Abstract
Hearing loss, the most common neurological disorder and the fourth leading cause of years lived with disability, can have profound effects on quality of life. The impact of this “invisible disability,” with significant consequences, economic and personal, is most substantial in low- and middle-income countries, where >80% of affected people live. Given the importance of hearing for communication, enjoyment, and safety, with up to 500 million affected globally at a cost of nearly $800 billion/year, research on new approaches toward prevention and treatment is attracting increased attention. The consequences of noise pollution are largely preventable, but irreversible hearing loss can result from aging, disease, or drug side effects. Once damage occurs, treatment relies on hearing aids and cochlear implants. Preventing, delaying, or reducing some degree of hearing loss may be possible by avoiding excessive noise and addressing major contributory factors such as cardiovascular risk. However, given the magnitude of the problem, these interventions alone are unlikely to be sufficient. Recent advances in understanding principal mechanisms that govern hearing function, together with new drug discovery paradigms designed to identify efficacious therapies, bode well for pharmaceutical intervention. This review surveys various causes of loss of auditory function and discusses potential neurological underpinnings, including mitochondrial dysfunction. Mitochondria mitigate cell protection, survival, and function and may succumb to cumulative degradation of energy production and performance; the end result is cell death. Energy-demanding neurons and vestibulocochlear hair cells are vulnerable to mitochondrial dysfunction, and hearing impairment and deafness are characteristic of neurodegenerative mitochondrial disease phenotypes. Beyond acting as cellular powerhouses, mitochondria regulate immune responses to infections, and studies of this phenomenon have aided in identifying nuclear factor kappa B and nuclear factor erythroid 2-related factor 2/antioxidant response element signaling as targets for discovery of otologic drugs, respectively, suppressing or upregulating these pathways. Treatment with free radical scavenging antioxidants is one therapeutic approach, with lipoic acid and corresponding carnitine esters exhibiting improved biodistribution and other features showing promise. These compounds are also histone deacetylase (HDAC) inhibitors, adding epigenetic modulation to the mechanistic milieu through which they act. These data suggest that new drugs targeting mitochondrial dysfunction and modulating epigenetic pathways via HDAC inhibition or other mechanisms hold great promise.
Background
Vision is the pre-eminent sensory means by which we navigate the world around us, 1 while the ability to hear endows us with the power of voice communication. It enriches our lives with the sound of music and alerts us to imminent danger that can be heard although perhaps not seen. The Greek physician-philosopher Alcmaeon of Croton2–5 and his two protagonist followers, Praxagoras of Kos3,6 and the great Alexandrian physician of Chalcedon, 7 Herophilus,2,3 propounded that hearing is a construct of the brain, where external sound channeled to it through the ears (transduced into sensorineural signals) is interpreted. 6
Hearing loss is the most common neurological disorder affecting people worldwide. 8 The World Health Organization (WHO) estimates that the annual cost of unaddressed hearing loss is in the range of $750–790 billion globally. 9 In the “Global Burden of Disease,” impaired hearing represents the fourth leading cause of disability worldwide.10–12 About 5–7% of the world's population (∼360–500 million people) has a hearing disability12,13 that is severe enough in more than 80% of people older than 85 years to interfere with their ability to communicate effectively. 10 In children, impaired hearing can impinge on their academic potential and social development 13 with lifelong adverse consequences. 14 Hearing loss due to mechanical (conductive) and/or anatomical issues in the outer and/or middle ear (Fig. 1) is less prevalent than that resulting from dysfunction in the cochlea and/or the auditory nerves (sensorineural) in the inner ear or a mixture of conductive and sensorineural components.10,15
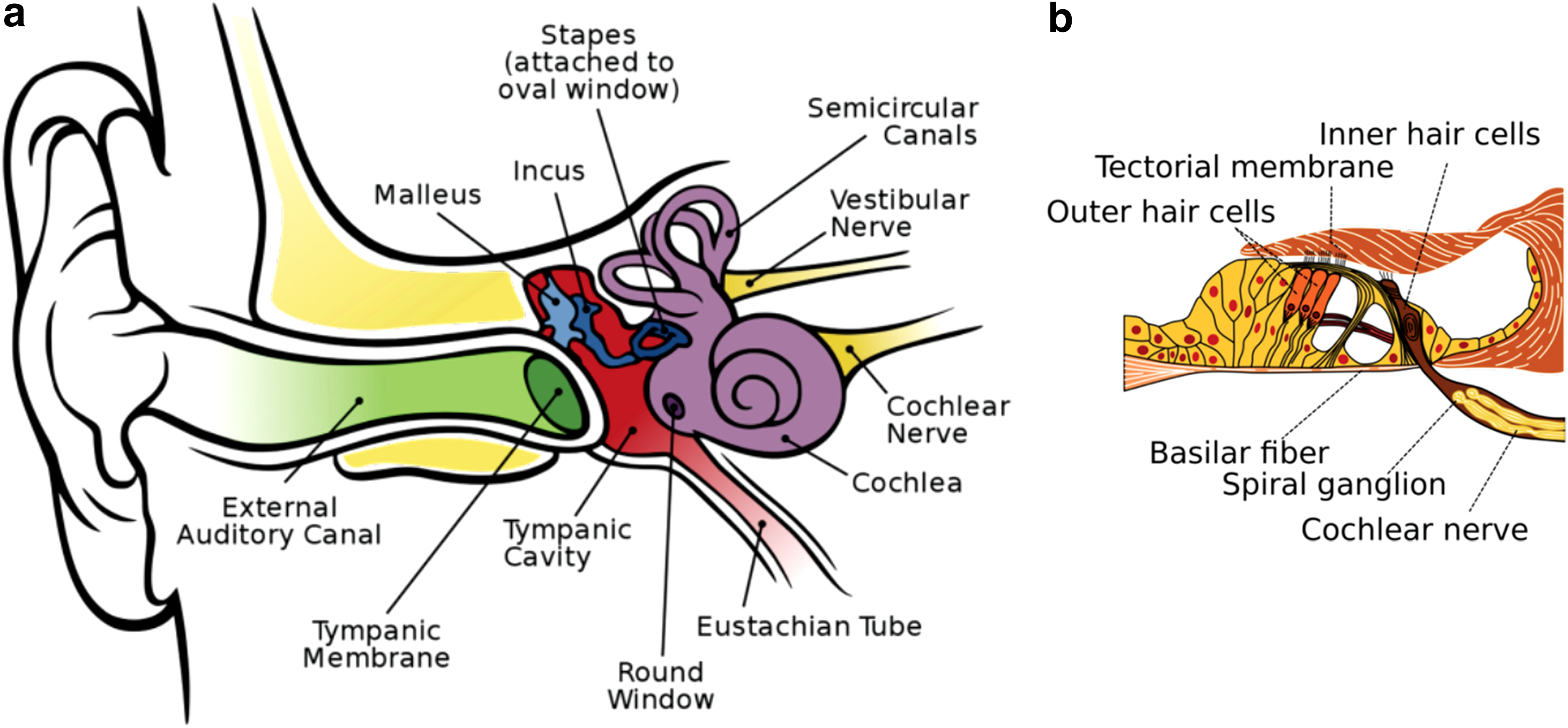
Anatomy of the human ear.
Although the array of techniques used in the diagnosis of sensorineural hearing loss (SNHL, the predominant form of hearing loss worldwide) is progressively being expanded and refined, 14 developing therapeutics to treat the onset of SNHL is proving to be considerably more difficult to realize13,14—thus placing a premium emphasis on prevention.12,16 A compelling case for prevention is noise-induced hearing loss (NIHL), one of the most common types of SNHL.17,18 Noise pollution is a growing health problem around the world.19–22 For example, in the United States alone, it is estimated that more than 25% of the adult population has measurable hearing loss caused by exposure to harmful noise. 10 Irreversible hearing loss can also be the product of disease12,16,23,24 and is often an unfortunate side effect of the aminoglycoside antibiotics25–30 and platin-based anticancer drugs.10,27,28,30–36 Regrettably, once damage has occurred, hearing aids and cochlear implantation are the only compensatory options presently available for affected individuals. 37
There is a substantial and growing worldwide unmet medical need for a pharmaceutical approach to treating hearing impairment.38,39 SNHL results from damage to the organ of Corti (Fig. 1), causing the degeneration of spiral ganglion neurons due to excessive injury and/or untoward death of cochlear hair cells in the inner ear,10,40,41 in most cases caused by internal or external pathologic factors, including infectious or inflammatory processes, ototoxic drugs, noise overstimulation, as well as the normal aging process.13,16,25,40,42 Unexpectedly, a recent meta-analysis of numerous epidemiological studies exposed a possible link between presbycusis (also known as age-related hearing loss, ARHL) and cognitive decline, cognitive impairment, and dementia—suggesting that ARHL may be a relevant biomarker and a targetable modifiable risk factor for dementia.43–46 However, given that advanced age itself is a leading risk factor for dementia,1,47–49 the meta-analysis correlation is not surprising.
Mitochondrial Dysfunction in Hearing Loss
Mitochondria mitigate cell protection, survival, and function1,50–52 (Hoffman ME, Augsburger BN, Foradori CD, et al. Neuroprotective effects of carnitinoid compounds in rodent cellular and in vivo models of mitochondrial complex I dysfunction. 2018; submitted) and, over time, they succumb to an increasing cumulative degradation of their cellular energy production and performance—driving the cell toward death (apoptosis) and/or premature senescence (Hoffman et al., submitted).40,47,48,53–59 Energy-demanding cells such as neurons and the vestibulocochlear hair cells (Fig. 1) are especially vulnerable to mitochondrial dysfunction60,61 and, consequently, hearing impairment/deafness is a characteristic clinical symptom of several neurodegenerative mitochondrial disease phenotypes.24,25,44,62–66
Focusing on the genetic basis of hearing loss, it is worth noting that many mutations in the mitochondrial genome (mitochondrial DNA, mtDNA), as well as in the nuclear genome (nuclear DNA, nDNA), are known to cause hearing deficits (Table 1).24,62,63,65–67 Alterations in certain regions of mtDNA associated with deafness are also associated with a host of other disorders, such as myopathy including cardiomyopathy, diabetes, and parkinsonism.68–72
Unless otherwise noted.
In addition, mitochondria are key regulators of our innate and adaptive immune responses to viral infections.50,73–76 Often overlooked among the many causes of impaired hearing, including deafness, is virus-induced hearing loss.77–79 Although the mechanisms of hearing loss/deafness associated with viral infections remain largely undefined,78,80 viral infections activate a cascade of mitochondrial antiviral innate immune responses that include nuclear factor kappa B (NF-κB)50,75,76,80,81 and nuclear factor erythroid 2-related factor 2 (Nrf2)/antioxidant response element (ARE) signaling pathways.1,50,76
Unmitigated oxidative stress (which is primarily caused by mitochondrial dysfunction) and epigenetically altered expression of genes sensing oxidative stress are significant contributors to the pathogenicity of neurodegenerative disorders (Hoffman et al., submitted).47,52,54,76,82,83 While a full understanding of these pathways awaits further study, it seems clear that epigenetics plays a significant role. Mammalian vestibulocochlear hair cells are a stress-sensitive, nonregenerative cell type and, like the retinal cells of the eye, 1 are not replaced when they are injured or die.10,13,14,29,57,60,82,84–86 Interestingly, emerging research indicates we selectively amplify directional sound in a noisy environment by unconsciously utilizing eye–ear coordination to integrate visual cues with the auditory information.87,88 Assessing vision and oculomotor function is essential in the diagnostic evaluation of vestibulocochlear auditory impairments, 89 particularly in patients with idiopathic etiology.90,91
α-Lipoic Acid, l-Carnitine, and Butyrate
Impressive advances in gene therapy8,14,92–94 and regenerative medicine are making inroads toward regenerating hair cells with the aim of reversing hearing loss.41,95 Some of the achievements demonstrate therapeutic potential,8,86 but a clinical application is still a long way off.24,39 In the more immediate future, shielding hair cells from oxidative damage and/or rescuing injured hair cells from falling into apoptosis by pharmacological treatment with free radical scavenging antioxidant compounds portend a promising therapeutic approach.13,14,29,30,57,96–99
Upregulating Nrf2/ARE gene expression pathways and/or suppressing NF-κB signaling are cogent targets for pharmaceutical intervention strategies. 34 Many natural and synthetic compounds are known inhibitors of NF-κB signaling 100 —butyric acid (butyrate)50,101–105 and α-lipoic acid (5-[(3R)-1,2-dithiolan-3-yl]pentanoic acid)50,106–110 (Fig. 2) are among them. Importantly, butyric acid and α-lipoic acid (ALA), as well as their respective corresponding l-carnitine esters PMX550DBr and PMX500FI (Fig. 2), also act as antioxidant histone deacetylase (HDAC) inhibitors (HDACi) (Hoffman et al., submitted).47,50,54 noted for effecting favorable epigenetic modulation of the cell survival protein, B cell lymphoma 2 (Bcl-2) with respect to the proapoptotic protein, Bcl-2-associated X protein (BAX) in a BAX/Bcl-2 ratio of 1:2.54,110–113 Again, note the implied potential of drugs that modulate epigenetic pathways.

Chemical structures of butyric acid, ALA, and their corresponding carnitine esters. ALA, α-lipoic acid.
ALA has been extensively researched as a neuroprotectant,114–119 acting on signaling mechanisms through both receptor-mediated pathways and nonreceptor-mediated antioxidant processes in a variety of cell types110,115,116,119,120—including cochlear hair cells.31,121,122 In humans, ALA is a functionally versatile endogenous molecule enzymatically synthesized in mitochondria from octanoic acid. 123 It is a key cofactor in the construction of vital metabolic multienzyme complexes, including pyruvate dehydrogenase and the glycine cleavage system. 123 It is also a strong antioxidant1,119,124,125 and anti-inflammatory109,119,125 agent capable of activating and modulating signal transduction pathways,109,119,126 upregulating the expression of nerve growth factor, and augmenting the conduction velocity of motor nerves. 127
The expression of ∼1% (∼200–250 genes) of the protein-coding human genome is modulated in concert with the Nrf2/ARE signaling pathway.117,128 ALA is a potent activator of Nrf2, a transcription factor encoded by NFE2L2 that helps regulate cellular redox balance and protective antioxidant and phase II detoxification responses in mammals. 50 Dietary antioxidant supplements are commonly sought by patients and caregivers for treating primary mitochondrial disorders.23,65 The role of antioxidants in prevention of age-related hearing loss has been reviewed by Tavanai and Mohammadkhani. 129 In one of the reviewed studies, C57BL/6 mice fed with control diet or diet containing 1 of 17 antioxidant compounds (acetyl-l-carnitine, N-acetyl-l-cysteine (NAC), ALA, carotene, carnosine, coenzyme Q10, curcumin, tocopherol, epigallocatechin-3-gallate, gallic acid, lutein, lycopene, melatonin, proanthocyanidin, quercetin, resveratrol, or tannic acid), ARHL was nearly completely prevented by ALA and coenzyme Q10 and partially by NAC, but not by the other compounds. 130 Unfortunately, this strategy showed no significant benefit in an interventional human study. 131
However, the results from the Polanski and Cruz 131 study may not truly address the ability of antioxidants to prevent ARHL because the design of the study was not directed toward prevention, and damaged cochlear hair cells are not restored by antioxidants. 129 In studies aimed at preventing hearing loss in aged animals, ALA was shown to confer significant hearing preservation.34,108 Similar results between human and animal studies 99 were also observed with the use of l-carnitine—an endogenously synthesized molecule mostly obtained from the diet. 65
NF-κB is a transcription factor that regulates the expression of a variety of genes involved in inflammation and immunity.81,104,105 Sodium butyrate is a well-documented HDAC inhibitor18,27,54,101,105 that has demonstrated anti-inflammatory NF-κB inhibition properties.50,101–105 Butyrate mediates NF-κB activation by rescuing the redox machinery and controlling reactive oxygen species 105 that are highly injurious to hair cells18,132 by suppressing the NF-κB signaling pathways. 105
Although ALA and butyrate are common food and diet supplements that can be safely taken in high doses, their bioavailability is not prolonged or sustained at an effective therapeutic level. 50 Furthermore, a recent Phase I clinical trial in age-related macular degeneration evaluating the safety and tolerability of ALA in 15 subjects, 65 years of age or older, showed that high doses (800–1200 mg) of racemic ALA cannot be tolerated very well by patients. 133 Thus, in the treatment of hearing loss, a need for ALA and butyrate derivatives having more clinically suitable pharmacokinetics is a challenging pharmaceutical objective.
Concluding Remarks
Hearing impairment is a major global health concern; its massive impact seemingly unrecognized until recently, and the affected population largely untreated. Preventing, or at least delaying or reducing, some hearing loss may be possible by avoiding excessive noise exposure and addressing contributory factors such as cardiovascular risk, infectious diseases, neurological disorders, and drug toxicity. However, these interventions will not be sufficient given the sheer magnitude of the problem. Thus, in view of recent advances in our understanding of the underlying mechanistic pathways—both mitochondrial and epigenetic—that govern hearing function, coupled with new drug discovery paradigms that can today be exploited to identify new and effective therapies, the time is ripe to tackle hearing loss with novel medicines. Alcmaeon of Croton remarked that vision and hearing are constructs of the brain. We see and hear in our dreams and in some aspects of disease conditions, such as high fever, schizophrenia, psychosis, or the later stages of dementia, and our dreams may blend into our conscious state immersed in auditory and/or visual hallucinations and delusions. The most common hallucination in schizophrenia is hearing voices.134,135 Finally, readers are directed to Table 2 for a summary of key points related to otologic disorders.
GPCR, G-protein-coupled receptor; LMICs, low- and middle-income countries; NSAID, nonsteroidal anti-inflammatory drug; RP, retinitis pigmentosa; TBI, traumatic brain injury; YLDs, years lived with disability.

Wide ranging chemical structures of potentially ototoxic drugs.
Footnotes
Acknowledgments
We gratefully acknowledge the generous financial support from the MitoCure Foundation and thank Dr. Robert J. Zamboni (Zamboni Chem Solutions, McGill University) for his invaluable advice and helpful discussions in the preparation of this article.
Authors' Contributions
All authors contributed to the writing of this article and agreed to its final content.
Author Disclosure Statement
K.S. owns shares in PhenoMatriX, Inc. K.K. and W.H.M. consult with and/or serve on the boards of various biotechnology and pharmaceutical companies from time to time, where they may receive compensation including stock options, and they receive compensation from ShangPharma Innovation, Inc., a healthcare venture capital firm. For all other authors, no competing financial interests exist.
