Abstract
The Perseverance rover is meant to collect samples of the martian surface for eventual return to Earth. The headspace gas present over the solid samples within the sample tubes will be of significant scientific interest for what it reveals about the interactions of the solid samples with the trapped atmosphere and for what it will reveal about the martian atmosphere itself. However, establishing the composition of the martian atmosphere will require other dedicated samples. The headspace gas as the sole atmospheric sample is problematic for many reasons. The quantity of gas present within the sample tube volume is insufficient for many investigations, and there will be exchange between solid samples, headspace gas, and tube walls. Importantly, the sample tube materials and preparation were not designed for optimal Mars atmospheric gas collection and storage as they were not sent to Mars in a degassed evacuated state and have been exposed to both Earth's and Mars' atmospheres. Additionally, there is a risk of unconstrained seal leakage in transit back to Earth, which would allow fractionation of the sample (leak-out) and contamination (leak-in). The science return can be improved significantly (and, in some cases, dramatically) by adding one or more of several strategies listed here in increasing order of effectiveness and difficulty of implementation: (1) Having Perseverance collect a gas sample in an empty sample tube, (2) Collecting gas in a newly-designed, valved, sample-tube-sized vessel that is flown on either the Sample Fetch Rover (SFR) or the Sample Retrieval Lander (SRL), (3) Adding a larger (50-100 cc) dedicated gas sampling volume to the Orbiting Sample container (OS), (4) Adding a larger (50-100 cc) dedicated gas sampling volume to the OS that can be filled with compressed martian atmosphere.
Context and Conclusions
The Mars Sample Return (MSR) Program has as a current draft Level 1 Requirement:
“The MSR Campaign shall be capable of delivering to Earth at least one dedicated atmospheric gas sample of at least 1.9 × 10−5 mole of martian atmospheric gas molecules that is collected and stored in a clean and sealed volume.”
This team has been tasked with studying the scientific implications of returning, instead, one or more sample tubes from Perseverance, which would be sealed on Mars in the presence of martian atmosphere. This report was initially prepared for the MSR Standing Review Board.
We address two specific issues: An individual sample tube on Perseverance would collect approximately 20% of the amount in the current draft Level 1 requirement, so we address the minimum amount of atmospheric sample needed to constrain high-priority scientific questions. The sample tubes on Perseverance have been designed for collecting rocks, not atmosphere, so we analyze how that aspect might be different from a container designed for atmospheric sample return, and how that affects our ability to answer scientific questions.
We conclude that both the size and history of the sample tubes would cause a major loss in scientific value, compared to a sample collected in accordance with the draft Level 1 requirement. However, the return of a single sample tube sealed with only atmosphere inside would likely provide significant scientific value beyond that of the head space gas trapped in tubes containing rock samples, and we
The difference in the amount of sample returned between the two options is problematic. There are critical measurements (
There are critical measurements, including the relative elemental abundances of Kr and Xe and the isotopic composition of N2, for which the history of manufacturing and exposure to atmosphere, water vapor, and dust the sample tubes have experienced, or will experience, would call into question measurements collected using the Perseverance sample tubes. The tubes that have been sent to Mars were not pre-treated in the same way that gas-collecting materials normally are in terrestrial situations (baked under high-vacuum conditions at >200°C for multiple days and then sealed until use). The materials used, notably the TiN coating of the inner walls, could potentially outgas or adsorb material before and/or after sampling, compromising the purity of the sample.
For the head space in tubes that contain samples, the situation is even worse than collection in a dedicated sample tube because there will be a smaller amount of martian gas, and each of the samples themselves will have interacted with that gas, likely in various ways that will make the interpretation of any head space gas aliquot (or even the pooling of several gas samples) difficult.
Calculations of the amount of leakage that will occur from a sealed sample tube have large uncertainties, though a best estimate based on limited data suggests that no more than a few percent is likely to be lost in most cases. That is more than would be desirable, and while that level of loss might be acceptable, if the leak rate is underestimated, it is possible that a sealed sample tube might not return any usable sample. Leakage in the other direction on Earth (
Although this team was tasked with determining how well the draft Level 1 Requirements would be addressed by sealing and returning one of the sample tubes already on Perseverance, and this report focuses on that, there are other options for returning samples of Mars atmosphere that would provide different levels of important scientific return.
At one extreme, head space gas will be present in the tubes that contain the collected solid samples, and that head space gas will be derived largely from the martian atmosphere. However, for this scenario, all the problems listed above will be exacerbated compared to an empty sample tube, that is, the volume will be smaller, the samples themselves will provide multiple opportunities for exchange with the head space gas, and the process of putting the solid samples in the tubes will make leakage more likely.
If NASA and ESA decide to remove the draft Level 1 Requirement, we would
At the other end of the spectrum, a compressed sample of the same volume as that of the draft Level 1 Requirement would have significantly more scientific value than one that simply meets the draft Level 1 Requirement.
An intermediate option is to collect gas in a newly designed sample-tube sized vessel that is valved with two valves and flown on either SFR or SRL. Such a vessel would not require additional space to return it compared to a typical sealed empty sample tube, although it would require development efforts. This strategy would alleviate concerns about the manufacturing and history of the collection container and provide a most robust mechanism for sealing (and testing the seal upon return), and while it would not meet the draft Level 1 requirement for volume, the inevitable advances in detection technology that will occur before the return of the samples would mitigate this.
Findings
When considering the head space gas in tubes with samples in them, we conclude that the relatively small volume, potential of seal leakage, and especially the likelihood of interaction of atmospheric gas with the sample and tube will make it difficult to answer any fundamental questions about the martian atmosphere.
Although there are potential problems, most notably due to the possibility of interaction of martian atmospheric gas with sample tubes that were not manufactured, stored, or treated specifically for the task of atmospheric sampling, we conclude that a sealed sample tube containing only atmosphere (i.e., devoid of solid samples) recovered from Perseverance would likely provide significant fundamental science return about the martian atmosphere. Such a sample would also provide a much-needed baseline for interpreting the head space gas in tubes with solid samples. If taken, this option should be accompanied by a program to study the interaction of the tube materials with simulated martian atmosphere and their outgassing behavior.
With consideration of upcoming Mars-targeted missions, we conclude that gas collected in a newly designed and purpose-built valved sample-tube sized vessel, which could be flown on either SFR or SRL, would be considered of higher priority than either the head space gas or a sealed M2020 sample tube. Conceptually, this vessel would require no more physical space to return than a sealed empty sample tube and alleviate concerns about the manufacturing and history of a non-purpose-built vessel, and the valving would provide a more robust mechanism for sealing the vessel and testing the seal upon return.
Structure of This Report
In Section 3, we discuss the species under consideration, calculate the amounts that would be acquired under various scenarios, and compare it to the amounts that would be needed for high-quality measurements using today's state-of-the-art techniques.
Section 4 discusses the effects of the container, including preparation, surface composition, storage history, and the possibility of leakage.
Section 5 is a summary of the basic points of the report.
Appendix 1 gives a history of previous groups' suggestions of requirements for a returned atmospheric sample.
Appendix 2 is a more detailed discussion of the scientific reasons for returning a sample of the martian atmosphere.
Appendix 3 is a detailed calculation of the effects of leak rates, based on measurements provided to us.
Species under Consideration and Amounts Needed
More details about the scientific rationale for measuring each of the species listed in Table 1 are given in Appendix 2, but the basic reasons driving the desire to measure each of these is as follows. The noble gases (He, Ne, Ar, Kr, Xe) are valuable for tracing the history of the atmosphere, including the original source of the atmosphere, additions to the atmosphere through martian mantle outgassing, and major processes such as past, catastrophic, and ongoing atmospheric loss. The triple oxygen isotopes in CO2, O2, CO, and H2O are necessary to understand the triple oxygen isotope variations in secondary minerals within rock samples and thus provide valuable information about interactions between the atmosphere and rocks (and water). The isotopes of nitrogen are heavily fractionated in the martian atmosphere; detailed measurements of the isotopic composition, coupled with measurements in rock samples, can provide information about atmospheric loss, as well as outgassing from the interior. In many cases, these measurements are complementary. As a known marker for biological activity on Earth, methane (CH4) in the martian atmosphere is of considerable interest. The amounts measured by Curiosity vary greatly (Webster
Quantities of Atmosphere per Container, Assuming a Sampling Atmospheric Pressure of 7.36 mbar.
All values assume atmospheric pressure of 7.36 mbar; fractions from Trainer et al. (2019) for CO2, N2, Ar, O2, CO; Krasnopolsky and Gladstone (2005) for He; Owen et al. (1977) for Ne, Kr, Xe, and H2O.
Assuming 50% of sample saved for posterity, amount of aliquot available for triplicate analyses by each of three laboratories.
Reference laboratories. Note that these are chosen to represent 2020 state-of-the-art laboratories, and do not imply that these will be the laboratories that will actually analyze the samples.
Quantities of Atmosphere per Container, Assuming a Sampling Atmospheric Pressure of 7.36 mbar.
All values assume atmospheric pressure of 7.36 mbar; fractions from Trainer et al. (2019) for CO2, N2, Ar, O2, CO; Krasnopolsky and Gladstone (2005) for He; Owen et al. (1977) for Ne, Kr, Xe, and H2O.
Assuming 50% of sample saved for posterity, amount of aliquot available for triplicate analyses by each of three laboratories.
Reference laboratories. Note that these are chosen to represent 2020 state-of-the-art laboratories, and do not imply that these will be the laboratories that will actually analyze the samples.
The highest priority measurements are as follows:
To evaluate what constitutes a necessary volume of atmospheric sample to achieve these priority measurements, we estimate in Table 1 the amounts of various species available in a sample tube (or a container of the same size as a sample tube), a 50 cc container, and a 100 cc container, each filled at ambient Mars atmospheric pressure. Note that a 50 cc container only provides approximately 85% of the draft Level 1 requirement so is used as a convenient size for purposes of example. Next, the abundance of the species in a single aliquot is calculated, assuming that, of the original sample, half is saved while the other half is divided into nine aliquots, to allow each of three laboratories to perform three analyses targeting the species of interest. Finally, the amount of sample required for a high-quality measurement in a 2020 state-of-the-art laboratory is listed. This group was not tasked with determining the accuracy required to answer specific questions, so we have used “high-quality” to denote the amounts and conditions various specific laboratories consider adequate for publishable measurements. If the amount of gas available is less than what is considered necessary for high-quality measurements, it is, of course, possible to do fewer replicates (which reduces confidence in the accuracy of the measurements) or to make measurements with lower precision. In that case, those making measurements on the returned samples will have to make these difficult choices, but our purpose is to determine the amounts needed to make measurements at the current state-of-the-art.
In practice, some measurements require a separate aliquot, because measuring one species precludes measuring others—mass spectrometers consume the sample that they measure. In other cases, multiple species can be measured on a single aliquot in a mass spectrometer, or the measurement can be made by a technique that preserves the sample. Measurements with a tunable laser spectrometer (TLS), as would be preferred for CO2, CO, and H2O, do not consume sample, hence are a preferred method for initial atmosphere characterization. Mass spectrometric measurements of all the noble gases could be done on a single aliquot. Measurements of the isotopes of molecular oxygen (O2) and nitrogen (N2) could also be analyzed on a single aliquot, though not the same aliquot as the noble gases. Hence, to make all the measurements discussed above would require twice as many aliquots as any single measurement, which would mean that each aliquot available might be only half as large as stated in Table 1.
In terms of the amount of sample needed, it is clear that a single sample tube would provide sufficient sample to measure the noble gases and triple oxygen isotopes in CO2, although CO2 and the noble gas Xe are both within a factor of two of the amount available for a sample tube.
For CO and H2O, the amounts collected for even a 100 cc container are far too small to acquire meaningful triple oxygen isotopic ratios. For O2 and N2, the amounts required are roughly three times the amount in a single aliquot of a 100 cc container. A precise measurement could be made on fewer aliquots with present-day technology, but only by sacrificing a significant fraction of the sample, which would preclude measuring noble gas isotopes. We anticipate that with a decade of technical improvement, such measurements could be feasible. The situation is worse by nearly an order of magnitude for an empty sample tube (and worse still for those containing martian solids), which means that the instrument developmental efforts required to measure these would be even larger.
An alternate solution to filling a single tube and/or utilizing the head space gas of sample tubes would be to collect a 100 cc sample and compress it by a factor of 100, as proposed by Jakosky
Another option is to rely on analysis of martian atmospheric gases trapped in the head space of every sample tube filled with rock or regolith samples. There is likely to be valuable science that can be done with these, particularly with respect to improving our understanding of the interactions between the atmosphere and the returned rock or regolith samples. However, because the head space gas will have interacted with the geochemically different samples in many cases, it will be highly beneficial to know what the starting atmospheric composition would have been. The ability to extrapolate this information from the head space gas of sample tubes whose seals could leak at different times during MSR and with variable amounts of volume remains a key challenge for these types of atmospheric samples. Such head space gas samples likely will not enable us to answer key questions about the atmosphere. Additional limitations associated with the manufacturing and environmental history of the sample tubes, enumerated below, also apply. These complications are exacerbated by having a smaller volume of gas and more materials (the samples themselves) with which the gas can potentially interact.
In Section 3, we showed that collecting a sample of martian atmosphere in a sample tube would result in an amount that makes some measurements marginal or impossible with current technology, but is not dramatically different from the draft Level 1 requirement. However, the materials and history of the sample tubes are a larger consideration. Tube construction and exposure to varying conditions over time could alter many or all of the measurements of the atmospheric sample in ways that might not be possible to detect or address. These concerns are as follows:
The sample tubes were coated with titanium nitride, in a nitrogen atmosphere. Titanium nitride is far from an inert material at the atomic level (
In addition, the sample tubes have been open since their manufacture and would not be sealed until some point on the surface of Mars. Therefore, sample tubes were not degassed at temperatures above 200°C under high vacuum pumping, as is required and done for lines and vessels used in noble gas and nitrogen laboratories. Baking under high vacuum is required to remove adsorbed terrestrial atmosphere contaminants, which otherwise will contribute terrestrial contamination to martian gases. This also holds for parts of the seals, such as the Au plating that will be in contact with the sample. Plating can be the source of species, particularly Kr and Xe, which can be removed only by severe baking. This means that they will have been exposed to the terrestrial and martian atmospheres for months or years, while also spending many months in the vacuum of space, all the while undergoing thermal cycles in which gases might be trapped or outgas. This is potentially a problem for N2, which will be difficult to measure anyway because of its low abundance, as well as for Kr and Xe, which readily adsorb onto surfaces. However, this would be an even worse problem if technical improvements make it possible to measure H2O, since the water available for exchange might be either terrestrial or martian or a mixture of both. Typical preparation protocols for containers for use in high-precision gas measurements include high-temperature bakeout under vacuum followed by sealing until the desired gas is sampled, which is not possible for the sample tubes.
Finally, extensive tests of the seals on the sample tubes have led to the conclusion that there is a 95% chance that 80% or more of the tubes will leak He at a rate of 1 × 10−8 scc/s or less. In Appendix 3, we calculate the amount of leakage that could occur during a two-year period when exposed to the vacuum of space and find that 5% of the gas would be lost, assuming a He leak rate of 1 × 10−8 scc/s. This amount of loss would be a concern, but marginally acceptable. The tests performed to date suggest that it is likely that most tubes will have lower leakage rates, which would diminish the problem. However, we would not know the leak rate of a specific seal until the sample is returned to Earth, if then, and if only 80% of the sample tube seals are that tight, then 20% would be at higher levels. Furthermore, the likely event of dust from the martian environment settling onto the seals will increase the likelihood of leakage. So, it is also possible that virtually all the sample could be lost, rendering the effort useless. In addition, after arrival in the Earth's atmosphere, the pressure of Earth atmosphere will be two orders of magnitude higher than the content of the sample tube, with the partial pressure of nitrogen displaying an even larger difference, as nitrogen is a trace constituent in the martian atmosphere and the major constituent in Earth's atmosphere. Contamination, even with a small leak, could render some of the most important measurements impossible. For a dedicated container with a valve, the leak rate of the actual container plus valve could be measured on Earth before it was sent to Mars. The dust risk increases with the duration of time the tubes are on Mars before being sealed, so if the atmospheric sample will be an empty tube on Mars 2020, earlier sampling would be preferable.
In summary, the draft Level 1 requirement for a “clean and sealed” volume appears critical for understanding any measurements of an atmospheric sample.
Summary
In terms of the size of the sample:
The amount of Ar, Kr, Ne, and He in a sample tube would easily be sufficient to make replicate, high-quality isotopic measurements. For He, we do not know how much 3He is in the martian atmosphere, but an amount below detection limits would set a scientifically important upper limit.
The amount of CO2 and Xe in a sample tube would be sufficient to make the desired number of replicate elemental measurements, but the amount is within a factor or two of the minimum required for accurate measurements; larger samples, such as those required to meet the draft Level 1 requirement, would make it possible to make more measurements and/or measurements of larger aliquots of any given species, increasing the number of high-priority questions about the atmosphere that we will be able to answer.
The amount of O2 and N2 in a sample tube would be insufficient to make any high-quality isotopic measurements. The amount in a 50 cc or 100 cc container would barely provide enough gas to make the minimum number of desired measurements, but could be sufficient with technical development.
Without major technical advances, the amounts of CO, H2O, and CH4 of even a 100 cc container would be too small to make isotopic measurements that are precise enough to be meaningful.
In terms of the container:
The sample tubes are coated with titanium nitride, a potentially active surface that could either adsorb sample gas or desorb contamination. The long-term net-effect of these two competing processes, particularly once the tubes are sealed on Mars, could be difficult to assess.
The titanium nitride coating was applied in a nitrogen atmosphere, which could lead to outgassing of nitrogen and/or outgassing of trace gases within the coating atmosphere.
The sealing mechanism contains Au-plated parts, which will have incorporated (terrestrial) gases present during the plating process. These gases can be a source of contamination if the equipment is not baked at a few hundred degrees centigrade under ultra-high vacuum conditions.
The sample tubes have been open since their manufacture and will remain open until they are sealed on Mars, which will have provided ample opportunity for contamination through exposure to two different planetary atmospheres. There would be no opportunity to bake the tubes or the parts of the sealing mechanism that will be exposed to the sample before the sample is acquired.
Adsorption and desorption onto the container surfaces will vary from species to species, and effects on elemental ratios of the species undergoing adsorption-desorption cycles is complex and might not be fully resolvable, calling any measurement, particularly an unexpectedly anomalous measurement, into question.
The leak rate of the sample tubes, while low, is not constrained to be low enough for a single tube to prevent loss of a substantial amount of an atmospheric sample in space or contamination by terrestrial atmosphere after return.
Appendix 1: Prior Descriptions of the Requirement for a Dedicated Atmospheric Sample
A series of reports and studies of Mars Sample Return discussed the return of an atmospheric sample, leading to the current draft Level 1 Requirement of a dedicated atmospheric sample.
A Mars Exploration Program Analysis Group (MEPAG) Next Decade Science Analysis Group report (2008) said, “… it should be possible to recover the headspace gas . … , although this gas will be significantly less useful for scientific purposes than a sample that has been isolated.” They did not use the word “dedicated.”
The report of the International Mars Architecture for Return of Samples Working Group (iMARS, 2008) did not use the term “dedicated,” but said (p. 20), “The gas sample should be isolated from the rock samples.” iMARS (2008) also wrote a set of draft requirements for an international MSR mission, and Requirement #9 is that “MSR will collect at least 1 gas sample of 10 cc at a pressure of 0.5 bar.” Note that this gas quantity is approximately one order of magnitude larger than the current draft Level 1 Requirement.
The term “Dedicated” was first introduced by the End-to-End International Science Analysis Group (McLennan
All of this was input to the Planetary Science decadal survey,
Appendix 2: Scientific Questions Driving Atmospheric Sample Return
The scientific rationale for returning samples of martian atmosphere was addressed in the last Planetary Science decadal survey,
Noble gas isotopic abundances
Noble gases (He, Ne, Ar, Kr, and Xe) are chemically unreactive, which makes them key tracers of planetary outgassing, atmospheric formation, and atmospheric evolution. In addition, they are relatively rare, so radioactive and other nuclear processes that produce a noble gas from a more common element can easily be traced.
Atmospheric isotopic compositions of all noble gases except He have been measured in martian meteorites (see review by Swindle, 2002), although corrections for the history of the meteorite often have to be made (
Martian Xe is strongly fractionated isotopically relative to solar (nebula) Xe in a manner similar to terrestrial Xe. On Earth, this appears to be the result of a continuous process during the Hadean and Archean and is linked to H escape and therefore H2O. Earth's atmosphere also contains recognizable contributions from the decay of 129I and 244Pu. Mars' atmosphere contains a contribution from 129I, but apparently lacks a contribution from Pu or U fission. Alternatively, the original noble gas reservoirs of the two planets might have been different. Higher precision measurements of the martian atmosphere are thus needed. In addition, the Curiosity measurement of martian Xe appears to show a puzzling contribution from cosmic-ray-spallation reactions within surface rocks producing 124Xe and 126Xe, a contribution that is not seen in the terrestrial atmosphere. For Kr, the major puzzle is that, while martian Ar and Xe appear to have been isotopically fractionated relative to solar compositions, martian Kr does not.
SAM was unable to measure Ne isotopes. In this case, there are contrasting interpretations of the meteorite gases at the tens-of-percent level for the ratio of the two most abundant isotopes, 20Ne and 22Ne. This value, coupled with the known measurement of 38Ar/36Ar, may make it possible to identify what processes are in action—thermal and/or non-thermal escape processes. In addition, the relative abundance of the least abundant isotope, 21Ne, is even less well known from measurements made to date. Since 21Ne can be produced both by nucleogenic processes in the martian mantle and by cosmic-ray spallation reactions in the very surface regolith layers, a precisely measured 21Ne/22Ne ratio could be coupled with models of outgassing of radioactive decay products in Ar and Xe and/or with models of cosmic-ray produced atmospheric excesses in Xe. Both relevant Ne isotope ratios (20Ne/20Ne, 21Ne/22Ne) should be measurable with sufficient precision (<2%) in a container the size of a sample tube.
The abundance of He in the lower atmosphere is estimated to be (10 ± 6) ppmv, based on the latest analysis of the Extreme Ultraviolet Explorer and Phobos data (Krasnopolsky and Gladstone, 2005). This estimate is based on a detection in the upper atmosphere above the homopause, coupled with a radiative transfer model, to give the value in the lower atmosphere. The isotopic ratio 3He/4He has not been measured (3He at the ppbv level or below is well below the detection limit by telescopic methods). With a returned sample, the abundance of 4He can be measured with high precision, and even 3He/4He can be determined if the ratio is in the same range as that of the terrestrial atmosphere. Both these values will be invaluable for modeling degassing from the martian interior.
Heavy noble gas elemental abundances
The heavy noble gases Kr and Xe provide a fascinating conundrum: the martian meteorites do not show the same atmospheric Kr to Xe elemental ratio. Figure 1 highlights the different martian noble gas reservoirs and shows where the fractionated component lies: martian atmosphere as measured in the shergottite meteorites (MA; Bogard
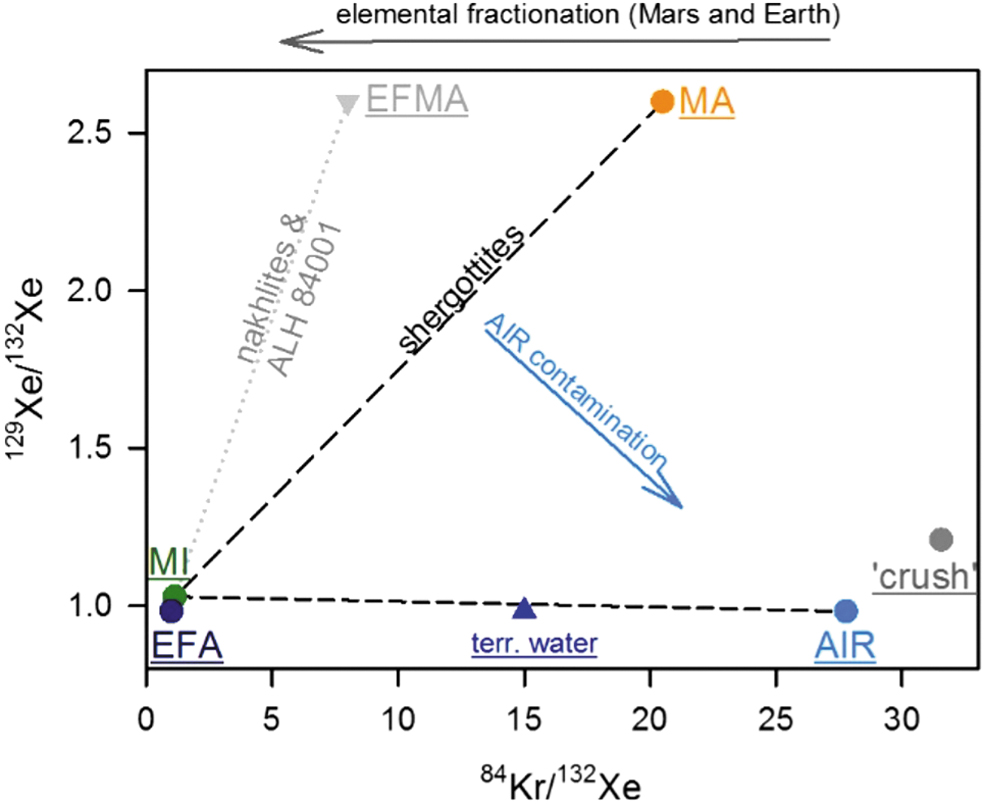
Endmembers as commonly used in Martian meteorite heavy noble gas research. Endmembers are underlined, mixing and fractionation trends are indicated by arrows and lines.
The origin of the elementally fractionated martian atmosphere in the nakhlite meteorites remains unclear, even though, by now, it has been investigated for over three decades (Ott, 1988; Mathew
Oxygen isotopes in CO2, O2, CO, and H2O
The Δ17O of igneous silicate rocks from Mars is 0.3‰ (Franchi
The martian atmosphere is very different from Earth's atmosphere as it is dominated by CO2, has only a pressure of ∼650 Pa, has undergone considerable escape to space, and is optically thin for UV light. The low density and absence of biologic O2 allows UV radiation to penetrate through the atmosphere down to ground level. Photolysis of CO2 to CO and O2 can result in large 100‰ anomalies in Δ17O in the reaction products (CO2 photolysis at 184.9 nm; experiments by Bhattacharya
The triple oxygen isotope composition of atmospheric CO2, O2, H2O, and CO would provide a unique picture of martian atmospheric photochemistry and allow an understanding of the anomalous signatures in martian minerals and water. If the anomalies observed in low-
If, however, atmospheric components with distinct isotope anomalies can be identified, the presence of variable anomalous oxygen isotope signatures in low-
Nitrogen isotopes in N2
The nitrogen isotopic composition of the martian atmosphere shows strong enrichment in 15N, δ15N = 572 ± 82‰ as measured by Curiosity (Wong
The terrestrial atmosphere shows a 19‰ excess of 15N15N (expressed as Δ30) in air relative to a random distribution of nitrogen isotopes, attributed to gas-phase chemistry in the thermosphere (Yeung
Nitrogen is a critical component required by all life on Earth, and the availability of nitrogen in the modern ocean strongly controls biological productivity (
Appendix 3: Leakage during Return
The following calculation is an attempt to address how much leakage might occur from a sample tube sealed on Mars and returned to Earth. Our information on the experiments on the seals comes from the M2020 Sample Tube Seal V&V Evidence Package.
Assuming molecular flow, the system should obey the Knudsen equation, which is
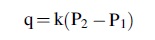
where q is the flow rate (leak rate), k includes various parameters relating to the size and shape of the leak, and P1 and P2 are the pressures outside and inside the volume. In our case, we are most worried about leaks to the vacuum of space, so P1 is effectively zero, and the equation becomes
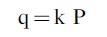
But q is proportional to the derivative with time of the pressure, dP/dt, so we can rewrite as
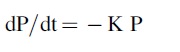
where K is some new constant, and the negative sign indicates that the pressure is dropping with time.
PV = nRT, so P is proportional to n, the number of moles, and it should be the same constant of proportionality on both sides, so

This equation is the basic radioactive decay equation, which can be solved as
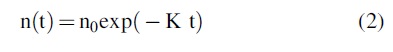
where n0 is the initial abundance.
Now we need to find K and t. We are given a leak rate of 1 × 10−8 scc/s of He, but we do not know what pressure (hence number of atoms or moles) that corresponds to. Let us assume the test is done with 1 bar of He on one side of the seal and a vacuum on the other (we will consider the implications of this below). In that case, the appropriate K for a sample tube (which is 13 cc), would be (substituting into eqn. 1)
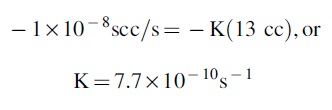
We will assume that the time in vacuum (the time in orbit, plus the trip time to Earth) is 2 years, or 6.3 × 107 s
So we substitute this value into eqn. 2 and get
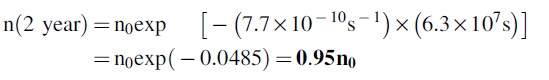
This would indicate that 5% of the original He sample would be lost. By itself, this number is troubling (we would much prefer retention of 99% or more of the sample), but the larger concern is that there are order of magnitude assumptions made in the calculation, so the actual number could be significantly different in either direction.
In a series of tests with and without dust or abrasion, and with variable forces applied to the seal, it was determined that abrasion was more important than dust, unless there was enough material to seriously affect the geometry of the seal, a circumstance unlikely to occur for a blank tube (but possible for the tubes with rocks in them). Subsequent tests with various stresses, such as thermal and vibration, of seals made in conditions designed to replicate the conditions on Perseverance, have so far returned leak rates below 1 × 10−10 scc/s.
We do not know the applicable pressure for the leak rate test. Our understanding is that the tests were done in a standard manner, with instrumentation under vacuum on the inside of the tube, and He atmosphere (under a “hat”) on the outside. Since the gas leaking into the tube is likely to be close to atmospheric pressure, but is not pure He, the partial pressure of He is likely to be less than one atmosphere, so K is likely to be larger than in our calculations, and the amount of gas retained is likely correspondingly smaller.
Our understanding is that tests were done only for gas leaking from the outside of the seal in, as opposed to the direction we have been considering. These are likely to be similar, if not identical, but it is not guaranteed. We have not presented an equivalent calculation for the leakage into the container after return to Earth, but the pressure differential is two orders of magnitude more, so the leak will be that much faster, adding a complicating effect. Actually, the difference in partial pressure for nitrogen is even more of a concern, since N2 is the dominant species in the terrestrial atmosphere but occurs at the percent level in the martian atmosphere, with a different isotopic composition. Although Knudsen flow may not be applicable in this case, the equivalent calculation shows that an exposure on the order of minutes in the terrestrial atmosphere, or in a nitrogen-filled storage container, could change the N isotopic composition by more than 1‰, the desired measurement uncertainty.
If the leak rate really is 1 × 10−10 scc/s, then with all other parameters kept the same, this calculation would give a retention of 99.95%, which is not a problem. On the other hand, if the leak rate is, instead, two orders of magnitude higher, this calculation would give a retention of <1%, leading to gas amounts that would not be measurable in many cases and to isotopic and elemental ratios that are fractionated. However, we will not know what the actual leak rate is until after the samples are returned, if then. If a dedicated container with a valve were constructed, the leak rate of the actual container plus valve could be measured both beforehand and afterwards.
Footnotes
Acknowledgments
We are deeply indebted to Mark Thiemens, Ulrich Ott, and John Eiler for thorough and thoroughly useful readings of an earlier version of this report. We also appreciate detailed discussions of tests of sealing mechanisms with Eric Kulczycki and Kristo Kreichbaum, who tried to explain everything that had been tried and why—we hope that we have accurately represented their extensive efforts. Finally, we appreciate the efforts of Dave Beaty, Michael Meyer, Gerhard Kminek, and, especially, Brandi Carrier, in shepherding this process through and making the appropriate connections.
The decision to implement Mars Sample Return will not be finalized until NASA's completion of the National Environmental Policy Act (NEPA) process. This document is being made available for information purposes only.
Disclosure Statement
No competing interests.
Funding Information
None
