Abstract
The notion of liquid water beneath the ice layer at the south polar layered deposits (SPLD) of Mars is an interesting possibility given the implications for astrobiology and possible human habitation. A body of liquid water located at a depth of 1.5 km has been inferred from radar data in the South Polar Cap. However, the high temperatures that would facilitate the existence of liquid water or brine at that depth are not consistent with estimations of heat flow that are based on the lithosphere’s flexure. Attempts to reconcile both issues have been inconclusive or otherwise unsuccessful. Here, we analyze the possible role(s) of subsurface ammonia and/or methanol in maintaining water in a liquid state at subsurface temperatures that are compatible with the lithosphere strength. Our results indicate that the presence of these compounds at the base of the SPLD can reconcile the existence of liquid water with previous estimations of surface heat flow.
Introduction
Data obtained from the radar subsurface sounder aboard the Mars Express spacecraft that orbits Mars reveals bright subsurface reflections in the Planum Australe region of the planet. These reflections cover an area that is about 20-km wide and is located at 193°E, 81°S (Fig. 1). The data are interpreted as an extant body of liquid water located beneath the south polar layered deposits (SPLD) at a depth of 1.5 km (Orosei et al., 2018). The existence of liquid water on Mars is particularly interesting because it might facilitate the existence of life. Even at temperatures too low to allow the function of terrestrial microbes, aqueous milieu can apparently preserve cells in a viable condition over vast periods, likely hundreds of millions of years (Hallsworth, 2022). Furthermore, bodies of liquid water would be valuable as a resource to enable human habitation (Seedhouse, 2009; Rummel et al., 2014).
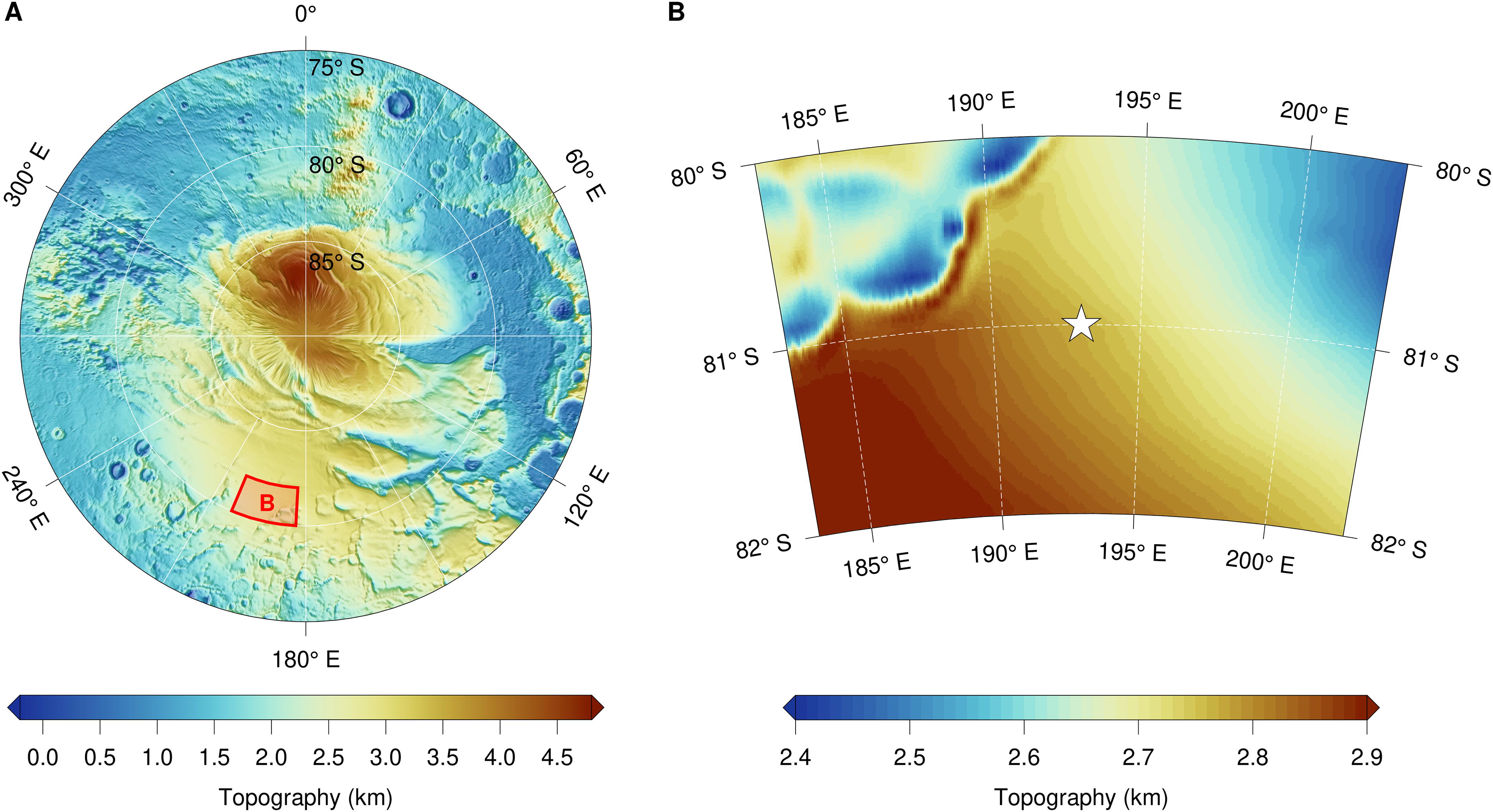
Location of the putative liquid water. Mars Orbiter Laser Altimeter (MOLA) topographic maps of the area considered in the current study (https://astrogeology.usgs.gov/search/map/Mars/GlobalSurveyor/MOLA/Mars_MGS_MOLA_ClrShade_merge_global_463m). (
Locally elevated heat flow contributes significantly to the formation of subglacial liquid water beneath the West Antarctic Ice Sheet (Fisher et al., 2015). However, the inference of liquid water beneath the SPLD, based on the interpretation of the bright reflections, is controversial because this liquid state would require temperatures that imply local surface heat flows higher than those predicted from lithosphere strength or those predicted using thermal history models (e.g., Sori and Bramson, 2019). There are alternative interpretations to liquid water that explain the bright reflections: electrically conductive minerals (Bierson et al., 2021), constructive interference of CO2 layers (Lalich et al., 2022), and hydrated clays (Smith et al., 2021), but each of these explanations has been disputed (Mattei et al., 2022; Lauro et al., 2022; Orosei et al., 2022).
Some authors have suggested the presence of salts that depress the melting point of water to reconcile the evidence suggesting the existence of liquid water with the estimations of the surface heat flow (e.g., Sori and Bramson, 2019; Mattei et al., 2022), as occurs in the subglacial lake complex of the Devon Ice Cap in the Canadian Arctic, where hypersalinity allows the existence of liquid water at subzero temperatures (Rutishauser et al., 2018).
Perchlorates have been favored as the most-likely salts because they are widespread on Mars, including the North Polar Cap (Hecht et al., 2009). However, surface heat flows calculated assuming the existence of perchlorates within the SPLD are still higher than expected (Sori and Bramson, 2019). It has been argued that the eutectic point of the SPLD ice can be low enough to provide the expected heat flow if diverse perchlorate salts are concomitantly present in the ice layer (Nair and Unnikrishnan, 2020; Mattei et al., 2022). In such a case, the presence of magnesium perchlorate and sodium perchlorate in water lowers the melting point temperature to 180 K (Nair and Unnikrishnan, 2020). However, the amounts of perchlorates needed to reduce the melting point to values compatible with the predicted surface heat flow are high for binary and ternary solutions (one or two salts in water): ∼20–50% by mass (Pestova et al., 2005; Nair and Unnikrishnan, 2020).
There are reasons to think that, at depth, the concentration of perchlorates at the SLPD is relatively low. First, the source for the perchlorates (from chlorine) is thought to be linked to surface radiation and/or atmospheric processes (Catling et al., 2010). Second, perchlorates are not found at high levels in martian meteorites that sample deeper materials (<ppm levels), and third, a concentration of perchlorates of 20–50% by mass would involve an excess of mass at the SPLD (Ojha et al., 2021). Furthermore, even if the required proportion of perchlorates exists, the thermal analysis of the region where the putative liquid water is located indicates that the overlying ice layer should be extremely porous to permit the basal melting of a mixture of perchlorates and water ice at the heat flows predicted from lithospheric strength (Egea-González et al., 2022). This is difficult to reconcile with the high density of the SPLD and with ice compaction at depth.
A possible analogy to explain the origin of the putative liquid water on Mars can be found in Lake Vostok, Antarctica. In this case, it has been proposed that the lake existed before glaciation and remained liquid beneath the ice over time (Duxbury et al., 2001). Similarly, one option for the liquid water on Mars is that it was formed in the planet’s past, when conditions were warmer, and has remained in a metastable liquid state since then (Stillman et al., 2021). However, it is still unclear if supercooled perchlorate–H2O solutions are stable over geologic timescales. (Ojha et al., 2021). Thus, the presence of perchlorates within the SPLD cannot reconcile the existence of liquid water with the heat flow estimations. Hence, there is an ongoing conflict between evidence for liquid under the SPLD, the thermal requirements to facilitate and stabilize this liquid state, and the thermal properties of the layer above the putative liquid body.
Here, we explore the possible presence of non-salt antifreezes in the putative liquid body, which could allow the liquid state to persist at low temperatures of 170–180 K (−103 to −93°C). We show that the antifreezes ammonia and methanol would eliminate the conflict between this liquid state and the heat flow estimates. We also discuss the implications of the existence of ammonia and methanol for the habitability of the liquid water body or lake under the SPLD.
Surface heat flow from the effective elastic thickness of the lithosphere
For reference purposes, we have re-evaluated the background (i.e., regional) heat flow at the South Pole from the effective elastic thickness of the lithosphere (T e), following the procedure described by Ruiz et al. (2010, 2011). The lithosphere below the South Pole shows small deflection under the weight of the polar ice cap, which indicates a cold lithosphere. T e is not easy to estimate for this region due to the small flexure. The minimum acceptable value for T e would be 150 km according to Broquet et al. (2021), which is comparable to (or lower than) the minimum T e values obtained by other authors (Plaut et al., 2007; Wieczorek, 2008; Ding et al., 2019). Thus, because of the inverse relation between lithosphere strength and heat flow, we use T e = 150 km in order to derive an upper limit for the surface heat flow in the South Pole. For comparison, we calculated heat flows for best-fit T e values in the range between 255 and 360 km (Ding et al., 2019; Broquet et al., 2021), whereas accurate heat flow lower limits cannot be obtained in this region due to the limited flexure.
We assume a mean crustal thickness of 70 km for the south polar region, based on the updated crustal thickness model by Wieczorek et al. (2022). Because the thickness of the crust is much lower than the effective elastic thickness of the lithosphere, the crust must be mechanically welded to the mantle lithosphere. Given this and the absence of clear lithosphere flexure, it is sufficient to consider the mechanical properties of the mantle lithosphere in the calculations. Thus, we assumed zero plate curvature, rheological parameters for olivine from Zhao et al. (2009), and a strain rate of 10−14 s−1 appropriate for the temporal scale of polar cap loading (Phillips et al., 2008).
The radioactive crustal heat production is taken to be the mean estimated for Mars from Mars Odyssey Gamma Ray Spectrometer measurements (4.89 × 10−11 W/kg; Hahn et al., 2011), whereas for the mantle lithosphere we assume 0.1 times the crustal heat production value (e.g., Taylor and McLennan, 2009). Densities of 2900 and 3500 kg/m3 are used, respectively, for the crust and the mantle lithosphere. The polar cap thickness is taken as 3 km (Plaut et al., 2007), and it is assumed to be composed of 85% water ice and 15% basaltic dust by volume (Zuber et al., 2007; Wieczorek, 2008; Broquet et al., 2021).
For the thermal conductivity of the crust, we consider two possibilities, 2 and 2.5 W/(m·K); the first value would be appropriate for a basaltic crust, whereas the second could be considered an upper limit if the crust includes significant amounts of granite-type materials (see Jiménez-Díaz et al., 2021 for a review). For the mantle lithosphere, we use a temperature-dependent thermal conductivity (see Ruiz et al., 2011). The thermal conductivity of the polar cap is estimated from a volumetric mean (geometrically pondered; see Beardsmore and Cull, 2001; Egea-González et al., 2022) between those of water ice (which is temperature-dependent; Fukusako, 1990) and basalt (the ice layer properties are described in detail in the next section). Finally, the surface temperature is taken as 155 K (−118°C) (Wieczorek, 2008); note that for the calculation of regional heat flow we consider values representative for the region loaded by the polar cap, not for the lake position. For further discussion of lithosphere parameters, see Ruiz et al. (2011) and Egea-González et al. (2017).
Surface heat flow and temperature at the base of the SPLD
To enable an analysis of the dependence of surface heat flows on the temperature at the depth of the putative liquid water, we have solved the one-dimensional steady-state heat conduction equation for the overlying ice layer. The heat conduction equation resolution requires specification of the thermal properties and the boundary conditions in the studied area.
The putative liquid water is located at a depth of 1.5 km under the SPLD, which is thought to be formed by a mixture of dust and water ice with a 1-m-thick layer of CO2 ice on top (Aharonson et al., 2004; Litvak et al., 2007). The thermal effect of the thin layer of CO2 ice is negligible in a layer with a total thickness of 1.5 km, so we did not include CO2 ice in our calculations. The thermal conductivity of the layer formed by dust and water ice is a required parameter to solve the heat conduction equation. We use a temperature-dependent thermal conductivity for the water ice given by the equation (Fukusako, 1990): k ice = 1.16 [1.91 − 8.66 · 10−3 T + 2.97 · 10−5 T 2], where k ice is the water ice conductivity in W/(m·K), and T is the temperature of the ice expressed in °C. For the dust component, we assume a thermal conductivity for basalt rocks of 2 W/(m·K) (Beardsmore and Cull, 2001), and we obtain the thermal conductivity of the mixture of water ice and dust as a geometric mean of both components assuming a dust proportion of 15% by volume (Zuber, et al., 2007; Wieczorek, 2008).
The surface and basal temperatures are boundary conditions for the heat conduction equation. The annual average surface temperature at the location of the putative subsurface liquid body is constrained to be ∼162 K (i.e., about −111°C) (Sori and Bramson, 2019), and then we calculated surface heat flow for a range of basal temperatures between 165 and 190 K (−108 and −83°C) to identify those that are consistent with the predicted surface heat flows. The heat conduction equation has been solved through the Simscale cloud-based platform (https://www.simscale.com/), which allows complex simulations without the need for large computer resources and has a friendly graphical user interface. We show the surface heat flow (F s) obtained from the basal temperature range in Fig. 2.
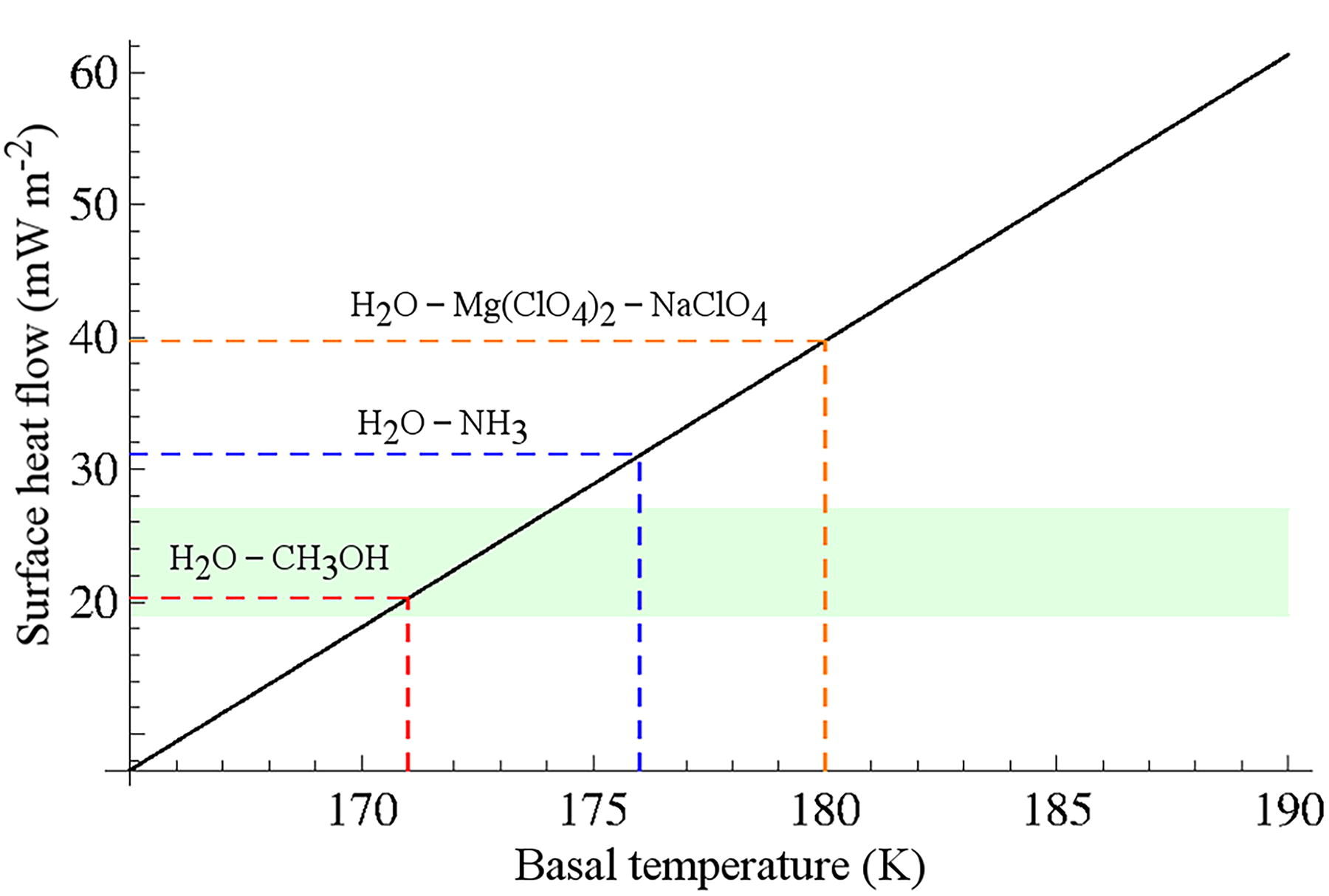
Surface heat flow vs. basal temperature at the location of the putative liquid water. Surface heat flow obtained after solving the heat equation for the SPLD ice layer and considering a range of temperatures at the depth of the putative liquid water between 165 and 190 K (black line). The green shaded area indicates the surface heat flow interval calculated from the elastic thickness of the lithosphere (T e). The dashed lines correspond to the freezing point temperature of an ammonia–water ice mixture (blue), methanol–water ice mixture (red), and for a mixture of sodium perchlorate and magnesium perchlorate in water (orange) (Nair and Unnikrishnan, 2020).
Note that the resolution of the heat conduction equation depends on several free parameters: the basal and surface temperatures, the thickness of the dust-water ice layer, the thermal conductivity of the dust, the thermal conductivity of the water ice, and the proportion of dust in the dust-water ice layer. The values selected for these parameters are based on results obtained in previous work and are widely accepted for Mars.
The background heat flow at the south polar region
Previous studies have used the relation between the temperature profile and the mechanical strength of the lithosphere to calculate the upper limits to the present-day heat flow of Mars from the effective elastic thickness of the lithosphere. The surface heat flows proposed following this approach are upper limits because the small flexure in this region introduces uncertainty in the deflection calculation (Phillips et al., 2008; Broquet et al., 2021). The maximum surface heat flow estimated in previous work ranges between 23 and 32 mW/m2 (Ruiz et al., 2010; Parro et al., 2017; Broquet et al., 2021).
For comparison purposes, we have re-evaluated the regional heat flow at the south polar region from the effective elastic thickness of the lithosphere (T e) following Section 2. We have calculated an upper limit for the surface heat flow at the south polar region using a minimum value of T e = 150 km, and we have also calculated heat flows for T e values in the range 255 to 360 km, which are the best-fit T e values according to Ding et al. (2019) and Broquet et al. (2021). We summarize our results in Table 1. The obtained upper limit for the heat flow is 27 mW/m2, although for T e best-fit values of the heat flow would be between 19 and 22 mW/m2. We therefore consider a range between 19 and 27 mW/m2 for the background heat flow in the south polar region of Mars. This range is consistent with the heat flow upper limit of 23.5 mW/m2 proposed in previous work (Parro et al., 2017; Broquet et al., 2021).
Surface Heat Flow Obtained from the Effective Elastic Thickness of the Lithosphere
Surface Heat Flow Obtained from the Effective Elastic Thickness of the Lithosphere
Surface heat flows were calculated for the south polar region from estimates of the effective elastic thickness of the lithosphere (T e). Lower and upper values of the heat flow range were calculated for thermal conductivities of 2 and 2.5 W/(m·K), corresponding, respectively, to a basaltic crust and to a crust including a substantial felsic component. See Section 2 for details.
These values are much lower than the lower limit of ∼70 mW/m2 that was deduced in previous work that considers perchlorates as antifreeze for maintaining a liquid body within the polar cap (Sori and Bramson, 2019). Even in optimal conditions, it is very difficult to obtain a heat flow lower than 35 mW/m2 being compatible with a liquid layer (Egea-González et al., 2022).
To identify the range of temperatures at the base of the SPLD that are compatible with the surface heat flow calculated from flexural models, we have calculated the surface heat flows (F s) that correspond with basal temperatures between 165 and 190 K (−103 and −83°C). The surface heat flow (F s) obtained from this basal temperature range is shown in Fig. 2.
Figure 2 indicates that the low surface heat flows necessary to account for the limited flexion of the lithosphere under the South Polar Cap require a basal temperature well below 180 K (−93°C), which is much lower than the melting points of mixtures formed by perchlorates and water ice. Thus, the apparent presence of liquid water under the SPLD suggests the potential presence of one or more substances that act as antifreeze, such as ammonia or methanol.
The presence of ammonia reduces the solidus of the ammonia–water system to 176 K (−97°C) even when the concentration of ammonia is low (Kargel, 1992). Table 2 summarizes possible candidates to reduce the SPLD ice layer melting point. If we assume that the ammonia is located at the base of the ice layer, the surface heat flow required for melting is 31.2 mW/m2, which is compatible with a surface heat flow of 19–27 mW/m2 if we take into account some realistic porosity in the ice layer. In the case of a mixture of methanol and water ice, the melting point decreases to 171 K (−102°C) for a wide range of methanol concentrations (Table 2). The heat conduction equation resolution provides a surface heat flow of 20.3 mW/m2 for a basal temperature of 171 K. These results for ammonia and methanol are consistent with the low surface heat flow that is predicted using flexural models for this region and may indicate the presence of compounds under the SPLD that are not usually considered at the surface of Mars.
Solidus Temperature for Candidates to Reduce the SPLD Ice Layer Freezing Point
Solidus Temperature for Candidates to Reduce the SPLD Ice Layer Freezing Point
Solidus temperature and required concentrations for water–ammonia and water–methanol systems together with binary and ternary solutions formed by the dissolution in water of perchlorates of sodium, magnesium, and calcium (NaClO4, Mg((ClO4)2), Ca((ClO4)2), respectively).
The influence of the overlayer pressure on the reduction of the dust-water ice melting point is negligible. The layer above the liquid water has a thickness of 1.5 km and a density of 1220 kg/m3. Taking martian gravity into account, the resulting basal pressure is calculated to be 6.8 MPa. This pressure is too low to cause a substantial change in the melting point.
Basal temperatures that are compatible with the expected range of surface heat flow increase when the porosity of the layer is taken into account. However, previous work (Egea-González et al., 2022) has studied the effect of porosity on the temperature at the base of the ice layer and shows that an extremely porous layer is required to obtain surface heat flows less than 35 mW/m2 with basal temperatures higher than 180 K. Such a porous layer is not compatible with the high density of the SPLD, which points to a compact ice layer with low porosity. Thus, the effect of realistic porosity in our result will not modify our conclusions.
Ammonia and methanol as antifreeze on Mars
The high effective elastic thickness of the lithosphere under the South Polar Cap is consistent with a cold lithosphere with upper surface heat flow limits ranging between 19 and 27 mW/m2. The temperature at the base of the SPLD should be much lower than 180 K (−93°C) to be compatible with these surface heat flow estimations. Therefore, the presence of substances that greatly reduce the melting point of water ice is required to render the layer of liquid water plausible. In particular, ammonia- and methanol-rich ices have melting points that are consistent with the predicted surface heat flows.
The notion of ammonia–water mixtures on Mars is one that has not been previously considered. There have been no definitive identifications of ammonia on the surface and atmosphere of Mars. Data from the ExoMars Trace Gas Orbiter (TGO) suggest that the concentration of atmospheric ammonia is likely below 14 ppbv (Trokhimovskiy et al., 2024). This absence of ammonia could be because ultraviolet radiation converts atmospheric ammonia to N2 (Atreya, 2010).
Derivatized ammonia has been detected on sand from the Bagnold Dunes on Mars’ Gale crater through the Sample Analysis at Mars (SAM) instrument, but it is not clear if it has an endogenic origin (Millan et al., 2021). However, we do know that ammonia plays an important role in the presence of liquid water in many icy bodies and is abundant in the solar system (e.g., Desch et al., 2009). There are also suggestions of ammonia as a primordial molecule on Mars (Atreya, 2010). The analysis of martian meteorites suggests that ammonia has been preserved in the subsurface since the Noachian period (Koike et al., 2020), where it has been protected from ultraviolet radiation and cosmic rays. Furthermore, there may be ongoing subsurface sources (biotic and abiotic) for ammonia (Summers et al., 2012; Barge et al., 2020). Thus, the hypothesis of the presence of ammonia is not implausible. In this sense, if there are ammonia seeps under the ice, then the ice slab could protect it from ultraviolet radiation, and ammonia could accumulate. In the case of leaks from the subsurface into the atmosphere, ammonia would be quickly destroyed and be essentially undetectable. Moreover, the presence of ammonia would be a source of isotopically primordial N and could help explain the 14N/15N in the martian atmosphere (Manning et al., 2008).
Regarding methanol, it has been proposed as a primordial molecule on Titan (Deschamps et al., 2010), and it could be also present in the early martian environment (Tang et al., 2006). Furthermore, methanol could be produced on Mars from methane (e.g., Wong et al., 2004). Methane has been detected in small abundances in the martian atmosphere (e.g., Yung et al., 2018). Measurements from the Curiosity rover reveal a significant seasonal variation in atmospheric methane levels, with peaks occurring during the martian summer (Webster et al., 2015). This was further supported by independent observations from the Mars Express orbiter, which detected 15.5 ± 2.5 ppbv of methane over the Gale crater just 1 day after Curiosity recorded a peak of 5.78 ± 2.27 ppbv (Giuranna et al., 2019). In contrast, the TGO has not detected methane, setting an upper limit of 0.05 ppbv in the martian atmosphere (Korablev et al., 2019; Knutsen et al., 2021). Methane on Mars appears sporadic, and it is likely being replenished by sources from the planet’s interior (Viúdez-Moreiras et al., 2020). Perhaps the supply of methane needed for the potential production of methanol could occur directly below the surface and even in the liquid body. As in the case of ammonia, the low proportion of methanol that is required to reduce the melting point, together with the short photochemical lifetime in the martian atmosphere, roughly 74 days (Wong et al., 2004), could complicate its detection in the case of leaks to the atmosphere.
Moreover, the presence of ammonia or methanol within the liquid body aligns with the findings of Orosei et al. (2018) and Lauro et al. (2020). Their studies identified several subglacial water bodies characterized by exceptionally high relative dielectric permittivity values, with one region reaching up to 40. Given the estimated cryogenic temperatures of these environments, the proposed solutes, ammonia and methanol, could achieve such high relative dielectric permittivity.
Implications and perspectives for habitability
Studies conducted in the Antarctic ice sheet have demonstrated the presence of viable microorganisms under extreme conditions, which suggests the possibility of life on other celestial bodies with similar characteristics (Abyzov et al., 2006). Ammonia and some ammonium salts entropically destabilize the cell membranes and biomacromolecules of terrestrial microbes, a property known as chaotropicity (de Lima Alves et al., 2015; Cray et al., 2013; Ball and Hallsworth, 2015). In contrast, methanol is known to be biophilic because in this regard its behavior is close to that of water, so it is relatively neutral (Cray et al., 2013; Cray et al., 2015). This said, chaotropic substances at subzero temperatures that rigidify biomacromolecular structures can facilitate the flexibility of cellular macromolecules and so facilitate, rather than inhibit, the biotic activities of microbial systems (Cray et al., 2015; Chin et al., 2010).
The concept of habitability is based on current knowledge of life on Earth (Cockell et al., 2024). Although some terrestrial microbes are known to produce methane, a mechanism to explain the origin of these compounds under the SPLD is yet to be determined. Nevertheless, the perfect match between the thermal characteristics of these systems and the predicted thermal state of the South Polar Cap seems remarkable and should be considered as a possible solution to the thermal problem for the presence of liquid water under the SPLD.
Conclusions
The results presented in this study indicate that the existence of liquid water below the South Polar Cap at the temperatures predicted from the lithosphere strength could suggest the presence of compounds under the ice slab that are rarely considered on Mars. Whereas data from geochemical analyses of martian surface materials do not indicate the presence of methanol or ammonia, their presence in the subsurface of the SPLD is plausible. These compounds reduce the melting point of the ice to values of 171 and 176 K. These temperatures lead to surface heat flow of 31.2 and 20.3 mW/m2 for ammonia- and methanol-rich ice, respectively. The obtained surface heat flows agree with the range of 19–27 mW/m2 that is obtained from the limited flexion of the lithosphere. In addition, the presence of ammonia and methanol is also consistent with the relative dielectric permittivity reported in the area (Orosei et al., 2018; Lauro et al., 2020). Although a mechanism to explain the origin of these compounds under the SPLD is yet to be determined, the perfect match between the thermal characteristics of these systems and the predicted thermal state of the South Polar Cap is remarkable and should be considered as a possible solution for the thermal problem that supposes the presence of liquid water under the SPLD.
Footnotes
Acknowledgments
The authors are thankful to an editor and two anonymous reviewers for their constructive and helpful comments. C.P.M. acknowledges support from the NASA Astrobiology Program. I.E.-G., J.R., and A.J.-D. are supported by Project PID2022-140686NB-I00 from the Spanish Ministry of Science and Universities (I.E.-G., J.R., and A.J.-D.).
Authors’ Contributions
Conceptualization: C.P.M. and J.R. Methodology: I.E.-G., J.R., and A.J.-D. Investigation: I.E.-G., J.R., C.P.M., and J.E.H. Visualization: I.E.-G., J.R., J.E.H., C.P.M., and A.J.-D. Supervision: J.E.H., J.R., C.P.M., A.J.-D. Writing—original draft: I.E.-G. and J.R. Writing—review and editing: J.E.H., J.R., C.P.M., and A.J.-D.
Data and Materials Availability
Author Disclosure Statement
The authors declare that they have no conflicts of interest.
Funding Information
C.P.M. received funding from the NASA Astrobiology Program. I.E.-G., J.R., and A.J.-D. received funding from the Spanish Ministry of Science and Universities (Project PID2022-140686NB-I00). I.E.-G. received funding from Plan Propio–UCA 2025-2027.
Associate Editor: Sherry L. Cady
