Abstract
Objective:
To summarize the evidence and identify gaps in the use of point-of-care (POC) techniques that hold potential to mitigate early, unplanned lactation cessation in term mother/infant dyads.
Methods:
An integrative review was conducted using the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. The search was conducted in the following electronic databases: MEDLINE (through PubMed), CINAHL (Cumulative Index to Nursing and Allied Health Literature), and Web of Science.
Results:
Of the 2,888 articles retrieved, 19 met the eligibility criteria and were included in this review. POC techniques included infant scales and a bedside ketone meter to measure infant weight change (n = 4); infant scales to measure mothers’ own milk (MOM) transfer (test-weight) (n = 15); and ion-selective electrode probes and somatic cell counter to measure mammary gland function and health (n = 2). MOM transfer, as measured with infant scales, was the most frequent POC technique and was used in four primary ways: outcome variable, accurate standard to test other measures of MOM transfer, personalize intake of MOM components, and predict later lactation outcomes. Commercially available ion-selective electrode probes provide accurate and objective measures of paracellular pathway patency in the mammary gland, and have relevance for measurement of secretory activation achievement.
Conclusion:
Infant scales to measure weight change and MOM transfer and ion-selective electrodes are ideal POC measures to identify and mitigate lactation risk in the early postpartum period for term dyads. Translational studies that inform risk detection algorithms and novel models of lactation care that include these POC techniques are needed.
Introduction
Early, unplanned lactation cessation affects over half of lactating mothers, 1 and is often related to perceived or real insufficient mothers’ own milk (MOM) volume with origins in the first 14 days postpartum.2–4 This early postpartum period represents a critical window for mammary gland programming and includes complex biological processes that must be achieved to optimize long-term exclusive breastfeeding.4–6 Secretory activation (SA), triggered by delivery of the placenta, involves the upregulation of four transcellular pathways and the closure of one paracellular pathway. 6 Once achieved, paracellular pathway closure must be maintained while autocrine/paracrine mechanisms assume control of MOM synthesis and secretion.7,8 By 4–7 days postpartum, healthy breastfeeding mothers of term infants achieve a threshold MOM volume of approximately 500–600 mL/day, considered adequate for infant growth. 9
Concerns about insufficient MOM volume are often perceived rather than real and are commonly related to normal infant behaviors such as fussiness and frequent feeds, as well as normally small colostrum volumes.3,4,10,11 Especially prominent in marginalized populations with lack of generational breastfeeding experience and support, these perceptions often lead to formula supplementation with resultant downregulation of lactation processes and real insufficient MOM volume.12,13 Infant feeding behaviors, such as excessive sleepiness or failure to maintain an effective latch, can also downregulate MOM synthesis and secretion due to ineffective and/or inefficient mammary gland stimulation and MOM removal. 14 Ineffective and inefficient feeding behaviors are common among late preterm and early term infants cared for in the maternity setting as well as in infants with hypotonia and orofacial problems.14,15 These perceptions of insufficient MOM volume and ineffective/inefficient infant breastfeeding behaviors are often modifiable but can lead to real insufficient MOM volume if not identified and managed with targeted lactation care. 16 However, there is an increasing number of mothers of otherwise healthy infants who experience real insufficient MOM volume in the early postpartum days, including those with metabolic, inflammatory, and genetic/epigenetic health problems.17–19 This subgroup is at greater risk for impaired secretory differentiation, delayed or impaired SA, lack of maintenance of paracellular pathway closure after SA achievement, and lack of achieving an adequate threshold MOM volume.17–19 Although these mothers need highly specific care to preserve lactation potential, they are easily overlooked in the maternity setting because the infant is not preterm or ill.17–19
Addressing early, unplanned lactation cessation among term breastfeeding dyads would optimize lactation outcomes, improve health for both mothers and infants, and reduce costs associated with suboptimal lactation.20,21 A promising first step is leveraging objective, accurate point-of-care (POC) technologies that can be integrated into guidelines and algorithms to identify and manage early lactation risk, regardless of whether insufficient MOM volume is perceived or real. To this end, we undertook an integrative review to summarize the current evidence and identify research gaps in the use of POC technologies for addressing early, unplanned lactation cessation among term mother/infant dyads.
Methods
Using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, 22 we comprehensively searched the literature for studies involving POC techniques used to evaluate lactation outcomes within the first 3 months postpartum among mothers of term infants. POC testing was defined as clinical laboratory testing conducted close to the site of patient care and providing rapid turnaround of test results. 23
Eligibility criteria
Eligible studies used a quantitative design, were available in full text, included human participants, reported on a POC technique that measured a lactation outcome, included only term mother/infant dyads (defined as ≥37 weeks gestational age, including early term infants) or reported data separately for term versus preterm dyads, included only dyads who were ≤3 months of age or reported the data separately for dyads ≤3 months, were published between January 2013 and May 2024, were peer reviewed, and reported in English. Excluded studies included data from human milk banks that did not report donor characteristics (gestational age at birth and infant age at the time of donation). Also excluded were commentaries, editorials, and dissertations.
Search strategy
A search was conducted in October 2024 of the electronic databases MEDLINE (through PubMed), CINAHL (Cumulative Index to Nursing and Allied Health Literature), and Web of Science. The search terms used were Medical Subject Headings (MeSH) headings, text words, and word variations related to the key concepts of POC testing (“point-of-care testing,” “portable electrode,” “test weigh,” etc.) (Supplementary Table S1) and lactation outcomes (“lactation,” breastfeeding,” “pumped breastmilk,” etc.) (Supplementary Table S2). The search strings utilized for each electronic database are reported in Supplementary Table S3.
Using Covidence, a systematic review management software (Melbourne, Australia), 24 duplicates were removed. Titles, abstracts, and full text articles were independently screened by five authors (L.H.B., S.D.J., K.T.K., C.M.P., and A.E.Z.). After full text screening, data on the following fields were extracted by the same five authors for articles that met eligibility criteria: first author, year, and country; study design and purpose; infant gestational and chronological age; POC technique; timing of the POC technique; and key findings of the study.
Data analysis
The authors synthesized data from each type of POC technique and reported on the following: the POC technique device, the number of measures taken, who performed the collection of data and/or interpreted findings, and the timing of the measures. The articles clustered into three categories based on POC measurement of specific lactation outcomes: infant weight change, infant MOM transfer, and mammary gland function and health. All discrepancies between reviewers regarding article screening, data extraction, synthesis, and interpretation were discussed until resolved.
Results
A total of 2,888 articles was retrieved from the electronic databases (Fig. 1). After removing 513 duplicates, 2,375 titles and abstracts were screened, of which 2,260 did not meet eligibility criteria, and 115 full text articles were screened. Of the 115 full text articles reviewed, 75 were excluded, leaving 40 articles meeting eligibility criteria. Of these 40 articles, 21 studies using infrared analysis of macronutrient content were excluded from the analysis reported here because they focused on measuring MOM composition rather than addressing early, unplanned lactation cessation. Thus, 19 articles remained and were grouped by specific POC techniques and the lactation outcome they sought to measure. POC techniques included infant scales and a bedside ketone meter to measure infant weight change; infant scales to measure MOM transfer during breastfeeding (test-weights); ion-selective electrodes and somatic cell counter to measure mammary gland function and health (Table 1). Of the 19 articles, 2 used infant scales for both infant weight change and MOM transfer so are reported in both categories.25,26 These 19 studies are summarized in Tables 2, 3, and 4.
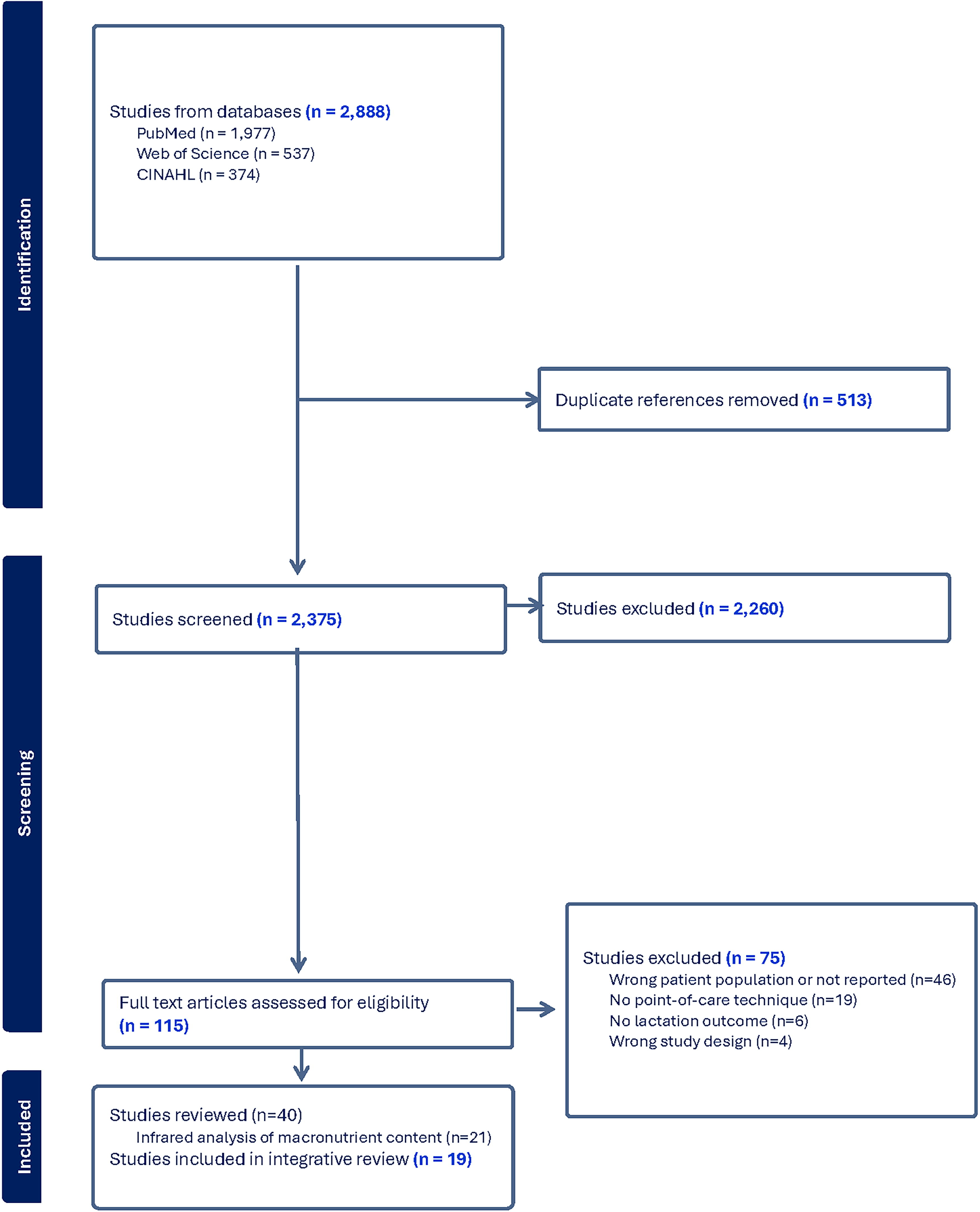
PRISMA flow diagram for an integrative review of point-of-care measures to identify risk for early, unplanned cessation of lactation.
Type and Frequency of Articles Reporting Point-of-Care Techniques
MOM, mothers’ own milk.
Lactation Outcome: Infant Weight Change
g, gram; GA, gestational age; MOM, mother’s own milk; NR, not reported; PNA, perinatal age; R, reported; r, correlation coefficient; SD, standard deviation.
Lactation Outcome: MOM Transfer
EDA, epidural anesthesia; g, gram; GA, gestational age; GDM, gestational diabetes mellitus; LATCH, latch, audible swallowing, type of nipple, comfort, and hold; LCFA, long-chain fatty acid; MCFA, mid-chain fatty acid; mL, milliliters; MLMS, moderate/normal mother’s own milk output; MOM, mother’s own milk; MPR, mother’s own milk production rate; NICU, neonatal intensive care unit; NR, not reported; PNA, perinatal age; R, reported; SD, standard deviation; SLMS, severely low mother’s own milk output.
Lactation Outcome: Mammary Gland Function and Health
CI, confidence interval; GA, gestational age; ICP-OES, inductively coupled plasma optical emission spectrometry; ISE, ion-selective electrode; K, potassium; mM, millimole; MOM: mother’s own milk; Na, sodium; OR, odds ratio; PNA, perinatal age; POC, point-of-care.
Infant weight change
Four articles described two POC techniques used to evaluate infant weight change during the first 14 days postpartum (Table 2).25–28 DiTomasso et al. and Roznowski et al. incorporated both research site and in-home measurement of infant weight change, and Carrega et al. used in-home weights only.25–27 Although DiTomasso’s primary objective was to examine infant weight gain and loss in the first 14 days postpartum in exclusively breastfed term dyads, Roznowski et al. and Carrega et al. used infant weight change as a secondary measure in studies for which MOM transfer was the primary outcome of interest.25–27 The second POC technique used infant weight obtained with a digital scale to serve as the accurate standard to test a novel device to predict excessive infant weight loss during the early postpartum period. 28
DiTomasso et al. sought to establish a relationship between early postpartum infant weight loss and exclusive breastfeeding at 14 days postpartum. 27 Using a prospective observational cohort design, DiTomasso divided 151 healthy, term infants into 2 groups based on postbirth weight loss of ≤7% versus >7%. 27 Infant weight was measured daily during the maternity hospitalization by nurses and postdischarge by mothers. 27 Eight mothers (5%) did not complete the entire 14-day weight measurement protocol. 27 Investigators acknowledged potential error in weighing procedures both by nurses and mothers. 27
Futatani et al. used the infant scale as the accurate infant weight change standard to examine the diagnostic accuracy of the bedside ketone meter (Precision Xceed, Abbott, Tokyo, Japan), a novel POC technique for predicting excessive weight loss and/or hypernatremic dehydration in term breastfeeding infants. 28 The bedside ketone meter measures serum beta-hydroxybutyrate, which is elevated when nutrient intake is inadequate. 28 Futatani et al. enrolled 585 exclusively breastfed term infants who were weighed daily on the same electronic scale within the same 2-hour interval during the maternity hospitalization. 28 Immediately following infant weight measurement, serum beta-hydroxybutyrate was sampled via heel stick and measured POC using the bedside ketone meter. 28 POC serum ketone values were positively correlated with infant weight loss percentage and were highly predictive of excess weight loss, hypernatremia, and acid-base imbalance among full-term, exclusively breastfed infants. 28 Although investigators conclude that the bedside ketone meter could serve as a useful clinical POC device to detect markers of inadequate MOM transfer in the early postpartum period, no data were provided to inform feasibility or cost. 28 Thus, it is unclear whether the bedside ketone meter is superior to infant scale measures of weight change and/or real measurement of MOM transfer, both of which are likely less costly, more widely available, and do not involve infant blood sampling. 28
Roznowski et al. and Carrega et al. reported intermittent and/or short-term weight change measures,25,26 but neither article included infant weight change as a primary study objective. Roznowski et al. used a combination of in-home and research site infant weight measurements primarily to establish a baseline weight gain trajectory prior to and during a 2-day pumping intervention aimed at increasing the MOM production rate. 25 Weight change became relevant upon finding inadequate baseline infant weight gain in 25% of the enrolled infants, despite mothers’ reports of exclusive breastfeeding. 25 Also unexpected, the rate of infant weight gain decreased during the 48-hour protocol, halting that study intervention. 25 Carrega et al. sought to address MOM transfer as a function of maternal sleep characteristics but also used the scale to measure infant weight change at 2 weeks postpartum. 26 This article did not explain why the weight was performed or how it related to primary study objectives. 26
MOM transfer
Fifteen studies measured MOM transfer using test-weights,10,18,25,26,29–39 a valid and reliable technique in which infants are weighed (grams) on an electronic scale before and after a breastfeeding session, under exactly the same conditions (e.g., clothing, blankets, pacifier), with 1 gram of post-feed weight gain = 1 mL of MOM intake.42,43 Eight studies used test-weights to measure MOM transfer as an outcome variable in observational and randomized research.18,26,30,31,33,35,37,39 Three studies used test-weights as the accurate standard to which alternative measures of MOM transfer or related measures (e.g., hourly MOM production) were compared or validated.10,25,29 Two studies used test-weights to personalize the absolute amount of MOM components consumed by the infant on the basis of ingested MOM volume,32,34 and two studies used test-weights to describe and/or predict MOM transfer trajectories (Table 3).36,38
Test-weighing procedures varied considerably across studies with respect to postpartum timing and total duration of measurements, where and by whom measurements were performed, subject compliance with procedures, and investigator descriptions of rigor with respect to user instructions and in evaluating acquired test-weight data.10,18,25,26,29–39 Single test-weights were performed in two studies either to evaluate pre-post differences in MOM transfer, 33 following an intervention, or to validate an alternative measure of MOM transfer. 29 Those who reported average daily MOM transfer included protocols ranging from 3 test-weights in 1 day (to estimate 24-hour intake) to 96 consecutive hours.10,18,25,26,31,32,34–39 Of the 15 studies, 11 included all or some test-weights performed in the home by mothers,10,18,25,26,31,32,34,36–39 with the remaining 4 studies performing test-weights in-hospital or at a research site.29,30,33,35 Subject compliance with completing a study’s specified test-weighing protocol was reported by all 11 authors in studies where test-weights were performed at home by mothers, and ranged from 62% to 100%.10,18,25,26,31,32,34,36–39
Test-weights to measure MOM transfer as an outcome variable in observational and randomized studies
Eight studies, ranging from randomized interventions to observational and proof of concept approaches, used test-weights as an outcome variable.18,26,30,31,33,35,37,39 These studies sought to establish associations or causality across a variety of independent and/or predictor variables, including galactagogues,31,35 common maternal intrapartum and postpartum health concerns (maternal sleep quality, epidural use, maternal metabolic risk),18,26,37,39and infant conditions (hyperbilirubinemia, tongue and lip-tie repair).30,33 Studies enrolled dyads as early as 3 days postpartum and as late as 3 months postpartum, and the test-weighing duration ranged from a single measure of MOM transfer pre- and postintervention to the in-home use of test-weights for 24–48 consecutive hours.18,26,30,31,33,35,37,39 Three studies used MOM transfer measures somewhat differently than others.26,33,35 Carrega et al. estimated 24-hour MOM transfer by using only 3 consecutive test-weights on a single day, and multiplying the mean intake by the number of daily breastfeeds. 26 Gharhari et al. performed a single test-weight pre- and postprocedures for tongue-tie and lip-tie release to compare the efficiency (mLs/min) of MOM transfer. 33 Paritakul et al. used test-weights to measure MOM transfer at 3 days postpartum and to estimate MOM production at 7 days postpartum using a breast pumping protocol. 35
Although most investigators included varying amounts of information about maternal instruction and monitoring of the appropriate use of scales for test-weighing, only Nommsen-Rivers et al. detailed the research rigor to assure that in-home measures were accurate and reliable. 18 Specifically, this included critical examination of collected data to address biological plausibility of intake (e.g., >200 mL or <2 g per breast), identifying measures with odd versus even numbers of transferred MOM volume (scale weighed only in even numbers), and other indices of error. 18
Test-weights as the accurate standard to test other estimates of MOM transfer
Three studies used test-weights as the accurate standard to test the concurrent or predictive accuracy of an alternative measure of MOM transfer, including the LATCH score, 29 hourly MOM production, 25 and maternal perceptions of insufficient MOM. 10 A common premise of these studies was that although test-weighing was an accurate and reliable measure of MOM transfer, simpler POC alternatives that were also less burdensome and more widely available merited consideration.10,25,29
In a blinded, observational study, Altunaş et al. performed test-weights and LATCH scores for a single breastfeeding session in 66 hospitalized infants, 33 (50%) of whom were term (≥37 weeks), and included in this review. 29 Investigators also calculated expected intake, defined as the three-hourly minimum volume the infant should consume, based upon weight and postnatal age, and compared expected intake to both MOM transfer by test-weights and LATCH scores. 29 A trend among higher latch scores, increasing MOM transfer and a greater proportion of expected MOM intake was reported. 29 Although investigators acknowledged marked variability in individual differences between LATCH scores and MOM transfer, findings were presented only in aggregate, so the magnitude of individual differences could not be seen. 29 Thus, the question of whether LATCH scores could substitute for test-weights in term infants was not adequately addressed in this study. 29 Furthermore, although term, these infants were hospitalized in the neonatal intensive care unit (NICU) at the time of study, which may influence findings. 29
Roznowski et al. measured test-weights to address one of four objectives in a study designed primarily to determine whether MOM production capacity could be increased by adding a short-term, frequent pumping regimen to on-cue breastfeeds. 25 A total of 23 mother-term infant dyads were studied at a mean of 54 days postpartum, and a subset of 16 mothers performed in-home test-weights for 48 consecutive hours. 25 Researchers estimated individual mothers’ 24-hour MOM production rates on the basis of the short-term pumping intervention, and used Bland–Altman plots to examine paired differences between this estimate and real 24-hour MOM transfer. Estimated 24-hour MOM production rate overestimated real 24-hour MOM transfer by 368 ± 234 g, leading investigators to conclude that MOM production rate cannot substitute for test-weights if the variable of interest is MOM transfer. 25
Kent et al. leveraged in-home test-weights to validate mothers’ perceptions of insufficient MOM production during the first 4 weeks postpartum. 10 Mothers with (n = 103) and without (n = 13) perceived insufficient MOM were enrolled between 6–28 days postpartum, and were provided a digital scale to perform one set of in-home, 24-hour test-weights during the study, 62% of whom complied. 10 Mean MOM transfer was significantly lower in mothers with versus without perceived insufficient MOM (693 mL versus 399 mL; p < 0.001). 10 However, nearly half (48%) of mothers who perceived insufficient MOM volume between 6 and 13 days postpartum and the majority (69%) who perceived insufficient MOM between 14 and 28 days were actually transferring adequate MOM volumes (≥440 mLs daily). 10 Kent et al. concluded that in-home measurement of MOM transfer via test-weights can provide valuable, actionable data to address early unplanned weaning in otherwise healthy dyads. 10
Test-weights to personalize infant-specific intake of MOM components
Two studies used test-weights to determine infant-specific amounts of MOM components ingested, rather than assuming a default intake based on generic concentrations.32,34 Both studies involved collecting MOM samples to measure the actual concentration of the component of interest, then personalizing this concentration to the absolute MOM amount ingested by the infant.32,34 George et al. enrolled 20 mothers of healthy term infants who provided MOM samples and completed 24-hour test-weights at 3 months postpartum in order to determine whether estimated MOM lipid intake, using generic references, approximated actual lipid intake by the infant. 32 Lipid content was measured via creamatocrit, an accurate and reliable POC technology that requires only drops of MOM. 44 Grote et al., who enrolled 174 healthy term dyads between birth and 12 months postpartum, measured 3-day test-weights along with 3-day maternal dietary history. 34 A subset of 30 mothers also provided MOM samples over a 3-day period at 1, 2, 3, and 6 months postpartum. 34 Unlike the George et al. study, in which test-weights and MOM samples were performed in “real time” for all subjects, Grote et al. relied on imputed data from the subset of 30 mothers to analyze data and make conclusions for the sample of 174 mothers.32,34
Test-weights to inform or predict adequacy of MOM transfer
Two studies focused on describing or predicting later MOM volume using early postpartum 24-hour test-weights.36,38 Takahata et al. used test-weights on 3–4 days postpartum and at 1 month postpartum to predict exclusive breastfeeding at 4 months postpartum. 38 Investigators concluded that while test-weighing remains “controversial,” it informs and predicts lactation risk. 38 Suwaydi et al. reported early lactation outcomes in a case series of 8 mothers with gestational diabetes mellitus (GDM) who were breastfeeding healthy term infants. 36 Multiple lactation outcomes were measured at 1 and 7 days postpartum, and 24-hour in-home test-weights were performed at 21 days postpartum by all 8 mothers. 36 Investigators concluded that GDM is associated with sub-optimal lactation outcomes, including low MOM transfer, despite high maternal intent and frequent breastfeeds and/or pumpings. 36
Mammary gland function and health
Two studies incorporated two different POC technologies to measure specific mammary gland function and/or health (Table 4).40,41 Both studies used LAQUAtwin (Horiba, Japan) ion-selective electrode probes to assess paracellular pathway patency in the mammary gland using MOM biomarkers of sodium (Na) concentration and sodium-to-potassium ratio (Na:K ratio).40,41 These studies were based on established lactation biology about closed and patent paracellular pathways.6,40,41 Specifically, when paracellular pathways are closed, as during established lactation in the healthy breast, MOM Na and Na:K ratio are low, although actual values for each may vary slightly according to the study and population. 6 When paracellular pathways are patent, MOM Na and Na:K ratio are elevated, as they are prior to the achievement of SA, during weaning and with mastitis. 6
In a blinded study using 92 MOM samples collected at 10 days postpartum, Esquerra-Zwiers et al. compared the accuracy of Na and K concentrations measured by ion-selective electrodes with values obtained using inductively-coupled plasma optical emission spectrometry (ICP-OES), a non-POC technology that is accurate and reliable.40,45,46 No statistically significant differences in Na concentration between methods was noted, but K concentrations measured by the ion-selective electrode probe over-estimated the ICP-OES, impacting the Na:K ratio. 40 Investigators concluded that although POC ion-selective electrodes accurately measure Na concentration and can be leveraged in clinical practice, additional refinement for POC K measurement is indicated. 40
Pace et al. leveraged two POC technologies to study clinical and subclinical mastitis over the first 6 weeks postpartum in a prospective cohort of 41 lactating women who provided 489 usable MOM samples. 41 Ion-selective electrodes were used to measure MOM Na and Na:K ratio, and somatic cell count was quantified using an automated cell counter (DeLaval cell counter, DeLaval International AB, Tumba, Sweden). 41 Somatic cell counts, a long-standing diagnostic technique in the dairy industry, reflect an immune response to the presence of bacteria, with lower counts reflecting a healthy mammary gland and elevated counts reflecting inflammation, infection, or mastitis. 41 Although associations between elevated Na, Na:K ratio and somatic cell count and both subclinical and clinical mastitis were reported, investigators concluded that the ability of these POC measures to serve as prognostic biomarkers was weak. 41
Discussion
Many POC lactation technologies have their origins in human milk field studies conducted without access to standard laboratory equipment and measures, 6 including portable infant scales to measure weight change and MOM transfer, 47 MOM biomarkers of paracellular pathway patency, creamatocrits to measure MOM lipid content, and others.48,49 The accuracy and reliability of these technologies were established in classical lactation studies decades ago.6,44,47,50 More recently, many technologies have undergone additional translational research to embed them into routine NICU practice where objective and quantifiable measures are fundamental to evidence-based lactation care for mothers and NICU infants.51–54 However, to our knowledge, this is the first integrative review to examine the application of these POC measures to the term dyad with the aim of addressing early, unplanned lactation cessation.
Of the 19 articles included in this review,10,18,25–41 16 focused on the use of digital scales for measurement of infant weight change and/or infant MOM transfer,10,18,25–27,29–39 with an overall consensus that this technology is noninvasive, inexpensive, portable, and widely available. Studies also indicate that mothers are able to perform in-home test-weights,10,18,25,26,31,32,34,36–39 which is consistent with findings from a classical study conducted by Kent et al. in 2015 that found in-home test weights improved maternal confidence. 12 In that study, Kent et al. enrolled 203 healthy term mother-infant dyads between 2 and 13 weeks postpartum who were either confident (N = 135) or not confident (N = 68) about their breastfeeding experiences. 12 Mothers completed 24-hour test-weights and pre-and post-surveys assessing specific breastfeeding concerns. 12 Overall, mothers were positive about the informational value of test-weights. 12 Those who were initially confident remained confident, indicating that test-weights did not undermine breastfeeding. 12 For mothers who were not confident and/or were supplementing frequently, test-weights revealed either adequate MOM transfer or identified those dyads with real insufficient MOM transfer. 12 An important takeaway from Kent et al. is that many mothers with and without confidence were previously unconvinced that their breastfeeding experience was normal, despite visualizing adequate weekly infant weight gain and receiving assurance from health care providers. 12 Hurst et al. reported similar data about maternal experiences with in-home test-weighing during the first month after NICU discharge, 55 and the literature is replete with older studies in discharged preterm infants that test-weights are acceptable to mothers. 56
The findings from this review and older studies suggest that in-home and/or in-office test-weights may be an ideal POC technology to help distinguish between perceived and real insufficient MOM volume to address early, unplanned lactation cessation. Contrary to the concerns of many lactation supporters, our findings did not support the assumption that POC test-weights are stressful for mothers and/or undermine breastfeeding.10,18,25,26,31,32,34,36–39 Furthermore, our review adds to the existing literature that use of visual cues to estimate breastfeeding effectiveness does not translate into accurate measurement of MOM transfer in term or preterm dyads.29,52,54 Also relevant is that assurances from pediatric and/or lactation specialists are insufficient for some mothers to feel confident in their breastfeeding.
Although only two studies in this review reported on the use of ion-selective electrode technology to detect aberrations in paracellular pathway patency in healthy term dyads, there is an extensive history of using MOM biomarkers for this purpose. 6 Originating in the dairy industry for detection of mastitis, 41 MOM Na concentration and Na:K ratio were subsequently used in human field studies (albeit without POC measures) to assess clinical and subclinical mastitis, specifically in studies related to HIV transmission via breastfeeding. 57 Neville et al. have published extensively on the use of Na concentration and Na:K ratio (not POC) to measure paracellular pathway closure as a fundamental step in the achievement of SA.58,59
The portable ion-selective electrode probe (LAQUAtwin Na: B-722, K: B-731; Horiba, Japan) has recently become available for measuring MOM Na and K concentrations, and accuracy of the Na concentration has been validated against ICP-OES.40,45 The feasibility of this POC technology is underscored by its affordability, portability, and easy maintenance and use, indicating its potential utility for practice and research. 51 Multiple studies have used these ion-selective electrode probes in the NICU population, primarily as a mechanism to detect and manage delayed/impaired SA achievement and lack of permanence of paracellular pathway closure after SA achievement.51,60 However, additional research needs to address whether Na:K ratio adds to the Na concentration alone, and whether inter-feed/pump interval and within-breast differences affect these MOM biomarkers. 61
Alternatively, the accuracy of a new POC device that purports to measure SA progress for the individual mother has been recently reported. 62 Evolving from the dairy industry and measuring MOM conductivity on the basis of electrolyte concentration, 63 the Mylee is an indirect measure of MOM Na concentration. 62 At the time of this writing, few data are available to inform its feasibility and/or outcomes related to research or clinical use.
Our review has highlighted major research gaps in the use of POC technologies to address early, unplanned lactation cessation in term dyads. An overarching research priority is the development and testing of evidence-based algorithms that distinguish between perceived and real insufficient MOM volume within the first postpartum week. A second priority is the detection of modifiable maternal and infant risk factors that can lead to real insufficient MOM volume if left unaddressed, including maternal metabolic disease and late preterm/early term birth. Relevant POC technologies might include infant weight change, MOM transfer, and MOM Na measurement of SA achievement. In addition, barriers to the use of these technologies, such as unfounded assumptions about maternal stress and burden, must be addressed. Finally, testing innovative models of lactation care that incorporate these POC technologies, such as breastfeeding peer counselors and doulas that visit mothers in the home, should be developed and tested. Central to these priorities is the inclusion of cost analyses to determine the economic impact of POC technologies and lactation practice models on the prevention of early, unplanned lactation cessation.
Summary and Conclusions
Perceived and real insufficient MOM volume with origins in the first 14 days postpartum is a major risk for early, unplanned lactation cessation in term dyads. Promising POC technologies that identify and mitigate risk in this population include digital scales to measure infant weight change and/or MOM transfer and ion-selective electrode probes that measure SA achievement and maintenance of paracellular pathway closure. Research priorities include development and testing of algorithms that integrate these POC technologies into evidence-based lactation care during the early postpartum period. In addition, the expansion of this technology into existing models of lactation care, such as breastfeeding peer counselors and doulas, hold potential as cost-effective strategies, especially in underserved populations.
Footnotes
Authors’ Contributions
L.H.B.: Conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, writing—review and editing, visualization, project administration. S.D.-J.: Methodology, formal analysis, writing—original draft. K.T.K.: Methodology, formal analysis, writing—original draft. C.M.-P.: Methodology, formal analysis, writing—original draft. L.A.P.: Conceptualization, methodology, writing—review and editing, supervision. A.L.E.-Z.: Conceptualization, methodology, formal analysis, investigation, data curation, writing—original draft, writing—review and editing, visualization, project administration, supervision.
Disclosure Statement
L.A.P. is a consultant for Medela AG and Otsuka Pharmaceutical Factory, Inc.
Funding Information
The authors have no funding to disclose.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
