Abstract
A balance between self-renewal and differentiation of spermatogonial stem cells (SSCs) is required to maintain sperm production throughout male life. The seminiferous epithelium is organized into stages of spermatogenesis based on the complement of germ cell types within a tubular section of the testis. The stages exist in close physical proximity and foster diverse phases of germ cell development despite exposure to a similar endocrine milieu that supports coordinated spermatogenesis. The objective of the current study was to identify the population dynamics of SSCs in vivo. We hypothesized that SSC populations and their niches are specifically distributed across the mature seminiferous epithelium in the mouse testis. To test this hypothesis, we conducted stem cell transplantation of germ cells obtained from stage-specific clusters of seminiferous tubules representing areas of high responsiveness to follicle-stimulating hormone (IX–I), androgen (II–IV), and retinoid (V–VIII) signaling. Similarly, we analyzed the expression of genes linked with SSC activity in these groups of stages. No stage-specific differences in the colonization efficiency or the colony number were detected after SSC transplantation, indicating that SSCs are equally distributed across all stages of the seminiferous tubule. In contrast, SSCs obtained from donor stages IX–IV established larger donor-derived colonies due to increased colony expansion. SSCs originating from different stages have varying degrees of stem cell activity in vivo, a notion consistent with Gdnf, Ret, and Bcl6b expression data. These results support the conclusion of a stage-specific, microenvironment-regulating SSC self-renewal and suggest the presence of a transit-amplifying population of undifferentiated spermatogonia in vivo.
Introduction
Adult stem cells are organized in specialized microenvironments called niches that are composed of somatic cells and extracellular matrix components that provide intrinsic and extrinsic factors involved in regulating cell fate decisions. A coordinated balance between self-renewal and differentiation is required to meet the demands of tissue function and maintenance. Spermatogonial stem cells (SSCs) are responsible for continual sperm production in mammals, and are the only adult stem cell population capable of transmitting genes to offspring. SSCs exist in seminiferous tubules of the testes and are in close proximity to somatic Sertoli cells that are involved in supporting their function. In fact, Sertoli cells are equally capable of producing paracrine factors that either contribute to self-renewal of SSCs, in the case of glial cell line-derived neurotrophic factor (GDNF)1,2 and basic fibroblast growth factor (FGF2), 3 or stimulate their differentiation via the actions of activin and bone morphogenetic protein 4. 4 This signaling coordinates a delicate balance between self-renewal and differentiation of SSCs that is required to maintain sperm production throughout life in males.
SSCs are a population of undifferentiated spermatogonia capable of self-renewal and producing donor-derived spermatogenesis in sterile recipients after transplantation.5,6 Although the activity of cells making up the undifferentiated spermatogonial population continues to be defined, single spermatogonia (As) are suggested to be SSCs that either self-renew or give rise to paired spermatogonia (Apr) connected by cytoplasmic bridges. Apr spermatogonia follow a clonal progression and differentiate into chains of 4, 8, and potentially 16 A-aligned (Aal) spermatogonia before becoming A1 spermatogonia. However, the identification of molecules and sources of intrinsic and extrinsic factors that regulate SSC cell fate remains incompletely explained due to the complex cell biology of the testis and the lack of bona fide SSC markers. 7 Thus, germ cell transplantation is the only reliable means to investigate factors that regulate in vivo SSC homeostasis. 8
Morphological studies have suggested a nonrandom distribution of undifferentiated spermatogonia with the interstitial compartment. 9 This area of testis contains blood vessels, lymphatics, and testosterone-producing Leydig cells. Similarly, researchers using a transgenic mouse expressing green fluorescent protein (GFP) under the control of a neurogenin 3 (NEUROG3) regulatory sequences suggest that SSCs exist within a vascular-associated niche. 10 Interestingly, the preferential localization of undifferentiated spermatogonia in these studies has been linked with specific stages of the mature seminiferous epithelium. This leads to the question of whether a microenvironment enriched for SSCs and their respective niches are associated with particular stages or regions of the mature seminiferous epithelium in the testis.
The mature seminiferous epithelium is organized into stages of spermatogenesis based on the presence and precise complement of germ cell types within a tubular section of the testis, including (1) spermatogonial cell type present, (2) specific phase of meiosis observed, and (3) step of spermatid development.11,12 This unique organization allows for precise regulation of spermatogenesis, such that gene products can orchestrate specific events important for highly differentiated germ cells, without impairing less-mature germ cells during maturation or development. For instance, gradients of mitogenic and differentiation signals are found in a spatial and temporal manner that are directly correlated to cellular events such as mitosis, meiotic entry, and spermiation, so that efficient sperm production can occur. For example, regions of high responsiveness to follicle-stimulating hormone (FSH; stages IX–I), 13 androgen (stages II–IV), 14 and retinoic acid (stages V–VIII) 15 have been described. Each portion of the seminiferous tubule will progress through all stages of spermatogenesis over time, and each stage of the cycle follows in an orderly sequence along the length of the tubule, a phenomenon called the spermatogenic wave, which is thought to initiate at postnatal day 10 in response to androgen and retinoid signaling in Sertoli cells. 16 It is known that Aal spermatogonia differentiate into A1 spermatogonia during stages VII–VIII of the epithelial cycle 17 concurrent with spermiation12,18,19 and high responsiveness to retinoid signaling. 15
No functional studies have been used to identify the specific location and distribution of SSCs in the seminiferous tubule. Therefore, this study was designed to isolate and characterize specific regions of the mature seminiferous epithelium of the testes to determine if a microenvironment enriched for SSCs is associated with particular stages or regions of the seminiferous epithelium. To achieve this objective, we isolated clusters of seminiferous tubules using transillumination-assisted microdissection to obtain segments of tubules that represent areas of high responsiveness to FSH (IX–I), androgen (II–IV), and retinoid (V–VIII) signaling. We hypothesized that SSC populations are static and are randomly distributed across the mature seminiferous epithelium in the mouse testis. To test this hypothesis, we (1) evaluated gene expression of SSCs and SSC-niche–associated factors within specific stages using quantitative real-time polymerase chain reaction (PCR), and (2) assayed the SSC activity and SSC proliferation kinetics of SSCs obtained from these stage-specific clusters with the use of germ cell transplantation.
Materials and Methods
Animal care and treatments
All animal experiments were approved by the Washington State University Animal Care and Use Committee and were conducted in accordance with the Guiding Principles for the Care and Use of Research Animals of the National Institutes of Health. Animals were housed in a standard animal facility with and provided ad libitum access to food and water.
Stage-specific tubule dissection of the adult seminiferous epithelium
Stage-specific seminiferous tubule clusters were collected from adult (postnatal 8–12 weeks) mice using the transillumination-assisted microdissection technique as described previously. 12 Briefly, donor testes were removed, placed in Hanks buffered salt solution, and then detunicated. Segments of tubule clusters that represent areas of high responsiveness to FSH (IX–I), androgen (II–IV), and retinoid (V–VIII) signaling were harvested and placed in an alpha-minimum essential medium (α-MEM). Tubule segments representing all stages (I–XII) in normal adult mice were used as a positive control. Randomly selected tubule segments from mice treated with busulfan (intraperitoneally; 40 mg/kg body weight) to eliminate endogenous germ cells were harvested as a negative control. Transgenic B6.129S7-GtRosa26 (Rosa26) mice from Jackson Laboratory (Bar Harbor, ME) that express β-galactosidase in all testicular germ cells were used as donors for all transplantation experiments. After dissection, tubule clusters from C57Bl/6 (C57) mice were left intact and processed for RNA isolation of cDNA production using TRIzol (Life Technologies, Grand Island, NY). At least three donors were used for each tubule cluster evaluated (n>3), and multiple independent dissection replicates were performed for each donor.
Preparation of donor germ cells
Rosa26 seminiferous tubule clusters were enzymatically digested as described by McLean and coworkers, 8 with minor modifications. Briefly, tubule clusters were transferred to a dish containing a digestion medium, which consisted of 0.18 mg/mL trypsin (GibcoBRL, Bethesda, MD), 0.16 mg/mL collagenase type IV (Sigma, St. Louis, MO), and 0.6 mg/mL DNase (Sigma) in an α-MEM, pH 7.4, and incubated for 10 min at 37°C. After incubation and addition of fetal bovine serum (10% v/v), tubule segments were dispersed by gentle pipetting, and the testis cell suspension was centrifuged at 600 g for 7 min at 4°C. After several washes, donor cells were resuspended in an MEM containing 0.03% trypan blue (GibcoBRL) to obtain germ cells for transplantation at a concentration of 107 cells/mL.
Germ cell transplantation and analysis of recipient testes
To determine stem cell activity, ∼7.8×104 donor cells were transplanted into seminiferous tubules of busulfan-treated immunologically compatible 129SvCP (129)×C57BL/6 F1 hybrid recipient mice. Recipient mice were sacrificed 8 weeks after transplantation, and the colonization efficiency of Rosa26 SSCs was determined by counting the number and measuring the length of blue colonies after staining with 5-bromo-4-chloro-3-indoyl β-D-galactoside (Xgal).
Quantitative real-time reverse transcriptase–PCR analysis
Tubule clusters obtained from adult C57 mouse testis tissue were homogenized in TRIzol (Life Technologies) according to the manufacturer's protocol. RNA concentration and purity were determined using a Nanodrop ND-1000 spectrophotometer (Thermo Fisher Scientific, Schwerte, Germany), and only samples with a 260/280 ratio of ≥1.8, but ≤2.1, were selected for analysis of gene expression as described. cDNA was prepared from 1 μg of total RNA, and real-time PCR was performed as previously described 21 using an iCycler iQ (Bio-Rad, Hercules, CA) detection system. Samples were assayed in duplicate, and comparisons were made by normalizing the expression of the target gene to that of ribosomal protein S2 using the Q-Gene method. 22 A list and description of TaqMan probes used in the current study are shown (Table 1). The PCR reaction mixture contained 100 ng cDNA, 12.5 μL of 2× TaqMan Universal PCR Master Mix, 1.25 μL 20× TaqMan gene-specific probes (Table 1), and 9.25 μL of nuclease-free water for a total volume of 25 μL. Data are presented as fold change (mean expression levels±standard error of the mean [SEM]) relative to the expression levels of seminiferous tubules with all stages of the mature seminiferous epithelium present.
TaqMan Polymerase Chain Reaction Probes Used for Quantitative Real-Time Reverse Transcriptase–Polymerase Chain Reaction Assays
Immunohistochemistry methods
Immunohistochemistry was performed on paraffin-embedded testis tissue sections (5-μm thickness) obtained from normal and busulfan-treated (intraperitoneally; 40 mg/kg body weight) intact mice using the avidin–biotin complex method and sodium citrate antigen retrieval (pH=6.0), as described. 23 After quenching endogenous peroxidase activity and washing slides in phosphate-buffered saline (PBS), slides were incubated for 30 min in a blocking solution (10% normal serum v/v in PBS). After blocking, tissue sections were incubated Armenian hamster anti–promyelocytic leukemia zinc finger (PLZF) monoclonal antibody (1:600; a gift from the Hobbs Lab) and rabbit anti-RET-phosphoY1062 antibody (1:200) for 12 h at 4°C. After washing, sections were incubated with biotinylated secondary antibodies for 60 min at 25°C, then washed, and incubated with a streptavidin–horseradish peroxidase substrate. Immunoreactivity was detected after a 5-min incubation in 3,3′-diaminobenzidine, and sections were counterstained with hematoxylin. As a negative control, serial sections were processed without primary antibody.
Statistical analysis
All datasets are presented as the mean±SEM, and differences between tubule clusters were considered significant at p<0.05. The effect of seminiferous tubule microenvironment on stem cell activity and expression of SSC-niche–associated factors were analyzed using a one-way analysis of variance, and pairwise comparisons were evaluated with a Dunn's multiple-range test. Nonparametric data were analyzed using a Kruskal–Wallis test.
Results
Isolation and germ cell transplantation of stage-specific segments of the seminiferous epithelium obtained by transillumination-assisted microdissection
Stage-specific clusters of the adult seminiferous tubules were isolated based on the transillumination pattern described by Kotaja et al. 20 These groups of tubules were (1) stages IX–I, (2) stages II–IV, and (3) stages V–VIII. Transillumination can be employed to dissect seminiferous tubules based on stages due to the different germ cells present at each stage. The different morphology of differentiating germ cells, primarily spermatids, alters the amount of light passing through the tissue such that consistent sections can be obtained (Supplementary Fig. S1). Seminiferous tubules with branches were not used to avoid transitional regions without segmental stages (Supplementary Fig. S1). After dissection, stage-specific tubule segments from adult Rosa26+ mice were digested to obtain a single-cell suspension of germ cells for transplantation into the testes of infertile, busulfan-treated recipient mice. The cell suspensions obtained from each group of seminiferous stages contained cells of consistent size and morphology (Supplementary Fig. S1). Germ cell transplantation provides the ability to directly determine the SSCs in a cell suspension that are capable of generating donor-derived spermatogenesis. Eight weeks after transplantation, recipient mice were sacrificed, and testes were stained with Xgal, and the number of donor-derived stem cell colonies was determined. Germ cells obtained from stage-specific clusters (IX–I, II–IV, and V–VIII) colonize recipient testis and generate donor-derived spermatogenesis at similar rates (Fig. 1). These data indicate that adult SSC populations and their niches are equally distributed throughout the seminiferous epithelium.

Germ cell transplantation analysis of stage-specific SSCs obtained from adult seminiferous tubules of mice.
The colonization efficiency of testicular cells isolated from randomly selected seminiferous tubules was not different from the germ cells isolated from the selected stage groups. We also transplanted testicular cells isolated from busulfan-treated mice as a second control. Busulfan treatment eliminates differentiating germ cells from the testis, resulting in seminiferous tubules devoid of germ cells, and donor cells for transplantation were obtained 4 weeks after busulfan treatment (Supplementary Fig. S1). Not surprisingly, the colonization efficiency of cells obtained from busulfan-treated donors was significantly impaired compared to cells from randomly selected seminiferous tubules and groups of stages, respectively (Fig. 2). Although busulfan treatment eliminates many SSCs, some do survive the treatment and can initiate donor-derived spermatogenesis after transplantation. However, the SSC recovery we observed was lower than reported results. 24

Germ cell transplantation analysis of stage-specific SSCs obtained from randomly selected seminiferous tubules of normal and busulfan-treated donor mice.
The extent of colony expansion was also evaluated to ascertain the proliferation kinetics and regeneration potential of SSCs obtained from specific groups of stages within the mature seminiferous tubule. Colony expansion of donor-derived spermatogenesis was significantly greater for donor cells obtained from stages IX–I and II–IV when compared to stages V–VIII (Fig. 3). Therefore, specific stages within seminiferous tubules support SSCs or undifferentiated spermatogonia, such that the colonization potential of these cells varies between stages. These data suggest that a dynamic SSC niche may be present in the testis that is important for regulating the biological activity of SSCs and their ability to proliferate and self-renew. Busulfan treatment had no effect on the expansion rate of SSCs after transplantation, despite significantly reducing their numbers (data not shown).

Analysis of SSC-niche–associated factors in stage-specific segments of the seminiferous epithelium
The differential germ cell-derived colony expansion observed after transplantation of SSCs from different stages of the seminiferous epithelium suggests that the SSC niche or regulation may vary in different stages. To test this hypothesis, we investigated the expression of SSC-niche–associated factors and potential regulation of these factors. Germ cell differentiation and thus the stages of the seminiferous epithelium are primarily regulated by FSH, testosterone, and retinoic acid. Our first step was to determine the expression of Fshr and Ar mRNA transcripts using quantitative real-time reverse transcriptase–PCR in the groups of stages we dissected for germ cell transplantation to determine the prevailing regulatory factor for each group of stages. Although not statistically significant, Fshr expression was biologically highest in stages IX–I, whereas AR expression was biologically highest in stages II–IV (Fig. 4A). Stra8 is induced by retinoic acid, so the expression of this gene was used to evaluated retinoic acid signaling in the groups of stage. The expression of Stra8 was biologically higher in stages V–VIII when compared to the other groups of stages (Fig. 4). High cellular responsiveness to retinoic acid is associated with stages in which SSCs have lower colony expansion potential (Fig. 3).

Gene expression analysis of endocrine and SSC-niche-associated factors in stage-specific segments of the seminiferous epithelium. The mean expression values of
A link between SSC homeostasis associated with SSC colony expansion and endocrine regulation of the seminiferous epithelium is lacking. Thus, we investigated the expression of SSC-niche–associated factors produced by germ and Sertoli cells in stage-specific clusters of the adult seminiferous epithelium. The GDNF signaling pathway, including its receptors RET and GFRA1, supports the maintenance and self-renewal of SSCs both in vitro and in vivo.1,25 The mRNA expression of Gdnf was significantly higher in stages IX–I and II–IV of the seminiferous epithelium compared to stages V–VIII (Fig. 4). Ret expression was significantly lower in stages V–VIII of the seminiferous epithelium compared to all stages (not shown), but not when compared to stages IX–I and II–IV (Fig. 4). No stage-specific expression pattern was observed for Gfra1 (Fig. 4).
To determine specific locations of the seminiferous tubule that may be associated with cell fate decisions and mechanisms directing SSC homeostasis in vivo, we assayed the mRNA expression of intrinsic regulators of SSC self-renewal, including B-cell CLL/lymphoma 6 member B protein (BCL6B) and nanos homolog 2 (NANOS2). The expression of Bcl6b was significantly higher (p<0.01) in stages IX–I when compared to other tubule clusters (Fig. 4). Specifically, a stage-specific decline in Bcl6b expression was detected between tubule segments obtained from stages II–IV and V–VIII. No stage-specific mRNA expression patterns were observed for Nanos2 (Fig. 4).
Busulfan treatment significantly reduces the number of differentiating germ cells and SSCs in the testis (Fig. 2). In addition, Sertoli and germ cell interactions are disrupted after busulfan treatment, causing the loss of seminiferous epithelial stage organization. We investigated how the loss of germ cells and stage organization affected the expression of the SSC niche factors to evaluate the stage-specific SSC colony expansion. Interestingly, Ret expression was not different in the testes of busulfan-treated mice compared to stages I–XII from untreated controls (Fig. 5). In contrast, expression of Bcl6b and Gfra1 was significantly lower in busulfan-treated samples compared to controls (Fig. 5). Gdnf expression was significantly higher in the seminiferous tubules of busulfan-treated animals compared to controls (Fig. 5). The similarity of the Ret expression in the seminiferous tubules of busulfan-treated and control samples suggests that somatic cells express RET. Immunohistochemical analysis confirmed the Sertoli cell expression of RET (not shown) and activated p-RETY1062 (Ret phosphorylated on Y-1062) in testis tissue of normal (Fig. 5) and busulfan-treated (Fig. 5) mice. These data suggest that GDNF signaling is active in regulating the SSC niche through both paracrine and autocrine mechanisms.
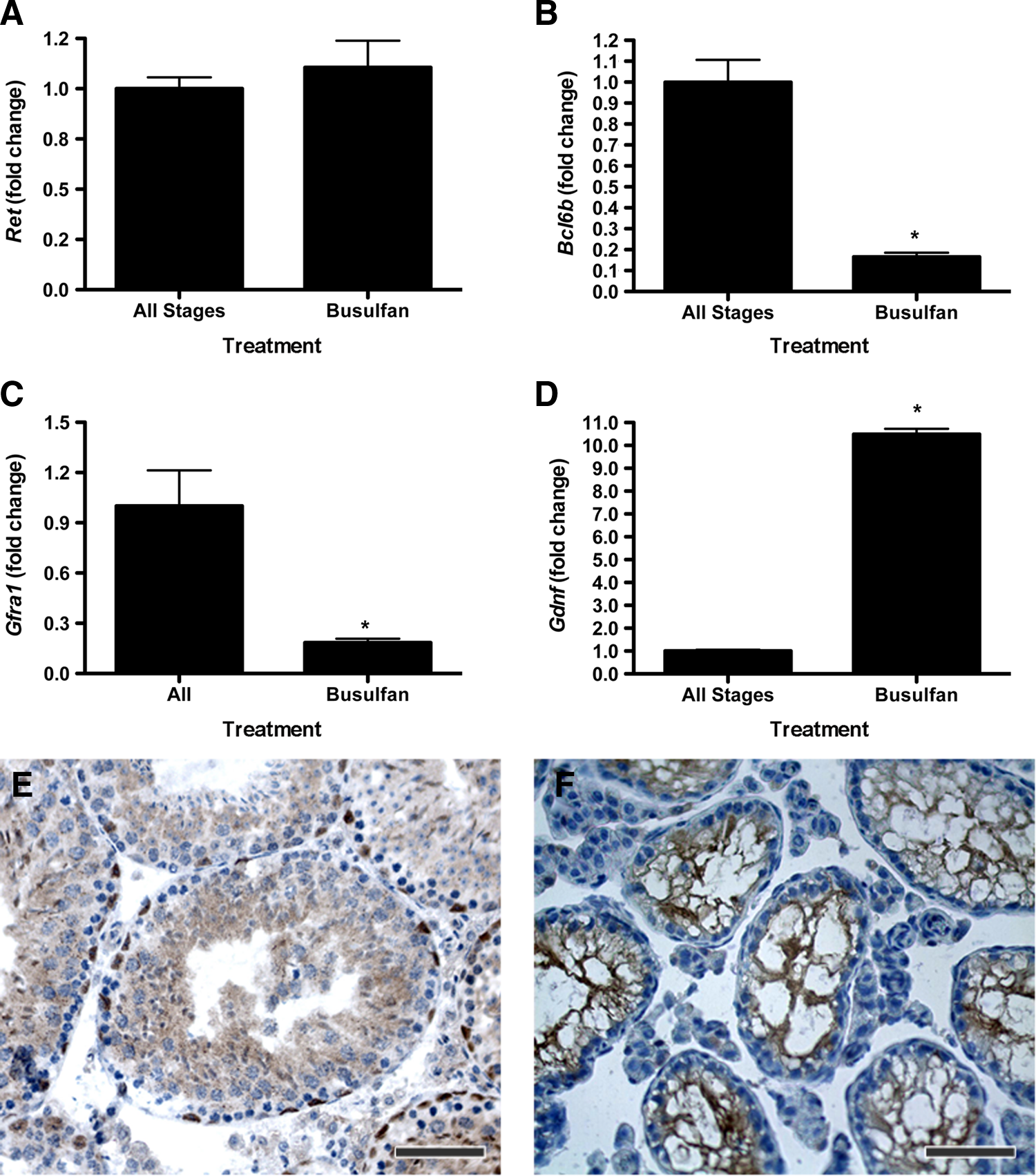
Busulfan treatment eliminates stages of the seminiferous epithelium and germ cell–Sertoli cell interactions altering expression of SSC niche factors. Real-time reverse transcriptase–polymerase chain reaction analysis was performed with RNA from busulfan-treated testes and randomly selected seminiferous tubules of untreated control mice. Expression analysis of
Discussion
The in vivo mechanisms regulating the biological activity and cell fate decisions of SSCs remain poorly understood. SSCs, like other adult stem cells, are thought to reside in niches, yet the structural and functional components of the niche microenvironment are undefined in the testis. The adult seminiferous epithelium is organized into stages of spermatogenesis classified based on the precise complement of differentiating germ cells within a specific segment of a tubule, and occurs in a spatial and temporal pattern of development. Further, the seminiferous epithelium cycle is associated with areas of high responsiveness to FSH, androgen, and retinoid signaling, despite exposure to a similar endocrine milieu. Several reports suggest that undifferentiated spermatogonia are preferentially located near the interstitial compartment 9 and vasculature 10 within the testis in a stage-specific (IV–VI) manner. This raises the question as to whether SSCs and their respective niches are randomly distributed or preferentially located within areas of the seminiferous tubule.
No functional studies have been used to (1) identify the specific location and distribution of SSCs in the seminiferous tubule, or (2) investigate the specific in vivo effects of local microenvironment on the biological activity of SSCs to self-renew or differentiate. In this study, we demonstrate that SSCs are randomly distributed across stages of the mature seminiferous epithelium in mice. We also observed stage-specific differences in the rate of colony growth and expansion after transplantation. Donor cells obtained from stages IX–IV produced larger colonies as characterized by a greater rate of postcolonization expansion when compared to SSCs from stages V–VIII. This suggests that the adult SSC population is characterized by varying degrees, or subsets, of stem cell activity, and indicates that a specific tubule microenvironment is associated with SSC activity in vivo.
Factors and protein markers associated with SSCs have been used to investigate the transition of a SSC into differentiating spermatogonia. For example, Sertoli cells produce GDNF in response to FSH,26,27 whereas testosterone stimulates the expression of GDNF by peritubular myoid cells.28,29 Responsiveness to FSH, androgen, and retinoic acid is highest between stages IX–I,13,30 II–IV, 14 and V–VIII 15 respectively, and this pattern correlated with the stage-specific expression of GDNF. A strict, stage-specific expression pattern was also observed for Bcl6b, a GDNF-inducible regulator of SSC maintenance and self-renewal in vitro. Bcl6b expression was observed with high, intermediate, and low expression in stages IX–I, II–IV, and V–VIII, respectively. Thus, the stage-specific responsiveness to FSH, androgen, and retinoids may provide local gradients of signal transduction necessary to precisely direct the cell fate decisions of SSCs, either directly or through the somatic microenvironment (see model; Fig. 6).

Model of SSC cell fate decisions within the seminiferous tubule of adult mice developed with the use of the data presented in this article. The stage-specific microenvironment regulates SSC self-renewal and differentiation, but regulates production and potentially bioavailability of SSC niche factors such as GDNF. Factors such as retinoic acid stimulate spermatogonial differentiation and alter the stem cell potential of the undifferentiated pool of spermatogonia, such that spermatogonia may have actual and potential stem cell activity resulting in differential colonization and expansion capability after transplantation. Large gray ovals represent Sertoli cells, and blue circles represent differentiating germ cells (large round: spermatocytes; small round: round spermatids; slender elliptical: elongating spermatids) and spermatozoa.
The differential SSC expansion means that undifferentiated spermatogonia (actual SSCs) may signal and stimulate Sertoli cells to produce factors important for establishing a niche microenvironment for SSC expansion and self-renewal. Another population of undifferentiated spermatogonia (transit-amplifying SSCs) may have a reduced ability to exert feedback on Sertoli cells, thus negatively impacting or delaying their expansion after colonization. In support, donor cells obtained from stages IX–I and II–IV demonstrated larger postcolonization expansion than cells originating from V–VIII. This indicates the population of actual SSCs from stages IX–IV colonizes and initiates expansion before differentiation, accounting for the larger colonies observed in recipients. In contrast, the population of transit-amplifying spermatogonia from stages V–VIII may rapidly initiate differentiation after colonization and contribute toward a new population of A1 spermatogonia.
Additional alternative explanations for the increase in size of colonies from transplanted SSCs obtained from stages IX–IV include that these SSCs have enhanced proliferation capacity or accelerated migration along the length of the seminiferous tubule after transplantation. These differential SSC colonization kinetics could be due to the combination of the atypical seminiferous epithelial environment in busulfan-treated recipients and unique characteristics of SSCs from stages IX–IV. For example, Gdnf expression is significantly higher in the testis of busulfan-treated mice than untreated controls, whereas Ret expression is not different in these groups. The observed increase in GDNF expression is likely due to the loss of differentiating germ cell RNA resulting in a proportional increase in Sertoli cell-expressed messages, but this does provide a potentially highly receptive environment for SSC colonization. Similar expression of Ret in testes from busulfan-treated and control mice explains why we did not observe stage-specific expression of Ret. Sertoli cell expression of Ret suggests that GDNF acts through paracrine and autocrine mechanisms in the seminiferous tubule. The combination of high expression of GDNF and RET in the seminiferous tubules of busulfan-treated mice may provide an environment where SSCs from stages IX–IV have enhanced proliferation or self-renewal, while SSCs from stages V–VIII have enhanced migration or differentiation.
The development of protocols to enrich for SSCs demonstrates that heterogeneity exists in the pool of undifferentiated spermatogonia. For example, GFRA1 is expressed on the surface of undifferentiated spermatogonia 8 and is necessary to mediate the effects of GDNF-RET signal transduction. 31 However, Thy1+, (selected) GFRA1-negative spermatogonia demonstrate enriched stem cell activity when compared to GFRA1+ population after transplantation, and ∼50% of the GFRA1+ population are also positive for KIT, 32 a marker of spermatogonial differentiation associated with decreased stem cell potential. 33 Further, the expression of PLZF (ZBTB16), a transcription repressor whose expression is restricted to undifferentiated spermatogonia, 34 was similar across stages (data not shown). Quantitative real-time reverse transcriptase–PCR indicated no stage-specific effect on the expression Gfra1 and Nanos2, two genes associated with SSCs. The significant stage-specific expression difference for Gdnf and Bcl6b supports the conclusion that the regulation of the SSC activity is at the level of GDNF production or bioavailability.
Germ cell transplantation analysis signifies a strong, functional relationship between SSC microenvironment and stem cell activity in vivo. The equal distribution of SSCs (and their niches) across the mature seminiferous epithelium was confirmed by transplantation, as no stage-specific effect on colonization efficiency was observed, and is supported by morphometric analysis. 8 Together, these findings indicate that stem cell activity is independent of a static niche, as SSCs and their niches are equally distributed across all portions of the seminiferous tubule in the adult testis. The findings of Yoshida et al. 35 suggest that a population (subset) of Aundiff spermatogonia retains stem cell activity as a transit-amplifying cell. These, taken together with our data here, indicate that the pool of transit-amplifying Aundiff spermatogonia is largest in stages IX–I and II–IV (where GDNF expression is highest), whereas extrinsic signals in stages V–VIII stimulate the pool of transit-amplifying Aundiff spermatogonia toward differentiation. Therefore, the cycle of the seminiferous epithelium may regulate the dynamics of transit-amplification and stem cell activity in populations of Aundiff spermatogonia. Based on these data, we present a revised model summarizing the cellular kinetics of SSCs and their microenvironment within the mature seminiferous epithelium in the mouse (Fig. 6).
To our knowledge, this is the first functional study regarding the population dynamics and stem cell activity of adult SSCs in vivo. Moreover, our findings provide evidence that SSC self-renewal is regulated by a distinct microenvironment independent of a resident stem cell niche in the seminiferous tubule of sexually mature mice. The complex cell biology of the testis necessitates a better understanding of the regulatory networks supporting and maintaining stage-specific microenvironments for SSCs. Elucidation of these mechanisms may offer new insights into the regulation of other tissue-specific, adult stem cell populations. Identifying the balance of extrinsic and intrinsic cues that directly regulate the biological activity of SSCs and functional and structural components of the tubule microenvironment will be the focus of future in vivo experimentation and may provide novel approaches for treating male-factor infertility for agricultural and human biomedical purposes.
Footnotes
Acknowledgment
We extend our appreciation to Dr. Robin Hobbs for the PLZF antibody.
Author Disclosure Statement
The authors have nothing to disclose.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
