Abstract
Earth is expected to have acquired a reduced proto-atmosphere enriched in H2 and CH4 through the accretion of building blocks that contain metallic Fe and/or the gravitational trapping of surrounding nebula gas. Such an early, wet, reduced atmosphere that covers a proto-ocean would then ultimately evolve toward oxidized chemical compositions through photochemical processes that involve reactions with H2O-derived oxidant radicals and the selective escape of hydrogen to space. During this time, atmospheric CH4 could be photochemically reprocessed to generate not only C-bearing oxides but also organics. However, the branching ratio between organic matter formation and oxidation remains unknown despite its significance on the abiotic chemical evolution of early Earth. Here, we show via numerical analyses that UV absorptions by gaseous hydrocarbons such as C2H2 and C3H4 significantly suppress H2O photolysis and subsequent CH4 oxidation during the photochemical evolution of a wet proto-atmosphere enriched in H2 and CH4. As a result, nearly half of the initial CH4 converted to heavier organics along with the deposition of prebiotically essential molecules such as HCN and H2CO on the surface of a primordial ocean for a geological timescale order of 10–100 Myr. Our results suggest that the accumulation of organics and prebiotically important molecules in the proto-ocean could produce a soup enriched in various organics, which might have eventually led to the emergence of living organisms.
Introduction
Recent cosmochemical studies that have compared isotopic compositions among Earth’s materials and primitive meteorites indicate that most of Earth’s accreting materials closely resembled enstatite chondrites, which have the most reduced oxidation state among primitive meteorites, throughout the accretion phase (e.g., Dauphas, 2017; Dauphas et al., 2024; Halliday and Canup, 2023; and references therein). This suggests that the volatile elements accreted on Earth were chemically reduced by reactions with metallic Fe in the accreting materials, forming an impact-generated atmosphere enriched in reduced gases such as H2 and CH4 (Urey, 1952; Kuramoto and Matsui, 1996; Schaefer and Fegley, 2010; Genda et al., 2017a; Genda et al., 2017b; Benner et al., 2020; Zahnle et al., 2020; Itcovitz et al., 2022; Pearce et al., 2022; Wogan et al., 2023). In addition, planet formation theories and the primordial isotopic compositions of hydrogen and noble gases in igneous rocks derived from the deep mantle suggest that proto-Earth also obtained the H2-dominated solar nebula gas as its proto-atmosphere (Hallis et al., 2015; Olson and Sharp, 2019; Saito and Kuramoto, 2020).
Such a highly reduced atmosphere covering a proto-ocean has been thought to be a candidate site for the synthesis of organic matter and prebiotically important molecules such as HCN and H2CO that precede the emergence of life (e.g., Miller, 1953; Miller and Urey, 1959; Schlesinger and Miller, 1983; Benner et al., 2020; Zahnle et al., 2020; Pearce et al., 2022; Wogan et al., 2023). These molecules can be produced through chemical reactions induced by the irradiation of high-energy photons and particles (e.g., Zahnle, 1986; Airapetian et al., 2016), electric discharge (e.g., Miller, 1953; Schlesinger and Miller, 1983), and/or high-velocity impacts (Furukawa et al., 2009; Ferus et al., 2020; 2022). Among these energy sources, solar UV irradiation is expected to be the most potentially available for the synthesis of these compounds (Chyba and Sagan, 1992). Synthesized HCN and H2CO could be precursors of amino acids and part of nucleobases and sugars: various amino acids can be produced from HCN, aldehydes, and NH3 through Strecker reactions (Miller and Urey, 1959). Adenine, which is one of the nucleobases, can be generated in the solution of HCN and NH3 (Oró, 1960), and ribose, a building block of nucleotides, can be produced from H2CO through the formose reactions (Butlerow, 1861). NH2CHO, produced through the hydrolysis of HCN, can serve as a precursor to all canonical nucleobases (e.g., Saladino et al., 2012). Organic haze aerosols produced in reduced atmospheres might be deposited on the surface and form a water-insoluble organics layer, part of which could hydrolyze to produce various amino acids and nucleobases (e.g., Khare et al., 1986; Poch et al., 2012; Pearce et al., 2024).
Atmospheric photochemistry that involves H2O-derived oxidant radicals, with hydrogen escaping into space, may have evolved an early Earth’s reduced atmosphere toward oxidized chemical compositions, although its rate was possibly slow. The hydrogen escape rate is estimated to asymptote to the diffusion-limited flux even under the enhanced X-ray and extreme ultraviolet (XUV) irradiation from the young Sun when considering the radiative cooling effects by CH4 and radiatively active photochemically derived products (Yoshida and Kuramoto, 2020; 2021). This prolongs the duration of the H2-rich condition, possibly up to several hundred million years, which is significantly longer than in pure hydrogen atmospheres (e.g., Sekiya et al., 1980; Erkaev et al., 2013; Lammer et al., 2014). On the other hand, CH4 would become oxidized to CO and CO2 through reactions that involve oxidant radicals in parallel with its photochemical conversion to HCN, H2CO, and other heavier organics.
The production rate of oxidant radicals primarily supplied by H2O photolysis depends not only on H2O concentration but also on the availability of UV photons at H2O photolysis wavelengths. While UV shielding by organic haze may suppress molecular photolysis as suggested for the protection of NH3 in ancient Earth’s atmosphere (Sagan and Chyba, 1997), it remains unknown how low molecular weight organic compounds synthesized prior to the organic haze formation may affect the rates of H2O and CH4 photolysis and subsequent chemical reactions. Specifically, UV shielding effects of gaseous hydrocarbons that contain more than two carbons have been given little consideration.
In the present study, we employed a one-dimensional (1D) atmospheric photochemical model for an early reduced Earth’s atmosphere mainly composed of H2 and CH4 to clarify the UV shielding effects of gaseous hydrocarbons on the production of carbon oxides and organic matter, and photochemical evolution of the early Earth’s atmosphere.
This contribution is organized as follows. In Section 2, we describe the outline of our photochemical model. In Section 3, we show the numerical results of the atmospheric profile, production/loss rates of each chemical species, and deposition rates of prebiotically important molecules. In Section 4.1, we show the comparison with the results without the UV absorption by hydrocarbons to clarify their UV shielding effects. In Sections 4.2 and 4.3, we analyze the effects of hydrocarbon accumulation and CO2 degassing on the production and loss rates of major species. In Section 4.4, we discuss the dependence of the calculation results on uncertain parameters. In Section 4.5, we show possible evolutionary tracks of the early reduced Earth’s atmosphere based on our calculation results.
Model Description
We developed a 1D photochemical model for a reduced Earth’s atmosphere mainly composed of H2 and CH4 based on PROTEUS (Nakamura et al., 2023a). So far, PROTEUS has been successfully applied to the Jovian ionosphere (Nakamura et al., 2022), the present-day Martian atmosphere (Nakamura et al., 2023b; Yoshida et al., 2023), an early Martian atmosphere (Koyama et al., 2024), and an H2O-dominated atmosphere in the runaway greenhouse condition (Kawamura et al., 2024 ). The details of PROTEUS are described in the work of Nakamura et al. (2023a). Below, we present the outline of the model in this study.
As for the chemical processes, we consider 342 chemical reactions (Supplementary Table S1) for 61 chemical species composed of H, O, C, and N; H2, CH4, N2, H2O, O2, O, O(1D), H, OH, O3, H2O2, CO2, CO, H2CO, HCO, HO2,1CH2, 3CH2, CH3, C2H6, C2H2, C2H4, HNO2, NO, HNO3, NO2, N, HNO, NH3, NH2, N2H4, N2H3, NH, C2H, C2, C3H8, C3H6, C2H5, CH2CCH2, CH, C, CH2CO, CH3CHO, C2H5CHO, C3H3, C3H2, CH3C2H, HCN, CN, HNCO, NCO, C3H7, C2H3, C3H5, CH3O2, CH3CO, C2H2OH, C2H4OH, CH3O, HCNOH, and H2CN. Referring to the chemical reaction list of Tian et al. (2011), we consider C-bearing species with up to three carbons, excluding S-bearing species and organic haze aerosols, to clarify the role of UV shielding by gaseous hydrocarbons.
The model considers the vertical transport of each chemical species due to molecular diffusion and eddy diffusion. For the molecular diffusion coefficient, we use the formula derived by Banks and Kockarts (1973):
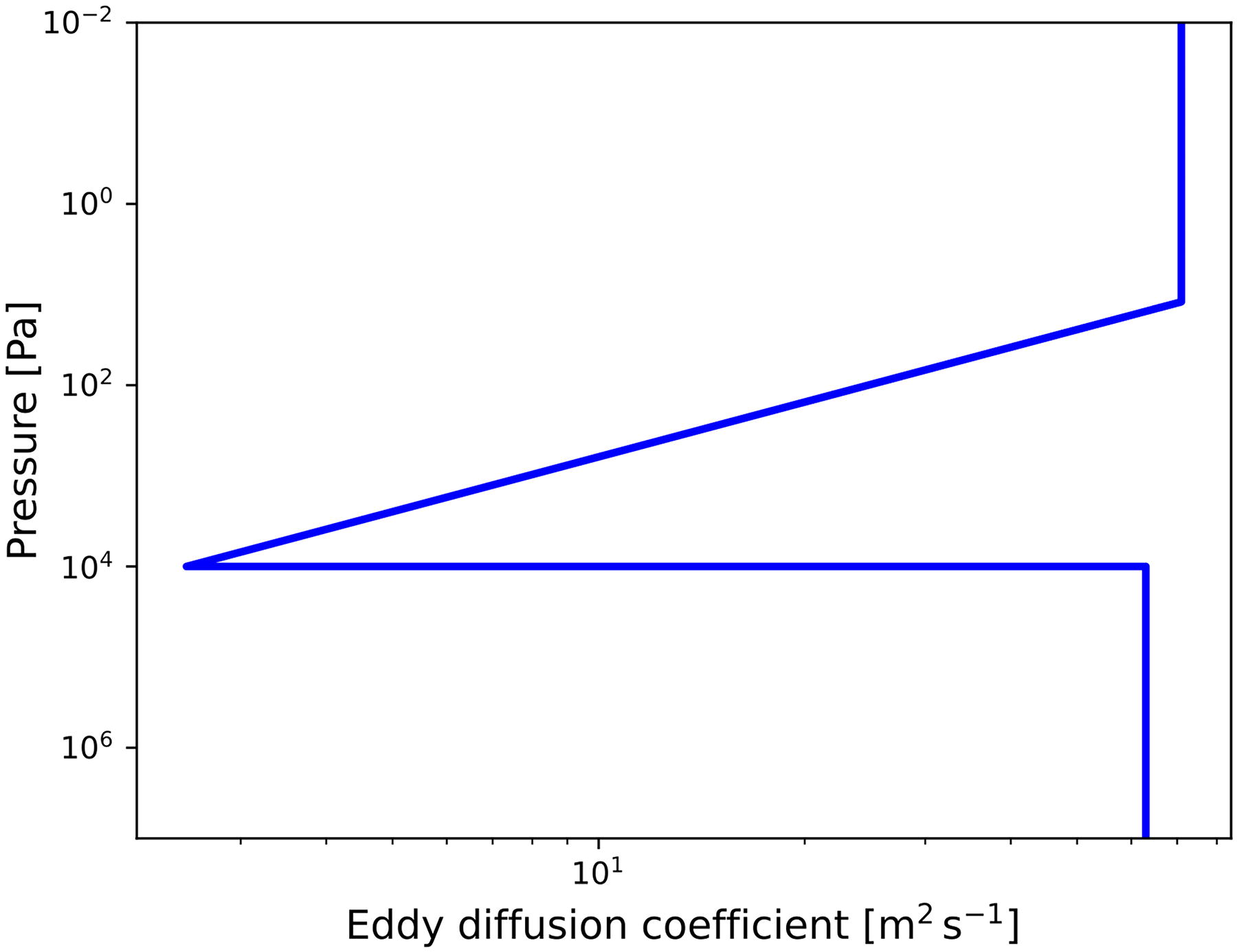
Eddy diffusion coefficient profile.
To calculate the profiles of the photolysis rates, we adopt the UV spectrum from 100 to 1000 nm estimated for the young Sun at the age of 100 Myr (Claire et al., 2012), and solve the radiative transfer by considering the absorption of the solar irradiation by chemical species assuming the solar zenith angle to be 57.3 degrees as indicated by Hu et al. (2012). For the absorption cross-sections, we mainly refer to the MPI-Mainz-UV-VIS Spectral Atlas of Gaseous Molecules (Keller-Rudek et al., 2013) and the JPL publication (Burkholder et al., 2015).
The temperature profile at the troposphere is assumed to follow adiabat, and that above the tropopause is assumed to be kept at the skin temperature (Zahnle et al., 2020). Here, we put the tropopause at 0.1 bar as it is in most solar system planets with atmospheres (Robinson and Catling, 2014). The water vapor profile at the troposphere is assumed to follow the saturation vapor pressure as a function of temperature. As the standard case, the mixing ratio of water vapor at the stratosphere is fixed at 1 ppm by adjusting the planetary albedo and the skin temperature at 175K, which is consistent with the stratospheric temperature under the faint young Sun (Tian et al., 2011; Zahnle et al., 2020). The mixing ratio of water vapor in the stratosphere is an important parameter in the photochemistry of the reduced atmosphere since water vapor is often the major oxidant. We discuss the effect of its change on the calculated atmospheric profile in Section 4.4.
At the lower boundary, which corresponds to the surface, the number densities of H2, CH4, and N2 are fixed. Here, the basal number densities of CH4 and N2 are set to satisfy the column abundances equivalent to the amount of carbon on the present-day Earth’s surface layer (
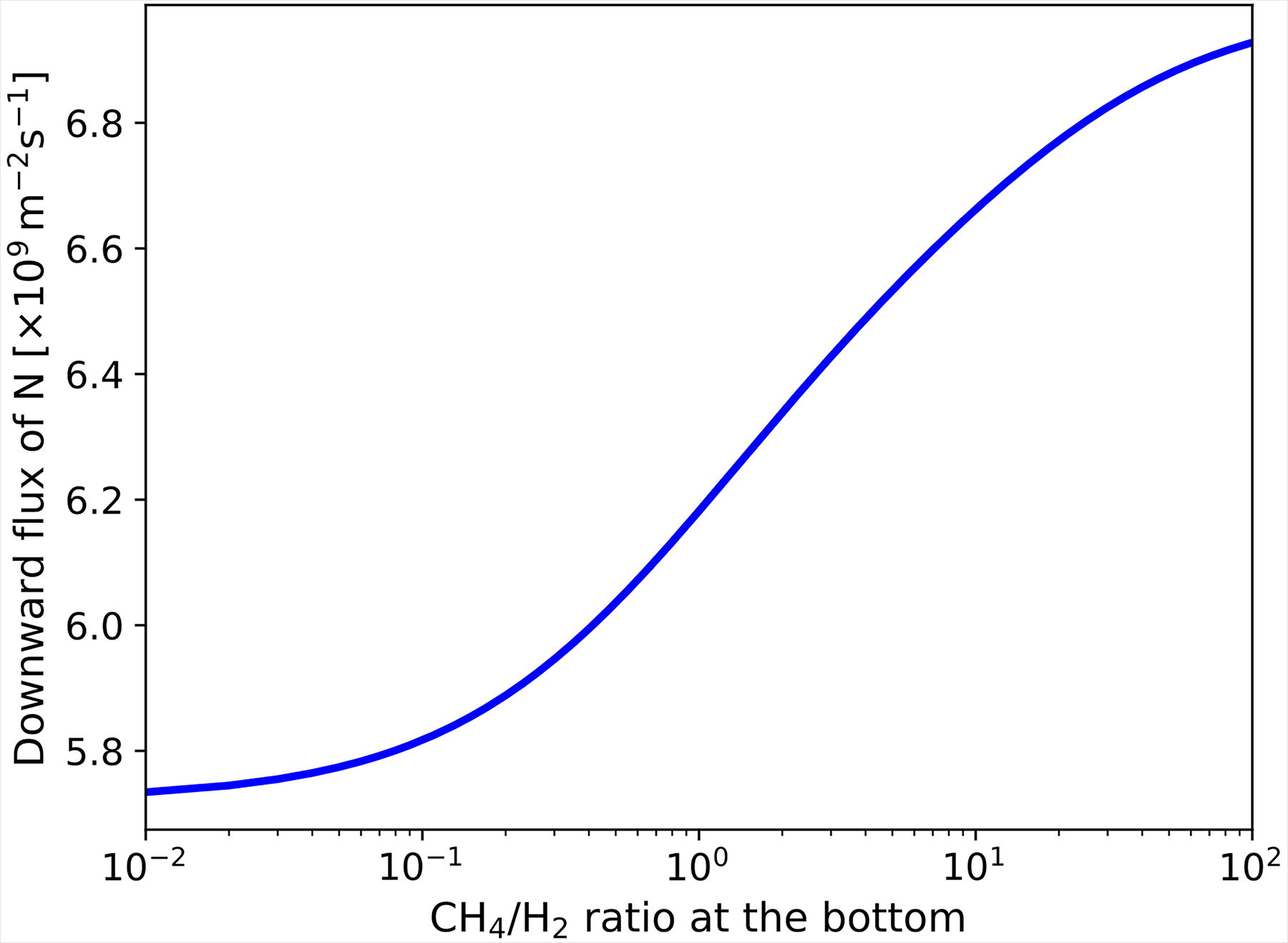
Downward flux of N depending on the basal CH4/H2 ratio.
The number density profiles are shown in Figure 3 for basal CH4/H2 ratios of 0.1 and 10. Here, various C-bearing species, including both organics and oxides, are produced from CH4. While the abundance of hydrocarbons such as C2H6 and C3H8 is large when CH4/H2=10, C-bearing oxides such as CO and CO2 are depleted in this case compared with those in the H2-dominated condition.
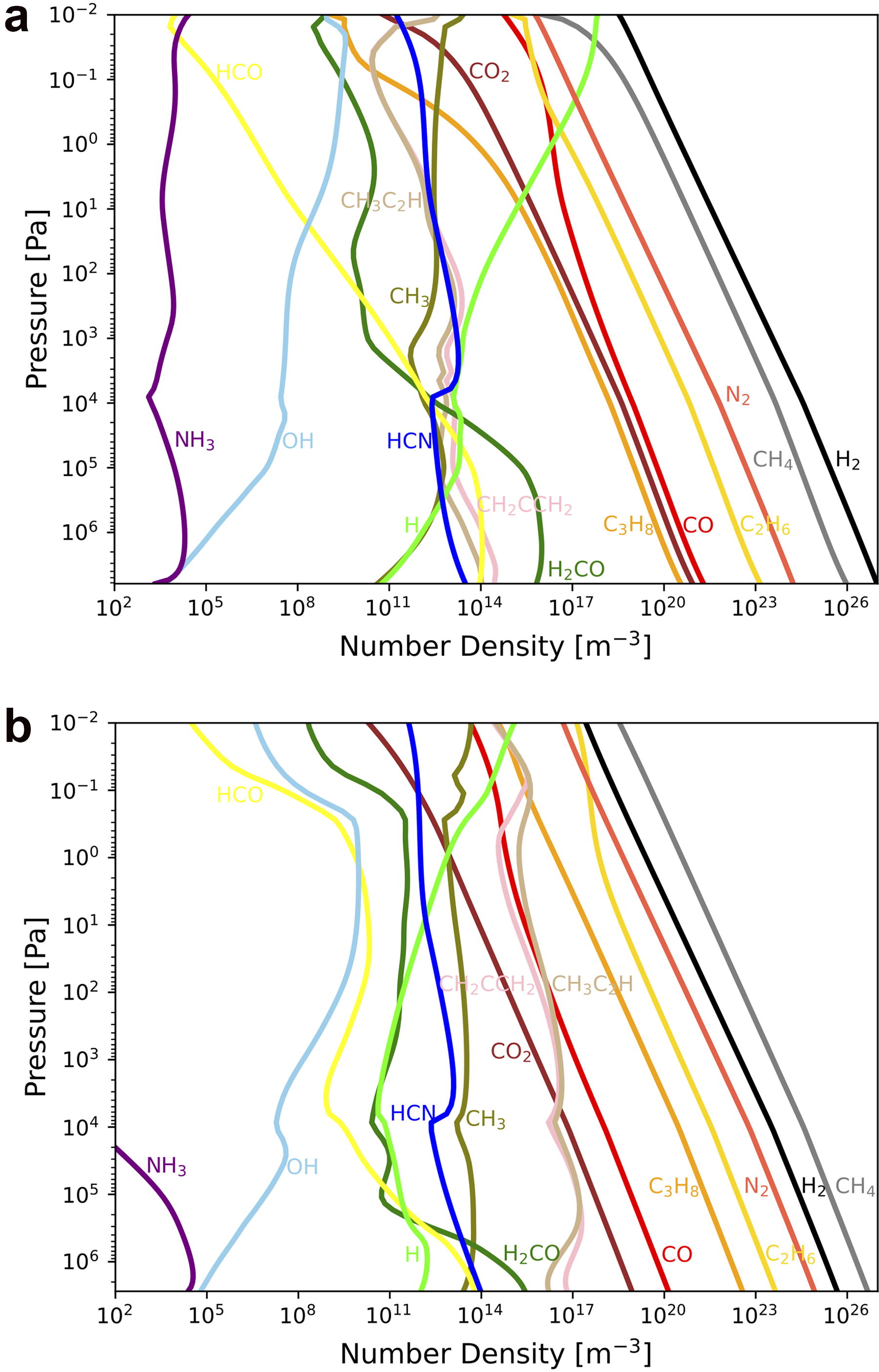
Number density profiles when the basal CH4/H2 ratio is 0.1
As indicated by the number density profiles, the production rates of hydrocarbons increase and far exceed those of carbon oxides under higher CH4/H2 ratios (Fig. 4a). As the mixing ratio of CH4 increases, the production of hydrocarbons increases with the net CH4 decomposition rate (Fig. 4a,b). Concurrently, the production of oxidant radicals by photolysis of H2O vapor, given saturated at the tropopause level, becomes suppressed (Fig. 5a) because the photochemically produced hydrocarbons such as C2H2 and CH2CCH2 effectively shields UV in wavelength range photolyzing H2O as shown in the absorption cross-section of each species in Figure 5b.
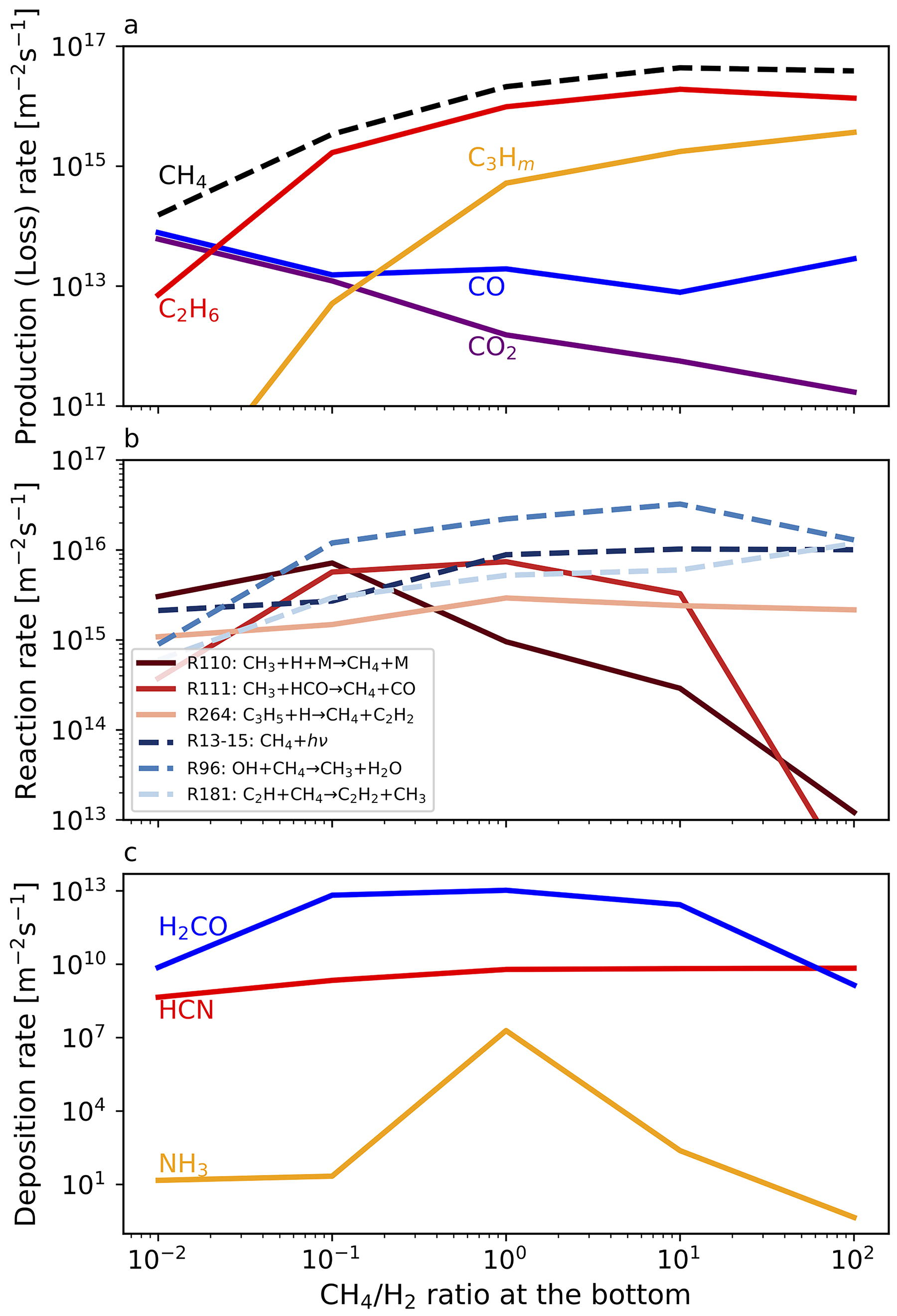
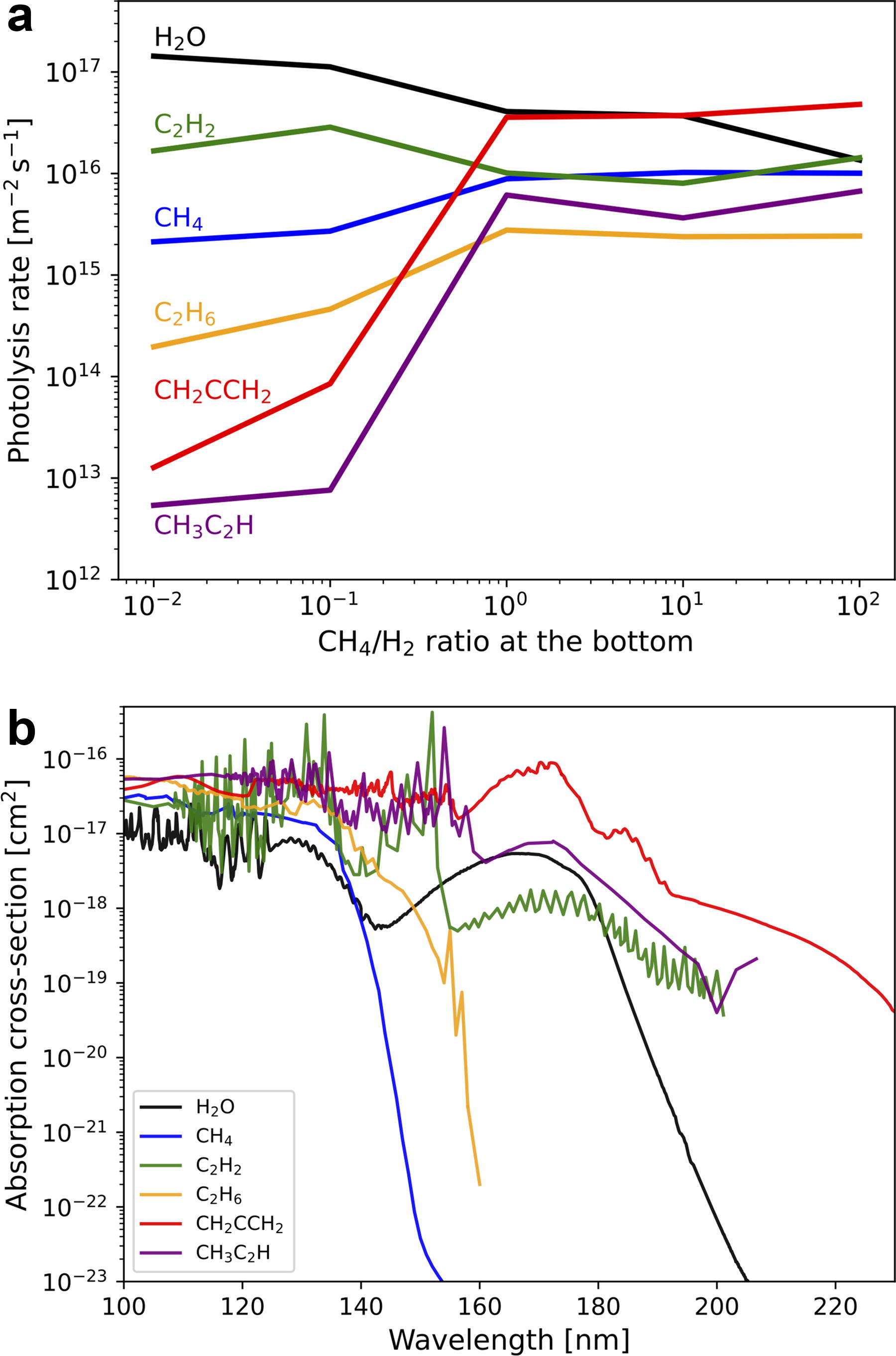
Atmospheric escape would progressively increase the CH4/H2 ratio in the proto-atmosphere because CH4 escape is likely negligible (Yoshida and Kuramoto, 2021), and the H2 loss would proceed with a timescale order of 10–100 Myr, which is shorter than the timescale of photochemical CH4 loss. CH4 is more stable, particularly in early H2-rich conditions: the timescale for CH4 loss obtained from the CH4 column density divided by its net column decomposition rate is as long as ∼1 Gyr when CH4/H2 ∼0.01. This long timescale primarily results from the effective CH4 reformation reactions, such as CH3+H + M→CH4+M (R110) (Fig. 4b), which is well known for the mesosphere regions of giant planets (Yung and DeMore, 1998). The effectiveness of the CH4 reformation reactions on H2-rich atmospheres, supposing primitive Earth, is also shown by Wogan et al. (2023). Moreover, the reactions that oxidize CH4 such as OH+CH4→CH3+H2O (R96) are suppressed in H2-rich conditions (Fig. 4b) because OH is depleted by the reaction with H2(R62: H2+OH→H2O+H), which also contributes to stabilizing CH4. As the atmospheric CH4/H2 ratio increases through the hydrogen escape to space, the photochemical production of organics by the polymerization of CH4-derived species becomes enhanced due to the slowing of hydrogenation reactions.
H2CO and HCN, which are considered crucial in prebiotic chemical evolution, can be continuously synthesized and accumulated in the proto-ocean during the evolution of the reduced atmosphere. In our photochemical model that contains background N2 equivalent to 0.8 bar at the surface, the deposition rate of H2CO peaks at around CH4/H2=1 (Fig. 4c), where hydrogenation of HCO proceeds efficiently near the surface. The maximum deposition rate of H2CO is at most ∼1014 molecules m−2 s−1, slightly lower than estimates for a weakly reduced N2-dominated atmosphere (2.8
Comparison with the results without the UV absorption by hydrocarbons
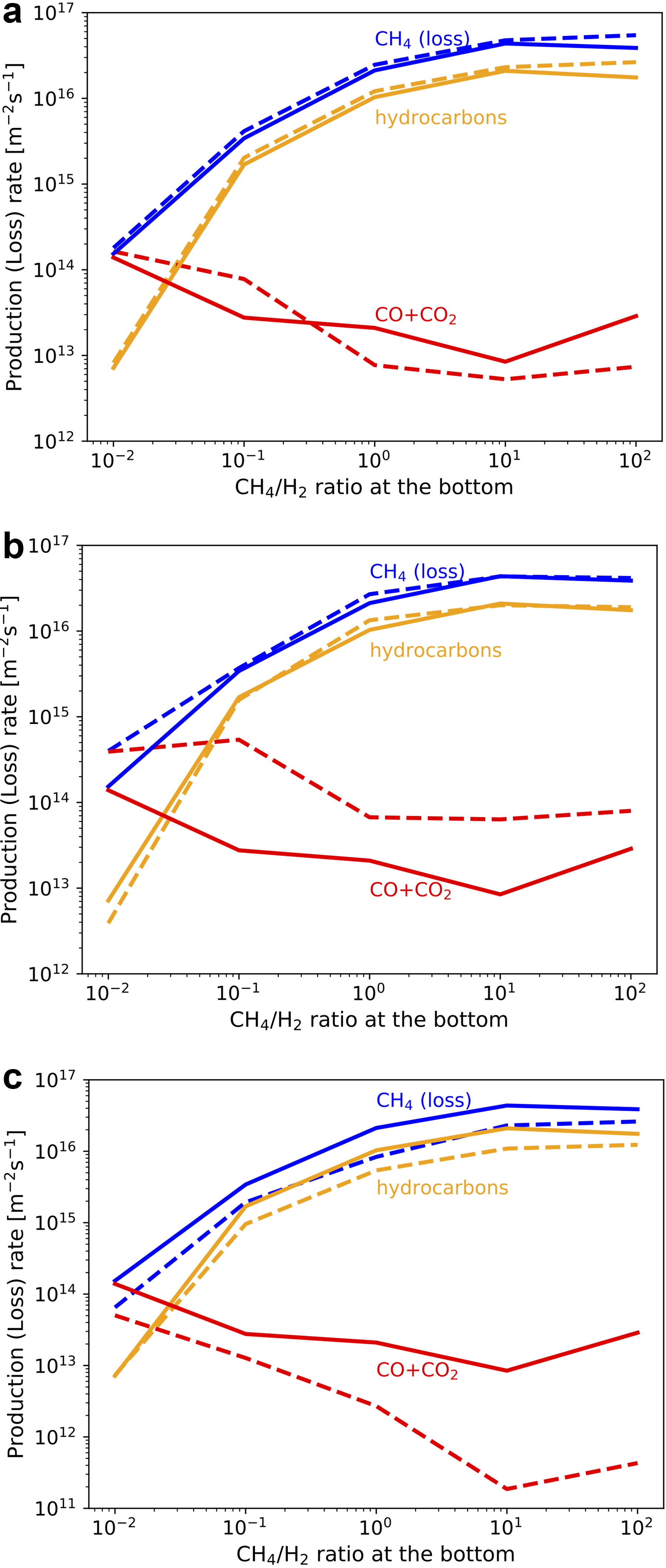
To clarify the UV shielding effects of hydrocarbons, the comparison with the simulation results without the UV absorption by hydrocarbons other than CH4 (Fig. 6a) and C3Hm neglected by previous studies such as Pearce et al. (2022) and Wogan et al. (2023) (Fig. 6b) is shown. The production rate of carbon oxides increases while that of C3Hm relatively decreases without UV absorption by hydrocarbons since the formation of oxidant radicals by H2O photolysis becomes enhanced. The UV shielding by C3Hm becomes effective, especially in CH4-dominated conditions (Fig. 6b). The production of C2H6 is a little enhanced in Figure 6a due to the more efficient CH4 photolysis. These results confirm that UV shielding by gaseous hydrocarbons significantly increases the branching ratio of organics production through CH4 photolysis compared with estimates by the previous studies.
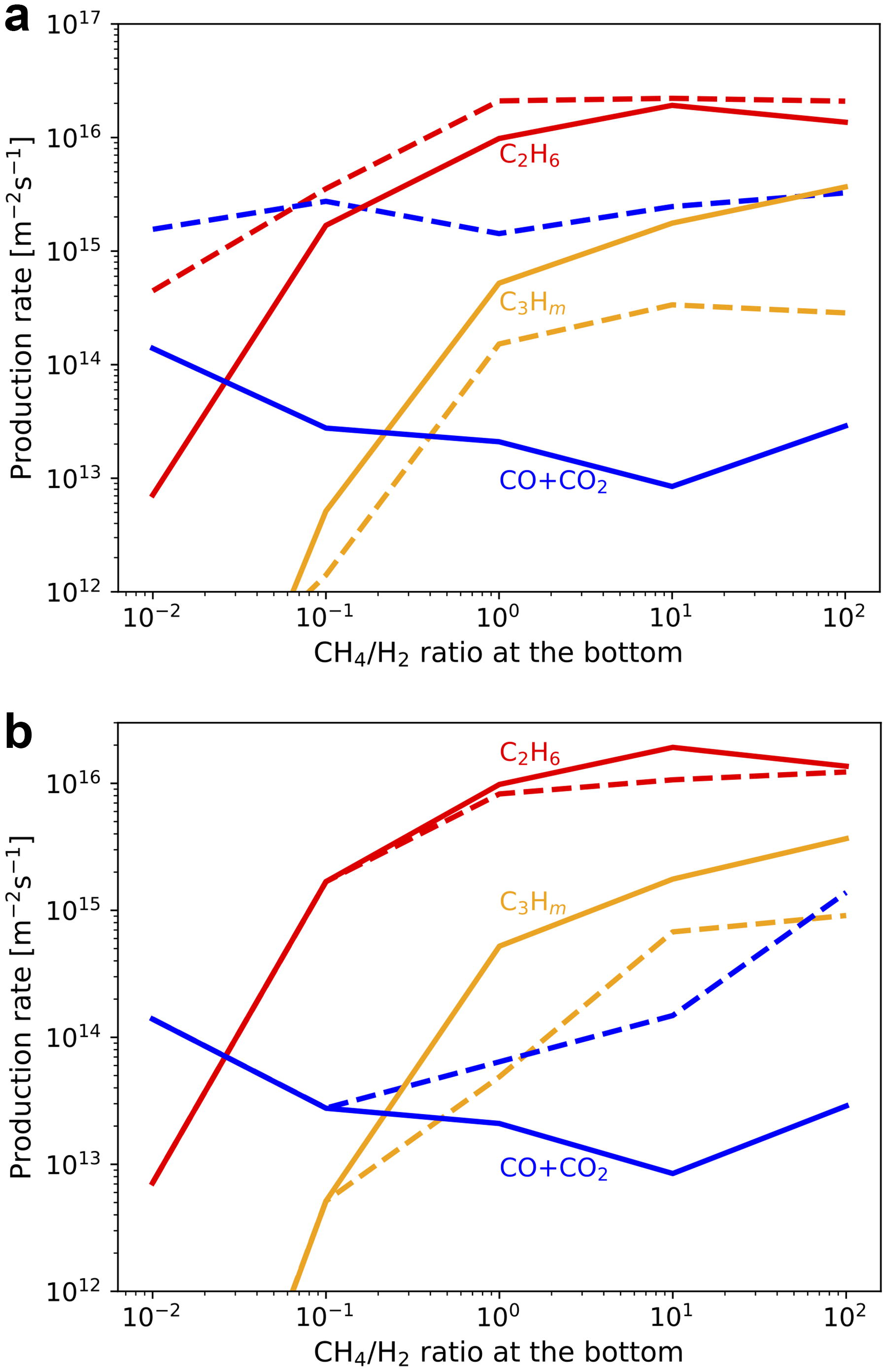
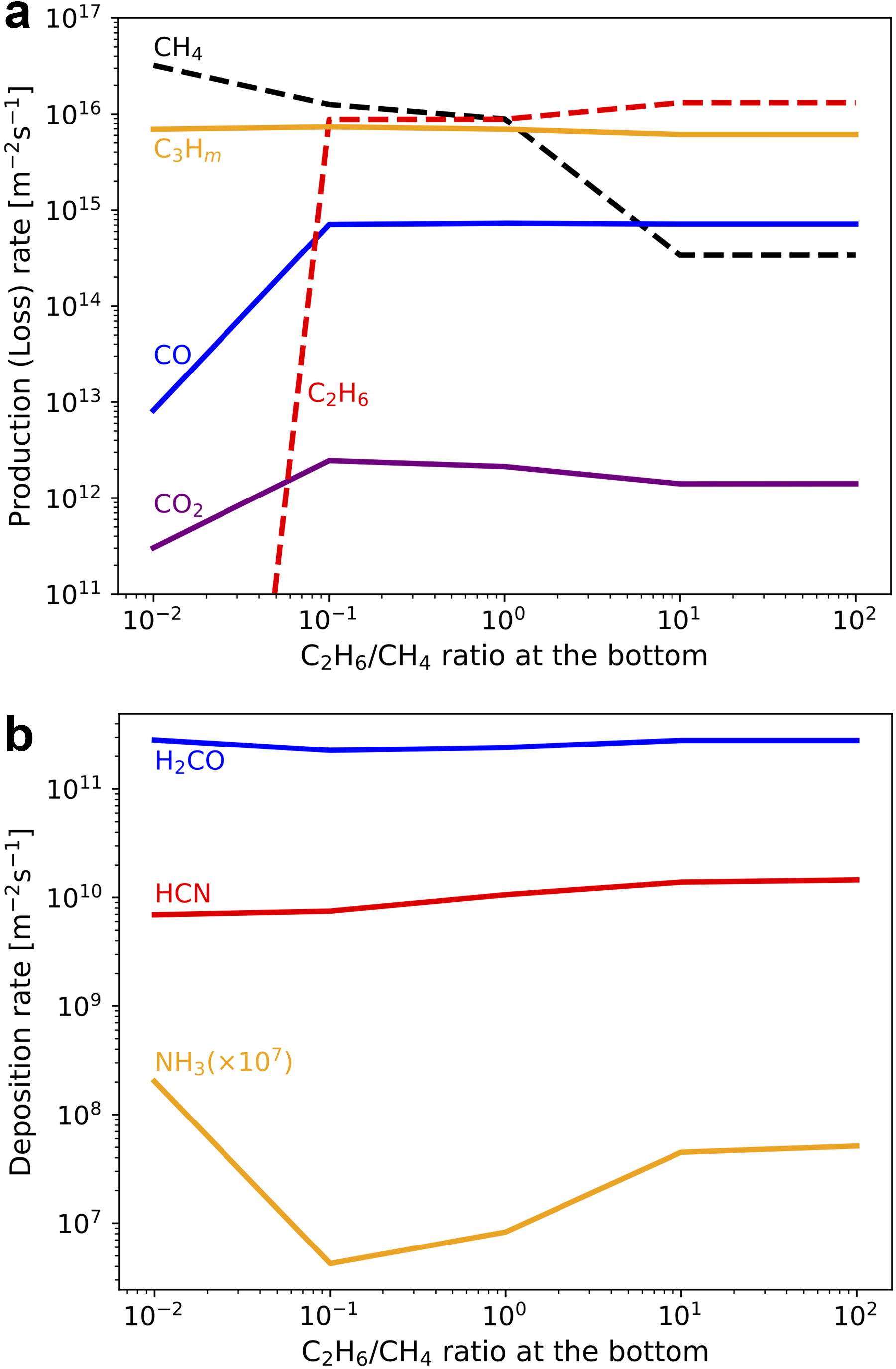
Our results show that hydrocarbons are efficiently produced from CH4 due to their UV shielding effects. The production rate of C2H6 is especially high among C-bearing chemical products (Fig. 4a). Since C2H6 is not a radical species, it may accumulate in the proto-atmosphere. The effects of the accumulation of C2H6 on the production and deposition rates of other relevant chemical species are examined through simulations with various atmospheric C2H6 abundances, assuming a fraction of CH4 is converted to C2H6 while fixing the total carbon amount equivalent to the present-day Earth’s surface layer. C2H6 is photolyzed to generate mainly heavier hydrocarbons such as C3H8 when the basal C2H6/CH4 is larger than ∼0.1 (Fig. 7a). The production rates of CO and CO2 are relatively small compared with that of heavier hydrocarbons due to the UV shielding by hydrocarbons as in CH4-rich conditions. The deposition rates of HCN, H2CO, and NH3 are comparable with those in CH4-dominated conditions (Fig. 7b). These results indicate that effective organics production continued even after C2H6 accumulation proceeded as shown in the atmospheric evolutionary track in Section 4.5.
We suppose that the initial atmosphere is mainly composed of H2 and CH4. On the other hand, oxidized gases such as CO2 may have been provided by volcanic degassing if the upper mantle had a similar redox state to today. The effects of CO2 supply on the production/loss rates of major chemical species are shown by simulations given various basal CO2/CH4 ratios in Figure 8. The CO production rate increases with the CO2/CH4 ratio (Fig. 8a). CO2 photolysis becomes the major source of oxidant radicals when CO2/CH4 >∼10−5, which can be maintained by CO2 degassing with rates higher than the present-day value (∼1013 m−2 s−1; Catling and Kasting, 2017). In these CO2-rich conditions, about half of CO is derived directly from CO2 photolysis, and the other half is produced through CH4 oxidation by oxidant radicals mainly provided from CO2 photolysis. CO becomes the major chemical product when CO2/CH4 >∼0.01, while the CO2 degassing rate needs to be more than two orders of magnitude higher than the present-day value to maintain the CO2 abundance. The result that CO becomes the major CH4-derived chemical product in CO2-dominated conditions is consistent with the photochemical calculation by Wogan et al. (2023) for an H2-CH4-CO2 atmosphere. These results indicate that the CH4 oxidation may have been promoted by volcanic CO2 supply as long as the supply was high enough to make CO2 the main source of oxidant radicals, although the degassing rate from proto-Earth is highly uncertain.
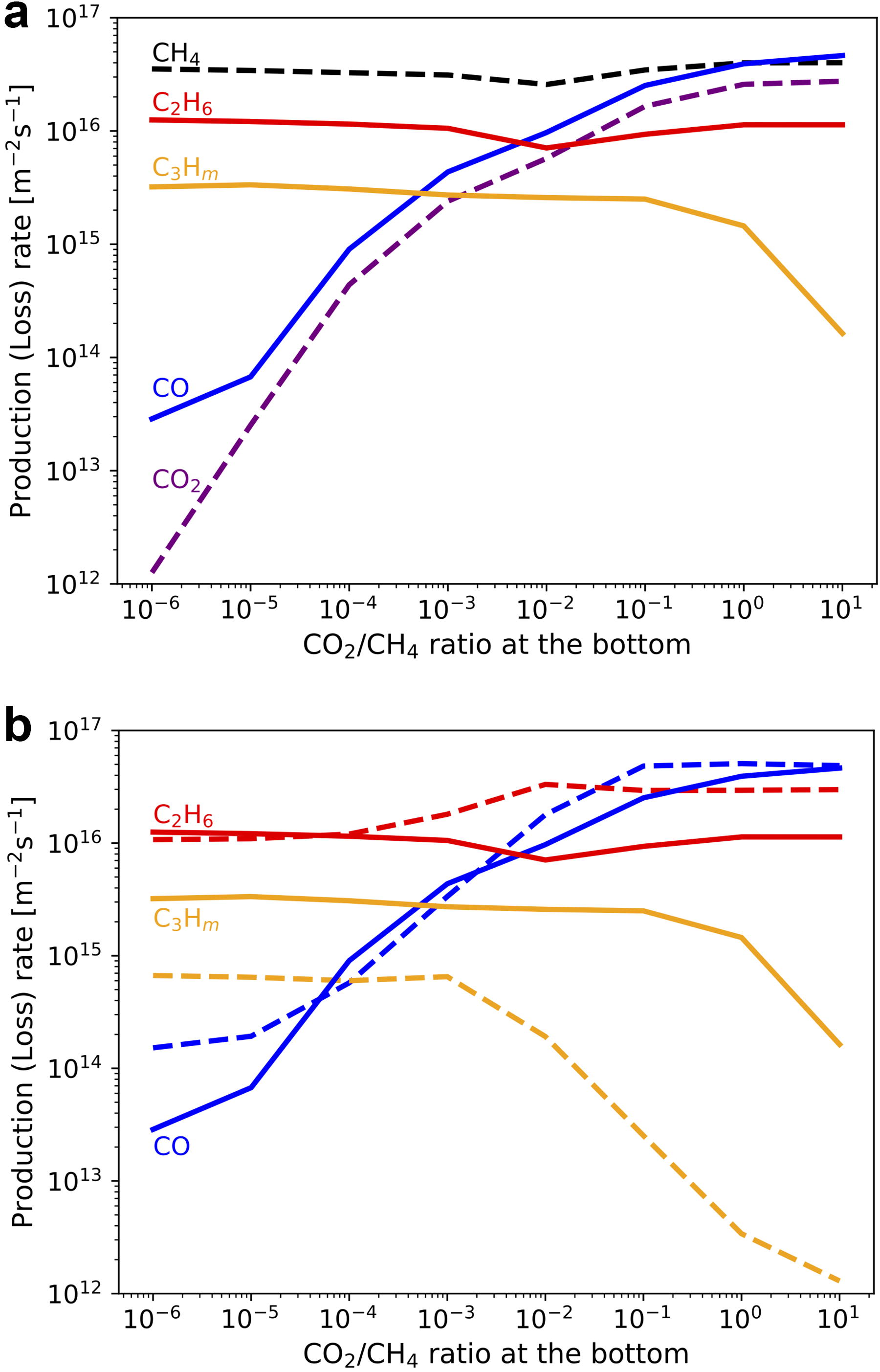
Efficient production of organics by their UV shielding occurs even in CO2-rich conditions; their production rate changes a little in the wide atmospheric composition range with CO2/CH4 <∼1 (Fig. 8a). To clarify the effects of UV absorption by C3Hm neglected by the previous studies, the comparison with the simulation results without their UV absorption is shown in Figure 8b. The production rate of CO becomes high, while that of C3Hm is significantly suppressed compared with the standard case due to the enhancement of H2O photolysis, which shows that UV shielding by C3Hm can enhance organics production in CO2-rich conditions. The production rate of C2H6 is slightly large without UV shielding by C3Hm because of the more efficient CH4 photolysis.
4.4.1. Dependence on the eddy diffusion coefficient
The profile of the eddy diffusion coefficient in the early Earth’s upper atmosphere is highly uncertain, although the setting should affect the calculation results. Figure 9a shows the comparison with the production/loss rates of major species under the eddy diffusion coefficient 10 times as large as the standard setting. In the higher eddy diffusion condition, the abundance of CH4 in the uppermost region increases due to the efficient vertical transport. This slightly enhances the CH4 photolysis and the following production of hydrocarbons. The production of C-bearing oxides is also slightly enhanced when the basal CH4/H2 ratio is small but becomes suppressed as the CH4/H2 ratio increases. The overall dependence of the production of CH4-derived major products on the atmospheric composition is little affected by the increased eddy diffusion, which indicates that the UV shielding effect of hydrocarbons plays a primary role in controlling this behavior.
4.4.2. Dependence on the stratospheric H2O abundance
Since H2O is the major source of oxidant radicals through its photolysis, its stratospheric abundance is a key parameter in the oxidation of reduced atmospheres. We suppose that the stratospheric H2O mixing ratio is 1 ppm, which is slightly lower than the present value, considering a colder stratosphere under the faint young Sun. On the other hand, the stratospheric temperature and H2O mixing ratio are variable depending on the uncertain planetary albedo on early Earth. Figure 9b shows the production/loss rates of major species when the stratospheric H2O mixing ratio is 10 times as large as the standard setting. The production rates of CO and CO2 increase in the abundant stratospheric H2O condition due to the increase in oxidant radicals such as OH and O. Despite the production enhancement of C-bearing oxides, however, the efficient production of hydrocarbons has little changed in the CH4-rich conditions due to their strong UV shielding effect.
4.4.3. Dependence on the UV flux
While our nominal simulation used a fixed UV flux estimated for the young Sun at the age of about 100 Myr, they should have changed with time. In particular, the flux of UV with a wavelength shorter than about 200 nm is estimated to decline rapidly (Claire et al., 2012). Figure 9c shows the production/loss rates of major chemical species when the whole UV flux is half as high as the standard case with the fixed spectrum profile. The net loss rate of CH4 decreases proportionally with the UV flux, while the relative production rates of major chemical products change a little. Considering the change in the UV flux with time, the timescale for CH4 decomposition should be prolonged compared with the results in Section 3.
Based on the production and loss rates of chemical species calculated with varying background atmospheric composition, possible evolutionary tracks are drawn for the early reduced Earth’s atmosphere. The initial atmosphere is composed of H2 CH4, and 0.8 bar N2 with taking the amount of CH4 equivalent to the C content in the present Earth’s surface layer (=
An example of atmospheric evolutionary tracks is shown in Figure 10 where the initial H2 partial pressure is taken at 300 bar, which corresponds to the delivery of H2 associated with the present ocean mass of H2O under equilibrium with accreting metallic Fe and mantle FeO (Kuramoto and Matsui, 1996). While the atmosphere is H2-dominated, CO and C2H6 are produced from CH4 slowly due to effective CH4 reformation. After CH4 becomes dominant as H2 is lost by escape, CH4 and C2H6 become efficiently photolyzed to generate heavier hydrocarbons. It takes ∼300 Myr until the partial pressures of H2 and light hydrocarbons CH4 and C2H6 decrease below 0.1 bar through their loss by escape, oxidation, and heavier hydrocarbon production. Although the depositions of H2CO, HCN, and NH3 little affect the change in atmospheric mass, their net mass reaches ∼1017 kg. Production of heavier hydrocarbon is a major branch of the transformation of C during the evolution of the reduced atmosphere.
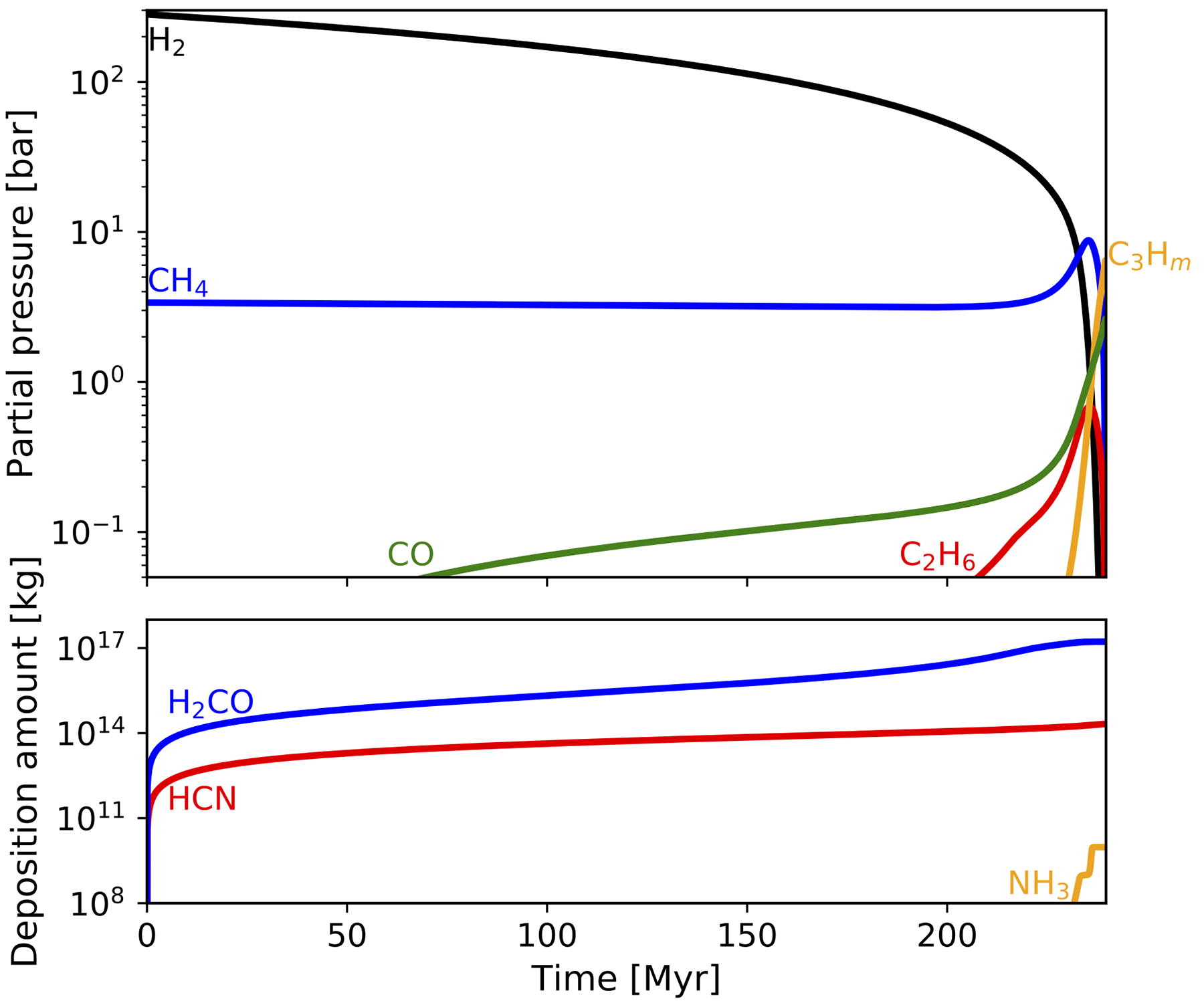
Possible change in the partial pressure and deposition amount of each chemical species with time when the initial H2 partial pressure is 300 bar.
The major atmospheric components that remain when H2 and light hydrocarbons become largely lost are heavier hydrocarbons regardless of the initial CH4/H2 ratio (Fig. 11a) since CH4 tends to be converted to hydrocarbons by their UV shielding effect. The final partial pressures of CO and CO2 are a little large when the initial CH4/H2 ratio is small because CO and CO2 are major photochemical products in the long-lasting H2-dominated phase (Figs. 4a and 10).
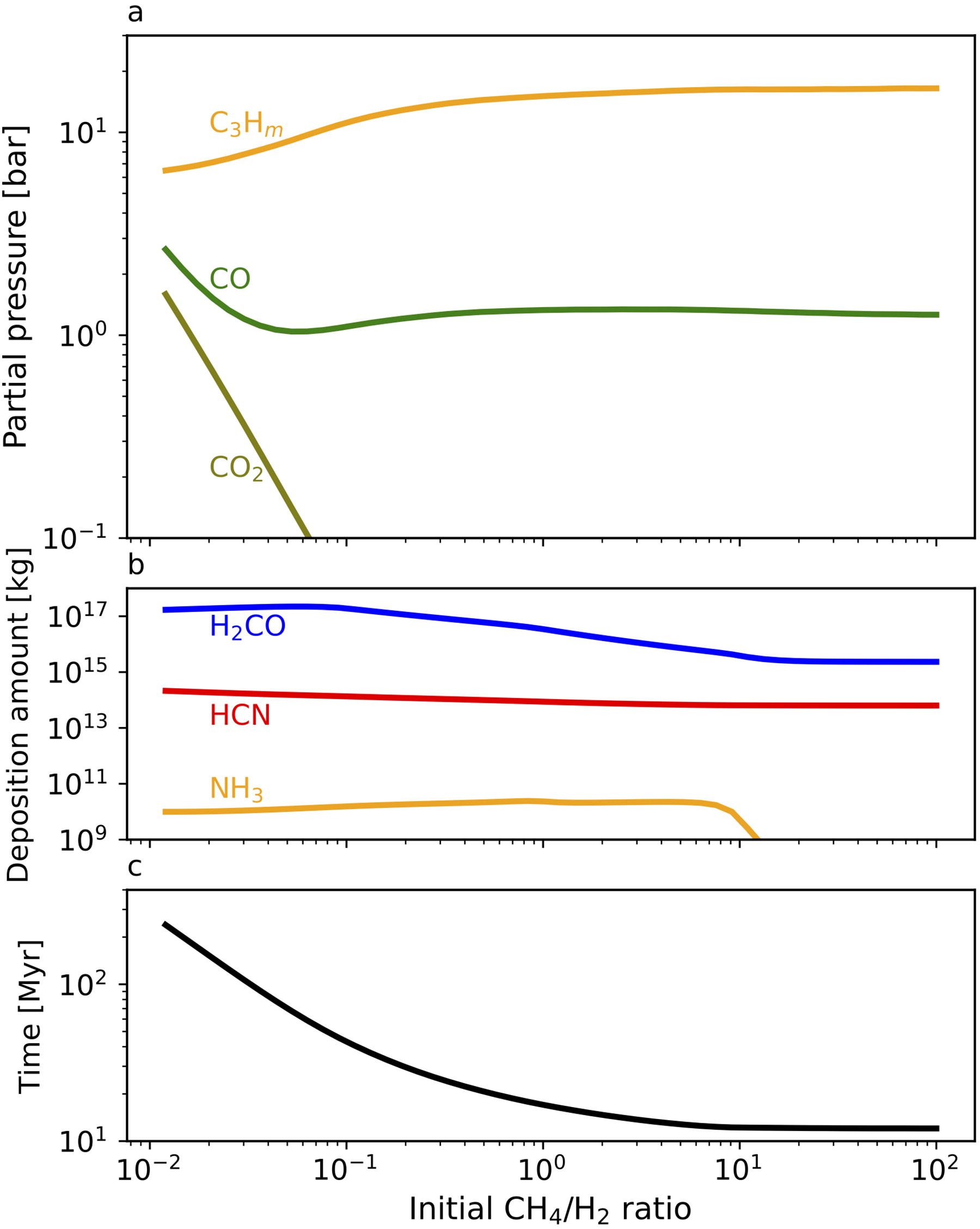
When the initial CH4/H2 ratio >∼1, the duration time of the atmospheric state enriched in H2 and/or light hydrocarbons (Fig. 11c) is almost constant ∼10 Myr, which is determined by the time for the decomposition of CH4 and C2H6. The duration time increases as the initial atmosphere is H2-rich due to the prolongation of time for H2 loss. The formation of a massive H2-rich atmosphere with a mass of several hundred bars on proto-Earth has been suggested from the chemistry of Earth’s building blocks involving metallic Fe (e.g., Dauphas, 2017). Late accretion may have produced H2 with a mass comparable to that of the seawater through the accretion of enstatite-chondrite-like materials with a total mass of ∼0.5% of Earth’s mass (e.g., Genda et al., 2017; Zahnle et al., 2020; Itcovitz et al., 2022).
The continuous deposition of H2CO and HCN during the long-lasting atmospheric evolution results in their total supply of ∼1017 kg and ∼1014 kg, respectively (Fig. 11b). This is equivalent to an H2CO concentration of ∼1M and an HCN concentration of ∼0.01M in the current volume of seawater; the former is much larger than the minimum H2CO concentration ∼10−2–10−3 M required for the occurrence of formose-type reactions (e.g., Bada and Miller, 1968; Reid and Orgel, 1967; Gabel and Ponnamperuma, 1967; Schwartz and De Graaf, 1993), and the latter is near the level at which HCN hydrolysis and oligomerization may be competing in prebiotic ocean (e.g., Sanchez et al., 1966; Miyakawa et al., 2002). The total deposition of NH3 is small, but NH3 can be produced through the hydrolysis of HCN (Zahnle, 1986; Tian et al., 2011) and the chemical reduction of N2 by accreting metallic Fe (Zahnle et al., 2020; Wogan et al., 2023). HCONH2 can also be produced through the hydrolysis of HCN (e.g., Saladino et al., 2012). The continuous supply of these prebiotically important molecules could potentially lead to the synthesis of amino acids, nucleobases, sugars, and their polymers. When combined with the hot spring hypothesis (Damer and Deamer, 2020), our results indicate that high concentrations of these organic compounds in hot spring pools can be achieved through atmospheric synthesis, as well as other supply processes such as organics production in hydrothermal vents (e.g., Martin et al., 2008) and exogenous delivery (e.g., Chyba and Sagan, 1992). Although part of the deposited organic compounds suffered from several decomposition processes such as hydrolysis and photolysis (Catling and Kasting, 2017; Pearce et al., 2022), these results suggest that the early atmosphere served as a major source of the prebiotically important molecules.
The efficient production of heavier organics with a total mass equivalent to ∼10 bar (Fig. 11a) may also have played essential roles in the supply of prebiotic compounds. In CH4-dominated conditions, ∼80% of organics with three carbons are expected to be polymerized to organic haze aerosols supposing that the branching ratio of their production from C3Hm can be approximated by
An atmospheric evolutionary scenario estimated by our calculation is schematically shown in Figure 12. First, an H2-dominated environment lasted for up to several hundred million years. Just after most H2 escaped to space, the production of heavier organics including complex organic haze aerosols became intensified through the enhanced CH4 photolysis and hydrocarbon self-UV shielding. The accumulation of such organics and the prebiotically essential molecules such as HCN and H2CO at this phase can produce various prebiotic compounds as mentioned above. Eventually, the production of biologically essential matters originating from the early atmosphere may have led to the emergence of living organisms.
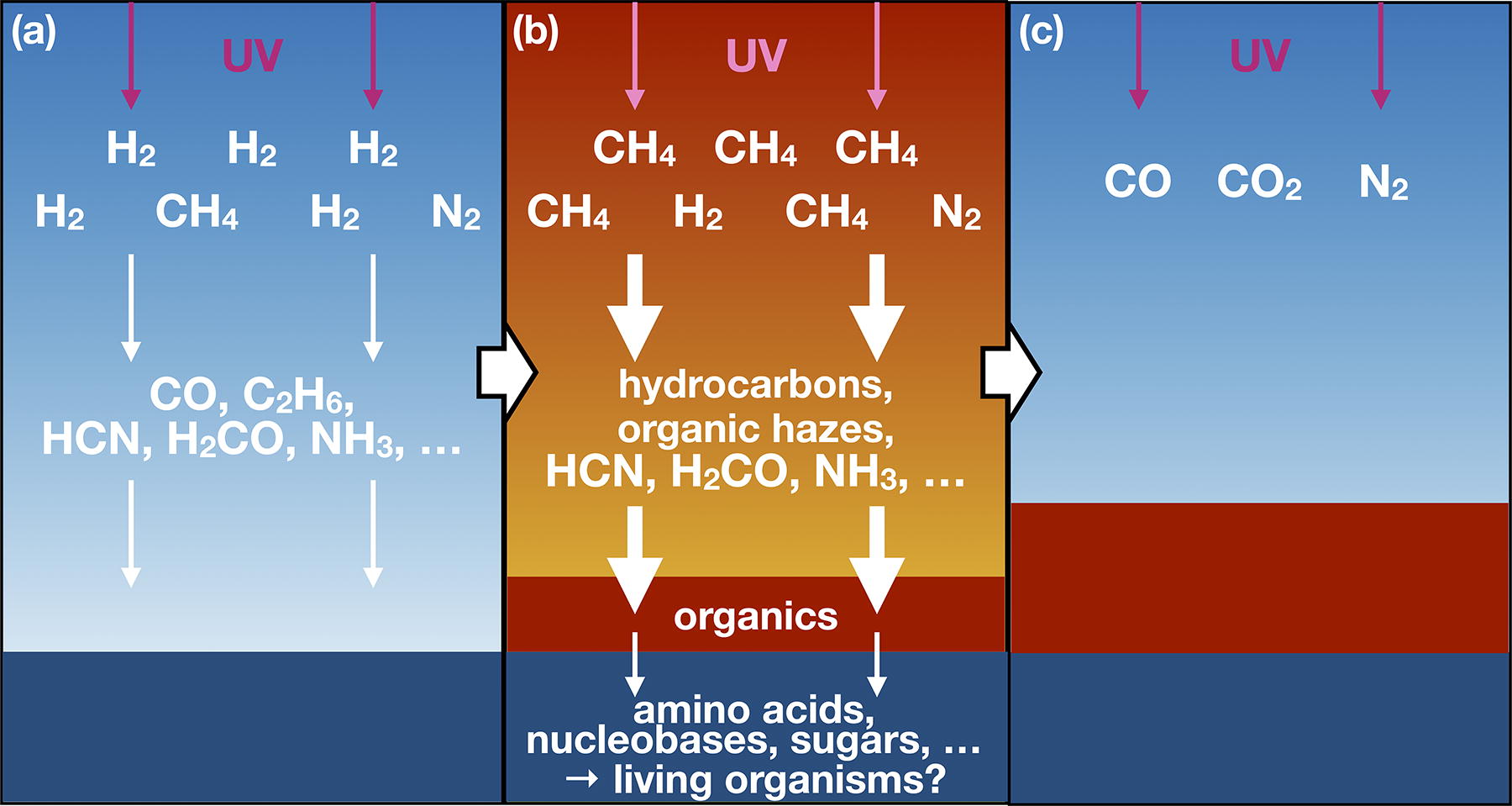
An atmospheric evolution scenario estimated by our calculation.
We applied our 1D photochemical model to Earth’s early reduced atmosphere, which was mainly composed of H2 and CH4, to clarify the UV shielding effects of gaseous hydrocarbons on the production of carbon oxides and organic matter. According to our results, UV absorptions by gaseous hydrocarbons such as C2H2 and C3H4 significantly suppress the H2O photolysis and following CH4 oxidation. Accordingly, nearly half of initial CH4 possibly becomes converted to heavier organics along with deposition of prebiotically essential molecules such as HCN and H2CO on the surface of a primordial ocean for a geological timescale order of 10–100 Myr. These results suggest that the accumulation of organics and prebiotically important molecules in the proto-ocean could have produced a soup enriched in various organics, which might have eventually led to the emergence of living organisms.
Authors’ Contributions
T.Y., N.T., and K.K. designed the study. T.Y. performed the model calculations. T.Y., S.K., and Y.N. developed the photochemical model. T.Y. took the lead in writing the manuscript. All authors interpreted the results and improved the manuscript.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported by JSPS KAKENHI grant Nos. JP23KJ0093, JP23H04645, JP22KK0044, JP22H00164, JP22KJ0314, JP24KJ0066, JP21K03638, JP24K07110, JP22K21344, and the International Joint Graduate Program in Earth and Environmental Sciences, Tohoku University (GP-EES).
Footnotes
Acknowledgment
The authors thank the anonymous reviewer whose comments greatly improved the article.
Supplementary Material
Supplementary Table S1
Supplementary Data S1
Abbreviations Used
Associate Editor: Nita Sahai
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
