Abstract
The preferential synthesis or destruction of a single enantiomer by ultraviolet circularly polarized light (UV-CPL) has been proposed as a possible triggering mechanism for the extraterrestrial origin of homochirality. Herein, we investigate the photoabsorption property of propylene oxide (c-C3H6O) for UV-CPL in the Lyman-α region. Our calculations show that c-C3H6O was produced by CH3 + and CH3CH(OH)CH3 or C3H7 • and O (triplet). The computed electronic circular dichroism spectra show that c-C3H6O and the intermediate (CH3CH(OH)CH2 +) could absorb the UV-CPL originating from the Lyman-α emitter spectrum, suggesting that the photolysis of c-C3H6O or CH3CH(OH)CH2 + upon irradiation could induce chiral symmetry breakage.
Introduction
Chiral organic molecules cannot be brought into congruence by translation, rotation, or conformational changes, while they are distinguished by two mirror images. The enantiomers of chiral molecules have two absolute configurations denoted as R and S (
Since the discovery of the enantiomeric excess (ee) of amino acids, including that in the Murchison meteorite (Cronin and Pizzarello, 1997; Engel and Macko, 1997), the cosmic origin of homochirality has been the focus of several studies. Various enantiomer formation and amplification mechanisms have been suggested and discussed (Kondepudi et al., 1990; Bonner, 1991; Soai et al., 1995; Cronin and Pizzarello, 1997; Engel and Macko, 1997; Bailey, 2000; Kawasaki et al., 2006; Fletcher et al., 2007; Garcia et al., 2019; Glavin et al., 2020). One of these mechanisms is the preferential synthesis or destruction of a single enantiomer through exposure to ultraviolet circularly polarized light (UV-CPL). This mechanism has been proposed as a possible triggering mechanism to induce asymmetry in amino acids (Bailey et al., 1998; Garcia et al., 2019; Glavin et al., 2020) as a physical process, which is widely recognized as one of the extraterrestrial origins of homochirality. Indeed, the selective destruction of enantiomers by UV-CPL has been confirmed in laboratory experiments (Flores et al., 1977; Meierhenrich et al., 2005, 2010; Nuevo et al., 2007; Meinert et al., 2014, 2015; Tia et al., 2014). In addition, infrared CPL of up to 17% was detected within the high-mass star-forming regions of the Orion molecular cloud (OMC-1) (Bailey et al., 1998). The infrared CP image shows that the infrared CPL region was spatially extended around young stellar objects (Fukue et al., 2010; Kwon et al., 2013, 2014, 2016, 2018), which were significantly larger than the size of our solar system. Recently, high infrared circular polarization induced by scatterings from dust grains aligned in magnetic fields has been explored by radiative transfer calculations (Fukushima et al., 2020). Such experiments and measurements are consistent with the astrophysical scenario of the origin of homochirality. In particular, in the early phase of the galactic evolution, the strongest emission in the pan-galactic light is the Lyman-α (Lyα) line with an emission of 10.2 eV (121.6 nm). This emission is caused by relaxation from the first electronic excited state to the ground state of a hydrogen atom. Such galaxies emitting Lyα radiation are observed as Lyman-α emitters (LAEs) (Shibuya et al., 2014). Therefore, the consideration of the photolysis of chiral molecules by Lyα irradiation can provide insights into a possible triggering mechanism to induce asymmetry in amino acids.
McGuire et al. (2016) detected a chiral molecule—propylene oxide (c-C3H6O)—for the first time in the Sagittarius B2 star-forming region using a telescope. However, the possibility of the existence of ee in the case of c-C3H6O could not be determined. Nevertheless, knowing the possibility of its existence is of significant importance to deeply understand the origin of homochirality. Considering the astrophysical scenario of the destruction of enantiomers through CPL irradiation, herein, we investigated the possibility of ee generation for c-C3H6O using quantum chemical calculations. We determined the formation pathways of c-C3H6O based on those of ethylene oxide (c-C2H4O) (Dickens et al., 1997; Turner and Apponi, 2001; Bennett et al., 2005). Furthermore, we calculated the oscillator and rotational strengths of electronic excitation for the chiral species during the formation processes to discuss the possibility of photolysis by UV-CPL absorption in the Lyα region.
Methods
Computational details
All calculations were performed by using the density functional theory (DFT) and post-Hartree-Fock (post-HF) methods implemented in the Gaussian 16 program package (Frisch et al., 2016). We performed full geometry optimizations using DFT calculations with the B3LYP functional and the aug-cc-pVTZ basis set, and then determined the energies using post-HF calculations with the CCSD(T) level of theory and the aug-cc-pVTZ basis set for the optimized geometries. Our previous DFT calculations confirmed that the B3LYP functional suitably reproduced the geometrical structures and total energies, which were in close agreement with the reliable CCSD(T) results (Kayanuma et al., 2017; Sato et al., 2018; Shoji et al., 2022). By calculating the analytical harmonic vibrational frequencies, we confirmed that the obtained local minima and transition states have no and one imaginary frequency mode, respectively.
Electronic excitation energies, oscillator strengths, and rotational strengths were calculated for the chiral species during the synthesis of c-C3H6O by using time-dependent density functional theory (TD)-DFT calculations with the CAM-B3LYP functional and the daug-cc-pVQZ basis set. We calculated 100 excited states to obtain the electronic circular dichroism (CD) spectra, in which Gaussian functions with a bandwidth of 0.1 eV for each excitation position (Rizzo and Vahtras, 2011) were used in constructing the spectra.
Selection of reaction pathways
We focused on the formation of ethylene oxide (c-C2H4O) to investigate the reaction pathway of c-C3H6O formation. In previous studies, some c-C2H4O formation pathways, based on the interstellar environment in hot-core and star-forming regions such as Sagittarius B2(N), have been proposed (Dickens et al., 1997; Turner and Apponi, 2001; Bennett et al., 2005). The three types of proposed formation pathways are as follows.
(Dickens et al., 1997),
(Turner and Apponi, 2001), and
(Bennett et al., 2005). No details on path 3 are available in the literature.
Based on the above pathways, the following three types of c-C3H6O formation pathways were investigated:
denoted as path
denoted as path
denoted as path
Reaction pathways
Path A
Path
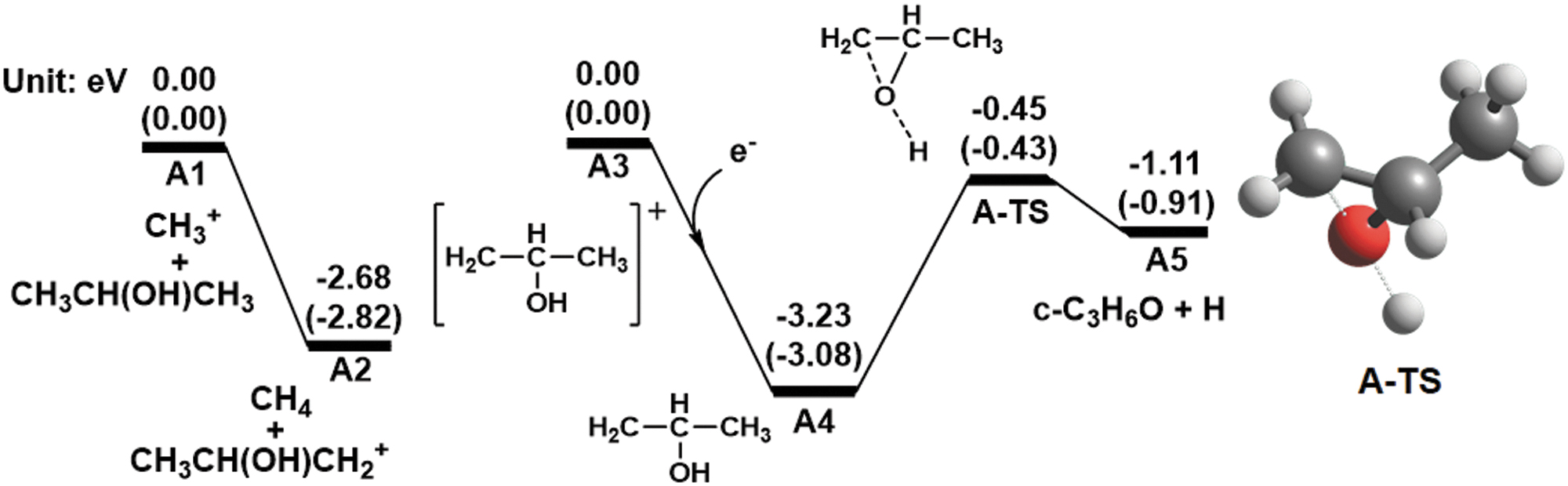
Computed energy diagrams for path
Path B
We considered path
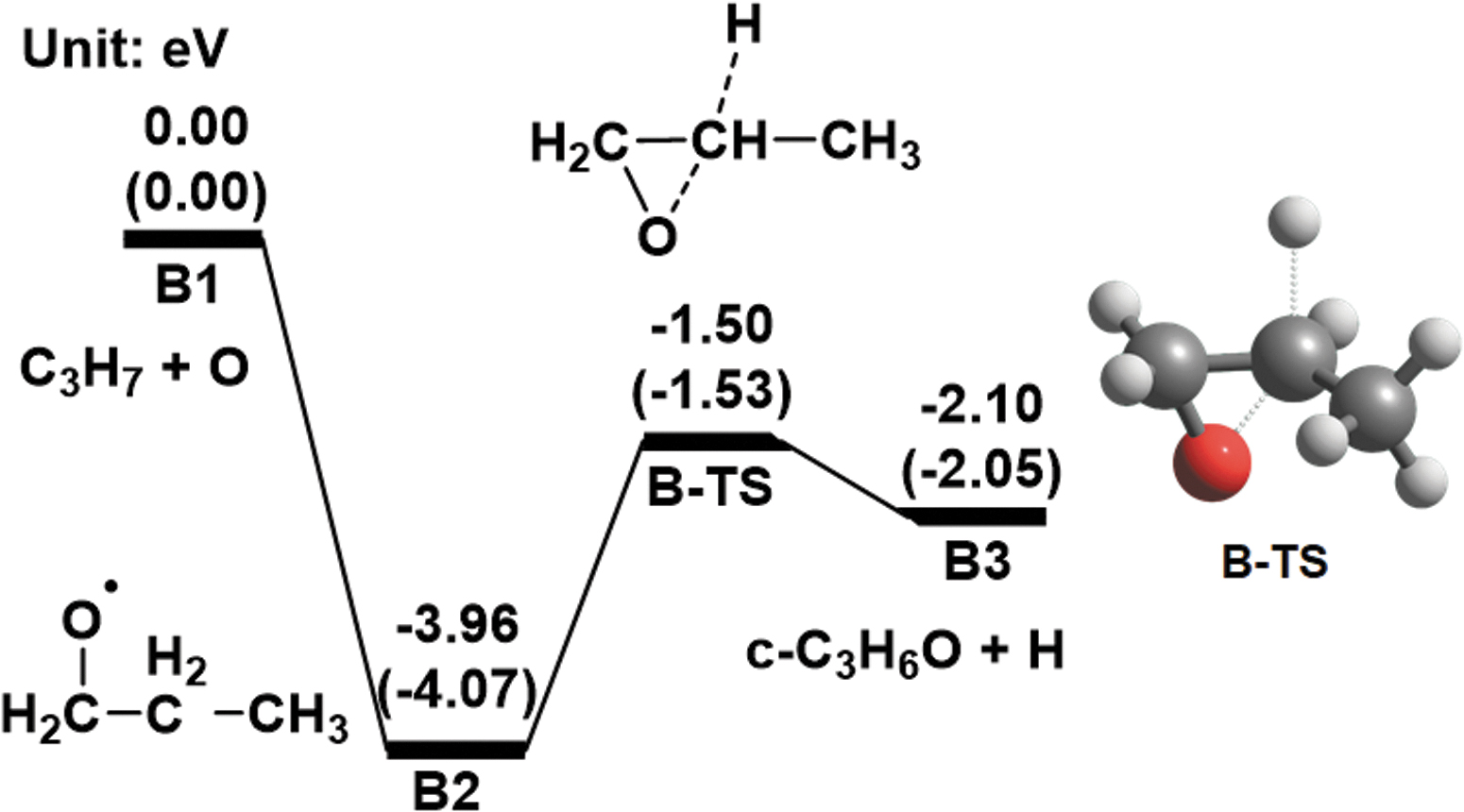
Computed energy diagrams for path
Path C
Finally, we considered path
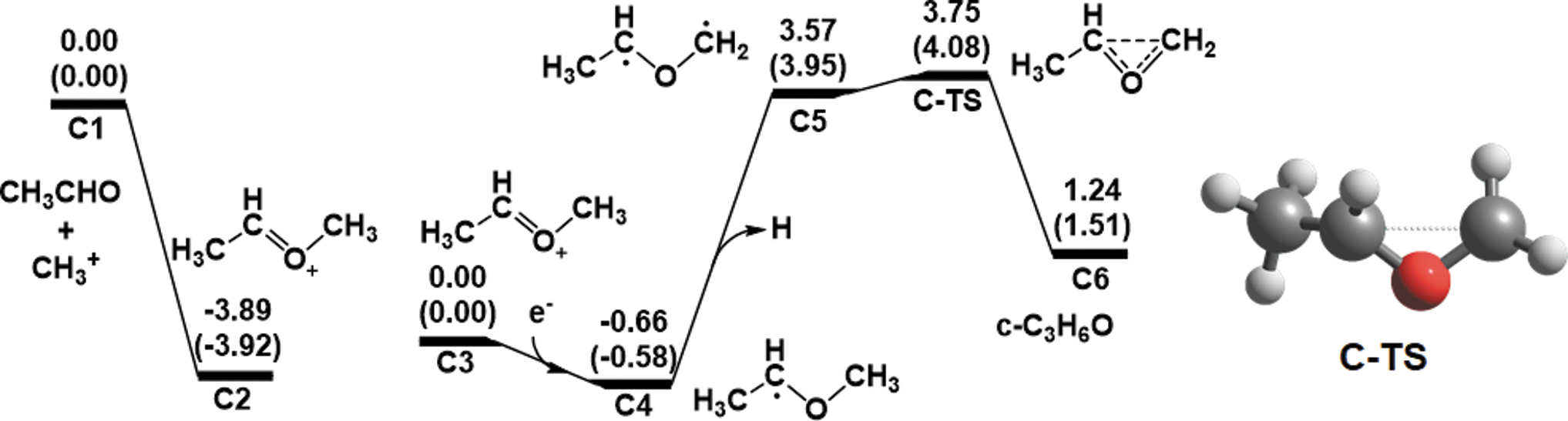
Computed energy diagrams for path
We investigated the photo-absorption property of the chiral species for Lyα with an emission of 10.2 eV (121.6 nm) and discussed the possibility of photolysis through the absorption of CPL in the Lyα region.
In previous works, the CD spectrum of c-C3H6O was measured in the vacuum ultraviolet region up to 8.2 eV (151 nm) (Carnell et al., 1991) and 9.0 eV (138 nm) (Breest et al., 1994). In the spectrum, three band maxima appeared at 7.1 eV (175 nm), 7.7 eV (161 nm), and 8.4 eV (148 nm), and the CD peak sign at 7.7 eV was opposite to the other peak signs at 7.1 and 8.4 eV (Breest et al., 1994). The simulated CD spectrum was also obtained from DFT and post-HF calculations (Carnell et al., 1991; Turner and Apponi, 2001; Miyahara et al., 2009; Varsano et al., 2009; Kröner, 2015). The results were qualitatively in agreement with the experimental CD spectrum. However, even the SAC-CI calculations, which provided a good theoretical description of the first two spectral bands, overestimated the rotatory strengths for the state corresponding to the third band (Miyahara et al., 2009; Kröner, 2015). According to the benchmark studies on CD simulations using TD-DFT calculations (Turner and Apponi, 2001; Jang et al., 2018), the M06-2X, CAM-B3LYP, and ωB97X-D functionals are feasible (Jang et al., 2018). Moreover, rotatory strengths require doubly augmented basis sets of at least triple zeta quality to reach a similar degree of convergence with the CAM-B3LYP functional (Turner and Apponi, 2001). The electronic excitation energies, oscillator strengths, and rotational strengths of c-C3H6O and CH3CH(OH)CH2
+ (
Figure 4a shows the computed CD and UV spectra of (R)-c-C3H6O. Three main peaks between 145 and 180 nm, with one positive and two negative signs, are observed and are consistent with the experimental observations (Breest et al., 1994). Herein, we focus on the photoabsorption property of the LAE spectrum with an intense peak at 10.2 eV (121.6 nm), although we cannot compare these results with the experimental results around the higher energy region because of the lack of experimental observations in this region. The calculated UV spectrum shows that c-C3H6O absorbs light at approximately 120 nm because of its strong oscillator strength. The calculated CD spectrum has a slightly negative sign at 121.6 nm. Figure 4b also shows the computed CD and UV spectra of (R)-CH3CH(OH)CH2 +. We consider the convolution of our CD spectra and the LAE spectrum (Shibuya et al., 2014), as shown in Fig. 5. In both cases, intense peaks near 121.6 nm are observed. Therefore, we suggest that both c-C3H6O and CH3CH(OH)CH2 + can absorb the Lyα line.
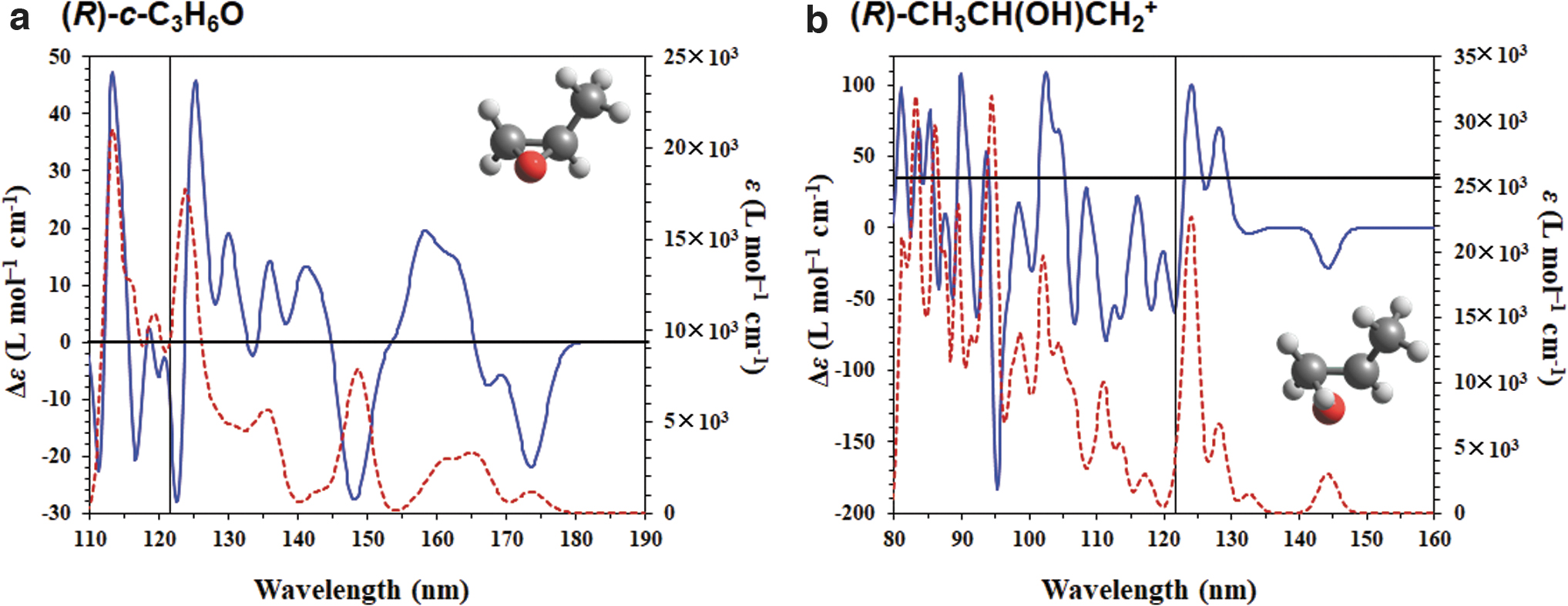
Simulated CD (blue line) and UV (red dashed line) spectra of (
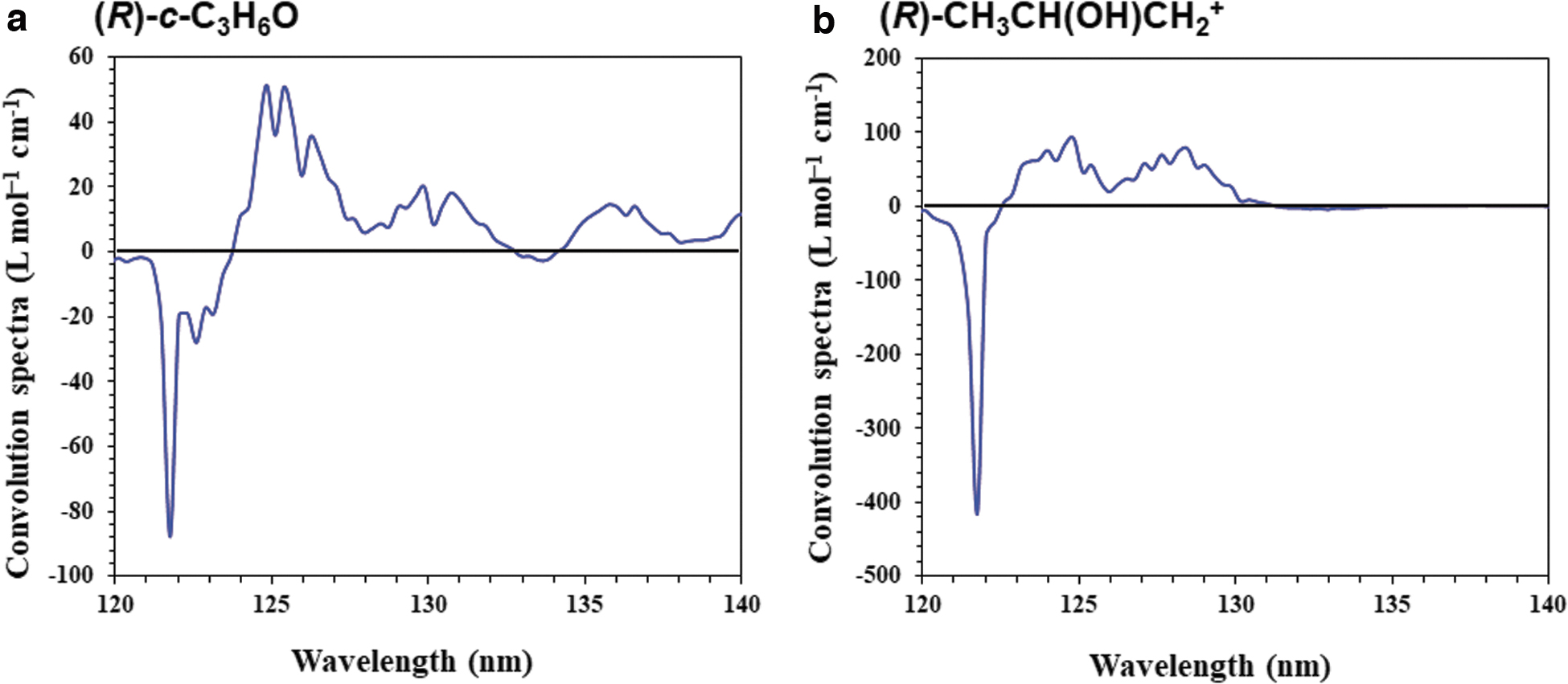
Convolution of the observational LAE spectrum and calculated CD spectra of (
A previous experimental study demonstrated that irradiating c-C3H6O in the gas phase at 185 nm generated propanal and acetone (Paulson et al., 1977). The experimental results and our computational results of the CD spectrum imply the occurrence of photolysis of c-C3H6O by Lyα, although the wavelength is different from that used in the previous experimental measurements. Determining the detailed photolysis reaction mechanism is a future challenge; however, the present results provide a possibility of the formation of c-C3H6O homochirality in space. Experimental verification is required to examine the photolysis by CPL irradiation in detail.
In this study, we investigated the formation and possible photolysis of c-C3H6O, a chiral molecule first detected in the Sagittarius B2 star-forming region (McGuire et al., 2016), under interstellar conditions. We focused on the previously proposed c-C2H4O formation mechanism (Dickens et al., 1997; Turner and Apponi, 2001; Bennett et al., 2005) to investigate the reaction pathways of c-C3H6O formation at the atomic level, which cannot be detected by space observation due to the short lifetime of most of the intermediates. The computed energy diagrams show two energetically downhill pathways for c-C3H6O formation. One pathway consisted of two reactions: (1) C3H7O+ and CH4 generation from CH3 + and CH3CH(OH)CH3, and (2) c-C3H6O formation from C3H7O+ and e−. The other pathway produced c-C3H6O from C3H7 • and O (triplet). Therefore, c-C3H6O was produced by CH3 + and CH3CH(OH)CH3 or C3H7 • and O (triplet), as indicated by our quantum chemical calculations. Because the reactants, the product, and several intermediates with a long lifetime can be detected by observation, the pathway proposed by our calculations is plausible in an interstellar environment. Furthermore, the CD spectra obtained from DFT and post-HF calculations indicate that c-C3H6O and CH3CH(OH)CH2 + could absorb UV-CPL from the LAE spectrum. This suggests that the photolysis of c-C3H6O or CH3CH(OH)CH2 + under CPL irradiation could induce chiral symmetry breakage. Infrared CPL was detected within the high-mass star-forming regions of OMC-1 (Bailey et al., 1998), and the infrared CP image also shows that the infrared CPL region was spatially extended around young stellar objects (Fukue et al., 2010; Kwon et al., 2013, 2014, 2016, 2018). In addition, LAEs were observed in the early phase of the galactic evolution (Shibuya et al., 2014). These experiments and measurements, in addition to our present calculation results, are consistent with the astrophysical scenario of the origin of homochirality. To prove this scenario, further experimental verification is required to examine the photolysis by CPL irradiation in detail. The short-lived intermediates discovered in our calculations, the formation energies of various reactions, and other calculated spectroscopic data are expected to be beneficial for their experimental verification.
Footnotes
Acknowledgments
This work was supported in part by the Multidisciplinary Cooperative Research Program at the Center for Computational Sciences, University of Tsukuba. Some of the computations were performed using the computer facilities at the Research Institute for Information Technology, Kyushu University, and the Research Center for Computational Science, Okazaki, Japan (Project: 22-IMS-C122). We would like to thank Editage (
Authorship Confirmation Statement
Y.H., H.N., and T.S. conducted the computations and analyses and interpreted the data with the help of N.W., M.K., M.S., M.U., and Y.S. Y.H. wrote the paper, and the others edited it. All the authors contributed to the final manuscript.
Authors' Disclosure Statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding Statement
This work was partly supported by JSPS Grants-in-Aid for Scientific Research (JP19K15524 and JP19H00697), MEXT Grants-in-Aid for Scientific Research on Innovative Areas (JP21H00014 and JP21H05419), the Cooperative Research Program of “Network Joint Research Center for Materials and Devices,” and a research grant from The Mazda Foundation.
Supplementary Material
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Abbreviations Used
Associate Editor: Christopher McKay
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
