Abstract
Background:
Systemic sclerosis (scleroderma) is a rare progressive autoimmune disease that affects the gastrointestinal (GI) system. Given the increasing prevalence of obesity, selecting the appropriate metabolic and bariatric operation (MBS) in these patients present a unique challenge to bariatric surgeons.
Objectives:
To evaluate the long-term outcomes of MBS in patients with scleroderma.
Methods:
We report 1-year outcomes (esophageal dysmotility, weight loss, and complications) of two patients with scleroderma and obesity who underwent sleeve gastrectomy (SG) and Roux-en-Y gastric bypass (RYGB) at our institution. We also review the literature regarding benign upper GI surgery in patients with scleroderma.
Setting:
This study was performed in University hospital settings at Kingston Health Sciences Center, Canada.
Results:
We identified two patients with severe obesity and scleroderma. Patient #1 was a 61-year-old female with a body mass index (BMI) of 41.6 kg/m2. She had mild esophageal dysmotility without clear evidence of scleroderma esophagus and underwent a SG. On follow-up, she developed scleroderma esophagus with progressive dysphagia and recurrent aspirations. She was converted to RYGB with satisfactory weight loss and improvement in her GI symptoms. Patient #2 was a 61-year-old female with a BMI of 46 kg/m2. She had scleroderma esophagus on initial assessment and underwent a RYGB with excellent control of her GI symptoms and satisfactory weight loss.
Conclusion:
RYGB is the preferred MBS in patients with scleroderma and obesity given the progressive nature of scleroderma. SG should be avoided as patients will likely go on to develop scleroderma esophagus and present with worsening reflux, dysphagia, and aspirations.
Key Points
Systemic sclerosis is a rare progressive autoimmune disease.
Systemic sclerosis is associated with almost universal esophageal involvement. Roux-en-Y gastric bypass is the preferred metabolic and bariatric surgery given the progressive nature of the disease. Sleeve gastrectomy should be avoided as patients will likely go on to develop scleroderma esophagus.
Introduction
Scleroderma is a rare progressive rheumatological autoimmune disease affecting less than 1/10,000 patients. 1 Its main characteristic is diffuse fibrosis of the skin, internal organs, and/or vasculopathy resulting in the highest mortality rate between rheumatological diseases. 2 It can be classified into limited cutaneous systemic sclerosis and diffuse cutaneous systemic sclerosis. Limited cutaneous systemic sclerosis is characterized by restricted skin involvement affecting the limbs distal to the elbows or knees, with or without face and neck involvement, long history of Raynaud’s phenomenon, pulmonary arterial hypertension, and severe disease of the gastrointestinal (GI) tract. Diffuse cutaneous systemic sclerosis is characterized by proximal limb or trunk skin involvement with skin sclerosis, short history of Raynaud’s phenomenon, increased risk of renal crisis and cardiac involvement, and high frequency of severe lung fibrosis. 1 The pathophysiology of the disease remains unknown; however, a combination of genetic predisposition and environmental factors leading to neural dysfunction, muscle atrophy, and fibrosis has been proposed.3,4 Histopathological changes include chronic inflammatory cell infiltration, collagen deposition, muscular atrophy, axonal, and vascular degeneration. 4
Systemic sclerosis can affect any system including cardiac, pulmonary, renal, and musculoskeletal systems. 4 The GI system involvement is very common, in up to 90% of patients, with all parts of the GI system being affected.5,6 Half of the patients with GI involvement have significant GI symptoms affecting quality of life and up to 8% of them develop life-threating complications such as malnutrition and pseudo-obstruction.6,7 The esophagus is the most commonly affected part of the GI tract in systemic sclerosis, with up to 90% involvement. The main feature of scleroderma esophagus is dysmotility with decreased/absent peristalsis and lax lower esophageal sphincter (LES) leading to gastroesophageal reflux, regurgitation, aspiration, and dysphagia. Esophageal stenosis, Barrett esophagus, and esophageal cancer occur more frequently in patients with systemic sclerosis. 8 Manometric findings of esophageal hypoperistalsis and aperistalsis have been reported in 81% and 52% of patients, respectively. 8 Gastroparesis is another common sequela of the disease, with up to 50% of patients being affected. 9 Patients also may present with anemia from esophagitis, gastritis, and gastric antral vascular ectasia (GAVE). The involvement of the GI system is progressive and irreversible.10,11
The prevalence of obesity, defined as body mass index greater or equal to 30 kg/m2, has tripled worldwide over the last 50 years. 12 Given the universal progressive and irreversible involvement of the GI tract with systemic sclerosis, specifically esophageal dysmotility and gastroparesis, plans for metabolic and bariatric surgery (MBS) in patients with severe obesity and systemic sclerosis deserve careful consideration regarding the type of MBS to perform. We describe a case series of two patients with systemic sclerosis and severe obesity who underwent MBS. We report on their 1-year outcomes and GI symptoms postoperatively.
Case #1
This is a 61-year-old female with limited cutaneous systemic sclerosis and severe obesity (Ht = 167.4 cm, Wt = 116.7 kg, body mass index [BMI] = 41.6 kg/m2). She presented to our Bariatric Center of Excellence seeking MBS in 2019. Her past medical history was significant for rheumatoid arthritis, obstructive sleep apnea, pulmonary hypertension among others. She reported mild gastroesophageal reflux symptoms and nonprogressive dysphagia to solids. Her medications included domperidone, esomeprazole, golimumab, hydroxychloroquine, methotrexate, and prednisone.
The patient underwent a multidisciplinary assessment at our Bariatric Center of Excellence along with dedicated studies of her upper GI tract including esophagogastroduodenoscopy (EGD), barium swallow with marshmallow, and esophageal manometry. EGD demonstrated laxity of lower esophageal sphincter (Hill Grade III), no esophagitis, no Barrett’s esophagus, and no GAVE. Barium swallow demonstrated distal esophageal dysmotility with tertiary contractions and transient holdup of mini marshmallow in the mid esophagus reproducing patient’s symptoms as shown in Figure 1. Esophageal manometry in 2016 and 2018 demonstrated nonspecific motor disorder without features of scleroderma esophagus.
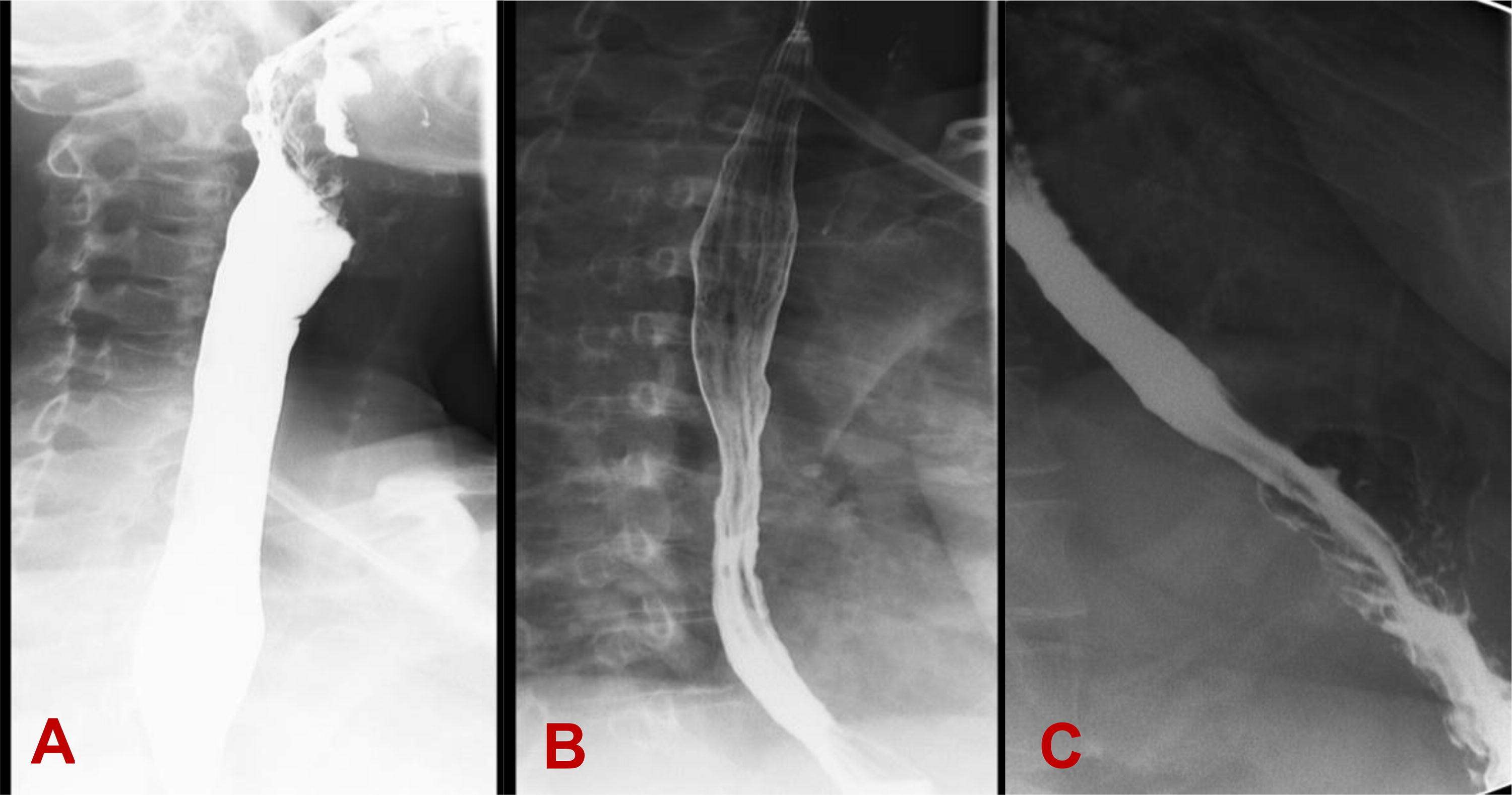
Barium swallow showing
Following multidisciplinary discussion and with input from gastroenterology and rheumatology teams this patient elected to proceed with a laparoscopic sleeve gastrectomy (SG). A Roux-en-Y gastric bypass (RYGB) was not recommended because of increased risk of anastomotic leak in the setting of chronic immunosuppression from steroid use. She underwent a laparoscopic SG with concurrent cholecystectomy for recurrent biliary colic in October 2020. Her weight on the day of surgery was 92 kg, BMI 34 kg/m2. SG was performed over 40F bougie with staple line terminating 6 cm proximal to the pylorus. The staple line was oversewn using a seromuscular Lembert-type 3-0 PDS (absorbable monofilament) running suture. Intraoperative endoscopy demonstrated a straight sleeve without narrowing or twisting, patent pylorus and negative air leak test. On postoperative days (POD) 1, 2, and 3, she reported dysphagia to liquids prompting an upper GI series to be done. Upper GI series on POD 3 demonstrated delayed transit of esophageal contrast into the stomach at the level of proximal gastric body as shown in Figure 2. Majority of contrast column refluxed into the esophagus, and only a portion of contrast was cleared from the esophagus after 5 min. A diagnosis of postoperative edema in the proximal stomach was made, and patient was discharged home on POD 5 tolerating liquids.
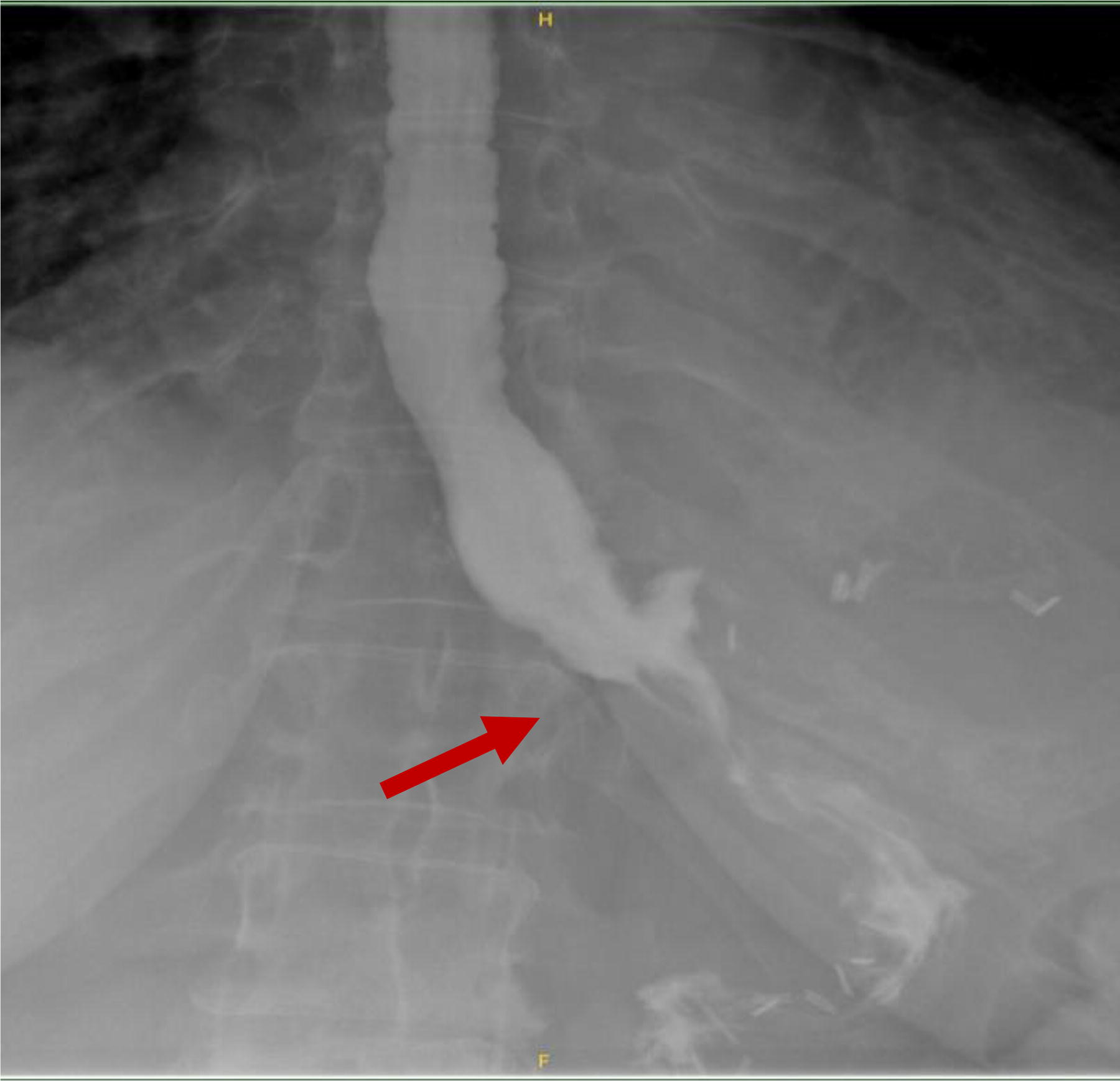
UGI demonstrating delayed transit of esophageal contrast into the gastric sleeve.
Three months later, patient reported developing progressive dysphagia to solids. Her weight decreased to 74.6 kg (BMI = 27.4 kg/m2) with percent excess weight loss (EWL) of 79.5%. Repeat EGD (February 2021) showed LA class A esophagitis with bile gastritis and normal anatomy of the SG. She was started on lansoprazole 30 mg PO BID and famotidine 20 mg PO BID with resolution of her dysphagia symptoms.
Patient presented back to our clinic in February of 2022 with multiple episodes of aspiration pneumonia. Upper endoscopy demonstrated no esophagitis or hiatus hernia but did show cartwheel gastritis (Fig. 3). Pharyngoesophagram showed a normal oral phase of swallowing and marked esophageal dysmotility with holdup of majority of the ingested barium at the level of the lower esophageal sphincter (Fig. 4). Esophageal manometry revealed absent contractility of the esophagus, consistent with scleroderma esophagus, with relaxation of LES and high-pressure zone in the proximal stomach (Fig. 5). Twenty-four-hour pH study demonstrated good control of acid exposure on BID dose of Proton Pump Inhibitors (PPI). Given patient’s progression to esophageal aperistalsis (scleroderma esophagus), and recurrent aspiration pneumonias requiring hospital admissions, we recommended proceeding with conversion to a RYGB and placement of a feeding gastrostomy tube into the gastric remnant following multidisciplinary discussion with gastroenterology. Conversion to RYGB was recommended in an effort to decrease the pressure gradient across the gastroesophageal junction to facilitate emptying of the luminal esophageal contents from the esophagus into the low-pressure system of the RYGB. Patient underwent the conversion procedure to a RYGB (75 cm Roux limb and 45 cm biliopancreatic limb) on July 2022, 21 months after the primary SG operation. Her weight was 92 kg with BMI of 35 kg/m2 on the day of the conversion operation. There were no intraoperative or postoperative complications. Following the conversion procedure, she is able to tolerate PO diet with less dysphagia and was using the G-tube for medications only. There were no hospital admissions for aspiration pneumonia for 12 months since the conversion procedure.
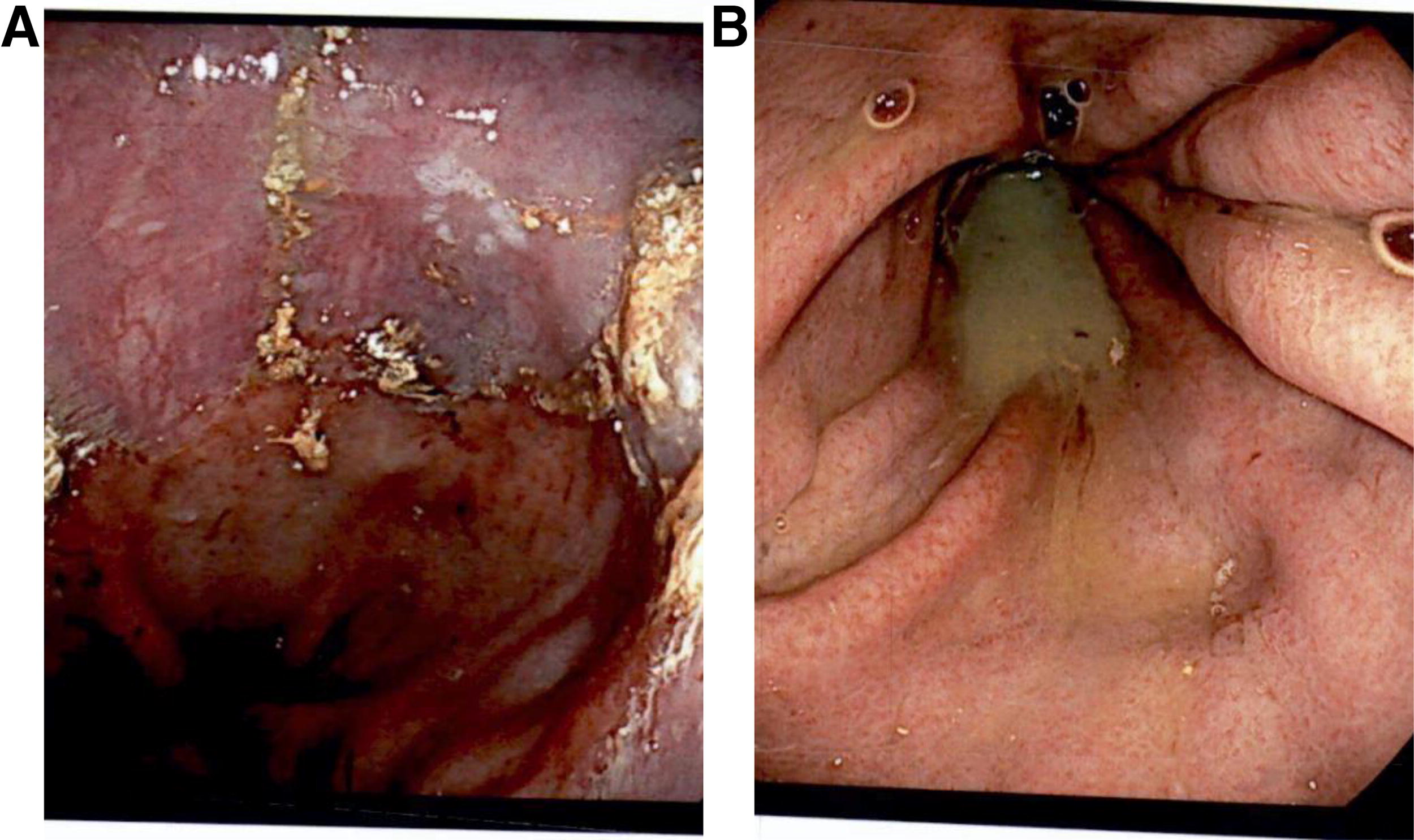
EGD showing squamocolumnar junction without esophagitis
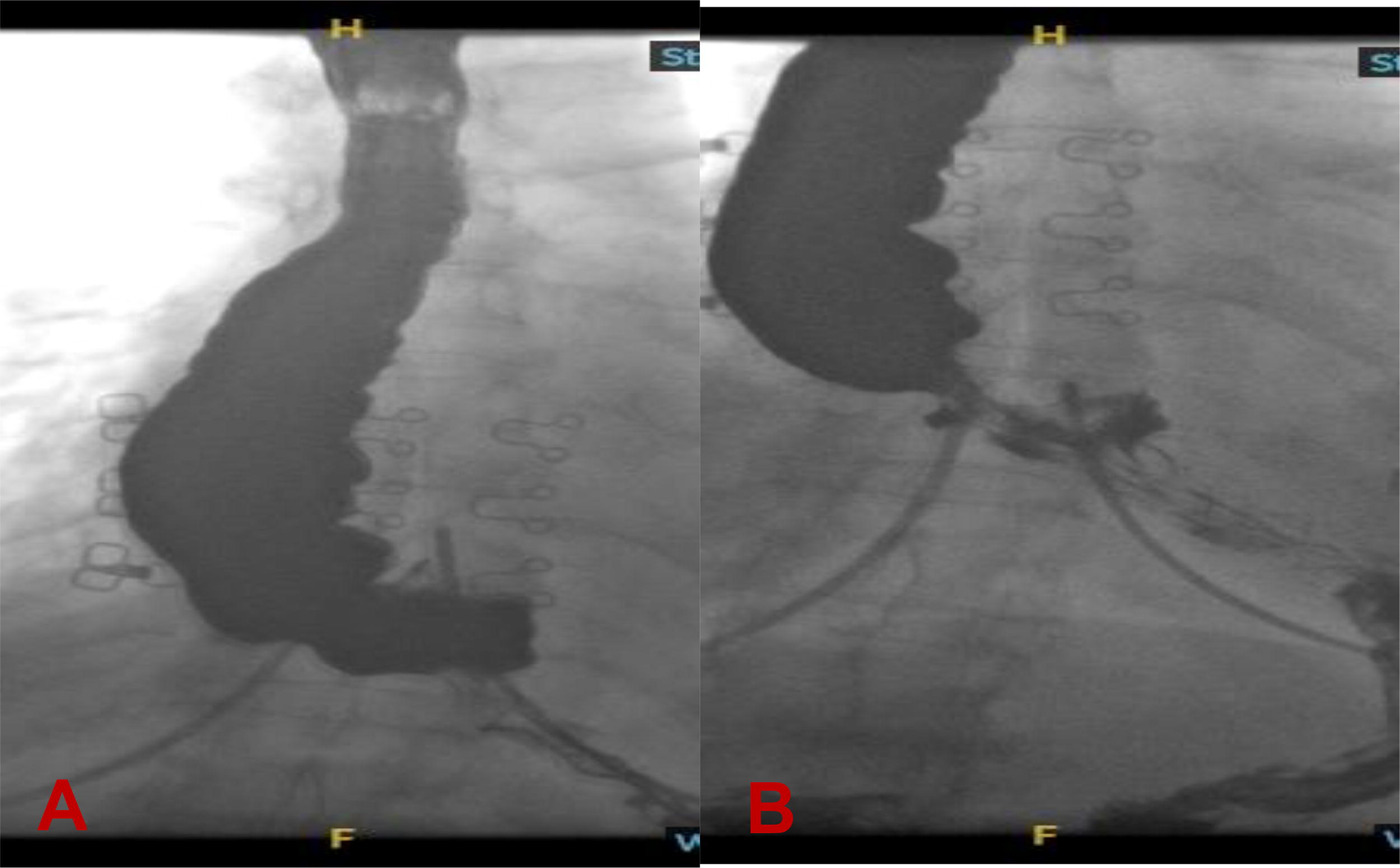
Pharyngoesophagram showing
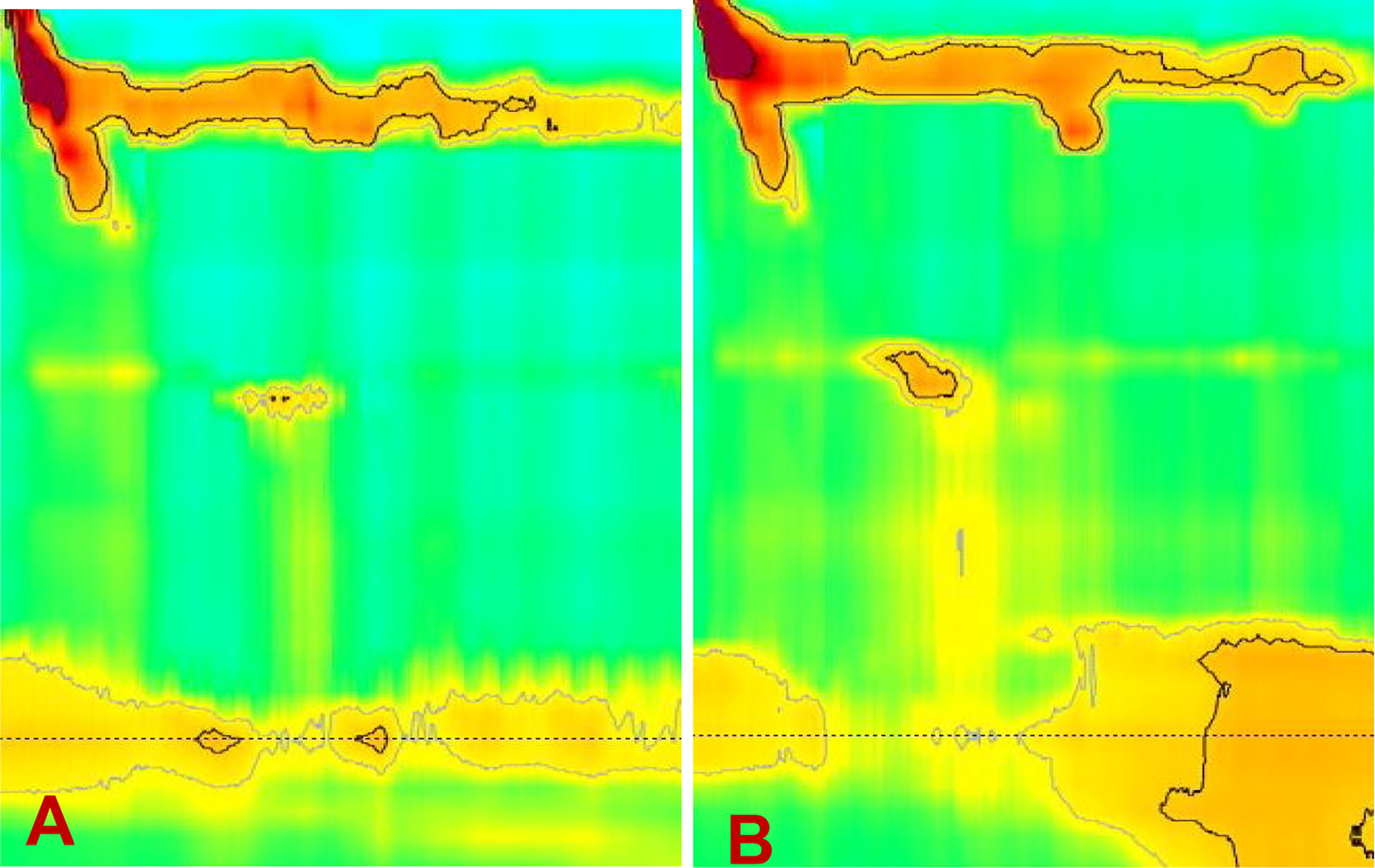
Esophageal manometry showing
Case #2
This is a 61-year-old-female with longstanding history of severe obesity (Ht 154 cm, Wt 110 kg, BMI 46 kg/m2) and limited cutaneous systemic sclerosis systemic sclerosis with esophageal involvement. She was referred to our Bariatric Center of Excellence in May of 2021. Her past medical history was significant for hypertension, type 2 diabetes mellitus, and gastroesophageal reflux symptoms among others. Her medications included methotrexate, plaquenil, pantoprazole, and domperidone.
Preoperative investigations included EGD, barium swallow, esophageal manometry, and a gastric emptying study. EGD (May 2021) demonstrated no esophagitis, no hiatus hernia, with endoscopic appearance of gastroparesis. Gastric emptying study (December 2021) demonstrated delayed gastric emptying at 4 h. Barium swallow (June 2021) demonstrated reduced primary peristalsis and moderate distension of thoracic esophagus with relaxation of LES consistent with scleroderma esophagus with dysmotility. Esophageal manometry (December 2021) demonstrated failed esophageal peristalsis on all wet and viscous swallows with normal relaxation of LES, consistent with scleroderma esophagus (Fig. 6).
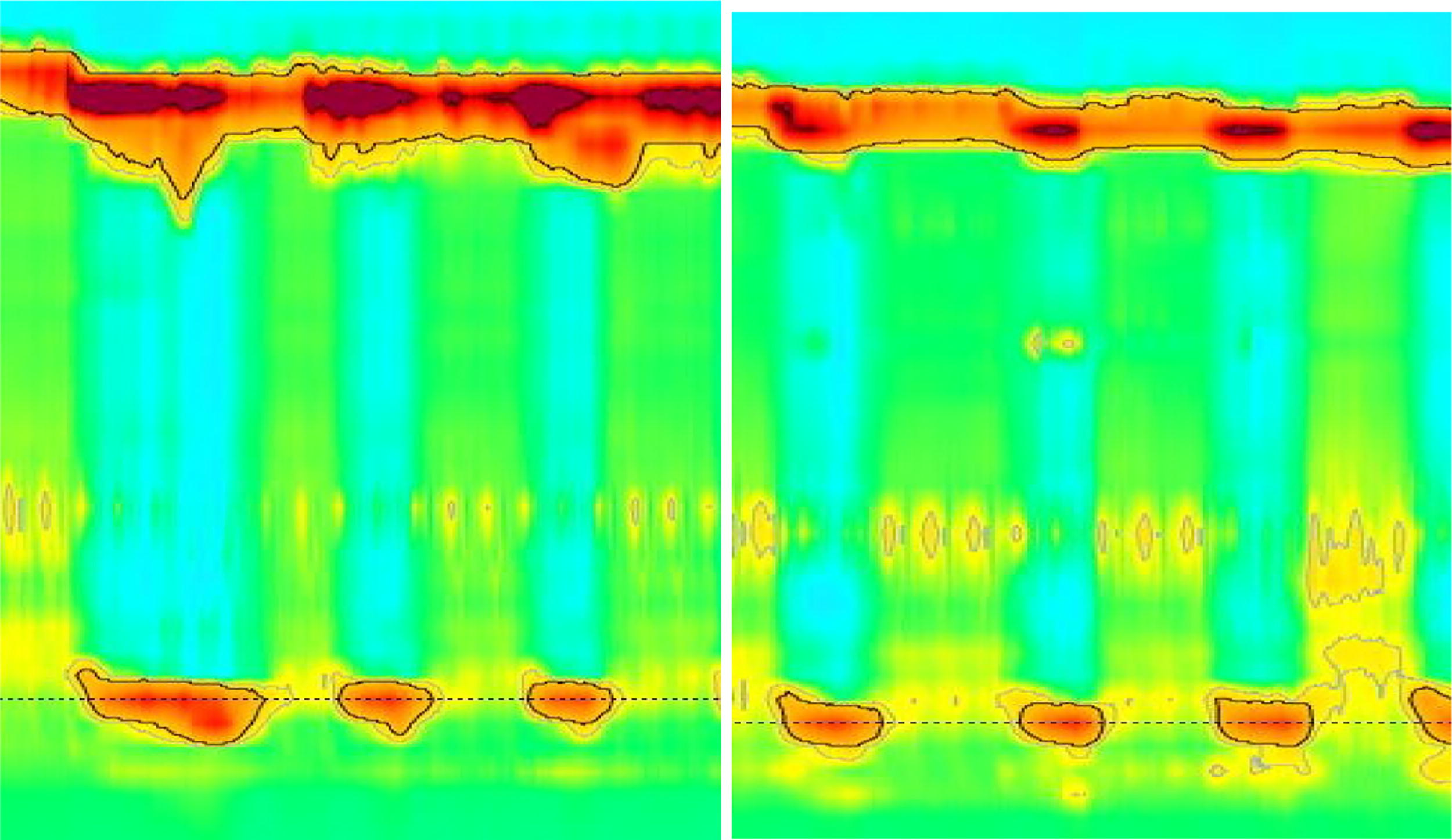
Esophageal manometry demonstrating failed esophageal peristalsis on wet and viscous swallows consistent with scleroderma esophagus.
After undergoing a multidisciplinary assessment in the Bariatric Center of Excellence and following consultation with gastroenterology, she underwent a laparoscopic RYGB (125 cm Roux limb and 75 cm biliopancreatic limb) in February of 2022. The procedure and postoperative course were uncomplicated. At 12 postoperative months, she denied dysphagia symptoms and reported good control of her gastroesophageal reflux symptoms on domperidone 10 mg PO daily without PPI or H2 blocker. Her weight was 67 kg with BMI of 28.3 kg/m2 with EWL of 84.8% at 12 postoperative months.
Discussion
We presented two patients with systemic sclerosis and severe obesity who underwent MBS: one patient undergoing a SG and the other undergoing a RYGB. Patient in case #1 did not have esophageal involvement from systemic sclerosis at the time of assessment for MBS, underwent a SG and developed scleroderma esophagus postoperatively. Her dysphagia symptoms and aspiration pneumonias improved with conversion to RYGB. The patient in case # 2 had systemic sclerosis with scleroderma esophagus at the time of assessment for MBS, underwent a RYGB and had excellent response to surgery without postoperative symptoms of dysphagia.
Despite being a very rare disease, systemic sclerosis is progressive disease with universal involvement of the esophagus over time and high likelihood of developing gastroparesis. This presents a challenge for the bariatric surgeon as patients with systemic sclerosis and severe obesity may be seeking MBS to improve their health, quality of life and to resolve obesity-related comorbidities. To our knowledge, this is the first case series describing outcomes of MBS in patients with systemic sclerosis. Lomas et al. reported a case of a women with severe obesity and limited cutaneous systemic sclerosis without esophageal involvement as preoperative manometry only showed a laxity of lower esophageal sphincter. Patient underwent a RYGB with acceptable weight loss and without complications. 13 Kasza et al. reported a case of limited cutaneous systemic sclerosis without esophageal involvement. 14 Patient underwent a SG with good weight loss outcomes over 12 months follow-up, but postoperative upper GI symptoms were not reported in their case report. 14
Around 50% of patients with systemic sclerosis will develop gastroparesis, which must be a consideration during MBS. 9 In RYGB, the pylorus is bypassed with rapid emptying of gastric pouch contents into the jejunum. In SG, the decreased distensibility of the gastric sleeve result in higher intraluminal pressure within the stomach with improved gastric emptying.15,16 In a cross sectional study assessing gastric emptying in 12 patients undergoing either SG or RYGB compared with controls, gastric emptying was significantly faster after RYGB (1.49 ± 0.27 min) and SG (32 ± 14.3 min) compared with controls (65.8 ± 13.6 min). There was no significant difference in gastric emptying between RYGB and SG. 16 Masclee et al. published a systematic review with 222 patients on the role of gastric bypass and SG as a treatment for gastroparesis. Although all studies reported symptoms improvement, gastric emptying was noted to be better after RYGB vs SG. 17 Moreover, the effects of the high-pressure system of the gastric sleeve on esophageal motility are not clear. In a review of nine studies assessing esophageal motility in patients post SG had conflicting results, possibly related to different surgical techniques for the SG. 18 The lower esophageal sphincter pressure was higher in two studies, lower in four studies, and the same in two studies. Amplitude pressure was higher in one study, lower in two studies, and unchanged in one study. Ineffective peristalsis was increased in three studies and unchanged in one study. 18
RYGB has been reported as surgical treatment for gastroesophageal reflux disease in patients with systemic sclerosis.19–21 Surjan et al. reported a case of severe gastroesophageal reflux disease in a patient with scleroderma esophagus who underwent a short limb gastric bypass (35 cm Roux limb and 40 cm biliopancreatic limb) with complete resolution of reflux symptoms at 2 years of follow-up. 21 Yan et al. compared the role of fundoplication and RYGB for management of gastroesophageal reflux disease in 14 patients with systemic sclerosis. Thirteen patients underwent preoperative manometry, 8 patients in both groups had absent contractility and four patients had ineffective esophageal motility. All patients in the RYGB group had complete resolution of reflux symptoms compared with only 50% in the fundoplication group. 20 In the case series of 33 scleroderma patients with gastroesophageal reflux disease undergoing fundoplication, RYGB, or esophagectomy, Kent et al. reported that RYGB was associated with fewer symptoms of dysphagia and gastroesophageal reflux compared with fundoplication. 19
Based on our experience and the available literature, we recommend against a SG in patients with systemic sclerosis and severe obesity. The high-pressure system of the gastric sleeve will likely worsen patients’ gastroesophageal reflux and dysphagia symptoms. RYGB is the preferred operation for patients with systemic sclerosis and severe obesity as it creates a low-pressure system within the gastric pouch with rapid emptying across the gastrojejunal anastomosis. Understanding the progressive disease of systemic sclerosis is also important. GI tract involvement in systemic sclerosis appears to progress in an orderly series of steps from neural dysfunction to smooth muscle atrophy to smooth muscle fibrosis resulting in permanent dysfunction. The mechanism underlying neural dysfunction remains unclear, but two hypotheses include arteriolar changes in the supply to the nerve or compression of the nerve secondary to collagen deposition. 3 In the early stage of the disease, the nerves are affected, but smooth muscle contractility is not. As a result, the patient may be asymptomatic or have very mild symptoms. It is possible that patient in case #1 was in this early stage of her esophageal disease at the time of her consultation for MBS as the manometric findings within the esophagus were normal. In the second stage of the disease, smooth muscle beings to weaken and atrophy, and patients begin to present with dysphagia symptoms. Finally, at the end stage of the disease, there is fibrosis of the muscle, and it is no longer possible to restore motor function. SG creates a high-pressure system distal to a lax lower esophageal sphincter and likely impairs emptying from the aperistaltic esophagus into the stomach. 22
Conclusion
Patients living with severe obesity and a diagnosis of systemic sclerosis, with or without esophageal involvement, should be approached with caution by the bariatric surgeon and with a clear understanding of the progressive nature of systemic sclerosis as a disease. SG should be avoided in these patient as it will likely worsen gastroesophageal reflux disease and dysphagia and increase risk of aspiration pneumonia and pulmonary fibrosis. RYGB appears to be the preferred MBS; however, further studies examining outcomes of MBS for patients with systemic sclerosis and severe obesity are required to make more definitive recommendations and conclusions.
Footnotes
Authors’ Contributions
M.A.: Writing—original draft, data curation, and investigation. M.R.: Writing—review and editing and resources. B.Z.: Conceptualization, methodology, project administration, and supervision.
Author Disclosure Statement
All authors have no financial or personal relationships to disclose that may affect the article.
Funding Information
No funding was received for this article.
