Abstract
Serious bone injuries have devastating effects on the lives of patients including limiting working ability and high cost. Orthopedic implants can aid in healing injuries to an extent that exceeds the natural regenerative capabilities of bone to repair fractures or large bone defects. Autografts and allografts are the standard implants used, but disadvantages such as donor site complications, a limited quantity of transplantable bone, and high costs have led to an increased demand for synthetic bone graft substitutes. However, replicating the complex physiological properties of biological bone, much less recapitulating its complex tissue functions, is challenging. Extensive efforts to design biocompatible implants that mimic the natural healing processes in bone have led to the investigation of piezoelectric smart materials because the bone has natural piezoelectric properties. Piezoelectric materials facilitate bone regeneration either by accumulating electric charge in response to mechanical stress, which mimics bioelectric signals through the direct piezoelectric effect or by providing mechanical stimulation in response to electrical stimulation through the converse piezoelectric effect. Although both effects are beneficial, the converse piezoelectric effect can address bone atrophy from stress shielding and immobility by improving the mechanical response of a healing defect. Mechanical stimulation has a positive impact on bone regeneration by activating cellular pathways that increase bone formation and decrease bone resorption. This review will highlight the potential of the converse piezoelectric effect to enhance bone regeneration by discussing the activation of beneficial cellular pathways, the properties of piezoelectric biomaterials, and the potential for the more effective administration of the converse piezoelectric effect using wireless control.
Introduction
Large bone defects can result from trauma, infection, complex nonunions, and disease,1–3 and can be especially detrimental to the elderly people and those with other underlying physical pathologies, such as osteoporosis. 4 In large bone defects where the bone regeneration is impaired or the demands of restoration cannot be met, surgical intervention and specialized treatments are essential to promote healing in response to injury. Autografts and allografts facilitate healing of these large defects, but with challenges such as donor site complications and restrictions on the amount of transferable bone.2,3 Thus, synthetic materials are appealing alternatives to biological tissues for bone implants. However, it is challenging for synthetic bone implants to mimic all the regenerative properties of the native tissue. Synthetic substitutes must overcome the problem of biocompatibility and also promote the natural signaling processes involved in bone healing. Therefore, it is an ongoing challenge to create a bone substitute with biological and mechanical properties comparable with bone. 2
The gap in the functionality of bone implants can be filled by the incorporation of smart materials that can enhance bone regeneration for improved orthopedic care. Smart materials are distinguished by their ability to reverse, change, or generate a response from external stimuli in their environment.4,5 Among the available smart materials, piezoelectric materials are utilized for bone regeneration because of their ability to electrically stimulate and mechanically actuate the healing bone. Piezoelectric materials can exhibit electromechanical responsivity to an external stimulus in either a direct or converse piezoelectric configuration. 6 The direct piezoelectric effect is defined as the generation of an electric voltage from applied mechanical stress, and the converse piezoelectric effect is the reverse scenario, where an applied electric voltage results in a mechanical response4,7 (Fig. 1). The bone itself has inherent piezoelectric properties and can respond to mechanical activity to produce electrical and biochemical signals that enhance bone growth.8,9 Although both the direct and converse piezoelectric effects are important, only the direct piezoelectric effect has been extensively studied for bone-implant applications. The direct piezoelectric effect requires movement to generate electrical stimulation, which is not practical in treating immobilized patients. 9 Incorporation of mechanical stimulation into bone-implant designs can be beneficial for enhancing healing in the absence of movement, and similar results could potentially be achieved through the converse piezoelectric effect, which is the focus of this review. 9 In addition to providing mechanical stimulus to the implanted site, the converse piezoelectric effect could also prevent stress shielding of implants. Stress shielding occurs when increased stiffness of the implant leads to decreased mechanical loading on the surrounding bone. 10
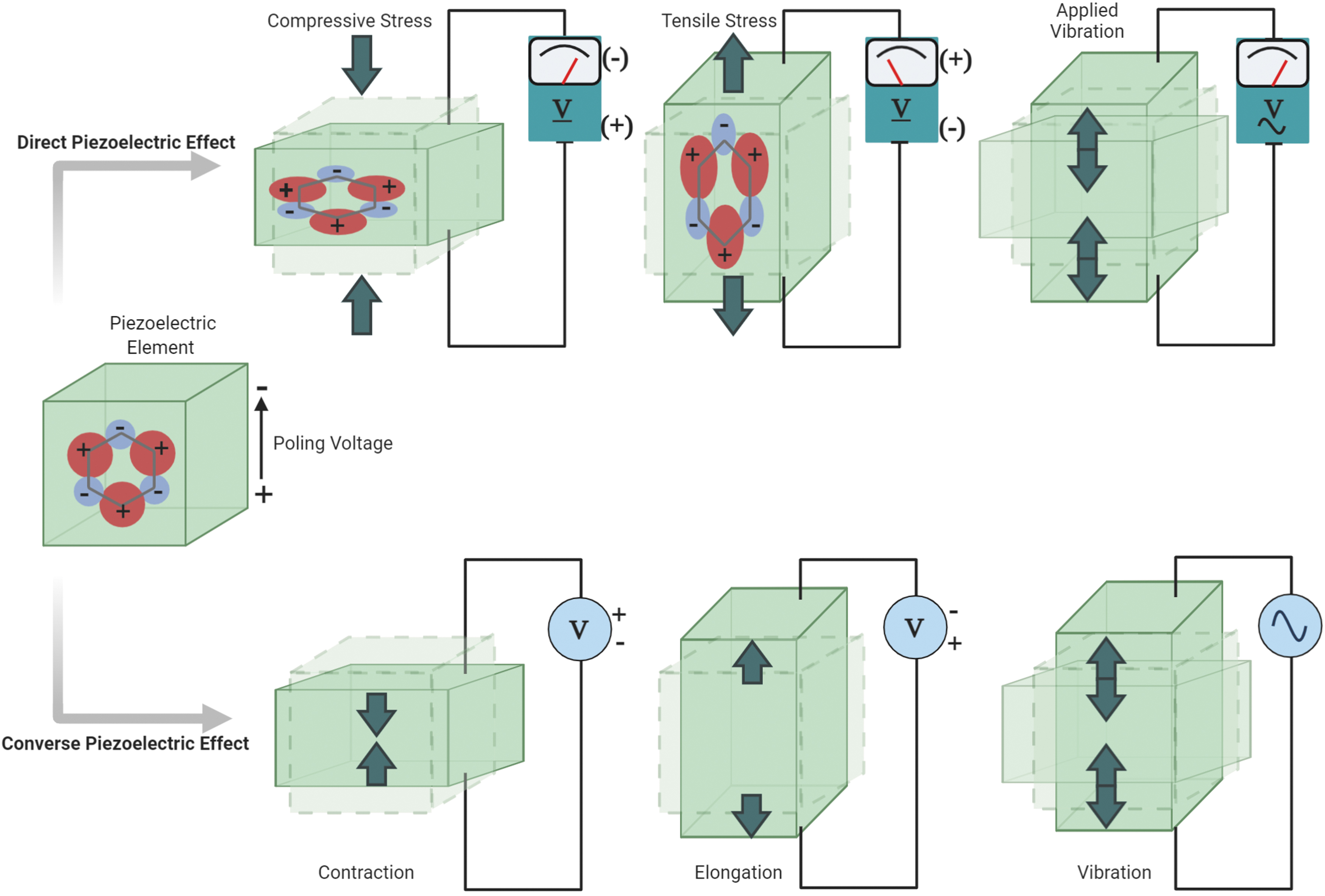
The Piezoelectric effect. The direct piezoelectric effect generates an electric voltage in response to a mechanical force, whereas the converse piezoelectric effect produces mechanical stimulation in response to an electric voltage.
This review will cover (i) mechanisms currently used to describe how mechanical stimulation through the converse piezoelectric effect can enhance bone growth and evaluate the utility of the converse piezoelectric effect in bone-implant applications. (ii) characteristics of piezoelectric materials used in biomedical applications and the desirable features of bone implants for a successful promotion of bone regeneration, and (iii) the potential of a wirelessly controlled piezoelectric implant to stimulate bone formation. All the topics discussed in this review are related to the importance of mechanical stimulation in the process of bone regeneration and how the converse piezoelectric effect exerted in a bone implant might enhance bone repair.
Bone and the Role of the Piezoelectric Effect in Bone Regeneration
Mimicking the extracellular matrix (ECM) of osseous tissue has provided the basis for creating materials for inducing bone regeneration. 11 Bone ECM is 30% organic components, 60% inorganic components, and 10% water. 12 Proteins, especially type I collagen, comprise the main organic constituent, whereas hydroxyapatite (HA) crystals comprise the primary inorganic component.11–13 The collagen is largely responsible for improving the toughness of the structure, whereas bone mineral provides strength and stiffness.14,15 These components ensure strength with some flexibility so that the bone does not simply snap under force.
When a bone breaks, the type of fracture, the stability of fixation, and the type of loading on the repaired structure contribute to the mechanical environment of the healing bone. 16 These factors can dictate whether the bone heals by direct (primary) or indirect (secondary) fracture healing. Primary fracture healing is characterized by direct remodeling of the lamellar bone and blood vessels, without the formation of a callus. 17 Primary fracture healing does not commonly occur because it requires realignment of the bone fracture ends and highly stable fixation.16,17 Secondary fracture healing is more common and consists of callus formation and both endochondral and intramembranous repair.17,18 This form of fracture healing is enhanced by weight bearing 17 ; however, more research is needed to determine the parameters for this positive effect. Too much load bearing during rehabilitation can cause pain and discomfort, but reduced bone mass or osteopenia can result from immobilization or strict fixation. 19 The use of the converse piezoelectric effect is promising in both primary and secondary fracture healing because the amount of mechanical stimulation supplied by the implant is directly proportional to the current that powers it and can be carefully controlled at the fracture site.
Bone has piezoelectric properties because of the highly oriented and patterned structure of collagen, and collagen's ability to respond to mechanical loads.20,21 When a shearing force is applied to collagen fibers and they slip past each other, a piezoelectric charge is generated. 22 Collagen also has significantly lower elastic moduli than the bone's corresponding mineral component, which makes collagen experience the greatest load when strained. Experiencing the greatest load under force deforms collagen fibers, and this deformation leads to the piezoelectric effect. 20 The role of collagen's piezoelectricity in bone regeneration and remodeling is difficult to decipher. Collagen's piezoelectricity is potentially an additional mechanism for osteocytes to sense areas with more stress; the generated piezoelectric charge would be greater in stressed areas, and this electrical signal can also stimulate osteoblasts to enhance bone formation. 20
There is a controversy around the presence of the piezoelectricity of the bone and its role in regeneration. Most likely, besides the piezoelectric effect, the fluid flow and streaming potentials were reported to contribute to bone healing and regeneration. 23 Although more research is needed to understand the extent that each play in the repair process, it is important to note that mechanical loading induces both bone-promoting responses.
Piezoelectric Effects on Cells that Promote Bone Growth and Repair
To develop a more complete understanding of either the converse or direct piezoelectric effects on bone repair, it is essential to understand the impact on cell function as well.
Direct piezoelectric effect
Cell signaling pathways are induced in bone repair models by mechanically induced electrical stimulation (direct piezoelectric effect). Compression on bone is shown to produce a negative electric charge owing to collagen reorienting its dipole moment, whereas traction produces a positive charge.4,24,25 Production of the negative electric charge induces cell membrane hyperpolarization, and the positive electric charge produces cell membrane depolarization. 25 The hyperpolarization from compressive force can promote osteogenesis and osteogenic differentiation of bone marrow cells from Ras activation.26,27 The generated negative charge can also electrically stimulate bone cells through the opening of voltage-gated calcium channels. 4 Opening these channels increases the intracellular calcium concentration, activating calmodulin to promotes nucleotide synthesis and cell proliferation.28–31
Activating the calcium/calmodulin pathway leads to the dephosphorylation of the nuclear factor of activated cells (NF-AT), which will translocate into the cell nucleus and bind with other transcription factors. 6 Binding facilitates gene expression of transforming growth factor β (TGF-β) and bone morphogenic protein (BMP).4,6 TGF-β promotes cell growth and differentiation by promoting osteoblast proliferation. 32 TGF-β is involved in many regulatory pathways such as mitogen-activated protein kinase (MAPK) and mothers against the decapentaplegic family of proteins (SMAD) that enhance tissue repair, regulate ECM production, promote mesenchymal stem cell (MSC) differentiation, and regulate bone homeostasis. 32 BMPs are osteogenic agents that are used to accelerate fracture healing, 32 and they are even used to treat complex bone defects with evidence of quicker bone formation. 3
Many in vivo and in vitro studies have shown that the direct piezoelectric effect expedites bone repair and regeneration. A summary of selected studies is provided in Table 1, which includes the materials and methods used and the highlights of results.
Relevant Studies of the Direct Piezoelectric Effect
Studies were chosen based on their usage of the direct piezoelectric effect or a similar mechanism providing mechanical stimulation for bone applications.
PVDF, polyvinylidene fluoride; GMC, goat marrow cells; HA, hydroxyapatite; qALP, alkaline phosphatase quantification assay; P(VDF-TrFE), polyvinylidene fluoride-trifluoroethylene; MSC, mesenchymal stem cells; RUNX2, runt-related transcription factor 2; ALP, alkaline phosphatase; PLLA, poly-
Converse piezoelectric effect
The shear strain on collagen and the piezoelectric effect, combined with shear strain from the fluid flow, is hypothesized to regulate mechanotransduction signaling in bone. 23 Osteocytes are immediately strained by mechanical loading and stretching of the surrounding bone tissue. The pressure generated from the fluid flow is hypothesized to magnify the strain on osteocytes and stimulates these cells. 33 This hypothesis is supported by the evidence that the amount of strain that the whole bone experiences in vivo from locomotion is typically 0.04–0.3% and rarely exceeds 0.1%, yet in vitro osteocytes need ∼1–10% strain to be activated.33–35 Pericellular organic matrix fills the space between the osteocyte cell processes and the canalicular wall; when the bone is deformed, the fluid flows through this pericellular space and there is a resulting drag force. 33 The strain amplification factor increases with loading frequency and increasing loading strain. 36 Thus, the structure and properties of bone facilitate the ability for mechanical stimulation to be sensed by bone cells, which subsequently strengthen the tissue and maintain bone health.
Mechanically activated osteocytes release signaling molecules, such as calcium ions, nitric oxide (NO), adenosine triphosphate (ATP), growth factors, Wnts, and prostaglandin E2 (PGE2), all regulate the activities of bone. 37 The following sections, which are summarized in Figure 2, outlines the current information on mechanisms and pathways relevant to these signaling molecules and also suggest a possible connection with the converse piezoelectric effect.
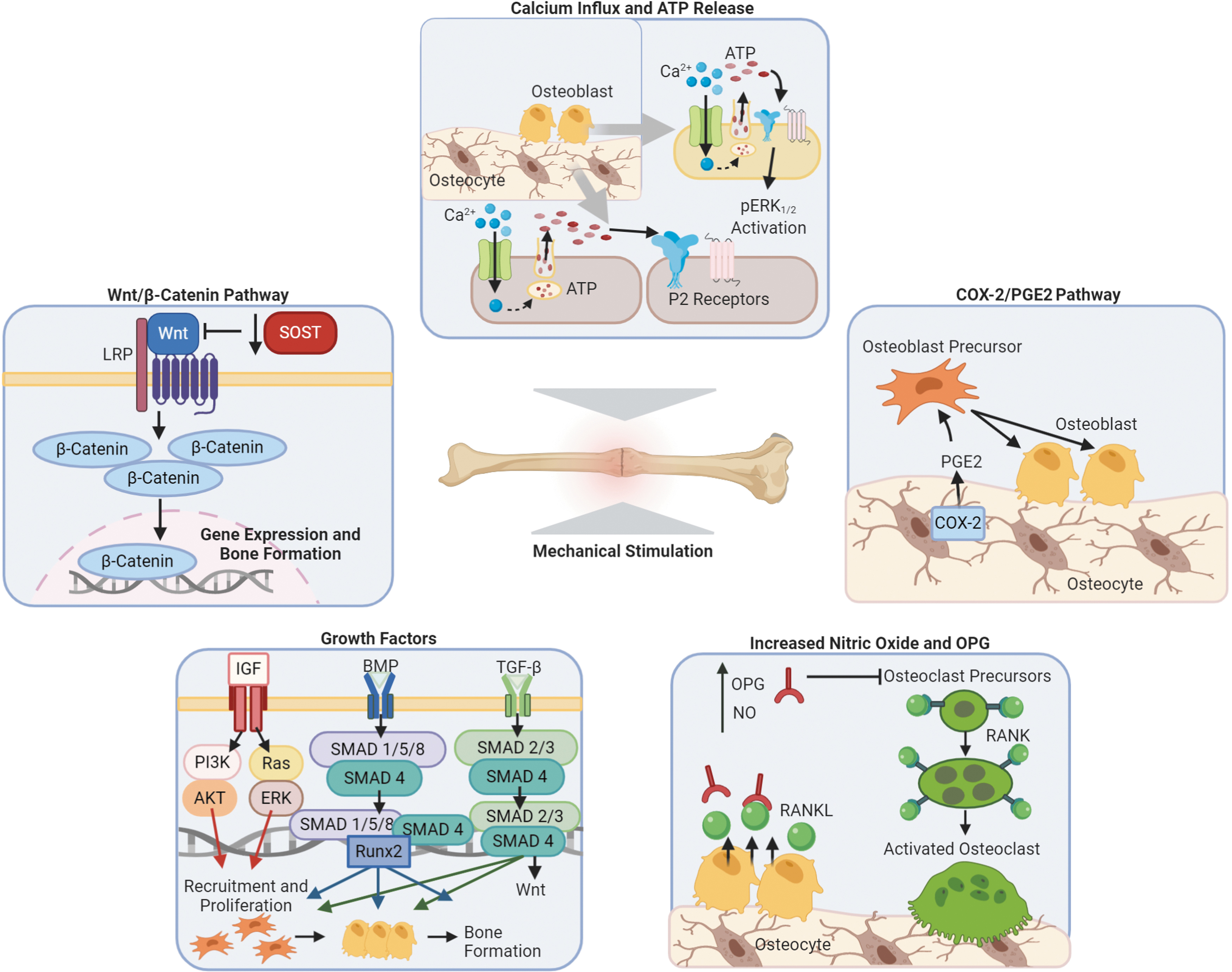
Mechanisms of bone cell response to mechanical stimulation. Defined bone cell mechanisms for the influx of calcium and ATP release, COX-2/PGE2 pathway, increased nitric oxide and OPG, growth factors, and the Wnt/β-catenin pathway. Overall, the effects of mechanical stimulation on bone show increased regeneration and decreased resorption. ATP, adenosine triphosphate; pERK1/2, phosphorylated (active)-extracellular signal-regulated kinases; P2 receptors, paracrine purinergic; COX-2, cyclooxygenase-2; PGE2, prostaglandin E2; NO, nitric oxide; OPG, osteoprotegerin; RANK, receptor activator of nuclear factor-κB; RANKL, nuclear kappa-B ligand; IGF, insulin-like growth factors; TGF-β, transforming growth factor β; BMP, bone morphogenic protein; PI3K, phosphoinositide 3-kinases; AKT, protein kinase B; Ras/MAPK, mitogen-activated protein kinase; ERK, extracellular signal-regulated kinases; Runx2, runt-related transcription factor 2; Wnt, wingless-related integration site; SOST, sclerostin encoding gene; LRP, lipoprotein receptor-related protein.
Influx of calcium ions and release of ATP
The influx of calcium ions is one of the earliest responses measured once osteocytes undergo mechanical loading.38–40 The influx of calcium ions occurs through the activation of mechanosensitive and voltage-gated calcium channels in the plasma membrane.38,39 Opening the voltage-gated calcium channels and the influx of calcium ions is needed for ATP release.41,42 ATP release and activation of the P2 receptor leads to phosphorylation of ERK1/2, which is activated by oscillating fluid shear stress in osteoblasts,42,43 as well as MAPK activation in osteoblasts. 44 The MAPK pathway is believed to regulate cell growth and differentiation of osteoblasts. 43
Cyclooxygenase-2 and PGE2
Inhibiting cyclooxygenase-2 (COX-2) in rats through NS-398 led to the conclusion that the induction of COX-2 is important for the anabolic response of bone to mechanical strain. 45 COX-2 is the enzyme that creates PGE2, which is released by mechanically loaded osteocytes. PGE2 is an important signaling molecule in the bone because it recruits and promotes the differentiation of osteoblast precursor cells, and also enhances the function of existing osteoblasts. 46 The ability to improve osteogenesis through stem cell differentiation is an important facet of the autograft, so having a piezoelectric implant capable of naturally recruiting osteoblast precursors would provide an innovative approach to bone procedures.
NO and osteoprotegerin
Bacabac et al. showed that MC3T3-E1 preosteoblasts subjected to fluid flow will produce an amount of NO linearly proportional to the rate of fluid shear stress. 47 In addition, the rate of mechanical loading is what enhances bone formation, rather than the magnitude of strain. High frequency, but low-magnitude mechanical stimulation can produce a high rate of loading, which enhances bone formation. 48 NO inhibits bone resorption by decreasing the expression of receptor activator nuclear kappa-B ligand (RANKL). RANKL is important for osteoclast formation, and NO decreases this activity. 49 Osteoprotegerin (OPG) is a decoy receptor for RANKL that prevents it from binding to the RANK receptor on osteoclasts. 50 Mechanical stimulation can regulate the gene expression of OPG and increase its prevalence to prevent bone resorption of a healing bone. 37
NO is a target indicator for testing the ability of the converse piezoelectric effect to mechanically stimulate the bone cells. Because NO production is considered proportional to the rate of fluid shear stress, the effects of mechanical stimulation from the converse piezoelectric effect can be assessed.
Growth factors: insulin-like growth factor, TGF-β, and BMP
Another early response to mechanical loading is the release of insulin-like growth factors (IGFs). Both IGF-1 and IGF-2 stimulate proliferation and differentiation of bone cells.51–53 IGF-1 is also important in regulating peak bone mass and maintaining bone mineral density. 54 An in vivo study demonstrated that mechanical loading of rat tibia induced IGF-1 expression in osteocytes. 55 IGF-1 induces differentiation of MSCs into osteoblasts by activating mTOR through the PI3K-Akt pathway. 54 Osteoblast-like cells derived from rat long bones exposed to dynamic strain increased the smallest transcript of IGF-2 (IGF-2 T3). 56
Fluid shear stress can also increase TGF-β in osteoblastic cells, 57 and mechanical stimulation on osteocytes can upregulate TGF-β.50,58,59 TGF-β is produced by osteoblasts and regulates the mineral component of the bone matrix. 60 TGF-β activation stimulates osteoblasts to create a new bone matrix, and this phenomenon is critical in the fracture healing and bone repair mechanisms. 61 BMP-2, 4, 6, 7, and 9 are involved in bone morphogenesis.62,63 Mechanical strain promotes the osteoinductive potential (in mouse MC3T3-E1 cells) by enhancing BMP-2 and BMP-4 levels in the ECM. 64 Mechanical stimulation also induces BMP-7 release by osteocytes, which promotes bone formation and also protects cells from apoptosis. 65
The BMP signals through SMAD phosphorylation, thus there is a direct correlation between SMAD and BMP. 66 Tan et al. showed that without SMAD4 in osteoblasts, there was a decrease in osteoblast proliferation and a concomitant decrease in bone volume. 67 This study reveals the importance of SMAD in maintaining bone growth, and the early stages of SMAD activation can be enhanced by applied mechanical force. 63 In addition to SMAD, ion channels have been shown to impact BMP signaling. 66 Specifically, inwardly rectifying potassium (Kir2.1) as a type of ion channel is believed to impact the BMP signaling. The loss of Kir2.1 in mice affected bone formation similar to the loss of BMP2, 4, or 7. 66 In addition, the loss of Kir2.1 leads to a decrease in SMAD phosphorylation, meaning the BMP signal is not efficiently transmitted without this channel. 66 BMPs are used in conjunction with bone grafts for their ability to promote growth and healing, so it would be beneficial to enact treatments that can naturally stimulate BMP production. Testing the converse piezoelectric effect for optimization of BMPs and other growth factors could lead to a more cost-effective and less invasive approach to bone repair.
Wingless type/β-catenin pathway
The Wingless type (Wnt)/β-catenin pathway is a regulator of bone homeostasis. 68 Osteocytes can respond to fluid shear stress in vitro through mRNA expression of Wnt signaling molecules, 69 indicating that bone is capable of adapting to mechanical stimulus through the Wnt/β-catenin pathway. Mechanical stimulation enhances the lipoprotein receptor-related protein 5, which is essential for Wnt signaling. 70 The SOST gene is downregulated during mechanical loading, and this gene encodes the protein sclerostin, 68 an inhibitor of bone formation through the Wnt/β-catenin pathway. 37 Thus, it is evident that mechanical stimulation plays a substantial role in bone regeneration because it results in the upregulation of Wnt signaling molecules and the downregulation of inhibitors of bone formation. Studying this relationship could provide insight for creating treatments that enhance gene expression leading to increased bone formation.
In observing the use of the converse piezoelectric effect, these cellular mechanisms described above should be tested to confirm effective mechanical stimulation. There are very few studies both in vivo and in vitro that test the converse piezoelectric effect compared with the direct piezoelectric effect, and these studies are summarized in Table 2.
Relevant Studies of the Converse Piezoelectric Effect
Studies were chosen based on their usage of the converse piezoelectric effect or a similar mechanism providing mechanical stimulation for bone applications.
PVDF, polyvinylidene fluoride; P(VDF-TrFE), polyvinylidene fluoride-trifluoroethylene; Li, lithium; OPN, osteopontin.
Properties of Piezoelectric Scaffolds Affecting Bone Growth
Biomaterials for bone regeneration are usually evaluated based on their potential for osteoinduction, osteoconduction, and osteogenesis. 3 An osteoinductive scaffold can induce bone formation by promoting the MSCs to form active osteoblasts, and this can be achieved by the inclusion of growth factors such as BMP.71,72 An osteoconductive scaffold guides bone growth on its surface and within its structure by allowing cells and tissue to attach.71,72 An osteogenic scaffold promotes the synthesis of new bone from cells within the structure. 72 The ability of a piezoelectric scaffold to provide mechanical stimulation to the tissue has the potential to fulfill all these characteristics of effective bone treatments. The mechanical strain on supporting scaffolds has been shown to improve the osteoinductive potential of the osteoblastic MC3T3-E1 cells in vitro, 64 and promote ECM-induced osteogenic differentiation of human MSCs in vitro. 73 High osteoconductive properties of a piezoelectric scaffold can be achieved depending on the materials used. Using a composite of bone materials such as collagen, HA, or even synthetic materials mimicking bone in a piezoelectric scaffold may enhance healing when combined with the converse piezoelectric effect to integrate bone growth with the scaffold. 9
The mechanical properties of bone and Young's modulus are also important to consider when constructing a scaffold. If the implant material is too flexible, mechanical loading from movement or daily activities will exert pressure onto the surrounding tissue and lead to excessive ossification. 21 Alternatively, if the materials used are much stiffer than bone, stress shielding will lead to bone resorption and implant loosening. 74 Optimizing the converse piezoelectric effect could provide the ideal mechanical stimulus needed to prevent such damage because the mechanical stimulation provided can be adjusted by the amount of electrical stimulation applied to the material.
Enhancing properties of piezoelectric materials in a scaffold for bone regeneration can also provide innovative solutions to the demands of orthopedic treatments, such as creating less invasive procedures for providing bone cell adhesion, mechanical stimulation, antimicrobial properties, and drug delivery. Creating a scaffold that can biodegrade in the body matching the schedule of the healing fracture is an impressive approach to improve current procedures. 75 Biodegradable scaffolds can be less invasive, more comfortable for patients, and cost-efficient because they would require surgical removal. The rate the scaffold degrades would need to be prolonged to match the healing rate of the fracture, which could vary depending on the individual. 76 The right blend of materials for a scaffold could provide the piezoelectric potential to optimize repair and the biodegradability to be absorbed once the bone has healed. 77
Antimicrobial properties are another area of opportunity for creating scaffolds for bone repair because there is a risk of contamination in invasive treatments. A scaffolding design capable of providing mechanical stimulation to bone through the converse piezoelectric effect could also release both antibiotics and appropriate cell signaling molecules for orthopedic repair.
Piezoelectric Biomaterials for Bone Regeneration
Piezoelectric materials used for bone regeneration applications can be classified as inorganic or organic materials. This distinction is not only based on composition but also extends to how piezoelectricity is expressed within each material. Inorganic piezoelectric crystals lack a center of symmetry and undergo structural shifts under mechanical stress as ions inside the inorganic crystals are displaced to generate a dipole moment. 76 Thus, the inorganic crystals develop electric polarization from the applied mechanical stress. In contrast, organic piezoelectric materials are mainly polymers and experience piezoelectric properties derived from their molecular structure. 76 Polymers may be semi-crystalline or noncrystalline, and frequently need to undergo a poling process or exposure to a high electric field to achieve piezoelectric properties.78,79 Organic materials are promising for bone regeneration because they can be biocompatible and even biodegradable. However, organic piezoelectric materials express a much lower piezoelectric coefficient than inorganic piezoelectric materials.6,76 There are lead-free inorganic piezoelectric materials for bone regeneration, but they need to be made more biocompatible by encapsulation 76 or by combining with other materials to form a composite. 9 In this section, common piezoelectric materials for bone regeneration are discussed further through the distinction of being piezoceramics, polymers, or composites (Fig. 3). Their properties will be described, along with their potential for creating a scaffold capable of delivering the mechanical stimulation needed by bone cells to enhance growth.
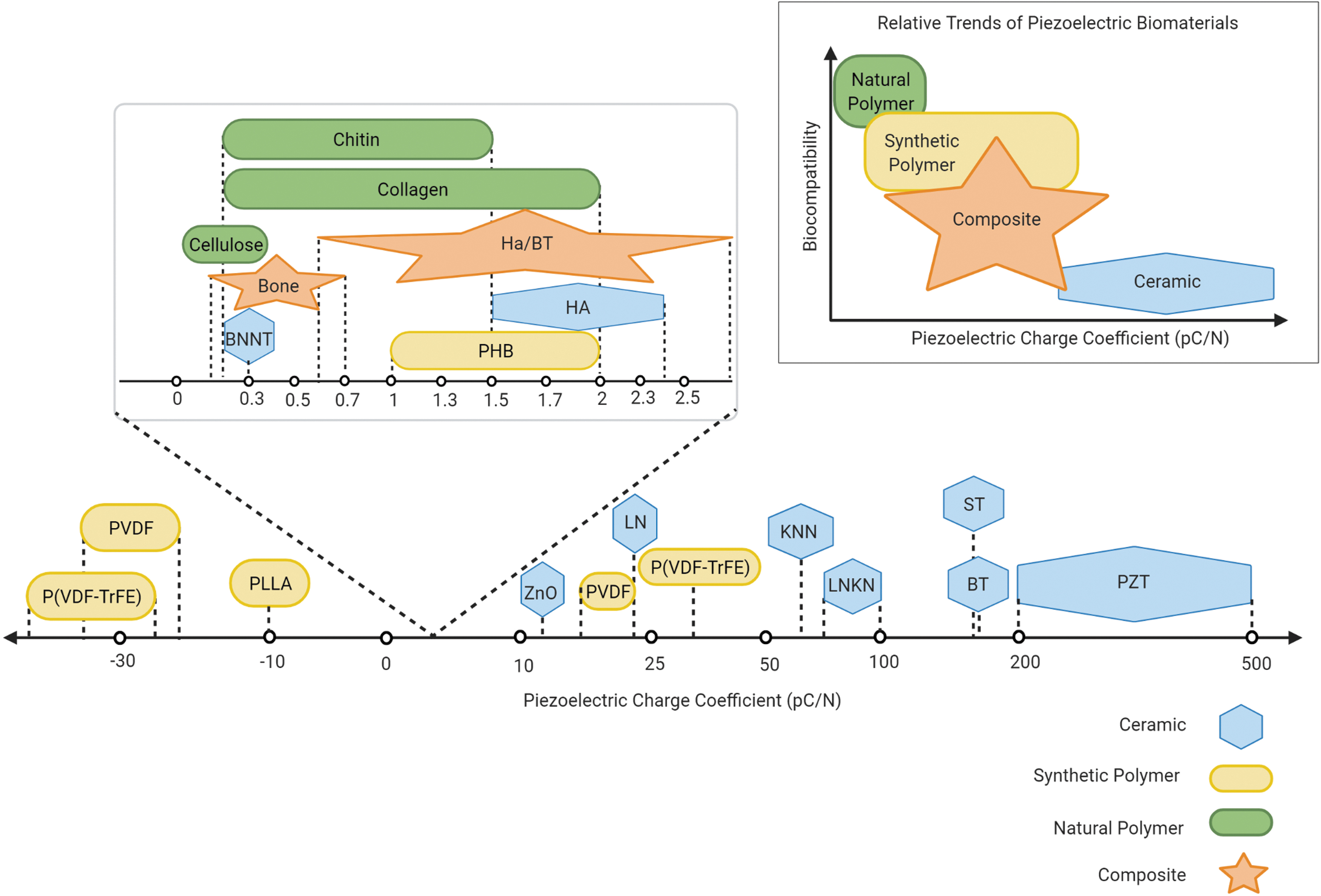
Relative trends observed in piezoelectric materials. Piezoelectric charge coefficients are plotted for various piezoelectric ceramics, synthetic and natural polymers, and composites. The general trend that polymers tend to be more biocompatible with lower piezoelectric charge coefficients, ceramics tend to be less biocompatible with higher piezoelectric charge coefficients, and composites can display a range of characteristics is depicted conceptually above. PZT, lead zirconate titanate; BT, barium titanate; ST, strontium titanate; LNKN, lithium sodium potassium niobate; KNN, potassium sodium niobate; P(VDF-TrFE), polyvinylidene fluoride-trifluoroethylene; PVDF, polyvinylidene fluoride; LN, lithium niobate; ZnO, zinc oxide; HA, hydroxyapatite; PLLA, poly-
Piezoceramics
Inorganic piezoceramics have high piezoelectric coefficients, which is advantageous for use in biomedical applications (Fig. 3). 6 Piezoceramics used as biomaterials are categorized mainly as lead-based, lead-free, or titanates. Although lead-based piezoceramics such as lead zirconium titanate (PZT) are sought after for their high piezoelectric output, lead is toxic. 80 Lead-free ceramics still generally exhibit higher piezoelectric coefficients than organic materials and are being explored for tissue engineering. Lead-free, biocompatible ceramics include zinc oxide (ZnO), potassium sodium niobate (KNN), lithium niobate (LN), lithium sodium potassium niobate (LNKN), and boron nitride nanotubes (BNNT),9,81 but the lead-free titanates, such as barium titanate (BT), have received the most attention for bone regeneration. 9
When using a piezoceramic for an implant, the issue with biocompatibility can arise because the ceramic releases ions when placed in the body. 81 However, a piezoceramic can be developed that releases ions that are osteoinductive or beneficial to the bone. One study demonstrated that using calcium titanate, strontium titanate, and barium titanate supported osteoblast proliferation in vitro, and Ca2+, Sr2+, and Ba2+ ions were all released from the scaffold. 82 This ion release from ceramic materials could be developed to provide an alternative to expensive growth factors. The release of beneficial ions can be supported by the scaffold and timed for a slow release, whereas growth factors can degrade readily if not supported properly by a scaffold. 82
As mentioned previously, piezoelectric materials lacking a center of symmetry can generate a net dipole moment under mechanical stress. However, HA is a centrosymmetric ceramic that also exhibits piezoelectricity because its nanoscale crystals can switch to a polar, noncentrosymmetric order when supplied with appropriate high-energy conditions. 83 Synthetic HA is commonly used because it mimics the natural inorganic component of bone. 84 However, on its own, HA is very brittle and thus not suitable for bearing high loads. 84 For this reason, Ha and other ceramics are often combined with other materials to improve their mechanical properties. Piezoceramics ultimately show potential for delivering high piezoelectric output, mimicking the inorganic crystal naturally found in bone, and releasing osteoinductive ions. Thus, piezoceramics could be used to optimize devices that provide mechanical stimulation through the converse piezoelectric effect. More research is needed to explore each of these properties in the context of piezoelectric implants, as well as for structural and biochemical support of bone regeneration.
Piezoelectric polymers
Piezoelectric polymers exhibit piezoelectricity derived from their molecular structure, producing a net charge when they undergo mechanical stress. Piezoelectric polymers are advantageous for bone implants because they are less expensive,
7
easier to produce, and lightweight compared with piezoelectric ceramic composites.
85
However, piezoelectric polymers have significantly lower piezoelectric coefficients than inorganic materials. Piezoelectric polymers are characterized by being ductile and flexible
85
and can be of synthetic or natural origin. Common synthetic piezoelectric polymers for bone applications are poly(vinylidene fluoride) (PVDF), poly(vinylidene fluoride-trifluoroethylene) (PVDF-TrFE), poly-
PVDF is a semi-crystalline polymer that exists in five phases, but only the polar β-phase has high piezoelectric properties. 79 The β-phase is a transconformation that can generate a dipole moment.79,81 Poled PVDF films have a greater piezoelectric coefficient than other polymers and are very popular in tissue-engineering applications. Ribeiro et al. showed that human adipose stem cells, when placed on an electroactive poled β-PVDF and subjected to dynamic conditions, had more osteogenic differentiation than under static conditions, or using nonpoled films. 87 PVDF has also shown the potential to induce bone formation configured to exert a converse piezoelectric effect. In a model treating cuts in sheep femur and tibia, Reis et al. demonstrated that the converse piezoelectric effect could be implemented using PVDF and an applied voltage to mechanically stimulate bone growth. 88
Another synthetic piezoelectric polymer is PVDF-TrFE. PVDF-TrFE is a copolymer of PVDF that demonstrates the highest piezoelectric coefficient of piezoelectric polymers because it exists in a transconformation that automatically demonstrates the β-phase. 89 Electrospinning PVDF-TrFE into a nanofiber scaffold improved the piezoelectric coefficient of the material. 90 This material enhanced wound healing in a rat model and the proliferation of fibroblast cells in vivo. 90 Although PVDF and PVDF-TrFE promote bone formation and are used in tissue-engineering applications, they are not biodegradable.
PLLA is a biodegradable and biocompatible polymer, so a scaffold made of this material would not need to be surgically removed from the body. PLLA has strong mechanical properties, so it is used in orthopedic devices, such as screws, pins, and plates. 91 The PLLA can be engineered to degrade slowly over time and gradual stress can be returned to the surrounding bone, so bone atrophy from stress shielding can be avoided. 92 In addition, electrospun PLLA nanofibers can mimic the structure of ECM fibers, making PLLA matrices especially useful for bone implants and bone graft substitutes. 93 Finally, electrospun PLLA has been used in sensor and actuator applications, 94 suggesting its utility for active piezoelectric devices.
Composites
Bone is a composite made of both inorganic and organic components to provide its dynamic load-sensing characteristics. This concept can be used to create scaffolds that combine inorganic piezoceramics and organic piezoelectric polymers with properties different from the individual constituents. An HA/BT composite achieved better biocompatibility and bone-forming activity when subject to cyclic loading than HA alone. 95 A P(VDF-TrFE)/BT composite supported bone formation in vivo and even prevented bone resorption through a decrease in RANKL expression, which limits the activity of osteoclasts. 96 To create synthetic bone graft substitutes, composite scaffolds may provide an effective material option. A biomimetic scaffold created by a nanohydroxyapatite(nHAp)/collagen/PLLA composite had a degradation rate that matched the rate of bone formation in rabbits, 97 demonstrating the principle that a biodegradable scaffold can be achieved with composites, eliminating invasive removal surgery.
Wireless Piezoelectric Effect
The implementation of the converse piezoelectric effect requires an electric source to induce mechanical stimulation, and more research is needed to create a biocompatible design that optimally powers this technology. Using Li-ion batteries to provide power has shown positive results in inducing the converse piezoelectric effect and providing mechanical stimulation.88,98 However, in vivo use of Li-ion batteries runs the risk of harmful contents leaking into the surrounding tissue. 99 Wireless control of the converse piezoelectric material could provide both a more biocompatible power source and automated feedback signaling. The result would be more control over the electrical stimulation and optimal mechanical stimulation of the tissue over time.
Wireless stimulation to enhance bone repair is an evolving area of study with the potential to create safer and more accessible treatments. Ultrasound has been used to provide wireless stimulation to piezoelectric materials. A nanomaterial made from ZnO nanowires and BT nanoparticles created an output current when stimulated by ultrasound, and this effect led to a calcium ion influx that enhanced neural differentiation. 100 Similarly, ultrasound has been used to wirelessly stimulate β-PVDF, inducing polarization of this piezoelectric material to enhance neurite differentiation in vitro. 101 Ultrasound activation of a PLLA nanofiber scaffold provided electrical stimulation to promote the healing of bone defects in mice. 102 Low-intensity pulsed ultrasound has increasing evidence supporting its utility for mechanically stimulating bone and inducing repair.103,104 Moreover, using a biodegradable PLLA scaffold, Das et al. showed that the combined effects of ultrasound and the generated piezoelectric charge enhanced bone healing. 102 In addition, PLLA nanofibers can act as an ultrasonic transducer, as demonstrated by Curry et al., and can be used to facilitate the delivery of drugs to the brain and wirelessly monitor vital physiological pressures. 105 Ultrasound is also an attractive option for wireless power transfer because even at a small wavelength it can still penetrate tissue. 106 Tsai et al. demonstrated that their design of ultrasonic wireless power transmission could safely deliver a power level of 15.91 mW to an implant in the body. 106
A different approach used magnetic force as a wireless source to stimulate preosteoblast cells using the piezoelectric composite P(VDF-TrFE)/Terfenol-D. 107 However, to the author's knowledge, there has not been a model created that explicitly tests the effects of the converse piezoelectric effect to mechanically stimulate bone growth. This could be owing to the difficulty in distinguishing between the occurrence of the direct and converse piezoelectric effects using stimulation methods such as ultrasound and also the risks associated with studying the transmission of electrical power inside the body.
Wireless control of a scaffold using the converse piezoelectric is an innovative approach to bone healing, not only because is there a potential to provide the dynamic mechanical stimulus needed to promote bone formation, but also because osteoinductive techniques and stem cell recruiting factors can be integrated into a single repair material. Thus, one could envision a synthetic bone graft substitute capable of enacting the proper stimuli to the fracture repair site, with mechanostimulation exerted through remote control.
Wireless power transmission has been investigated in providing a remote power supply to biomedical implants to eliminate the need for uncomfortable wires and batteries 108 ; however, it has not been used for the application of the converse piezoelectric effect. Issues with developing novel wireless power sources for in vivo applications include ensuring that the devices minimize discomfort and function in the biological environment.
Radiofrequency (RF) is a common method for wirelessly powering implants 109 that has not been explored in the field of piezoelectric stimulation. RF power transmission has been shown to maximize the power transfer efficiency of millimeter-sized implants. 110 In addition, low-frequency power transfer technology can be used to avoid RF radiation hazards or toxic effects because of its low operating frequency. 109 Resonant resistor–inductor–capacitor (RLC) circuits are often used on the implant surface, which has an integrated coil that can amplify voltage. 111 An AC/DC converter is used to create a constant DC supply from the amplified voltage, which could then power the implant. 111
The need for a biocompatible wireless power supply has also been assessed for completely biodegradable implants that avoid invasive removal surgery.111,112 Biodegradable wireless control can be achieved by using metals that can resorb in the body, such as magnesium or iron, to construct a device. 111 A biodegradable RLC resonator from the biodegradable PLLA-PPy and PCL-PPy polymer composites and the metals Mg, Fe, along with Mg− and Fe− alloys, could be the basis of a completely biodegradable and wirelessly powered scaffold. 113 Optimizing RF wireless power transmission to generate the converse piezoelectric effect could not only enhance the repair process in bone but also support the transmission of data such as level of mechanical stimulation or temperature to a receiver. 114 Harnessing the technology of a wireless power supply that can relay information about the implant site and merging this with wireless control of the converse piezoelectric effect has exciting potential for clinical applications.
Conclusion
To mitigate the disadvantages of bone grafts to repair serious bone injuries, including high cost and limited availability, synthetic bone graft substitutes are being explored. However, it is difficult for these substitutes to promote the natural signaling processes in bone healing. Smart piezoelectric materials have gained special attention to address these problems because they can mimic natural bone bioelectric signals through either the direct piezoelectric effect or inversely provide mechanical stimulation through the converse piezoelectric effect. The direct piezoelectric effect has been extensively studied, whereas the converse effect, although important, has been neglected. This review has focused on several reasons why the underrepresented potential of implementing the converse piezoelectric effect into the design of bone implants is worth further exploration.
The converse piezoelectric effect can prevent bone atrophy from immobilization and stress shielding because it can provide more control over the mechanical stimulation supplied to the implanted site. Mechanical stimulation in bone has a therapeutic effect through cellular mechanisms. The influx of calcium ions, release of ATP, increased levels of NO, and release of PGE2 by increased activation of COX-2 provoke key mechanisms that promote the differentiation of osteoblasts. Mechanical stimulation can also enhance growth factors such as IGF, TGF-β, and BMP, which are osteoinductive agents, increase signaling through the Wnt/β-catenin pathway and downregulate inhibitors of bone formation. There is a gap in the literature showing proof that each listed signaling pathway involved in mechanical stimulation is increased during stimulation by the converse piezoelectric effect, so future work in this area must involve these pathways being observed closely both in in vitro and in vivo.
Another important reason for exploring the potential of the converse piezoelectric effect is that there is a wide range of piezoelectric biomaterials from which to create biocompatible and biodegradable scaffolds. In addition, brittle inorganic piezoceramics and flexible piezoelectric polymers can be combined to create composites that recapitulate the natural structure of bone. Although the traditional electrical power sources such as the lithium-ion battery and bulky implants have limited the implementation of the converse piezoelectric effect into bone implants, advances in wireless control of the converse piezoelectric effect can provide a safer alternative. Further research to advance this area would involve employing new wireless sources such as RF and measuring the mechanical stimulation produced by a piezoelectric scaffold.
Thus, utilizing the converse piezoelectric effect in bone graft scaffolds is a promising line of research. However, understanding the cellular mechanisms triggered by converse piezoelectric stimulation, testing novel composite biomaterials in preclinical trials, and creating a proper and patient-accessible design for wireless control of the converse piezoelectric effect are all necessary steps to advance this technology.
Footnotes
Authors' Contributions
The authors confirm contributions to the article as follows: draft article preparation, literature review, analysis, and figure and table preparation: A.C., A.M., and K.P.; draft article preparation and revision: A.G., F.S.L., and K.C. All co-authors have reviewed and approved this article before submission to this Journal. This article has been submitted solely to this Journal and is not published, in the press, or submitted elsewhere.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Funding and support were provided by the Comparative Medicine Institute (CMI) through the National Institute of Health grant T34GM131947 and the CMI's Summer Interdisciplinary Research Initiative (SIRI) and the Ross M. Lampe Chair of Biomedical Engineering.
