Abstract
Adenosine is a nucleoside displaying various biological effects via stimulation of four G-protein–coupled receptors, A1, A2A, A2B, and A3. Adenosine also modulates voltage-gated (Kv) and small conductance calcium-activated (SKCa) potassium channels. The effect of these potassium channels on the expression of adenosine receptors is poorly understood. We evaluated the action of BgK (a natural Kv channel blocker) and Lei-Dab7 (a synthetic SKCa channel blocker) on the expression of adenosine A2A receptors (A2AR) in Jurkat human T cells. We found that Lei-Dab7, but not BgK, increased the maximal binding value of the tritiated ligand ZM241385 to A2AR in a dose-dependent manner (+45% at 5 nM; +70% at 50 nM as compared to control). These results were further confirmed by Western blotting using a specific monoclonal antibody to human A2AR. The ligand affinity-related dissociation constant and A2AR mRNA amount were not significantly modified by either drug. We suggest that modulation of SKCa channels can influence membrane expression of A2AR and thus has a therapeutic potential.
Introduction
Adenosine is a ubiquitous nucleoside produced by ATP hydrolysis that is released by most cells into extracellular spaces where it binds G-protein–coupled receptors, namely, A1, A2A, A2B, and A3.1–4 These receptors display various primary sequences and pharmacological properties. 5 Adenosine modulates cAMP production of target cells 6 and consequently the cAMP-dependent release of neurotransmitters. 5 Activation of A1 and A3 subtypes inhibits cAMP production, 7 but activation of A2A and A2B subtypes enhances cAMP production,8,9 facilitating the cAMP-dependent neurotransmitter discharge. 10 Although adenosine and adenosine receptor agonists inhibit voltage-activated calcium channels (especially N and L types) of smooth muscle cells through the βγ G-protein complex leading to vasodilation and hypotension,11–15 they also modulate potassium channels.16–20 These potassium channels are subdivided into voltage-gated (Kv), calcium-activated (KCa), inward rectifier (including those that are ATP sensitive, KATP), and two-pore channels.21–24 KCa channels are further classified as big (BKCa), intermediate (IKCa), and small (SKCa) conductance channels.25–27 These channels are activated by increased cytoplasmic concentration of Ca2+,28–33 BKCa channels also being voltage sensitive. SKCa channels are subdivided into three classes (SKCa1–3).34,35 Most potassium channel subtypes are modulated by adenosine through activation of adenosine receptors.5,15 In contrast, vasodilation of arteries induced by CGS21680, an adenosine A2A receptor (A2AR) agonist, 36 is inhibited by a cocktail of nonspecific potassium channel blockers, 37 and smooth cell relaxation induced by activation of A2AR occurs partly through the modulation of KCa channels.38,39 Furthermore, iberiotoxin, an Eastern Indian red scorpion (Buthus tamulus) toxin, which is a blocker of BKCa channels,40,41 attenuated the release of nitric oxide by endothelial cells that occurs during A2AR activation, suggesting that both BKCa and SKCa are coupled to A2AR. 42 CGS21680 also induces enhanced mRNA expression of Kv1.3 and increased production of Kv protein, while the A2AR antagonist ZM241385 inhibits these effects. 43 Among SK channels, SKCa1 and SKCa2 are involved in the physiological effects of CGS21680. 44 Numerous studies dealing with the consequences of A2AR activation on potassium channel modulation have been reported, but little is known about the influence of potassium channels modulation on the expression of A2AR. The aim of this study was thus to evaluate the influence of Kv and SKCa channel blockers on A2AR expression in a human T-cell line as measured by ligand binding saturation curves, Western blots and mRNA amount.
Materials and Methods
Materials
BgK, a potassium-channel toxin from the sea anemone Bunodosoma granulifera, an inhibitor for Kv1 channels, was purchased from Latoxan®, (Valence, France). Caffeine was from Sigma-Aldrich (Saint Quentin Fallavier, France). [3H]-ZM241385 was from Tocris Cookson (Bristol, United Kingdom; 23.5 mCi/mmol), and ZM241385 was from Fischer Bioblock Scientific (Ilkirsch, France). Lei-Dab7 was synthesized by the stepwise solid-phase method 45 by using a peptide synthesizer (Model 433A, Applied Biosystems Inc., Foster City, CA). The side-chain protecting groups used for trifunctional residues were: 2,2,5,7,8-pentamethylchromane-6-sulfonyl for Arg and homoarginine; tert-butyloxycarbonyl for Orn, Lys, and homolysine; and 1-(4,4-dimethyl-2,6-dioxocyclohex-1-yliden)-3-methylbutyl for Dab and diaminopropionic acid (Dapa). The reduced peptides were stirred under air at 1 mM in 0.2 M Tris-HCl buffer, pH 8.3 for 48 h at 25°C to allow folding/oxidation. The folded/oxidized toxins and their structural analogs were purified to homogeneity by reverse-phase high-pressure liquid chromatography onto a C-18 ODS Aquapore column (PerkinElmer Life Sciences, Waltham, MA).
Cell membrane preparation
The Jurkat human T-cell line was cultured in RPMI-1640 medium supplemented with 10% heat-inactivated fetal calf serum and 1% glutamine at 37°C with 5% CO2 at about 1×106 cells/mL. Cells were cultured for 24 h alone and in the presence of BgK or Lei-Dab7 (both at 1, 5, and 50 nM) and caffeine (at 50 and 300 nM). Cells were centrifuged, and 9×109 cells were suspended in 4.5 mL of binding buffer (50 mM Tris-HCl, 10 mM MgCl2, pH 7.4), homogenized for 30 s using an Ultra Turrax (Staufen, Germany), and immediately used in binding assay. Protein concentration was determined using a Beckman Synchron LX apparatus (Villepinte, France).
Binding assay
The methodology described by Varani et al. 46 was used with some modifications: 47 saturation binding experiments were performed in triplicate, using the selective A2AR ligand [3H]-ZM241385, by incubating 200 μL of cell membrane homogenates for 90 min at 4°C with increasing concentrations of tritiated ligand (0.5–6 nM). Bound and free signals were separated using vacuum filtration of the sample through Whatman GF/C glass-fiber filters. Cold binding buffer (1 mL) was added to the sample before filtering. The filter was extensively washed and the signal associated with the filter was determined using a Beckman LS-1800 liquid scintillation spectrometer. A weighted, nonlinear, least-square curve-fitting program, Graph Pad Prism (Graph Pad Software Inc., San Diego, CA) was used for maximal binding (Bmax) and dissociation constant (KD) determination. Nonspecific binding of [3H]-ZM241385 was measured in the presence of an excess (10 μM) of unlabeled ligand.
Western blotting
Cells (0.5×106) were solubilized with 30 μL of 62.5 mM Tris-HCl buffer, pH 8.3, containing 2% sodium dodecyl sulfate, 10% glycerol, 0.01% bromophenol blue, and 5% mercaptoethanol. Solubilizates were sonicated for 10 min at 47 kHz and loaded onto a 12% acrylamide, 60 mm×90 mm, 1.5-mm-thick minigel. Cell proteins were submitted to standard electrophoresis procedure. Separated proteins in the gel were directly electrotransferred onto a 0.45-μm polyvinylidene difluoride membrane. Blotted membrane was saturated with nonfat dried milk and incubated with Adonis, a mouse monoclonal antibody to human A2AR. 48 Anti-glyceraldehyde 3-phosphate dehydrogenase (anti-GAPDH) mouse monoclonal antibody (Clone GAPDH-71.1, Sigma, St. Louis, MO) was added to Adonis as loading control for normalizing blot results to GAPDH. Blots were visualized by horseradish peroxidase–labeled anti-mouse antibodies and enhanced chemiluminescence substrate (SuperSignal West Femto, Pierce Biotechnology, Rockford, IL) using a Kodak Image Station 440CF (Eastman Kodak Company, Rochester, NY). The staining intensities of the bands were measured by densitometry using ImageJ 1.42q software (National Institutes of Health, Bethesda, MD) and results were expressed as the pixel ratio of the A2AR/GAPDH bands.
Quantification of A2AR mRNA
Total RNA from Jurkat cells was extracted with the MagAttract RNA tissue Mini M48 kit (Qiagen, Hilden, Germany) according to the manufacturer's recommendations (Bio-Robot M48). cDNA was prepared from 250 ng of total RNA. Real-time polymerase chain reaction was performed with a Light-Cycler (Roche, Neuilly sur Seine, France) using the Light-Cycler FastStart DNA Master plus SYBR green I kit. The 18S rRNA primers were 5′-GGT-GAC-GCG-GAA-TCA-GG-3′ (forward) and 5′-GCT-GCT-GGC-ACC-AGA-C-3′ (reverse), generating a 218-bp DNA fragment. The A2AR primers were 5′-TCC-CAT-GCT-AGG-TTG-GAA-CA-3′ (forward) and 5′-GGA-AGA-TCC-GCA-AAT-AGA-CAC-3′ (reverse), generating a 191-bp DNA fragment. The amount of mRNA encoding A2AR was expressed in arbitrary units (AU) defined as the amount ratio of A2AR mRNA/18S rRNA.
Statistical analysis
A two-way analysis of variance (ANOVA) was used for intergroup comparisons. A p-value<0.05 was considered to be significant.
Results and Discussion
SKCa channels are expressed in various cells of the central nervous49,50 and cardiovascular systems51,52 where they play a key role in all excitable cells. SKCa channels are involved in the “after hyperpolarization process,” 53 a period in which the membrane potential is negative to the normal resting potential and then slowly returns to baseline. The activation of SKCa channels causes membrane hyperpolarization, which protects excitable cells against repetitive action potential. 54 SKCa channels are also expressed in hematopoietic cells, where they modulate reactive oxygen species by neutrophils. 55 In lymphocytes, several types of potassium channels (especially Kv1.3 and IKCa1) were reported with particular roles in mitogen- and antigen-specific proliferation, cell volume regulation, and apoptosis.56,57
Lei-Dab7 is a synthetic analogue of Leiurus quinquestriatus scorpion toxin, which is a highly selective inhibitor of SKCa2. 58 It was previously reported that Lei-Dab7 decreases the analgesic effects of CGS21680, an A2AR agonist, in a dose-dependent manner. 44 This suggests that SKCa2 mediates, at least in part, the biological properties of CGS21680. In order to determine the impact of potassium channel blockers on the amount of and the ligand affinity for A2AR expressed on the cell membrane, Bmax and KD values were estimated, respectively, from saturation curves of [3H]-ZM241385 binding to Jurkat T cells. Representative dose–response curves performed with and without 50 nM of Lei-Dab7 (a SKCa channel blocker), BgK (a Kv channel blocker), and caffeine (used here as a positive control) are shown in Figure 1. Lei-Dab7 treatment increased the Bmax value of A2AR in a dose-dependent manner (+45% at 5 nM and +70% at 50 nM as compared to control), BgK did not significantly modulate Bmax values at the concentrations tested, and caffeine increased Bmax values by 77% and 104%, as compared to controls, at 50 and 300 nM, respectively (Fig. 2A). The specificity of these results was further confirmed by Western blotting using Adonis, a specific monoclonal antibody directed to the second extracellular loop of the human A2AR. 48 Figure 2B shows that Adonis revealed similar expressions of A2AR in cells incubated with or without 50 nM BgK (mean pixels ratio of A2AR/GAPDH bands±SD of triplicates: 0.13±0.08 and 0.09±0.05, respectively) and higher expressions of A2AR in cells incubated with 50 nM Lei-Dad7 and caffeine (0.63±0.11 and 0.76±0.16, respectively). The mean KD value (±SD of triplicates) of ZM241385 for A2AR in the presence of 50 nM of BgK, Lei-Dab7, and caffeine was 5.2±0.9, 3.2±2.2, and 6.3±2.7 nM, respectively, as compared to control (without drug): 3.7±1.1 nM. The amount of mRNA encoding A2AR in the presence of 50 nM of BgK, Lei-Dab7, and caffeine was 17±0.9, 17.2±0.7, and 18±0.9 AU, respectively, as compared to control (17.6±0.6 AU). The statistical analysis of these results indicated that both the KD value of ZM241385 for A2AR and the amount of mRNA encoding A2AR were not significantly modified by Lei-Dab7, BgK, and caffeine treatment.
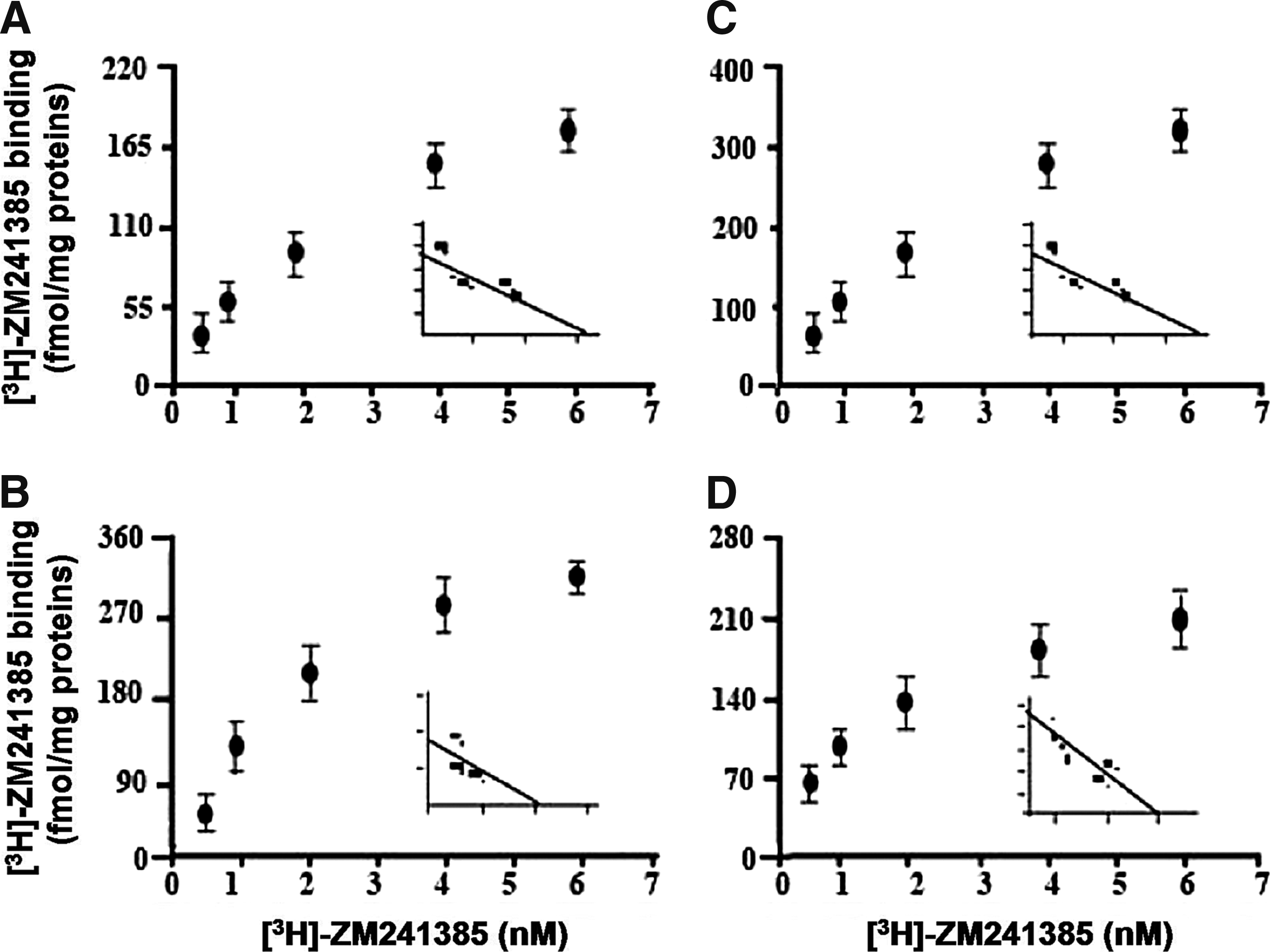
Representative dose-response curves of [3H]-ZM241385 binding to adenosine A2A receptor (A2AR) of Jurkat T-cell membrane. Cells cultured without drug
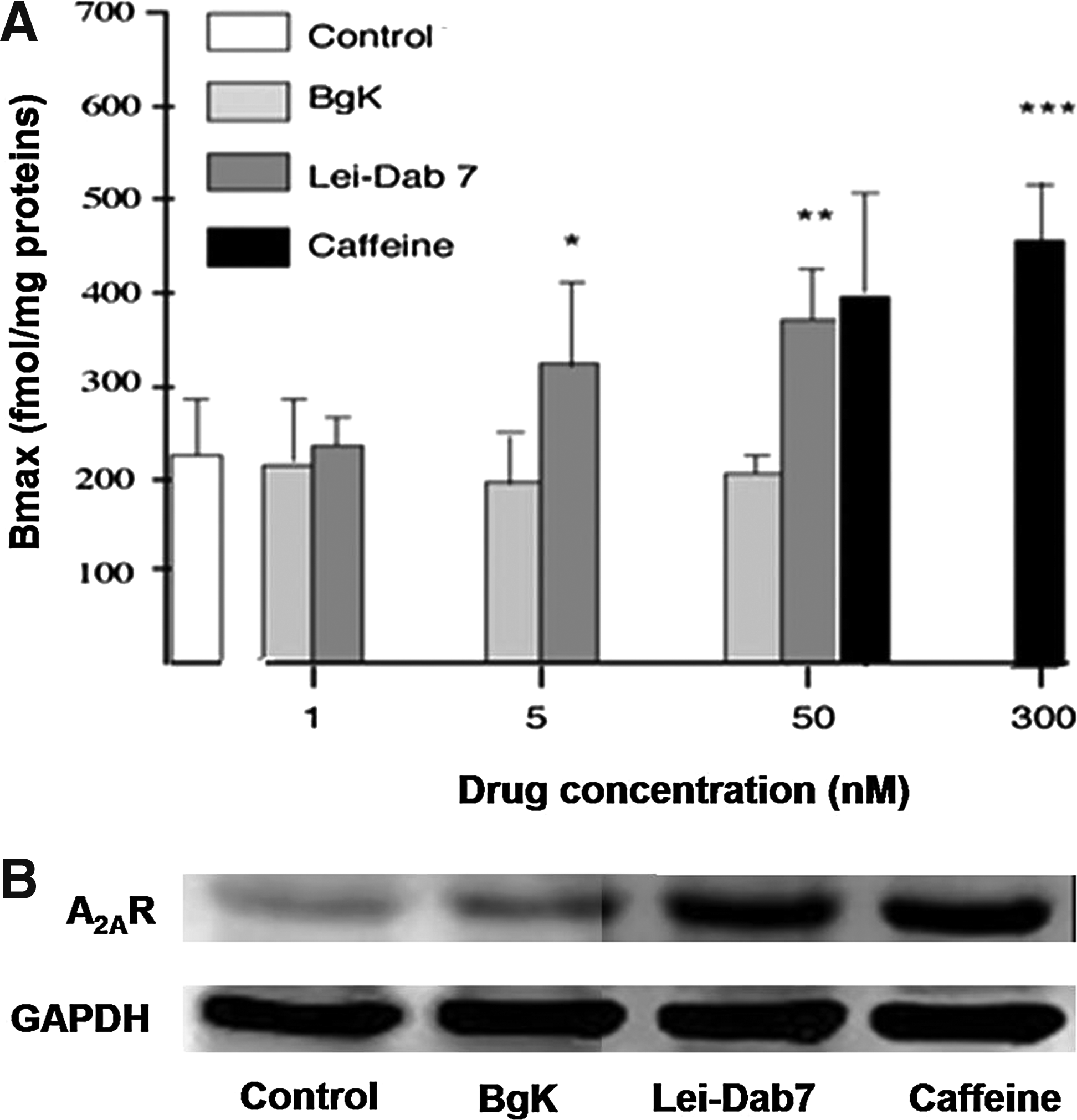
Here, we found that inhibition of SKCa2 enhances A2AR expression at the cell membrane in a dose-dependent manner. This increase mimics those obtained using caffeine, which is known to induce an up-regulation of A2AR.48,59 The specificity of our results was confirmed by using BgK, which blocks Kv1 potassium channels60,61 and did not modify A2AR expression even at high (50 nM) levels. The A2AR increase we observed did not result from increased A2AR mRNA cellular expression; however, a lack of change in mRNA expression did not exclude an increase in A2AR protein synthesis. Also, it was previously reported that glibenclamide, a KATP channel blocker, modifies the affinity of CGS21680 for A2AR. 38 Here, the potassium channel blockers Lei-Dab7 and BgK, like caffeine, did not change the KD of ZM241385 for A2AR and, consequently, did not affect the A2AR ligand affinity.
A2AR are coupled to G-proteins to activate adenyl cyclase, resulting in increased cAMP production that, in turn, can activate protein kinase A (PKA) 62 and stimulate potassium channels. 63 This signaling pathway was reported in vasodilatation of rat preglomerular microvessels 64 to be coupled to a release of epoxyeicosatrienoic acids, which were also reported to have cardioprotective effects by modulation of mitochondrial ion channels like KATP and signaling through phosphoinositide 3-kinase pathways. 65 In immune cells, A2AR-mediated activation of cAMP-dependent PKA induces phosphorylation of cAMP-responsive element-binding proteins, which activates numerous genes transcription via their promoters and consequently alters cell growth and cytokines expression. 66 Another downstream target of PKA is the L-type calcium channel that allows Ca2+ to enter into T cells for their activation and function. 67 Alternately, in PC12 cells, prolonged A2AR stimulation activates novel protein kinase C (PKC) through a pertussis toxin–sensitive G-protein and phosphorylates and suppresses the type VI adenyl cyclase, which causes negative feedback regulation of the cAMP signal induced by the A2AR. 68 The signaling pathways of the A2AR appear complex and interconnected. It is not surprising that A2AR interferes with potassium channels and calcium cellular fluxes and vice versa. T-cell activation requires sustained calcium signaling provided by a concerted interaction between potassium channels and the calcium release-activated calcium channels. 69 Thus, by acting via A2AR to inhibit stimulated T cells, potassium channels can be viewed as potential targets for immunosuppression.70,71
The present results suggest that the blockade of SKCa channels in Jurkat T cells may influence the membrane expression of A2AR by a mechanism that remains to be revealed. We hypothesize that blockage of SKCa channels increases intracellular calcium levels ([Ca2+]i), inducing activation of calcium-dependent protein kinases that, in turn, increase the expression of cell-membrane proteins such as A2AR. The modulation in A2AR expression induced by SKCa channels may help to fine-tune the immunosuppressive signaling in T cells and, in this respect, has an interesting therapeutic potential. However, this mechanism needs to be confirmed by further functional investigations in immune as well as excitable neuronal and cardiac cells.
Footnotes
Acknowledgments
Y. By is a recipient of grant from the Assistance Publique, Hôpitaux de Marseille, France.
