Abstract
Background:
An estimated 60% of mothers experience early, unplanned lactation cessation and do not meet their breastfeeding goals, increasing the risk of maternal and infant health problems and their associated costs. Perceived or real insufficient mothers’ own milk (MOM) during the first 14 postpartum days is a frequently reported reason for early, unplanned lactation cessation. These first 14 postpartum days include the lactation phases of secretory activation (SA) and coming to volume (CTV; ≥500 mL MOM daily) achievements and are affected by biological, behavioral, and economic factors.
Methods:
A framework for studying early, unplanned lactation cessation was developed by researchers with expertise in lactation science, neonatal research, healthy population research, public health, basic sciences, and economics.
Results:
Current research and clinical models primarily address behavioral factors, including maternal perceptions of insufficient MOM volume and social support from family or peers. While important, these models do not include biological and economic factors known to affect early, unplanned lactation cessation. The proposed integrated framework incorporates biological, behavioral, and economic factors with the centrality of mammary gland stimulation and MOM removal.
Conclusion:
This framework may be used to develop and test innovative research and clinical models and may especially be useful in addressing inequities among populations at higher risk for early, unplanned lactation cessation.
Introduction
In the United States, 83% of mothers initiate lactation, but 60% do not achieve their lactation goals, often due to concerns about insufficient mothers’ own milk (MOM) volume.1,2 For many mothers, perceived or real insufficient MOM volume originates in the first 14 postpartum days, which is a critical window for biological processes that program the mammary gland for long-term regulation of milk synthesis and secretion. 3 Perceptions of insufficient MOM volume during this early postpartum period are frequently related to the normally small amounts of MOM produced and infant behaviors such as fussiness and frequent feeds. 4 Although potentially modifiable, perceived insufficient MOM volume often leads to suboptimal lactation outcomes, including formula supplementation, partial rather than exclusive breastfeeding, and early, unplanned lactation cessation. 4
There is a growing number of women who experience real insufficient MOM volume, many of whom have unmodifiable biological lactation risk factors such as preterm birth, obesity, diabetes, hypertensive disorders of pregnancy (HDP), and other comorbidities.5,6 Some mothers may find that the economic trade-offs necessary to achieve early lactation goals are not feasible, especially when balancing paid and unpaid work with frequent breastfeeding. 7 These biological and economic risks disproportionately impact Black, Hispanic, and other systemically marginalized populations and likely explain some of the inequities in early, unplanned lactation cessation.7–9 However, previous research and clinical models that address early lactation problems in healthy and at-risk populations have focused primarily on behavioral factors, such as motivation, support, and education.
Decades ago, the biological importance of the early postpartum period was characterized by Chapman and Perez-Escamilla as a public health priority for the achievement of long-term lactation goals. 10 Two fundamental, highly complex lactation phases must be completed during this time. First is the achievement of secretory activation (SA; lactogenesis II, MOM coming in), which is primarily a hormonal process triggered by the birth of the placenta. 3 Second is the switch to the autocrine/paracrine control of lactation, which results in the achievement of a threshold MOM volume to support infant growth and development, also known as coming to volume (CTV; ≥500 mL MOM by 14 postpartum days).3,11 All too often the critical nature of this early postpartum period is not understood, even by credentialed practitioners, nor is it routinely controlled for in original research when MOM volume or exclusive breastfeeding serves as an outcome. 12 The purpose of this article is to briefly outline the normal biology of SA and CTV achievements and to propose a framework for research and practice that addresses early, unplanned lactation cessation. Research and practice gaps, as well as implications for disparities and equity, are highlighted throughout.
Framework for Addressing Early, Unplanned Lactation Cessation
Figure 1 depicts the biology of the first 14 postpartum days for both healthy mothers and those who are breast pump-dependent with preterm infants in the neonatal intensive care unit (NICU). Figure 2 depicts the complex interplay of factors affecting early, unplanned lactation cessation, defined here as discontinuing the provision of MOM via breastfeeding or pumping during the early postpartum period, especially within the first 14–30 days. Central to this framework is the important role of mammary gland stimulation and MOM removal, which should be optimized as a first step for both research and practice.
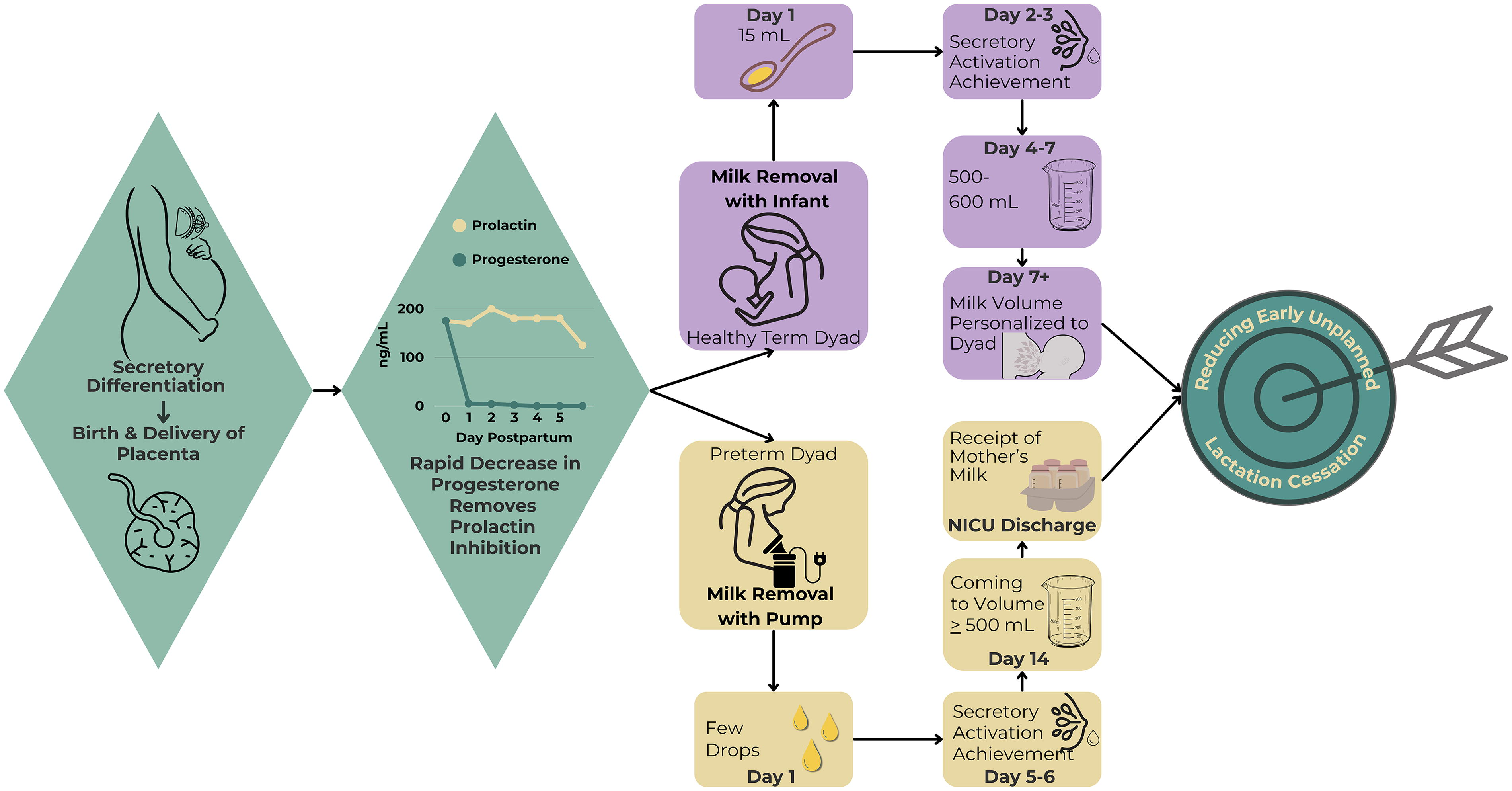
Critical lactation milestones in the first 14 postpartum days.
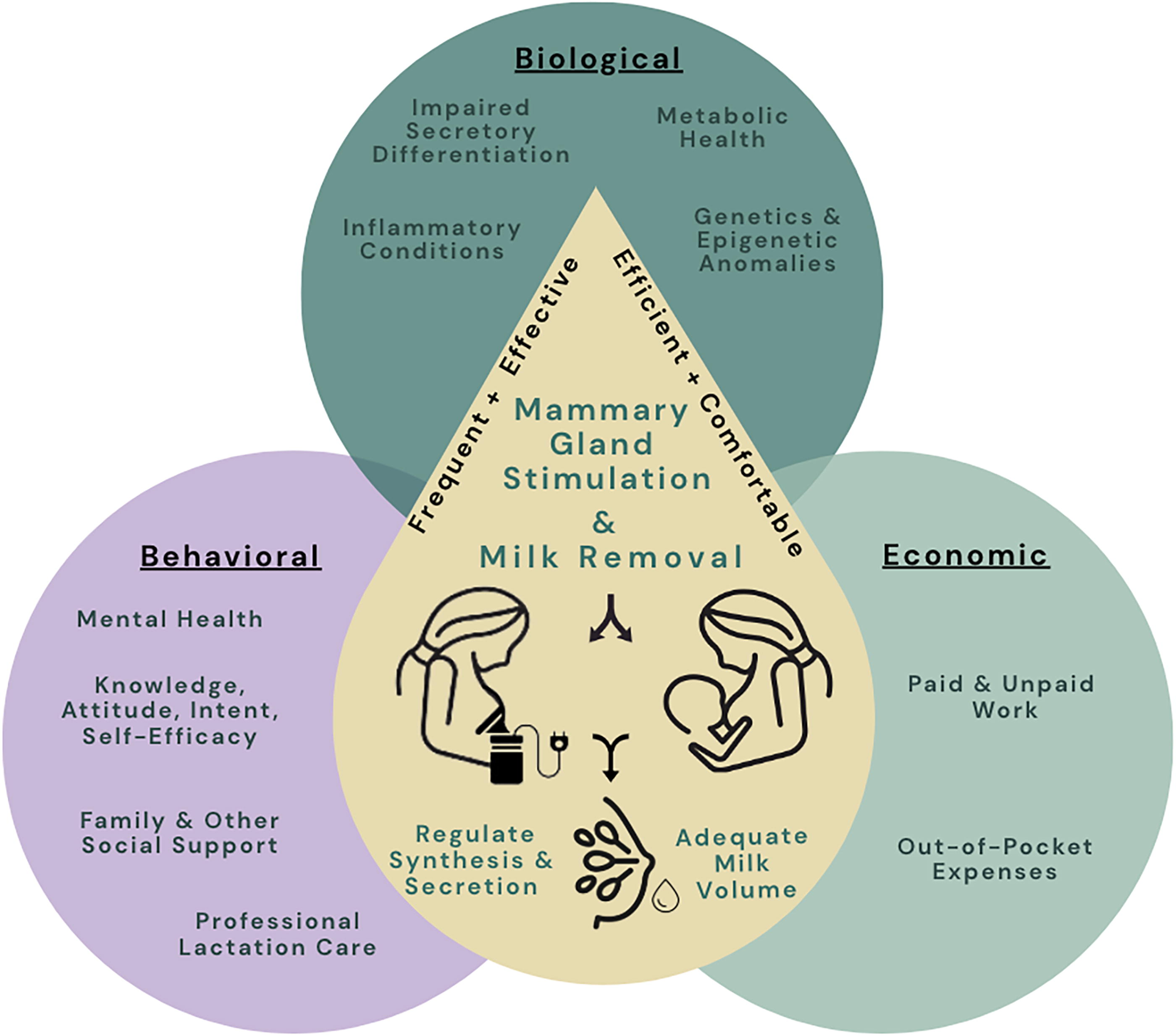
Theoretical framework of the factors affecting early, unplanned lactation cessation.
Mammary gland stimulation and MOM removal
Prior to the achievement of SA, when little MOM volume is available, frequent infant suckling or use of an initiation breast pump suction pattern provides human-specific stimulation to the mammary gland, catalyzing SA achievement.13,14 SA involves the upregulation of four transcellular pathways: exocytosis (protein and lactose), milk-fat globule (fat), secretion across the apical membrane (ions and water), and pinocytosis/exocytosis (immunoglobins). 15 Additionally, during SA the paracellular pathway closes, allowing lactose, the primary osmolyte in MOM, to remain in the gland and upregulate MOM volume. 3
Upon achievement of SA, effective MOM removal (i.e., removing as much available MOM as possible) stimulates the release of prolactin from the anterior pituitary gland, resulting in a two- to threefold increase in circulating prolactin within 30–45 minutes of the start of MOM removal. 16 Differences between baseline and suckling-induced peak prolactin concentrations are the highest during the first 10 postpartum days, signaling the importance and perhaps the programming nature of this time frame. 16 Ineffective MOM removal triggers downregulation of prolactin via multiple autocrine/paracrine mechanisms.17–19 Efficient MOM removal (i.e., optimal rate of milk flow) increases the likelihood of timely SA and CTV achievements as well as infant satiety and maternal perceptions of “normalcy” of breastfeeding.20–22 Comfortable MOM removal, whether by infant or pump, is essential because pain triggers the dopamine-like prolactin inhibiting factor and is linked to early, unplanned lactation cessation. 23 Continued MOM synthesis is regulated through autocrine and paracrine feedback mechanisms related to MOM removal, ductal stretching, and local signaling mechanisms.17,21 Therefore, frequent, effective, efficient, and comfortable MOM removal, or lack thereof, drives a feedback loop, which is fundamental to increasing, decreasing, or maintaining MOM volume. 21 Biological, behavioral, and economic factors interact with and impact this fundamental mechanism.
Recent studies with mothers of preterm infants have reported impermanence in mammary gland tight junction closure following SA achievement during the first 14 postpartum days, likely associated with infrequent or ineffective milk removal.24,25 Importantly, MOM removal can be frequent but ineffective, as in the case of infant conditions that compromise latch and sucking behaviors, such as with late preterm/early term birth, oral malformations, restrictive tethered oral tissues, and hyperbilirubinemia.12,26,27 Similarly, breast pump-dependent mothers may remove MOM frequently but ineffectively due to inferior breast pumps, incorrectly sized breast shields, single versus double pumping, and other improper pumping techniques. 28 Not to be overlooked are other common behavioral factors contributing to ineffective and inefficient MOM removal, such as postponing or shortening feeding or pumping sessions due to the presence of visitors or the needs of other family members. 29
Biological Factors Influencing Early, Unplanned Lactation Cessation
An array of biological factors may influence early lactation processes, beginning during pregnancy when mammary epithelial cells change from their undifferentiated ductal stage to specialized alveolar mammary epithelial cells, a process called secretory differentiation. 21 Animal and other basic science research has identified lactation risks related to impaired secretory differentiation, maternal metabolic health, maternal inflammatory conditions, and genetic and epigenetic anomalies.17,30–34 While some of these concepts have been studied in humans, this area represents a priority research gap, given the number of mothers with these comorbidities of pregnancy and their clinical link to early lactation problems.
Impaired secretory differentiation
Several maternal health conditions potentially affect mammary gland development during pregnancy, including metabolic disorders and inflammatory conditions such as obesity and HDP.5,6 Additionally, mammary development is interrupted by preterm birth, and mothers who deliver preterm infants often have comorbidities, including obesity and HPD, that impact secretory differentiation and may manifest clinically as delayed and/or impaired SA. 35
Placental growth impacts mammary gland development and is associated with MOM volume as evidenced by animal models. 30 For example, placental size and number of placental cotyledons have been associated with MOM volume in dairy cattle. 30 However, very few human studies have investigated the role of the placenta in lactation. In a sub-analysis from the Mothers, Infants, and Lactation Quality study, researchers found that maternal blood lipids, insulin, and leptin measured prenatally at 28 weeks of gestation were associated with both duration of exclusive breastfeeding and placental size. 36 However, they did not directly investigate associations between the placenta and lactation. 36 Although further human studies are needed, it is hypothesized that placental inflammation interferes with placental development resulting in reduced synthesis of lactogens, growth hormones, oxytocin, serotonin, and other hormones required for mammary gland development (Fig. 3).30,36,37 Prenatal growth in breast volume may be a marker of mammary glandular tissue growth and differentiation and is a strong predictor of MOM volume at 1–6 postpartum months. 37 Because of the placenta’s critical role in mammary development, it is a research priority to understand the biological environment of pregnancy to inform the biology of early, unplanned lactation cessation.30,36,37
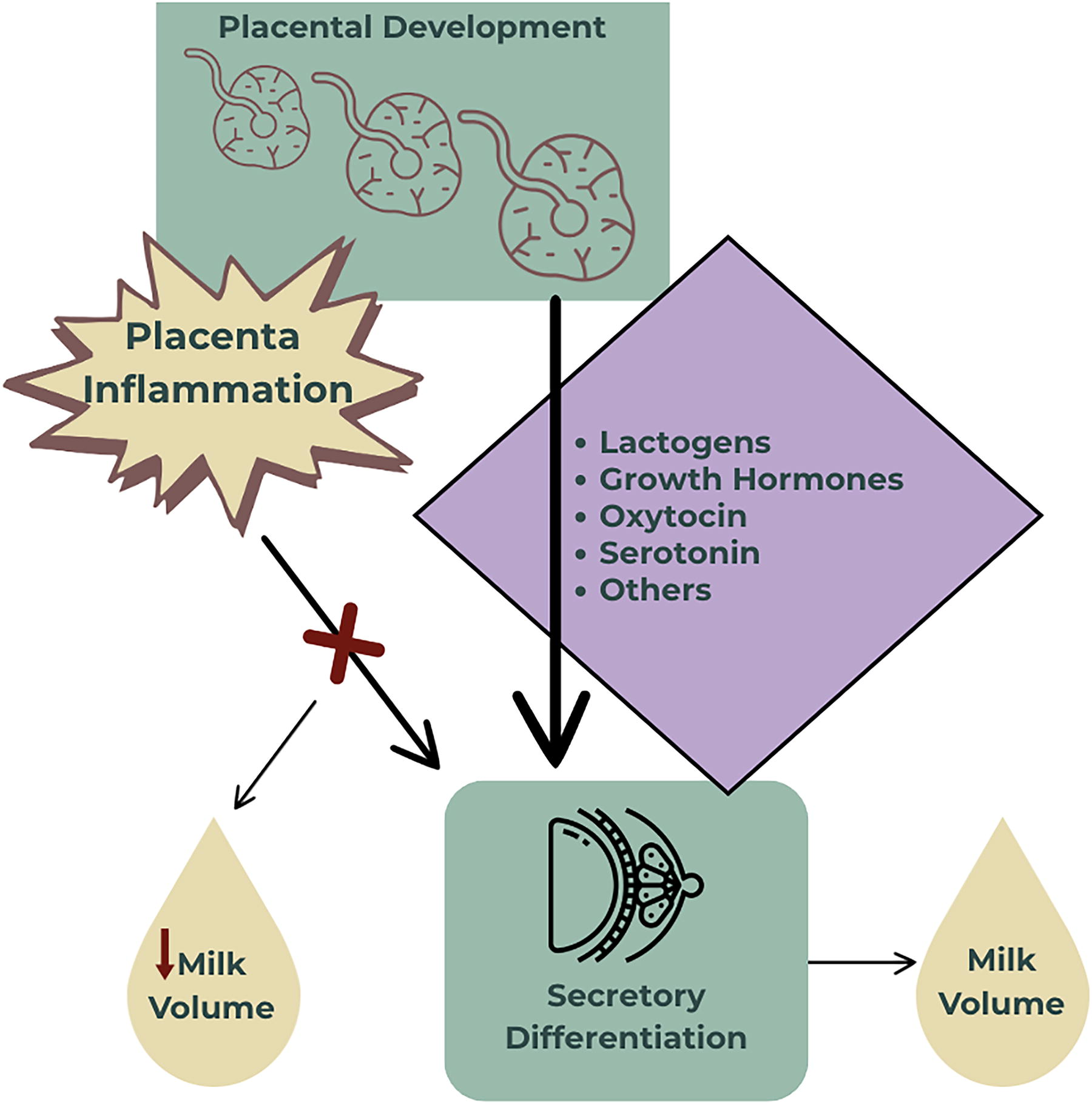
Theoretical effect of placental inflammation on secretory differentiation and subsequent milk volume.
Maternal metabolic health
Excess adiposity (measured as obesity and overweight by body mass index [BMI]; obesity ≥30 kg/m2 and overweight 25 to <30 kg/m2) 38 and excess gestational weight gain are key risk factors for delayed SA and shorter lactation duration, independent of behavioral interventions and frequent breast emptying.39–41 Obesity affects various metabolic signaling pathways, is a cause of inflammation, and can result in insulin resistance and impaired glucose tolerance. 6 Early animal studies suggest that excess adiposity alters endocrine regulation of mammary development and function 42 ; specifically, rats with excess adiposity had less pronounced increases in prolactin after birth. 43 In the only human study, Rasmussen and Kjolhede found that maternal obesity was associated with a blunted prolactin response to infant suckling. 31 However, this study did not measure infant sucking effectiveness with respect to MOM removal, a potentially important confounder. 31 To our knowledge these results have not been replicated in a more controlled design and may best be accomplished via breast pump use where suction rates, pressures, and rhythms can be standardized across subjects in a way that is not possible with breastfeeding infants. 12
Progesterone inhibits the action of prolactin, likely through cross-talk in the signal transducer and activation of transcription 5 (STAT5) signaling pathway regulated by both prolactin and progesterone receptors. 44 Although serum progesterone does not differ with BMI, 31 progesterone is found in high concentrations in both human and bovine adipose tissue, and sequestration by the adipose tissue is thought to be a primary mechanism of progesterone clearance from blood.44,45 Therefore, researchers have proposed a promising hypothesis that high levels of retained progesterone in the mammary adipose tissue postpartum may delay SA achievement.44,46 To date, this hypothesis has not been tested experimentally in humans.
Recent studies have identified the role that overweight and obesity have in MOM fat metabolism and synthesis which impacts SA achievement.47,48 The large CHILD cohort study in Canada found that overweight and obesity were positively associated with MOM fatty acid profiles that were high in mid-chain fatty acids and negatively associated with exclusive breastfeeding. 48 This study also reported that MOM mid-chain fatty acids were positively associated with a common mutation in the FADS2 gene (rs174575—GG), which codes for the delta-6 desaturase enzyme involved in the production of long-chain omega-3 and omega-6 fatty acids. 48 Other researchers have shown the upregulation of lipid metabolism genes in the early weeks of lactation, which is reflected in the MOM fatty acid profile in healthy, lactating mothers. 47 These findings suggest that gene-regulated lipid metabolism and synthesis of the MOM fat globule are crucial to SA achievement and the establishment and maintenance of lactation.47,48
In addition to obesity, other metabolic factors associated with low MOM production may result from insulin resistance-related delays in SA achievement.6,49 A recent case–control study found that mothers with very low MOM production (<300 mL/day) had a more metabolic syndrome-like phenotype, including elevated fasting plasma glucose, fasting plasma triglycerides, blood pressure, increased waist circumference, and lower high-density lipoprotein cholesterol compared with exclusively breastfeeding controls. 6 While no universal BMI threshold has been definitively linked with suboptimal lactation outcomes in mothers of term or preterm infants, 5 Medina-Poeliniz et al. found significantly delayed and/or impaired SA in breast pump-dependent mothers of preterm infants with BMI ≥27 versus those with BMI <27. 5 Further research is needed to determine whether BMI is the optimal measure of adiposity for assessing obesity-related lactation risk.
Maternal inflammatory conditions
Inflammatory signaling in the mammary gland is associated with reduced MOM production. 50 Specifically, the pro-inflammatory cytokine tumor necrosis factor-α (TNF-α) is elevated in obese mice and has been shown to suppress lactose synthesis in cultured mammary epithelial cells, likely through decreased activation of the transcription factor, STAT5. 50 TNF-α also downregulates glucose transporter 12 (GLUT12) expression and enhances the degradation of GLUT1, transporters necessary for carbohydrate transport within the mammary epithelial cells. 50 In humans, MOM TNF-α is elevated in subclinical mastitis, which is an inflammatory condition, and in mothers with very low MOM volume compared with exclusively breastfeeding controls. 51 Furthermore, mothers with very low MOM volume have disrupted transfer of fatty acids from blood to milk. 51 Mammary uptake of fatty acids is mediated by lipoprotein lipase, 52 which TNF-α inhibits. 53 Together, these studies suggest that inflammation, such as that seen with overweight and obesity, disrupts the synthesis of carbohydrates and lipids in the mammary gland, potentially leading to low MOM volume.51–53
Genetic and epigenetic anomalies
Genetic predispositions may influence the production of critical components in MOM, affecting MOM volume. 32 Genome-wide association studies with dairy animals identified multiple genomic regions associated with MOM volume.54–57 However, to date, no large-scale genomic study associates human MOM production with maternal genetics.
Individual mutations in prolactin (PRL) and its receptor (PRLR) have been identified in women with both low and no MOM production, suggesting that genetic variations in these genes may contribute to suboptimal lactation outcomes, including early, unplanned lactation cessation.32,58 In addition, mutations in Zinc transporter 2 (ZnT2) have been associated with low MOM zinc concentration, which may be related to low MOM production.59,60 In a study evaluating the association between specific maternal genotypes (single nucleotide polymorphisms) and low MOM production, investigators identified a specific genetic variant (rs2271714) in MOM fat globule epidermal growth factor and factor V/VIII domain-containing gene (MFGE8) associated with self-reported insufficient MOM volume and shorter breastfeeding duration. 61 Finally, the minor allele of the oxytocin receptor (OXTR) gene (rs53576) may be associated with breast and nipple pain, 62 potentially contributing to early, unplanned lactation cessation. Results from these studies suggest genetic variations may be important in determining the adequacy of MOM production and lactation outcomes and represent an important research priority.
Animal models suggest that epigenetic changes, such as chromatin accessibility, DNA methylation, and microRNA gene expression regulation, may affect MOM production and lactation outcomes. 33 Only a few human cohorts have addressed the relationship between maternal epigenetic changes and MOM volume. Hicks et al. found significantly higher MOM levels of micro-ribonucleic acids (miRNAs) let-7g-5p and miR-22-3p on seven postpartum days in mothers with self-reported low MOM volume suggesting that MOM miRNA might be used as a biomarker for low MOM production. 33
Behavioral Factors Influencing Early, Unplanned Lactation Cessation
The various maternal mental health, knowledge/attitudes/intent/self-efficacy, family and other social support, and professional lactation care factors that are linked with early, unplanned lactation cessation have received more study than either biological or economic factors.63–74 However, behavioral factors can be heavily intertwined with biological and economic risks, such as lack of social support alongside maternal obesity and low household income.39–41,72,75,76 Focus on a singular behavioral factor may mask the impact of compounded risks in both research and practice.
Maternal mental health
Maternal mental health, including perceived stress, depression, and anxiety, are associated with impaired lactation. 64 Multiple biological mechanisms may explain this relationship, including inhibition of prolactin, oxytocin release interference, reduction of insulin sensitivity, and dysregulation of the hypothalamic–pituitary–adrenocortical axis. 64 Mood disorders are more common in systematically marginalized populations and in mothers who experience unstable housing, have no private space to breastfeed/pump, experience food insecurity, return to work soon after birth, care for other children or family members, or experience interpersonal violence.8,64,77 These burdens often lead to infrequent and/or ineffective MOM removal, downregulating MOM synthesis. 8 Similarly, mothers who deliver preterm infants commonly experience distress symptomatology, especially post-traumatic stress disorder, which can interfere with the abovementioned MOM synthesis and secretion mechanisms as well as frequent breast pump use. 78
Maternal knowledge, attitude, intent, and self-efficacy
Knowledge, attitude, intent, and self-efficacy are interrelated and modifiable factors known to affect lactation outcomes.65–71 While prenatal lactation education has been associated with increased knowledge, positive attitude, and self-efficacy, it may not always translate into improved lactation outcomes, often related to postpartum complications that require professional lactation care. 66 Attitude, often influenced by knowledge, is the intensity of a mother’s beliefs about the benefits of providing MOM and is strongly associated with lactation intent.67,68 Although prenatal lactation intent strongly predicts exclusive breastfeeding within the first 3 postpartum days, few studies have examined the association between prenatal intent and exclusive breastfeeding up to 6 postpartum months.69,79 Breastfeeding self-efficacy has been well studied among term dyads and shows that mothers with low self-efficacy during the first few postpartum weeks are more likely to have perceived insufficient MOM volume that leads to early, unplanned lactation cessation. 71 Although one study found that objectively measuring infant MOM intake using test weights improved maternal confidence, this finding has not been routinely implemented into clinical practice. 65 More translational work is needed to integrate objective measures such as test weights and MOM biomarkers of SA into routine clinical interventions that address lactation self-efficacy. 65
Family and other social support
Family members, particularly partners and maternal grandmothers, are highly influential in the initiation and maintenance of lactation, including early, in-hospital exclusive breastfeeding.72,73 Due to forced wet nursing during slavery, targeted marketing of formula, and other factors related to structural and interpersonal racism, many Black families did not breastfeed for multiple generations, resulting in minimal lactation experience and less social support for current generations.72,75,76 Limited social support can reinforce maternal perceptions of low MOM volume, especially in the early postpartum period. 72 A recent integrative review revealed an inverse relationship between maternal perceptions of “support” from the Black maternal grandmother and continued MOM provision in Black mothers of preterm infants cared for in the NICU. 80 Although the Black family matriarch, especially the maternal grandmother, is often regarded as the expert for childcare, she may not have experience with a NICU infant and may offer support that does not align with the needs of the breastfeeding mother. 80
An innovative strategy may be the integration of breastfeeding peer counselors into settings including the NICU, prenatal clinics, postpartum units, and the Special Supplemental Nutrition Program for Women, Infants, and Children.72,81,82 Additionally, doulas, who are now reimbursed through Medicaid in some states, may be important providers of lactation care.83–85 While doula support has been associated with higher lactation initiation rates, few studies have examined the relationship between doula care and continued lactation or exclusive MOM provision. 85 A research opportunity exists to equip breastfeeding peer counselors and doulas who serve marginalized communities with additional tools to address early, unplanned lactation cessation in the home environment, including the use of point-of-care technologies such as selective ion electrodes to measure MOM biomarkers of SA achievement and test-weights to measure infant MOM intake. 86
Professional lactation care
Physicians, advanced practice providers, nurses, nutritionists, and dietitians who work in maternal and infant health receive lactation education during training programs and provide varying levels of clinical breastfeeding care. 74 Certified Lactation Consultants and Certified Lactation Specialists receive advanced lactation training and can provide general lactation care, including emotional, informational, and basic hands-on assistance. 87 International Board Certified Lactation Consultants can develop individualized plans of care to assist families in meeting their lactation goals. 87 However, given the increasing constellation of biological risks, the expertise of lactation specialists is essential, especially breastfeeding medicine practitioners (physicians and advanced practice providers), who can provide directed lactation care, including procedures and prescription medications. 88 Many of the risk factors for early, unplanned lactation cessation can be mitigated with targeted professional lactation care in the first postpartum weeks, 63 so evidence-based protocols that identify and refer vulnerable mothers during the maternity hospitalization are a research priority. 88 Unfortunately, limited access to professional lactation care often exists due to geographic and economic barriers, leaving practitioners not trained in lactation care to intervene in complicated lactation scenarios. 89
Economic Factors Influencing Early, Unplanned Lactation Cessation
Although multiple studies reveal that high-dose MOM reduces the risk of potentially modifiable health problems and their costs for term and preterm infants and their mothers, MOM provision is not cost-free.90–92 Mothers and their families bear these costs, which may affect biological lactation processes that lead to early, unplanned lactation cessation. 93
Paid and unpaid workload
Breastfeeding and pumping MOM require time, an often-overlooked barrier only the mother encounters, and more time spent providing MOM means less time for other paid or unpaid activities.94,95 Thus, new mothers face decisions about allocating their time between breastfeeding or pumping versus other unpaid activities, such as family care, household tasks, and paid work. 7 These time allocation decisions are critical in the early postpartum period because each additional hour spent pumping over the first 5 postpartum days directly affects SA achievement and pumped MOM volume in breast pump-dependent mothers of preterm infants. 96 Although comparably granular data are not available for healthy term dyads, mothers are encouraged to breastfeed their infants 8–12 times per day during this time frame. 97 One study reported that mothers of exclusively breastfed term infants spent 16 hours per week (approximately 2.3 hours per day) in feeding activities at 3 months postpartum, 94 and mothers of very low birth weight (<1,500 g birth weight) infants spend an average of 70–120 minutes per day pumping MOM and cleaning equipment over their infant’s NICU stay.94,95 Although perceived lack of time and trade-offs with paid and unpaid activities likely contribute to early, unplanned lactation cessation, few interventions have been aimed at overcoming these barriers in the early postpartum period.
Out-of-pocket expenses
The economic burden of MOM provision extends beyond opportunity costs, including out-of-pocket expenses, such as nursing bras and pads, milk storage containers, and breast pump purchase or rental. Breast pump-dependent mothers must often self-pay for the rental of hospital-grade electric breast pumps, despite data showing their effectiveness, efficiency, and comfort. 23 The Patient Protection and Affordable Care Act increased access to breast pumps for Medicaid recipients with the intent of increasing equity in MOM provision. 98 However, these pumps are not intended for breast pump dependency, and multiple barriers exist to accessing them in a timely manner for all populations.98,99 Requirements such as physical proof of infant birth and prior authorization practices often result in substantial delays during the critical first 14 postpartum days, affecting SA and CTV achievements and maintenance.98,99 These barriers disproportionately affect mothers with low incomes and likely contribute to inequities in this field.
Summary and Conclusions
Biological, behavioral, and economic risk factors impact basic mammary gland stimulation and MOM removal processes, especially during the early postpartum period when complex biological lactation phases must be achieved. Our framework proposes an integrated approach to conceptualizing these risk factors for researchers and clinicians. For researchers, this framework may be used to develop and test innovative models that examine individual factors simultaneously, focus on the intersectionality of factors, and guide the study of mediating and moderating effects. For both researchers and clinicians, this framework may especially be useful in addressing inequities among populations at higher risk for early, unplanned lactation cessation, including mothers of preterm and critically ill infants, those with maternal health risk factors, and systemically marginalized populations.5,6,72,75,83 Implementing this framework will require interdisciplinary collaboration among those who study these factors from bench to bedside and in community and public health settings. Often overlooked in both research and practice is the inclusion of translational lactation scientists to ensure that fundamental components of mammary gland stimulation and MOM removal are addressed and/or controlled so that the impact of other biological, behavioral, and economic factors can be understood.
Footnotes
Authors’ Contributions
L.H.B.: Conceptualization, methodology, writing—original draft, writing—review and editing, and project administration. S.D.-J.: Methodology and writing—original draft. A.E.-Z.: Methodology and writing—original draft. Y.G.M.: Methodology and writing—original draft. J.G.: Methodology, writing—original draft, visualization, and writing—review and editing. K.T.K.: Methodology and writing—original draft. C.M.P.: Methodology and writing—original draft. R.W.: Methodology, writing—original draft, and visualization. T.J.J.: Conceptualization, methodology, writing—original draft, writing—review and editing, and supervision. L.A.P.: Conceptualization, methodology, writing—review and editing, and supervision.
Disclosure Statement
R.W. is a member of the Board of Directors for the International Society for the Study of Fatty Acids and Lipids. T.J.J. is a consultant for Medela AG. L.A.P. is a consultant for Medela AG and Otsuka Pharmaceutical Factory, Inc.
Funding Information
The authors have no funding to disclose.
