase, and potassium channels in cells expressing CB1. The authors concluded that NADA did not activate Gi/o or Gs coupled signaling. Interestingly, NADA (10–30 μM) mediated an activation of Gq/11 subunit of CB1, which led to an elevation in [Ca]i and induced an internalization of CB1. 45
NADA (Ki=250 nM) has an affinity to CB1 in the [3H]SR141716A binding inhibition assay, even stronger than AEA (Ki=0.8 μM). 5 Other compounds such as PALDA and STEARDA were inactive at concentrations smaller than 5 μM. OLDA exhibited some activity on CB1 receptor (Ki=1.6 μM) in the mentioned assay 7 (Table 1). In autaptic hippocampal neurons, NADA did not inhibit the excitatory postsynaptic current (EPSC) through CB1 in comparison with 2-AG.46,47
Effects on TRP channels
NADA and OLDA increase intracellular calcium/induced calcium influx through activation of TRP receptors, especially the nonselective cation channel TRPV1.6,7 Not only NADA and AEA but also lipooxygenase products of arachidonic acid and NADA metabolites activated the TRPV1 receptors.26,48,49 Nevertheless, distribution studies have revealed inconsistent results, beginning with the presence of this receptor in the brain tissue. Data about the localization in dentate gyrus, hippocampal pyramidal neurons, are still a matter of debate. Therefore, it is difficult to assess the results describing the action of NADA on TRPV1. The use of unspecific antibodies or the lack of suitable, reliable controls in immunohistological and Western blot analysis may result in such findings. Even in electrophysiological experiments, it is possible to trigger unspecific effects by using nonphysiological high ligand concentrations. Over the years, Trpv1 mRNA was shown to be widely expressed in primary sensory fibers and in diverse areas of the central nervous system, most abundant in the limbic system, striatum, hypothalamus, centromedian and paraventricular thalamic nuclei, substantia nigra, reticular formation, and cerebellum.48,50–55 However, later studies provide strong evidence for the presence of TRPV1 in low levels in the brain. 56 Although the expression of trpv1 mRNA in the brain was confirmed by many authors, the functionally active TRPV1 proteins seem to be missing in some cells. The issues of distribution of functionally active TRPV1 remain open, more studies are needed to confirm the role of TRPV1 in the brain.
To study the pharmacology of TRPV1 receptor and NADA-mediated effects, heterologous expression systems, such as transfected Chinese hamster ovary and HEK-293 cells with the TRPV1 (HEK-293-TRPV1) receptor, were used. Activation of the TRPV1 receptor led to nonselective cation influx, calcium influx, membrane depolarization, and glutamate release and cell death.48,52 The phosphorylation of the TRPV1 receptor induced also a sensitization, whereas desensitization was caused by dephosphorylation. 52 NADA has been shown to activate both human and rat TRPV1 overexpressed in HEK-293 cells (EC50≈50 nM).6,46,57 OLDA activated TPRV1 in human embryonic kidney (HEK) cells overexpressing human TRPV1 (EC50≈36 nM) because PALDA and STERDA were inactive. 7 In TRPV1 knockout murine trigeminal ganglion (TG) cells, NADA did not induce a current contrary to AEA, indicating the agonistic nature of NADA on TRPV1. 58 Other ECs such as PALDA and STERDA, mediated an entourage effect on NADA-mediated actions, indicating an enhanced calcium mobilization through TRPV1 when coapplied with NADA. 59
NADA binds to the intracellular domain of TRPV1 and requires a transport across the cell membrane.54,60,61 It has been reported that NADA and capsaicin were equipotent in rat neonatal DRG neurons. 6 However, we demonstrated that the response of NADA (10 μM) application was relatively small in comparison with capsaicin (10 μM) in patch clamp experiments on HEK-293-TRPV1. 46 Our results were supported by electrophysiological approaches in DRG neurons and in isolated guinea pig bronchi and urinary bladder62,63 (Table 2).
AMT, anandamide membrane transporter; CGRP, calcitonin gene-related peptide; CHO, Chinese hamster ovary; DRGs, dorsal root ganglia; GABA, gamma-aminobutyric acid; HUVECs, human umbilical vein endothelial cells; sEPSCs, spontaneous excitatory postsynaptic currents; sIPSC, spontaneous inhibitory postsynaptic current; TG, trigeminal ganglion.
Cross-talk between CB1 and TRPV1 receptors
The interaction between CB1 and TRPV1 has been postulated in several studies.64–66 There is an evidence of a high degree of colocalization of CB1 and TRPV1 in DRG, and in neuron-enriched mesencephalic cultures, hippocampus, and cerebellum.65,67–70 To our knowledge, none of the studies have demonstrated the colocalization of both receptors at synaptic levels. It was suggested that the activation of CB1 receptor may inhibit TRPV1-mediated toxic events. 66
In adult rat DRG neurons, NADA evoked significantly CB1- and TRPV1-dependent increases in intracellular calcium.6,63 It was postulated that blocking of CB1 receptor by the selective antagonist SR141716A alters NADA uptake into neurons and, thereby, reduces the ability of NADA to activate TRPV1. 63 In the presence of an antagonist, CB1 may block AMT and prevent thereby the NADA binding to the active site of TRPV1.
Application of capsaicin or NADA (1 μM) evoked increases in intracellular calcium concentration in DRG neurons through activation of TRPV1. This response was attenuated by both FAAH inhibitor (URB597) and AMT inhibitor (UCM707). Reduction in synthesis or uptake of NADA may explain this effect. 71 In substantia nigra pars compacta, NADA in the presence of one of the antagonists activated CB1 and TRPV1 in a concentration-dependent manner. 65 In patch clamp experiments, NADA led to an increase in glutamatergic transmission through TRPV1 but decreased the GABAergic transmission through CB1 in dopaminergic neurons measured as sEPSC (spontaneous excitatory postsynaptic currents), resulting in an excitatory effect. In contrast, NADA (1 μM)-mediated CB1 activation had an inhibitory effect measured as spontaneous inhibitory postsynaptic current on dopamine neurons after blockade of TRPV1. Furthermore, tonic inhibition of GABAergic transmission was mediated by NADA (1 μM) in a CB1-dependent way without the involvement of TRPV1. 10
Peroxisome proliferator-activated receptor-γ and NADA
Peroxisome proliferator-activated receptor-γ (PPARγ) is a nuclear receptor and transcription factor in the steroid superfamily. An increase in its transcriptional activity was induced by NADA among other cannabinoids. 72 NADA caused a time-dependent PPARγ-mediated, NO-dependent vasorelaxation of rat aorta. NADA's mentioned activity was inhibited by PPARγ antagonist (GW9662), CB1 receptor antagonist (AM251), and FAAH inhibitor (URB597). One possible explanation might be the involvement of FAAH in the synthesis of NADA. The inhibition of FAAH decreases NADA concentration and the dependent receptors remain inactivated. In addition, it results in an increased AEA concentration. Binding of NADA to CB1 receptor initiates also different intracellular pathways, like MAPK that presumably activates PPARγ.73,74
The Role of NADA in Tissue Function and Diseases
NADA and pain
Nociception is defined as a process by which thermal, mechanical, or chemical stimuli are detected by nociceptors. The cell bodies of the nociceptors are localized in DRG and TG. There are two major classes of nociceptors. One class is regulated by TRPV1 depending on changes in the local tissue and thermal chemical signals75,76 and activation of CB receptors.67,77 The other class of nociceptors, the peptidergic C nociceptors, release neuropeptides, such as substance P and calcitonin gene-related peptide (CGRP), and express TrkA receptor that binds nerve growth factor.78–80 It was demonstrated that the activation of antinociceptive CB1 by NADA reduces the pronociceptive actions evoked by TRPV1. 81
NADA combines features of ECs and endovanilloids, making it an interesting and potent therapeutic agent in development of analgesic drugs. NADA activates TRPV1 and CB1 receptor that have a well-established role in pain modulation and are present on DRG5,78,80 (Table 2).
Summarizing the data already mentioned, NADA induces both pro- and antinociceptive effects in the central and peripheral nervous system dependent on the concentration range. In thermal allodynia, NADA has been shown to relieve pain after topical administration in primates. 82 Injected intraperitoneally in mice, NADA induced hypothermia, hypolocomotion, and analgesia 5 similar to 20 mg kg−1 AEA. 83 Intraplantar injection of NADA (5 μg in 50 μL=0.07 mM) inhibited the smaller mechanically evoked innocuous responses of dorsal horn neurons through CB1 and for stronger mechanical stimuli through TRPV1. 63
High concentrations of NADA were pronociceptive in different model systems of male rats. When administered peripherally and intradermally, NADA caused behavioral thermal hyperalgesia through TRPV1 and CB1. 6 NADA (EC50=1.55 μg) administrated intradermally into the receptive fields of dorsal horn increased dose dependently both spontaneous and heat-evoked activity in spinal nociceptive neurons in laminae I-V of dorsal horn of the spinal cord in a TRPV1-dependent, CB1-independent manner, indicating pain sensation—thermal hyperalgesia. 8 Differences in the observations made by Huang et al., Huang and Walker, and Sagar et al. can be explained by a different kind of stimuli or different neurons, which were recorded. Coadministration of STERDA potentiated the induction of thermal hyperalgesia by NADA. 59 In contrast to capsaicin, subcutaneous injections of OLDA (EC50=0.72±0.36 μg) into rat hind paw induced a significant dose-dependent thermal hyperalgesia lasting for 3 h. No nocifensive behavior was observed (licking and lifting the hindpaw) till 30 min after injection. PALDA and STERDA alone had no effects in this study. 7
NADA displayed antihyperalgesic effects in models of inflammatory pain after intrathecal administration. These effects were reversed by antagonists of both CB1 (all NADA concentrations) and TRPV1 (high NADA concentrations). 84 NADA (5 μg) mimicked the action of TRPV1 antagonist and inhibited the neuronal responses to mechanical stimulation in electrophysiological recordings from the dorsal horn in anesthetized rats. 63 In contrast to previous findings, NADA evoked CGRP release from TG neurons 85 and from slices of rat dorsal horn spinal cord in a TRPV1-dependent manner, 6 resulting in neurogenic inflammation. NADA injected in TG or administrated intraocularly was excitatory, pungent, and evoked nocifensive responses. 85 It could also modulate different cation channels involved in pain sensation, like T-type calcium channels (Ca(V)3)86,87 (Tables 1 and 2). NADA strongly inhibits human recombinant T-type calcium channels (Ca(V)3 channels) expressed in HEK-293 cells and native mouse T-type, which was shown to play an important role in modulating peripheral and central pain processing in a variety of pain models.86,87 The role of calcium channels in NADA-mediated processes provides an explanation for the lack of complete blockade with CB1 as well as TRPV1 antagonists.
In the hippocampus as well, NADA produces opposite effects on Ca2+ entry. For example, NADA induced a rise of resting presynaptic Ca2+ and enhanced the release of gamma-aminobutyric acid (GABA) and glutamate. However, in low micromolar range, NADA inhibited the K+-evoked Ca2+ entry and K+-evoked Ca2+-dependent release of GABA and glutamate. These effects were not counteracted by JWH133 (CB2 antagonist), AM251 (CB1 antagonist), ruthenium red (TRPV antagonist), and sulpride (D2, D3, and D4 antagonist). Only TASK-3 inhibitors triggered the rise of resting intracellular Ca2+.
88
NADA was shown to inhibit voltage-gated sodium channel (veratridine-dependent), release of
Taken together, the mode of action of NADA mediated in the central nervous system needs further clarification. A wide choice of models covers a wide spectrum of physiological and pathological activities, but a complete characterization is missing. Colocalization studies of TRPV1 and CB1 in combination with electrophysiological and in vivo studies will help to clarify how NADA influences the function of nociceptors, ganglion, spinal cord neurons, and neuron–glia interactions.
Protection and toxicity
Cannabinoids have been shown to exert neuroprotective effects in different models. Neuroprotection is mainly associated with CB1 receptor activation,91,92 whereas neurotoxicity is associated with TRPV1 activation.66,93–95 After excitotoxic lesion in vivo as well as in vitro, CB1 receptor was found to prolong preservation of neurons.96,97 These results are in line with our data on NADA. 46 NADA (1 nM) was neuroprotective in organotypical hippocampal slice cultures after NMDA treatment, partly through CB1. In electrophysiological experiments, NADA (1, 10 μM) did not inhibit EPSCs in autaptic hippocampal neurons. 46 We assume that CB1-dependent decrease in intracellular calcium concentration does not mediate NADA's neuroprotective effects. High concentrations of NADA (10 μM) seem to activate additional mechanisms preventing the neuronal demise as the neuroprotection was independent of CB1, TRPV1, and abn-CBD receptors. 46 In addition, NADA showed protective effects in cultured cerebellar neurons by reducing oxidative stress induced by hydrogen peroxide 98 and in primary hippocampal neurons against hypoxia through CB1. 99 Pretreatment with NADA protected human neuroblastoma cell line SK-N-SH from hypoxia. 100 NADA (5 μM) induced cell death in human neuron-like cell line SH-SY5Y, stably expressing recombinant human TRPV1. 93 Despite similarities to an apoptotic process, the cell demise took place independent of caspase activity and was blocked by a TRPV1 antagonist. 93 In contrast, in vivo studies demonstrated protective effects of TRPV1 activation on neurons against excitotoxicity 101 or ischemia. 102 Little is known about the signaling pathways involved in NADA-mediated protection and toxicity. Also NOD seems to have a specific protective function in endothelial cells. In human umbilical vein endothelial cells, NOD was protective against cold preservation injury measured in lactate dehydrogenase test. 103 Furthermore, NOD improved the renal function in setting of ischemia in vivo by downregulation of NFκß and subsequent inhibition of vascular cell adhesion molecule 1 in proximal tubular epithelial cells. 104
Immune cells
At cellular level, microglia plays a critical role in brain damage. NADA has an anti-inflammatory potential acting through a mechanism that involves reduction in the synthesis of microsomal prostaglandin E synthase (mPGES-1) in lipopolysaccharide-activated microglia. NADA is a potent inhibitor of PGE2 synthesis, without modifying the expression or catalytic activity of COX-2, or the production of prostaglandin D2 that plays a central role during neuroinflammation.105–107 It had also the ability to prevent free radical formation in primary microglial cells. AEA and NADA had opposite effects on glial cells. 106
Furthermore, NADA specifically inhibits IL-2 and TNF-α gene transcription in Jurkat T cells and inhibits the signaling pathways mediating the activation of transcription factors NF-κB, NFAT, and AP-1 involved in the immune response. NFAT was shown to regulate the changes in microglial phenotype.108,109
NOD did not affect the early T cell activation (IL-2, TNF-α, and IFN-γ) but inhibited NFκB and AP-1 activation in phorbol 12-myristate 13-acetate/ionomycin-stimulated T cells. It decreased the proliferation of both naive and memory lymphocytes without any toxic effects. Moreover, in the presence of NOD, the number of T cells, which did not pass beyond the G0/G1 phase, increased. 110 NADA had an inhibitory activity on HIV-1 replication in Staphylococcal enterotoxin B-activated peripheral primary T cells, peripheral blood mononuclear cell, and in Jurkat T cell line. 111 This effect, independent of CB1 and FAAH, was believed to result from changes at the transcriptional level by affecting both Tat and NFκB-dependent transcription. NADA, OLDA, and PALDA also prevented the degranulation and release of TNFα and decreased the tnfα-mRNA in RBL-2H3 mast cells treated with an IgE-antigen complex. PALDA was the most potent antiallergic N-acyldopamine, which downregulates allergic mediators through multiple targets such as Syk, Akt, p44/42, cPLA2, and 5-LO pathways. 112
Human neutrophil migration in Boyden chamber assay was inhibited by NADA (nM), independent of CB1 and CB2. 113 We observed that NADA (100 pM and 1 μM) significantly reduced the number of isolectin B-positive microglial cells after excitotoxicity. 46 These observations support the anti-inflammatory effects of NADA directly on immune cells.
Effects on vascular system
Several lines of evidence indicate that the cardiovascular depressive effects of cannabinoids are mediated by CB1 receptors. Recent studies provide strong support for the existence of as-yet-undefined endothelial and cardiac receptors that mediate certain EC-induced cardiovascular effects. TRPV1 receptor was shown to be present on sensory neurons innervating smooth muscles in several organs and in arteriolar smooth muscle cells. 56 Besides, capsaicin-sensitive sensory nerves participate in regulation of the vascular tone, inter alia through the release of vasodilator neuropeptides, such as CGRP. NADA has been demonstrated to induce vasorelaxant effects in human small mesenteric vessels, the superior mesenteric artery, and in the aorta 114 (Table 2).
In small mesenteric vessels, NADA-mediated vasorelaxant effects were CB1, abn-CBD, and TRPV1 dependent and were probably mediated by activation of potassium channels and an intrinsic endothelial mechanism independent of dopamine (D1) receptors. 114
NADA-mediated vasorelaxation in superior mesenteric artery was CB1, capsaicin dependent, and independent of abn-CBD receptor. Moreover, NADA caused dose-dependent depressive effects in rats fed with a normal and high-sodium diet. These effects were reversed by the TRPV1 antagonist, capsazepine, and CGRP receptor antagonists but not the CB1 receptor antagonist, SR141716A. Interestingly, activation of TRPV1 by NADA mediated the CGRP release from mesenteric arteries. 115
Conclusions and Outlook
The aim of this review was to summarize the current knowledge on N-acyldopamines with special reference to the functional role of the endocannabinoid NADA, in brain, pain modulation, and in other organ systems. NADA acts mainly through CB and TRPV1 receptors participating in several physiological activities in the body. NADA is neuroprotective, acts on immune cells, and mediates vasorelaxation. NOD was also shown to inhibit T cell activation and could be used for the treatment of inflammatory diseases and in the transplantation medicine. NADA, NOD, and OLDA inhibited aggregation of human platelets. Further investigation is needed to explore their therapeutic application. NOD implementation in transplantology has been proposed several times. NADA seems to affect the proliferation/migration and actions of immune cells especially microglia; NOD inhibits the proliferation of T cells but does not impair T cell activation. NADA potentially mediates both anti- and pronociceptive responses depending on the balance between CB1 receptors and TRPV1 channel activation, and the kind of stimulus. TRPV1 desensitization might be a possible explanation for diversity of NADA-mediated actions. A better understanding of the mechanism behind NADA, NOD, and OLDA-mediated actions may lead to development of novel therapies in acute neurological disorders and in neuroinflammatory pain. However, we need to understand first how exactly their synthesis and degradation occur, in which cell type these process take place, and to learn more about the function of endogenous NADA. The majority of data originate from animal studies. It is possible that the conflicting data on NADA represent species and cell-specific differences. Even if the distribution of the receptors is conserved between species, the coupling to the signaling cascades and effectors is often different. It is still not known whether NADA concentration levels change after lesion or under pathological situations. Precise determination of NADA as a “tricky” compound seems to be difficult. Therefore, better, more reliable, and faster methods are urgently needed. Presumably, like other endocannabinoids, NADA is produced on demand and gets degraded very fast. Owing to NADA-mediated neuroprotection, two parallel directions need to be investigated. First, how exactly N-acyldopamines influence immune cells, especially microglia. The changes in microglial and lymphocytes morphology, migration, cytokine profile, mRNA, and microRNA expression need to be screened. Second, the better understanding of EC system under pathological conditions might help to establish NADA as a potential therapeutic agent, if the problems with its instability and oxidation are solved. Would chemical modifications make NADA's application possible? The role of NADA in the regulation of motor activity and in the Parkinson's disease needs further investigation.
Footnotes
Acknowledgments
The authors would like to deeply thank P. Chandra (Goethe-University, Frankfurt) for critical reading of the article and T. Hohmann (Martin Luther University, Halle-Wittenberg) for helpful discussions. Urszula Grabiec was supported by the Roux Program FKZ 26/27 and FKZ 29/18.
Author Disclosure Statement
No competing financial interests exist.
