Abstract
Many pathogens that can cause major public health, economic, and social damage are relatively easily accessible and could be used as biological weapons. Wildlife is a natural reservoir for many potential bioterrorism agents, and, as history has shown, eliminating a pathogen that has dispersed among wild fauna can be extremely challenging. Since a number of wild rodent species live close to humans, rodents constitute a vector for pathogens to circulate among wildlife, domestic animals, and humans. This article reviews the possible consequences of a deliberate spread of rodentborne pathogens. It is relatively easy to infect wild rodents with certain pathogens or to release infected rodents, and the action would be difficult to trace. Rodents can also function as reservoirs for diseases that have been spread during a bioterrorism attack and cause recurring disease outbreaks. As rats and mice are common in both urban and rural settlements, deliberately released rodentborne infections have the capacity to spread very rapidly. The majority of pathogens that are listed as potential agents of bioterrorism by the Centers for Disease Control and Prevention and the National Institute of Allergy and Infectious Diseases exploit rodents as vectors or reservoirs. In addition to zoonotic diseases, deliberately released rodentborne epizootics can have serious economic consequences for society, for example, in the area of international trade restrictions. The ability to rapidly detect introduced diseases and effectively communicate with the public in crisis situations enables a quick response and is essential for successful and cost-effective disease control.
Biological Weapons
Approximately 17 countries in the world, including the former USSR and 5 countries that are implicated as sponsors of international terrorism, have or have had an active biowarfare research program, and several international terrorist organizations are believed to have the financial resources and political contacts needed to gain access to modern bioweapon cultures and production technologies.2,3 Additionally, groups and individuals dissatisfied with a government or society have been known to use or plan to use biological weapons for personal reasons. 3
Current classification of potential biological weapons is based on agent characteristics, such as the ease of obtaining and dispersing the agent, its virulence, morbidity, mortality, and also the capacity for individual-to-individual transmission. On the web pages of the Centers for Disease Control and Prevention (CDC) and the National Institute of Allergy and Infectious Diseases (NIAID), potential biological weapons are classified, according to the criteria mentioned above, into A, B, and C categories. Category A lists the pathogens that are most feared in the context of bioterrorism events. 4 Most attention may need to be focused on combatting infections that have serious medical consequences, but in several cases, the economic damage caused by a pathogen may demand actions even though the medical effects are trivial. 1 Bioterrorists may not always be constrained by the need to target or predict their outcome precisely, and extensive epidemics are not a prerequisite for creating great public anxiety.4,5 Simple fear of a disease could be enough to cause economic problems for a region because of, for example, diminished interest in recreational activities and tourism. Consequently, virtually any pathogenic microbe could be used by bioterrorists.
Besides human health concerns, the agricultural sector is vulnerable to bioterrorist attacks because domestic animals rarely have an innate resistance to foreign pathogens and are usually not vaccinated against these diseases. 4 Even small outbreaks of exotic diseases in livestock could remove a country from the global market for its agricultural products. 4 Agricultural bioterrorism is especially insidious as animals themselves may become the primary vectors for transferring agents. 4 Furthermore, the majority of category A and B potential biological weapons on the CDC lists are of a zoonotic nature, 5 and thus an attack on animal populations could pose a health risk for humans as well.
Bioterrorism and Wildlife
Unless an area is intensively monitored for pathogens, a deliberate release of infectious agents will probably not be detected at the time of release, but only after people or domestic animals exhibit symptoms of infection. The incubation periods of different kinds of infectious diseases vary, but in many cases, days or even weeks can pass before an outbreak is noticed. This provides the pathogen with the opportunity to spread and infect other organisms that may not have been targeted by the bioattack, such as local wildlife.
Wildlife is a natural reservoir for many diseases, and, as seen in the emergence of West Nile virus in the United States, the existence of wildlife reservoirs makes eradication of a disease that has had time to establish itself unrealistic. 6 The damage can become extensive when the released agents manage to spread over large geographic areas before they are discovered. Many wildlife species are highly susceptible to high-priority bioterrorism agents, 7 and, consequently, pathogens released during a bioattack directed at humans or livestock can either jump to wildlife or persist in the environment and cause zoonotic epidemics for a long time. Furthermore, several diseases with serious medical or economic significance for humans (eg, many viral hemorrhagic fevers) can infect wildlife species without their exhibiting overt clinical signs of infection, which can make the source identification of outbreaks complicated. Control measures of infectious diseases often involve immunization, but in the case of wildlife infections in which humans and domestic animals are not the reservoirs, herd immunity is not achievable, and pathogens cannot be eliminated through immunization. 1
Rodents and Infectious Diseases
Rodents can function as reservoirs and vectors spreading infectious diseases. 8 Many rodent species are highly opportunistic, meaning that they can easily adapt to new conditions and are able to take advantage of temporarily suitable environmental conditions for rapid reproduction. These characteristics have allowed some species, such as the house mouse (Mus musculus) and brown rat (Rattus norvegicus), to spread over most of the Earth's terrestrial areas. Opportunistic qualities may also support a potential dispersal of other accidentally or deliberately introduced rodent species outside their endemic areas in the future. Moreover, the opportunistic species often have peridomestic affinities and often live in close proximity to human settlements, which makes them an important link for transmitting infections among wildlife, humans, and livestock. 9
The absolute numbers of infectious diseases associated with rodents are not known, mainly because of the lack of complete systematic surveys of rodents for pathogens. 9 However, according to Hugh-Jones et al., 10 rodents function as reservoirs for approximately 46% of all globally known zoonoses, and more people are believed to have died in the past thousand years because of rodentborne infections than wars. 8 The symptoms of infections that are transmissible by rodents vary from relatively trivial (ie, skin lesions at the site of infection in cases of cowpox) to commonly fatal ones (ie, bubonic plague). 1 Transmission of rodentborne infections (Figure 1) may happen directly from rodents to humans (ie, hantaviruses) or via an arthropod vector (ie, fleas in cases of plague, ticks in cases of Lyme disease, and sand flies in cases of leishmaniasis). 1 Studies have also shown a direct connection between the abundance of rodents in the wild and the number of rodentborne human infections. 11
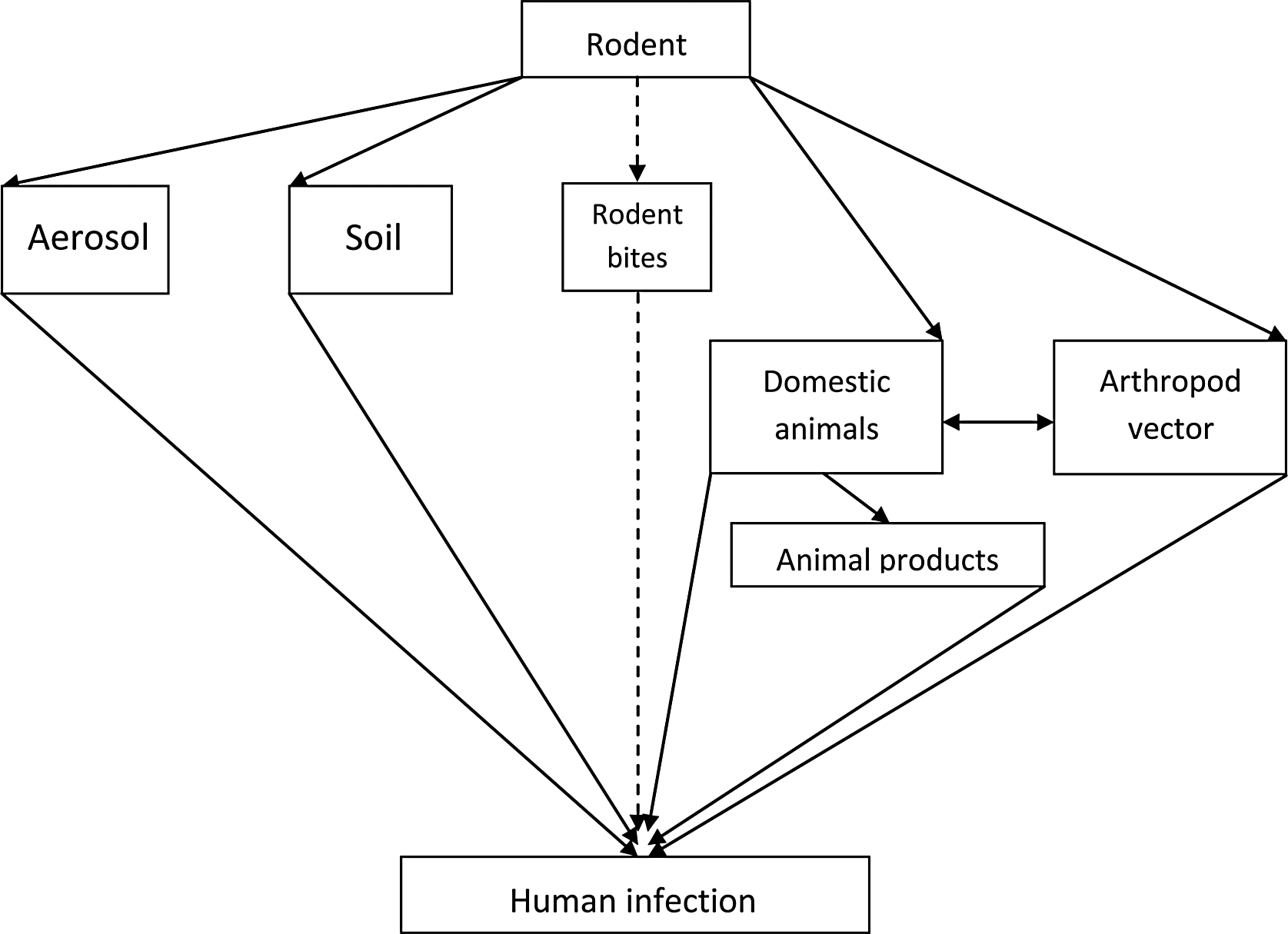
Examples of possible routes for human infection with rodentborne pathogens. Rodentborne pathogens in aerosolized form can be obtained directly through inhalation. Human infection can also occur by consuming contaminated water, or through skin and wounds when in contact with infected soil/water. Diseases can also be transmitted through rodent bites; however, compared to other sources this pathway is relatively uncommon. An indirect way to obtain pathogens is through contact with rodent-infected domestic animals or by consuming food products from these animals. Another route for indirect infection is through arthropod vectors (ticks, mosquitoes, flies, and so on) that after being infected from a rodent reservoir transmit the disease to humans when obtaining blood meals.
Rodents as a Dispersal Mechanism
A deliberate release of infected rodents or the contamination of wild rodents with pathogens would be a relatively easy way to threaten public health. For example, a diseased animal that is left on the ground or in a water source would be easily accessible to wild rodents and could cause a disease outbreak in an unexpected location. Many diseases are environmentally hardy and will be able to persist in organic or inorganic matrices for an extended period 4 and could thus potentially infect, for instance, burrowing rats. Subsequent contact of these animals with humans or domestic animals could spread the disease. No incidences of rodent-targeted bioterrorism have been described in modern times. But during World War II, the Japanese army, Unit 731, was reported to have experimented with plague by harvesting plague-infected fleas from infected rodents and dropping them over populated areas in China and Manchuria. 12
The likelihood of unintentionally infecting wild rodents during a bioattack targeted against humans or livestock is plausible. Most of the diseases caused by category A pathogens exploit rodents as vectors or reservoirs (eg, plague, tularemia, arena viruses, etc). This is also true for many of the diseases caused by category B pathogens (eg, toxoplasmosis, brucellosis, Q fever, etc). It might not be very likely that rodents come across anthrax letters targeted at certain individuals, but aerosolized pathogens that are released into the environment in a less targeted way could infect rodents, especially if the pathogen can survive in the environment for a long period. In cases of bioterrorism directed toward livestock, soil and water contamination by infected excreta from domestic livestock could spread pathogens to the sympatric rodent populations and in this way establish a rodent reservoir of infection. 13
Rodents as Disease Vectors
Humans often provide rodents with an abundance of supportive resources and favorable microclimatic conditions that allow them to grow in population size, especially in areas where humans have greatly reduced the number of their natural predators.14,15 In Europe, brown rats and house mice are among the most recognized urban rodents. These 2 species have quite different behavioral characteristics; while the house mouse is a highly inquisitive species, rats are cautious and prefer the familiarity of a known environment. 14 Rats also show a more dominant nature, suppressing and occasionally preying on house mice.
Urban rodent metapopulations usually consist of many small groups that may have little contact with each other except in cases of disturbance.14,16 However, disturbance in the form of road work or building activities is a common event in cities, and it is likely that these human activities enhance the transmission of pathogens between rodent groups. These activities could also increase the opportunities for contact among rodents, humans, and pets. In undisturbed rodent groups, young males typically leave their native groups to disperse into new areas. 16 Hence, young males can be expected to be responsible for spreading pathogens between rodent groups. Local overpopulation of rats in cities is most common in areas of failing hygiene, as, for example, near train and bus stations with abundant remains of food and other materials. 17 Additionally, unrestricted food supplies in gardens with compost heaps are thought to function as valuable shelters for rats. Just as train and bus stations might be a preferred target for a bioterrorism attack because of the potential for the maximum effect on public health, such areas would also be a prime place for the introduction of pathogens into rodent reservoirs.
The structure of rodent communities in rural areas is similar to that in urban ones, but the size of individual territories may be more flexible and may vary with the season and food availability. Furthermore, the structure of rodent communities depends on the local land use and differs, for example, from the areas that are used for livestock farming and cultivation of crops. The mobility of rats depends of the reliability of their food sources. For instance, brown rats can be immobile during food abundance but may roam more than 6 kilometers in other circumstances.18,19 According to Taylor, 18 rats that lived near reliable food sources in agricultural areas rarely moved more than 30 m from their home sites, but when the food was removed, they expanded their range considerably. Ironically, that means that rodent-proofing a farm after a bioterrorism event would ensure transmission between neighboring farms and probably even to wildlife species. In this case, if the goal is to prevent disease dispersal, it would be wiser to use trapping or rodenticides locally and follow recommendations from an ecological rodent management approach 20 rather than rodent-proofing a farm and withdrawing all food sources.
Ecological Context of Disease Dispersal
The lack of predators and low biodiversity are often ignored ecological parameters that greatly facilitate infectious disease dispersal. Modern cities have a low level of biodiversity, and natural competition and predation mechanisms are altered, allowing abnormal and unbridled growth of certain species. Generally, when a pristine ecosystem is disturbed, specialist species with specific food, nesting, or breeding requirements cannot survive. In the absence of such specialist competitors, opportunistic species with high fecundity will thrive. 21 As mentioned above, several opportunistic species also possess characteristics such as high mobility and high population densities that make them ideal hosts and transmitters of zoonotic pathogens. 21
In recent years, it has become increasingly evident that the loss of biodiversity tends to increase pathogen transmission and disease incidence.9,22 Consequently, the term “dilution effect” has been admixed into public health forums. The “dilution effect” hypothesis suggests that high biodiversity reduces the risk of transmission of certain diseases. 23 “Dilution effect” functions only when the pathogen is transmitted horizontally and when the reservoir competence for a certain pathogen varies among potential host species. Increased biodiversity would then increase the likelihood that infected organisms meet more individuals from a non–host species that would limit pathogen transmission. Indeed, epidemiologic and experimental studies have shown that lower diversity of small mammals increases the prevalence of hantaviruses in their hosts, leading to increased transmission risk to humans. 22 Consequently, as a deliberate release of pathogens can be expected to take place in areas of low biodiversity, this condition will considerably facilitate pathogen dispersal. However, in areas where municipalities are regularly performing rodent control, the dynamics of rodent populations and diseases may greatly differ from more degraded urban areas. Additionally, the increasing populations of urban foxes in many European cities may lower the rodent populations but, unfortunately, also add to the dispersal of parasites with complex life cycles, such as Echinococcus multilocularis and other helminths.24-26
The “One Health” Approach
Understanding disease management as a multifactorial issue is critical for preventing disease expansion including in the context of bioterrorism. Behavior and physiology of reservoir organisms (ie, organisms that can amplify a pathogen but are not necessarily a direct link to transmission, often rodents), including humans, wildlife, and domestic animals, and of the disease vectors (ie, organisms that actually transmit the disease, often arthropods but also rodents in some cases) are important factors contributing to infectious disease expansion. Dealing with an outbreak is therefore a task beyond medical and public health specialists alone. It demands the additional understanding of veterinary and environmental factors and knowledge of issues regarding human social behavior and political changes, basic science-related information about pathogen life cycles and evolution, and aspects related to vector/reservoir life history. 27 Cooperation between specialists with different expertise is necessary because the factors associated with disease expansion are not independent: For example, modifications in human behavior can change the ecology of wildlife reservoirs and/or disease vectors and in this way affect the transmission patterns of pathogens. It can be expected that, for example, family doctors and veterinarians in countries that are nonendemic for a particular disease will not be aware of its clinical manifestations. This can cause a delay in the diagnosis, leading to higher numbers of fatalities and increased dispersal, including to wildlife. In order to recognize the role of wildlife in the context of bioterrorism, it is important that veterinarians be aware of clinical signs of infection with biothreat agents in wildlife. 7 If disease outbreaks occur in nonendemic areas, outside the expected season, or in unexpectedly large numbers, these events could be investigated for the possibility of bioterrorism. 4
Examples of Rodent Reservoir Bioagents
Plague
The life cycle of plague, Yersinia pestis, involves a complex interaction between rodents and fleas, occasionally involving human infections. 28 The most important enzootic and epizootic reservoirs of plague are different kinds of rodents. 29 Plague agents are able to survive in the soil for long periods and can be transmitted to burrowing animals for extended periods.30,31 Plague occurs worldwide but primarily in certain locations in Africa and Asia. 7
The 3 major forms of Y. pestis infection in humans are classic bubonic plague, primary septicemic plague, and pneumonic plague. 28 Untreated bubonic plague has a fatality rate of 40% to 60%, whereas the pneumonic and septicemic forms are usually fatal. Human infection can be acquired by flea bites from infected fleas, but also by contact with fluids from infected animals and by respiratory droplets from infected humans or animals. 28
Human cases are most commonly associated with die-offs of rodent hosts that lead to fleas leaving the dead animal and seeking new hosts. The mouthparts of rodent fleas are often too weak to penetrate the human skin, but cat fleas can do it with ease. 31 Cats are highly sensitive to plague and can be infected both by flea bites and by ingestion of infected rodents. They are also the most likely animals to bring plague into human facilities.7,28 Clinical signs in humans develop 1 to 6 days after becoming infected, providing the pathogen with a time-lag that could be sufficient for both people and animals to move and spread the disease, making control of the disease more difficult. 7
Plague is a high-risk potential weapon of bioterrorism. Humans can be easily infected via aerosols, and aerosolized bacteria are easily transmitted between infected humans and animals. This disease has a high attack rate and produces severe clinical disease. 28 Moreover, antibiotic-resistant Y. pestis strains are known to exist, and, in aerosolized form, it would be a formidable weapon. 32 Plague still occurs naturally in several locations in the world, so it is a relatively easy agent to obtain. It can also be easily disseminated into wild rodent populations in both urban and rural areas.
Tularemia
At least 4 subspecies of Francisella tularensis (tularemia) are recognized, 2 of which are human pathogens: Francisella tularensis biovar tularensis and Francisella tularensis biovar palaeartica. 7 F. tularensis infection has been demonstrated in an impressive number of wildlife species, 33 but it is still mainly considered to be a disease of rodents and lagomorphs. Arthropods, such as ticks, biting flies, and mosquitoes, serve as vectors for tularemia and potentially even as long-term reservoirs. 33 Tularemia infections occur mainly in the northern hemisphere, most frequently in Scandinavia, Central Europe, North America, Japan, and Russia. 15
Tularemia is highly infectious and can be transmitted by several epidemiologic routes: through arthropod vectors, direct contact with infected blood and tissues, intact skin and wounds, ocular mucous membranes, inhalation, and ingestion.7,28 The infectious dose depends on the mode of transmission: Only 10 organisms are needed to cause disease by inhalation, whereas 108 organisms are required parenterally. 28 Six different forms of tularemia have been classified according to their clinical presentation and route of exposure, although the pneumonic form is the most likely bioterrorism agent because it can be transmitted as an aerosol and has 30% to 60% mortality if untreated. 34 F. tularensis has been weaponized by the United States and very likely by several other countries.33,34 The highly infectious nature of tularemia makes it possible to use it in the most “unsophisticated” ways of bioterrorism, as both rodents and humans are able to acquire infections through direct contact with infected rodent carcasses or by consumption of food or water that is contaminated by an infected animal. 15
Brucellosis
Six species with numerous biotypes of Brucellae have been identified, but only Brucella abortus, B. melitensis, B. suis, and B. canis can cause human disease. 28 Brucellosis is a worldwide zoonosis that mainly infects domesticated animals, but different wild mammals, including rodents (especially brown rats and even voles), act as reservoirs.35,36 In livestock, brucellosis is a lifelong infection. The bacteria localize in the reproductive tract and cause spontaneous abortions and sterility. 28 Transmission to humans can occur when open wounds are exposed to animal secretions, by infected aerosols, or via consumption of unpasteurized dairy products.
In the case of Brucellae, a major threat arises from agroterrorism—that is, the deliberate infection of domestic animals. Brucellosis is an economically important disease in production animals worldwide. 35 Countries that are presently free from animal brucellosis could suffer serious economic losses if brucellosis were to be introduced into their livestock. 35 Brucellae are a highly virulent pathogen in humans and animals and would be an effective biological weapon. 35 Only 10-100 aerosolized organisms are needed to cause disease, and the early symptoms of the disease are nonspecific, often leading to delayed diagnosis and ineffective treatment. Although Brucella is sensitive to heat and most disinfectants, it can survive in the environment for up to 2 years, thus constituting a continuous threat to both humans and animals. 37 In 1954, B. suis became the first agent to be weaponized by the United States, but several other countries have or are suspected to have weaponized this agent, including the United Kingdom.28,37
Q Fever
Coxiella burnetii, which causes Q fever, is a category B bioterrorism agent that is highly infectious in both humans and livestock. C. burnetii spores are extremely resistant to heat and desiccation but also to cold. 28 C. burnetii is a worldwide zoonosis that infects various mammals (including domestic goats, sheep, and cattle), birds, fish, and arthropods. Urine, feces, and birth material of infected animals are sources for C. burnetii contamination in the environment. Human infections usually occur after inhalation of aerosolized bacteria: A single inhaled organism is enough to produce clinical illness. 28 The placentas of infected animals can be heavily contaminated with these bacteria, and during parturition bacteria are released into the environment and may cause infection for up to 150 days. 28 The bacterium can survive in the environment for years and may travel long distances in the form of aerosol. 19
Although cultivation of Coxiella is rather laborious, large amounts of infectious material can be produced. 38 Coxiella may not cause high mortality, but if released as an aerosol, it could cause acute disabling disease and great economic losses in the agricultural sector. Acute infection of Q fever can be followed by fatal (eg, endocarditis) or debilitating (eg, chronic fatigue syndrome) disorders. Because of nonspecific symptoms, the diagnosis of Q fever might be delayed. Antibiotic treatment is available for the acute form of C. burnetii but not for the chronic complications. 38
Brown rats are thought to function as an important factor in the dissemination of endemically circulating Q fever between wild and domestic animals and humans. 19 Cats may have a role in these infection routes as well since they prey on the wild reservoirs.
Viral Hemorrhagic Fevers
Viral hemorrhagic fevers (VHFs) are caused by a group of viruses that require an animal or insect host reservoir for transmission. 39 They are usually geographically restricted to specific regions of the world where they create enzootic infections. Humans generally get infected after exposure to contaminated saliva, urine, or feces of infected animals or by rodent or insect bites. 39 VHFs include arenaviruses, such as Lassa fever and South American hemorrhagic fever viruses; bunyaviruses, such as hantaviral infections (HTVs) and Rift Valley fever virus (RVFV); flaviviruses, such as dengue hemorrhagic fever, tickborne encephalitis, and yellow fever viruses; and filoviruses, represented by Ebola and Marburg viruses. For most of these diseases, there are only limited treatment and vaccination options. Some VHFs have been weaponized by researchers in the former Soviet Union, the US, and possibly North Korea. 40
Arenaviruses
Arenaviruses are included in the category A pathogen list and as such pose a threat to public health if used as a bioterrorism agent. Traditionally, arenaviruses are divided into the Old and New World arenaviruses. The reservoir hosts of almost all arenaviruses are different kinds of commensal or semicommensal rodents that live close to humans or in cultivated fields. 41 In rodents, most of the arenaviruses cause a persistent, asymptomatic infection with chronic viremia and viruria. Lymphocytic choriomeningitis virus (LCMV) is an arenavirus with a worldwide distribution because of its association with the house mouse, Mus musculus. LCMV rarely causes severe infection except in infants and immunocompromised individuals.41,42 In contrast, Old World viruses (ie, Lassa virus and Lujo virus), which are found in Africa, and several New World viruses in South America (eg, Junin, Machupo, Sabia, Guanarito, and Chapere) can cause severe disease and hemorrhagic fever syndrome. 43 Rodent-to-human infections of arenaviruses are thought to occur through aerosols and fomites. 41 This indicates that these agents could potentially be used as bioweapons if propagated to high titer in cell culture and in aerosols. 43
Most cases of arenavirus transmission occur in rural, agricultural areas. The lack of rapid diagnostic methods for specific diagnoses of arenaviral hemorrhagic fevers means that diagnoses are usually made late in the illness and in most cases vaccination is not available. 43 Because arenaviruses are rarely transmitted from person-to-person and are not carried by arthropods, each arenaviral disease is tightly constrained in the geographic range of its rodent reservoir host. 43 This means that a bioterrorism attack could increase the infection prevalence in local rodent fauna in endemic areas only (although a possible spill-over infection in other species cannot be ruled out). When an attack also involves the release of infected rodents, the spread of the disease would depend on the ability of the infected host to establish itself in a nonendemic area. Given the opportunistic nature of many rodent species, it may be a possibility in some cases.
Hantaviral Infections
HTVs are transmitted to people via aerosols of infectious excreta (urine, feces, saliva) from chronically infected small wild rodents. 44 HTVs are often classified as either hantaviral fever with renal syndrome (HRS) in the Old World or hantaviral pulmonary syndrome (HPS) in the New World, but this dichotomy is not clear in all cases. 45 All HTVs are parasitic in small rodents, such as mice, rats, and voles, causing a relatively asymptomatic lifelong infection. HTVs are often species specific, sometimes infecting only a single rodent species. 39
HPSs cause severe, sometimes fatal, respiratory disease and are mainly associated with wild rodent species of the subfamily Sigmodontinae. 46 In the US and Canada, the Sin Nombre virus (also found in other parts of the Americas) is responsible for the majority of cases of HPS. The host of the Sin Nombre virus is the deer mouse (Peromyscus maniculatus), found throughout the western and central US and Canada but also southward in the Americas. In South America a myriad of different HTVs can be found: Andes virus has been reported in Argentina, Chile, and Uruguay; Laguna Negra virus in Paraguay and Bolivia; Rio Mamoré virus in Bolivia; Caño Delgadito virus in Venezuela; and Araraquara and Juquitiba viruses in Brazil. 47
The most severe form of HRS is caused by Hantaanvirus (ie, carried by wild urban rats Rattus norvegicus and Rattus rattus and Apodemus mice) and Dobrava-Belgrade virus (associated with Apodemus mice) that occur in Asia and Eastern Europe. A moderate form, the Seoul virus (carried by various species of rats), occurs only in Asia. The majority of mild hantavirus infections in Europe are caused by Puumala virus (primarily associated with the bank vole, Clethrionomys glareolus). 44 Mortality after exposure to the severe forms is approximately 5% to 15% and to the milder form, 1% to 2%.
Because HTVs exist worldwide and are mainly transmitted by aerosols, there is concern about their potential use as biological weapons, even if they are only listed as category C agents on the CDC's list of bioagents. Their potential threat is increased by the fact that immunity in populations is generally very low, many strains without cross-protection exist, and no vaccines are available. 39 Furthermore, diagnosis can be difficult when the disease occurs outside its typical range, as initial clinical symptoms are nonspecific. 39 Still, HTVs are notoriously difficult to isolate and produce, even with the means available in the most advanced laboratories, and they usually do not transmit from person-to-person. 44 The relative rodent host-specificity of the most virulent HTVs means that introduction of the disease to local wild rodents is possible only in endemic areas (although spill-over effects to local species outside of the endemic areas have not yet been investigated and must be considered a possibility). Introduced into rural or urban rodent fauna, HTVs have the potential to cause long-term medical episodes and to spread fear.
Filoviruses
Filoviruses, Marburg and Ebola, are highly virulent pathogens that have a high mortality rate. If terrorists were to cause even a small outbreak of Marburg or Ebola, the public perception of the threat alone could cause major social and economic disruption. 48 However, the life cycle of these viruses and the potential capacity of rodents as vectors or reservoirs for Ebola and Marburg is still largely unknown. Fruit bats are commonly suggested as the main reservoirs for these diseases, 49 but Morvan et al. 50 detected Ebola virus glycoprotein and polymerase gene sequences in rodents and shrews and suggested that common small terrestrial mammals living in peripheral forest areas could be reservoirs for Ebola. Even though these results have been debated, 49 the possibility that rodents would function as vectors or reservoirs for these viruses in the event of bioterrorism cannot be ruled out. Furthermore, since more than a week would elapse before the first symptoms of illness appear, and since Filoviruses survive at room temperature in liquid or dried material for a number of days, the release of these viruses in an urban area 48 could result in their transmission (eg, to rodents) before the disease is discovered.
Rift Valley Fever Virus
RVFV is a zoonotic arthropodborne pathogen native to Africa that increasingly causes severe morbidity and mortality in humans and livestock. 51 The lack of prophylactic and therapeutic methods, the potential for human-to-human transmission, and the significant threat to livestock associated with RVFV make this pathogen a serious bioterrorism threat. 52 RVFV is transmitted through a broad range of mosquito genera and by other vectors including sand flies. 51 This broad range of competent mosquito vectors, some with a worldwide distribution, makes the prospect of transmission and spread to native wildlife following a bioterrorism event possible. Rodents, among them brown rats, have shown RVFV antibodies in endemic areas and have been suggested to serve as potential intermediate amplifying hosts during periods of livestock immunity following an epizootic. In this way, rodents would play a crucial role in the maintenance of the virus's natural cycle. 53 Human RVFV infections are usually preceded by transmission from wild to domestic animal hosts. RVFV has a devastating effect on livestock: For example, in sheep, mortality in lambs under 2 weeks of age approaches 100%, in older animals 30%, and abortions approach 100%. Cattle also show high abortion rates (up to 100%), with adult mortality at approximately 10%. 51
Contagious Animal Diseases
Even if they do not affect human health, an outbreak of contagious animal diseases can have serious economic consequences for livestock. Consequently, enzootic livestock diseases represent a potentially serious threat against national agricultures as well as many wildlife species. 2 Several formerly devastating diseases that have been eradicated from livestock populations in the western world over the past century are still common and readily accessible elsewhere. 2 For some diseases, samples or cultures obtained from infected animals are all that would be required to initiate a serious outbreak. Foot-and-mouth disease, malignant catarrhal fever, Newcastle disease, and Rift Valley fever are examples of diseases that can pose major threats to livestock and wildlife and that have been cultivated and possibly weaponized according to Office International des Epizooties. 2 Furthermore, these diseases have been shown to infect or be transported by rodents. Other diseases that can infect rodents and could cause economic losses in agricultural settlements are classical swine fever, porcine parovirus, clinical encephalomyocarditis fever virus, and neosporosis. 15
Will Human Infections Be Detected in Time?
The degree and spread of human infections after a bioattack depends on local circumstances. In well-developed areas with good medical resources, the disease dispersal is likely to be noticed and stopped earlier than in poor and degraded areas. If an urban bioattack should result in roaming infected rodents, the most likely humans to first encounter the pathogens are people in degraded urban areas with poor hygiene and especially homeless people. Because of their isolation from the community, these groups can see the disease developing to full lethal proportions before it is discovered. If an affected person is alive when transported to the hospital, the hospital has a chance of performing diagnostics, and it is likely that the pathogen will be identified. But this depends on the standards and experience of the hospital staff, and in cases of bioattacks the previous experience and consequently the capacity to expect a disease may be lacking even if the hospital has high standards. When a person who died due to an unknown, uncommon disease is presented for autopsy, the chances are small that the pathologist will recognize it immediately, and there is a risk that no extra precaution will be taken initially, thus providing the pathogen with the potential to spread to those in the autopsy room. As is the case with tuberculosis transmission in autopsy rooms, 54 other pathogens can be aerosolized during an autopsy and spread to workers. If the body is still fresh at the autopsy, an infectious disease may be recognized, but if a body is already decomposing, there is no chance that a forensic pathologist will recognize an infection because the decomposition will hide every possible sign. In this scenario, the chances of transmitting the pathogen to, for instance, a pathologist or members from the police team, in addition to the wildlife that may have been previously rooting around the body, are significant. Additionally, no alarm will reach the public until days later when more infections have been discovered.
Prevention, Communication, and Education
It is obvious that the best way to, at least partly, avoid the dispersal of rodentborne diseases after a bioattack is to control the number of (commensal) rodents before the possible incidence. The efficacy of proactive rodent control programs is evident from several reports, but today complaint-based reactive pest control seems to be the most popular strategy for rodent management among policymakers, as these control actions are easy to document and demonstrate to those with a rodent problem. 55 Unfortunately, data from the UK show that if no proactive control is undertaken, it is unlikely that more than about 30% of infestations would be eliminated by a reactive complaints-based strategy. An alternative proactive rodent control strategy enables resources to be targeted where the risk is greatest and is more likely to achieve effective control. 55 One of the best examples of this strategy is the long-term urban rodent control program undertaken in Budapest, Hungary, where a strategy applied and monitored over 30 years has not only involved effective control methods but also an analysis of the behavior and habitat use of urban rats. 55 Furthermore, several ecological management approaches to control city rats, involving changing the location of food sources in buildings so that the environment is less predictable to rats and breaking links between populations in resource-rich patches have been demonstrated to be efficient in the long term. 20 However, resources for proactive work are in many places in the world being withdrawn in favor of less effective and poorly targeted reactive strategies. 55
When a bioattack happens, in an emergency, there is no time to develop an elaborate strategy for public health communication. If the situation has not been analyzed properly, officials will most likely improvise, and the risk of spreading incorrect information is great. 31 Methods of communication that function without inducing panic among the general public must therefore be developed in advance.
Local pest management and control agencies are valuable partners for officials in case of a rodentborne disease outbreak, since they most likely have the data about current rodent activity, and they are specialists in pest elimination methods. Consequently, they need to be involved in the lines of communication both with officials and the general public.
After a bioattack involving rodentborne pathogens, rats and mice, both dead and live, should be considered potentially infectious. 31 People should be advised not to touch dead animals and to make their homes, shelters, and cabins rodent-proof. Homes should also be kept clean, and food and garbage should be covered in rodent-proof containers. 7 However, as discussed above, rodent-proofing and cutting off the food flow of rodents in a facility that already contains infected rodents would just result in spreading the pathogens to other households as the rodents will try to find new food sources. Local trapping is recommended, but it is difficult to perform efficient trapping over large rural areas. Rodenticides may be a more efficient solution, but not many rodenticides are allowed in outdoor environments, as the side effects of them on nontarget species may be devastating and cause liver toxicity and increased parasite and pathogen burden.56-58 Furthermore, many rodent species have been shown to become resistant to the most commonly used rodenticides. 59 Consequently, developing an efficient and nature-friendly way to implement rodent control would be of high importance.
A failure to involve the public as a key partner in a public health crisis could hamper effective epidemic management. 60 Simple, clear language should be used for public instructions, making it easier for all groups of society to follow them. As described above, when a pathogen has been introduced among wildlife species, the eradication of it will be difficult, and recurring disease outbreaks can be expected in the future. Consequently, the recommendations for handling the situation must be made with a long-term perspective.
Tailored messages are needed for people with jobs that create special exposures. Special education may be provided for, for example, hunters and outdoor recreationists. Restricting activities in areas that are likely to harbor infected animals can also be an important preventive strategy. 7 People who are frequently outdoors should also make sure they wear tick-, flea-, and mosquito-proof clothes and wash their hands with clean water after contact with soil. Domestic pets should be treated for ectoparasites, as they often can bring disease home. 7
Active surveillance of wild animal populations both before and after a bioterrorism attack is necessary for identifying common baseline values for occurrence of certain diseases and sources of infection in the environment. Better approaches for intervention are needed to be able to prevent the spread of an introduced biological warfare agent into a wild animal population. 6 We also need additional research about relative susceptibilities and exposure pathways for animal species living near human populations. An ability to rapidly detect introduced diseases and react quickly to hazards is essential for successful and cost-effective disease control. Diagnostic methods that facilitate early detection (eg, analyses that can detect hundreds of pathogens in a single sample, multiplex assays) can help in the process.
Footnotes
Acknowledgments
Writing of this publication has been supported by the framework of the EU project AniBio Threat (Grant Agreement: Home/2009/ISEC/AG/191) with financial support from the Prevention of and Fight against Crime Programme of the European Union, European Commission—Directorate General Home Affairs. This publication reflects the views only of the authors, and the European Commission cannot be held responsible for any use that may be made of the information contained therein.
