Abstract
Many of the recently discovered Earth-like exoplanets are hosted by M and F stars, stars that emit intense UVC, especially during a flare. We studied whether such planets are nevertheless habitable by irradiating a desert lichen, Clavascidium lacinulatum, with 254-nm 55 W/m2 UVC nonstop for 3 months in the laboratory. Only 50% of its algal photobiont cells were inactivated. To put this in perspective, we used the same setup to challenge the photobiont cells but grown in pure culture, and Deinococcus radiodurans, the most radiation-resistant bacterium on Earth. Entire monolayers of hundreds of cells were inactivated in just 60 s. Further studies indicated that the cortex of the lichen was rendered UVC-opaque by deposits of phenolic secondary metabolites in its interstices. The lichen was injured only because, while most photochemical reactive oxygen species were quenched, photochemical ozone was not. We conclude that UVC-intense exoplanets are not necessarily uninhabitable to photosynthetic organisms.
1. Introduction
Earth-like exoplanets revolve around a main sequence star (Buccino et al., 2007; Kasting et al., 1993; Rushby et al., 2013; Scalo et al., 2007; Schwieterman et al., 2018). All main sequence stars, M and F types in particular, emit intense ultraviolet rays (UV), especially during a flare (Buccino et al., 2007; Spinelli et al., 2023). Short-wavelength UV (200–280 nm), or UVC, is biocidal. Thus, exoplanets may not be considered habitable unless we show that photosynthetic microorganisms are capable of withstanding UVC assaults on ecologically relevant timescales.
According to the pioneering work of the late Professor Richard Castenholz and his associates, cyanobacteria that possess a polysaccharide sheath may be such organisms (Garcia-Pichel et al., 1992; Garcia-Pichel and Castenholz, 1991, 1993; Proteau et al., 1993). When exposed to long-wavelength UV (315–400 nm), or UVA, these prokaryotic microorganisms melanize their polysaccharide sheaths with scytonemin, a broad-spectrum UV-screening pigment, until their sheaths are UV-opaque. Such UVA-acclimated cyanobacteria can survive 1.0 W/m2 254 nm UVC for at least one h (Dillon and Castenholz, 1999).
Available evidence suggests that crustose and foliose lichens grown in open habitats may be such organisms as well. These ∼1 mm thick flat growths, called thalli, contain in their interstices UV-attenuating deposits of phenolic secondary metabolites traditionally referred to as lichen acids (le Pogam et al., 2016; Molnár and Farkas, 2010; Nguyen et al., 2013; Radice et al., 2016). Such lichens are also desiccation tolerant and able to revive photosynthetically within minutes of rehydration and reach full capacity overnight. Exposure to simulated martian conditions or to outer space in Earth orbit, at least on the order of days, had little effect on this revival (Brandt et al., 2015; de la Torre Noetzel et al., 2007; de la Torre et al., 2010; de Vera et al., 2002, 2004; de Vera et al., 2010; de Vera et al., 2019; Lorenz, 2023; Millot et al., 2012; Nguyen et al., 2013; Raggio et al., 2011).
Exoplanets hosted by M and F stars receive far higher UVC photon fluxes, up to 100 W/m2 (Abrevaya et al., 2020; Kasting et al., 1993; Rugheimer et al., 2015; Welsh et al., 2007). To study if such fluxes are survivable, we exposed air-dried thalli of a squamulose lichen from the Mojave Desert, Clavascidium lacinulatum, to 55 W/m2 254-nm UVC in ambient air in our Las Vegas laboratory nonstop for 3 months. Remarkably, half of the algal photobiont cells in them remained viable and multiplied when plated on agar.
Next, we conducted two experiments that indicated that lichen acids are nature’s equivalent to aromatic additives in UV-resistant plastics, photostabilizers that absorb UV without being destroyed while keeping photochemical formations of reactive oxygen species (ROS) and ozone to a minimum (Yousif and Haddad, 2013). First, we showed that in the presence of N, N-dimethyl-p-nitrosoaniline (pNDA), a presumed photostabilizer, photochemical hydroxyl radical (•OH) formed under UVC but not under UVA. Second, we showed that UVC was less injurious to lichens in an anaerobic glovebox than to lichens in ambient air.
Materials and Methods
Soil-inhabiting squamulose lichen Clavascidium lacinulatum (basionym Placidium lacinulatum) (Prieto et al., 2012) was collected from the Mojave Desert (36°27'7.11"N, 114°42'33.34"W; permit #: MOJA-2020-SCI-0028) (Fig. 1). Its photobiont Myrmecia israelensis was isolated in culture using soil extract agar as described by Ahmadjian (1967). Soil extract was obtained by boiling desert soil in deionized water in 1:1 ratio (w/v) for one h, followed by centrifugation removal of particulates. For UVC tests, liquid cultures were raised in faster-growing mixotrophic Trebouxia medium (Starr, 1964). The bacterium Deinococcus radiodurans (B2906 Agriculture Research Service Culture Collection) was grown in liquid Luria-Bertani medium (Bertani, 1951).

Lichens are symbiotic associations between two microorganisms, an alga or a cyanobacterium (the photobiont) and a fungus (the mycobiont). Mycobiont (lichens are named after mycobionts) and photobiont were identified based on the nucleotide sequence of the V4 region of 18S rDNA. For the photobiont, the V3-V4 region of chloroplast 16S rDNA was sequenced as well. For DNA extraction, 50 mg of dry lichen was bead-beaten using a Mini-Beadbeater (BioSpec Products) at top speed with five 3-mm glass beads in a 2-mL vial for three rounds, each precooled in liquid nitrogen and lasting 30 s. The DNA extraction method of Park et al. (2014) was followed except that phenol-chloroform isoamyl isopropanol, instead of chloroform, was used as solvent. Paired-end sequencing of the V4 region of 18S rDNA and of the V3-V4 region of 16S rDNA was done by CD Genomics (New York, USA) using primers 528F-706R and 341F-806R, respectively. Operational taxonomic unit (OTU) construction was described by Saidi-Mehrabad et al. (2020). The BLAST search against the NCBI database produced a match between the most abundant 18S rDNA OTU and the photobiont Myrmecia sp. PA-3–3-2 (MT735207.1) with 100% sequence similarity; a match between the second most abundant 18S rDNA OTU and the mycobiont Clavascidium lacinulatum (EF689847.1) with 97.70% sequence similarity; and a match between the most abundant 16S rDNA OTU and the chloroplast of the photobiont Myrmecia israelensis (NC_025525.1) with 100% sequence similarity. The raw16S (SAMN40377171) and 18S rDNA sequences (SAMN40377172) were deposited in the NCBI Sequence Archive.
Injury-repair incubation
To ensure that lichens were injury-free going into a test, they were incubated on a moist cotton pad for 24 h and then air-dried. The revitalization protocol was based on daily measurement of quantum yield of lichens under the above condition for five days. The maximum quantum yield was reached by hour 24.
Quantum yield (QY)
A MINI-PAM-II pulsed amplitude modulated fluorometer (Heinz-Walz, Germany) was used to measure photosystem II QY, defined as follows (Schreiber, 2004):
QY0.5 and QY24 as measures of total and recoverable injury
QY24, taken after 24 h of rehydration, is a gauge of irrecoverable injury. QY0.5, taken at hour 0.5, is a measure of total injury. Figure 2 shows the relationship between QY0.5 and QY24 in lichens that were UVC irradiated at 55 W/m2 for 0, 1, 18, 30, and 60 days. It is clear that repair was not yet activated by hour 0.5 and that repairable injury was repaired by hour 24.
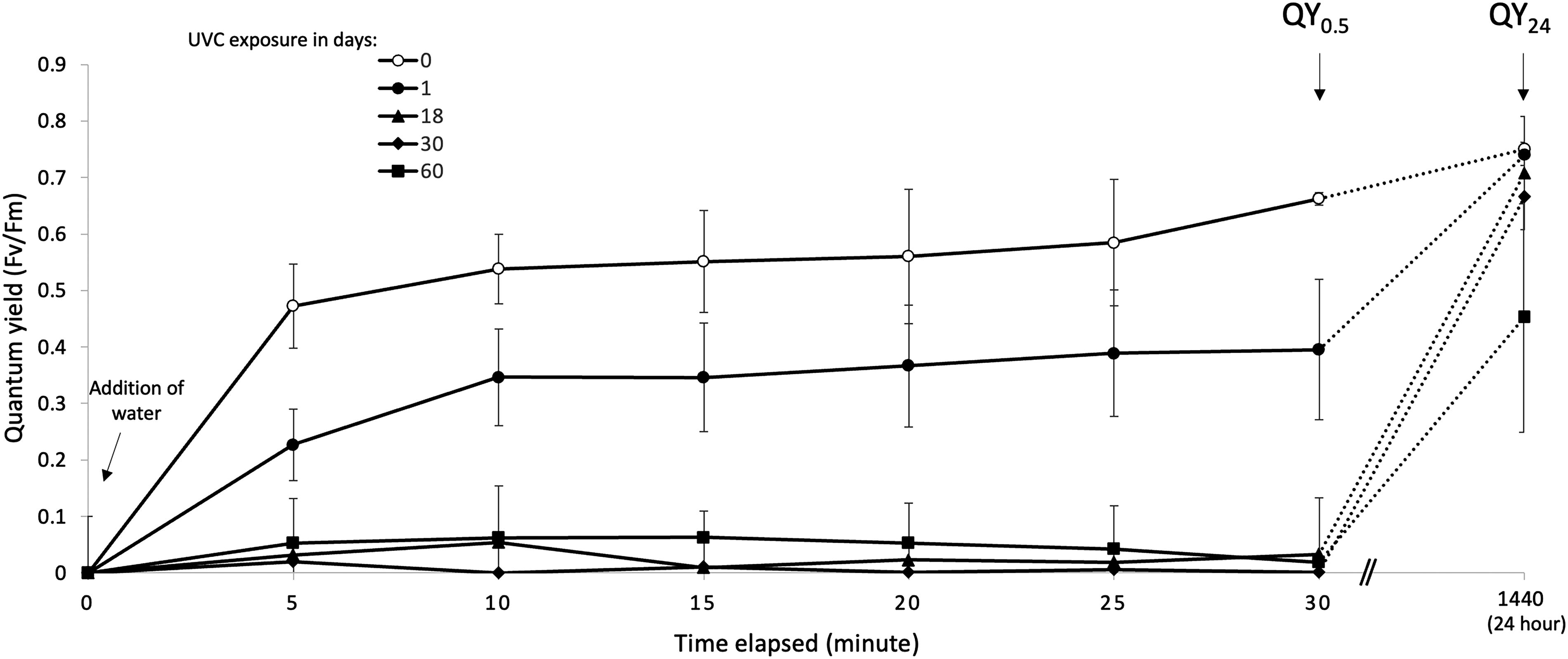
Photosynthetic recovery curves of lichens that were UVC-injured to varying degrees (N = 3, error bar = SD), showing that at hour 0.5 injury repair had yet to begin while at hour 24 repairable injury was repaired. That is, QY0.5, quantum yield taken at hour 0.5, is a gauge of total injury while QY24, quantum yield taken at hour 24, is a gauge of irrecoverable injury.
A tissue grinder was used to turn 5–10 mg (dw) lichen of measured thallus surface area in 5 mL water into a suspension of individual algal cells and tissue fragments that contained no more than 20 cells. Its cell/aggregate density was estimated using a light microscope and a hemocytometer to inform serial dilutions and plate counts to determine colony forming units (CFUs) per thallus surface area. For the bacterium D. radiodurans and the cultured photobiont M. israelensis, 0.1 mL of liquid culture of known cell density was spread on agar plates. Following irradiation with the covers off, the plates were incubated until colonies emerged in the unirradiated (control) plates.
UV lamps and gas composition in the anaerobic glovebox
We used a 115-watt germicidal 254-nm lamp and a 100-watt 365-nm lamp. Our anaerobic glovebox was filled with 90% N2, 5% H2, and 5% CO2.
pNDA as a model photostabilizer to demonstrate ROS from UVC but not UVA
N, N-dimethyl-p-nitrosoaniline (pNDA) is a colorimetric hydroxyl radical (•OH) reagent (Kraljić and Trumbore, 1965). Twenty milliliters of 10 µM solution in a 50 mL beaker was irradiated with UVA or UVC at 55 W/m2. Absorption at 440 nm was measured using a Hatch DR6000 spectrophotometer.
Liquid chromatography-UV-mass-spectrometry analysis of lichen acid extracts
Extracts were injected (1.00 μL unless otherwise described) on an Agilent Infinity 1260 II ultra-performance liquid chromatography system (LC). It was equipped with a diode-array UV-Vis detector (λ = 190–800 nm) coupled to a 6546A high-resolution quadrupole time-of-flight mass spectrometer (MS) with electrospray ionization in either positive or negative data-dependent MS/MS mode with collision energies set to 10, 20, and 40 eV per duty cycle. All organic solvents and mobile phase modifiers were Fisher Optima grade. Mobile phase solvents consisted of 18 MΩ water (solvent A) and 99% acetonitrile (aq, solvent B). Under acidic conditions both solvents were modified by adding 0.1% formic acid, and under neutral buffered conditions (pH = 7.0), water was modified by adding 10 mM ammonium acetate (NH4OAc) and solvent B was unmodified. For reversed-phase (RP) chromatography, we used an Agilent Poroshell EC-C18 column (2.1 × 50 mm, 1.9 μm). The RP gradient for both solvent compositions (0.4 mL/min) began with initial conditions of 0% B, hold for 2 min, ramped to 100% B (2–11 min), held 100% B (11–13 min), and reconditioned the column at 0% B (13.1–15 min). For aqueous normal-phase (ANP) chromatography, we used a MicroSolv Cogent 2.0 Diamond Hydride column (2.2 × 150 mm, 2.1 μm). The ANP gradient (0.4 mL/min) for both solvent compositions began with initial conditions of 90% B, ramped to 50% B (0–12 min), held 50% B (12–14 min), ramped to 50% B (14–15 min), and reconditioned the column at 90% B (15–25 min).
Results
Figure 3 shows the effect of UVC irradiation in ambient air on our lichen, on its algal photobiont Myrmecia israelensis grown in pure culture, and on the bacterium D. radiodurans. For the lichen, the effect was assessed in QY24 and in photobiont viability. Prior to irradiation, the QY24 was 0.75 ± 0.01, and the viability was 331 ± 45 CFUs per 0.2 mm2 thallus. After a 3-month exposure, a dose of about 430,000 kJ/m2, the QY24 was 0.44 ± 0.36, a 40% drop, and the viability was 213 ± 48 CFUs per 0.2 mm2 thallus, a 35% drop. In neither case was the drop statistically significant. The effect on monolayers of cells of Myrmecia israelensis and D. radiodurans was, in contrast, lethal, with all several hundred cells inactivated in just 60 s (Fig. 3). The photon flux through a cortex peel was measured using a photometer with 0.01 W/m2 sensitivity. The reading was zero.
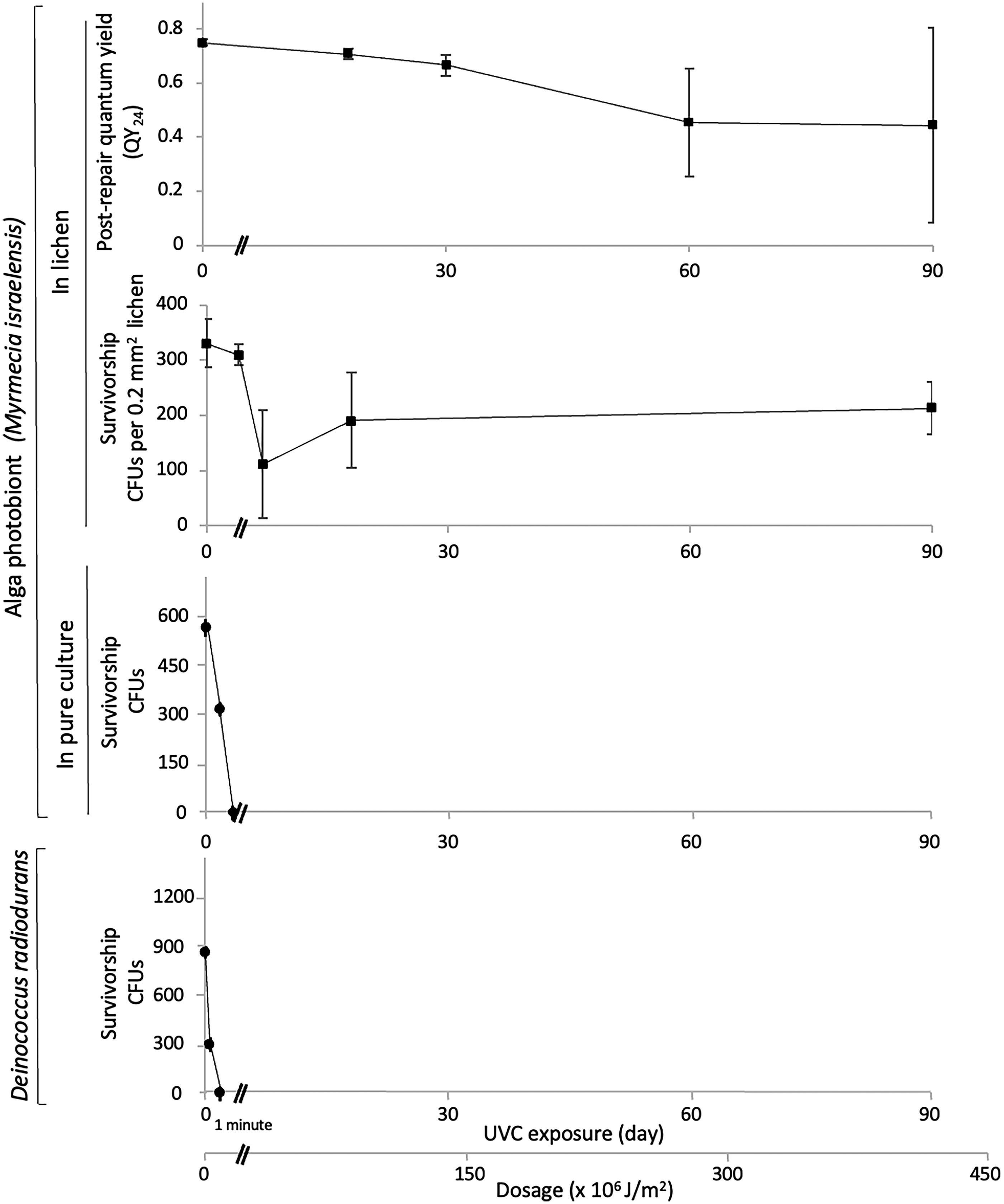
Effect of UVC irradiation on the lichen Clavascidium lacinulatum, on the algal photobiont Myrmecia israelensis in pure culture, and on the bacterium Deinococcus radiodurans in terms of QY24 (irrecoverable injury), viability, or both, showing that the lichen withstood the UVC assault for orders of magnitude longer than free-living algae and bacteria did (N = 3, error bar = SD).
LC-MS analyses of lichen extracts in methanol revealed a mildly acidic, carboxylated compound with the tentative molecular formula of C10H14N2O5 ([M + H]+ = 243.0977 Da, 0.63 ppm; [M-H]− = 241.0832 Da, 0.85 ppm, Supplementary Data S1). On-line ultraviolet-visible spectral analysis showed an absorption maximum of the compound at 254 nm (Fig. 4).
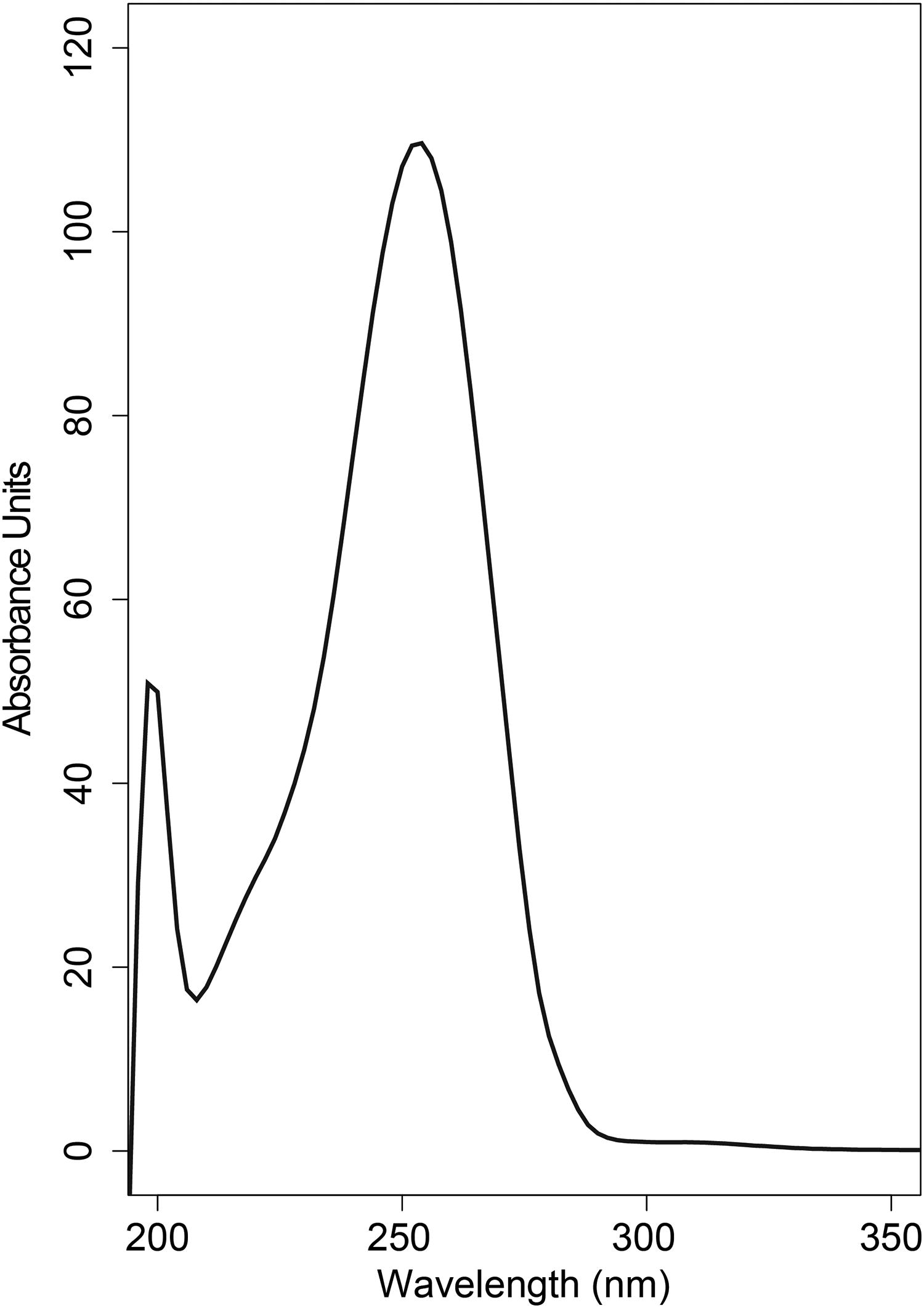
Solvent-subtracted UV-VIS absorption spectra of a secondary metabolite extract from the lichen Clavascidium lacinulatum.
A one-millimeter-thick mat of cultured algal cells was well short of the lichen cortex’s UVC screening efficacy. Prior to a 30-min exposure, the QY24, measured through the bottom of the mat, was 0.47 ± 0.19. After the exposure, the QY24 crashed to 0.07 ± 0.01 (Fig. 5). The photon flux through the mat was 4 W/m2, or 7% of the incident flux.
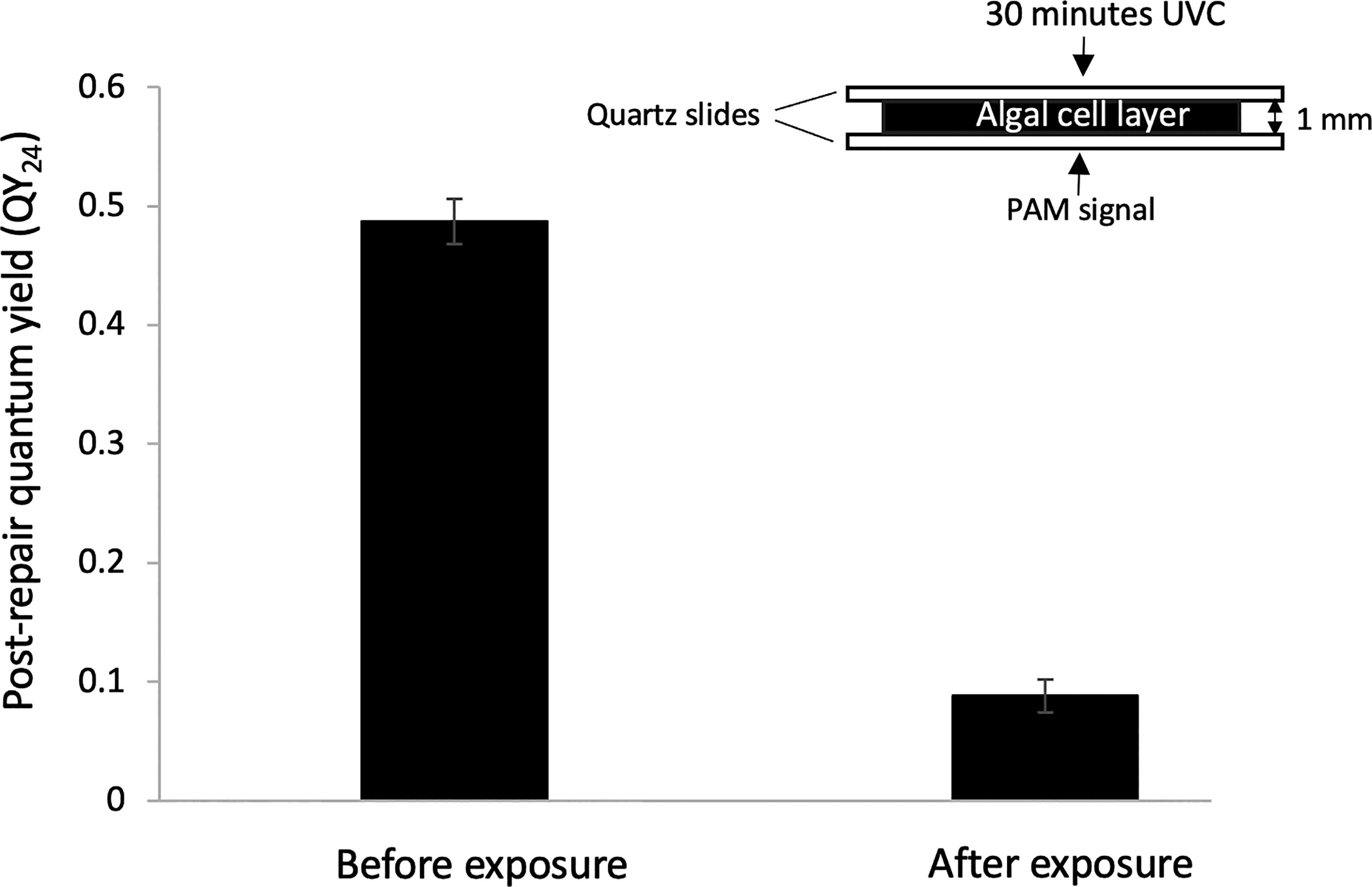
Lethal effect of a 30-min UVC exposure on a one-millimeter-thick mat of cultured algal cells, resulting in virtual elimination of photosynthetic capacity QY24 (p < 0.01, paired t-test, N = 3, error bar = SD), indicating that the lichen cortex would not be UV-opaque without the lichen acids.
UVA irradiation, despite being more penetrative and despite a longer exposure time used to compensate for its less energetic photons, was less injurious than UVC. Specifically, 72 h of UVC irradiation lowered the QY0.5 of the lichen, a measure of total injury, by 45%, from 0.71 ± 0.019 to 0.39 ± 0.08. Three times longer, 216 h of UVA irradiation at the same flux, in contrast, lowered the QY0.5 by a smaller amount of 28%, from 0.61 ± 0.02 to 0.44 ± 0.06 (Fig. 6).
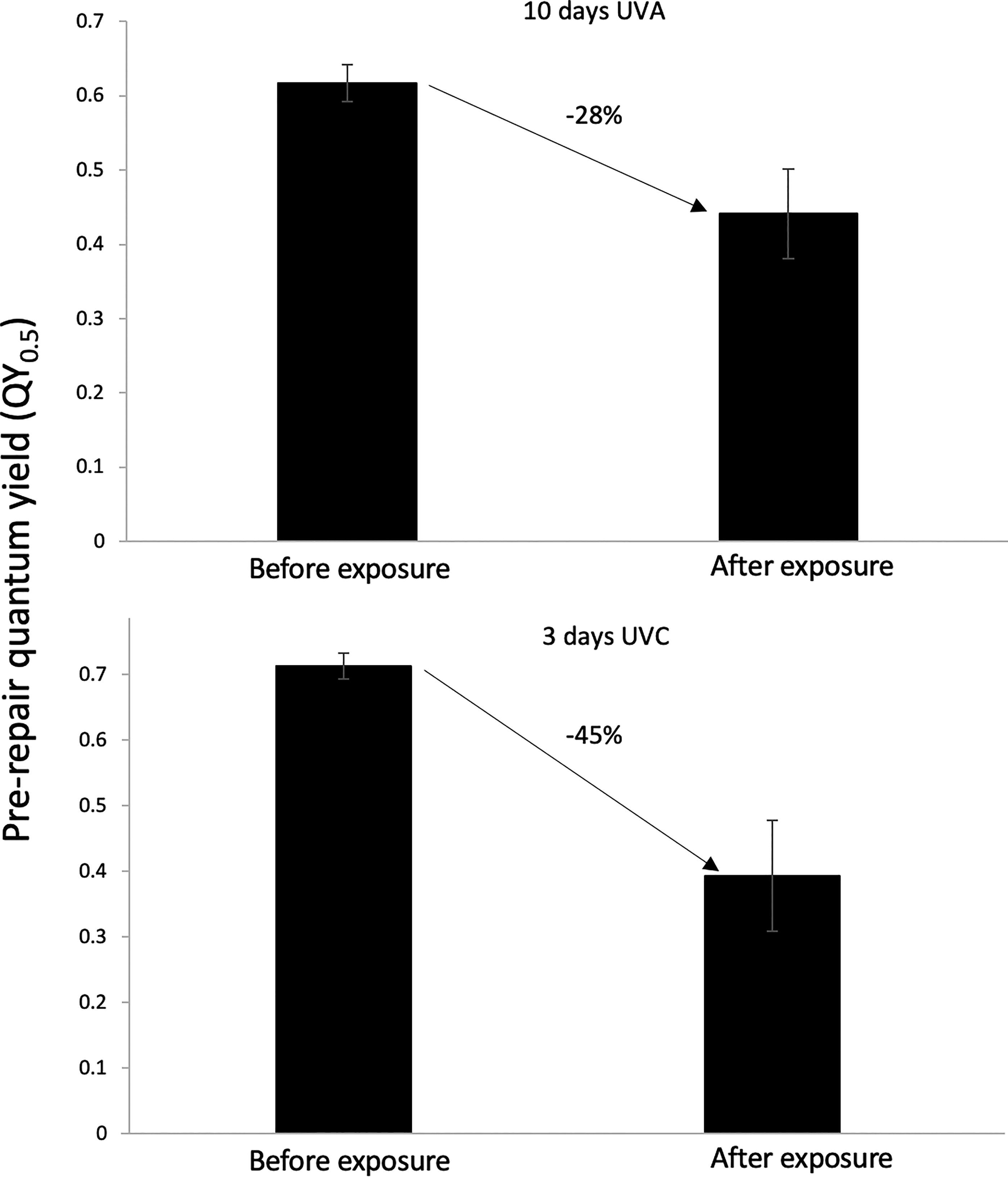
Effect of UVC versus UVA irradiation on the QY0.5 of the lichen Clavascidium lacinulatum, showing that UVA is less injurious despite being more penetrative and despite a 3-times longer exposure (p < 0.01, unpaired t-test, N = 5, error bar = SD).
UVC irradiation was less injurious to lichens housed in an anaerobic glovebox filled with 90% nitrogen, 5% carbon dioxide, and 5% hydrogen than to lichens in ambient air. Specifically, 432 h of UVC irradiation in ambient air lowered the QY0.5 by 97%, from 0.70 ± 0.05 to 0.02 ± 0.01. The same experiment but conducted inside the anaerobic glovebox lowered the QY0.5 by a smaller degree of 64%, from 0.69 ± 0.05 to 0.25 ± 0.01 (Fig. 7).
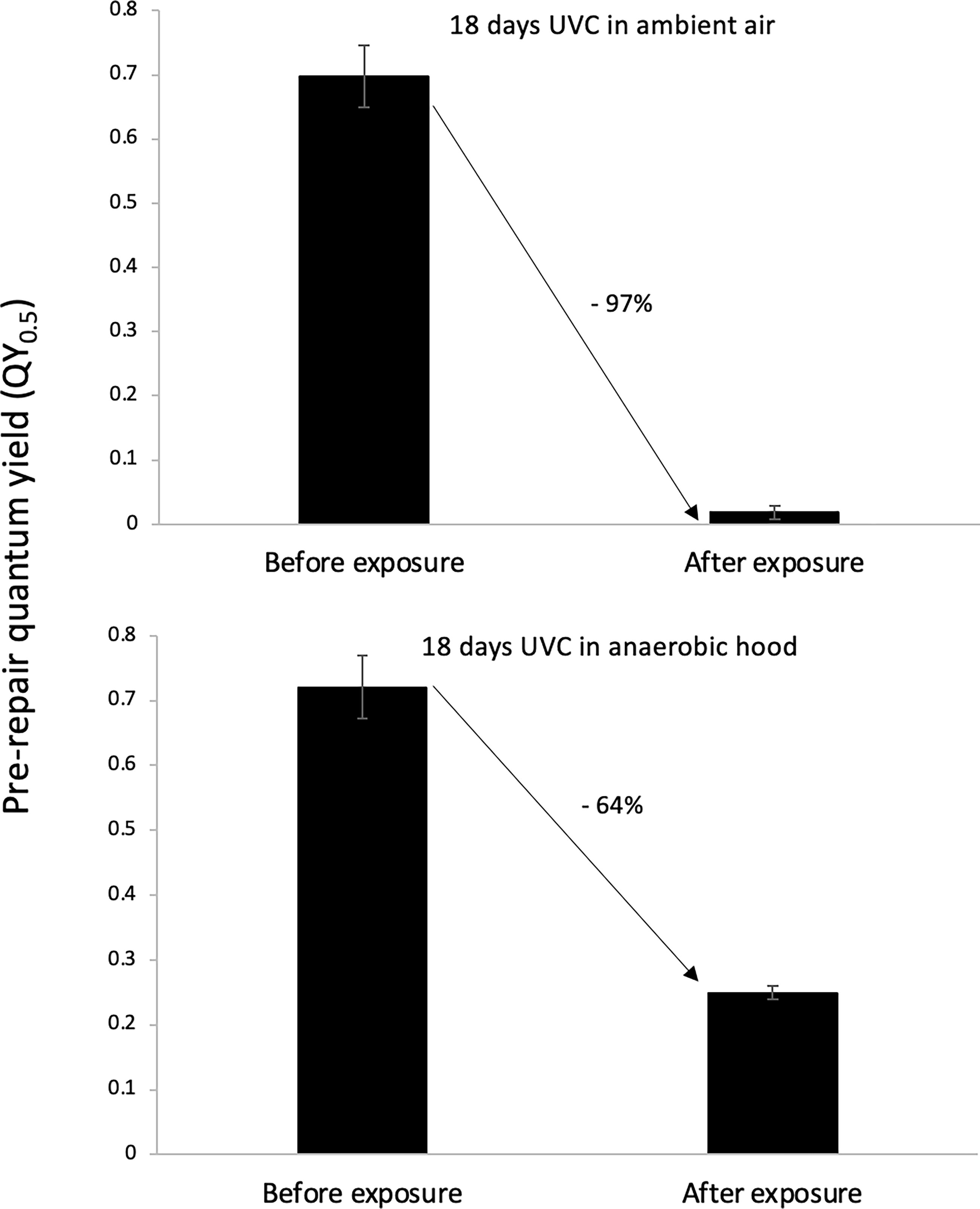
Effect of UVC irradiation on the QY0.5 of the lichen Clavascidium lacinulatum in ambient air versus in an anaerobic glovebox, showing that UVC is less injurious in the absence of O2 (p < 0.01, unpaired t-test, n = 3; error bar = SD).
The aromatic compound N, N-dimethyl-p-nitrosoaniline (pNDA) is a colorimetric •OH regent which, upon reacting with •OH, became more transmissive to photons at 440 nm (Kraljić and Trumbore, 1965). The change in absorbance occurred under UVC but not under UVA (Fig. 8).
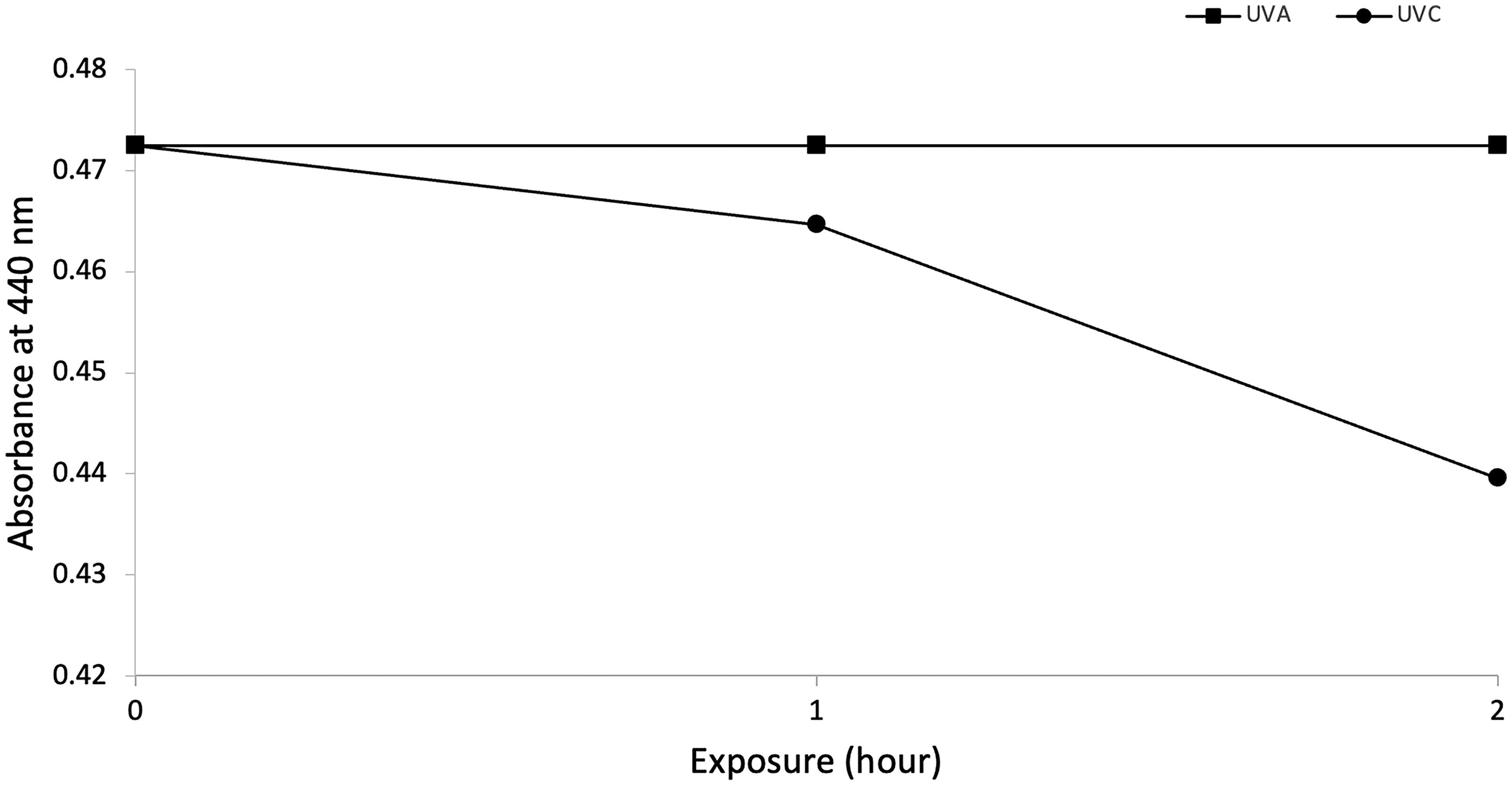
Photochemical •OH formation under UVC, but not UVA, in the presence of the model photostabilizer N, N-dimethyl-p-nitrosoaniline (pNDA).
At 55 W/m2, even a brief exposure to UVC, much less one that is ecologically relevant, is biocidal to unprotected microorganisms due to two injurious reactions. First, some cellular aromatic elements, such as tryptophan and riboflavin, are photosensitizers; they stimulate harmful ROS formations when photo-excited (Yoshimoto et al., 2020; Zhao et al., 2020). Second, and far more harmful, neighboring pyrimidine residues within a DNA strand undergo permanent bond rearrangements (Batista et al., 2009; Ravanat and Douki, 2016). Indeed, only seconds are required to render unprotected algae and bacteria nonviable (Fig. 3).
However, the cortex of our lichen is UV-opaque. Consequently, after a 3-month exposure, half of its algal content remained viable and photosynthetically intact (Fig. 3). The cortex contains two different sources of UV-attenuating aromatic compounds: the cellular components within the fungal hyphae and the lichen acids without. We suggest that the latter are photostabilizers. Indeed, the 254 nm absorption maximum of one of them, with the tentative molecular formula of C10H14N2O5, supports this conjecture (Fig. 4 and Supplementary Data). We also suggest that the fungal hyphae in the cortex are little more than a scaffold that holds the lichen acids in place. Indeed, an algal mat prepared from pure culture and devoid of lichen acids let through a significant amount of UVC photons. Its UVC screening efficacy did not even approach that of the cortex (Fig. 5). The proposed photostabilizing function of lichen acids is also supported by early studies. Specifically, de Vera et al. (2002) showed that the UVC tolerance in the crustose lichens Fulgensia bracteata and Xanthoria elegans decreased when their upper cortex was excised. Sánchez et al. (2014) found that a rinse in acetone, a lichen acid solvent, diminished the UVC tolerance of the fruticose lichen Circinaria gyrosa and of the crustose lichen Rhizocarpon geographicum. In a rare deviation, de La Torre et al. (2010) found that excising the cortex of R. geographicum had little effect on its tolerance for outer space in Earth orbit. This may be because outer space is a complex environment where UV is but one of many stressors.
Lichen acids are powerful antioxidants as well (Fernández-Moriano et al., 2016; Luo et al., 2009; Thadhani et al., 2011). In their presence, short-lived ROS, such as •OH radicals, are unlikely to diffuse far from the sites of their formation within the cortex before they are quenched. Photochemical ozone is far more stable, and it may form in the cortex given the presence of O2 and N2O in urban air and given the presence of photosensitizers in the fungal cells. UVA irradiation, let alone UVC, is a known source of photochemical ozone under such conditions (Calfapietra et al., 2013; Carter, 1994; Seinfeld and Pandis, 2016). Hence, UVA, which is photochemically less productive, is less injurious than UVC (Fig. 6), and UVC is less injurious to lichens in the anaerobic glovebox than to lichens in ambient air (Fig. 7). To the extent that pNDA is a photostabilizer, UVA is indeed photochemically less productive than UVC (Fig. 8). The fact that the lichen in the anaerobic glovebox was still injured suggests that stable non-oxygen radicals, such as hydrogen radicals, formed under our anaerobic gas environment. A photostabilized cortex is consistent with the challenges that the lichen faces in its natural habitat. The surface of a desert is exposed to mineral-adsorbed ROS, photochemical ozone, and solar UVA (Georgiou et al., 2015; Navarro-González et al., 2003; VanCuren, 2015).
While lichens come equipped with UV-screening photostabilizers, the ability to synthesize them is the result of evolutionary adaptation to dehydration-induced oxidative stress, not UV (Kranner et al., 2005, 2008; Kranner and Lutzoni, 2018; Vondrák et al., 2023). Indeed, lichen symbiosis is an ongoing evolutionary process that occurs on dim wood surfaces, such as the underside of felled logs on the floor of a forest, where algae and fungi grow next to one another (Vondrák et al., 2023). Like all aerobic microorganisms, algae and fungi are equipped with antioxidant enzymes, notably superoxide oxidase and catalase. However, the enzymatic defense is suspended in a desiccated organism. Hence, desiccation-tolerant microorganisms, including lichenized algae and fungi, are equipped with antioxidants as well as antioxidant enzymes (Farrar, 1988; Keunen et al., 2013). Lichen symbiosis enables a third layer of defense, a macroscopic thallus in which cells and tissues are coated in lichen acids to minimize exposure to atmospheric ROS (Mahmud et al., 2024). Thus, from an evolutionary perspective, the UV-screening property of lichen acids is secondary. It does not come into play until lichens enter open and exposed habitats.
Scytonemin, the photostabilizer in desert cyanobacteria and desert cyanolichens, is a genuine UV-screening pigment (Dillon and Castenholz, 1999; Garcia-Pichel, 1998; Garcia-Pichel and Castenholz, 1991). This is because Archean Earth, where its biosynthesis evolved, was a reduced environment with no oxidative stress. Much of the iron in today’s iron beds was reduced and dissolved in seawater then, which resulted in a high enough concentration to screen solar UVC entirely and screen solar UVA partially from the photic zone (Garcia-Pichel, 1998). The antioxidant property of scytonemin, therefore, did not come into play in cyanobacterial ecology until after free oxygen appeared in the atmosphere at around 2.0 billion years ago (Hidalgo et al., 1994; Matsui et al., 2012; Ručová et al., 2023).
Intracellular photostabilizers such as carotenoids are present in bacteria, including D. radiodurans (Frank et al., 2002; Oren and Gunde-Cimerman, 2007; Taylor, 1984). Studies of D. radiodurans suggest that such compounds help protect the cell against acute ionizing gamma radiation challenges (Ji, 2010; Lemee et al., 1997; Tian et al., 2009; Zhang et al., 2007). Yet they cannot shield the cell from UVC (Fig. 2). This suggests that photostabilizers are not an effective UV screen unless they are placed in the cell envelope or, in the case of lichens, in the cortex to allow for high density and a longer light path.
Given the results of our studies, it is not unreasonable to suggest that the lichens in the Mojave Desert would continue to persist without the stratospheric ozone shield. In the case of the cyano-lichens, which are essentially colonial cyanobacteria anchored to soil and rock by fungal hyphae, this is not a hypothetical scenario. The scenario, in fact, occurred on land about 2.6 billion years ago (Watanabe et al., 2000) and in the ocean following the Great Oxidation Event, around 2.0 billion years ago, when iron was all but stripped from seawater due to oxygenic photosynthesis (Anbar et al., 2007; Bekker et al., 2004; Poulton et al., 2021).
In conclusion, UVC-intensive exoplanets may not be considered uninhabitable. Rather, they may be teeming with colonial microorganisms that, like the lichens in the Mojave Desert (Fig. 1), are “tanned” and virtually immune to UVC stress.
Footnotes
Acknowledgment
The authors thank Dr. Ali Saidi-Mehrabad for his assistance with metagenomic data analysis, Dr. Brian Hedlund for access to his anaerobic glovebox, and Dr. Ravi Kopparapu for his comments on an early draft of our article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Funding for this work was provided by the NASA Habitable World Program (Grant# 18-HW18_2-0051) and by the NASA EPSCoR Office Rapid Response Research Program (Grant# NV-80NSSC23M0154).
Supplementary Material
Supplementary Data
Associate Editor: Petra Rettberg
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
