Abstract
The health of astronauts during space travel to new celestial bodies in the Solar System is a critical factor in the planning of a mission. Despite cleaning and decontamination protocols, microorganisms from the Earth have been and will be identified on spacecraft. This raises concerns for human safety and planetary protection, especially if these microorganisms can evolve and adapt to the new environment. In this study, we examined the tolerance of clinically relevant nonfastidious bacterial species that originate from environmental sources (Burkholderia cepacia, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Serratia marcescens) to simulated martian conditions. Our research showed changes in growth and survival of these species in the presence of perchlorates, under desiccating conditions, exposure to ultraviolet radiation, and exposure to martian atmospheric composition and pressure. In addition, our results demonstrate that growth was enhanced by the addition of a martian regolith simulant to the growth media. Additional future research is warranted to examine potential changes in the infectivity, pathogenicity, and virulence of these species with exposure to martian conditions.
Introduction
The survival of microorganisms in a simulated Mars environment has been investigated by a number of recent studies (Horneck, 2000; Beblo-Vranesevic et al., 2020, 2022; Horne et al., 2022). These laboratory-based simulations have shown that certain terrestrial microorganisms are capable of surviving under simulated Mars conditions. However, the challenges underlying this premise lie in the accurate representation of the martian environment: ultraviolet (UV) and ionizing radiation, dry and cold atmosphere, low water activity and accessibility, and salt/perchlorate-rich regolith (Horneck et al., 2001). Even though Mars is in the habitable zone of our Sun and relatively close to the Earth, the environmental conditions on its surface differ greatly from our planet.
The global mean annual surface pressure on Mars is 6 hPa with a composition of 95.32% CO2, 2.7% N2, 1.6% 40Ar, 0.1% O2, 0.07% CO, 0.03% (variable) H20, 2.5 ppm Ne, 0.3 ppm Kr, 0.08 ppm Xe, 0.04–0.2 ppm (variable) O3, compared with the 1013 hPa at sea level on Earth (Haberle, 2015). The surface of Mars also receives very harmful and deadly radiation. Orbital measurements have shown a total polychromatic UV flux (200–400 nm) of 49.95 W/m2 (Schuerger et al., 2003) and an ionizing radiation total dose rate of 233 ± 12 μGy/day (Matthiä et al., 2017), which is much higher compared with ∼0.575 μGy/day on Earth (BFS, 2022).
Furthermore, if liquid water is available, it would most likely resemble a brine made up of different salts, including perchlorates (Clark and Kounaves, 2016). It is important to investigate the effects of all these factors and try to mimic Mars conditions as accurately as possible.
Most of the research that aimed to understand which microorganisms can survive on Mars has focused on extremophilic microorganisms: halophiles, thermophiles, psychrophiles, xerophiles, and radioresistant organisms (de la Vega et al., 2007; Bauermeister et al., 2014; Panitz et al., 2019; Beblo-Vranesevic et al., 2020, 2022; Thombre et al., 2020). Such experiments rendered valuable information about the mechanisms of survival, the regulation of metabolism, and the regulation of different and/or shared genes (Horne et al., 2022). However, we know much less about the survivability of human pathogens that are derived from environmental sources to the conditions on Mars. This is of crucial importance as these microorganisms have been shown to be highly adaptable and therefore have a high chance of contaminating the surface of Mars particularly with crewed missions. In addition, these organisms are opportunistic pathogens and therefore have the potential to cause (re-)infection of astronauts during space travel.
Understanding what type of microorganisms are in spacecraft assembly facilities (SAFs) and on the International Space Station (ISS) is of critical importance both for human health and planetary protection, which is why the microbiome of these locations is continually studied (Singh et al., 2018; Mora et al., 2019; Kumar et al., 2022; Leo and Onofri, 2023). In these settings, despite the application of stringent decontamination approaches, bacteria and fungi have been identified, including human-associated as well as biosafety level 2 ones, an example is Klebsiella pneumoniae investigated in this study (Checinska Sielaff et al., 2016; Venkateswaran et al., 2017). Furthermore, these measures may facilitate the unwanted selection of disinfectant-resistant microbial species such as Bacillus pumilus, which has been isolated in SAFs several times (Nicholson et al., 2009; Mora et al., 2019).
For this reason, threshold levels have been implemented on the ISS for the maximum acceptable bioburden for bacteria and fungi (Williams, 2007; Holloway et al., 2003; Van Houdt et al., 2012). Therefore, we can assume that, if these microorganisms have been identified in SAFs and on the ISS, they can make their way to Mars during a human mission. Furthermore, when on Mars, the distribution of these, however, in small amounts, will be unavoidable due to spacesuit and/or spacecraft leakages (Gibson, 1971; Vogt, 1998). Additional sources of contamination could be through waste disposal, especially when human fecal matter will be processed, which is another source from which the organisms analyzed in this study can be found (Okuda et al., 2010; Ochieng et al., 2014; Schwab et al., 2014; Raffelsberger et al., 2021).
During the Apollo missions, the waste disposal system was a source of contamination as the inner and outer fecal disposal plastic bags, despite containing germicides (a mixture of sodium orthophenylphenol and sodium chlorophenylphenol) leaked inside the capsule (Sauer and Jorgensen, 1975). If the waste disposal on Mars will be carried out in a similar way the risks of astronauts coming into contact with these and other organisms are likely. Furthermore, the health risks to the astronauts coming into contact with the bacterial species investigated in this study are to be analyzed further, since the human immune system will be highly dysregulated during a long-duration space mission to Mars (Crucian et al., 2018; Capri et al., 2023).
Therefore, gaining new insights on the impact of Mars simulated conditions on clinically relevant nonfastidious pathogens is essential. In the present study, we focused on the following clinically relevant nonfastidious pathogens: Burkholderia cepacia, K. pneumoniae, Pseudomonas aeruginosa, and Serratia marcescens. We decided to study these four bacterial species as previous research has shown that these species are capable of utilizing and growing in minimal media with compounds identified in carbonaceous meteorites as sole carbon sources (Domínguez-Andrés et al., 2020). Exposing these bacterial species to Mars-like conditions will help our understanding of how nonextremophiles respond to such an environment. The investigation of heterotrophs has been highlighted as a very important aspect also in regard to habitability constraints (Matthews et al., 2023). We therefore strongly believe this research to be a starting point to understand the infectivity, pathogenicity, and virulence of these species upon exposure to a human host on Mars.
In addition, essential elements to consider are not only human safety but planetary protection as well. In this study, we define planetary protection as the responsibility to avoid the unwanted contamination of habitable sites of interest with terrestrial microorganisms also known as forward contamination, as well as the contamination of Earth with dangerous material, known as backward contamination (Coustenis et al., 2023).
Therefore, the assessment of Mars conditions on microorganisms relevant for human health is particularly important in the new age of space exploration, which could enable humans to explore “nearby” planetary bodies such as the Moon and Mars. With current spacecraft technologies, a trip to the Moon would last 3 days and to Mars ∼6 months. Studies have shown that, despite the different approaches to sterilization of spacecraft surfaces, biological contaminants can make their way beyond Earth (Rettberg et al., 2019). Undoubtedly, when humans take part in space travel, microbial contamination of the explored site or planetary body is very likely due to the ubiquitous number of microbes harboring in and on the human body, that is, the microbiome, which is an essential part of human health.
Furthermore, research into the antibiotic effectiveness and drug resistance of microbial species in human-supporting space stations can be traced to the 1980s (Klaus and Howard, 2006; Mora et al., 2019). Previous studies have shown that the human immune system is dysregulated in space due to a variety of factors, including the disruption of the circadian rhythm, differences in gravity, radiation, confinement, psychological stress, restricted diet, and limited sanitary measures (Stahn et al., 2017; Buchheim et al., 2019; Simões and Antunes, 2021). These factors, coupled with a lowered antibiotic efficacy and changes to the human microbiome, could have severe consequences on the health of astronauts, especially in the event of an infection with microorganisms potentially modified by the extraterrestrial planetary conditions (Klaus and Howard, 2006; Allen et al., 2022).
We want to highlight how our study is a starting point to evaluate the survival of human-associated pathogens to Mars-like conditions. Key questions this study investigates include the following: Will terrestrial microorganisms be able to survive the martian environment, especially that in the proximity of human habitats, which will be likely more favorable for microorganism survival? To what extent do they survive? Are there specific Mars conditions/characteristics that improve the survival of the bacteria? If they do, are they modified by the conditions on Mars and what will be the risks for human health upon re-exposure?
Materials and Methods
Growth in minimal media to select the optimal (single) carbon source
Bacterial species were ordered from Leibniz Institute DSMZ-German Collection of Microorganisms and Cell Cultures GmbH, and are the following: B. cepacia (DSM 7288), K. pneumoniae (DSM 30104), S. marcescens (DSM 30121), and P. aeruginosa (DSM 22644). All species were grown on nutrient-broth (NB) (Difco) agar (15 g/L), at 35°C overnight in biological triplicates. After incubation, the colonies were resuspended in M9-complete minimal salts media containing the following: 47.8 mM Na2HPO4, 22.0 mM KH2PO4, 8.6 mM NaCl, 3.7 mM NH4Cl, 2 mM MgSO4, 0.1 mM CaCl2, and 0.02 mM Fe(III)Cl3, without carbon source, as previously described by Domínguez-Andrés et al. (2020). The optical density at 600 nm (OD600), of the bacterial culture, was measured after dilution in the M9-complete media with 0.2% (w/v) of a carbon source.
The carbon sources used were
Starting cell numbers were the following: 2.85 × 106 cells of P. aeruginosa, 6.91 × 106 cells of B. cepacia, 1.93 × 106 cells of S. marcescens, and 6.05 × 106 cells of K. pneumoniae. Each well contained 190 μL of media with an additional 10 μL of cells grown in NB. In parallel, bacterial cultures were diluted in series with phosphate-buffered saline (PBS), plated on NB, and incubated overnight to determine colony-forming unit (CFU) counts.
Growth in the presence of sodium perchlorate at Mars-like concentrations
The bacteria were inoculated in M9 complete with
Growth with supplementation of Mars Global Simulant 1 to the growth media
To evaluate the survival and/or growth of the different bacterial species in the presence of Mars regolith, the cells were diluted in M9-gluconic with the addition of Mars Global Simulant 1 (MGS-1; Exolith Lab, FL), see Cannon et al. (2019) for regolith composition and Supplementary Tables S1 and S2, at different concentrations (% w/v) in 250 mL Erlenmeyer flasks and incubated overnight with 125 rpm orbital shaking at 35°C. Electron microscopy pictures were taken with the Hitachi™ 3000 (Hitachi, Tokyo, Japan) at 100 × , 400 × , and 1000 × magnification following the manufacturer's protocol, see Supplementary Fig. S2. The MGS-1 was presterilized by autoclaving before addition to the growth media. The concentration of MGS-1 was evaluated from 0.3% (w/v) to 12% (w/v) to determine whether there would be a toxic effect to these bacteria.
Monitoring cell survival and/or growth with OD600 measurements and microscopy cell counts was not possible due to the turbidity caused by the regolith simulant. Therefore, cell counts were determined by dilution plating on NB agar plates that were incubated overnight at 35°C.
Survival after desiccation treatment
The bacteria were grown in M9-gluconic overnight to have them in late-exponential phase. Aliquots of 200 μL were placed on sterile 1 cm diameter glass disks and left to dry for 24 h at room temperature under sterile conditions with a relative humidity of ca. 30% ± 10% in a working clean bench. The surface area of each glass disk is 0.7854 cm2 and based on this value and the average surface area of a cell of each species, we calculated the number of cells we have on each disk fit; we therefore assume they are in a monolayer. Table 1 summarizes the calculations performed.
Calculated Number of Cells on Glass Disks Used in the Desiccation Experiments
Calculated Number of Cells on Glass Disks Used in the Desiccation Experiments
To determine the survival rate, each glass disk with desiccated cells was placed in a 5 mL tube filled with 1 mL of PBS and lightly vortexed. A dilution series with PBS was performed to determine the survival rate based on CFU counts. The same experiment was performed with cells grown in M9-gluconic supplemented with 5% (w/v) MGS-1.
To understand how the bacteria survive to UV-C (254 nm), irradiation was performed by growing them in M9-gluconic overnight. A volume of 2700 μL of each bacterial culture was placed in closed sterile UV transmissible quartz cuvettes (0.5 cm path-length; Hellma GmbH & Co. KG, Muelheim, Germany) and placed vertically at 32.5 cm from the UV source described below. Each cuvette contained a small magnetic stirrer, which allowed the bacterial cultures to be stirred while being radiated, to have a uniform exposure of most of the bacterial cells. UV-C irradiation was produced by a low-pressure Hg lamp (Model NN 8/15; Heraeus, Berlin, Germany), which was allowed to warm up for >30 min before exposure (Supplementary Fig. S1). To measure the fluence of the lamp at the sample site, a regularly calibrated UVX radiometer with a UV sensor (UV-25) for 254 nm (both UVP Ultra-Violet Products, Cambridge, United Kingdom) was used.
The UV-C fluence rates were 170 μW/cm2 and to reach the maximum dose of 250 J/m2 at 32 cm distance from the light source it took 147 s. The same experiment was performed with polychromatic Mars-like UV (200–400 nm) radiation using the SOL2 polychromatic lamp, equipped with a UV 500S irradiation source (Dr. Hoenle AG; UV-Technologie, Germany) at the German Aerospace Center in Cologne, Germany (DLR, 2020). The fluence of the lamp was determined by using the Bentham DMc150 transportable spectroradiometer (Bentham Instruments Ltd., Reading, United Kingdom), with optics inside the simulation chamber as per Rabbow et al. (2005). The polychromatic UV fluence rates were 10.35 W/m2 at 112 cm distance from the light source, and to reach the maximum dose of 1200 J/m2, it took 116 s.
Survival was determined after exposure to UV-C and polychromatic-UV by dilution and plating of the bacterial cultures on NB agar plates and incubation overnight at 35°C. Before dilution plating, during total irradiation times, samples were briefly stored in the dark on ice (at 4°C). Media absorbance measurements were performed with the use of the Thermo Scientific™ NanoDrop™ 2000c, in a UV-transparent 1 mL cuvette. Media absorbance measurements were also taken to determine possible absorption effects.
Survival to polychromatic UV radiation in growth medium with and without supplementation of 5% (w/v) MGS-1
The same experimental procedure was performed with cells grown in M9-gluconic and with supplementation of 5% (w/v) MGS-1.
Survival after exposure to 1 day of desiccation and to polychromatic UV radiation
To determine the survival of the bacteria to the combined effect of two Mars-like conditions consecutively, late exponential cell cultures were exposed to desiccating conditions as described above. After 24 h of desiccating conditions, the glass disks with the dried cells were exposed to polychromatic UV irradiation. The disks were exposed to the desired UV fluences. Survival was determined by CFU counts.
Survival in growth medium supplemented with 5% (w/v) MGS-1 after desiccation and exposure to Mars atmosphere and pressure in combination with polychromatic UV radiation
The survival of the species to martian atmosphere and pressure was evaluated by first desiccating the cells in combination with 5% (w/v) MGS-1, as described in Section 2.6. The second step was to store the desiccated cells on glass disks in the adapted transport and exposure (TREX)-box, as described by Beblo-Vranesevic et al. (2017a) and Cortesão and Siems et al. (2021). The TREX-box was connected to a gas supply of Mars-like atmosphere (2.7% N2, 1.6% Ar, 0.15% O2 in CO2 vol/vol) and a vacuum pump (rotary vane pump DUO 035 and HIPace 700; Pfeiffer Vacuum GmbH, Germany) to apply the desired pressure (6 hPa). The TREX-box was monitored constantly during the exposure (TPG 262 Full Range Gauge; Pfeiffer Vacuum GmbH).
Samples were exposed to Mars-like atmosphere and pressure (6 hPa) for 24 h in the gas-tight TREX-box. The box, using a suprasil window, enabled full UV-visible exposure of the samples during polychromatic UV radiation while under desiccating conditions and Mars-like pressure and atmospheric composition. Similar Mars-like exposures with the use of the TREX box were performed by Cortesão and Siems et al. (2021). All figures were created with the use of the data visualization library Seaborn version 0.12.1 (Watson, 2021), an interface of Matplotlib (Hunter, 2007).
Results
Assessment of the growth on single extraterrestrial carbon sources
The study described by Domínguez-Andrés et al. (2020) showed that the selected nonfastidious bacterial species (B. cepacia, K. pneumoniae, P. aeruginosa, and S. marcescens) were able to tolerate and grow under minimal media conditions supplemented with carbon sources that are also present in carbonaceous meteorites. For the survival assays of this study, we first evaluated which carbon source (
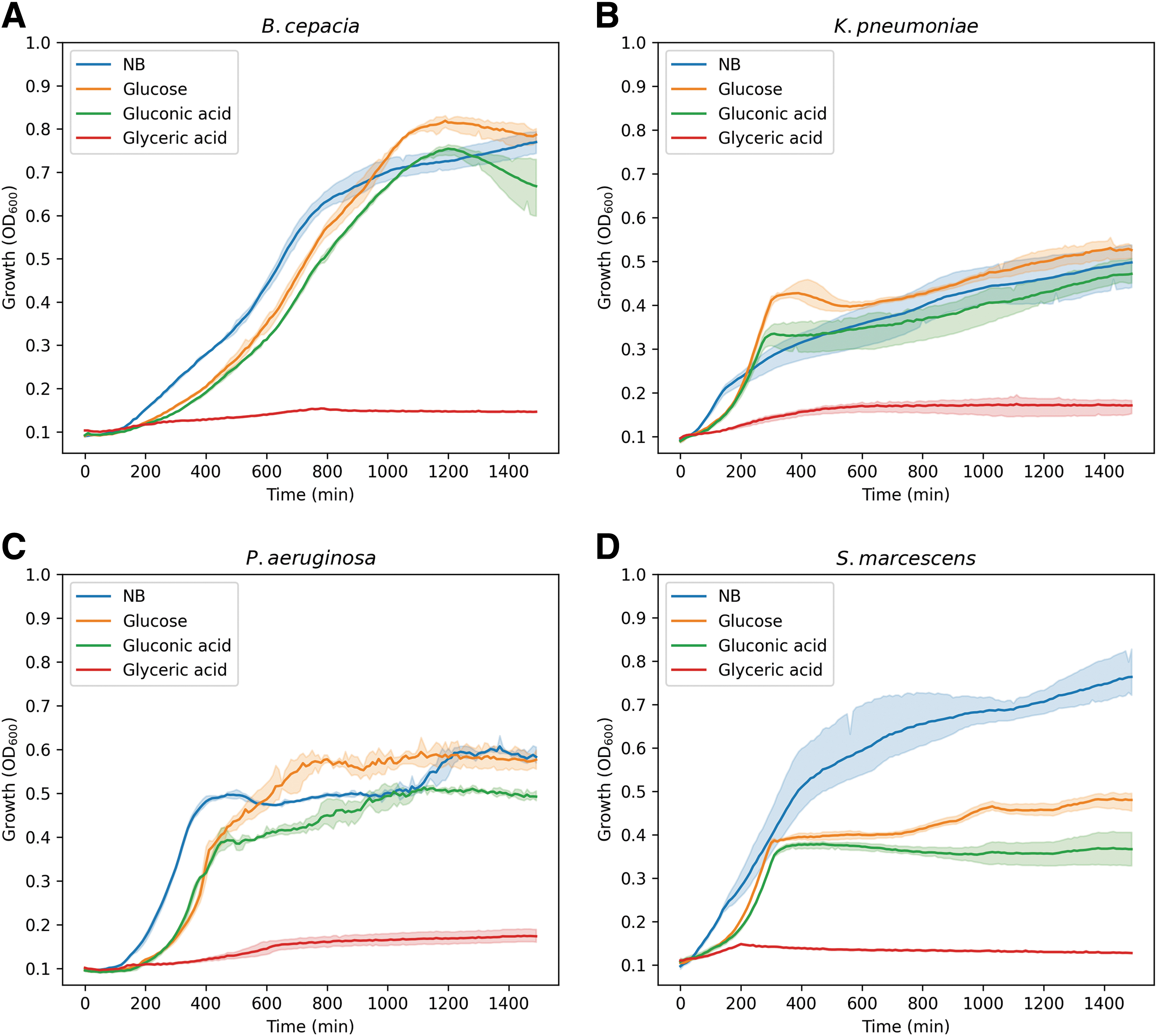
Nonfastidious bacteria are capable of growing in M9 supplemented with a single carbon source. Growth curves of the nonfastidious bacteria in M9 supplemented with a single carbon source. The graph shows the growth curves in nutrient broth (blue), M9-glucose (orange), M9-gluconic (green), and M9-glyceric (red). The curves represent the OD600 measurements taken over a period of 24 h at 35°C. The color shading represents the standard deviation of the OD600 values for the three biological triplicates. Burkholderia cepacia
To understand whether the bacteria would be capable of growing in M9 medium, with a single carbon source, in the presence of Mars-relevant sodium perchlorate concentrations of 35 and 70 mM (Clark and Kounaves, 2016), we inoculated M9-gluconic and M9-glucose (positive control) with 35 and 70 mM sodium perchlorate. Despite these challenging growth conditions, all species showed growth; however, a longer lag time before the exponential phase was observed (Fig. 2). Furthermore, none of the bacteria achieved the same maximum OD600 values as in the controls. When analyzing the results in more detail, one can observe that B. cepacia was able to grow in the presence of sodium perchlorate only when glucose was provided as a carbon source.
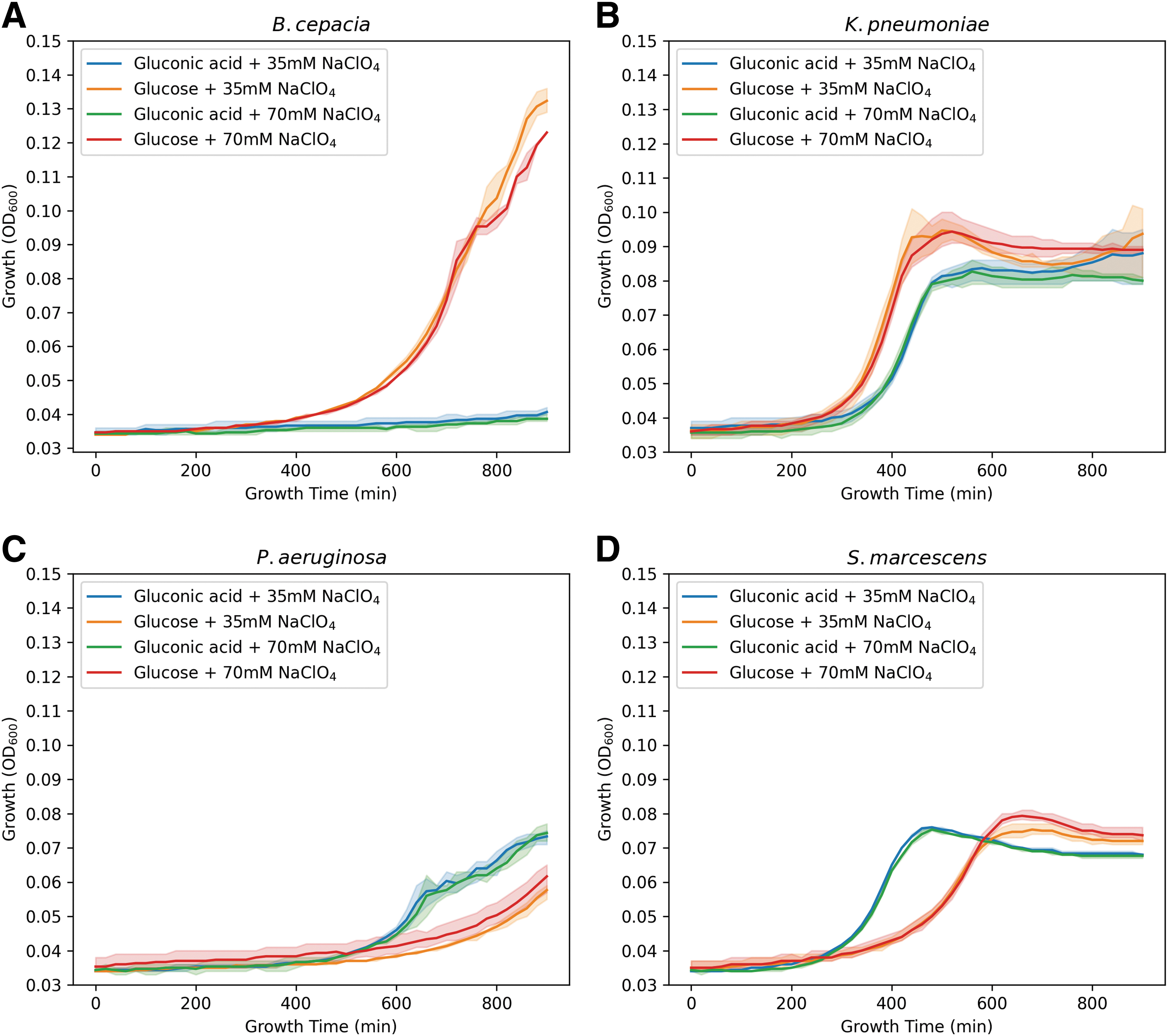
The bacteria are capable of showing growth with 35 and 70 mM of sodium perchlorate in M9. Growth curves of the bacteria with Mars-relevant concentrations of sodium perchlorate. Growth curves measured by OD600 of the bacteria inoculated in M9 with a single carbon source and the addition of sodium perchlorate inoculated over a period of 15 h at 35°C, Burkholderia cepacia
Growth of K. pneumoniae was hardly affected by the presence of sodium perchlorate; only an increase in the growth was observed in the media with glucose, as was also found for the other species (P. aeruginosa and S. marcescens). In general, growth of P. aeruginosa in all media was limited. Interestingly, in the case of S. marcescens, the cells were able to reach the stationary phase more rapidly in M9-gluconic acid, than in the media supplemented with glucose (Fig. 2). Similar growth rates were seen when the bacteria were incubated with increased sodium perchlorate concentrations, except for B. cepacia where hardly any growth was measured (Fig. 3). However, this oxidizing stressor caused an even greater reduction in growth speed and concentration.
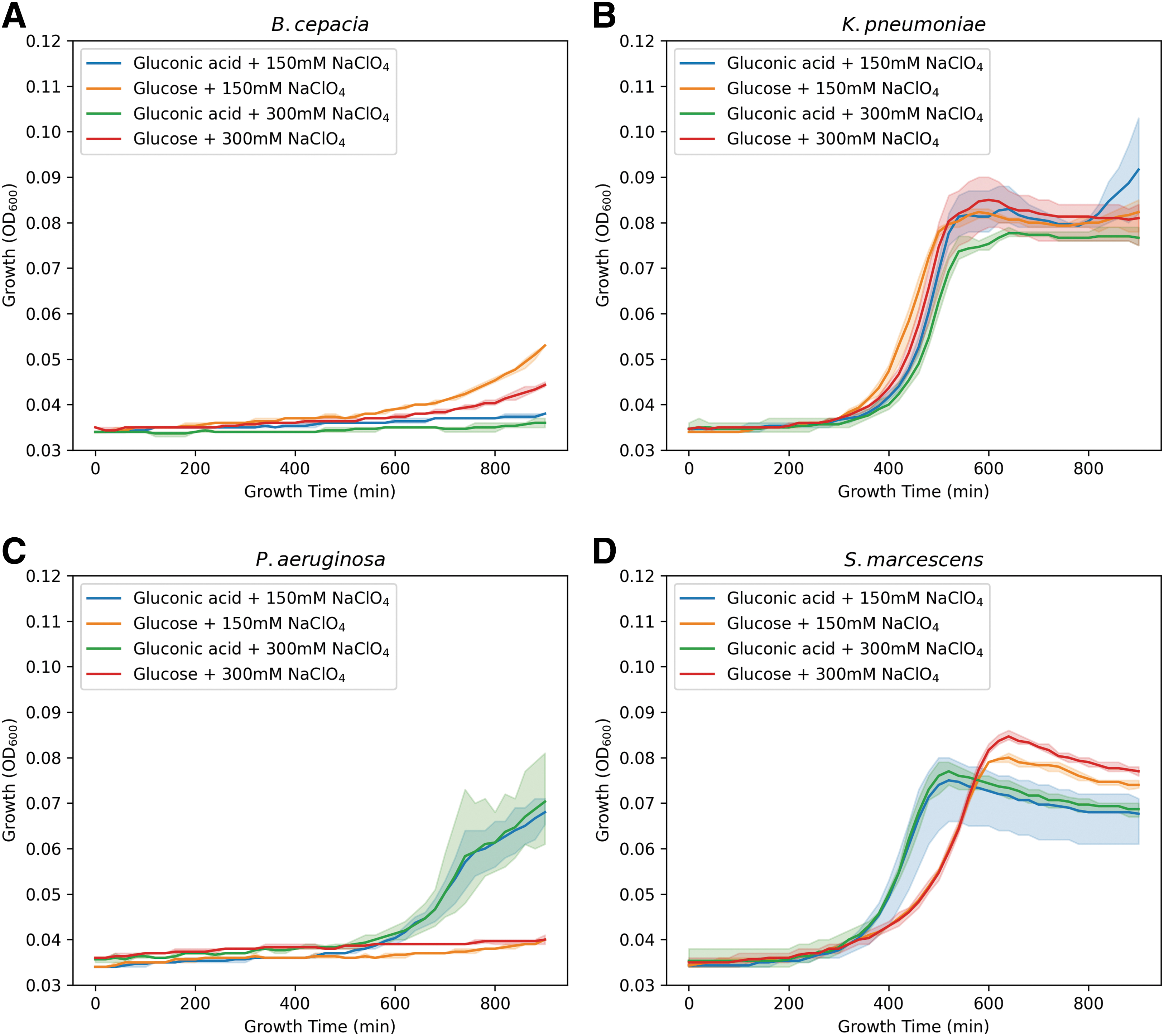
Evaluating the growth in the presence of higher perchlorate concentrations. Growth curves of the bacteria with increased concentrations of sodium perchlorate. Growth curves measured by OD600 of the bacteria inoculated in M9 with a single carbon source and the addition of sodium perchlorate inoculated over a period of 15 h at 35°C, Burkholderia cepacia
To understand whether the bacteria could grow in the presence of simulated Mars regolith, as found in martian brine or ponds, we incubated the species in M9-gluconic acid in the presence of different concentrations (0.3–12% w/v) of MGS-1 (Fig. 4). The control culture was not supplemented with MGS-1. It was found that most concentrations of MGS-1 stimulated rather than inhibited growth. In 1% (w/v) MGS-1, all species showed a higher colony count than the control. In the case of P. aeruginosa, the difference in CFU was almost four orders of magnitude higher. Concentrations of MGS-1 in the media were tested up to 12% (w/v) to understand whether the simulated regolith would be limiting for growth; however, this was not observed.
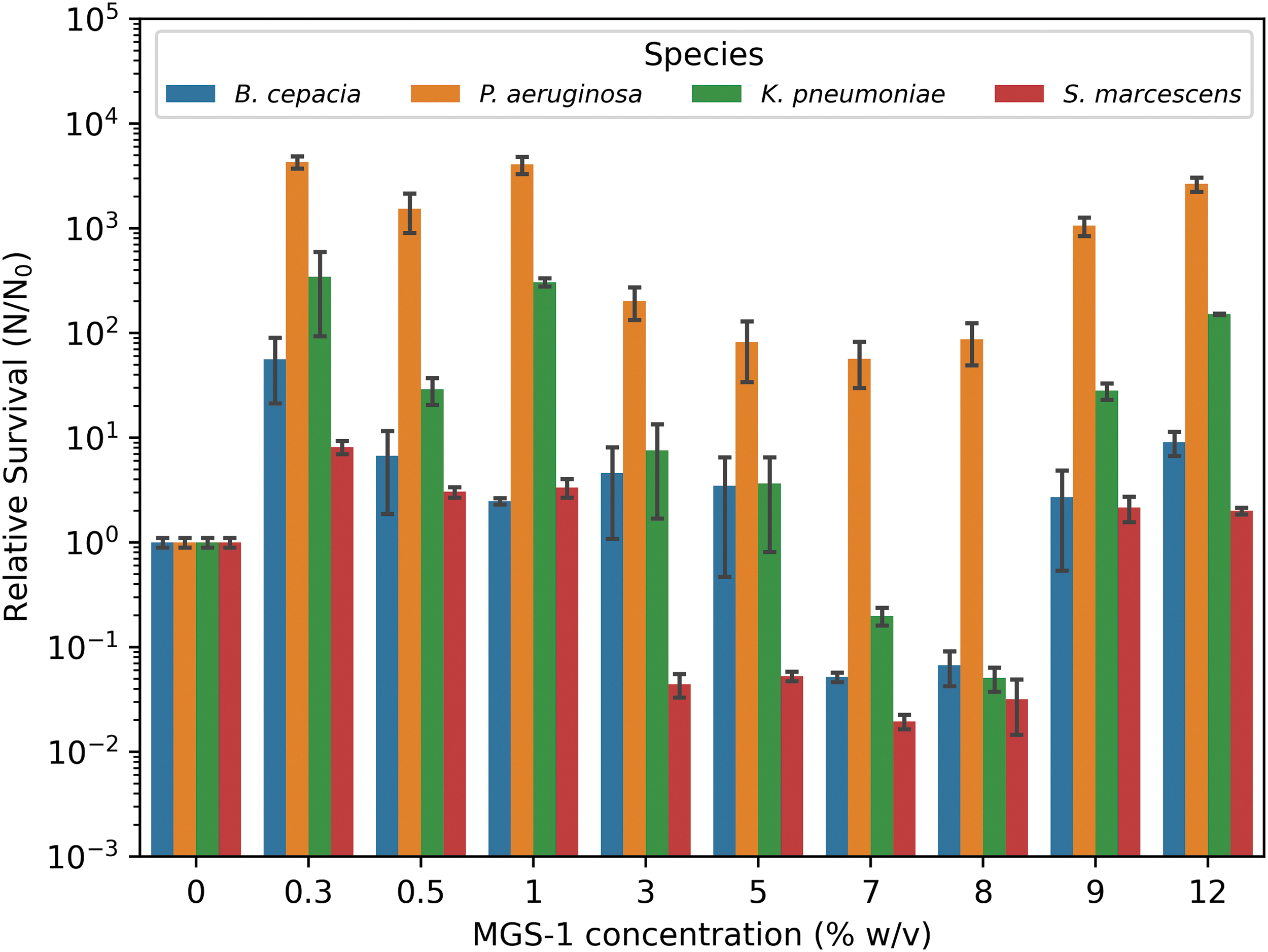
Evaluating the growth in the presence of MGS-1. Growth in the presence of MGS-1. The regolith simulant concentration was tested from 0.3% to a maximum of 12% (w/v) in liquid M9-gluconic media. The colored bars represent the colony counts of the bacteria inoculated with MGS-1 and incubated overnight at 35°C. MGS-1 = Mars Global Simulant 1.
For all the subsequent exposure experiments, an MGS-1 concentration of 5% (w/v) was chosen as this was the optimal concentration for making dilution series for CFU counting, enabling higher throughput experiments. A concentration of 5% (w/v) is considered relevant in terms of exposure of the bacterial species to the regolith components, based on which predictive conclusions can be drawn.
To understand how bacterial species from Earth would survive under such dry conditions, we assessed the desiccation of the different species. Before desiccation, cells were grown in M9-gluconic acid medium to late exponential phase, at room temperature (22°C ± 4°C). The only species that survived desiccation and showed survival up to a maximum of 16 days were K. pneumoniae and S. marcescens (Fig. 5). When bacteria were grown in combination with 5% (w/v) MGS-1, all species survived at least 13 days of desiccation. This is a substantial increase in survival especially for B. cepacia and P. aeruginosa, which previously did not survive any desiccation time frame. Furthermore, the maximum survival for both K. pneumoniae and S. marcescens increased from 16 to 22 days.
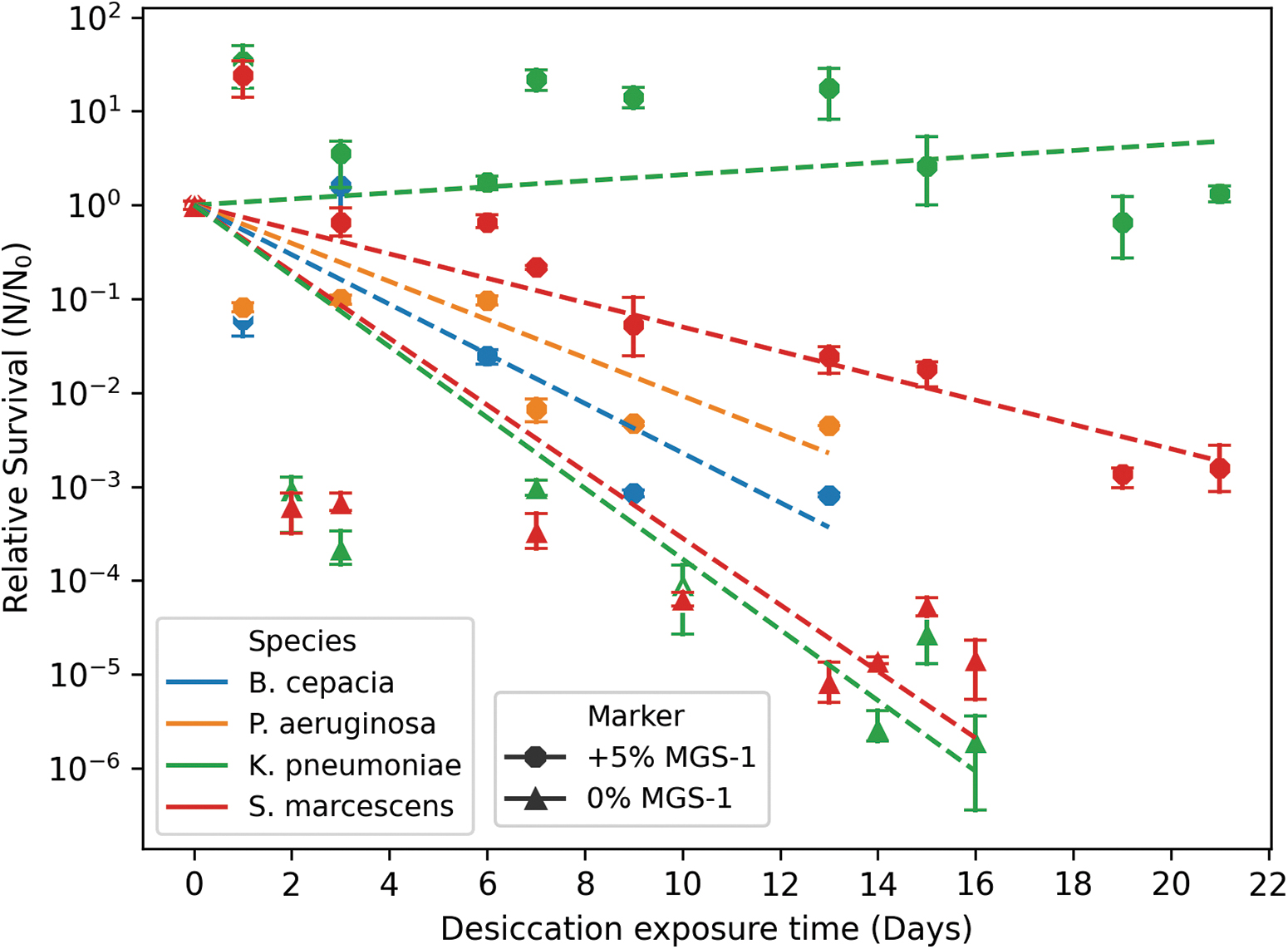
Desiccation survival of the bacteria in the presence of 5% (w/v) MGS-1. Relative survival of the bacteria when exposed to desiccating conditions. All the bacteria were able to survive this condition when supplemented with 5% (w/v) MGS-1. When grown in M9-gluconic, Klebsiella pneumoniae (green triangle) and Serratia marcescens (red triangle) were the only species surviving, while Burkholderia cepacia and Pseudomonas aeruginosa did not survive, points not shown. The survival with MGS-1 can be seen for all species, K. pneumoniae (green circle), S. marcescens (red circle), B. cepacia (blue circle), and P. aeruginosa (orange circle). The survival of all the species under all conditions was evaluated until 21 days. The displayed survival shows the limit to which it was detected.
To measure the resistance of bacteria against UV irradiation, we exposed the different species to UV-C (254 nm) and polychromatic UV (200–400 nm) radiation at different fluences. We found that P. aeruginosa and S. marcescens showed the highest resistance against both UV-C and polychromatic UV (Fig. 6). The calculated F10 fluences (fluence to which 10% of the population survives) support this finding. The calculated values were 146 and 102 J/m2 for UV-C and 736 and 737 J/m2 for polychromatic UV, for each species, respectively. The two other species, B. cepacia and K. pneumoniae, had a much lower survival limit, and the calculated F10 values were 23 and 45 J/m2 for UV-C and 69 and 35 J/m2 for polychromatic UV, respectively.
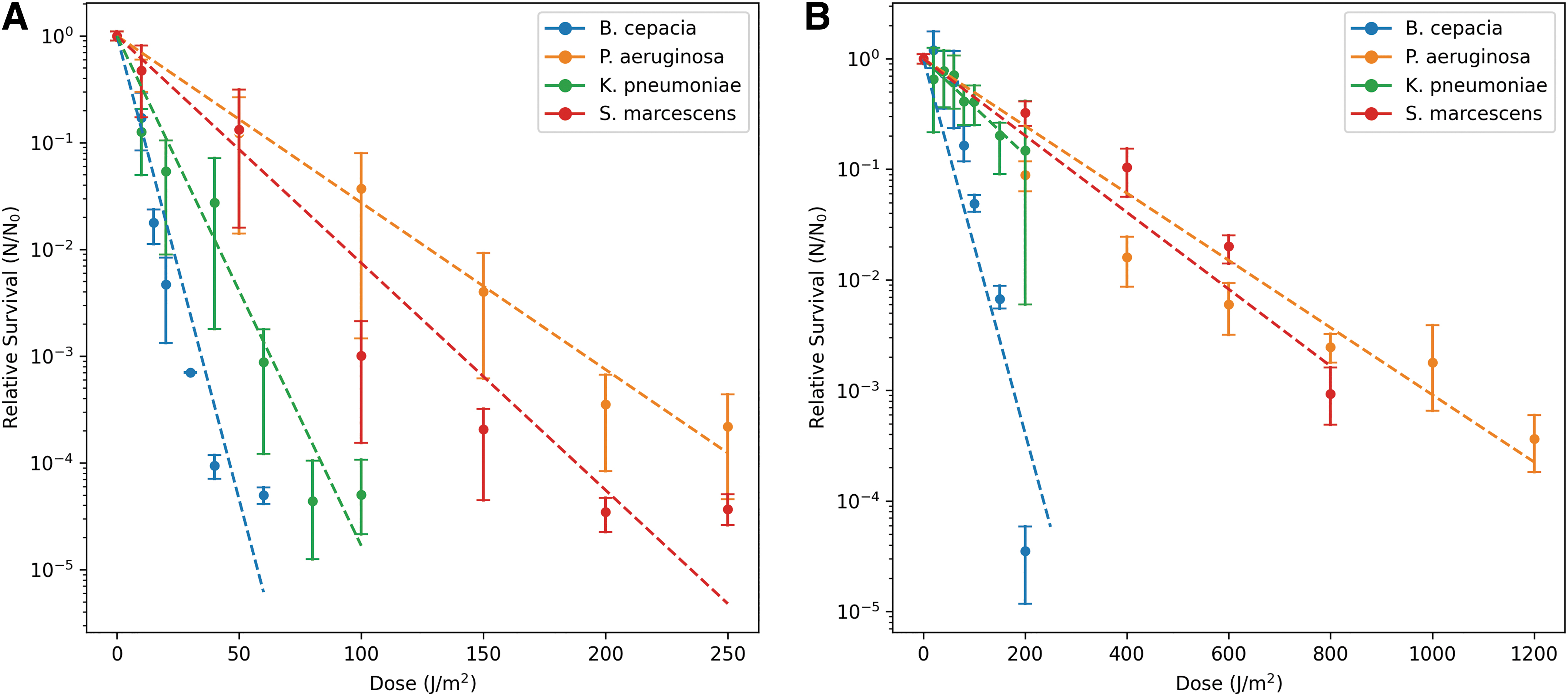
Relative survival of all the bacteria to UV radiation. Maximum radiation doses for UV-C (254 nm) was 250 J/m2
The bacteria were exposed to a combination of Mars-like conditions to closely replicate all the conditions present on Mars. The first experiment was conducted with K. pneumoniae and S. marcescens, the only species that showed survival to desiccation to a combination of desiccation with polychromatic UV radiation. Only S. marcescens was able to survive under these conditions (Fig. 7B). Comparing the F10 of the species exposed to polychromatic UV grown with and without MGS-1, there is a clear increase in survival when the media are supplemented with MGS-1 (Fig. 7A).
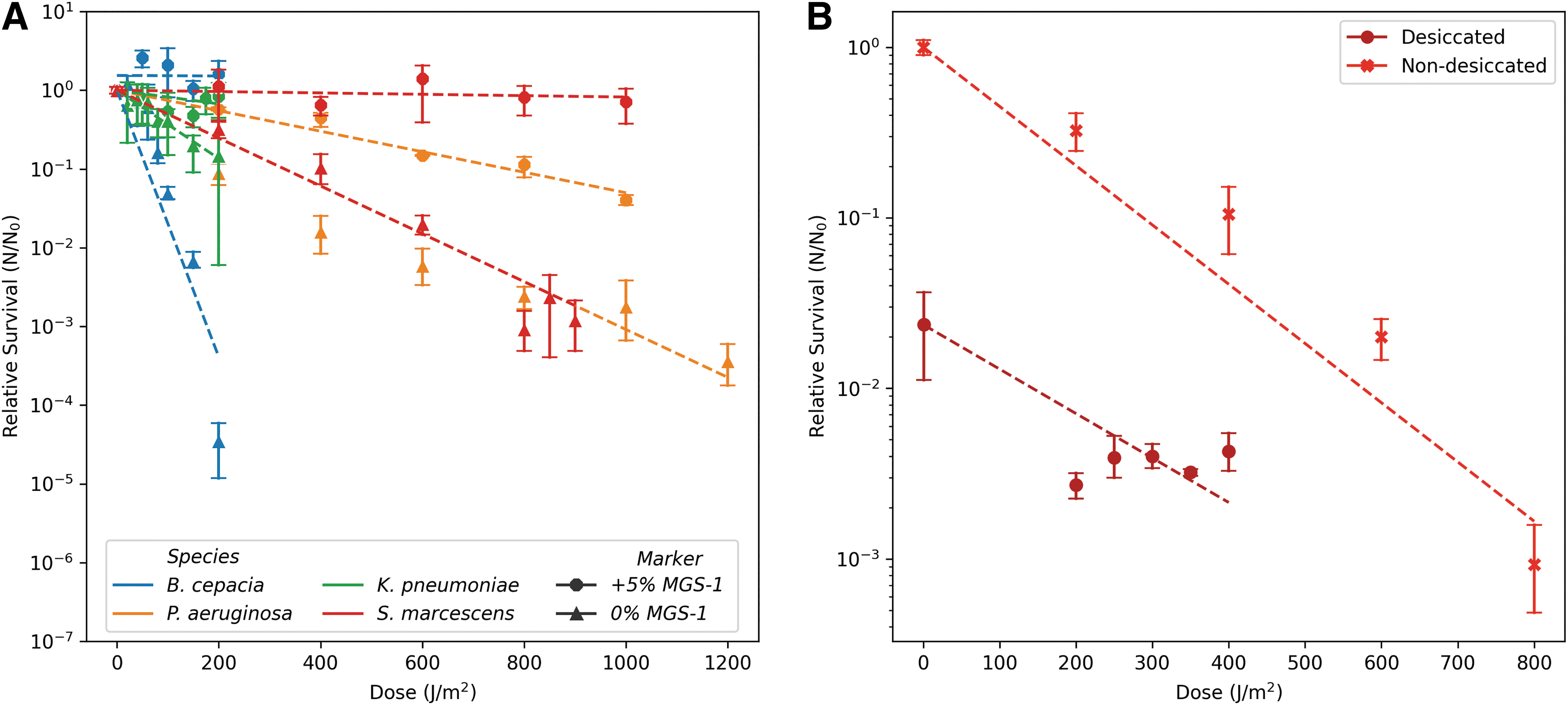
Survival of the bacteria to combined conditions. Relative survival of the bacteria to polychromatic UV radiation with or without MGS-1
Furthermore, we evaluated cell survival following exposure to the combined martian conditions. This included growth in combination with MGS-1, desiccation, UV irradiation, as well as martian atmosphere and pressure. Under these conditions the cells were desiccated overnight after being grown in combination with 5% (w/v) MGS-1, placed in a TREX-box with Mars-like atmosphere and pressure and irradiated with 150 J/m2 of polychromatic Mars-like UV radiation. This dose would be equivalent to ∼12 s (12 s × 12.49 W/m2) on the martian surface assuming the Mars solar constant is 49.95 W/m2, no absorption by the atmosphere, and that only half of the time one hemisphere of the planet is irradiated with polychromatic UV (200–400 nm) radiation (Schuerger et al., 2003). In Fig. 8, the survival of the species to these conditions is presented.
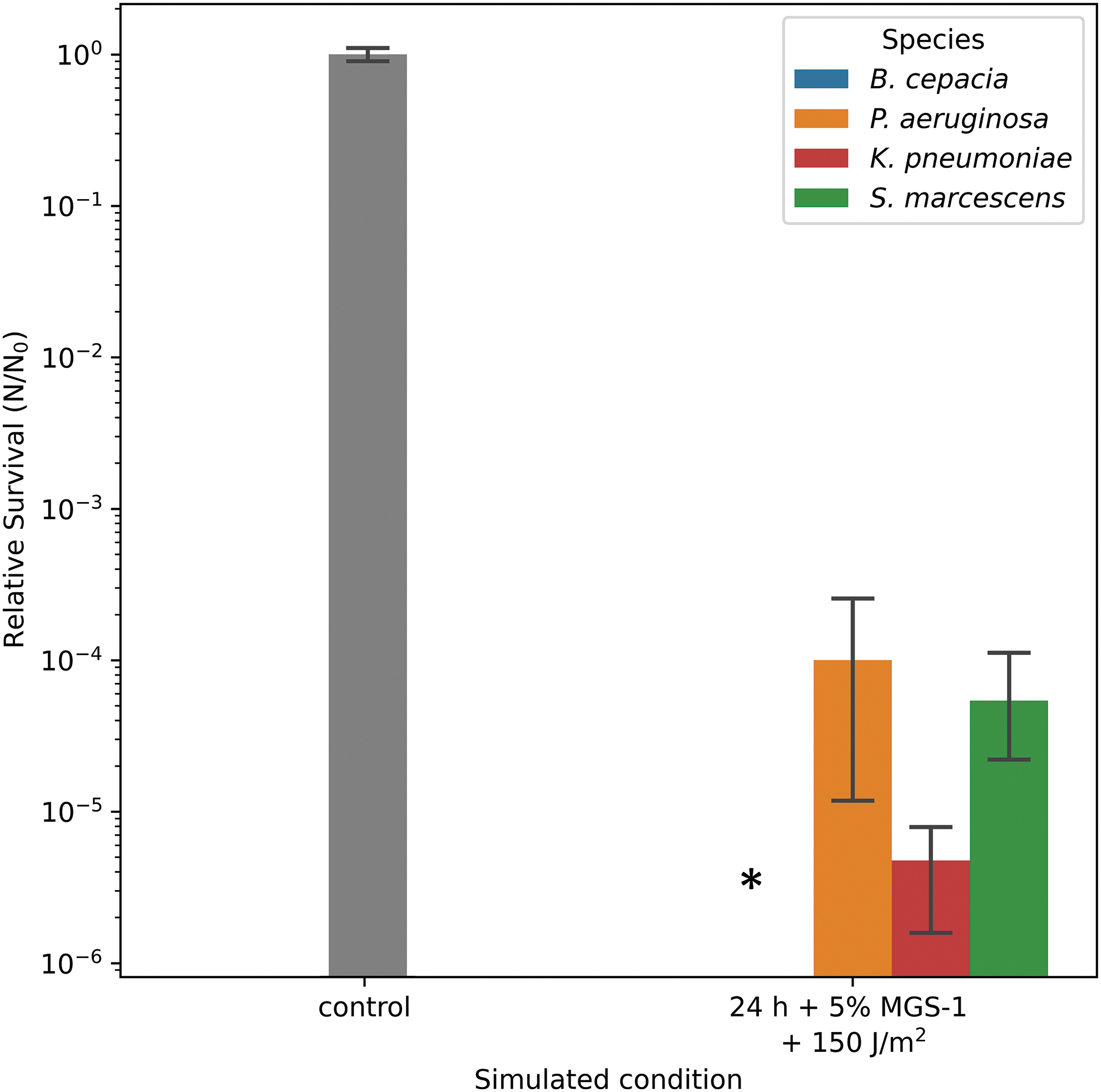
Survival of the bacteria to combined Mars conditions. All the bacteria were grown in M9-gluconic in combination with 5% (w/v) MGS-1, desiccated o/n, and exposed to martian atmosphere and pressure for 24 h. All the bacterial species were able to survive these combined conditions with the exception of Burkholderia cepacia (*).
All species, except B. cepacia, were able to survive these conditions, with P. aeruginosa and S. marcescens showing the highest degree of survival (one order of magnitude higher than K. pneumoniae). These results show that all bacterial species that we have analyzed are able to survive, to a certain extent, under martian simulated conditions. However, the amount of time the bacteria can survive in the simulated environment varies and is generally limited.
The Earth harbors many nonfastidious bacteria in the environment, a number of which can cause opportunistic infections. To protect the crews of future Mars missions, it is essential to assess the capabilities of these potential pathogens to survive the martian environment. Understanding their resistance to these extreme conditions can help with the selection of targeted disinfectants. Moreover, understanding the survival and likely changes that will be undergone by these bacteria will be very important to understand their future interaction with humans and the capacity to cause disease. Past literature has investigated the survival of different human-related bacteria to simulated Mars and interplanetary spacecraft conditions, including the food contaminant Bacillus subtilis (Schuerger, 2022; Schuerger et al., 2023), the pathogen Enterococcus faecalis (Godin et al., 2020), as well as Serratia liquefaciens (Mickol et al., 2017).
In the present study, we show that nonfastidious human pathogens can survive for days to weeks under Mars-like conditions. Importantly, it seems that the presence of Mars regolith improves the survival of these microorganisms.
We investigated the survival of the gram-negative bacteria B. cepacia, P. aeruginosa, K. pneumoniae, and S. marcescens to Mars-like conditions. Previous work with these species has shown that they are able to grow in different types of oligotrophic media with a range of singularly provided carbon sources (Domínguez-Andrés et al., 2020). These findings are particularly remarkable since these bacterial species are typically grown in rich media. However, being pathogenic they could also be well suited to grow or survive in adverse environments such as the human body. Our study reports concurrent findings for cells grown in M9-complete minimal media supplemented with
The effect, or lack thereof, can be explained due to glyceric acid having a lower amount of carbon atoms (3) than gluconic acid (6). Experiments with adjusted concentrations of carbon atoms as gluconic acid showed the same growth rate as the original concentration. It may be suggested that the bacteria would show a higher growth rate with gluconic acid due to its closer molecular similarity to glucose.
Data have shown that most of the liquid water that would be available on Mars would be in the form of perchlorate brines, within hydrated minerals, or as ice frozen beneath the surface (Martínez and Renno, 2013; Toner and Catling, 2018; Wray, 2021). Moreover, it is likely that the presence of water and temperature changes will change Mars conditions in the proximity of future human habitats on Mars. Therefore, it was important for us to first understand whether the selected species would be capable of surviving and/or growing in these environments. Analyzing the growth curves of the different species in simple salts medium (M9) supplemented with Mars-relevant sodium perchlorate concentrations and in oligotrophic media with only one C source, we can see how all the species differently tolerate this condition. Both K. pneumoniae and S. marcescens showed growth in media supplemented with glucose and gluconic acid.
However, B. cepacia showed growth only in glucose-supplemented media, while P. aeruginosa growth was greatly limited with both sugars. Genes responsible for perchlorate-resistance have been identified in the Atacama Desert, a Mars analog site on Earth, and research has shown that they can be expressed in bacteria such as Escherichia coli (Díaz-Rullo et al., 2021). A strain of S. marcescens NIIST5, isolated from a laboratory-scale fed-batch bioreactor, contained genes involved in the perchlorate reduction pathway, such as perchlorate reductase and chlorite dismutase (Vijaya Nadaraja et al., 2013). However, this strain is different from the one used in this study. Furthermore, we are not aware of other perchlorate reducing genes in the additional three investigated bacterial species. Nonetheless, this could suggest that a similar mechanism of perchlorate reduction could take place on Mars, for example, through the process of gene transfer.
Consequently, the transfer of perchlorate reducing genes could support even better growth of the bacteria in perchlorate brines. This is of evermore greater importance, as we have shown that the bacteria can tolerate higher concentrations of sodium perchlorate (Fig. 3). Earlier studies have investigated the resistance of S. marcescens to perchlorate (Vijaya Nadaraja et al., 2013), but this research did not simulate Mars-relevant concentrations. Further investigation is required into the growth potential of the species with other Mars-identified perchlorates.
Having shown that the species can grow in M9-gluconic and, to an extent, in the presence of sodium perchlorate, understanding how the species might interact with the regolith was the next step in replicating martian conditions. Interestingly, all the bacteria incubated up to 1% (w/v) MGS-1 showed increased cell proliferation compared with M9-gluconic acid alone. Research has shown that some bacteria can make use of rocks and minerals to obtain energy through redox reactions (Olsson-Francis et al., 2017; Ramkissoon et al., 2019). Increasing the MGS-1 concentration in the growth media past 1% (w/v) did not necessarily result in higher cell yields for all species studied. In our experiments, P. aeruginosa is the only species that showed a greater cell number after incubation in all MGS-1 concentrations, compared with the control.
In all further experiments where MGS-1 was supplemented, a concentration of 5% (w/v) MGS-1 was used to more closely represent the conditions on Mars, and to have more efficient control over the volumes. Due to the particular composition of the regolith simulant and the grain size distribution of <0.04 to 1000 μm, we speculate that the regolith could shield the bacteria from a wide range of adverse conditions. In addition, on Mars, the bacteria would be desiccated in a mixture with regolith. Under this assumption, we speculate that the structural composition of the MGS-1 could allow the retention of traces of water, which would enable the bacteria to have an increased survival (Supplementary Fig. S2). Undoubtedly, further investigations into the interaction of these species with the regolith are warranted.
Evaluating the survival of bacteria to desiccating conditions is an area of research that has been extensively investigated, considering its importance for the Mars environmental characteristics. Our results are particularly interesting as they show how two bacterial species, K. pneumoniae and S. marcescens, were able to survive desiccation for up to 16 days when grown in minimal media, M9-gluconic. The survival of these two species in this study is particularly remarkable when compared with other bacteria, which can only survive these conditions if grown in nutritionally rich media—E. coli O15:H7, for 8 days (Stasic et al., 2012) and Mycobacterium tuberculosis, 1 week (Potts, 1994). Other extremophilic microorganisms such as the (hyper-)thermophiles Hydrogenothermus marinus and Aquifex aeolicus can survive to the same desiccating conditions for 28 days, when cultivated in species-specific standard conditions (Beblo et al., 2009).
The survival of the two species analyzed is interesting when considering the cultivation conditions are in minimal media. When desiccation is combined with polychromatic UV exposure, the only species that survived was S. marcescens. The effects of desiccation alone can lead to changes to the cell structures, protein denaturation, and DNA strand-breaks (Beblo-Vranesevic et al., 2017b). The combined effects of polychromatic UV radiation and desiccation led to a maximum dry-state survival of 1 day. Due to the inability of the cells to be metabolically active in a dry state, the mechanisms of DNA repair most probably could not be activated and shielding of S. marcescens could only be provided with the available intracellular pigment.
Of further interest is the survival of all the species when cultivated in M9-gluconic supplemented with 5% (w/v) MGS-1. All the bacteria were able to survive at least up to 13 days. Furthermore, the bacteria that were able to survive desiccation without MGS-1 had an increase in their tolerance from 16 to 21 days. Other studies have also shown how archaea, used as model organisms for life on Mars, were able to survive more extensively to desiccation when supplemented with regolith simulants (Kral and Altheide, 2013; Schirmack et al., 2015). We can speculate that the regolith might enable the retention of water, which could support the survival of the bacteria for longer periods of time.
Despite Mars having a lower solar constant (49.95 W/m2) compared with Earth (116.16 W/m2), for total UV irradiance (200–400 nm), the thinner and differently composed atmosphere allows more of the short-wavelength solar radiation to reach the martian surface (Patel et al., 2004; Paulino-Lima et al., 2010). To understand how the bacteria can survive these conditions, we exposed them to UV-C (254 nm) and polychromatic Mars-like UV (200–400 nm) radiation. The UV spectrum of our SOL2 lamp (Supplementary Fig. S1) is not a replication of what can be seen on the surface of Mars as was seen by Delgado-Bonal et al. (2016) and Vicente-Retortillo et al. (2015); however, it shows us the effect of simulated UV irradiation on the survival of the bacterial species.
Both S. marcescens and P. aeruginosa have repair mechanisms that we wanted to limit in this study. These include S. marcescens’ photolyase enzyme, nucleotide excision repair (NER) (Fernández Zenoff et al., 2006), and P. aeruginosa's DNA polymerase IV mechanism (Sidorenko et al., 2017). To avoid the activation of these repair mechanisms, as much as possible, samples were briefly stored in the dark on ice, before dilution plating during total irradiation times. Our results have shown that the two most UV tolerant species, P. aeruginosa and S. marcescens, are also capable of repairing their DNA with enzymes activated with certain wavelengths of light, such as with photolyase.
The increased survival of these two species compared with the other bacteria in this study could be attributed to the red prodigiosin pigment of S. marcescens (El-Bialy and Abou El-Nour, 2015) and several pigments produced by P. aeruginosa, such as pyoverdine, pyocyanin, pyorubin, and pyomelanin (Kothari et al., 2022). The pigments, coupled with the NER and polymerase IV DNA repair mechanisms, can give the two bacteria a great advantage when exposed to UV radiation. This relationship can especially be seen when analyzing the F10 values of the organisms when exposed to UV-C and polychromatic UV. Some model organisms have been shown to have similar F10 UV-C doses such as Salinisphaera shabanensis (56 J/m2) and Buttiauxella sp. MASE-IM-9 (42 J/m2). However, others have demonstrated higher F10 values such as Deinococcus radiodurans, 660 J/m2 (Beblo-Vranesevic et al., 2022).
This study has shown that supplementing growth media with MGS-1 can increase the tolerance of certain species to several Mars simulated conditions. This effect is also present when bacteria are grown in 5% (w/v) MGS-1 and exposed to UV-C and polychromatic UV radiation. We suspect that regolith grains provide partial protection to the microorganisms. Literature has investigated the minimum thickness or depth on Mars, which can attenuate the biocidal effect of polychromatic UV radiation (Cockell et al., 2000; Moores et al., 2007). Furthermore, Schuerger et al. (2012) reported that a 0.5 mm layer of crushed basalt can attenuate >99% of UV penetration (200–800 nm). Despite the differences in the experimental setup, similar survival characteristics can be seen in our results with the exposed P. aeruginosa and S. marcescens when grown in 5% (w/v) MGS-1.
Combining the exposure of the strains to desiccation and Mars atmosphere could closely replicate the conditions to which the bacteria would be exposed to on the surface of Mars. The effect of exposing the cells to 24 h of Mars atmosphere and 150 J/m2 of polychromatic UV radiation following an overnight exposure to desiccation can be greatly deleterious to the cells. However, only B. cepacia could not survive these cumulative conditions, although the number of viable cells was strongly decreased for the other species as well. Despite 24 h being a limited period, we wanted to describe this result as it can still be a relevant time frame for astronauts reencountering the bacterial species during routine waste disposal tasks in a possible Mars colony.
The gram-negative Acinetobacter baumannii is also an opportunistic pathogen that can survive 26 days of desiccation in a mineral medium with one carbon source, a growth condition similar to that used in this study (Zeidler and Müller, 2019). Investigations into food safety standards with Salmonella enterica (Gruzdev et al., 2011), and nosocomial safety standards with Staphylococcus capitis (Chaibenjawong and Foster, 2011), have shown how these two species can also survive desiccation. We believe that the data from such studies could prove useful, not only in identifying possible similarities in survival mechanisms, such as with compatible solutes, but also to speculate on their potential survival to Mars-like conditions. Furthermore, our research could be a relevant starting point for investigating the long-term effects of deep space on the human immune system.
We also acknowledge a number of limitations of our study. First, practical aspects precluded us to study the impact of the factors studied for long periods of time, situation likely to be incurred in practice. Second, we investigated in the present study only the impact of each of the various factors at one time, while these conditions will be likely encountered in combination on the surface of Mars. Finally, these factors may argue that even harsher conditions will be encountered by these microorganisms on Mars. However, we should also point out that the Mars conditions in the proximity of future human habitats are likely to be milder and more conducive for bacterial survival, and this also needs to be considered. All these considerations argue that future studies are needed to extend this field of investigation, some which are currently planned by our group.
In conclusion, identifying the extent to which human pathogens can survive Mars simulated conditions is the first step to determine possible health threats to astronauts. The potential of the bacteria to grow in minimal media makes them a concern for human safety and planetary protection. As illustrated here, several species can survive a range of Mars-like environments, especially when their growth is supported by the regolith simulant, MGS-1. We highlight how among the species analyzed, P. aeruginosa and S. marcescens have the highest potential of surviving the combined stressors. Bacteria were exposed to desiccation repeatedly throughout the experiments. This condition is important as the exposure to Mars atmospheric conditions will likely bring desiccating conditions (Haberle, 2015). It can be suggested that, under specific conditions, the bacteria would be very protected from exposure to UV radiation; for example, when grown with MGS-1 or under a thin layer of regolith.
Undoubtedly, further investigations into the mechanisms of survival to desiccation of the species and other gram-negative human pathogens are warranted. The identification of potential shared desiccation resistance mechanisms could aid in the targeted development of biocides and drugs for use on Mars. Moreover, this research can also aid in updating sterilization techniques for space missions, as well as identifying treatment techniques for diseases. In the present study, we only investigated a relatively limited number of bacterial species for their resistance to Mars conditions. One could envisage the selection of more resistant microorganisms when considering the thousands of species that make up the human microbiome. Understanding the selection and evolutionary process of human microbiome in Mars conditions is of urgent importance in light of the planned human missions to Mars.
In addition, studies to investigate the interactions between the potentially modified microorganisms and the host immune system will be of crucial importance to determine whether the investigated bacteria could pose a greater human health risk after exposure to Mars-like conditions. This is of even higher importance to investigate as the human immune system is highly dysregulated during space travel. The probability of disease-causing organisms making their way to Mars is highly likely when human travelers are involved. While transporting humans to Mars will be a difficult challenge, transporting the microorganisms of the human microbiome will be inevitable. Further studies that investigate how these nonextremophile organisms will respond and change once exposed to the martian environment are essential to maintaining astronaut health, preventing microbial contamination of Mars, and ultimately ensuring successful missions to Mars.
Footnotes
Acknowledgments
We are grateful to Vincent Andrew Moore and Laura Dominika Los for their assistance with the English language editing of this article. We also thank Andre Parpart for the help with the Mars atmosphere and pressure experiment setup.
Authors' Contributions
T.Z., P.R., and K.B.-V. designed the experiments. T.Z. performed the experiments, analyzed the data, and wrote the first draft, with all the authors contributing by providing feedback. M.I.d.J., J.D.-A., and M.G.N. provided advice, guidance, and conceived ideas.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The study was supported by DLR internal funds, and this project was supported by The Netherlands Organization for Scientific Research (Off Road Grant 04510012010022 to Jorge Domínguez-Andrés).
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Table S1
Supplementary Table S2
Abbreviations Used
Associate Editor: John Rummel
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
