Abstract
Latin America (Mexico, the Caribbean, and Central and South America) is a megadiverse region. From Mexico and the Caribbean, through Central America to the Amazon and Andes, Latin America’s botanical diversity contributes significantly to global food production and security, as it is the region of domestication for crops essential to human survival, including maize, potato, cassava, sweet potato, and beans. However, Latin America is also one of the most threatened regions with massive loss of native habitat where cultivars of these domesticated crops and their wild relatives, invaluable for new traits important for the adaptation to the abiotic and biotic challenges from climate change, currently exist. Many of these crops are vegetatively propagated, and their unique allelic makeup is crucial to conserve. Unfortunately, these unique genotypes cannot be conserved as seed and are at risk of vanishing forever due to the lack of secure conservation methods. The Global Plant Cryopreservation Initiative (GPCI) is addressing this challenge by providing a secure, long-term method for conserving plant diversity which cannot be conserved as botanical seed. The Latin America hub for the GPCI, at the International Potato Center (CIP) in Peru, is a model for how to mobilize and build capacity at the country level through national programs to conserve their genetic resources. Starting with a regional in-person workshop in 2022, CIP has maintained momentum for a plant cryopreservation network in Latin America by holding annual regional virtual cryopreservation meetings. CIP provided in-person capacity training for Latin American researchers in plant genetic resources conservation, with a special focus on cryopreservation. CIP has also implemented national plant cryopreservation programs in Ecuador and Chile, which include backing up their accessions in CIP’s “cryo-vault.” These activities provide the foundation for the future of a strong, lasting plant cryopreservation community of practice throughout Latin America.
Introduction
The FAO Second Report on the State of the World’s Plant Genetic Resources for Food and Agriculture 1 estimates that over 1750 genebanks worldwide maintain approximately 7.4 million accessions of plant genetic resources. While the report estimates that only 25%−30% of these accessions are genetically unique, it is important to recognize that the diversity within these collections represents the lifeblood of current and future efforts to feed the world in a time of drastically changing climate, catastrophic dwindling soil and agricultural land, and a ballooning human population. Recent reports on climate change indicate that 2024 was the hottest year on record since temperature measurements began, 2 exceeding the 1.5°C limit for maximum temperature increase set by the Paris Agreement for the first time. 3 The need to conserve plant diversity is more dire today than it has ever been in the history of human civilization.
Ex situ plant genetic resources collections in genebanks primarily focus on conserving the diversity of agricultural crops and their crop wild relatives (CWR). The overwhelming majority of genebank holdings are stored long-term (decades) frozen as desiccated botanical seed (termed orthodox seed) at −18°C. Such long-term storage facilities for orthodox seed (seed that can withstand desiccation to below 10% moisture content) range from small regional genebanks using household freezers to very elaborate facilities with large walk-in freezers. One of the largest global genebanks for botanical seed is the Svalbard Global Seed Vault in Norway, which serves as a safety backup facility for global orthodox seed genetic resources collections. As of January 2025, the Svalbard Global Seed Vault holds 1,331,458 seed samples from 1184 genera and 6297 species of plants from 195 different global depositors of samples, 4 of which 33 countries (17%) are from Latin America (which, for the context of this article, refers to Mexico, the Caribbean, Central America, and South America).
It is estimated that approximately 88% of higher plant species produce orthodox (desiccation-tolerant) seed.5,6 These species typically originate from temperate environments, such as northern North America, the Mediterranean region, and desert and savannah ecosystems, which are characterized by periodic seasonal droughts, 7 and thus, the vast majority of plant diversity can be safely conserved as frozen botanical seed. However, these ecosystems, and the species that originated in them, are not ubiquitous or evenly distributed globally. In contrast, the remaining ∼12% of higher plant species that do not produce orthodox desiccation-tolerant seed (termed recalcitrant seed) generally are found in humid, moist, and warm ecosystems with infrequent and minimal seasonal variation typical of tropical regions and the equatorial zones of the world, including Latin America. However, the boundary between orthodox and recalcitrant seed is not clear-cut. The boundary is gray and filled with plant species that are either partially desiccation tolerant or are desiccation tolerant but short-lived (<20-year half-life) at −18°C (a subset of this group is often termed as seed with intermediate storage behavior). For clarity in this article, we refer to seed which is either partially desiccation tolerant or has a half-life of <20 years when desiccated and stored at −18°C as short-lived orthodox seed. As will become evident, it is important to clearly draw this distinction because both recalcitrant seed and short-lived orthodox seed are priority candidates for long-term cryopreservation in genebanks.
Moreover, it is critical that we introduce a fourth category (in addition to orthodox seed, short-lived orthodox seed, and recalcitrant seed) in need of long-term conservation: plant genetic resource collections of clonal or vegetatively propagated material. Five (50%) of the top 10 crops critical for human survival 8 are clonally propagated (potato, cassava, sweet potato, yam, banana/plantain), and three of these crops (potato, cassava, and sweet potato) are native to and were domesticated in Latin America. For vegetatively propagated crops, it is the unique allelic combination in each individual which is important to conserve. Thus, genetic resource collections of these crops are overwhelmingly conserved as clones or living plants. Therefore, these crops (clones) are maintained either in field plantings, glass/screenhouses, or in vitro in sterile tissue culture, which needs to be regenerated every 1 to 3 years depending on the crop, increasing the risk of mixture or contamination. The risk of loss to these collections is highest when maintained in the field due to both biotic and abiotic challenges. However, the maintenance cost of a field collection is usually lower than the maintenance cost of an in vitro collection. For this reason, many plant genebanks, particularly in the Global South, can only maintain their collections as field collections, where they are subject to the highest risk of loss. Unfortunately, for most clonal genetic resources collections in Latin America, the establishment of an in vitro culture is often a prerequisite for successful cryopreservation.
The Global Plant Cryopreservation Initiative
The Global Plant Cryopreservation Initiative (GPCI) was conceived to provide safety backup capacity for the most at-risk recalcitrant and short-lived seed and clonal crop genetic resources collections in the Global South. It is important to emphasize that these collections cannot be safely backed up in the Svalbard Global Seed Vault, and thus there is no safety net for these irreplaceable genetic resources if they are lost at their primary site of storage. A study was commissioned in 2017
9
to determine the feasibility and need for the creation of a global cryopreservation safety back-up for these at-risk genetic resource collections, akin to the Svalbard Global Seed Vault. The primary recommendations from the study were:
Cryopreservation is the best long-term conservation option for clonal and recalcitrant seed crop collections.
Cryopreservation has lower running costs, increased longevity and security in storage, and provides greater genetic stability. However, the study recognized that cryopreservation has higher initial costs, as well as a need for a higher level of skill and technical challenges. Cryopreservation has huge benefits for the long-term secure backup of plant genetic resources collections.
Maintenance of collections in vitro is costly, requires continual replenishment, and is difficult to transport, compared to seed, for safety backup. The infrastructure needed to implement and maintain a safety backup cryofacility would be modest (5K accessions), but this need is expected to double in 10 years. There is an immediate need to accelerate the development and implementation of cryopreservation to safeguard clonal and recalcitrant seed crop collections.
Approximately 100,000 Annex 1 accessions (Annex 1 crops are crops covered by the Multilateral System of the International Treaty on Plant Genetic Resources for Food and Agriculture [ITPGRFA]) are currently at risk in field and in vitro genebanks, which could be securely backed-up in a cryopreservation facility. The Consultative Group on International Agricultural Research (CGIAR) is ideally positioned to develop proposals and to seek donor sponsorship for a cryopreservation backup facility for clonal and recalcitrant seed crop genetic resource collections.
These recommendations very concisely spell out the need for and the path forward for a GPCI. Clonal, recalcitrant seed, and short-lived orthodox seed genetic resources collections are critical to human survival now and in the future. Currently, non-cryopreserved methodologies for their long-term conservation are neither secure nor sustainable. The need is greatest in the most megadiverse regions in the world, where technology, funding, and policy for conservation are most lacking. Although not a panacea, nor a simple cure-all, cryopreservation of these at-risk plant genetic resources collections would provide the only secure method for long-term conservation. The need is urgent, as, in the long-term, a mechanism is required to provide the technology, expertise, skill, and where-with-all for the cryopreservation of more than 100,000 invaluable accessions in global collections!
The GPCI is the global community’s answer and response to this call. The GPCI is focused on the most vulnerable, at-risk, and in-need plant genetic resources collections, including recalcitrant and short-lived seed and clonal crops critical to human survival. The GPCI is initially centered around three cryohubs, each with a mandate to facilitate the cryopreservation of plant genetic resource collections in their regions (Latin America hub and Africa hub) or to use their expertise and facilities to enhance ongoing global cryopreservation efforts for plant collections that need additional capacity and help to implement cryopreservation programs for safeguarding at-risk plant genetic recourses collections (European hub). All three hubs are housed in CGIAR centers with cryopreservation infrastructure and know-how: the International Institute of Tropical Agriculture in Ibadan, Nigeria; Bioversity International in Leuven, Belgium; and the International Potato Center (CIP) in Lima, Peru. The best example of how these hubs operate is the Latin America hub at CIP, where a new large-capacity cryobank was recently inaugurated (21 November 2024) as part of the GPCI.
Under the terms of the ITPGRFA, genetic resource collections must be available freely for use for training, breeding, and research. This component of plant genetic resources collections is key to maximizing value and return on investment for the collections, as they are not meant to be simply locked away but rather used for the betterment of human livelihoods and survival. Thus, the GPCI has a novel and unique key “use” element for the genetic resources cryopreserved, which has been trialed for over two decades by CIP, 10 where material is collected from farmers’ fields under the terms of the ITPGRFA and transferred to CIP for long-term secure storage in the in-trust potato collection for use by humanity. Once at CIP, the material is phytosanitary cleaned and repatriated to smallholder farmers throughout Peru for planting. Between 1997 and 2020, almost 15,000 samples from over 1500 accessions were repatriated to 139 communities in Peru. This “Clean and Share” model is continuing in Peru and has been incorporated as part of a Darwin Initiative funded program with sweet potato in Madagascar and Zambia in support of the GPCI (D. Ellis, personal communication).
Latin American is a diversity hotspot
Latin America is critical because it is home to a number of extremely diverse agroecological regions that harbor a high concentration of crop diversity. 11 Latin America includes two of Vavilov’s original eight centers of crop diversity—Mexico-Guatemala and South America/Andean regions (in three subgroups, Peru-Ecuador-Bolivia, Southern Chile, and Southern Brazil)—which have been maintained through subsequent revisions.12–13 Harlin, 14 using Vavilov’s teaching, revised the concept of centers of crop diversity by identifying three global regions in which the domestication of plants originated. This included Central America as one global region and South America as a “non-center,” signifying a region that is more dispersed, interacting with the primary center.15,16 A recent update of the global diversity “hotspots,” regions with a high diversity of plants found nowhere else in the world (at least 1500 endemic vascular plants) and which are threatened with only 30% or less of their original vegetation remaining, described Latin America as containing eight of the 35 global biodiversity hotspots. 17 Furthermore, the Tropical Andes (the Andean mountains of Venezuela, Colombia, Ecuador, Peru, Bolivia, and the northern tropical regions of Argentina and Chile) is listed as the most diverse hotspot in the world. 18 Obviously, Latin America is treasured for its biodiversity. However gradual, yet continual degradation and destruction of native landscapes has continued for at least the past 500 years. Native plant ecosystems have been cleared for agriculture, grazing, timber, firewood, minerals, and urbanization, with over 90% of the original Atlantic rainforest cleared, 6 not to mention the impacts of climate change on the native biodiversity. A study by Kew Botanical Gardens 19 estimated that approximately one-third of the world’s threatened plant species listed in the IUCN Sampled Red List Index for Plants are contained within Latin America and the Caribbean region.
Clearly Latin America is a prime candidate and in dire need of secure conservation of its genetic resources to ensure these irreplaceable resources are available for future generations. Crops native to and domesticated in Latin America contribute an estimated 35% of global food production, a percentage higher than any other single global region. In addition, an estimated 56% of Africa’s food basis corresponds to species from Latin America. 20 Of the four species that combined feed more people than all other species put together, two are native to Latin America (maize and potato). Furthermore, one of these four species, potato, is clonally propagated, meaning its genetic diversity collections cannot be securely safeguarded in the conditions at the Svalbard Global Seed Vault.
Although the Convention on Biological Diversity (CBD) has provided significant attention, legal rights, and obligations for parties, including virtually every country in Latin America, to conserve their genetic resources, these legal instruments have thus far only provided limited stimulus and funding for the conservation and sustainable use of plant genetic resources in Latin America. Interestingly, if one takes the seven food species with the largest quantity of samples in ex situ genebanks that originated in Latin America, only one, maize, has the highest percentage of ex situ samples (12%) conserved in a national genebank, Mexico’s national genebank. For the other six crops, the largest genetic resources collections are not in the countries national collections but are instead held in the international IRACs (CGIAR) genebanks, including Phaseolus (15%) and cassava (21%) at the International Center for Tropical Agriculture (CIAT) in Colombia and potatoes (20%) and sweet potatoes (20%) by CIP in Peru, in addition to major national programs in the Global North such as peanuts (27%) and tomatoes (30%) by the USDA in the United States. 20 It is important to note that the general lack of large national genetic resources collections of their native plants by Latin American countries is not due to a lack of interest but rather a lack of investment in infrastructure for national programs, as well as a lack of sustained support for their collection and conservation. Under the CBD, the Global Environmental Facility (GEF) is the institutional structure to dispense funds for conservation and sustainable use initiatives at the national level. However, from 1992 to 2012, Latin America received only 1.8% of the overall GEF funds, and only seven of 211 projects were allocated to the region. 11 Thus, in the absence of these countries having the ability to securely protect their genetic resources for future generations, there is an even greater urgent need to securely safeguard these precious resources for future generations before they vanish forever.
An example that illustrates how increased resources could significantly enhance the conservation of these genetic resources, which are so vital for food security, can be found in Brazil. The country possesses a well-established infrastructure for cryopreservation, housed at one of its research centers, Embrapa Genetic Resources and Biotechnology (CENARGEN). This center is strategically positioned to serve as the primary facility for the secure storage of long-term duplicates from germplasm banks across the country. In addition, within the framework of the Latin American Cryopreservation Network proposal, CENARGEN could operate as a secondary hub, with the capacity to support other countries in the development and implementation of cryopreservation methods for multiple crops, thereby consolidating its role as one of the leaders in cryobanking in Latin America and the Caribbean, alongside CIP. However, the lack of investment and limited resources for routine operations have hindered progress in advancing these initiatives. This challenge is also faced by other Latin American national programs, which, despite having basic infrastructure for cryopreservation, could play a much more significant role if adequately supported.
CIP as a center of excellence in plant cryopreservation in Latin America
The Latin American hub for the GPCI, located at CIP in Lima, Peru, is collaborating with national programs to ensure options for the secure long-term ex situ conservation of these critical botanical genetic resources through cryopreservation. This approach is particularly important for those resources that cannot be conserved as botanical seed. CIP is responsible for maintaining the global collections of potato, sweet potato, and nine other Andean Root and Tuber Crops (ARTCs)—achira, arracacha, oca, mashua, mauka, ulluco, yacon, ahipa, and maca—which are held in trust for humanity under the ITPGRFA. All the in trust genetic resources collections managed by the CIP genebank, except for ahipa and maca, are vegetatively propagated, and more than 90% are maintained as in vitro cultures. These cultures are costly to maintain (∼100 USD/year/accession), susceptible to widespread contamination (i.e., thrips, mites, microbes), and prone to human error due to the constant need for subculturing. 21 While the initial cost of cryopreserving a potato accession is relatively high (∼$450–500 USD/accession), the annual maintenance cost once in cryo is extremely low (∼$7 USD/accession/year, which includes periodic tank and liquid nitrogen generator replacement). This is in stark contrast to the routine cost (∼$100 USD/year/accession) of maintaining an accession in tissue culture. 22 It is evident that after just 5–6 years the upfront cost for cryopreservation is fully recovered. From this point forward, the annual maintenance cost for an accession is below a tenth of the cost of maintaining it in vitro, which is a significant savings considering that cryopreserved collections will remain viable and usable for hundreds of years or more. And there is the added benefit in cryopreservation with the drastic reduction in the risk of genetic changes (somaclonal variation and epigenetic changes) in an accession due to long-term maintenance and serial transfer of in vitro plant material.
There are numerous reports from many labs in Latin America on the success with cryopreservation in various plant species,23,24 however, the vast majority of these reports are small-scale protocol development research articles focused on a limited number of genotypes. This is likely due to funding limitations as well as the genotype-specific response to existing plant cryopreservation protocols. Finally, the evaluation of plant recovery after thawing from liquid nitrogen has evolved over the years. Some results that were categorized as “recovered” 1–2 decades ago would now only be classified as “survived” samples, as they did not develop into complete in vitro plants that could survive when moved out of in vitro (i.e., containing a functional apex, stem, roots, and leaves). As a result, reports of large-scale use of cryopreservation for genetic resources collections in the region remain very limited. It is important to note that Vollmer et al. 22 lists only six major cryopreserved plant genetic resources collections globally, so Latin American national programs are not unique in the limited number of genotypes cryopreserved. CIP, however, is an exception in not only having a large cryobank with ample vials per accession to accommodate use, but it also has a long history in cryopreservation dating back over 30 years. 25 Due to its legacy in cryopreservation and status as the custodian of the in trust collections for potato, sweet potato, and ARTCs, CIP is a logical choice as the GPCI hub for Latin America. Additionally, as a global International Agricultural Research Center (IARC), CIP possesses the necessary cryopreservation expertise with the largest plant cryopreservation lab in terms of personnel and achievements worldwide. Therefore, it has the infrastructure, long-term funding outlook, and mandate for capacity building and enhancing the long-term conservation of valuable plant genetic resources in the mega-diverse Latin American continent.
The newly constructed state-of-the-art cryobank at CIP is earthquake resistant and offers significant expansion capacity to meet growing needs. In 2013, CIP launched a comprehensive and innovative plant cryopreservation quality management system for their cryobank, which now serves as a global model for plant cryobanks. It is important to note that the purpose of cryobanking plant genetic resources collections is to preserve living material for future generations, ensuring it can be accessed, regenerated, and used as viable living plant material as needed. Thus, verified, accurate, and trusted viability data for whole plant regeneration is essential, as is having a sufficient number of individual vials available for multiple pulls from the cryobank to ensure a sustainable supply to meet breeding and research needs for decades or centuries to come. Finally, the legacy of plant cryobanks in safeguarding genetic diversity is to guarantee that future cryobank managers, centuries from now, will know exactly what is stored in the cryobank, how to access and regenerate it, and what to expect when retrieving a vial out from the cryobank for regeneration.
It is important to note that since the first successful report of the survival of a complex shoot meristem from cryopreservation, 26 the cryopreservation of plants has undergone a long history of optimization. However, this progress has been achieved with relatively little coordination, and research efforts have largely been carried out independently in various laboratories worldwide. As a result, plant cryobanks now store a vast assortment of different types of material, employing a variety of different cryopreservation methods, each with evolving and ever-changing quality management systems. Despite there being only three main cryopreservation methodologies for plants (vitrification, dehydration, and two-step [or controlled-rate] freezing), each method incorporates a myriad of variables. These include options such as ±encapsulation, different pretreatments, and varying approaches to thawing and regeneration of whole plants. Looking at the progress of plant cryopreservation over the past 50 years, it is evident that the field has evolved significantly. In the 1980’s plant cryobiologists were satisfied recovering a cell that could divide. This focus shifted in the 1990s to defining success as the recovery of material that was green, indicating the capacity to be photosynthetically active. By the early 2000’s, the standard for success had evolved to the recovery of meristematic tissue, and it is now generally recognized that success, particularly for the cryopreservation of genetic resources, requires the recovery of whole plants that can survive outside the in vitro environment and grow normally. Although the standard for recovering whole autotrophic plants is largely accepted worldwide, there is huge variation in how this standard is interpreted. This has led to significant differences in the types of material stored in various plant cryobanks. Today, one of the challenges facing plant cryobanks is that with limited funding for the cryopreservation of new material, many global plant cryobanks are likely to have a high percentage of subpar material in their cryobanks, which will unfortunately be passed on to future generations, often without clear transparent data.
The best example of this to our knowledge was in 2013, after CIP established very strict written guidelines for parameters that would qualify for long-term conservation in their cryobank, 27 CIP took the unprecedented step of evaluating everything that had been stored in their cryobank prior to 2013. The authors say “unprecedented” because, to our knowledge, no other plant cryobank has taken the major step to do such a thorough evaluation of their existing cryobank and rationalized the stored material based on updated quality standards. To put the 2013 reevaluation of CIP’s cryobank into perspective, it is important to outline the quality management guidelines that were put in place for the CIP cryobank.
In general, prior to qualifying for cryopreservation at CIP, an accession must be fully curated (contain complete passport data available), be genetically unique within the collection, have DNA fingerprints for long-term quality control, and be certified as phytosanitary clean. Once an accession meets these criteria, CIP places 10 vials per accession into long-term cryoconservation, with 10 shoot tips per vial, into its main cryobank at the CIP headquarters in Lima, Peru. In addition, 2–4 vials, depending on the baseline recovery rate, are set aside for international safety backup. More recently, a supplementary national safety backup cryobank was established in the highlands of Peru (Huancayo, >3200 m.a.s.l), (currently: 1800 accessions). The minimum accepted full-plant recovery rate is 30% or higher, based on a sample size of 30 shoot tips for baseline testing. Exceptionally, recovery rates of 20%–30% are accepted, but only on the condition that a new cryo-run is processed to increase the total sample size and mitigate risks. Cryo-runs with an average recovery rate of less than 20% or signs of contamination are discarded and never transferred to the cryobank. The average full-plant recovery rate per vial of CIP’s potato cryo-collection is >70%, which aligns with our philosophy of requiring the removal of only a single vial from LN (liquid nitrogen) to recover plants when necessary, maintaining the cryobanked stock at an optimal level. The viability of all accessions in the cryobank is reassessed after at least 1 year in LN by thawing one vial to ensure no errors occurred during sample transfer and no hidden contamination is present. Each step of the cryopreservation protocol—starting from in vitro culture, through pre-conditioning (i.e., cold acclimatization), shoot tip excision, cryoprotection, freezing in LN, thawing, media used, recovery assessment, transitory and final storage (location and quantity), viability reassessment, and safety backup—is recorded in a Laboratory Information Management System (LIMS) database using real-time tracking with QR codes. As a result, an uninterrupted chain of process and record tracking has been established (Fig. 1).
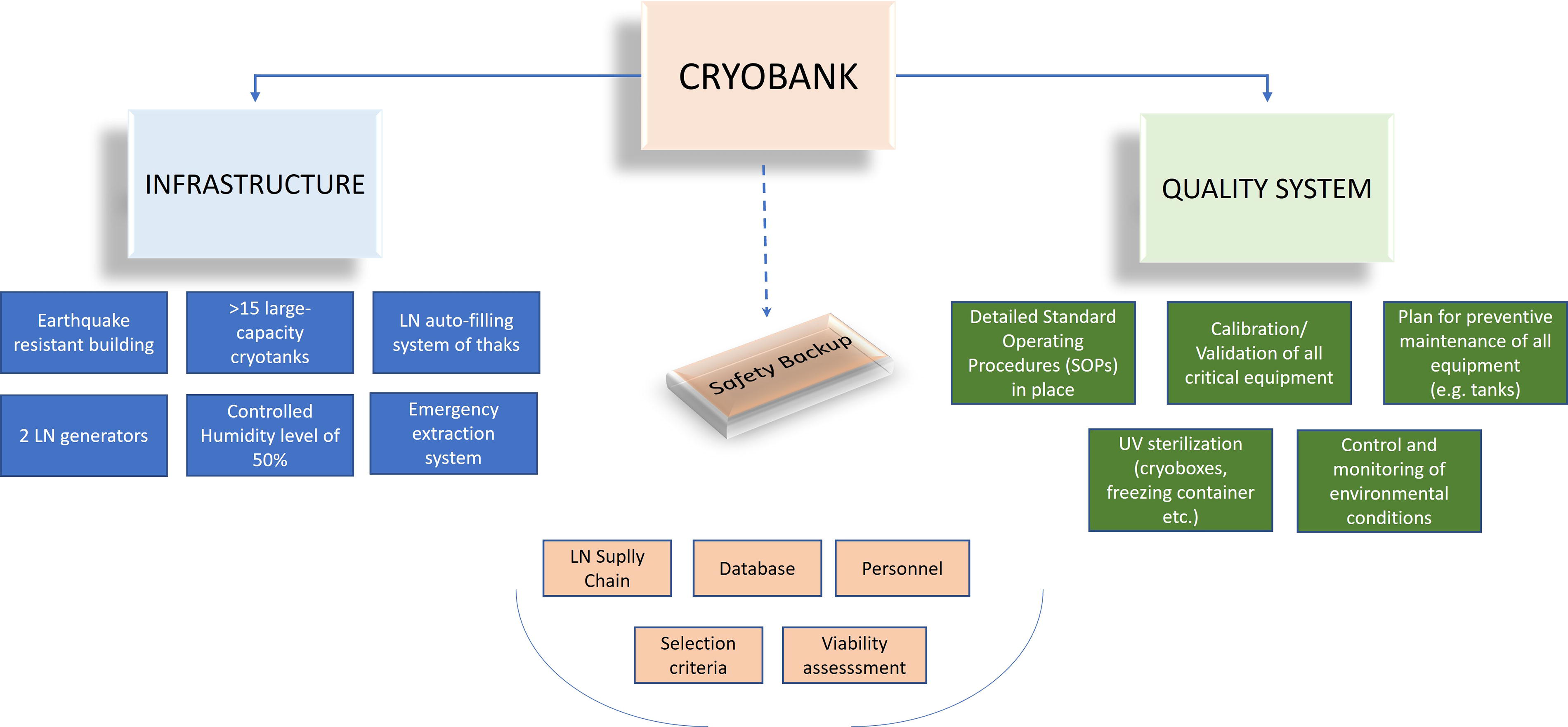
Schematic of the process chain and key elements in the CIP cryopreservation program.
The pre-2013 CIP potato cryobank contained material categorized into five groups based on the year the material was put into the cryobank and the method(s) used to cryopreserve the material. In summary:
Group 1 (1996–2000); This group consisted of materials cryopreserved using the vitrification methodology of Steponkus et al.
28
where excised potato axillary shoot tips were vitrified in a solution containing ethylene glycol: sorbitol: bovine serum albumin (50:15:6 wt%) for 50 minutes at room temperature.
25
A variety of variables were tested in a non-replicated manner with 1-5 straws/accession from 288 accessions stored in the cryobank. Group 2 (2004–2006); This group consisted of potato shoot tips vitrified in plant vitrification solution-2 (PVS2), which contains 30% glycerol (w/v), 15% ethylene glycol (w/v), and 15% DMSO (Dimethylsulfoxide) (w/v) mixed with 0.4 M sucrose (w/v) dissolved in liquid Murashige and Skoog medium.
29
The shoot tips were exposed to varying periods of time in PVS2, with each exposure replicated 3 times with a single straw. 61 accessions were conserved in the cryobank in this time period. Group 3 (2006–2009); This group consisted of potato shoot tips cryopreserved using the droplet PVS2 vitrification method. The time variables were the same as in Group 2, but in this case, shoot tips were vitrified in droplets of PVS2 on aluminum foil strips, which were then placed in cryovials for cryopreservation.30–32
Experiments were conducted in triplicate, with 146 accessions cryopreserved using 1–3 vials/accession. Group 4 (2006–2009); This group consisted of potato shoot tips that were either cold acclimated or not prior to shoot tip isolation and cryopreservation after a 50-minute exposure to PVS2, as described for Group 3. Experiments were done in duplicate, with 492 accessions using 1–2 vials cryopreserved per accession. Group 5 (2009–2012); This group consisted of 507 potato cold acclimated accessions. After shoot tip excision, the shoot tips were placed in a droplet of PVS2 on an aluminum foil strip prior to cryopreservation. Cryopreservation runs had a variable number of replications per accession (1–2) and each accession was represented by 1–4 vials in the cryobank.
A priori, it was known that the majority of the pre-2013 material in the potato cryobank did not meet the new standards. The vast variation in methodology used over nearly two decades would likely have left a legacy of dramatic inconsistencies, particularly in the predictability of results when thawing individual vials. This would have posed significant challenges for future managers wanting to use the materials in the cryobank. Looking at the collective data from these five groups, 27% of the accessions had viabilities below 20%, which were therefore deemed unacceptable by current standards. In addition, the number of vials per accession in all groups was well below the required amount for a plant genetic resources cryobank. Any accessions deemed worthy of preserving would have to be recryopreserved to ensure ample vials for the future. Nevertheless, the reevaluation of the material was initiated on the assumption that some of the material might still meet the current standards for long-term storage and inclusion in the CIP cryobank.
For Group 1, 30% of the material in the potato cryobank lacked historical records, and thus, a 17% subset (48 accessions) was randomly selected for testing. From this subsample, 35% of the accessions were found to be contaminated upon thawing, and plants were only recovered from 4 accessions (∼8%). With these results, it was determined that the remaining cryopreserved material in this group was not worth the time to test and thus was removed from the cryobank and discarded. For Group 2, there was considerable inconsistency in the number of cryopreserved straws, and a small subset of 6 accessions (10%) were thawed. Not a single plant was recovered from these accessions, and again, it was decided that none of the accessions from this Group should be retained. Groups 3 and 4 were complicated by the use of multiple aluminum strips in each cryovial. A subsample of 98 vials (18%) was thawed. 12% of the thawed samples were found to be contaminated, and again due to poor recovery, small sample size, multiple methods used, and multiple aluminum strips per vial, these accessions were also deemed unsuitable for long-term storage and were removed from the cryobank and discarded.
In summary, all material cryopreserved prior to 2010, along with >50% of the remaining potato cryobank in 2013, was deemed unacceptable under the 2013 guidelines and discarded. This reduced the CIP potato cryobank from 1205 accessions to <400 accessions (baseline for 2013). Fortunately, material in Group 5, cryopreserved from 2010 to 2012, contained good documentation and was deemed 72% acceptable despite low numbers of vials and multiple foil strips being present in a single vial. In cases where multiple foil strips were present, these were separated under LN, and a single strip was placed in a new vial to ensure quality control in future processing. In cases where the number of vials was below the standard (10 vials in the cryobanks + 2–4 for safety backup), these accessions were recryopreserved to ensure an adequate number of vials were available in the cryobank for the future. It should be noted that for readers unfamiliar with plant genetic resource collections, all (100%) of accessions discarded from the cryobank were still maintained as in vitro cultures; hence, no genetic diversity was irreversibly lost. Moreover, the discarded accessions were later reintroduced to cryo following the 2013 guidelines. Table 1. summarizes the evolution of the CIP cryobank.
The Evolution of the CIP Potato Cryobank (1996–2025)

Quality Management System.
Three cryovials from LN at 1, 2, 4, and 8 years and assessed for viability (whole plant recovery). The plan is to continue removing vials and assessing viability at 16-, 32-, and 64-years post-cryopreservation.
As part of the newly established cryobank quality guidelines, CIP introduced a careful monitoring program of processes, which led to an increase in the numbers of plants regenerated from each vial. As a result, the percentage of vials (10 shoot tips/vial) with over 50% of the shoot tips regenerating into whole plants from the CIP cryobank increased from approximately 35% in 2012 to over 60% in 2018. Furthermore, the number of accessions cryopreserved per year increased from around 100 in 2012 to >500 in 2018. At present, the CIP cryobank holds 4660 potato accessions, representing about 90% of its total collection, with an average full-plant recovery rate of over 70% (+LN). Approximately 1850 of these accessions (∼40%) also have a safety back-up as cryopreserved material at the national level (in Huancayo, 3250 m.a.s.l). It is planned to fully back-up CIP’s potato cryo-collection on two levels—one nationally and another internationally within the next 3 years. The new quality guidelines empowered all staff to strive for excellence, leading to efficiencies gained in all areas of the cryopreservation process, from pretreatment of material before cryopreservation to all phases of thawing and plantlet recovery. The higher throughput translated into cost savings and enhanced long-term confidence and security for the entrusted collections.
Another unique aspect of the cryopreservation quality management guidelines at CIP is the inclusion of both short-term and long-term viability monitoring programs for cryopreserved materials. In plant genebanks, accessions stored as botanical seed are germination-tested on a periodic basis (i.e., every 10–20 years) to assess when or if germination viability falls below an acceptable level. This is critical because once a botanical seed begins to lose viability, it usually declines very rapidly. 33 Therefore, monitoring the decrease in viability is essential so that fresh, vigorous seed can be rapidly regenerated prior to a loss of potentially valuable alleles due to the rapid decline in viability. While this is standard practice for seed genebanks, it is not routinely done in plant cryobanks. Since there is limited information on the longevity of organized plant tissues in cryopreservation, CIP drew on lessons from seed genebanks and initiated a viability monitoring program for all cryopreserved accessions in their cryobank. The routine guidelines at CIP involve cryopreserving 15 vials of 10 shoot tips per vial per accession. Three vials are removed from cryopreservation after a minimum of 24 hours for an initial, baseline evaluation of viability. As previously mentioned, any potato accessions with less than 20% of the shoot tips regenerating into whole plants are recryopreserved, and that first cryo run is discarded. After 1 year, 100% of cryopreserved accessions undergo a short-term viability reassessment by thawing a single vial to ensure there is no hidden contamination and that the viability of this vial falls within the standard deviation of the initial baseline viability. Currently, 99% of cryopreserved accessions (>500 per year) pass the short-term, 1-year viability retest. After both the initial viability test and the 1-year viability test, 11 vials/accessions remain. Two vials are set aside for international safety backup storage in a geographically remote location from the main cryobank, and nine vials remain in the cryobank for future use.
In 2013 CIP initiated a long-term cryobanking monitoring program cryopreserving an additional 240 shoot tips (24 vials) from 8–14 randomly selected accessions annually for long-term viability monitoring. In addition to thawing the three initial vials to establish a baseline viability for each accession, three additional vials are thawed from LN at 2, 4, and 8 years (30 shoot tips/time point) and assessed for viability (whole plant recovery). The plan is to continue removing vials and assessing viability at 16, 32, and 64 years post-cryopreservation. After this the current manager of the cryobank will decide at what intervals viability assessments are needed. As of 2022, 101 potato accessions in the long-term monitoring experiment have been assessed at 2-, 4-, and 8-years (76, 52, and 17 accessions, respectively), and no significant differences compared with the baseline viability were detected confirming the stability of potato shoot tips in the cryobank. 22
Latin America plant cryopreservation network
The cryobank at CIP stands out as a regional and global center of excellence in cryopreservation. We are unaware of any other plant cryopreservation program globally that is of sufficient scale to cryopreserve 500+ accessions annually while also running a long-term viability/recovery monitoring program that provides statistically verifiable data for future generations. With this capacity and knowledge, CIP has taken the lead in organizing a Latin America plant cryopreservation network, initially focused on national programs, to raise awareness of the need for a unified effort to conserve invaluable and vulnerable plant genetic resources in the region, which cannot be conserved long-term as botanical seed.
On August 23–24, 2022, the inaugural workshop of the Latin America Plant Cryopreservation Network (1er Taller de Criopreservación de Recursos Genéticos Vegetales en Latinoamérica) was hosted by CIP, with funding from CGIAR to support the GPCI. The workshop saw great enthusiasm from the region, as evidenced by the participation of the national programs from 14 Latin American and Caribbean nations, along with plant cryopreservation experts from Spain and the U.S., a representative from the ITPGRFA (Italy), the Global Crop Diversity Trust (Germany), and representatives from CIP’s sister CGIAR center, CIAT (International Center for Tropical Agriculture [Centro Internacional de Agricultura Tropical]) in Cali, Colombia (Table 2, Fig. 2). Each national program presented the status of their plant genetic resources conservation programs, and group discussions focused on the needs of individual countries as well as the region. While all countries reported having in vitro programs, only seven countries (Argentina, Brazil, Chile, Cuba, Ecuador, Mexico, and Uruguay) possess cryopreservation capacity at the national level. Virtually all countries reported lacking adequate cryopreservation equipment and supplies, with most identifying capacity building as a priority for the regional plant cryopreservation network.
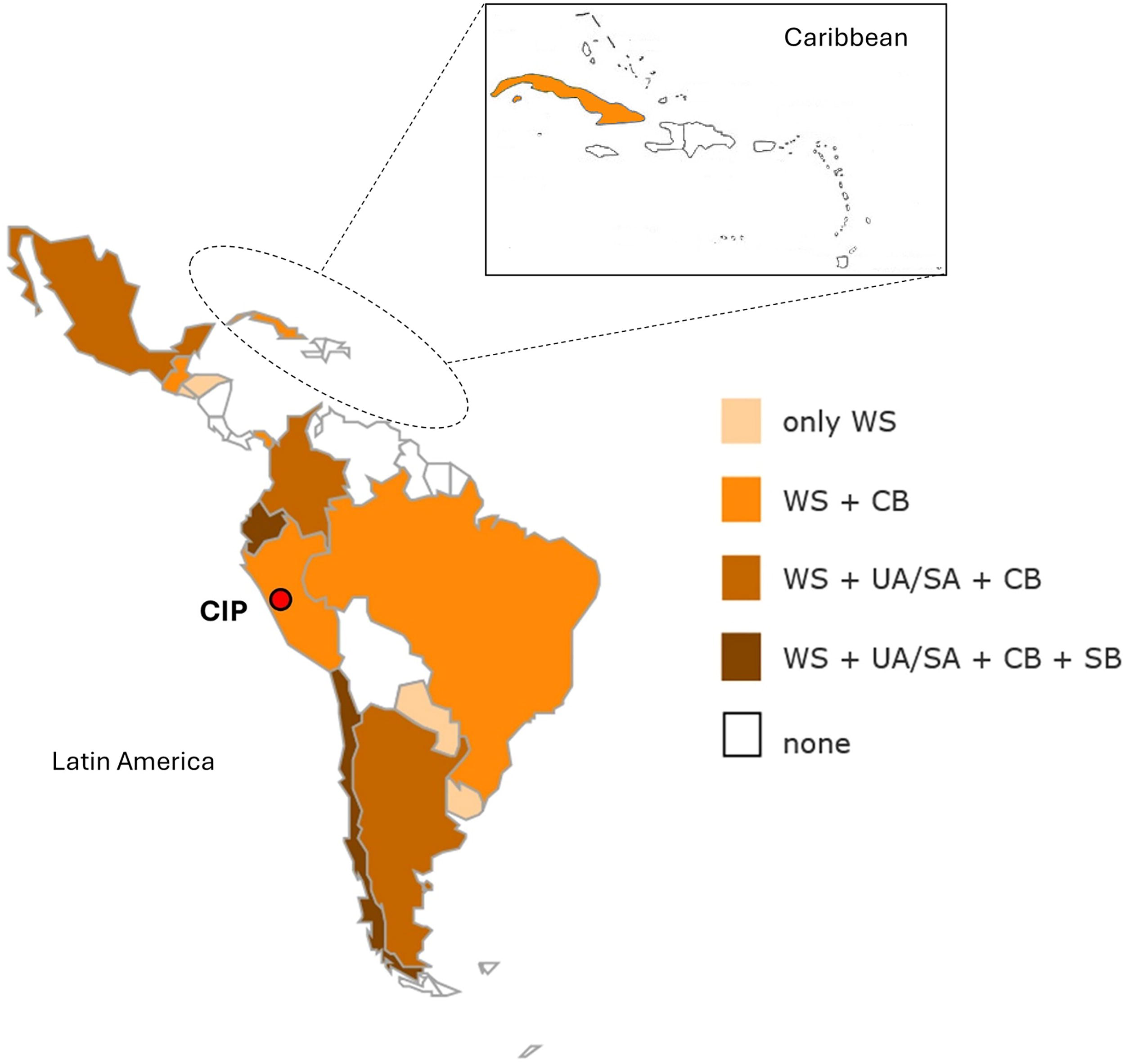
National Agricultural Research Systems (NARS) from Latin America that participated in the Inaugural Workshop of the Latin America Plant Cryopreservation Network (WS), received intensive capacity building on cryopreservation at CIP from 2022 to 2024 (CB), signed umbrella and/or specific collaboration agreements with CIP (UA/SA), and agreed to deposit a safety copy of cryopreserved material from their country at CIP (under the terms of a black-box agreement) (SB).
Attending Organizations at the 1er Taller de Criopreservación de Recursos Genéticos Vegetales en Latinoamérica (First Workshop on Cryopreservation of Plant Genetic Resources in Latin America) Held in Lima, Peru, 23–24 August 2022

Since the 2022 workshop, the Latin American Plant Cryopreservation Network has held three virtual meetings, and interest continues to grow (Fig. 3). CIP has also hosted in-person capacity-building workshops (“hands-on training”) attended by 27 researchers from 10 countries, focusing on plant cryopreservation, though the workshops covered all aspects of clonal and seed plant genetic resources conservation. Limited financial support remains the primary barrier to advancing genetic resources conservation in general and plant cryopreservation specifically in the region.
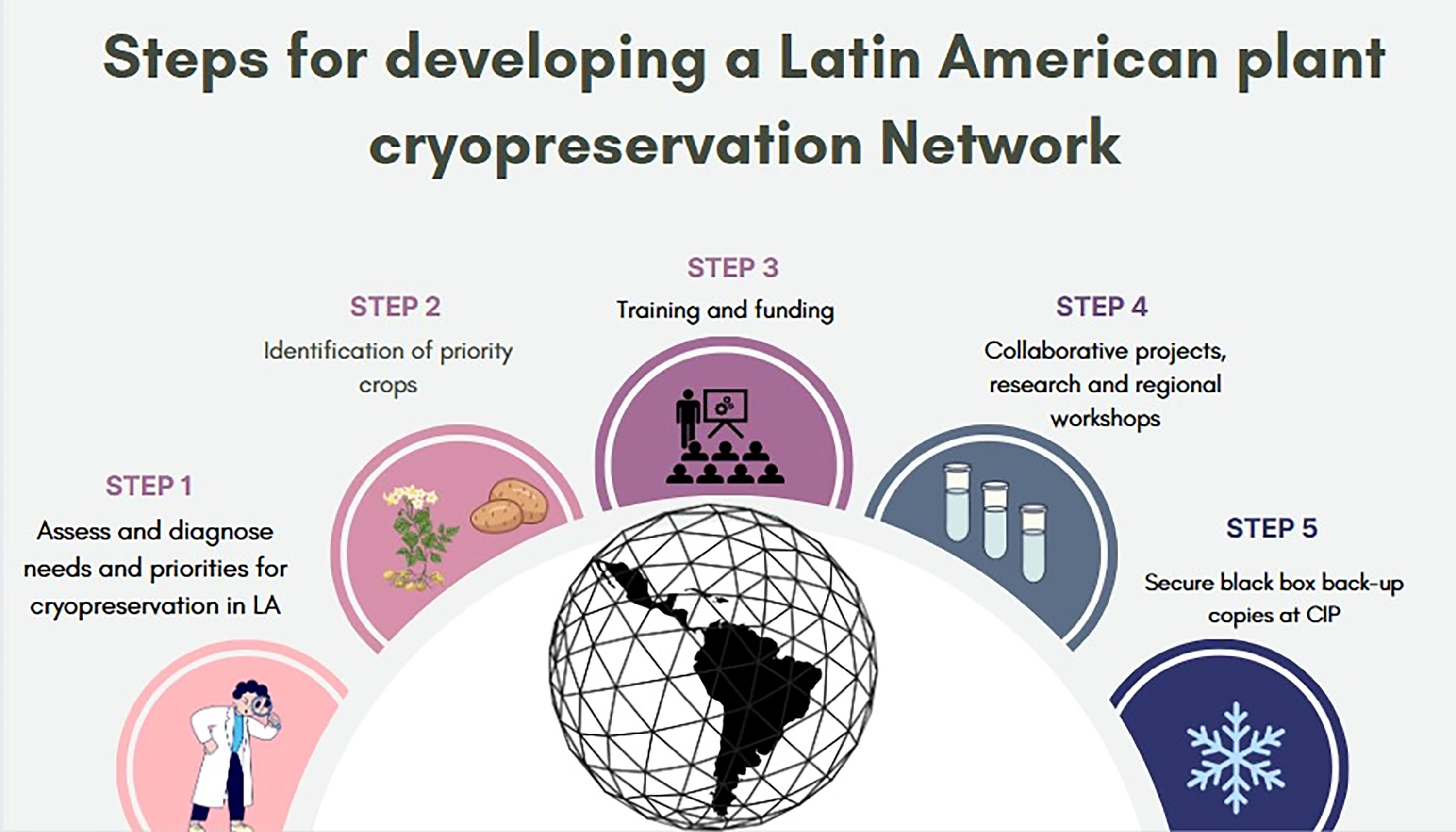
Steps currently identified for implementation of the Latin America Plant Cryopreservation Network.
In this context, CIP has used a small amount of available cross-cutting funds to help implement plant cryopreservation programs in the national programs of Chile and Ecuador. However, much greater financial support is needed during the initial phase of implementation (∼USD 1.5 million per year) to ensure the systematic, secure, and reliable conservation of plant genetic resources at the national level using the cutting-edge method of cryopreservation. As previously mentioned, once cryopreservation is implemented, costs for maintenance of at-risk plant genetic resources collections drop dramatically. These institutes have committed to depositing safety backups of their cryopreserved potato, garlic, and groundnut accessions at CIP (>100 accessions). Facilitating a safety back up at a secure location with high-quality standards is one of the most critical aspects of the Latin American Plant Cryopreservation Network, as, for example, Brazil and Argentina lost portions of their cryo-collections in recent years due to the COVID crisis and hyperinflation, respectively. A core issue with the limited and sustained cryopreservation progress for most Latin American countries is insufficient resources and investment. A critical challenge is the general lack of awareness regarding the importance of cryopreservation of plant genetic resources collections, which is reflected in the limited support cryopreservation receives from funding agencies. Despite this, in collaboration with INIAP in Ecuador, CIP contributed to the publication of a protocol for the cryopreservation of embryonic axes of groundnut. 34 A collaboration agreement is also being developed between Brazil, represented by EMBRAPA, and CIP to provide technical support for the establishment of cryobanks for cassava, banana, and pineapple, along with the safety backup of collections of other crops such as potato, sweet potato, and grapes.
Major crops of interest for cryopreservation in the Latin American region include potato, cassava, sweet potato, banana, ARTCs, pineapple, strawberry, fruit trees, forest trees, cacao, coffee, garlic, groundnut, sugarcane, grapes, and yam (Dioscorea sp.). Several countries also have specialty crops of local importance, such as vanilla, palms, and citrus. Publications on the successful cryopreservation of all these crops exist35–41 and many of these crops have large genetic resources collections stored in cryopreservation outside Latin America. Only a few crops (such as potato, cassava, sweet potato, and coffee) have more than 100 accessions in cryobanks in Latin America. Most of the crops on the priority list are vegetatively propagated, meaning they would require in vitro shoot culture as a starting material for cryopreservation. This should not be a limitation, as all countries have stated they possess in vitro capacity.
Evidence of cryopreservation efforts within Latin America, and by scientists in the region on these crops, is documented in the literature, providing a strong foundation for the GPCI to build on. Using the list of crops identified at the Latin America Plant Cryopreservation Network workshop, examples include significant efforts within the IARCs with potato,22,42,43 sweetpotato, 40 and ARTCs (Vollmer personal communication) at CIP; cassava 44 at CIAT; sugarcane and coffee45,46 at the Centro Agronómico Tropical de Investigación y Enseñanza. In addition cryopreservation research, or collaborations on such research, by scientists in national programs or universities in Latin America countries includes pineapple47–49 at EMBRAPA in Brazil; strawberry50,51 with INIA in Chile and INIFAP in Mexico; sugarcane, banana, and citrus 35 at Universidad de La Habana, in Cuba; tropical fruits including mangaba 52 by Universidade Federal de Sergipe, Brazil, and passion fruit53,54 by Universidade Do Estado De Mato Grosso and EMBRAPA, in Brazil; forest trees including eucalyptus 37 by EMBRAPA in Brazil and Cedrela odorata 55 at the Instituto Nacional de Investigaciones Forestales Agrícolas y Pecuarias, Mexico; and coffee56,57 at the National Institute of Agricultural Sciences, Cuba and Universidade Federal de Lavras, Brazil. These are some examples of many cryopreservation projects that are ongoing or being developed in several countries in Latin America and the Caribbean.
These examples provide a glimpse into the extensive interest and work being done to conserve the rich and invaluable plant genetic resources of Latin America, as well as the growing interest and need to build cryopreservation capacity for the region’s diverse flora. The region, which is under severe pressure due to climate change, is experiencing a surge of awareness and commitment to developing a strong cryopreservation program, as evidenced by ongoing engagement with the Latin American Plant Cryopreservation network and the GPCI.
Authors’ Contributions
D.D.E.: Lead writer, concept development, researcher, and editor; R.V. scientific contributor, reviewer, and editor; F.V.D.S.: Content contributor, reviewer, and editor; V.C.R.A.: Content contributor, reviewer, and editor.
Footnotes
Acknowledgments
The authors thank the CGIAR and the Global Crop Diversity Trust for support in launching the Global Plant Cryopreservation Initiative and the German Agency for International Cooperation (GIZ) for financial support for the CIP cyrovault. The authors also thank Margarita Hernandez-Ellis for thoughtful and critical review and comments on the article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
