Abstract
Introduction:
Human milk is the preferred source of enteral nutrition for very low birthweight (VLBW) infants, and it possibly decreases dependence on parenteral nutrition (PN) and reduces incidence of late-onset sepsis (LOS). No systematic review to date has specifically addressed the value of early versus late introduction of human milk diet (HMD) on duration of PN and incidence of LOS among VLBW infants.
Objective:
To review the evidence for an early versus late introduction of HMD on duration of PN and incidence of LOS in VLBW infants.
Method:
Preferred reporting items for systematic reviews and meta-analysis—guided search of EMBASE and PubMed/Medline databases was conducted for this systematic review using phrases addressing population, intervention, comparator, and outcome framework to identify articles published over the past two decades without language restrictions. Full-text articles (both observational and randomized) that studied an early versus late initiation of HMD were included. Mean difference (MD) and relative risk (RR) with 95% confidence intervals (CIs) were calculated for PN and LOS. Quality of evidence was analyzed using UK National Service Framework and the risk-of-bias was assessed using Robvis®.
Results:
One randomized controlled trial (RCT) and two observational studies (two English and one Chinese) recruited 474 VLBW infants (455 analyzed). Among an intrauterine growth–restricted cohort enrolled in the RCT (n = 72), early HMD resulted in statistically significant reduction in PN dependence. However, no statistically significant difference was found in LOS. Two observational studies found similar reductions in PN duration and LOS incidence among the early HMD cohort. One observational study reported significant PN reduction; however, the incidence of LOS did not reach statistical significance in either case.
Conclusion:
An early HMD may reduce the duration of PN for a growth-restricted VLBW cohort. Observational studies suggesting reduced PN and LOS from early HMD endorse the need for bioactivity-focused human milk research. Variations in feeding guidelines among VLBW infants have the potential to influence neonatal outcomes significantly.
Introduction
It is estimated that 8.6% of livebirths in Europe are premature. 1 On a global scale, premature births and associated complications represent the most common contributors to under-five mortality. 2 The more preterm an infant, the more the likelihood of prolonged neonatal intensive care, gestation-associated morbidities, increased mortality, subsequent hospitalizations, and the potential for childhood disability. 3 Therefore, this global health conundrum is an economic and social encumbrance.
The very preterm (VPT < 32 weeks gestational age [GA]) and very low birthweight (VLBW < 1500 g) infants possess an immature gastrointestinal tract, which hinders the optimal tolerance of enteral feeding and results in inadequate absorption of nutrients, often leading to growth failure and increased morbidity. 4 Nutrition of the VPT infant is thus a critical element of care in the early postnatal period.
The American Academy of Pediatrics and the World Health Organization (WHO) recommend mother’s own milk (MOM) as the ideal source of nutrition for newborn infants. If MOM is unavailable or insufficient, the next best recommended enteral feeding for VLBW infant would be pasteurized donor human milk (DHM). 5 The traditional pasteurization process (Holder method, heating to 62.5°C for 30 minutes) and subsequent freezing and storage of DHM not only inactivates viruses like HIV and CMV but also alters the nutritional composition and immunological properties of breast milk.6–10 Systematic reviews and meta-analysis of the effect of DHM versus cow milk–based preterm formula (PTF) have revealed reduction in incidence of necrotizing enterocolitis (NEC), duration of parenteral nutrition (PN), and the time-to-attainment of full enteral feeding in preterm infants who received DHM over formula.4,11
General recommendation from WHO is to commence breastfeeding within an hour of birth. The initiation of enteral feeding for preterm infants with a human milk diet (HMD), comprising DHM or MOM, can be divided into early or late. An early HMD is initiated from birth up until 72 hours of life for the infant.12,13 In comparison, the late initiation of HMD denotes enteral feeding commencing >72 hours after birth and often from 120 to 144 hours after birth or even beyond. 13 Early HM feeding has been suggested to stimulate gastrointestinal motility, enteric hormone secretion, early and appropriate intestinal microbial colonization, and feeding tolerance.14,15
As mentioned, HMD-containing MOM or DHM is the preferred nutritional source for VLBW infants. However, during the early postnatal window, many VLBW infants require PN in order to meet the optimal nutritional requirements, ensure tissue accretion, prevent excessive weight loss, reduce extra-uterine growth restriction, and allow for optimal growth and reduce mortality. 9 Unfortunately, PN comes with reported risks such as late-onset sepsis (LOS), some of which could be central line–associated bloodstream infections (CLABSI), electrolyte disturbances, nutrient deficiencies, loss of intravenous access, line-associated thrombophlebitis and thrombosis, extravasation of PN, line displacements requiring multiple attempts, and unnecessary handling of vulnerable infants. 16 Neonatal central line for PN is commonly in the form of an umbilical venous catheter (UVC) or peripherally inserted long line. PN is often started as soon as the infant is born via a central or peripheral line. 17 Duration of PN depends on the rate of progression and tolerance of enteral feeding or if the infant’s intestines need to be “rested” in cases of suspected NEC or in the context of surgical gastrointestinal pathologies. In the former case, it is a “bridge” to allow full attainment of enteral feeding, for example, while the enteral HM feeding advances, the need for PN decreases and eventually stops. 18 Maintenance of UVC/peripherally inserted long line and the duration of PN are suggested risk factor for the development of LOS in VLBW infants. The development of neonatal sepsis lengthens hospital stay by an average of 7 days and can cost an extra 3700–29,000 USD per infection. 19
Systematic reviews have already been conducted in relation to HM versus PTF,20,21 DHM versus PTF, 11 and MOM versus DHM, 22 assessing the incidence of NEC and other major neonatal morbidities. With the increasing access to donor milk banks, most neonatal units now encourage the use of HM (MOM or DHM) as the primary enteral nutritional choice for VLBW infants. Therefore, in this context, the value of early introduction of MOM or DHM against the late introduction and costly dependence on PN, and the potential for the inherent risk of an increased incidence of LOS is worth analyzing. Such a systematic review has not yet been conducted.
Methods
This systematic review was designed in line with the criteria suggested by Cochrane collaboration. It was conducted in accordance with the preferred reporting items for systematic reviews and meta-analysis (PRISMA) guidelines of 2021. 23
Search strategy
Search phrases were developed, and an electronic database search was performed using two health-related databases (EMBASE and PubMed/Medline) to identify relevant articles that investigated the influence of an early introduction of HM diet for VLBW infants. The keywords identified were, breast milk, donor human milk, mother’s own milk, very-low birthweight infant, very preterm infant, human milk diet, parenteral nutrition, late-onset sepsis, and late-onset neonatal sepsis.
In agreement with the population, intervention, comparison, outcome framework, search phrases were determined through pilot screening of previously known literature to identify terms relevant to the population, feeding intervention, comparator groups, and outcome. 24 Search terms and Boolean operators (AND, OR, and NOT) used in the database search are presented in Table 1. Both VLBW and extremely low birthweight (ELBW, <1000 g) were searched collectively. Additional records were identified from the bibliographies of eligible studies. Attempts were made to contact authors where full-text articles of selected studies were not available. The database search was limited to articles published from January 2000 up to and including June 2023, and no language restrictions were applied.
Database Literature Search Strategy
Study criteria
Studies were selected on VPT and VLBW infants based on the inclusion and exclusion criteria defined later in the article.
Participants
The inclusion criteria were as follows: (1) VPT infants born <32 weeks GA; (2) VLBW infants weighing <1500 g (including ELBW <1000 g); (3) appropriate for gestational age (AGA) or small for gestational age (SGA), as long as criteria (1) and (2) are met; (4) infants fed an early or late HMD; (5) infants fed a diet of DHM or MOM; and (6) infant feeding commenced with DHM or MOM, irrespective of subsequent fortifier usage (cow milk–derived or human milk–derived).
The exclusion criteria included the following: (1) infants admitted to hospital born >24 hours ago; (2) infants with any congenital/lethal anomaly; and (3) infants fed a diet of preterm or term infant formula (fully or partially) of bovine origin.
Type of study
The inclusion criteria included the following: randomized controlled trials (RCTs), longitudinal observational studies, epidemiological studies, cross-sectional studies, case–control studies, clinical trials, cohort studies, quality improvement projects (QIP) that follows any of the above, and when full-length articles of eligible studies were available.
The exclusion criteria included the following: case reports, case studies, letters to the editor, qualitative thematic analysis, narrative reviews, systematic reviews, literature reviews, expert opinions, abstract-only publications, conference proceedings, books, and book chapters.
Comparison groups or comparator
The inclusion criteria included the following: studies with VPT, VLBW infants fed MOM or DHM, and studies with VPT, VLBW infants fed an early HMD compared with a delayed (late) HMD introduction.
The exclusion criteria included the following: studies restricted to human milk feeding only, and no comment on early versus late, studies restricted to formula (term or preterm) feeding, comparing early versus late introduction, and studies including both HMD and PTF.
Language and publication time
Full-text articles published in all languages from January 2000 to June 2023 that studied early versus late commencement of human milk feed to VPT infants with VLBW infants and influence on the duration of PN and the incidence of LOS. Google Translate was used for the translation of non-English abstracts and full text of eligible studies.
Outcomes
Eligible studies, which analyzed the effect of early and late HMD on VPT, VLBW infants on the duration of PN, and the subsequent development of LOS, either as primary or secondary outcomes in the analysis were included.
Study selection
Records from each database search were imported to reference management software (Zotero®, USA). Before screening, duplicated records were deleted. The remaining records were screened in a two-step manner by the primary reviewer (R.C.). To start, studies were screened according to their title and the abstract. The second step involved screening the full text of the articles. A second reviewer (W.H.) confirmed study selection. The third reviewer (R.K.P.) independently screened the abstracts and full-text articles and acted as an arbitrator when primary and secondary reviewers did not agree or needed clarity. List of excluded studies is provided in Supplementary Material Appendix S1.
Data extraction
Data from the relevant studies were extracted by the primary reviewer onto an Excel spreadsheet. The following information was extracted from each of the selected studies: general study information (i.e., author, setting, study period, objective), descriptive information on the participants (i.e., inclusion and exclusion criteria for the given study, number randomized and analyzed, GA, birthweight), and nutritional management (enteral nutrition, PN, and fortifier usage) and their definitions of the feeding intervention (Table 2).
Characteristics of the Included Studies
BM, breast milk; BW, birthweight; CMDF, cow’s milk derived fortifier; DHM, donor human milk; GA, gestational age; IUGR, intrauterine growth retardation; IVH, interventricular hemorrhage; MOM, mother’s own milk; NEC, necrotizing enterocolitis; PICC, peripherally inserted central catheter; PN, parenteral nutrition; ROP, retinopathy of prematurity; SD, standard deviation; UVC, umbilical venous catheter; VLBW, very low birthweight.
Quality of evidence appraisal
The quality of the included studies was assessed by two of the reviewers (R.C. and R.K.P.) using the UK NSF 27 for cohort or case–control studies. This tool ensured steps were followed in order to appropriately appraise the quality of the studies identified. The evidence from the retrieved studies was given a rating based on the following three categories: (1) the design of the study involved categories such as primary research-based evidence, secondary research-based evidence, or review-based evidence; (2) the quality of the study was assessed by asking five questions to which the answer was yes, no, or in part and resulted in high-, medium-, or poor-quality scores; and (3) applicability that was classified on the basis of context into direct and indirect categories, whereby direct categories involved studies that focus primarily on the effect of an early HMD among VLBW infants.
Risk-of-bias assessment
Risk-of-bias visualization (Robvis®) was used to evaluate the risk-of-bias in the included studies of this review. 28 (Robvis®), a structured and validated internet-based assessment checklist app based on the Cochrane risk-of-bias tool, was used to evaluate the domains for potential bias. This application provided a checklist, based on the Cochrane risk-of-bias checklist, comprising of seven evidence-based domains evaluating different forms of bias: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other bias. A traffic light plot of risk-of-bias is summarized in Figure 1.
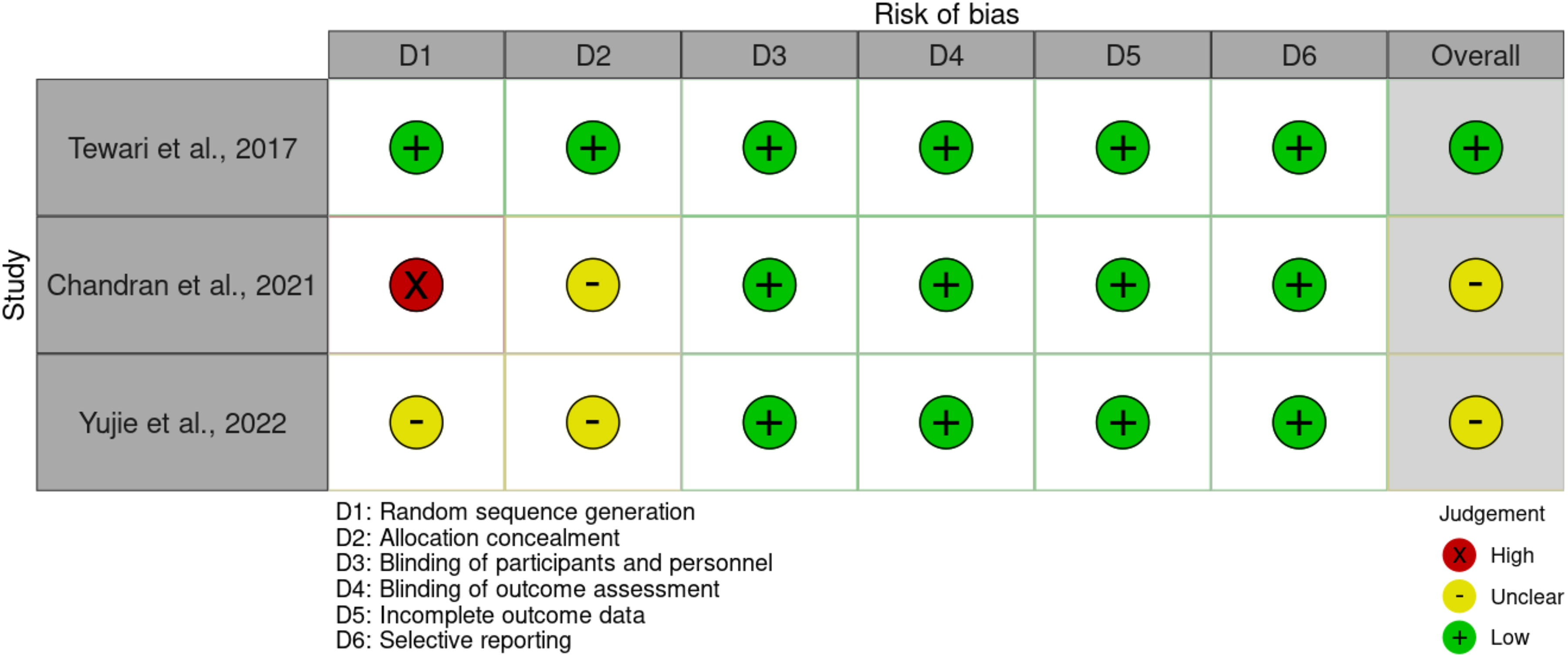
Risk-of-bias assessment (Robvis®).
Synthesis of results
All included studies were described in a study characteristic table and summarized narratively by the outcome. 29 Continuous data (duration of PN) were summarized using mean difference (MD) between groups with 95% confidence interval (CI), where a negative MD indicating a decrease in duration of PN was associated with early HMD. 30 , 31 Dichotomous data (incidence of LOS) were summarized using RR ratio with 95% CI, where an RR < 1 indicates a lower incidence of LOS in the early HMD group. Meta-analysis of the data was not appropriate owing to heterogeneity of study designs among the eligible studies (one RCT and two observational). 29
Results
PRISMA flow diagram in Figure 2 represents the selection process. A total of three unique studies (two English and one Chinese) that met the predetermined inclusion criteria were identified for this review: one randomized controlled study, 13 one prospective study, 32 and one quality improvement initiative. 33
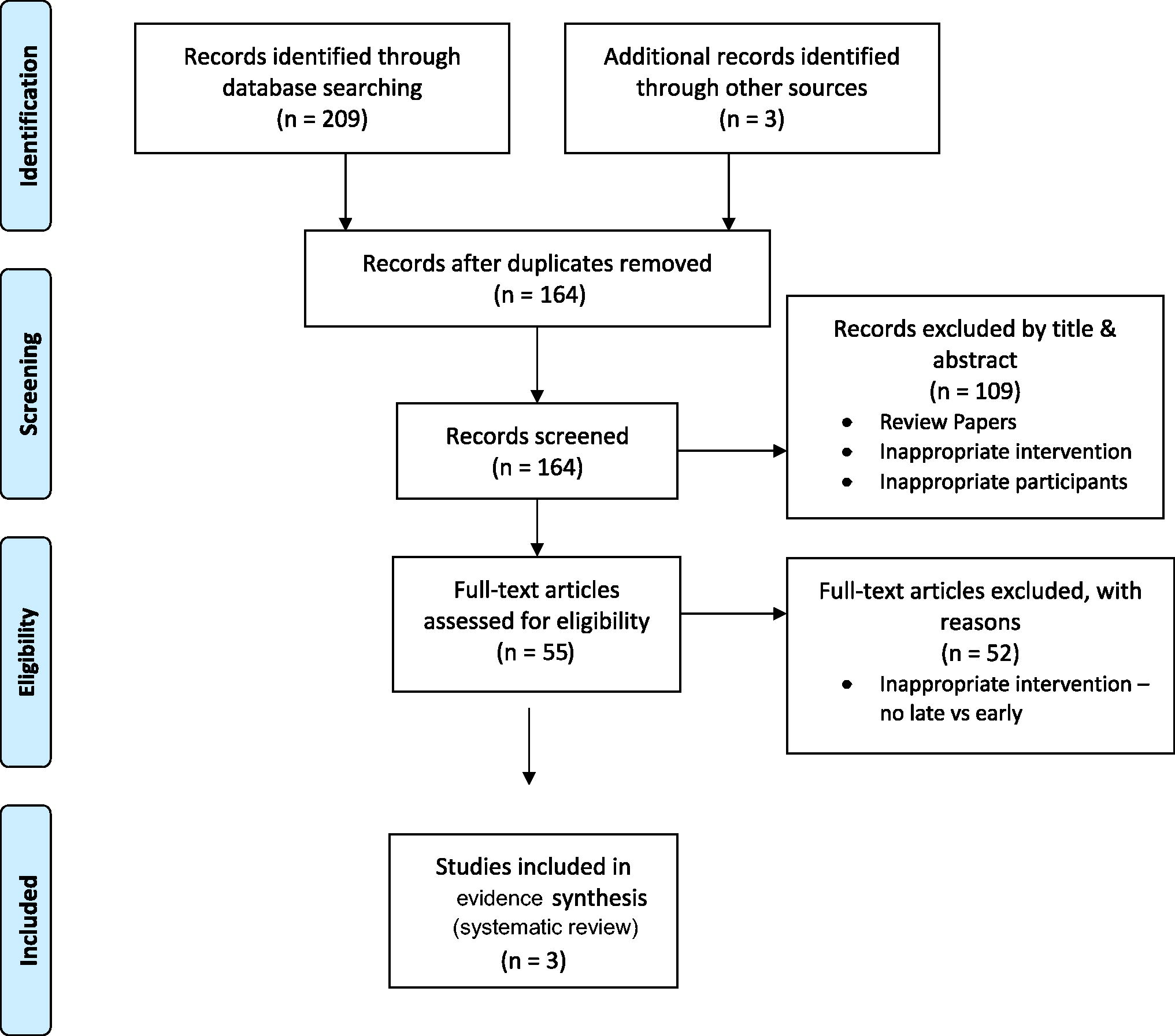
PRISMA flow diagram.
Risk-of-bias
Sequence generation was not reported in the selected RCT, and blinding (of personnel and participants) was not possible in any; however, it was judged that the outcome was unlikely to be influenced by lack of blinding, as it was objective. Visual representation is summarized in Figure 1.
Quality assessment
The three studies generated high quality of evidence following application of the NSF quality assessment tool. The three studies were rated Grade A research, with all having a high-quality score of >7/10.
Study characteristics
Table 2 represents a description of the individual studies. The three studies13,32,33 represent a total of 474 infants included in this review. Only one RCT was eligible for inclusion (Tewari et al. 13 ) with both arms of the trial selected from an IUGR cohort with umbilical Doppler flow concerns. Among the total number of infants, Chandran et al. 32 study represented the highest number and Yujie et al. 33 included the smallest number, and all studies were published between 2015 and 2020. One study was conducted in China, 33 one in Singapore, 32 and one in India. 13 The GA (mean ± SD) of the infants was distributed as follows: 28.7 ± 1.1 weeks in the early intervention group and 28.5 ± 0.8 in the late intervention group for Yujie et al., 33 31 weeks 3 days ± 5 days in the early intervention group and 31 weeks 2 days ± 5 days in the late intervention group for Tewari et al., 13 and 29.4 ± 2.72 for the early intervention group and 29.2 ± 2.61 for the late intervention group for Chandran et al. 32 Birthweight (g, mean ± SD) was recorded as between 1160 ± 165 g and 1232.61 ± 119.90 g. HM, either MOM or DHM, was used in all three studies. Cow milk–derived fortifier was added to the milk in two studies during the subsequent neonatal course, guided by standard recommendations in neonatal practice to fortify when infants reach 80–100 mL/kg of daily HM intake. 34 None of the studies used the relatively new human milk-based fortification to offer an exclusive human milk diet (EHMD). 9
Findings of the included studies
Tables 3 and 4 represent the main results of the studies included in this systematic review.
Influence of an Early HMD Versus Late HMD in VLBW Infants on the Incidence of Late-Onset Sepsis
CI, confidence interval; HMD, human milk diet; RCT, randomized controlled trial; RR, relative risk; VLBW, very low birthweight.
Influence of an Early HMD Versus Late HMD in VLBW Infants on the Duration of Parenteral Nutrition
Values in bold are significant.
CI, confidence interval; HMD, human milk diet; RCT, randomized controlled trial; SD, standard deviation; VLBW, very low birthweight.
Effect on the incidence of LOS
Three studies reported on the incidence of LOS in the VPT, VLBW infants (Table 3). Similar measures of effect were observed in the two observational studies, with risk ratios (RRs) of 0.51 and 0.62 as a result of lower observed incidence of LOS in the early HMD group compared with the late HMD group. Additionally, the RCT highlighted a RR of 0.67. While the RR values were <1 in all studies, none were found to be statistically significant.
Effect on the duration of PN
The studies reported on the duration of PN in the VPT infants (Table 4). The observed mean duration of PN was lower in the early HMD group across both observational studies, with MD of −5.00 and −5.82 days. However, statistical significance was only achieved in one study. 32 Similarly, in the RCT, the mean duration of PN was also lower in the early HMD group, and this reduction was statistically significant.
Discussion
Reflections from the main results
The findings of this review suggest that an early introduction of HMD may be associated with a lower incidence of LOS in comparison with a late introduction (Table 3). Although all studies found lower observed incidences of LOS in the early intervention group with RR ranging from 0.51 to 0.67, none were statistically significant.
The three studies reported early HMD having shorter mean duration of PN (Table 4) compared with late HMD, represented by an MD ranging from −4.00 days to −5.82 days. The reduction in duration of PN in the early HMD was statistically significant in two out of the three studies. The fewer days an infant receives PN, the quicker that infant attains full enteral feed. 34 As PN is costly and has the potential for complications, 17 any intervention contributing to a significant reduction in the duration of PN is desirable.
Among the VPT infants, neonatal sepsis is a significant cause of morbidity, mortality, and impaired neurodevelopmental outcomes, including cerebral palsy.35,36 LOS requires treatment with prolonged antibiotic courses, which can be harmful to the immature gastrointestinal tract and developing microbiome of the VPT infant, 37 predisposing the infant to inflammatory conditions such as NEC15,38,39 and contributing to antimicrobial resistance.25,26 Central lines are needed in VPT infants as a means of receiving medications, fluids, and PN. Unfortunately, they act as a means for microorganisms to enter the bloodstream from the infant’s own skin, carer’s skin, or outside environment. Tewari et al. 13 reported that the earlier removal of the central line following the discontinuation of PN resulted in the decreased incidence of LOS. Therefore, a decrease in the duration of central line maintenance and PN could result in the decreased risk of LOS and CLABSI. As this review is based on a niche and novel topic, our search for robust data observes that studies addressing an early versus delayed or late initiation of HMD are scarce which highlights the need for more research.
Strengths of this study
As the first systematic review specifically addressing the value of an early versus late HMD initiation to VLBW infants in reducing the duration of PN and incidence of LOS, this evidence synthesis offers further evidence base to address potentially modifiable nutritional risk factors in the care of VPT infants. By including all eligible studies over the past two decades, without any applied language restrictions, and following the PRISMA guidelines, risk-of-bias analysis, and quality assessment, authors attempt to fill a specific and critical gap in the evidence supporting the optimal enteral feeding practices. The strength of this systematic review is that it has summarized the available limited evidence of the benefits of an early HMD as a means of offering some protection to VLBW infants against LOS and decreasing the duration of PN.
Limitations of this study
The authors wish to highlight the following limitations. (1) Only two outcomes (duration of PN and incidence of LOS) were included, and the sample size for the two observational studies (one prospective and one QIP) was small. (2) Only one RCT was included, with both arms of the trial selected from an IUGR cohort with umbilical Doppler flow concerns, thus limiting the generalizability of the results to the wider VLBW population. (3) All three included studies used bovine-origin fortifiers during the neonatal course. Thus, the added benefits suggested by the use of HMDF and EHMD could not be evaluated. 40 (4) The definition of early and late, in relation to the timing of the introduction of HMD is not fixed; therefore, there was heterogeneity in the time-to-commence milk feeding among the three studies. (5) Due to the paucity of data, selected studies could not be further analyzed for the value of buccal colostrum application, rate of increase in the volume (slow versus fast) of early feeds, transition from trophic feeding to sequential daily increments, bolus (intermittent) feeding versus continuous gavage feeding, or for the use of probiotics along with the feeding regimen. (6) Authors have included in the LOS those subspecified as CLABSI as well, while acknowledging that these are not always interchangeable. (7) Evidence drawn from studies conducted prior to 2000 was not included. (8) Health economic evaluation through the avoidance of PN and LOS was not included as part of this review. (9) While an International Prospective Register of Systematic Reviews (PROSPERO) registration was initially envisaged, reaudit of a time-bound QIP aimed at reducing the rate of LOS in our unit warranted an early analysis. 41 The next phase of this systematic review addressing the specific role of early and EHMD (with human milk–derived fortification) has been initiated via PROSPERO (ID No: 456853). (10) Priori subgroup analysis was not conducted to ascertain the differential contribution of MOM versus DHM (i.e., early MOM versus early DHM and late MOM versus late DHM). (11) Progression of this systematic review to include a meta-analysis of the data was not appropriate given that two of the three studies were observational studies and not randomized. Inclusion of results from non-randomized studies to a meta-analysis poses the risk that VLBW infants fed earlier could be healthier, reaching full feeds faster, and thus the resultant low LOS. In this context, we have summarized the estimates from the three studies, including the two observational studies as recommended by the Cochrane handbook. 42
Conclusion
The influence of an early HMD comprising MOM or DHM in VPT, VLBW infants on the duration of PN and incidence of LOS has been evaluated in this systematic review. Early introduction of HMD in RCT has shown a reduction in the duration of PN among a growth-restricted VLBW cohort. Observational studies suggesting reduction in PN and LOS from early HMD endorse the need for further research in bioactivity-focused enteral nutrition of VLBW infants during the critical postnatal window, in order to strengthen evidence for the non-nutritional and extra-gastrointestinal benefits associated with bioactivity and immunogenicity of human milk. Further research into the early introduction of HMD on the duration of PN and the incidence of LOS is recommended.
Footnotes
Acknowledgments
The authors acknowledge the support offered by the patient representative of Limerick Neonatal Charity in this project. Support offered by the Library at University Hospital Limerick and University of Limerick is also gracefully acknowledged.
Authors’ Contributions
R.K.P. conceptualized, designed, and supervised the systematic review. R.C. and R.K.P. searched the literature and collected the data. R.C. and W.H. were responsible for accessing, reviewing, and verifying eligible studies and for the integrity of datasets used. R.C. prepared the first draft article, and W.H. performed the initial statistical analysis of the data. W.H. and H.P. contributed to the methodology of systematic review and statistical analysis of the data. H.P. contributed to the intellectual statistical content and edited the article. DM edited the article and contributed to the scientific content. C.P.D. contributed to the intellectual content, structured the article, and critically appraised the pooled observations. All authors participated in the interpretation of the data and writing of the article. All authors confirm that they had full access to all the data used in the study and accepted the responsibility to submit for publication. The final submitted version of the article was reviewed and approved by all authors. R.K.P. acted as the guarantor responsible for the overall content of the published work.
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Patient and Public Involvement (PPI)
While direct patient and public involvement was not applicable for conduct of systematic review of published literature, representation by the designated patient delegate of our Limerick Neonatal Charity (Irish Charity Registration No: 20073732) was ensured as an independent observer and offered parents’ views from the very outset.
Ethics Approval
Formal approval from research ethics committee (REC) was not sought as the systematic review included only published literature and analysis of deidentified data.
Author Disclosure Statement
All authors completed the ICMJE forms and disclosed any conflicts. RKP received honorariums/travel expenses for conference presentations from Nestle®, Prolacta bioscience®, Astra Zeneca®, and Sanofi®. No conflict of interest for the other authors.
Funding Information
The authors have not declared a specific grant for this research from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
