Abstract
Background and Objectives:
As cannabis use increases among reproductive-aged women, there is a growing need to better understand the presence of cannabinoids in milk produced by women using cannabis. It is unclear how concentrations of cannabinoids such as delta-9-tetrahydrocannabinol (Δ9-THC) persist in milk after cannabis use and what factors contribute to variation in milk Δ9-THC concentrations. Our objectives were to measure cannabinoids in human milk following cannabis abstention, after single and repeated instances of cannabis use, and identify factors contributing to concentration variation.
Methods:
The Lactation and Cannabis (LAC) Study prospectively observed 20 breastfeeding participants who frequently used cannabis (≥1/week), had enrolled <6 months postpartum, were feeding their infant their milk ≥5 times/day, and were not using any illicit drugs. Participants collected a baseline milk sample after ≥12 hours of abstaining from cannabis and five milk samples at set intervals over 8–12 hours after initial cannabis use. Participants completed surveys and recorded self-directed cannabis use during the study period.
Results:
Δ9-THC peaked 120 minutes after a single instance of cannabis use (median, n = 9). More instances of cannabis use during the study period were associated with greater Δ9-THC area-under-the-curve concentrations (ρ = 0.65, p = 0.002), indicating Δ9-THC bioaccumulation in most participants. Baseline Δ9-THC logged concentration was positively associated with self-reported frequency of cannabis use (b = 0.57, p = 0.01).
Conclusions:
Cannabinoids are measurable in human milk following cannabis use, and concentrations remain elevated with repeated cannabis use over a day. Substantial variation in Δ9-THC milk concentrations reflects individual differences in characteristics and behavior, including average postpartum frequency of cannabis use.
Introduction
There has been recent and rapid expansion of cannabis legalization in the United States. During this time, cannabis use has increased, and perceptions of risk associated with use have decreased among reproductive-aged women.1,2 However, maternal cannabis use in pregnancy is associated with increased risk of preterm birth, infants being small-for-gestational-age at birth, perinatal mortality, and low birth weight, even when controlling for use of tobacco and other drugs.3,4
Less is known about how cannabis use and cannabinoids, which have been measured in the milk produced by women who use cannabis,5,6 affect human milk composition. This knowledge is critical because human milk is frequently the sole source of nutrition in early life. Importantly, many cannabinoids, including delta-9-tetrahydrocannabinol (Δ9-THC, the dominant psychoactive component of cannabis), are lipophilic, and lipid-rich human milk and mammary tissue may be cannabinoid repositories.7,8 Previous research on 54 samples donated to a human milk biorepository from donors who used cannabis postnatally detected Δ9-THC in milk up to 48 hours after last use; in one sample, it was still detected approximately 140 hours after last use. 5 Another study on seven participants with test-confirmed prenatal cannabis use estimated that Δ9-THC may be detectable in milk for >6 weeks after last use. 6 In paired milk and plasma samples from 20 breastfeeding mothers who reported cannabis use a median of 1–2 times/day via inhalation and/or via edibles or topically, median Δ9-THC concentrations were seven times higher in milk than in plasma, suggesting that Δ9-THC may bioaccumulate in the mammary gland and/or is preferentially taken up from circulation. 9 Only one study assessed the pharmacokinetics of milk Δ9-THC concentrations after a single, controlled dose of cannabis, 10 but no research has yet characterized how cannabinoid concentrations in milk change over time following self-directed or repeated maternal cannabis use.
We conducted a prospective study among breastfeeding individuals reporting frequent cannabis use to (1) characterize Δ9-THC concentrations in human milk after abstention and self-directed cannabis use (including repeated instances of use), (2) estimate total infant Δ9-THC exposure via human milk, and (3) identify possible associations between Δ9-THC concentrations in milk and selected maternal factors such as body mass index (BMI, an indicator of adiposity), rate of postpartum weight loss (an indicator of negative energy balance and adipose tissue mobilization), time postpartum, and the frequency, amount, and type of cannabis used.
Materials/Subjects and Methods
Participants and study design
Twenty breastfeeding individuals and their infants residing in Washington and Oregon, where the purchase of cannabis is legal, were enrolled and completed the Lactation and Cannabis (LAC) Study in 2022. Inclusion criteria were as follows: ≥21 years of age, have an infant <180 days old born full term (≥37 weeks gestational age) at enrollment, report using cannabis at least weekly, breastfeeding and/or providing their pumped milk to their infant ≥5 times per day, and provided pumped milk to their infant previously. Exclusion criteria included use of illicit drugs, use of opioid use disorder treatments or opioids in the past 5 years (excluding any administered/prescribed opioids relating to delivery or cesarean-sections), symptoms of illness in the last 7 days (i.e., diarrhea, vomiting, severe cough, rapid breathing, or fever) in the participant or infant, and/or indications of a breast infection (i.e., fever, red streaks or hard red portions of the breast, reported breast pain, discomfort, or lumps on the breast not typical during breastfeeding). Inclusion criteria were determined through self-report. The Washington State University Institutional Review Board approved all study procedures. A Certificate of Confidentiality was obtained from the National Institutes of Health.
In this prospective study (Fig. 1), participants were instructed to observe a 12-hour abstention from cannabis prior to milk sample collection. Following abstention, participants collected a baseline milk sample (S1). After baseline milk collection, participants administered their own cannabis at least once. They collected five additional milk samples (S2–S6) within timed intervals (30 to ≤40 minutes, 1 to <2 hours, 2 to <4 hours, 4 to <6 hours, and 8 to <12 hours) after initial cannabis use. Participants used cannabis how they normally would after abstention, potentially including multiple uses throughout the milk collection period.
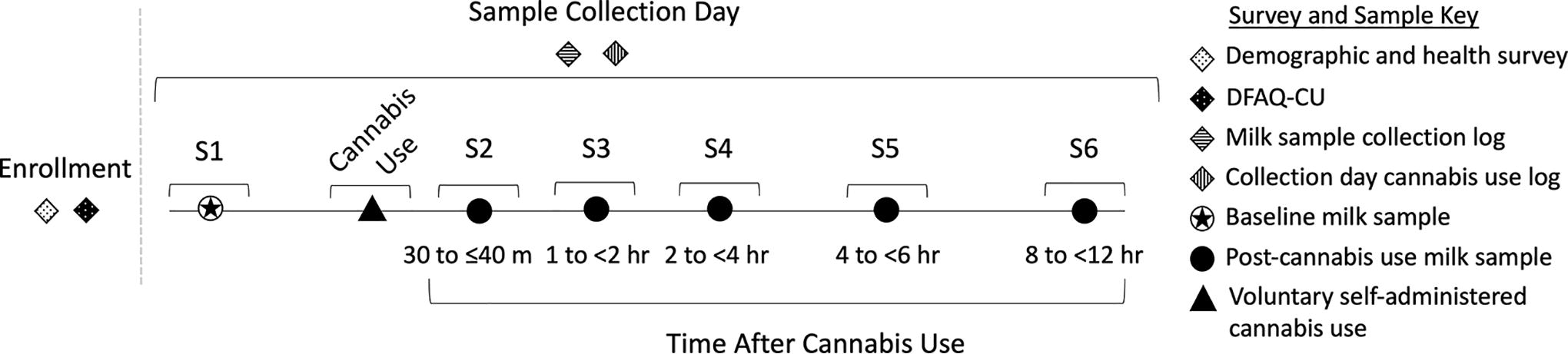
Sample and data collection strategy. Sample 1 (S1, baseline) was collected after at least 12 hours of abstention from cannabis. Cannabis was used at a time and place of the participant’s choosing and was self-administered. Samples 2–6 (S2–S6) were collected at the listed post-cannabis use intervals (30 to ≤40 minutes, 1 to <2 hours, 2 to <4 hours, 4 to <6 hours, and 8 to <12 hours). Milk collection logs were recorded by participants at each collection, and cannabis use logs were recorded from first use after baseline and throughout the day, if participants used cannabis more than once.
Metadata, surveys, and cannabis use data collection
Following enrollment, participants completed online surveys using REDCap 11 that assessed demographics, health, prenatal and postpartum substance use, and cannabis use (Daily Sessions, Frequency, Age of Onset, and Quantity of Cannabis Use Inventory [DFAQ-CU] 12 ). On the collection day, participants self-recorded cannabis abstention and use (see Supplementary Data S1 for metadata collection details).
Milk sample collection and quantification of cannabinoids
Participants collected full breast expressions into collection bottles and aliquoted up to 15 mL of milk for analysis. For cannabinoid analysis, sample preparation was carried out according to Britch et al. 13 with minor modifications described in Supplementary Data S1. Milk samples were analyzed using an Acquity liquid chromatography system and a Synapt G2-S mass spectrometer. Milk (390 µL) was spiked with 10 µL of a solution containing 400 ppb each of the deuterated standards (THC-d3, 11-OH-THC-d3, COOH-THC-d3, CBN-d3, CBD-d3, 7-OH-CBD-d3, 7-COOH-CBD-d3; Cerilliant, Round Rock, TX, USA). Acetonitrile (ACN, 800 µL) was used for protein precipitation, and 1.3 mL of 1% ammonium hydroxide was used for pH adjustment prior to loading on Oasis MAX 3cc cartridges (Waters, Ireland). Cartridges were washed with 0.75 mL of 35% ACN. In the final step, cannabinoids were eluted with 2.0 mL of the eluent solvent. 13
Analysis was run on three randomly assigned batches, and standard curves were generated for each batch. Quantitation was based on an internal standard that corrects for changes in instrument performance and analyte losses during sample preparation, to correct for potential batch effects. Data were analyzed by the TargetLynx module of MassLynx v. 1.4 (Waters, Milford, MA, USA). Levels of detection (ng/mL) were determined to be as follows: THC-d3 = 0.58, CBD-d3 = 0.43, COOH-THC-d3 = 0.60, CBN-d3 = 0.65, 11-OH-THC-d3 = 0.69, 7-OH-CBD-d3 = 1.30, and 7-COOH-CBD-d3 = 1.5. The level of quantification was 1.00 ng/mL for all compounds, except 7-OH-CBD-d3 and 7-COOH-CBD-d3, which were 2.00 ng/mL.
Details on sample collection, storage, and analysis are included in Supplementary Data S1.
Statistical analyses
Statistical analyses were conducted in R 4.2.2. 14 Statistical approaches including t-tests, Spearman correlations, and regression models are detailed in Supplementary Data S1. Significance was declared at p < 0.05. Given the limited data on cannabinoids in milk and the exploratory nature of the analyses, results of p < 0.1 are noted but not considered significant. For analyses requiring samples to be collected within a specific time frame, samples were within acceptable time ranges if they were collected 30 to ≤40 minutes, 1 to <2 hours, 2 to <4 hours, 4 to <6 hours, and 8 to <12 hours after initial cannabis use (Fig. 1). In all other analyses, time was treated continuously including all measurable samples.
Results
Participant and sample characteristics
Mean participant age and time postpartum at sample collection were 26.6 (SD = 3.5) years and 106.8 (50.9) days, respectively (Table 1). Five participants did not report valid (≤13 days before sample collection day) postpartum weight; consequently, their postpartum weight loss rate and BMI were not calculated. Most (65%) participants reported using cannabis on average at least once daily. On the study day, 12 of the 20 participants used cannabis more than once. Eighteen participants smoked cannabis at least once, two participants only vaped, and seven participants both smoked and vaped during the study day. Participants provided a total of 114 milk samples (Supplementary Fig. S1).
Summary of Demographics, Anthropometrics, Cannabis Use Patterns, and Milk Δ9-THC Concentrations for the Women and Infants (N = 20)
Participant specified “other” as “concentrates.”
One sample was below the level of quantification, so the midpoint between the level of quantification and level of detection (0.79) was imputed for this sample.
Study day estimates of THC intake are based on maternal subjective interpretation of amount of cannabis smoked/vaped as well as product packaging's THC concentration information. tetrahydrocannabinol. THC concentration information as provided by the manufacturer.
BMI, body mass index; SD, standard deviation; Δ9-THC, delta-9-tetrahydrocannabinol.
Milk Δ9-THC concentrations
Mean baseline milk Δ9-THC concentration after the 12-hour cannabis abstention was 36.5 (48.0) ng/mL (Table 1, Fig. 2). Mean concentration after initial cannabis use (post-cannabis use) was 104.4 (126.3) ng/mL. Participants’ average Δ9-THC concentration in milk after cannabis use ranged from 2.9 to 312.6 ng/mL (Fig. 2). Supplementary Table S1 provides descriptive statistics for 11-nor-9-carboxy-Δ9-tetrahydrocannabinol, 11-hydroxy-Δ9-tetrahydrocannabinol, cannabinol, 7-hydroxy cannabidiol, cannabidiol, and 7-carboxy-cannabidiol for baseline and post-use samples.
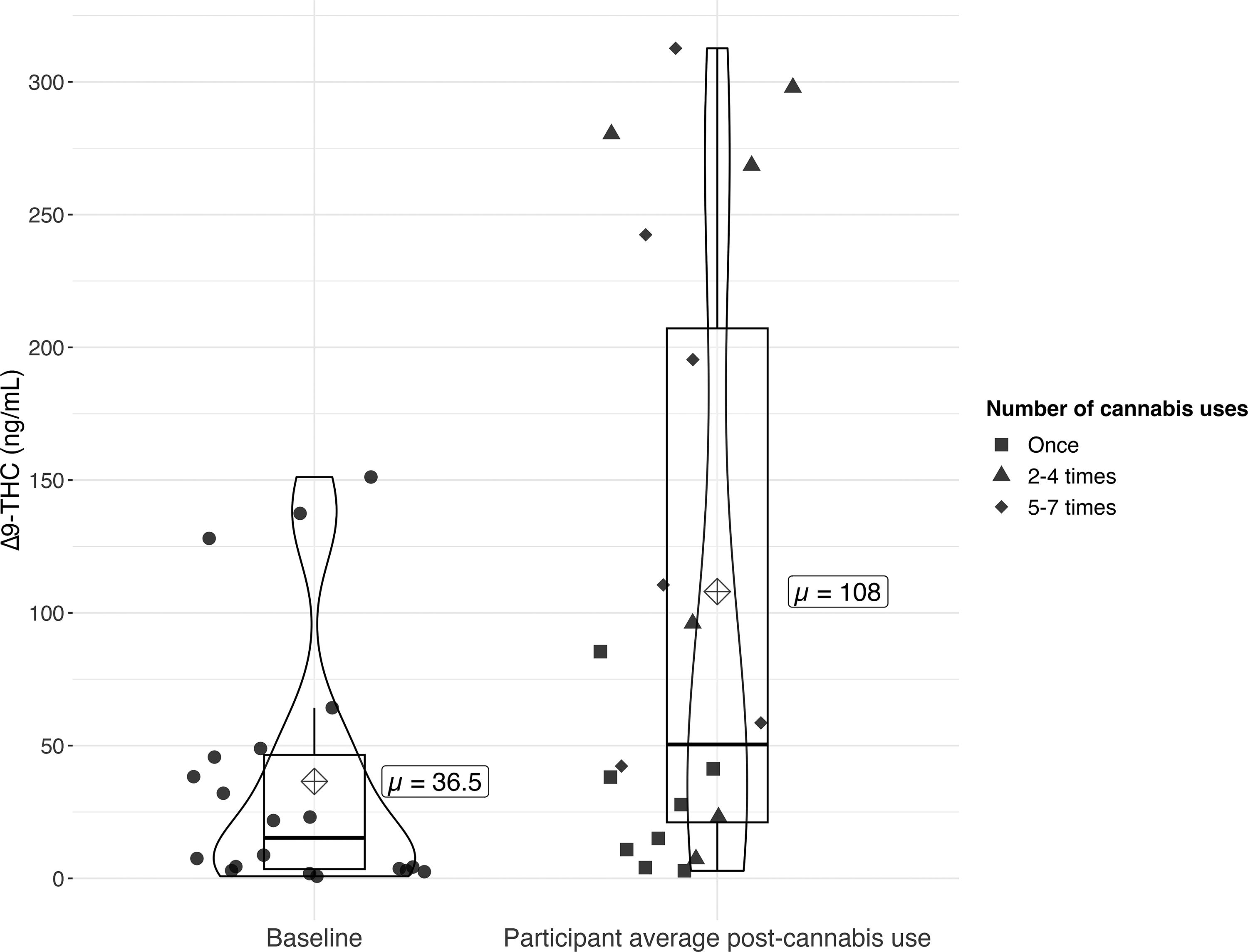
Δ9-THC concentrations in milk at baseline (S1, n = 20) and after cannabis use (mean of S2–S6, n = 94). Circle points represent Δ9-THC concentrations in milk in baseline samples (collected at least 12 hours following last cannabis use). Shape points (square, triangle, and diamond) represent participant average Δ9-THC concentration in milk following cannabis use (average of samples S2–S6). Point shape is determined by the number of times the participant used cannabis from the first cannabis use to the end of the study period. Horizontal line indicates median; box hinges indicate 25th and 75th percentiles; whiskers indicate 1.5 * interquartile range; dots are individual participant datapoints. Δ9-THC, delta-9-tetrahydrocannabinol.
Associations between baseline milk Δ9-THC concentrations and maternal characteristics
In linear regression models controlling for time since last cannabis use, natural-logged baseline Δ9-THC concentrations in milk were positively predicted by frequency of cannabis use (b = 0.57, p = 0.01; Supplementary Table S2). Baseline Δ9-THC concentrations and postpartum weight loss rate were not significantly correlated (ρ = –0.24, p = 0.40; Supplementary Fig. S2). In addition, when controlling for time since last cannabis use, baseline Δ9-THC concentrations were not significantly predicted by postpartum weight loss rate (b = –6.09, p = 0.06). Baseline Δ9-THC concentrations were not significantly predicted by maternal BMI, time postpartum, total lifetime length of cannabis use, or time between last breastfeed and baseline milk sample collection when controlling for time since last cannabis use. Mean baseline Δ9-THC concentration did not differ by the primary form of cannabis use [marijuana/leaf/flower = 22.4 (22.6) ng/mL; concentrates = 57.6 (67.9) ng/mL; Welch t-test p = 0.19] or by the primary method of cannabis use [n = 15; bong = 55.8 (54.6) ng/mL; joints = 15.9 (24.0) ng/mL; t-test equal variances assumed p = 0.10].
Estimation of Δ9-THC half-life in milk
In regression models of natural-logged baseline Δ9-THC, each hour since last cannabis use was associated with a 0.05876 ng/mL (unlogged 6.05%) decrease in baseline Δ9-THC concentrations (Supplementary Table S2). Using the unlogged estimate as the decay constant, the half-life of Δ9-THC in milk was estimated to be 11.45 hours.
Milk Δ9-THC concentrations after a single instance of cannabis use
The timing of peak Δ9-THC concentration in milk following single cannabis use was notably variable across the nine participants in this subset (x̄ = 96.4 minutes; range = 30–135 minutes; Fig. 3, Supplementary Table S3, Supplementary Fig. S3). Nonetheless, most participants (n = 5) in this subset had their highest Δ9-THC concentration in the milk sample collected 120–135 minutes after cannabis use. Mean Δ9-THC peak concentration was 56.9 ng/mL (range = 4.2–129.6 ng/mL). There was no uniform pattern for changes in milk Δ9-THC concentration after a single use of cannabis: in some participants, it peaked 30–35 minutes after use; in others, 85–93 minutes after use; and in the rest, 120–135 minutes after use (Supplementary Table S3, Supplementary Data S1, Supplementary Figs. S3 and S4).
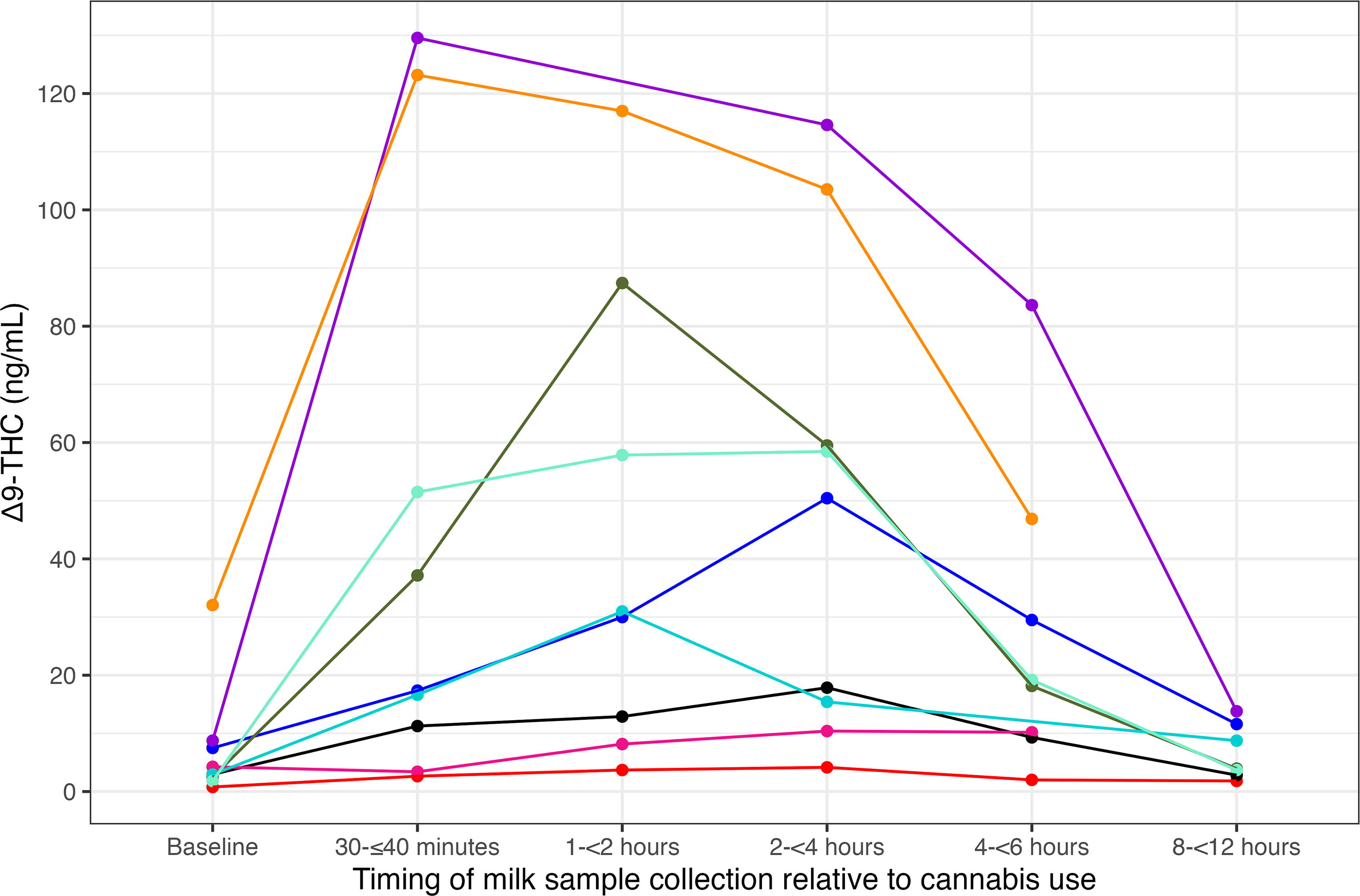
Δ9-THC concentration in milk samples among those participants who only used cannabis once on the study day or prior to collection of sample 6 (S6) (n = 9). Each color represents a different participant. One participant’s sample 5 is not plotted in this figure, as it was collected outside the acceptable time window (7.8 hours), but was retained in analyses. Two participants’ sixth samples were excluded from this single-use cannabis analysis, as they used cannabis again between their fifth and sixth milk sample collections. One participant who used cannabis once during the study was excluded from this analysis, as they did not have the required number of post-use samples to evaluate changes in Δ9-THC concentrations after single use. Δ9-THC, delta-9-tetrahydrocannabinol.
Repeated cannabis use and Δ9-THC concentrations in milk
Participants who used cannabis multiple times across the period of milk sample collection (n = 12) displayed different patterns of Δ9-THC concentrations (Fig. 4) from those who used cannabis once. In contrast to the clear rise and fall of Δ9-THC concentration after a single use of cannabis, most participants (n = 7, 58%) who used cannabis multiple times had higher Δ9-THC concentrations in their last milk sample than in the sample collected ∼30 minutes after first cannabis use. Generally, repeated cannabis use resulted in elevated milk Δ9-THC concentrations across the 8–12 hours of sample collection.
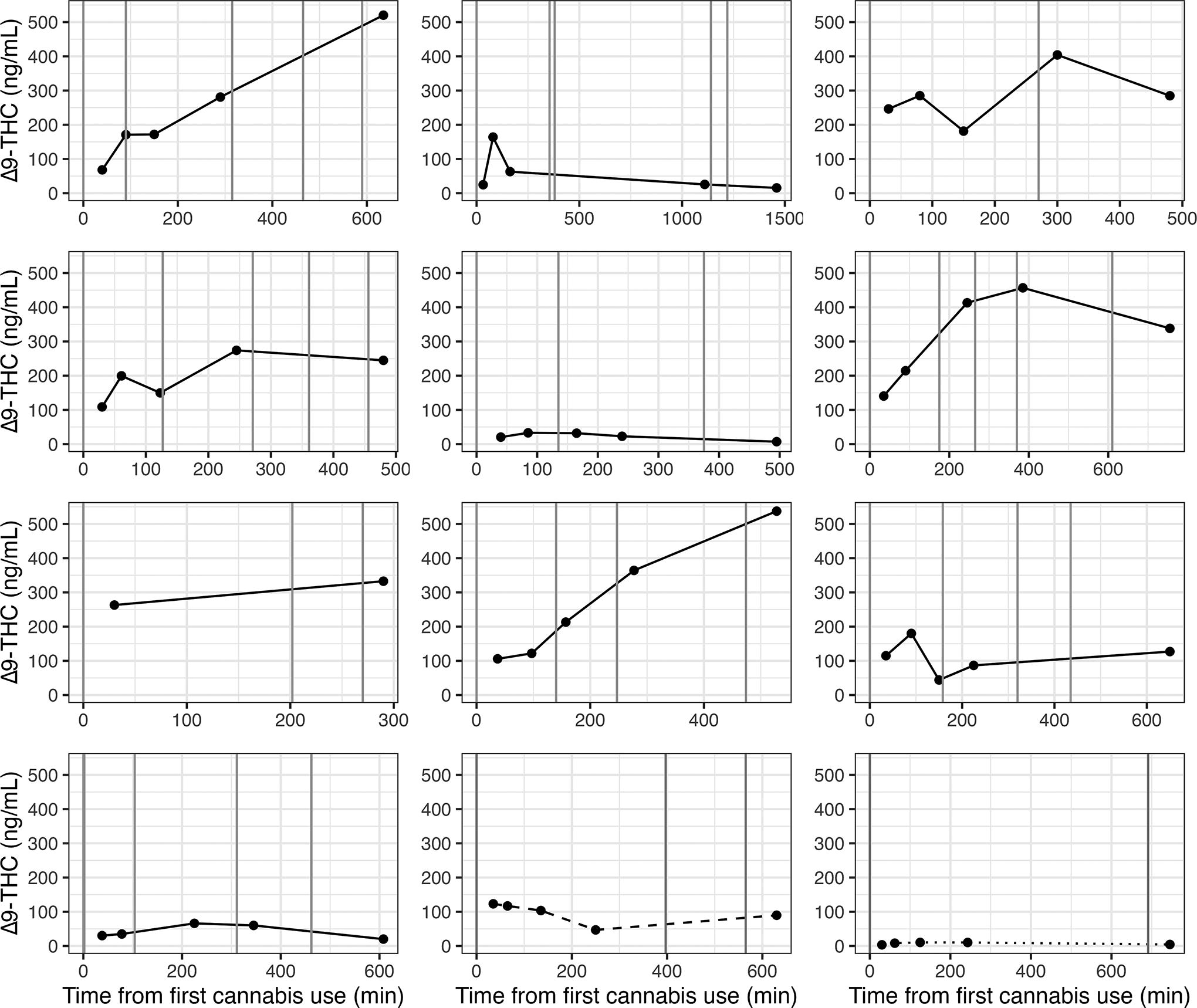
Δ9-THC concentrations in milk (ng/mL) after first cannabis use among participants who used cannabis more than once (n = 12). Each panel represents a different participant. Vertical lines indicate cannabis use times. Dashed and dotted lines in the last two boxes are participants whose samples, prior to second/third cannabis use, were included in Figure 3 and associated analysis. Dashed line corresponds to the yellow line, and dotted line corresponds to the pink line in Figure 3. Δ9-THC, delta-9-tetrahydrocannabinol.
Average milk Δ9-THC concentration after cannabis use and milk Δ9-THC AUC (area-under-the-curve, a measure of overall Δ9-THC concentration accounting for unobserved Δ9-THC concentrations between samples) were positively correlated with the total number of cannabis use instances on the study day (ρ = 0.62; p = 0.003 and ρ = 0.65; p = 0.002, respectively) and the total amount (g) smoked (ρ = 0.60; p = 0.01 and ρ = 0.52; p = 0.03, respectively) but not with total estimated THC inhaled or total number of vape hits during the study day (Supplementary Fig. S5).
Estimated per feed and daily infant Δ9-THC intake
Estimated mean infant Δ9-THC intake per feed across all milk samples was 0.0095 mg (range = 0.0001–0.0572 mg). The lowest mean estimate of Δ9-THC intake per feed occurred in the baseline milk samples (0.0037 mg). Each infant’s estimated average Δ9-THC intake per feed ranged from 0.0003 to 0.0280 mg. When scaled by body weight, each infant’s average Δ9-THC intake per feed ranged from 0.00004 to 0.0072 mg/kg (median = 0.0009 mg/kg, x̄ = 0.0017 mg/kg, Fig. 5A).
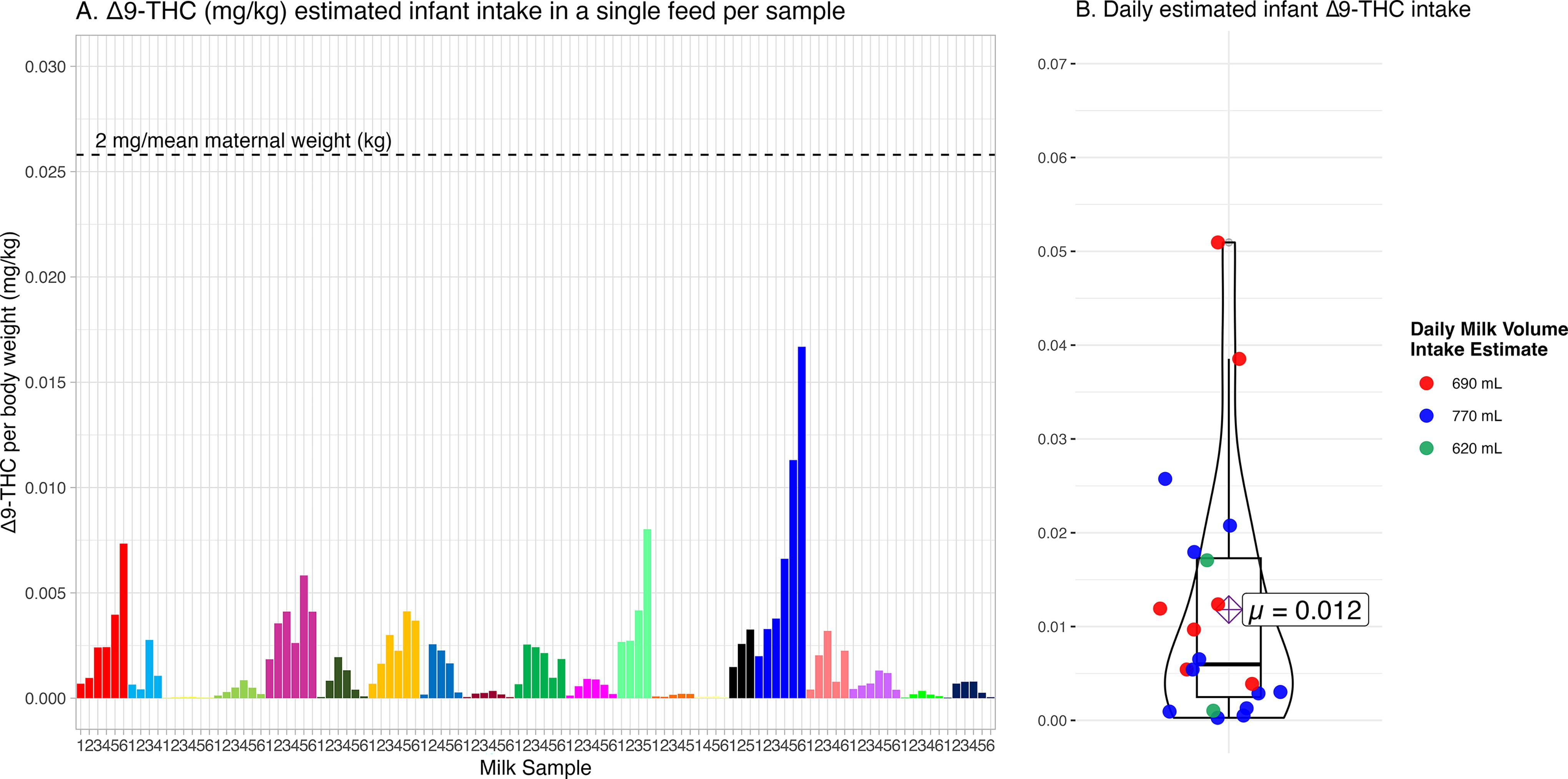
Estimated infant Δ9-THC intake based on milk Δ9-THC concentrations scaled to infant weight.
Estimated mean daily infant Δ9-THC intake was 0.0667 mg (range = 0.0006–0.4006 mg). Each infant’s estimated average daily Δ9-THC intake ranged from 0.0019 to 0.1963 mg/day (x̄ = 0.0677 mg). When scaled by their body weight, each infant’s estimated average daily intake ranged from 0.0003 to 0.0509 mg/kg (median = 0.0060 mg/kg, x̄ = 0.0118 mg/kg, Fig. 5B).
Based on Baker et al.’s 10 calculation assuming 150 mL/kg/day milk consumption, the estimated median infant Δ9-THC dose across all samples was 5.7 μg/kg/day (0.0057 mg/kg/day), and the mean of within-individual average infant Δ9-THC dose after maternal cannabis use was 16.2 μg/kg/day (0.0162 mg/kg/day).
Discussion
In this prospective study of self-directed cannabis use among breastfeeding individuals, more frequent cannabis use throughout the day was associated with higher, and increasing, Δ9-THC concentrations in milk. For participants who used cannabis one time during the study, there was a wide range of peak Δ9-THC timing and concentrations in milk, with peak Δ9-THC concentration occurring a median of 2 hours after cannabis use, followed by a decline in concentration.
After abstaining from cannabis for at least 12 hours, milk Δ9-THC concentrations were still measurable for all participants. Median Δ9-THC concentration at baseline (15.3 ng/mL) was notably lower than a previously published value obtained from analyzing single milk samples collected from cannabis-using mothers (22 ng/mL). 15 Similar to previous findings, 5 we found that variation in baseline Δ9-THC concentrations is positively associated with frequency of cannabis use, controlling for time since last cannabis use. Adipose tissue has been identified as a long-term cannabinoid storage depot16,17 with concentration ratios between adipose tissue and plasma of 104:1, 18 and mobilization of adipose tissue as would occur during negative energy balance (a condition particularly relevant in the early postpartum) may contribute to variation in Δ9-THC in milk. Although it was not statistically significant, more rapid weight loss in the postpartum period trended with lower baseline Δ9-THC concentrations in milk, indicating that the role of adipose tissue mobilization during this time could be another contributor to variation in milk Δ9-THC concentrations.
Participants who used cannabis once during the study period produced milk with substantial variation in the peak and trajectory of Δ9-THC concentration. Δ9-THC concentration peaked at a median of 120 minutes after cannabis use. This is later than Baker and colleagues’ 10 report that Δ9-THC in milk peaked 60 minutes after use. We found that mean Δ9-THC concentration at peak was 56.9 ng/mL, compared with 94 ng/mL mean peak reported by this same group. This difference in concentration at peak may be attributable to the different peak timings. We found that when Δ9-THC in milk peaked earlier, it peaked at a higher concentration compared with those who peaked later. The later overall peak timing in this study compared with that of Baker and colleagues 10 may also be because of differences in THC dose and method of cannabis use. Two of our participants vaped cannabis, rather than smoked. Participants in Baker and colleagues’ study were similar to our study’s participants in age, full-term births, and exclusion of other illicit drug use. However, different from our own study’s participants, participants in the Baker and colleagues’ study were instructed to purchase a specific cannabis strain from a preselected dispensary for the study. In addition, participants in our study used cannabis products averaging 35% THC, and those who smoked reported smoking an average of 0.33 g, compared with the controlled dose of 0.1 g of 23.18% Δ9-THC concentration in the study by Baker and colleagues. 10
Owing to sample size and the observational nature of our study, we cannot point definitively to individual factors that contribute to variation in the peak of Δ9-THC concentration in milk after cannabis use. However, we found that participants whose milk Δ9-THC concentrations peaked earliest (30–35 minutes post-cannabis use) were currently using cannabis more frequently and had been using at this more frequent rate for longer than participants whose milk Δ9-THC concentrations peaked later. Their time postpartum was also earlier, on average, 40 days, whereas the other peak groups were, on average, 127 and 130 days postpartum. It is possible that prenatal cannabis use may impact milk Δ9-THC concentration in the early postpartum period. Although we did not assess associations between prenatal variables on milk Δ9-THC concentrations, most participants did use cannabis during pregnancy and most reported the same frequency of cannabis use during pregnancy as during the postpartum period.
Our study estimated that an infant consuming 150 mL/kg/day of milk would consume a median 0.0057 mg/kg/day of Δ9-THC. This is comparable, although slightly greater, than some estimates (0.0041 mg/kg/day) from previous studies.9,10 As participants in our study self-directed their own cannabis use, this higher estimate likely reflects how self-directed and repeated cannabis use of various cannabis products and levels of THC can result in overall greater daily infant Δ9-THC intake.
Infant Δ9-THC intake based on age-appropriate milk volume consumption estimates and actual infant weight was estimated to be even higher (median 0.0052 mg/kg of Δ9-THC in a single feed). Our reference point of how much Δ9-THC mg/kg of body weight an average woman would consume via a 2 mg edible of 100% Δ9-THC was 15 times greater than the mean infant intake estimate per feed. For all infants, this estimate is below the lowest observed adverse-effect level of Δ9-THC of 0.036 mg/kg of body weight and the acute reference dose (ARfD) of 1 μg/kg. 19 However, no assumption of safety of this dose should be inferred. The effects of Δ9-THC consumption via milk on infant development, particularly the effect of repeated low-dose exposures, are unknown. Only two studies20,21 have assessed the association between maternal cannabis use during breastfeeding and infant development, and their results are inconsistent. Evidence from rodent studies indicates persistent mild effects on memory and learning after a single exposure to an ultralow dose that would be equivalent to 0.0002 mg/kg Δ9-THC in humans, 22 which is five times lower than the ARfD. In our study, 75% of the infants were estimated to consume more than this ultralow dose in their average feed. Human research is needed to examine associations with infant development.
Strengths of this study include participant self-directed cannabis use (following planned abstention prior to the baseline sample), which generated novel data on the concentration of Δ9-THC in milk over time given a range of cannabis use patterns. In addition, we had full breast expressions allowing assessment of total Δ9-THC concentration in milk, critical owing to the high lipid content in hind milk. Participants reported their cannabis use on the study day, reducing recall errors. Moreover, we used a validated survey 12 to assess cannabis use behaviors. Limitations are our sample including only people who used cannabis ≥1/week, potential for type II errors in bivariate associations because of small sample size, difficulties in participant estimation of their cannabis use, 23 variable dose, and inconsistent and inaccurate cannabis product THC labeling. 24 Future research should address these limitations.
There is an urgent need for clear guidance on cannabis use while breastfeeding. Although breastfeeding mothers have guidelines for managing some substance use, such as waiting 2 hours after consumption of a single drink of alcohol to breastfeed, 25 there are insufficient data about the persistence of cannabinoids in milk after cannabis use to create similar guidelines for cannabis. Current guidance for medical practitioners is to discourage cannabis use during lactation, given the absence of sufficient research.2,26,27 However, as some perinatal and breastfeeding women report using cannabis for therapeutic purposes,28,29 there is a critical need to develop guidelines accounting for frequency of cannabis use and concentration of cannabinoids in milk over time. Our results demonstrate that Δ9-THC is measurable in milk following cannabis use and that overall Δ9-THC concentration in milk is higher across the day with more instances of cannabis use. We also found that after a single use of cannabis, most participants’ milk Δ9-THC concentrations increased until 2 hours after cannabis use, after which it began to decrease. Although we do not know whether there is a safe threshold of Δ9-THC exposure for infants, these results indicate that there is no clear window of time for mothers to breastfeed after cannabis use without some degree of Δ9-THC exposure for the infant.
Conclusions
In this study of self-directed cannabis use among breastfeeding individuals, Δ9-THC concentration in milk was greater for participants who used cannabis more times during the 8- to 12-hour study period. For those who only used cannabis once during the study period, Δ9-THC concentration in milk peaked at variable times, as early as 30 minutes, but most commonly 2 hours after cannabis use. More frequent cannabis use was associated with greater Δ9-THC concentrations after abstaining 12 hours. These results indicate that infants consume measurable amounts of Δ9-THC from breastfeeding, even after 12 hours of abstaining from cannabis. Future research is needed to identify potential effects (or lack thereof) of frequent Δ9-THC consumption on infant development.
Footnotes
Acknowledgments
This study was made possible by the women and infants who participated in this study.
Authors’ Contributions
E.H.: Writing—original draft, writing—review and editing, data curation, formal analysis, investigation, and visualization. A.B.: Investigation and writing—review and editing. D.G.: Funding acquisition, investigation, resources, supervision, and writing—review and editing. J.W.: Writing—review and editing. C.S.: Investigation and writing—review and editing. B.C.: Investigation and writing—review and editing. O.B.: Investigation and writing—review and editing. C.B.-L.: Conceptualization, funding acquisition, and writing—review and editing. M.M.: Conceptualization and writing—review and editing. M.M.: Conceptualization, funding acquisition, and writing—review and editing. C.M.: Conceptualization, funding acquisition, investigation, project administration, resources, supervision, and writing—original draft, and writing—review and editing.
Author Disclosure Statement
The authors have no conflicts of interest relevant to this article to disclose.
Funding Information
This investigation was supported in part by funds provided for medical and biological research by the State of Washington Initiative Measure No. 171, the State of Washington Initiative Measure No. 502, and Washington State University Health Equity Research Center.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
