Abstract
Background:
Delayed and/or impaired secretory activation (SA) achievement is a risk factor for early unplanned lactation cessation. This pilot study (1) evaluated the feasibility of in-home daily milk collection; (2) compared daily maternal self-report of breast fullness with mother’s own milk (MOM) biomarkers of SA; and (3) examined SA patterns in association with lactation outcomes through 3 months postpartum.
Methods:
Twelve mother–infant dyads were enrolled at term (median gestational age (GA) = 39.9 weeks, range = 38.3–41.1 weeks). After birth, participants self-reported breast fullness and collected milk samples daily for a week. Infant weight gain and milk intake by test-weights were assessed postpartum during a week 1 routine infant outpatient visit and again during a week 2 home visit. Milk samples were tested for sodium (Na) and potassium (K) using portable ion selective electrodes. Timing of SA achievement was denoted as the first day breast fullness was self-rated ≥ 3, Na concentration was < 12 mmol/L, or the Na/K ratio was < 0.6. Breastfeeding duration and exclusivity were measured at 1, 2, and 3 months postpartum.
Results:
By day 6, all mothers self-reported SA achievement, while only 72–82% achieved SA using MOM biomarkers. Cohen’s kappa between the timing of SA by maternal self-report and MOM biomarkers showed lack of agreement (κ = 0.05 and 0.08), but was high between MOM Na and Na/K ratio (κ = 0.87). Three patterns of SA by MOM biomarkers emerged (mature and stable, impermanent, and delayed) with implications for infant weight loss/gain at 1 week postpartum. These patterns were not associated with breastfeeding exclusivity through 3 months postpartum in this highly motivated sample.
Conclusion:
MOM biomarkers of SA achievement are feasible and have significant potential for use in community settings.
Introduction
The first few weeks of lactation are a critical time for the establishment of an adequate milk supply. 1 In particular, the 34% incidence of delayed secretory activation (SA) in the United States is high 2 and has negative implications for excess neonatal weight loss 3 and early unplanned cessation of lactation.1,4 Delayed SA achievement may indicate impaired mammary epithelium function at a critical lactation stage, with the potential to disrupt future milk synthesis. This is significant because perceived insufficient milk supply is the most commonly cited reason for earlier than desired weaning. 5 Therefore, improved screening for SA achievement is needed, particularly in community settings, to better identify and manage mother–infant dyads at risk for early unplanned cessation of lactation and to reassure those for whom SA is progressing normally.6,7
Mother’s own milk (MOM) biomarkers are gaining attention as an objective measure of SA achievement, in part, because portable, accurate, and cost-effective measurement devices have become available.8,9 Sodium (Na), potassium (K), and the Na/K ratio in milk are increasingly being utilized to assess mammary epithelium permeability in mothers of term9–13 and preterm infants.14–16 Both MOM Na and Na/K ratio are highest in early colostrum, but decrease rapidly during the first few days postpartum in response to paracellular pathway closure, while potassium (K) ions accumulate.13,17 Although maternal perception of milk coming in is the most accessible measure of SA achievement in research and is frequently used in clinical practice in community settings, it has not been evaluated in relation to MOM biomarkers in term mother–infant dyads. 13
This pilot study sought to examine the feasibility of using point-of-care measures of SA achievement as predictors of infant health and lactation outcomes in community settings. The purpose of the study was (1) to evaluate the feasibility of in-home, daily milk sample collection for assessing SA achievement during the first week postpartum in healthy, term mother–infant dyads; (2) to compare daily maternal self-report of breast fullness with MOM biomarkers as indicators of SA achievement; and (3) to evaluate the timing and patterns of SA achievement in relation to infant weight gain and milk intake during the first 2 weeks, as well as to lactation duration and exclusivity through 3 months postpartum.
Methods
Study population
This study sought to enroll healthy mothers giving birth to term infants. Flyers for this IRB-approved study were posted in the University of New Mexico prenatal clinics between December 2022 and September 2023. Inclusion criteria were prenatal intent to breastfeed, planned vaginal birth, and term birth (≥ 37 weeks). Exclusion criteria included maternal age < 18 years, preexisting diabetes mellitus, uncontrolled thyroid disease, history of breast reduction surgery, known fetal anomalies that affect breastfeeding (e.g., cleft lip or palate), or an infant hospitalized in the intermediate care nursery or neonatal intensive care unit (NICU) during the entire first week. Written informed consent was obtained from all participants.
Study design
This multimethod, observational pilot study collected data from the time of birth through 3 months postpartum. Eligible participants were consented and enrolled at a baseline visit after 37 weeks of pregnancy but before giving birth. During week 1 postpartum, daily maternal perception of breast fullness was reported by electronic survey, and daily samples of milk were collected for subsequent MOM biomarker measurement. The laboratory technician conducting biomarker measurements was blinded to daily maternal self-report of breast fullness. Week 1 medical records from the infant outpatient visit were reviewed for infant weight loss/gain, milk intake, and feeding history. During postpartum week 2, a home visit was conducted to collect a fresh milk sample, determine infant weight loss/gain since birth, and to measure infant milk intake during breastfeeding. At 1, 2, and 3 months postpartum, breastfeeding duration and exclusivity were assessed by phone calls to mothers. The research assistant conducting these phone calls was blinded to day of SA achievement by self-report and MOM biomarkers.
Measures
Participant characteristics
Maternal demographics were collected by interview during the baseline visit. Information on participant health, pregnancy history, labor course, infant health, and lactation history was abstracted from the electronic medical record.
Breastfeeding intentions
The Infant Feeding Intentions (IFI) Scale 18 was administered electronically at the baseline visit. This validated, 5-item questionnaire was designed to assess the strength of breastfeeding intentions. Items are rated on a 5-point Likert scale, with a higher summary score indicating stronger intentions to breastfeed. The highest possible score is 16.
Maternal self-report of SA
During the first week postpartum, participants were sent electronic surveys each morning via REDCap (Vanderbilt University, Nashville, TN) to assess breast fullness as an indicator of SA achievement. Participants rated breast fullness on a 5-point Likert type scale (1 = no change, 3 = noticeably fuller, 5 = uncomfortably full). 3 SA by maternal self-report was recorded as the first day that breast fullness was rated ≥ 3, as per Dewey et al. 3
MOM biomarkers of SA
Following a morning breastfeed, milk samples were collected daily on postpartum days 2–6, using equipment provided during the hospital stay. Specifically, participants were provided a manual breast pump (Medela, McHenry, IL, USA) and five 10-mL sterile sample collection containers labeled with coded identifiers and postpartum day. Mothers were taught to express and collect approximately 10 mL of milk from the most recently emptied breast. Collected samples were stored in the home freezer until the week 2 home visit. Participants were informed they could collect smaller amounts of milk or forgo milk collection entirely if there were concerns about infant weight loss, satiety, or health.
During the week 2 home visit, a silicone milk collector (Haakaa Ladybug, Auckland, NZ) was placed on the opposite breast to passively collect milk during breastfeeding. The quantity of milk collected was measured and up to 10 mL of milk was retained for the study. At the end of the home visit, all milk samples (1 fresh, 5 frozen) were transported on ice from the participant’s home to the laboratory. Both fresh and frozen milk was stored at −80°C until Na and K analyses were performed as follows.
Immediately before measurement, whole frozen milk samples were gradually thawed to room temperature, gently mixed using an electronic shaker, and inverted three times. Portable ion selective electrodes (Horiba LAQUATwin Pocket Meters, Irvine, CA) were used to measure Na and K ions using methods described by Lai et al. 8 and Esquerra-Zwiers et al., 9 and as detailed in Box 1. All samples were tested and read in duplicate and results were entered into a spreadsheet for analysis. Mean [M] coefficients of variation were 1.90% for Na and 1.51% for K, which are within the acceptable range of < 10%. Finally, the mean values in ppm were converted to millimoles (mmol/L) using the molecular weights of 23 g/mol for Na and 39 g/mol for K. Na/K ratios were calculated by dividing mmol/L of Na by mmol/L of K, as described by Filteau et al. 19 A variety of thresholds for Na (range = 10 to 20 mmol/L)11,13,14,20,21 and the Na/K ratio (range = 0.6 to 1.0)8,10,11,13,14,21 in milk have been proposed for indicating milk maturity and prediction of future, healthy mammary gland function. The current study identified the earliest day in the available data that Na fell below 12 mmol/L and the Na/K ratio fell below 0.6 to indicate SA achievement, given the healthy, term sample of exclusively breastfeeding dyads.
Box 1. Ion Selective Electrode Analysis of Sodium (Na) and Potassium (K) Ions in Whole Milk
Infant weight loss/gain
Medical records from the week 1 infant outpatient visit were reviewed for infant weight loss/gain. During the week 2 home visit, the study team completed a lactation history and weighed the infant clothed only in a clean diaper using a digital scale that weighed to the nearest 2 g (Tanita BD-815U, Arlington Heights, IL, USA) tared with an identical clean diaper. This weight was used to calculate weight change since birth.
Milk intake during breastfeeding
Medical records from the week 1 infant outpatient visit were also reviewed for infant milk intake using test weights. During the week 2 home visit, a complete breastfeeding session was observed, and infant milk intake was measured using test-weights via the aforementioned scale. Infant milk intake was calculated by subtracting the postfeed weight from the prefeed weight, using a 1 g weight change to indicate 1 mL of milk intake. 22
Breastfeeding duration and exclusivity
Follow-up phone calls were made to the participant at 1, 2, and 3 months postpartum to evaluate infant feeding (i.e., exclusive breastfeeding, partial breastfeeding, or exclusive formula feeding). Participants were asked, “Which of the following things are you feeding your baby at this time?” Answer options included “milk from the breast, expressed or pumped milk, formula, water, or other.”
Statistical analysis
All data were recorded in REDCap and analyses were conducted in SPSS version 29. This pilot study was designed to inform future studies with larger samples sizes and thus was not powered for inferential statistics. Compliance with the study protocol was also evaluated as an indicator of the feasibility of study methods. Descriptive statistics were used for participant demographics and key study measures. Spearman correlations were used to test for rank-order associations between the indicators of SA. Cohen’s kappa was used to evaluate SA indicator agreement. Individual trajectories of Na and Na/K ratios during the first 2 weeks postpartum were plotted and examined for patterns. Exploratory analyses were conducted to examine associations between the measures of SA with infant weight loss/gain, infant milk intake, and lactation outcomes, including the duration and exclusivity of breastfeeding. One-way ANOVA, independent sample t-tests, Spearman correlations, or chi-square tests were used for these analyses. Given the small size of this pilot data set, findings with p values < 0.10 are reported in the text, but full results are provided in Supplementary Tables S1 and S2.
Results
Participant demographics
A total of 12 mother–infant dyads were enrolled. Two infants from this group were admitted to the NICU following birth. Of these, one infant was in the NICU for the first full week of life, thus this dyad was excluded from further analyses. The other infant was admitted for respiratory distress related to meconium-stained fluid and was discharged in stable condition on day 4 of life. Participant demographics for the remaining 11 mother–infant dyads are presented in Table 1. Of these 11 dyads, the majority of participants were primiparous (64%), identified as a person of color (55%), and had a prepregnancy body mass index (weight in kilograms/[height in meters] 2 ) in the normal range (median [Mdn] = 22.4, range = 19.2 to 24.9). Participants also had strong intentions to breastfeed with scores near the high end of the IFI scale (Mdn = 16.0, range = 13.0–16.0). Most participants experienced spontaneous vaginal birth (73%), but five mothers (46%) experienced a postpartum hemorrhage with a quantified blood loss >1,000 mL. Infants were generally healthy with a median gestational age of 39.9 weeks (range = 38.3 to 41.1), median birth weight of 3,230 grams (range = 2,755 to 4,630), and median infant weight loss of 4.2% before hospital discharge (range = −7.4% to −1.0%).
Descriptive Statistics for Demographics, Birth, and Lactation Outcomes Including Earliest Observed Day of Secretory Activation, Infant Weight Gain, Milk Intake, and Lactation Exclusivity at 3 Months
Findings are presented for the full sample and for sodium (Na) and sodium–potassium (Na/K) ratio secretory activation (SA) pattern subgroups.
N = number, Mdn = median.
*p < 0.05, bold values indicate a significant difference between groups.
N = 1.
Earliest observed day of SA by maternal self-report, Na, and Na/K ratio are based upon the available information and may not represent the absolute first day of SA if data collection began after that point for a given participant.
If the earliest observed day of SA occurred after day 6 and the actual date of SA was unknown, day of SA by Na or Na/K ratio is coded as the soonest day possible based on available information.
Achievement of SA
SA using maternal self-report
Descriptive statistics and raw data for maternal self-report of SA by day postpartum are presented in Table 2 and Supplementary Table S3. All mothers provided at least 5 consecutive days of self-report data, with 45% initiating their reporting on postpartum days 2 and 64% initiating reporting on day 3. The remaining 4 participants began reporting on postpartum days 4 or 5. All mothers achieved SA as indicated by maternal self-report of breast fullness ≥ 3 within the first 6 postpartum days, with a median of postpartum day 3 (range = 2–6). Out of the 7 mothers who self-reported breast fullness within the first 3 days, 100% achieved SA as indicated by self-report within 72 hours postpartum.
Indicators of Secretory Activation (Maternal Self-Report of Breast Fullness, Sodium and Potassium, and the Sodium–Potassium Ratio) by Day Postpartum
N = number, Mdn = median.
SA using MOM biomarkers
Descriptive statistics and raw data for Na, K, and the Na/K ratio are presented in Table 2 and Supplementary Table S3. At least five consecutive daily milk samples were provided by 91% of participants (N = 10), with 45% initiating sample collection on day 2 and 82% initiating sample collection by day 3. Two participants provided initial milk samples on days 4 and 6 due to maternal or infant complications. Three mothers did not achieve SA by either Na or Na/K despite providing milk samples through at least the first 6 days postpartum. For these mothers, the day of SA achievement by MOM biomarker was coded as the earliest possible day for the specific individual based on available information, either postpartum day 7 or 8.
Nine mothers (82%) achieved SA as indicated by Na concentration < 12 mmol/L within the first 6 postpartum days, with a median of postpartum day 4 (range = 2–8). Eight mothers (72%) achieved SA as indicated by the Na/K ratio < 0.6 within the first 6 postpartum days, with a median of postpartum day 3 (range = 2–8). Two mothers did not achieve SA by either MOM biomarker within the first 6 days postpartum.
Out of the nine mothers who provided milk samples on the first 3 days, four mothers (44%) did not achieve SA as indicated by Na within 72 hours postpartum. Similarly, three mothers (33%) did not achieve SA as indicated by Na/K within 72 hours postpartum.
Comparison of SA indicators
There were no associations between the day of SA achievement by maternal self-report and Na (r = 0.12, p = 0.73) or Na/K ratio (r = 0.10, p = 0.76). However, when comparing Na and the Na/K ratio, the days of SA achievement were highly correlated (r = 0.95, p < 0.001) (Fig. 1). The Cohen’s kappa between the day of SA achievement by maternal self-report and MOM biomarkers demonstrated lack of agreement (Cohen’s κ = 0.05 and 0.08, p = 0.72 and 0.62), whereas the day of SA achievement by MOM biomarkers showed strong agreement (Cohen’s κ = 0.87, p < 0.001).
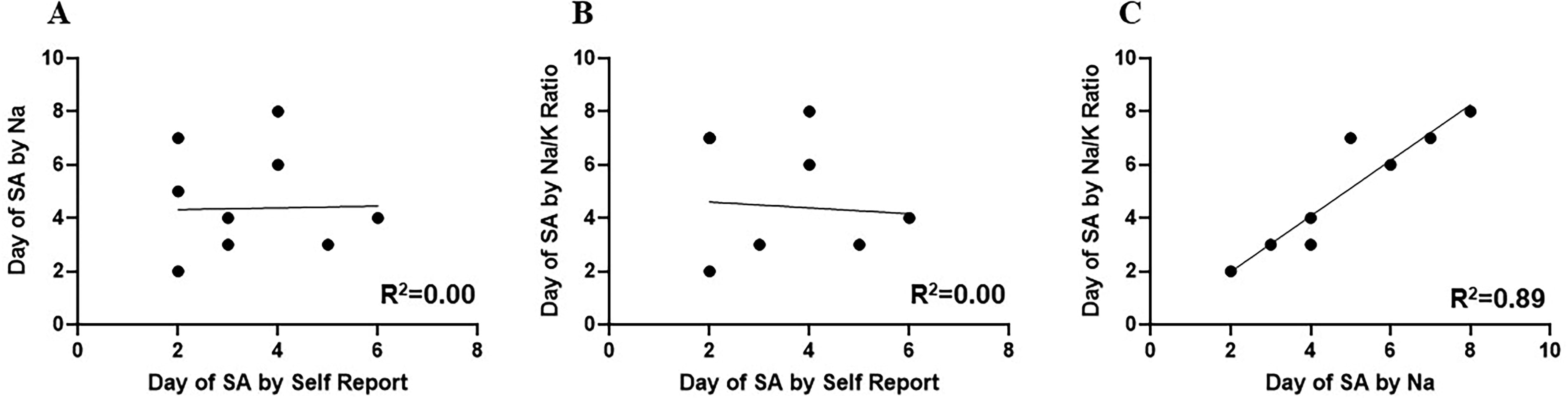
Scatterplots with best fit line comparing earliest day of secretory activation (SA) by maternal self-report of breast fullness ≥ 3, mother’s own milk (MOM) sodium (Na) level < 12 mmol/L, and MOM sodium–potassium (Na/K) ratio < 0.6:
Multiparas tended to self-report SA earlier than primiparas (M = 2.5 versus 3.9 days, p = 0.09). Participants experiencing cesarean birth (M = 5.0 versus 2.8 days, t = 4.25, p = 0.002) or postpartum hemorrhage (M = 4.2 days versus 2.7 days, t = 2.39, p = 0.04) achieved SA by maternal self-report later than those with uncomplicated births. Formula supplementation in the hospital was also associated with later maternal self-report of SA achievement (M = 4.8 versus 2.6 days, t = 4.94, p < 0.001). In contrast, none of these factors was associated with the day of SA achievement using MOM biomarkers.
Later SA achievement using Na as an indicator was associated with greater neonatal weight loss at the week 1 infant outpatient visit (r = −0.70, p = 0.02). Similarly, later SA achievement using the Na/K ratio trended toward an association with greater neonatal weight loss (r = −0.59, p = 0.06) and less infant milk intake during the week 1 infant outpatient visit (r = −0.59, p = 0.08). There were no associations between day of SA by maternal self-report and any infant weight or lactation outcomes. Of note, none of our participants reported overt signs of mastitis. There was a single retrospective report of plugged ducts at week 2.
Patterns of SA by MOM biomarker
Three trajectories of Na concentration change by postpartum day are plotted in Figure 2. For seven mothers (64%), Na concentration declined to < 12 mmol/L during the first 7 postpartum days and remained under this threshold once achieved, demonstrating a mature and stable SA pattern. Two participants (18%) showed an impermanent SA pattern where the Na concentration declined to < 12 mmol/L at least once during the first postpartum week, but did not remain consistently under this threshold. Finally, two participants (18%) displayed a delayed SA pattern, where Na remained consistently >12 mmol/L throughout the first postpartum week. Of note, there were no associations between these patterns and infant or lactation outcomes. Since Na < 12 mmol/L is a conservative threshold, we next examined whether the four mothers with impermanent or delayed SA patterns would be reclassified using a more generous SA achievement threshold of Na < 16 mmol/L. 14 Results revealed that both mothers with delayed patterns would be reclassified as impermanent. Of the two mothers classified as impermanent, one of the two would remain impermanent, while the other would be reclassified as mature at a threshold of Na < 16 mmol/L. At this threshold, there were still no associations between these patterns and infant or lactation outcomes.
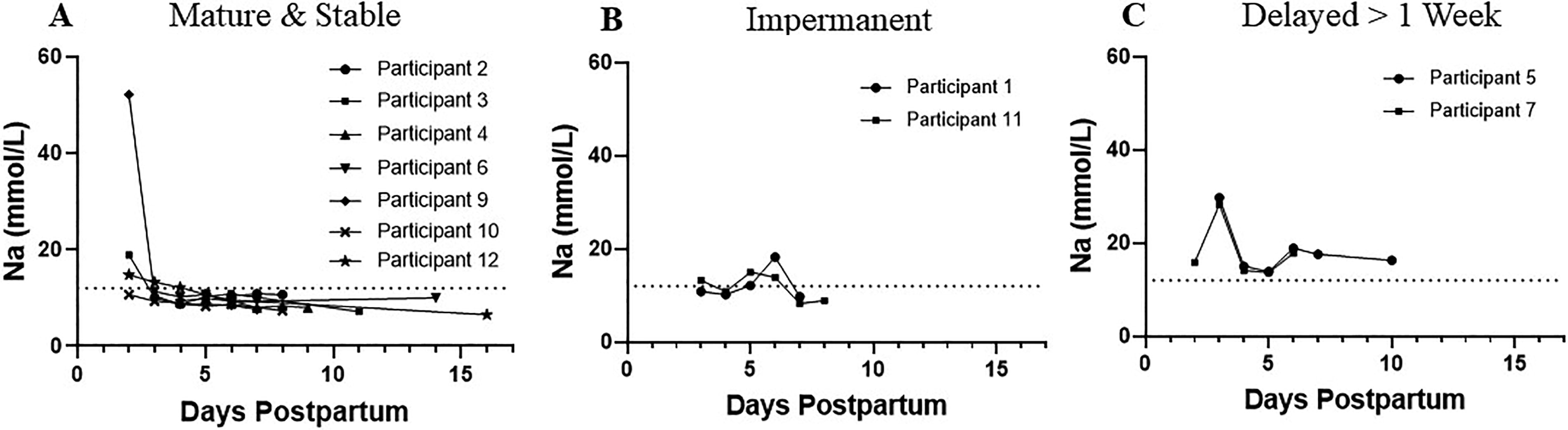
Three patterns of secretory activation (SA) as indicated by mother’s own milk (MOM) sodium (Na):
Three similar trajectories are depicted for the Na/K ratio in Figure 3. Although the same three patterns of SA achievement were noted, the frequencies differed. Four mothers (36%) demonstrated a mature and stable SA pattern, four mothers (36%) demonstrated an impermanent SA pattern, and three mothers (27%) demonstrated a delayed SA pattern. Significantly less weight loss (M = −0.2%) between birth and the week 1 infant outpatient visit was noted for infants from dyads with a mature and stable SA pattern compared with infants from dyads with an impermanent (M = −5.9%) or delayed SA pattern (M = −6.7%), F = 4.82, p = 0.04.
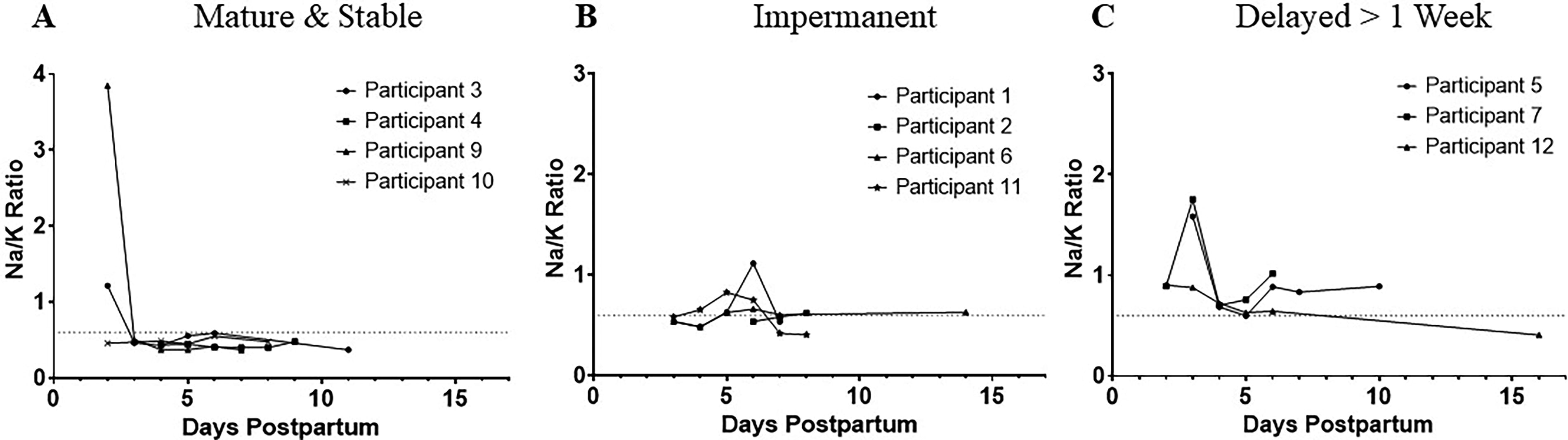
Three patterns of secretory activation (SA) as indicated by mother’s own milk (MOM) sodium–potassium (Na/K) ratio:
Since an Na/K ratio < 0.6 is a conservative threshold, we next examined whether the seven mothers with an impermanent or delayed SA would be reclassified using a more generous SA achievement threshold of Na/K ratio < 0.8. 14 Results revealed that two of the three mothers with a delayed pattern would be reclassified to impermanent, while one would be reclassified to mature. Of the four mothers classified as impermanent, two would remain impermanent, while two would be reclassified to mature at an Na/K ratio < 0.8. At this threshold, there were no associations between these patterns and infant or lactation outcomes.
Discussion
This pilot study is the first to demonstrate the feasibility of in-home, daily milk sample collection for the evaluation of biomarkers of SA achievement during the first postpartum week in healthy, term mother–infant dyads using commercially available portable ion selective electrodes (ISEs).8,9 We found better than expected participant compliance with study procedures, with the majority of participants beginning sample collection within 2–3 days postpartum and collecting at least 5 days of milk samples during the first postpartum week. We wish to acknowledge the efforts made by our participants during a week that is challenging for most postpartum patients. Although not presented here, we also piloted in-home testing for Na and K using portable ISEs and successfully measured Na concentrations and Na/K ratios using fresh whole milk in six participant homes. Our findings support those of other researchers demonstrating the feasibility of using point-of-care measures of breastfeeding in the home, such as test-weighing for measurement of infant milk intake. 22
This pilot study also highlights the potential importance of serial measures of MOM biomarkers compared with a single “spot” measure. While other studies have found a single measurement of Na or the Na/K ratio on day 7 or 10 to be predictive of early supplementation or weaning,10,12 it is possible that a single sample could miss concerning SA patterns depending on the day of sampling. Variability and impermanence in serial MOM biomarkers of SA achievement have been reported among breast pump-dependent mothers of preterm infants in the NICU.14,15,21 While the significance of these patterns in healthy, term dyads is unclear, this work informs the importance of serial versus single-day MOM biomarker collection in subsequent research.
Maternal self-report versus biomarker measurement of SA achievement
Maternal perception of milk coming in is the most accessible measure of SA achievement in research 13 and is frequently used in community settings. However, Medina-Poeliniz et al. 13 recently emphasized the need for validation of maternal perception of milk coming in using MOM biomarkers as the “gold standard” for comparison. A recent review confirms this point, highlighting that maternal perception of SA achievement does not concur with MOM biomarker data in pump-dependent mothers of preterm infants in the NICU. 21
In the current study, we compared daily self-report of breast fullness with daily measures of Na and Na/K ratio during the first postpartum week. We found that Cohen’s kappa between SA by maternal self-report and MOM biomarkers demonstrated lack of agreement, and that on average, SA achievement by maternal self-report occurred approximately a day earlier than for MOM biomarkers. While it is possible that a different clinical indicator for maternal self-report of SA achievement could have identified a stronger relationship with MOM biomarkers, the lack of concordance remains potentially problematic. Misperception of milk coming in could potentially result in delayed intervention for at-risk dyads. For example, two mothers in the current study achieved SA on day 7 or 8 postpartum using MOM biomarkers. However, both self-reported earlier SA achievement on days 2 or 4 and both infants tended toward greater infant weight loss in the first week postpartum (Mdn = −7.7%). Both dyads might have benefited from earlier identification and directed lactation care.
Of note, day of SA achievement using Na and Na/K ratio was highly correlated in the current study. Similar to other studies, this suggests that Na concentration alone might be used as a single MOM biomarker of SA achievement for routine screening.13,14,21 This finding is especially important for community settings because measurement of Na concentration alone is far easier to perform than is the Na/K ratio. Additional research is needed in larger and more diverse populations of women to determine which MOM biomarkers are the most predictive of lactation problems.
Although our sample was limited, we conducted exploratory analyses to examine associations between each of the indicators of SA achievement with inpatient experiences, infant weight, and lactation outcomes. Compared with uncomplicated inpatient experiences, cesarean birth, postpartum hemorrhage, and inpatient formula supplementation were each associated with delayed SA achievement by maternal self-report. This relationship was not present for SA achievement using MOM biomarkers. It is possible that awareness of lactation risks related to these inpatient experiences may have influenced mothers’ perceptions of SA achievement timing, especially considering that MOM biomarkers did not support this finding.
In contrast, delayed SA achievement by Na concentration was significantly correlated with greater infant weight loss during week 1 postpartum. A non-significant trend toward an association between delayed SA achievement using Na/K ratio and greater infant weight loss and less milk intake at the week 1 outpatient visit was also noted. These data add to the accumulating literature that suggests perception of SA achievement may be insufficiently accurate to enable timely and personalized lactation interventions in some populations.13,21
Patterns of SA achievement and clinical and lactation outcomes
Upon examining trends of decrease in Na concentration and Na/K ratio over the first postpartum week, we identified three distinct patterns of SA achievement in these otherwise healthy, term mothers: (1) mature and stable, (2) impermanent, and (3) delayed >1 week. Our small pilot study limits actionable recommendations based on associations between patterns of SA achievement and infant and lactation outcomes. In addition, minor deviations in the day of the postpartum week 1 and week 2 visits could have influenced infant weight outcomes. However, infants of mothers in the mature subset by Na/K ratio were generally thriving and had mostly regained or surpassed birth weight during the first postpartum week. In contrast, infants in both the impermanent and delayed SA subsets by Na/K ratio remained approximately 6% below their birth weight. Despite these early differences, 91% of mothers, including the four mothers with either impermanent or delayed trajectories, were exclusively breastfeeding at 3 months postpartum. All participants in this pilot study had very strong intentions to breastfeed, and it is likely that this internal motivation impacted maternal response to any early lactation challenges. The week 1 and 2 postpartum follow-up visits in this study may have also provided lactation support that enabled these longer term breastfeeding outcomes.
Strengths and limitations
This pilot study had several strengths, including the daily measurement of MOM biomarkers of SA achievement in a community setting. However, there were some limitations. Three different infant weight scales were used among settings (i.e., mother–baby unit, pediatric clinic, home visit), which could potentially affect the accuracy of infant weight measures. However, this is a common reality in community settings and all scales used in this study were electronic, minimizing measurement error. Mothers were not instructed to collect milk from both breasts or to specify which breast was represented by the individual milk sample. It is theoretically possible that interbreast differences in milk maturation may exist and may have played a part in the impermanent SA achievement pattern. This limitation should be addressed in subsequent research. Finally, the small sample size for this pilot study meant that it was underpowered for hypothesis testing. However, the intensive data collection protocol has generated more detailed research questions and has demonstrated the feasibility of integrating our methods into subsequent larger studies with more diverse community participants.
Conclusions
In conclusion, this pilot study demonstrates the feasibility of collecting daily milk samples in community settings for the testing of MOM biomarkers of SA achievement during early lactation, as a strategy to mitigate early, unplanned cessation of lactation. Given the limitations of maternal self-report of SA achievement, MOM biomarkers hold potential to more accurately assist in the screening and management of otherwise healthy, term breastfeeding dyads.
Footnotes
Acknowledgments
The authors thank the study participants and the clinical and research staff at the University of New Mexico Health Sciences Center.
Authors’ Contributions
K.T.K.: Conceptualization, methodology, formal analysis, investigation, resources, data curation, writing—original draft, visualization, supervision, project administration, and funding acquisition. J.L.: Investigation, data curation, and writing—review and editing. R.M.: Investigation, data curation, and writing—review and editing. S.C.: Investigation, data curation, and writing—review and editing. E.R.: Validation, investigation, data curation, and writing—review and editing.
Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Information
This project was supported, in part, by the Dedicated Health Research Funds of the University of New Mexico School of Medicine allocated to the Child Health Research Signature Program. Additional funding was provided by the National Institutes of Health (NIH) under grant numbers R01HD111200 and UL1TR001449.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
