Abstract
Background:
Biological material and health information from patients are valuable for medical research. Under a “broad” consent model, hospital patients in Norway can consent to their biological material and health information being stored in research biobanks and used for “specific, broadly defined research purposes” within a specified medical research area but not for medical research in general. Patients are asked to provide new consent each time researchers wish to use their material in a different medical research area. This study investigated patient representatives’ views on having a general consent for medical research without limitation to specific research purposes. We also investigated preferences for the storage of biological samples, the process of consent collection, and factors motivating or hindering consent.
Method:
An online, anonymous survey was shared with patient representatives from patient advisory councils at hospitals in Norway, who answered the survey on behalf of patients. A total of 157 representatives completed the survey (response rate of 41%).
Results:
A majority (66.2%) supported general consent for medical research and the use of surplus material for medical research in general (63.7%) without limitation to specific research purposes. A minority (35%) supported the use of surplus material without being informed. Sixty-five percent agreed that biological samples could be stored with no time limitation. Over half (56%) preferred to ask patients to consent prior to a hospital visit, and the majority (70.7%) supported the possibility of choosing between digital or paper consent. Factors motivating consent included the desire to contribute to medical research (89.8%) and faith in scientific progress (24.2%). Main hindrances included the fear that health information may be used for other purposes than research (49%), uncertainty regarding research uses (43.9%), and lack of information (31.8%).
Conclusion:
A move toward a general consent for medical research may better comply with patients’ wishes and maximize research potential.
Introduction
Biological material and health information from patients are valuable resources for medical research, playing a fundamental role in the development and improvement of medical practice. In Norway, patients receiving health care in public hospitals may be asked to donate extra biological material, for example, blood, urine, or tissue samples, for use within a clearly defined research project or for storage in a biobank and for use in various future projects, the details of which are not specified. In the latter case, the donor gives a “broad consent.” Broad consent is well-established in biobanking and has been argued to provide the most adequate protection of the participant’s rights when samples and health data are stored over time and used in several studies. 1 However, it has been a matter of debate how broad a consent may be without violating the principle that a valid consent must also be “informed.” The discussions have given rise to somewhat different definitions of broad consent in different countries, spanning from consent to an explicitly defined range of research projects to consent to unspecified future research. 2 While Germany has adopted a consent model supporting undefined future medical research purposes, 3 in Norway, the Norwegian Health Research Act section 14 defines broad consent as the use of human biological material and personal health data for “broadly defined research purposes within a specified area,” 4 such as in the areas of cancer or heart disease. The Norwegian Regional committees for medical and health care research ethics (REC) may add specific requirements for the use of broad consent and set limitations on its scope. If researchers wish to use the material in a project that does not fall within the wording of the existing consent, they must contact the donors and ask for a new consent. Usually, the biological material may only be stored as long as required for the completion of approved research projects and according to the terms of the consent. 4
It seems that the broad consent as practiced in Norway sets limitations to the use of biological material and health data, thereby representing a waste of valuable resources, which are sorely needed to support medical discovery. In Australia, researchers in oncology have highlighted the lost opportunities to advance cancer treatment due to the failure of current ad hoc consent procedures for biobanking in public tertiary hospitals.5–7 Studies in other countries have shown overwhelming support among patients for biological sample donation to medical research and biobanking practice in hospital settings, without a priori restrictions.5,6,8,9 Some European countries, such as Finland, already employ liberal consent models. The Finnish biobank legislation allows for the use of general consent for undefined medical and health research in research biobanks. 10 It is still an open question whether a general consent to research on biological samples and health data—with no limitation to research areas—would be acceptable to Norwegian hospital patients.
In addition to storing collected biological samples, large amounts of surplus material used for diagnostic purposes are stored in Norwegian diagnostic biobanks. Researchers can seek permission from the REC to the secondary use of such material for specific research projects without collecting informed consent, provided that the patient has been informed about the possibility and given the opportunity to opt out, according to the Norwegian Health Research Act Section 28. 4 A previous Norwegian study of patients’ opinions on research based on existing clinical data and biospecimens found that this type of “passive” consent for the secondary use of surplus material was favored by Norwegian hospital patients. 11 Nevertheless, it is not broadly practiced and most RECs require an active specific consent for each new research project planning to use surplus material, that is, the narrowest form of consent.
The main purpose of this study was to survey patient views on whether biological material and health data collected in the health service, including surplus material, ought to be used for medical research based on a general consent as opposed to the current restriction to defined research areas. We define “general consent” as “consent to medical research in general without limitations to certain diseases and diagnoses,” and we define “medical research” as “activities that are carried out using scientific methodology to acquire new knowledge about health and disease,” according to the Norwegian Health Research Act. 4 This article does not cover opinions on the legal or ethical issues related to consent as such but rather focuses on the acceptable extent of consent to research use of biological samples. Furthermore, this study investigates preferences for the storage of biological samples, consent collection and timing, and factors motivating or hindering consent.
Material and Methods
Data collection
The study was carried out by Biobank Norway, the Norwegian node of the Biobanking and Biomolecular Resources Research Infrastructure–European Research Infrastructure Consortium (BBMRI-ERIC), a European research infrastructure for biobanks. 12 A working group developed an online questionnaire using Nettskjema, a secure web survey solution developed and hosted by the University of Oslo. 13 The questionnaire was sent out in June 2023 by e-mail to 36 patient advisory councils at Norwegian hospitals, three regional advisory councils, and one council at the Norwegian Cancer Society, comprising a total of 380 patient representatives. Patient representatives were chosen because they are familiar with patients’ perspectives, and we did not have the possibility to contact patients directly. Patient representatives are members of patient advisory councils, who represent all patients and relatives in the health organization’s operations, and may be affiliated to patient organizations. 14 The advisory councils had members from organizations representing a variety of disorders and diseases, including mental health disorders, neurological disorders including Huntington’s disease, Parkinson’s and autism, skin disorders, back problems, diabetes, physiological disabilities, heart failure, and cancer. We contacted the leaders of each council and asked them to distribute the survey to their members. Most representatives have been patients themselves, and they were asked to answer on behalf of patients. A survey link with information was also available on the Biobank Norway website. 15
The questionnaire included 14 questions, of which 5 collected information about the representatives’ age, gender, experience as a patient representative and patient, and whether they had a background as a health care professional (see Supplementary Data S1, which was translated from Norwegian using ChatGPT 16 and revised by a native English speaking author). The first two questions investigated views on the use of collected biological samples and surplus material. Specifically, the first question explored whether patients think additionally collected biological samples should be used for a specific medical research project (reflecting a “specific” consent), or for medical research for multiple projects but limited to certain diseases and diagnoses, such as cancer research (reflecting a “broad” consent as currently used in Norway), or to medical research in general without limitations (reflecting a “general” consent). Question 2 investigated whether surplus biological material from diagnostics should be used for specific, broadly defined, or general medical research as in question one but under the premise that surplus material may be used with a passive consent (defined in our survey as the use of surplus biological material for research without obtaining the patient’s active consent, given that the patient has been informed and had the opportunity to opt out). Question 3 explored whether the use of surplus material for medical research without providing patients with any information would be acceptable. Question 4 focused on the duration of biological material storage, questions 5–7 on the timing and procedures for informed consent, and questions 8 and 9 on motivations and barriers to consenting to research. For each question, relevant information was provided about relevant ethical and legal requirements. All questions in the form were mandatory, and all had comment fields except for the demographic questions. The questionnaire did not allow for the identification of the respondents.
Two patient representatives in our network provided inputs to the questionnaire design and content. The study protocol and questionnaire were assessed by the Data Protection Officer of the Norwegian Institute of Public Health, who provided feedback on the content to ensure anonymity and deemed the study not to require approval by an ethics committee. According to the Norwegian Health Research Act, only medical and health research on human beings, human biological material or personal health data must seek pre-approval from an ethics committee. The representatives consented to the use of their responses for research when starting the survey. Data collection ended in July 2023.
Data analysis
We present the results of the survey using descriptive statistics and simple associations between groups. The text in the comment fields was summarized using a simple text analysis. 17 For each question with a free-text option, we coded the comments under each response into categories. Associations between groups have been analyzed with the Fisher exact test (gender, patient status, health care professional status) or Pearson chi-squared test (age groups) in IBM SPSS statistics v29. Only associations that were statistically significant (p < 0.05) are reported here.
Results
The survey collected 157 responses, which represents a response rate of 41%. See Table 1 for participant characteristics.
Participant Characteristics
Preferences for the use of biological material and health information research
Most representatives (66.2%) supported a general consent to the use of biological material and health data for medical research, without limitation to specific diseases and diagnoses (Q1, Fig. 1). Many (n = 29) mentioned in the comments section that this was important to be able to develop new treatments and make research more efficient. About the same proportion of patient representatives (63.7%) thought that surplus material from diagnostics should be used for medical research in general (Q2, Fig. 1), and it was especially those who had been patients themselves who supported this (66% vs. 44% in the group who had not been patients, p = 0.005, Fig. 2). Similar to the comments to question one, the majority who commented on question two said they supported the use of surplus material for medical research in general in order to advance medical research and not waste valuable material (n = 9). However, some (n = 5) also specified that it is acceptable provided the patient has been properly informed and has had the right to withdraw. A few (n = 3) also said they would prefer to have informed consent for the use of surplus material rather than a passive consent based on notification with the possibility to opt out.
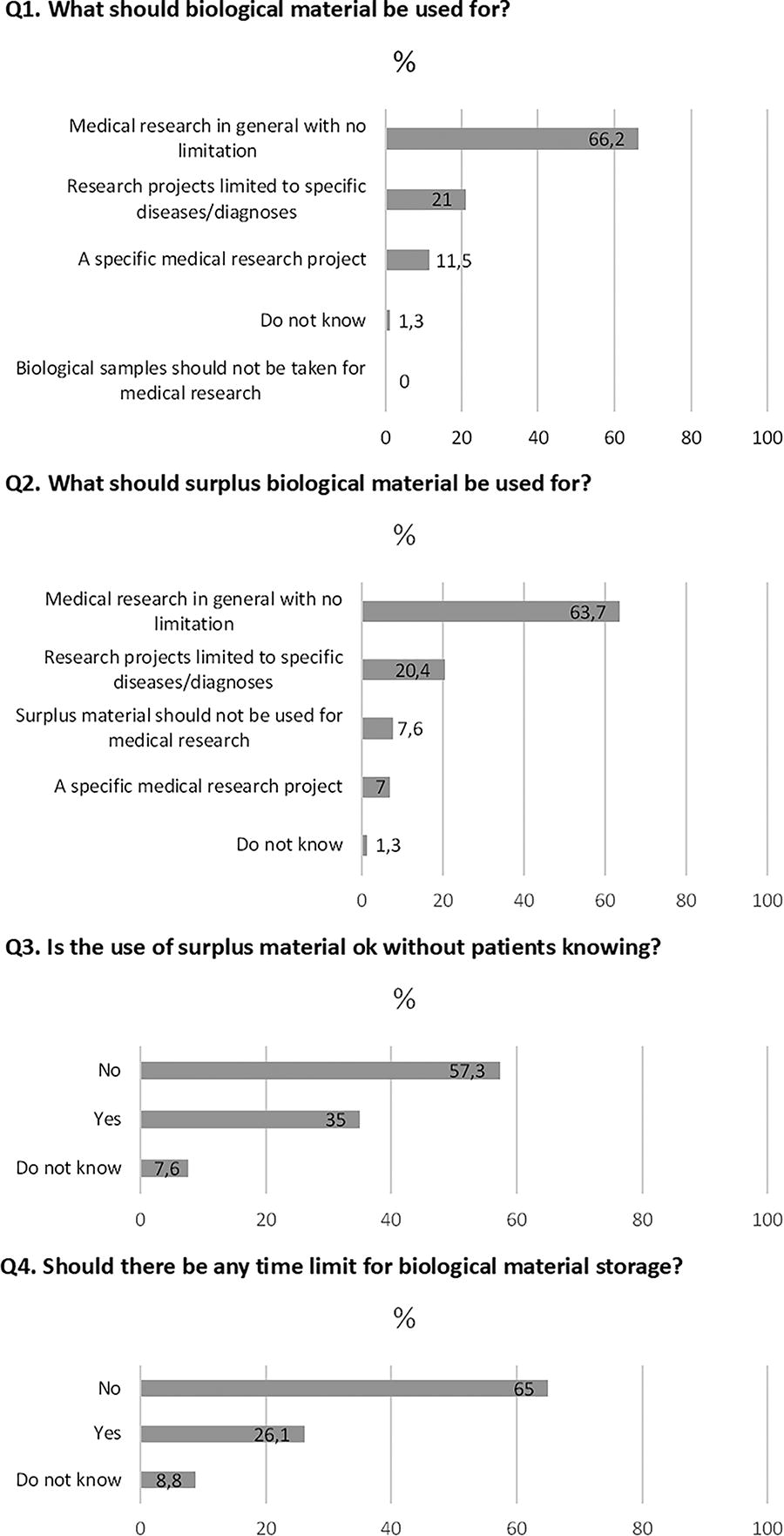
Preferences for the use of biological material and health information for medical research (Q1–Q4).
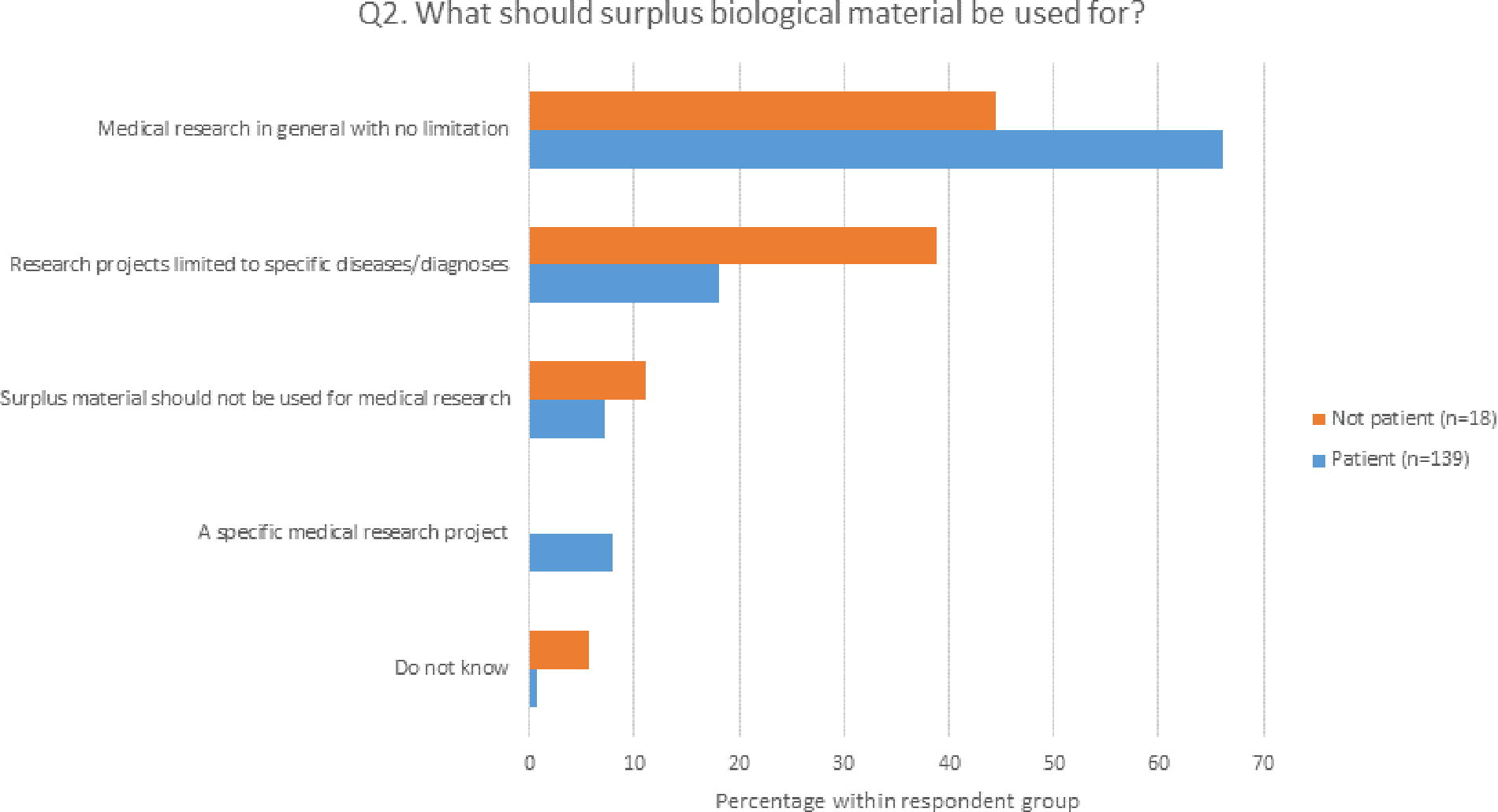
Differences in preferences between patients and non-patients for the use of surplus biological material (Q2). Patients from the past 5 years and previously were grouped together. Please note that the number of respondents in the non-patient group was much lower. Those who had previously been a patient were more supportive of the use of surplus material for medical research in general than those who had not been patients (p = 0.005).
A majority (57%) did not support the use of surplus material if the patient had not been informed about it (Q3, Fig. 1), and women were more against this than men (62% among the women and 50% among the men, p = 0.02, Fig. 3). In the comments section for question 3, the majority (n = 8) of those against stated that it is ethically correct to inform the patient that his or her surplus biological material could be used in future research, and several (n = 6) also said they would prefer informed consent for the use of surplus material rather than a passive consent based on notification with the possibility to opt out. Overall, most representatives (65%) supported the storage of biological material for research purposes without a time limit (Q4, Fig. 1), and several (n = 12) commented that this was important to ensure the optimal utilization of the material for future research. This opinion was, however, strongly correlated with age: 71% in the group 18–29 years wanted a time limit, while in the age groups over 30 years, the majority wanted no limit (p = 0.006, for the ranked age group trend, Fig. 4).
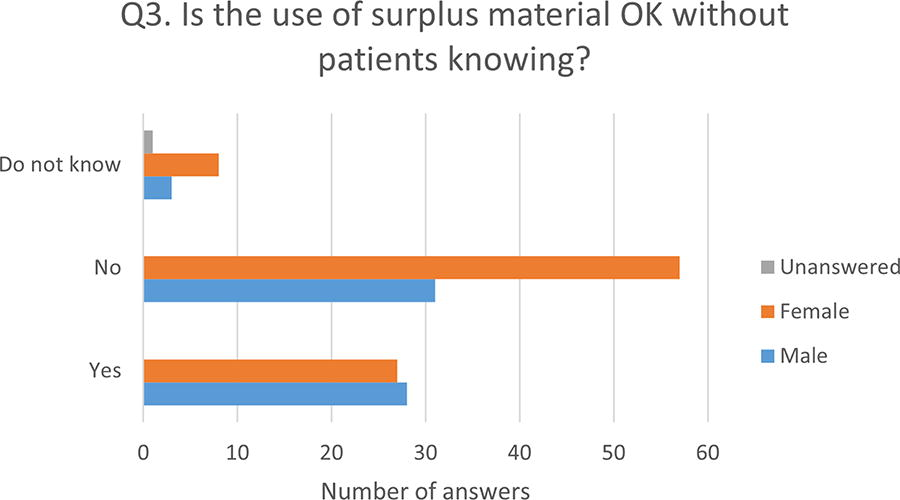
Differences in preferences between men and women for the use of surplus material without knowing (Q3). Women are more skeptical of the use of surplus material without being informed than men (p = 0.02).
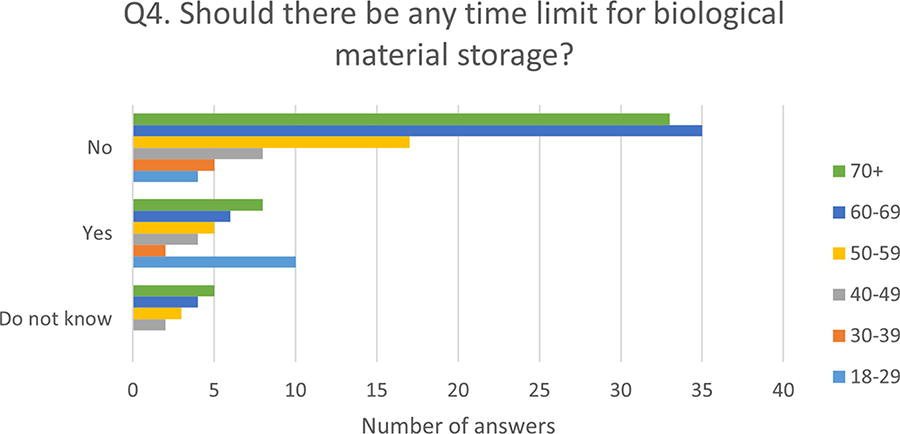
Different preferences among age groups for whether there should be a time limit for the storage of biological material (Q4). The youngest respondents are more skeptical to long-term storage (p = 0.006).
Patient preferences for consent processes
On the timing of consent (question 5), over half of the patient representatives favored being asked to consent in the invitation letter for a hospital visit (56.1%) (Fig. 5). However, many (n = 22) specified in the comments section that, ideally, they would like to receive information in advance of a hospital visit and then give consent at the time of attendance, which would give them a period to reflect, and allow them to ask questions in person at the hospital visit. For question 6, most representatives (70.7%) thought that patients should have the choice between paper-based or digital consent (Fig. 5). In the comments section for question 6, several respondents (n = 16) highlighted the importance of considering the needs of patients without digital skills. There was no significant difference in the response rates between the different sociodemographic categories for questions 5 and 6. For question 7, most respondents supported providing patients with information about the use of biological material for research on the digital platform Helsenorge.no (45.2%) followed by newsletters to patients (30.6%) and general information on the hospital website (9.6%), and this was significantly associated with age: those under 60 preferred the digital platform Helsenorge.no, while those over 60 preferred the newsletters and hospital website (p < 0.001) (Fig. 6). The preference for newsletters increased with age (p = 0.001). For those favoring the digital platform, several (n = 8) mentioned in the comments that they would also need information about how to use it, and be informed each time there is new information relevant for them in the portal.
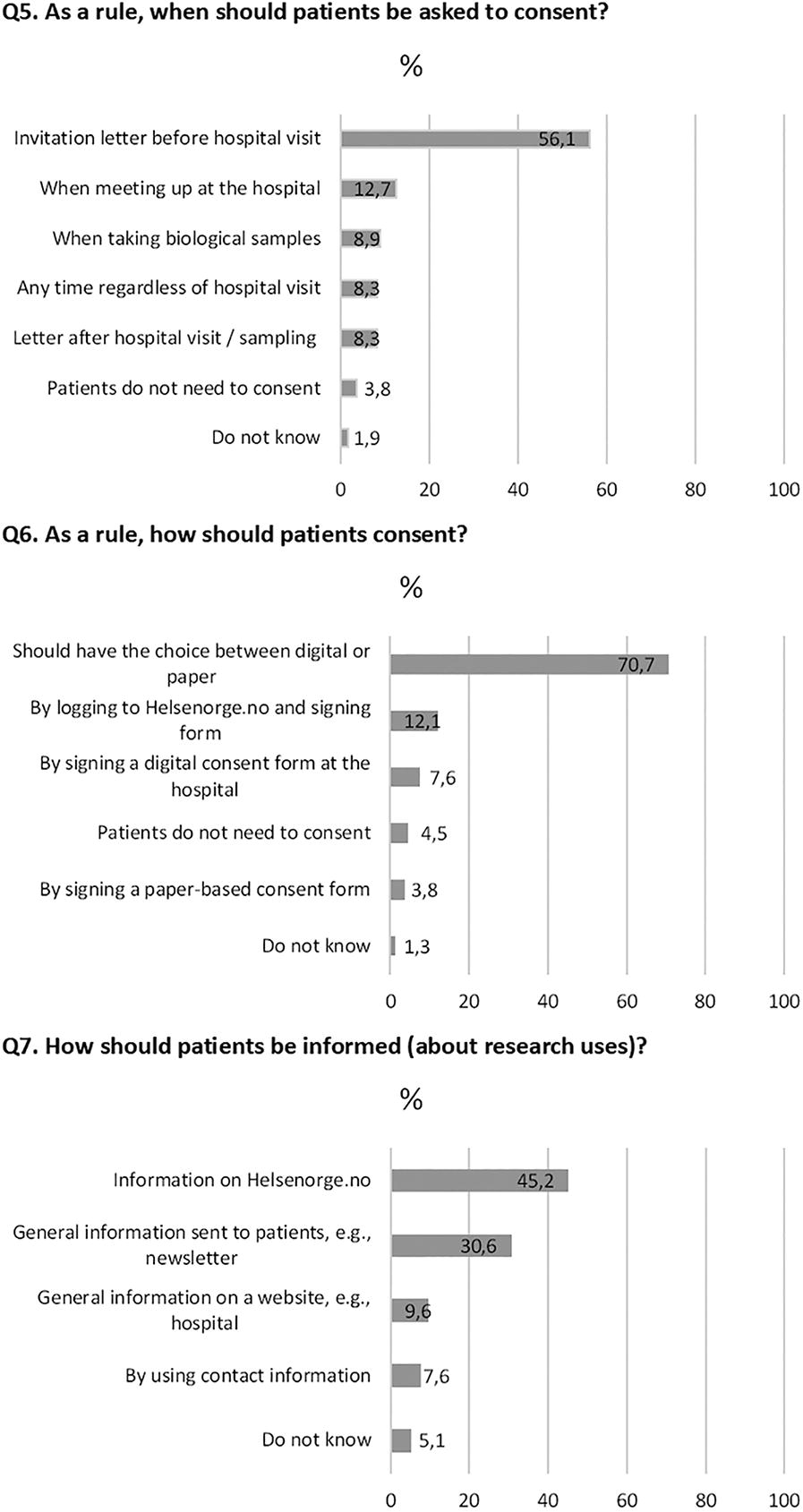
Patient preferences for consent processes (Q5–Q7).
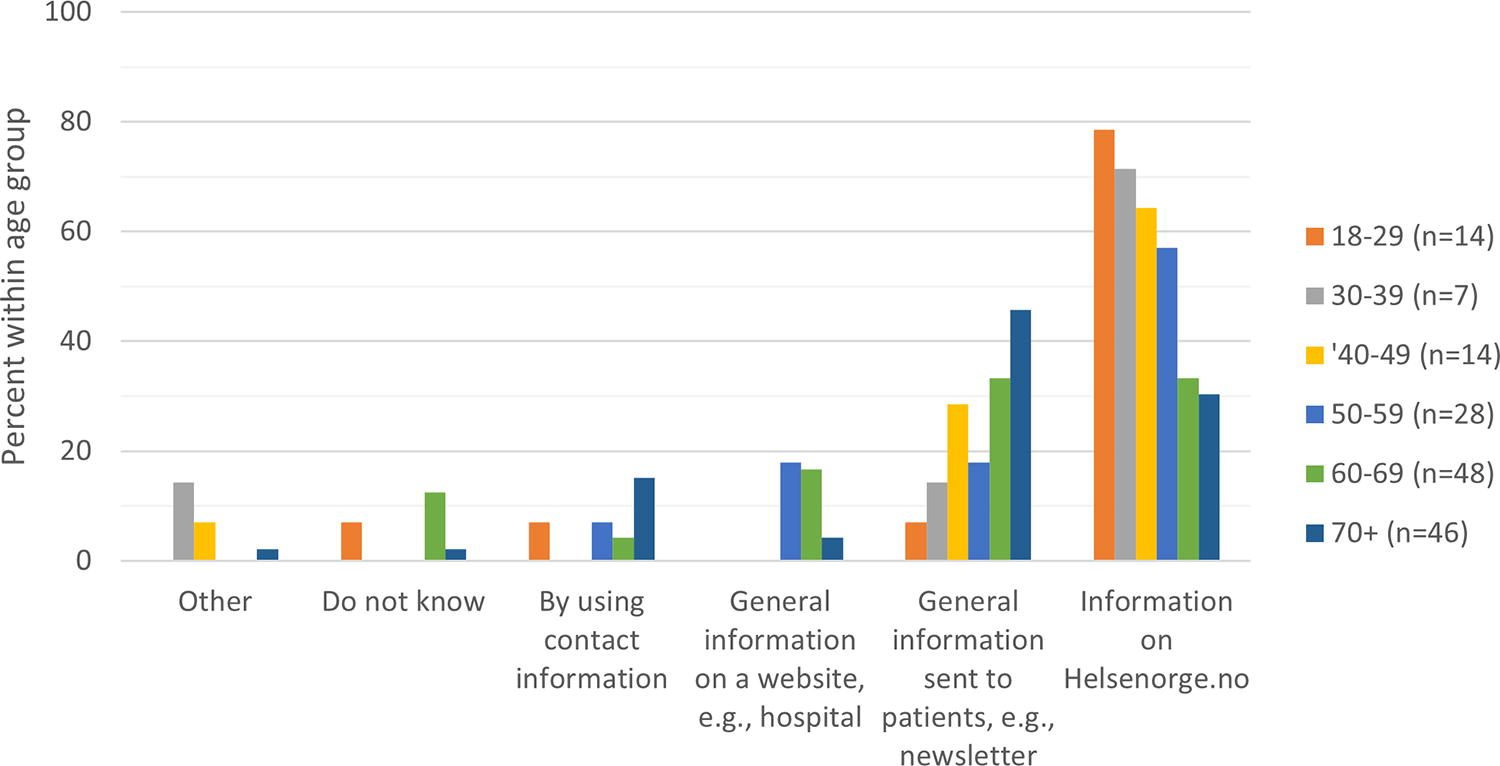
Different preferences among age groups for how patients should be informed about research uses. Those under 60 preferred the digital platform Helsenorge.no, while those over 60 preferred the newsletters and hospital website (p < 0.001).
Factors motivating and hindering patients to consent to medical research
For questions 8 and 9 regarding factors motivating or hindering consent, respondents could select up to two answers. Figure 7 shows that the desire to contribute to medical research and help future patients was considered the main motivating factor for consenting to research (89.8%), followed by optimism and faith in scientific progress (24.2%) and understanding that valuable biological material should not be wasted (16.6%). A total of 9.6% of representatives believed that the hope for personal benefit could motivate participation in research. Main factors believed to hinder consenting to medical research included the fear that health information may be used for something other than research (49%), uncertainty about the type of research to be conducted (43.9%), and poor communication (31.8%) (Fig. 7). There were very few comments to questions 8 and 9, but one respondent commented on question 8 that “the desire to contribute to medical research” and “belief in scientific progress” in the answer options were closely related.
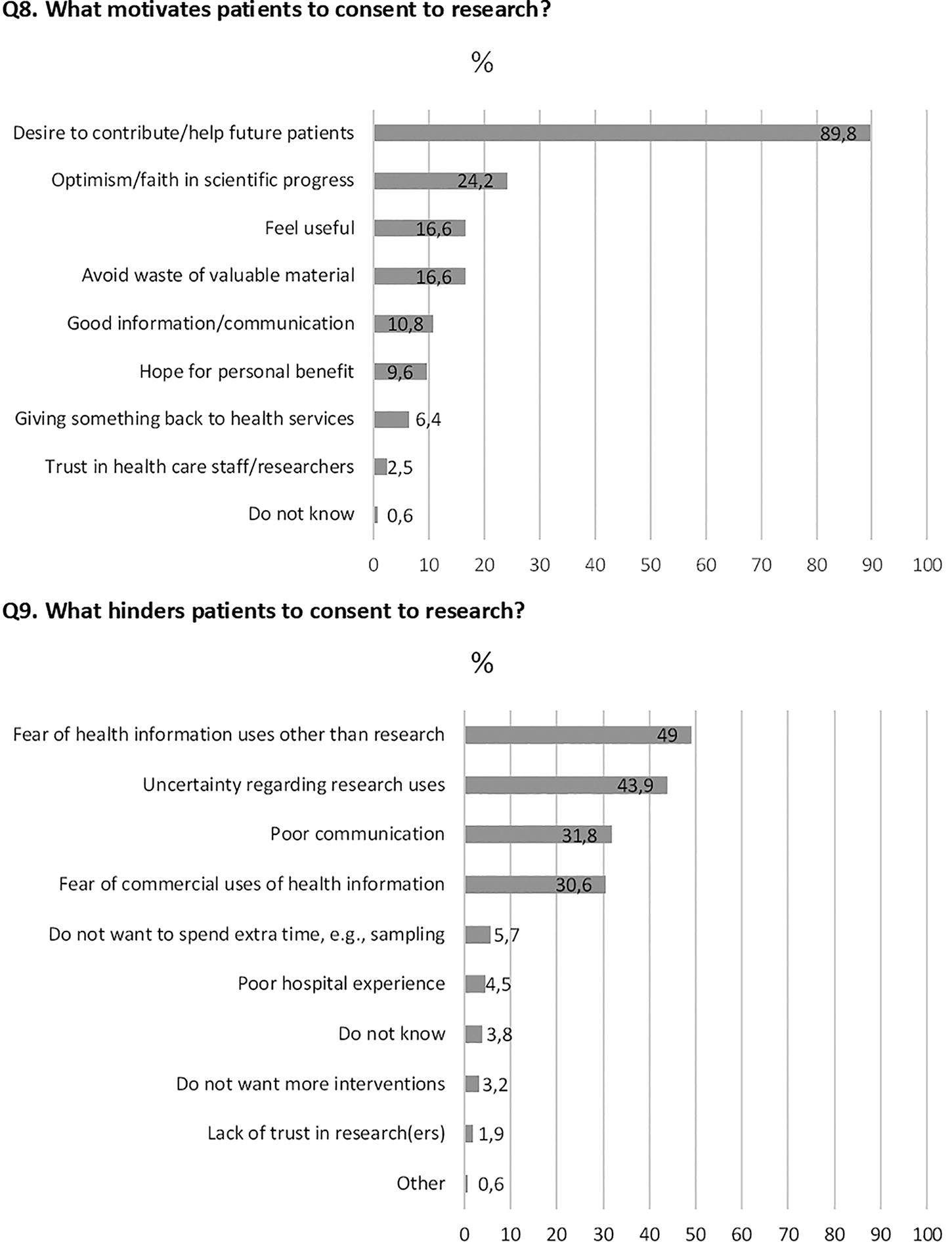
Factors motivating and hindering patients to consent to medical research (Q8 and 9).
Discussion
According to the patient representatives in our sample, Norwegian patients would likely support a broader use of their biological material and health information for medical research than currently practiced and possible. This corroborates results from a Swiss experimental study, which showed that the scope of consent (general vs cancer-oriented) may not play a vital role for patients and that other factors are more important, such as how the biological material is collected and how often data are needed. 18 Similar to other studies, 5 Norwegian patients are also likely to find the long-term storage of biological material for research acceptable, although the youngest patient representatives were less prone to think so. Another study identified similar concerns among young adults regarding the location and time length of the storage of their biological material. 19 It is, however, unclear whether this is because young people are more wary of privacy issues than older generations due to their exposure to digital technologies via, for instance, social media, or whether the older generations are less concerned than the younger ones about the uses of their data for health research. 20
Overall, our results corroborate findings from other studies showing a great desire among patients to consent to using their biological material and health data for medical research, to contribute to medical research and help future patients.5,8,21,22 However, fewer of our respondents were motivated by faith in scientific progress, despite the similarity between scientific progress and medical research. This may have been because the respondents were able to choose up to two categories for this option, but most of them chose only one. They may have favored the first category “contribute to medical research/help future patients” because it covered aspects of both medical (hence scientific) progress and altruism. Previous studies have shown altruism to be the main motivating factor for patients’ willingness to consent to biobanking research,5,8 and this may explain why most of our respondents favored this option.
Different informed consent models have been implemented in biobanking throughout the years, from specific to broad, and the models have varying abilities to inform participants about the risks and benefits of the research and to enable them to assess how the research aligns with their personal values. 1 The utilization of biological material for research is not fully optimized in Norway. Opening for a general consent for the future use of biological material—not limited to broadly defined research uses—would provide more flexibility to researchers who often are not able to foresee all potential future research needs at the time of biological material collection. 23
Concrete examples already exist of biobanks and research projects using a general consent. In Germany, procedures have been implemented to support hospital-based biobanking, to which patients can provide informed consent to future, undefined, research uses. 24 Any access by researchers to the stored biological material requires approval from an independent ethics committee and the biobank’s access committee. 24 The country has also recently adopted a nation-wide broad consent template document for the use of patient pseudonymized data for research. 3 In Switzerland, the Lausanne Institutional Biobank recently established general consent. Patients, who are usually approached by a team of recruiters, can consent to the open-ended use of their biological samples and health data, including genome analyses. 25 To fully maximize the use of biological resources for research, broad consent models ideally should be developed not only nationally but also at the international level to support cross-border exchange. In North America, the Global Alliance for Genomics and Health has developed model consent clauses that can be used by researchers conducting genomics research to facilitate international sample and data sharing, 26 and launching similar initiatives in Europe should be feasible. Broad consent is already widely practiced in European biobanking and is supported by the General Data Protection Regulation.27,28
For a general consent model to be ethically acceptable, several considerations need to be taken into account. Finding the right timing and setting to collect patient consent may be important. In a recent Swiss study, the researchers raised concerns that collecting consent in a hospital setting by direct contact with patients at a time when they may be influenceable and vulnerable could negatively influence patient autonomy. 21 Similarly, a study at the Lausanne Institutional Biobank found that patient willingness to consent depended on the relationship patients and recruiters establish during their meetings in the hospital setting. 22 In contrast, a German study showed that consent collection directly after patient admission was found acceptable by patients and seen as time efficient and practical. 29 The patient representatives in our study preferred that patients be given the possibility to consent to research before they attend the hospital. Research should be conducted to investigate which preferences Norwegian patients have regarding the timing and setting of consent collection.
Our results also show that patient willingness to support research does not rule out the desire to receive information that their surplus biological material may be used for research. This contrasts somewhat with results from another Norwegian study, which found that just over half (57%) of the patients did not think it necessary to consent or receive any information about their biospecimens being used for future research, while a third (35%) wanted passive consent. 11 However, the question used in their study was slightly different as it focused on the positive aspects of using surplus material and on what sort of consent patients would prefer. In contrast, our question was directly about whether they think it is acceptable to use surplus material without informing the patient about it, which may have elicited more skepticism on behalf of our respondents. Our general impression from the literature, however, is that most patients would like to be informed of the secondary use of biological material collected during hospital visits, and many support passive consent provided they have the possibility of opting out.8,11 This was also mentioned by the respondents in the free-text comments in our study. However, passive consent assumes that patients have received relevant information, and although some information about research uses may be provided on hospital websites or via posters in hospital corridors, the current practice in Norway is that patients must actively seek information themselves, so there is a risk that a considerable number of people are not well informed about research uses. Future studies may therefore investigate how well-informed patients are about the potential use of their surplus biomaterials, and how to communicate this information most appropriately.
Broadening consent to a general consent to medical research may mean in practice that the consent is collected only once. This could also lead patients to feel they have no say regarding how their samples are being used, such as for commercial uses, to which our results and other studies show some skepticism.30,31 However, a one-time consent to research does not mean giving up the right to be informed about how biological material and health data are being used for research. One way to address these concerns may be to raise awareness of the terms of the broad consent by conducting educational campaigns toward patients. For instance, a university hospital in Switzerland has developed brochures, videos, and a website to inform patients, in lay terms, about how consent to research is practiced and what it means to patients. 32 Another approach may be to use flexible consent and digital information platforms 33 to regularly provide patients with adequate information about which types of research their biological data and health information is being used, and to accommodate for patients’ changing preferences over time. Our representatives suggested using the Norwegian national online health services platform Helsenorge.no 34 for such information. The use of digital platforms for consent collection and interaction between researchers and patients is becoming more widespread and has shown promising results. 35 However, the use of such platforms should be combined with more traditional methods such as paper-based consent and newsletters, to take into consideration the needs of those with limited digital skills or access. Previous research has also found that a considerable proportion of patients (42%) still prefer to give consent on paper. 11 Although Norway is a relatively well-digitalized country, several groups of the population are still strongly dependent on paper-based solutions. 36
Providing sufficient and timely information to patients will require developing a national infrastructure that can keep track of how people’s health data and samples are being used, while simultaneously ensuring anonymity and equitable access to information tailored to each person’s capabilities and situation. Efforts are underway by the health authorities in Norway to make this possible. However, the development of such an infrastructure on a national scale is expected to be onerous. It will therefore be critical to identify the most pressing information needs of Norwegian patients regarding research uses of their biological information and health data to guide the cost-effective choice of consent management and information channels, including the use of digital platforms.
Limitations
This study has important limitations. We did not survey patients but patient representatives, who responded to our questions on behalf of patients. Although we believe that patient representatives have the necessary experience and knowledge to answer the survey, they are not representative of the average patient. This is because they likely have more knowledge about research, and possibly are more positive to research than the average patient due to their role in patient advisory councils. Furthermore, it is also possible that representatives from patient groups with cancer or heritable diseases that lack efficient treatments may be more motivated to provide wider access for research purposes than those representing patients with chronic and treatable diseases. We did not, however, ask the respondents to provide information in the questionnaire about which type of disease they represent, and we therefore do not know the proportion of respondents representing the different types of diseases. Future studies may consider whether differences in opinions and preferences exist between patient representatives working with different types of diseases.
Another limitation of our study is that most patient representatives were above 60 years of age, and we lack sufficient data among younger patient representatives. The respondents were also predominantly female, which may reflect a high interest in health issues among females. Finally, a possible limitation is that some of the questions may have assumed specific background knowledge. We aimed to provide enough necessary background information on the topic of consent and the Norwegian legislation in the survey but had to compromise somewhat on the amount of information to keep it simple and maintain survey readability.
Conclusions
Our results suggest that the time may be ripe to consider broadening the “broad consent” currently practiced in Norway and adopt the principle of general consent for the use of biological samples and health data in medical research. Future studies should investigate how a new consent model can be complemented with reliable and efficient mechanisms to secure patient rights and equitable access to information. A change in the Norwegian law that allows patients to give a general consent to the use of both collected biological material and surplus material for medical research, could support better utilization of biological material collected in the health service, and may also correspond better to the patients’ desire to contribute to medical research.
Footnotes
Acknowledgments
The authors would like to thank the patient representatives for their valuable time and input in answering the questionnaire. Special thanks go to legal counsel Birgitte Wirum Sand for providing insight into the legal aspects of Norwegian consent procedures and for providing valuable input in the development of the questionnaire. The authors also thank the two patient representatives who helped give feedback on the understandability of the questionnaire. The authors are also grateful to the Biobank Norway WP3 ELSI group for comments on the results and article draft.
Authors’ Contributions
R.B.C.: Conceptualization, data curation, formal analysis, investigation, methodology, project administration, visualization, writing—original draft, writing—review and editing. M.K.: Conceptualization, data curation, formal analysis, methodology, visualization, writing—review and editing. W.R.: Conceptualization, project administration, methodology, writing—review and editing. Ø.L.M.: Conceptualization, funding acquisition, project administration, visualization, writing—review and editing. S.K.: Visualization, writing—review and editing. J.H.: Visualization, writing—review and editing. I.B.-L.: Conceptualization, formal analysis, funding acquisition, investigation, methodology, project administration, resources, supervision, visualization, writing—original draft, writing—review and editing.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
