Abstract
Although human cardiomyocytes (CMs) are capable of some cell division, this response is neither sufficient to repair damaged cardiac tissue nor efficient to compensate for pathological stress. Danio rerio (zebrafish) CMs have been shown to have high proliferative capability to completely repair hearts after injury; however, no reports have focused on their physiological and cellular response to cardiac overload stress. We hypothesized that forced excessive long-term cardiac overload stress would elicit a proliferative response similar to regenerative cardiac repair in zebrafish. We completed a 10-week forced fast-speed swimming exercise regimen, comparing exercised hearts to nonexercised controls for physiological function and histological evidence of cell proliferation. Our results indicate that exercised heart ventricles are 33% larger, yet exhibit no significant changes in cardiac physiological function as evaluated by the heart rate and the percent shortening fraction. We found 8% more CM nuclei per cross-sectional area within exercised ventricular tissue, indicating that cardiomegaly was not due to individual cell hypertrophy, but due to hyperplasia. This novel zebrafish cardiac stress model may be used to identify genes or proteins with therapeutic potential for treating cardiac stress pathologies, as well as molecules that could be used as initiators of cardiac cell proliferation in humans.
Introduction
When human hearts are subjected to physiological and pathological stress, individual ventricular cardiomyocytes (CMs) undergo hypertrophy to increase in size, resulting in a larger heart with a thickened left ventricular wall.1–3 Cardiomegaly of mammalian hearts usually results in an inadequate performance and ultimately heart failure.4–6 Injured human cardiac tissues stimulate molecular mechanisms that produce fibrous scar tissues, 7 causing dangerous contractile dysfunctions with overload stress, eventually leading to cardiac death. 8 Although CM regeneration does occur at some low rate in human hearts, 9 this regenerative ability is unfortunately inefficient and neither adequately repairs damaged cardiac tissue 10 nor responds to demands of overload stress with an efficiently compensated heart. 4
In contrast, adult zebrafish (Danio rerio) efficiently and completely regenerate working heart tissue via hyperplasia—proliferation of existing cardiac cells.11,12 Thus, zebrafish have become a popular model for the study of cardiac tissue regeneration.7,13 So far, only their response to cardiac injury has been investigated using three models: apex amputation,11,14,15 cryoinjury,16–18 and genetic CM depletion. 19 These studies have begun to identify genetic targets for therapy in human cardiac regeneration.14,15,20 Because these models involve injury, altered gene expression may be due to cardiac damage and/or CM proliferation. 21 These injury models do not target responses to excessive cardiac overload stress; ultimately overload stress is what leads to heart failure in humans. Two other studies have described zebrafish heart enlargement via hyperplasia in response to rapid growth 22 and chronic anemia. 23
We present a novel approach—excessive intermittent forced swimming exercise—to study molecular and cellular pathways involved in the proliferative stress response of zebrafish hearts. Our hypothesis was that exposure of zebrafish to long-term excessive cardiac overload stress through intermittent forced swimming exercise would elicit a proliferative response similar to the regeneration process seen with cardiac injury models. We established that our exercise regimen results in overall heart enlargement, and that enlargement occurs via hyperplasia. We also evaluated the physiological efficiency of the enlarged, compensated zebrafish hearts.
Materials and Methods
Adult mixed-sex wild-type Danio rerio were handled in accordance with current National Institutes of Health (NIH)/National Research Council Guide for Care and Use of Laboratory Animals. Twenty wild-type zebrafish were housed in our fish exercise aquarium (exercised [EX]), and twenty wild-type zebrafish were housed in Barry University's Core Aquarium Research Facility (nonexercised [Control]). Fish density, water, and feeding were carefully controlled. We used a custom-built fish treadmill (unpublished; Bhuvasorakul, Faragasso, and Chase, Florida State University, Tallahassee, FL), equipped with an Emerson 1081 pool/aquarium pump (Dolphin Pumps, Pensacola, FL) to force exercise. The swimming rate was calculated at 25 cm/s—considered to be fast sustainable swimming speed for zebrafish24,25—by dividing length of the treadmill by the total time it took to empty during maximum flow. Fish were exercised for two 90-min exercise sessions per day for 10 weeks. Subsequently, zebrafish were anesthetized with an aqueous solution of tricane methane sulfonate (Finquel, Argent Chemical Laboratories, Redmond, WA) 0.168 g/L, (pH 7–7.5), for 10 min to achieve deep plane anesthesia. Zebrafish were euthanized by heart extraction. Ten EX and ten Control were sacrificed for physiological contractile studies; nine EX and eight Control were sacrificed for tissue sections.
Video images were recorded using a Q-Imaging Rolexa-XR Fast 1394 camera (Q-Imaging, Surrey, Canada) attached to a MZ8 Leica Dissection Scope (Leica Microsystems, Inc., Buffalo Grove, IL) with StreamPix Software (Norpix, Montreal, Canada). Videos were processed using Virtual Dub 1.6.17 (Avery Lee, 2007), and imported into ImageJ 1.39 s (NIH). Fluorescence microscopy was performed on an Olympus BX60 microscope (Olympus, Center Valley, PA) using an Olympus DP70 camera. Still images were processed using DP Controller Ver. 2.1.1.183 and DP Manager Ver. 2.2.1.195.
The total body length (BL; in cm) was averaged to obtain the mean. The ventricular surface area (VSA) was measured from still photos using ImageJ and a digital pen pad. Surface areas for three different angled photos of each heart were averaged to obtain the VSA. The VSA/BL ratio was calculated for each heart, and ratios were averaged to obtain means for each group. The VSA/BL ratio has previously been established as an index for the measurement of cardiomegaly.23,26 Heart rates of anesthetized fish were counted from video images and converted to beats per minute (BPM). To determine the percent shortening fraction, an open heart surgical procedure was performed on anesthetized fish to open the pericardium and expose the intact beating heart to the camera. 0.5–1 m video clips of intact, exposed, beating hearts were obtained. Stacks of frames were analyzed frame-by-frame to identify still images of end-diastole and end-systole for each heart. ImageJ was used to measure the length in pixels of the short axis across the ventricle at either stage. The percent shortening fraction was measured as the difference between mean end-diastolic and mean end-systolic ventricular dimensions divided by the mean end-diastolic dimension. Immunostained overlay images of ventricular tissue sections were obtained using standard immunohistochemistry, and then analyzed to specifically identify CM nuclei within a cross-sectional area (CSA). A primary rabbit polyclonal antibody to myocyte enhancement factor-2 (Mef2) and a secondary goat anti-rabbit IgG Texas red antibody (MEF-2 C-21sc-313 and sc-2780; Santa Cruz Biotechnology, Santa Cruz, CA) were used to fluorescently label Mef2-tagged CM. 4′-6-Diamidino-2-phenylindole (DAPI)-phosphate-buffered saline (100 ng/mL) was used to label nuclei. Sections were imaged with TRITC (for Mef2) and DAPI (for nuclei) filters on the same microscopic field, and then overlaid. To exclude nuclei from fibrocytes and nucleated red blood cells, only those nuclei specifically lying within CMs were counted and included in the analysis. A total of twenty 0.5-mm2 boxed areas at two different tissue depth levels were averaged to obtain each CM/CSA ratio.
Data analyses were performed with Excel (version 2010; Microsoft, Redmond, WA) and SigmaPlot 2009 (9.01; SPSS Inc., Chicago, IL). Statistical significance was determined using the Student's t-test.
Results and Discussion
We have investigated a novel, noninvasive method to study the process of cardiomegaly via CM hyperplasia in wild-type Danio rerio. After 10 weeks of forced excessive cardiac overload, we found no significant difference in average total BL between EX and Control (Fig. 1). However, EX fish developed significant cardiomegaly, with a 33% higher (p<0.01) VSA/BL as compared to Controls (Fig. 1). Contractile physiology of EX was essentially the same as Control: no significant difference in either heart rate (HR) or the percent shortening fraction was found (Fig. 1). Enlarged mammalian hearts tend to initially respond to overload stress with a compensatory stronger ventricular contraction, which steadily progresses to decompensated decreased output; 27 enlarged EX zebrafish hearts are capable of sustaining normal resting HR and contractile strength despite significant enlargement.

Zebrafish hearts enlarge significantly, but demonstrate no changes in contractile physiology in response to long-term excessive forced exercise stress. After 10 weeks of forced swimming exercise, there was no significant difference in body length (BL) between exercised zebrafish (EX) and Control fish (absolute values, EX=3.39 cm; Control=3.26 cm.), and the ventricular surface area/BL ratio indicates development of significant cardiomegaly in EX fish (absolute values, EX=0.32; Control=0.24). EX hearts were 33% larger than Control. Despite significant enlargement, there was no difference in heart rate (absolute values, both EX and Control=79.8 beats per minute) or strength of contraction as measured by the percent shortening fraction (absolute values, EX=8.5%; Control=9.1%). N=10 hearts for each data set. Error bars represent SEM. Statistical significance: **p<0.01.
At the cellular and tissue levels, an increase in cardiac mass can be explained by either collective increase in size of single cardiac cells (hypertrophy) or increase in the cardiac cell number (hyperplasia). 23 If tissue enlargement were due to hypertrophy, CM nuclei would distribute farther apart relative to each other as each cell enlarges, detectably decreasing the ratio of CM nuclei/CSA. However, if observed cardiomegaly were attributed to hyperplasia, there would be either unchanged distribution of CM nuclei (if tissue was fully remodeled and cells were mature), or increase in CM nuclei/CSA (if cells were still cycling and/or immature). We found 8% more CM nuclei/CSA in EX compared to Control (p<0.05) (Fig. 2). These data indicate that cardiomegaly in EX fish is due to hyperplasia—proliferation of CMs—rather than hypertrophy.
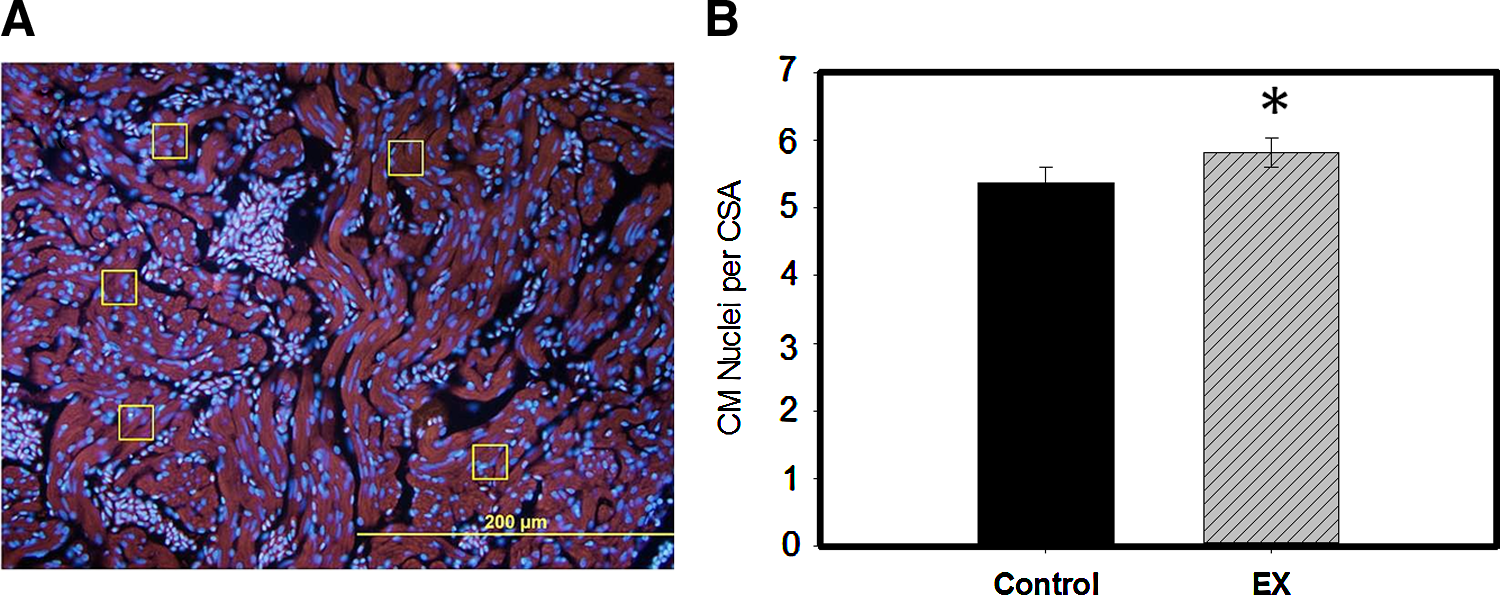
Cardiomegaly of exercised hearts occurs via hyperplasia, as indicated by an increased number of cardiomyocytes per cross-sectional area. There were 8% more CM nuclei per measured cardiomyocyte (CSA) in EX ventricular sections than Control.
Our model can be used to investigate proliferation initiation factors in zebrafish hearts. When human hearts enlarge in response to pathological stress overload, the physiological performance is altered.4,6 We found zebrafish compensatory heart enlargement to be a more efficient response to overload stress than hypertrophic enlargement in humans. Our model could potentially serve to investigate molecular targets for specifically treating human pathological cardiac overload stress conditions such as diabetes and hypertension.
Injury models have shown that zebrafish can fully repair heart tissue by 60 days postinjury. 16 Gene markers that were upregulated in the early stages of regeneration show downregulation as early as 7–14 days postinjury.15,20 Our data were collected following 70 days of excessive exercise, representing fully remodeled hearts. Future studies will examine hearts at early time points during proliferation to observe significant changes in cellular and molecular responses in actively dividing CM. Investigating the zebrafish regenerative response, whether in response to cardiac overload stress or injury, may identify genes with therapeutic potential to initiate human cardiac cell proliferation and repair.
Footnotes
Acknowledgments
Funding was provided by the NIH-NIGMS MBRS RISE: R25 GM059244 award to Barry University (supporting M.J., P.D., N.L., and J.T.) and the DOE: No.-DE-FG02-06CH11438 award to Barry University.
We thank: Ms. Janina L. Bhuvasorakul, Mr. Joseph Faragasso, and Dr. P. Bryant Chase from the Department of Biological Science at Florida State University for designing and building the custom fish treadmill exercise tank; Mr. Stephen Dunham, DARPA grant program coordinator from the College of Health Sciences at Barry University, for preparation of all histological tissue sections; the Histopathology Laboratory at Memorial Regional Hospital, Hollywood, FL, for the provision of supplies and equipment for preparation of histological tissue sections; Dr. Gerhild Packert, Associate Dean/Professor from the College of Health Sciences at Barry University, for the use of microscopic equipment; Mr. Nicholas Morales for his assistance with exercise and husbandry of zebrafish; and Dr. Christoph Hengartner, Assistant Professor of Biology at Barry University, for assistance in proofreading and editing the article.
Author Disclosure Statement
All authors declare that no competing financial interests exist.
