Abstract
Viruses constitute a significant part of the human microbiome, so wherever humans go, viruses are brought with them, even on space missions. In this mini review, we focus on the International Space Station (ISS) as the only current human habitat in space that has a diverse range of viral genera that infect microorganisms from bacteria to eukaryotes. Thus, we have reviewed the literature on the physical conditions of space habitats that have an impact on both virus transmissibility and interaction with their host, which include UV radiation, ionizing radiation, humidity, and microgravity. Also, we briefly comment on the practices used on space missions that reduce virus spread, that is, use of antimicrobial surfaces, spacecraft sterilization practices, and air filtration. Finally, we turn our attention to the health threats that viruses pose to space travel. Overall, even though efforts are taken to ensure safe conditions during human space travel, for example, preflight quarantines of astronauts, we reflect on the potential risks humans might be exposed to and how those risks might be aggravated in extraterrestrial habitats.
1. Introduction: Viruses in the Space Context
Space modules are enclosed, compact environments that harbor various microbial communities. Viruses are a significant part of such enclosed communities (Hjelmsø et al., 2019; Mora et al., 2019), some of which can be pathogenic to humans and pose a threat to individual and public health. However, viruses can also cause a range of other problems. For instance, bacteriophages can carry microbial virulence or antibiotic-resistance genes and spread them throughout bacterial populations on human bodies or in enclosed environments. Another example of the damage that viruses cause on Earth is the significant loss of crops grown for food, which is specific to plant viruses. Since human space missions are planned to mostly provide plant-based food to the astronauts, it is easy to see their threat to space travel. Therefore, plant and microbial viruses, along with human pathogenic viruses, represent a major issue for space travel. Space modules provide exceptional conditions for Earth's microbes to spread and grow (McKernan et al., 2008) due to high radiation doses, microgravity, and compact spaces (Fig. 1).
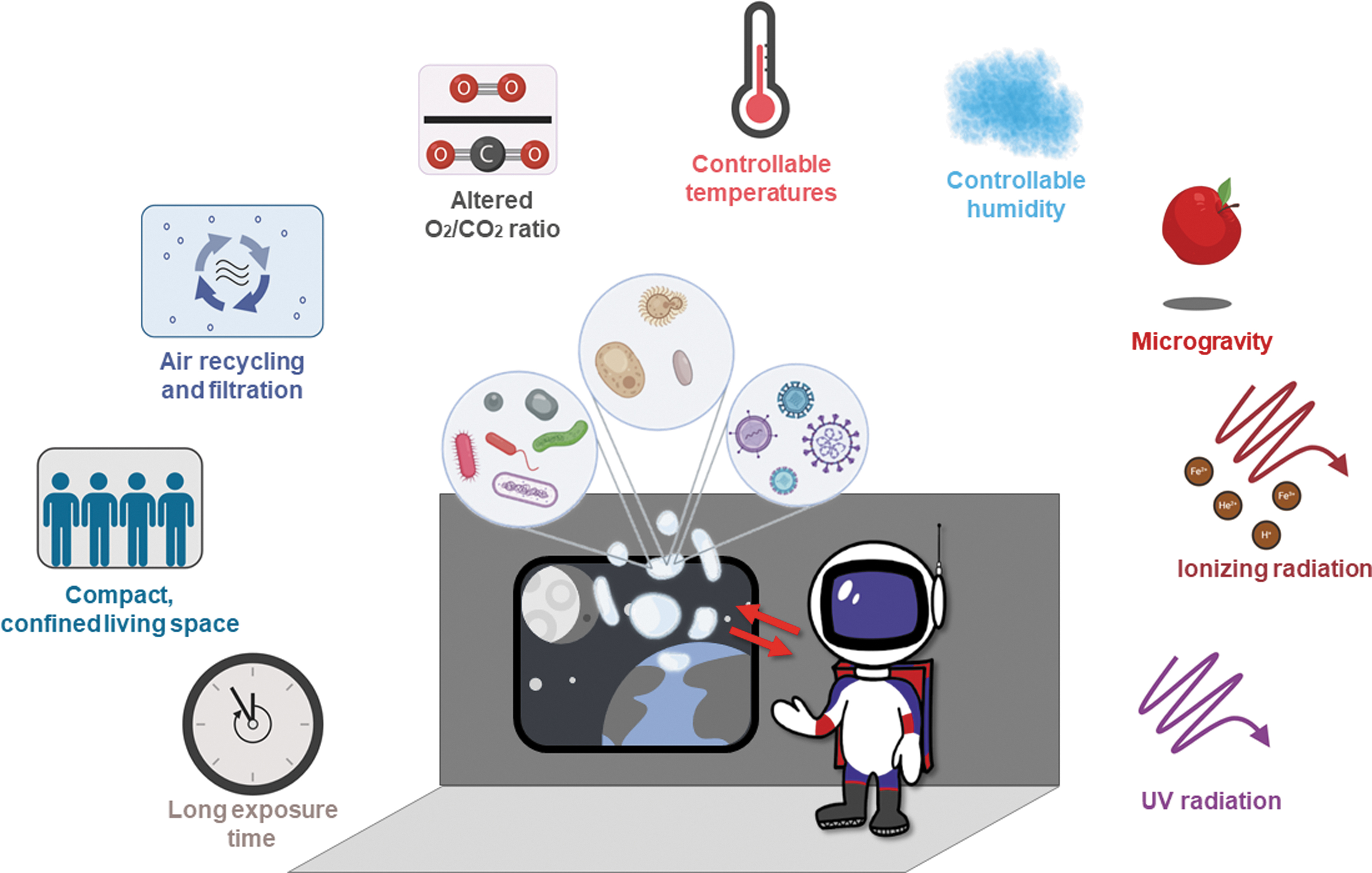
The environmental factors acting on microbes (including viruses), humans, and their interaction in space environments. Created with
Due to the extreme conditions in space, astronauts are especially vulnerable to infections given that cosmic radiation, microgravity, and psychological stress tend to compromise the human immune system (Crucian et al., 2015; Fernandez-Gonzalo et al., 2017; Akiyama et al., 2020). Before departure to the International Space Station (ISS), crew members go through a 7-day isolation known as the “Health Stabilization Program” (NASA, 2010). Crucian and colleagues reported the occurrence of microbial diseases, cold sores, and allergies among 50% of the crew members in 38 six-month missions (Crucian et al., 2016a).
Researching viruses is important in the space industry because the unique conditions of space travel can weaken human immunity. Therefore, viral infections during space travel may have a detrimental impact on the success of human missions. In contrast to the bacterial and fungal microbiomes, research on the virome variability in spacecraft is scarce (Berliner et al., 2018). The study of viruses is challenging due to the need for a host, a low biomass in the environment, and a complex phylogeny. Nevertheless, new methods for high-throughput DNA-sequencing enable the collection of high amounts of environmental sequence data, which illustrates viral diversity (Roux et al., 2017; Berliner et al., 2018; Nooij et al., 2018; Ann Gregory et al., 2019). Metagenomics allows the detection of previously unknown viruses (Delwart, 2007) and has revealed that viruses constitute a major part of most microbiomes on Earth (Rosario and Breitbart, 2011; Mokili et al., 2012). Additionally, other studies that have implemented both metagenomics and culture methods in buildings and transport vehicles have shown that most bacteria originated from the human skin both on surfaces and in the air of enclosed spaces (Tsai and Macher, 2005; Gibbons et al., 2015; Hsu et al., 2016; Stephens, 2016). Yet it is not clear whether the same assumption holds for viruses. Literature reports on viral diversity in closed environments vary, depending on the source of microbes (Prussin and Marr, 2015; Prussin et al., 2019, 2020). Prussin and Marr identified the outside environment as the major source of microbes in an indoor environment (Prussin and Marr, 2015). Also, metagenomic studies on the seasonality of microbial distribution in bioaerosols suggest that humans in the enclosed environments strongly affect the airborne viral communities (Prussin et al., 2019). On a space station, however, the source of microbes is represented by the interchange of astronaut and microbe cross-contamination from humans to equipment and subsequently from equipment to humans. The low numbers of passengers make the ISS a microbiologically controlled environment. Additionally, it allows for thorough microbial monitoring of equipment or the astronauts themselves by the implementation of strict hygiene measures, methods of sterilization, and food monitoring (Pierson et al., 2013). Nevertheless, the ISS might have a very dynamic virome.
To better understand how we can monitor and control viral spread in space travel, the present study addresses four main questions: What is the abundance and diversity of viruses in the ISS microbiome? How are viruses and their human hosts influenced by the environmental conditions of space travel? How can viruses be monitored and, in the case of harmful contaminations, decontaminated during space missions? Are there any health threats associated with viruses in the space context?
2. Viruses in the Microbiome of the ISS Surfaces
Our understanding of the viral microbiome dynamics on the ISS is sparse, mostly due to limited methodologies with which to study it. So far, there has only been one analysis of the ISS microbiome that included viruses (Mora et al., 2019). Therein, shotgun metagenomic sequencing of environmental surface swabs characterized the microbiome inside the ISS. The sequenced reads were then assigned based on sequence similarity to phylogenetic groups in virus genome databases. The reads similar to virus sequences made up 0.57% (21,415 out of 3,731,403) of all sequence reads. The majority of virus-related reads (∼95%) originated from bacteriophages, while ∼4% were derived from animal/human viruses, including herpesviruses, and the remaining were classified as reads related to plant and algal viruses or remained unclassified (Fig. 2). Among them, the reads similar to viruses from 72 different virus genera were identified to be distributed in 21 families, including the ones that contain human pathogens (Fig. 3). It is also worth noting that the metagenomic analysis was performed only on the pooled subset of environmental samples. The average length of reads was 126 bp. Those are relatively short reads. Therefore, some viruses might have been missed during the analysis.
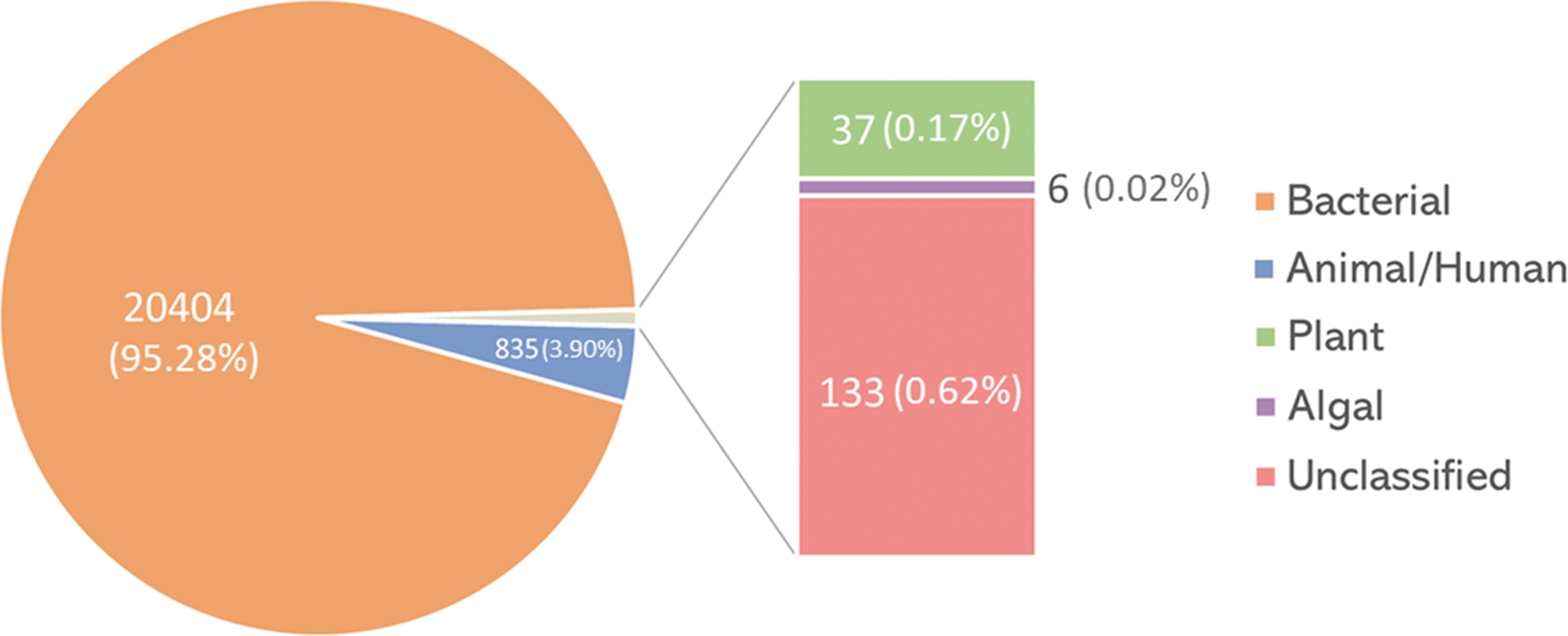
Distribution of viruses by the number of reads detected on ISS surfaces. The total number of detected reads is designated for every category. Data from the work of Mora et al. (2019). Created with MS Excel v2102.
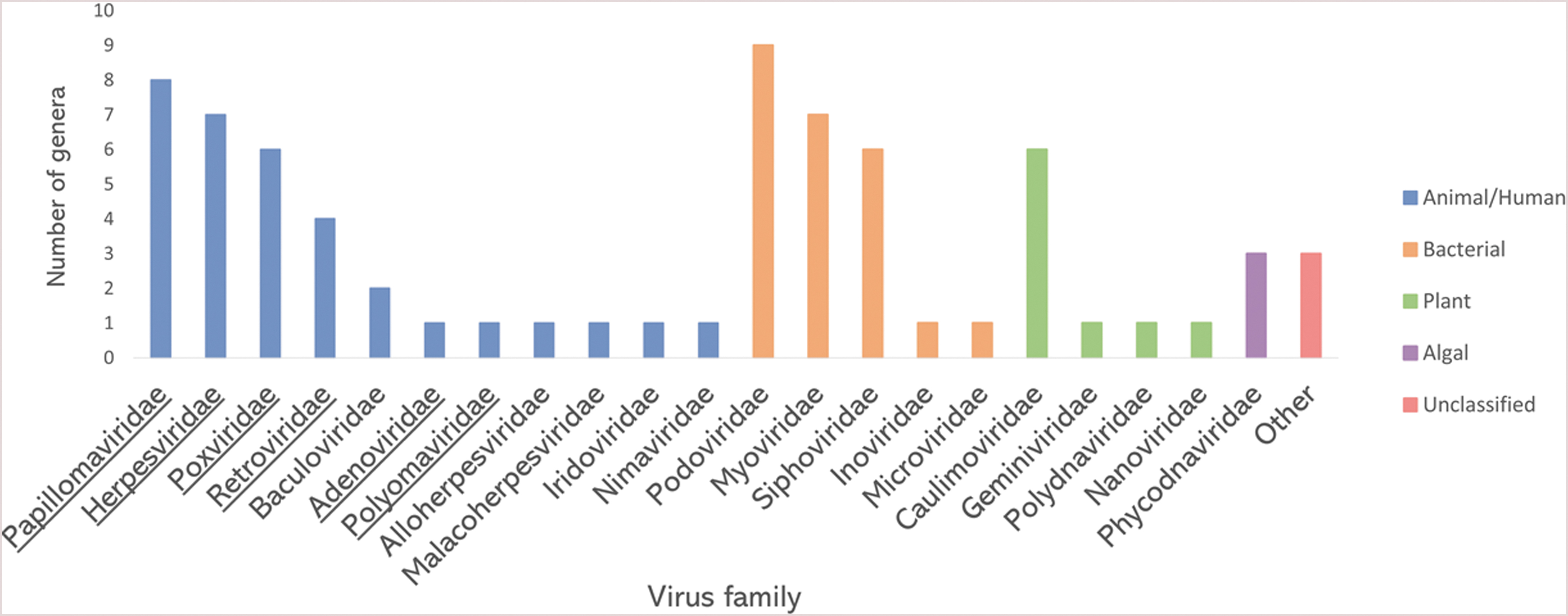
Virus families detected on ISS surfaces by shotgun metagenomic analysis. For every family, the number of detected genera is plotted. The families that contain human pathogens are underlined. Data from the work of Mora et al. (2019). Created with MS Excel v2102.
The low abundance of reads similar to virus sequences may be due to the highly sterile conditions on the ISS or caused by the decreased stability of virus samples in comparison to other microbes. Also, the viral genomes are underrepresented in genomic databases used for assigning sequences, so a great portion may remain unidentified. Bacteriophages influence the human microbiome and physiology by altering an organism's microbiome (Navarro and Muniesa, 2017) with potential impacts on the astronauts' health. Reads similar to animal viruses were distributed into 33 genera, 13 of which are known to infect humans and cause diseases of varying severity. They include a range of herpesviruses, which establish latency and can undergo reactivation (Pierson et al., 2005; Mehta et al., 2014, 2017; Rooney et al., 2019; Voorhies et al., 2019). These authors' analysis results indicate that pathogenic viruses were present in low abundance and unlikely to cause significant health problems on short-term space missions, even under conditions unfavorable to a healthy immune system. However, their impact on long-term missions remains unknown.
3. The Influence of Environmental Factors Related to Space Travel on Viruses and Their Hosts
During space travel, humans and their microbiome are exposed to conditions that significantly differ from those in their natural environment. As the ISS orbits Earth at around 420 km above sea level, exposure to cosmic and UV radiation is much higher than on the ground due to the filtration of UV by the ozone layer. There is also the additional stress of microgravity in space. Furthermore, space missions can last 3–6 months, and future missions could last up to a few years. These environmental factors can affect virus integrity directly and influence their stability or indirectly influence the host vulnerability to infection (Foster et al., 2014; Carratalà et al., 2017).
As human space missions are planned more robustly than ever, it is also necessary to consider the effects of the space environment on viral infectivity and environmental stability. This is a highly complex topic given that viruses are influenced by a range of environmental factors related to space travel, depending on the mission. Some examples include extreme and rapid temperature variations during day/night cycles on the Moon, extremely low pressures of the Moon and Mars, or microgravity in deep space. Also, the fine regolith dust of varying chemical composition, present on many rocky celestial bodies, can potentially affect the stability of a viral particle. Since astronauts will spend a long time confined in enclosed habitats, as they currently do on the ISS, the conditions inside them would be the most relevant for human health and virus spread. Therefore, for this mini review, we summarized the effects of physical conditions that affect viruses and their hosts in space habitats, that is, elevated levels of UV and ionizing radiation, humidity since it is an important factor of the enclosed environment that affects viral spread, and microgravity because it is currently impossible to control and has a considerable effect on virus-host interaction. A summary of studies addressing the impact of space-related stresses on virus stability is presented in Table 1. Also, the physiological stresses for humans in such isolated conditions include psychological stress, nutrient availability, close contact with other crew members, artificial light/dark cycles inside the habitat, among others. Even though these are important factors to consider in the future, they are not covered here as their consequences vary among individuals and bear a minimal source of concern in comparison to the physical factors explored in this review.
Summary of the Studies of Individual Simulated Stress Factors Acting on Viruses during Air and Space Travel
The routes of infection and host for each virus are specified. Herein are included bacteriophages due to the direct impact in bacterial symbionts affecting the host microbiome.
Over the course of spaceflight history, there has only been one reported outbreak in space—the common cold (head cold) outbreak among the three Apollo 7 astronauts, which spread rapidly and reduced the ability of the astronauts to cooperate with the control center (NASA, 1968). The reasons for the lack of reports are mainly pre/flight quarantine, normal mission duration of up to 6 months, but also the confidentiality of the astronaut medical data (Crucian et al., 2016a). However, reactivation of latent viruses has been documented in astronauts on the ISS, which caused skin rash and rhinitis in some exceptional cases (Crucian et al., 2016b). Varicella zoster virus (VZV) is one of the viruses reactivated in astronauts, which is known to cause significant pain and tissue damage in some cases. Therefore, vaccination of astronauts against VZV will be helpful to reduce the symptoms during space missions. Figure 4 illustrates the reported virus-related events over spaceflight history.
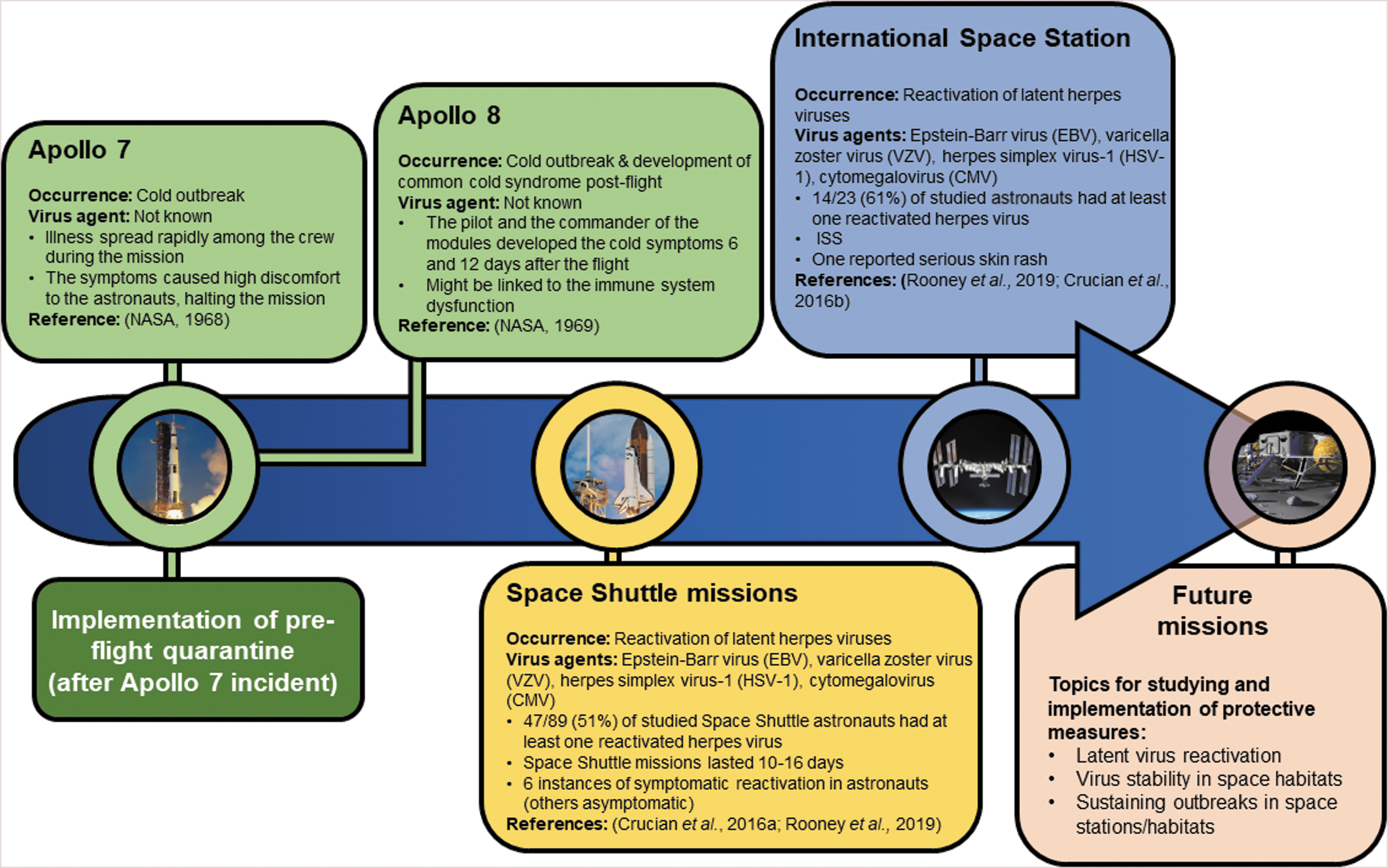
The occurrence of reported virus-related events over spaceflight history. The 7-day preflight quarantine of the astronauts helped sustain the infectious diseases on space missions as there are no reported outbreaks after its implementation. However, this might also be due to confidentiality of astronaut medical data. Currently, only reactivation of herpes viruses is being reported as a problem on space missions; though this is generally asymptomatic, it causes health issues, especially in the long term.
3.1. UV radiation
Most damaging UV radiation is filtered by Earth's atmosphere; therefore, all species on Earth are protected from most of the UVC, some UVB, and UVA to a lesser extent (De Gruijl and Van der Leun, 2000). UV radiation is one of the most threatening and damaging factors in the space environment for humans and microorganisms, along with ionizing radiation (gamma rays, X-rays, and fast charged particles). Those factors are also of concern on the Moon and Mars (Furukawa et al., 2020). UV has a major impact on viruses as discussed below. Interestingly, it has been proposed that viral populations can contain subgroups that are more resistant to UV inactivation as explained by the two-hit model of inactivation, which postulates that “two hits” of radiation rays are required to inactivate a virus particle (Kowalski et al., 2000; Cutler et al., 2011).
Ultraviolet radiation substantially reduces virus titers on surfaces and in the air (Tseng and Li, 2005, 2007; Sagripanti and Lytle, 2011). UVC radiation at 254 nm wavelength can induce damage to the viral genome and proteins (Beck et al., 2014, 2016, 2018). Overall, DNA viruses tend to be more resistant than RNA viruses, and those with double-stranded genomes are more resistant to UV radiation (Tseng and Li, 2007). Yet, independently of the nucleic acid type, genome damage is the most important factor for viral inactivation (Wang et al., 2004; Ye et al., 2018).
Also, UV radiation can reactivate dormant viruses in rodents (Garssen et al., 1995; El-Ghorr and Norval, 1996; Norval and El-Ghorr, 1996; Goade et al., 2001; Norval, 2006; Viarisio et al., 2011). Epidemiological data on papillomaviruses and herpesviruses suggest the same assumption holds for humans (Chen et al., 2008; Hampras et al., 2014; Uberoi and Lambert, 2017). The mechanism of this reactivation is related to how UV radiation suppresses the immune system (Norval and Halliday, 2011; Schwarz and Schwarz, 2011; Ullrich and Byrne, 2012) by activation of regulatory T cells (thymus cells, a type of lymphocyte) that produce immunosuppressive signals and thus inhibit the immune system (Rana et al., 2008). This paves the way for opportunistic disease-causing viruses to exploit a compromised immune system and provoke disease. Therefore, treatments that influence T cell signaling might impact the astronaut's health and contribute to virus control.
3.2. Ionizing radiation—gamma rays, X-rays, and charged particles
Ionizing radiation is another factor of space travel that influences living organisms, which are shielded from its effects on Earth due to the planet's magnetic field and the ozone layer. Those are gamma and X-rays that originate from the Sun or charged ions (H+, He2+, Fe ions) coming from interstellar space (Horneck et al., 2010; Chancellor et al., 2018). This type of radiation can induce significant damage to biomolecules by causing the formation of reactive oxygen species (ROS) and by breaking the molecular bonds (Reisz et al., 2014; Cortesão et al., 2020) and cause double-stranded breaks in DNA (Vignard et al., 2013).
Long-term exposure to low-intensity ionizing radiation increases the chances of developing cancer, especially of the skin and lungs (Cohen, 2002; WHO, 2016). Rodent models show that both gamma and X-rays have immunosuppressive effects that lead to the reactivation of herpes and rabies viruses and an increase in illness severity (Openshaw et al., 1979; Rodiriguez et al., 1990; Wang et al., 1990; Ceccaldi et al., 1996). One of the most dramatic effects is the apoptosis of dendritic cells, which neutralizes their defensive action that controls B and T cell immune response (Liu et al., 2011).
Ionizing photons also inactivate environmental viruses directly and indirectly (Johnson, 1965; Sullivan et al., 1971; Ward, 1980; Hume et al., 2016) mainly by damaging viral genomes and thus compromising viral replication (Summers and Szybalski, 1967; Ohshima et al., 1996; Lomax et al., 2013). Direct inactivation acts by damaging the viral genome, proteins, and lipids. The indirect mechanism of inactivation acts through the generation of hydroxyl radicals from water, ozone, and oxygen molecules, which originates ROS and which then damages nucleic acids, proteins, and lipids.
Studies suggest that different types of radiation can induce the lytic cycle of herpesviruses—Epstein–Barr virus (EBV) and human herpesviruses (HHV) types 1–3 (Ramirez-Fort et al., 2018; Mehta et al., 2018). Hence, while immunosuppression plays a role in the reactivation of herpesviruses, ionizing radiation can also directly activate lysogenic viruses (Ramirez-Fort et al., 2018).
3.3. Humidity
Humidity is another important environmental factor that can be controlled in space modules and has a known impact on viruses and other microbes (Yamaguchi et al., 2014). It also influences the virus-host interaction. Some studies have shown that relative humidity (RH) can be a predictor of viral stability (Shaman and Kohn, 2009; Tang, 2009). In these studies, stability depended on the presence of a lipid envelope. Viruses with an envelope conveyed more stability at lower RH (20–30%), while those without an envelope and a protein shell are more stable at higher RH (70–90%). However, recent studies on influenza and SARS-CoV-2 suggest that absolute humidity (AH) is a better predictor for stability (Deyle et al., 2016; Marr et al., 2019; Haque and Rahman, 2020; Wang et al., 2020). At higher AH, respiratory viruses show reduced transmission. This is because high AH can be only reached at higher temperatures, so the effect of AH is the combined action of RH and high temperature (Marr et al., 2019). In addition to inactivating airborne infectious viruses, high humidity thickens human mucosa, which acts as the barrier between the organism and environment, therefore reducing the possibility of viral infection from the air (Kudo et al., 2019). In the case of viral outbreaks in space habitats, humidity could be regulated to decelerate viral spread.
Hypothetically, viruses with lipid envelopes accumulate on the surface of water droplets in the air and are afterward inactivated by surface tension (Yang and Marr, 2012). At increased RH, droplets are larger because there is less evaporation that leads to a higher accumulation of lipid-enveloped viruses being inactivated by surface tension. On the other hand, non-enveloped viruses are repelled by water surfaces, which makes inactivation less efficient. Also, higher evaporation rates at lower RH decrease the pH of aerosol droplets, which induces conformational changes on surface proteins. This can make non-enveloped viruses less stable (Yang and Marr, 2012). However, these scenarios are currently only hypotheses, and more mechanistic research is needed to understand virus inactivation by RH and AH. Accordingly, bacteriophages with a protein envelope are better inactivated by UV radiation when exposed to low RH (Tseng and Li, 2005, 2007), but for a porcine reproductive and respiratory syndrome virus (PRRSV), a moderate RH (25–79%) enhances UV inactivation (Cutler et al., 2012) for unknown reasons.
3.4. Microgravity
In space travel, exposure to varying g-forces is very common. Spacecraft experience high g-forces during liftoff and are under the influence of microgravity once they reach orbit. Also, future celestial destinations like the Moon and Mars have lower gravitational force than Earth. Therefore, research of viruses in microgravity-like conditions is more relevant in the context of space travel, as well as hypergravity, which is relevant in the consideration of high g-forces experienced during launching into orbit and orbital transfers.
Research on virus-host interactions in microgravity scenarios indicates that it results in beneficial effects, contrary to other factors that induce viral reactivation (e.g., ionizing radiation), (Honda et al., 2020). Honda and colleagues found that cells infected with Kaposi's sarcoma-associated herpesvirus (KSHV) upregulate cell-intrinsic KSHV-inhibiting restriction factors in microgravity like CCCTC-binding factor (CTCF) or adenosine monophosphate–activated protein kinase (AMPK). This suggests that microgravity alone could partially counteract the damaging or debilitating effects of other space travel stress factors.
However, additional evidence displays a negative impact of microgravity. The intestinal epithelial cells have been shown to experience a decrease in their epithelial barrier function under microgravity (Alvarez et al., 2019). The gut harbors its microbiome, and it prevents viruses and other microbes from entering inside an organism. In this research, ion flux and fluorescein isothiocyanate-dextran (FITC-dextran) permeability of human epithelial barrier were measured in vitro, and it was found that the permeability increased, potentially allowing viral spread.
4. Control of Viruses in Space Travel
Innovative and efficient ways for virus identification, tracking, and inactivation are crucial to tackling the vast spectrum of problems they cause in public health, the economy, and agriculture. This is also important in spaceflight to prevent hindering missions due to viral infections. Various measures are taken to ensure the sanitary conditions aboard spacecraft, from microbial tracking to preflight astronaut isolation (Pierson et al., 2013).
Additional decontamination is applied to spacecraft with a special focus on bacterial and fungal spores, preventing microbial spread to other celestial bodies. Developing novel methods of disinfection and microbial control in space travel poses a challenge because they should efficiently fulfill the healthcare requirements compatible with human exposure without affecting the structural integrity of a spacecraft. For this reason, chemical disinfection is often not suitable for space applications.
4.1. Antimicrobial surfaces
Certain materials and compounds have antimicrobial properties that are exploited to inhibit or reduce microbial growth in environments where strict hygiene standards are necessary, such as airplanes, spaceflight, healthcare, or food production (Page et al., 2009; Mousavi Khaneghah et al., 2018).
Metals with antimicrobial properties are copper, silver, and their respective alloys, but also ions of mercury, iron, lead, zinc, and aluminum (Sreekumari, et al., 2005). Including a minimum of 55% of copper into composite materials (Mehtar et al., 2008) would contribute to ensuring safety during air and space travel as it efficaciously inactivates most viruses, including SARS-CoV-2 and other microbes (Noyce et al., 2007; Warnes et al., 2015; Schmidt et al., 2017; Bryant et al., 2021). There are also efforts to develop alternative antimicrobial surfaces for space travel such as AGXX (Van Loi et al., 2018), a silver/ruthenium surface coating that produces ROS, which inactivates most pathogens, including viruses.
4.2. Sterilization of spacecraft
There is a concern to protect other planets from contamination with Earth's microbes. Spacecraft surfaces are sterilized inside and out by intense treatment at high temperatures (145°C) for several days, which guarantees that no microbes or spores can survive. In recent years, plasma sterilization has been considered an effective alternative due to its more convenient application (Stapelmann et al., 2013). These methods of sterilization are collaterally highly effective against viruses (Bozkurt et al., 2015; Sakudo et al., 2019), making antimicrobial disinfection overly effective in preventing virus contaminations.
Interestingly, full microbial sterility of spacecraft in human missions is hard, if not impossible, to achieve because astronauts themselves represent a reservoir of microbes that can cross-contaminate the environment. Development of simple and easy-to-use detection tests such as SHERLOCK (Gootenberg et al., 2018) or DETECTR (Chen et al., 2018) for specific viral genomes would be helpful in space missions. Such tests utilize CRISPR-Cas9 technology to specifically detect viral sequences within minutes. Also, environmental nucleic acid sequencing techniques like Oxford Nanopore (Quick, 2019) are becoming more robust and easier for application in extreme environments such as space missions. This will vastly aid the detection and characterization of viromes in enclosed environments, not just in space habitats, but also on Earth.
4.3. Air filtration
Inside the ISS, there is constant air circulation and filtration with high-efficiency particulate air (HEPA) filters. They have been reported to efficiently filter out small aerosols (98% efficiency, particles 0.3–10 μm diameter; Mousavi et al., 2020). Even though virus sizes are in nanometer range, they travel in air-suspended droplets and aerosols that are micrometer-sized, being efficiently filtered by HEPA filters. Therefore, they protect the astronauts. Those filters also provide an interesting test sample for researching airborne microbial communities on the ISS. Probably, future space habitats will also include such filters due to the need for constant air recycling.
5. Health Threats Viruses Pose to Space Travel
5.1. Latent infections and viral reactivation
Even though disinfection on space missions is thorough, it is impossible to completely neutralize the disease-inducing factors due to their prevalence in the human hosts, since a large portion of humanity is already latently infected with specific viruses. Latent infections are caused by viruses that, upon a single inoculation, can establish lifelong infections, like herpesviruses. Table 2 lists herpesviruses that cause latent infection in humans regarding global prevalence, route of transmission, possible consequences, and site of persistence. As a result of immune deregulation in space, these viruses can undergo reactivation, potentially with increased severity due to the vulnerability of the host. Their reactivation in astronauts is probably caused by the stress factors discussed above, resulting in the changes in CD8+ T cell (T lymphocytes expressing cluster of differentiation 8 glycoprotein) and regulatory T cell function, which have been described to maintain viral latency (Mehta et al., 2013).
The Global Prevalence, Route of Transmission, Some Possible Consequences of Primary Infection and Reactivation, and Site of Persistence of Herpesviruses Causing Latent Infections in Humans: HSV-1 and HSV-2, VZV, EBV, HCMV, HHV-6 and HHV-7, and KSHV
B cells = bursa cells, a type of lymphocyte; EBV = Epstein–Barr virus; HCMV = human cytomegalovirus; HHV = human herpesvirus; HSV = herpes simplex virus; KSHV = Kaposi's sarcoma-associated herpesvirus; T cells = thymus cells, a type of lymphocyte; VZV = varicella zoster virus.
Multiple studies have detected reactivation and shedding of viruses in human space and analog missions and environments (Pierson et al., 2005, 2007; Mehta and Pierson, 2007; Mehta et al., 2014, 2017). Due to the prevalence of herpesviruses in the general population, reactivation events cannot be reliably avoided in space either by isolation or by medical treatment. Therefore, developing spaceflight countermeasures to attenuate viral reactivation outcomes such as preflight immunity enhancement to inhibit viruses is a factor to be considered. Though herpesviruses are not the only viruses that latently infect humans, they are the major focus in spaceflight (Rooney et al., 2019). Thus, further studies of latent viral infections are necessary to determine the vulnerability of astronauts to other latent infection viruses besides those belonging to the Herpesviridae family.
The reactivation of these viruses, some of which are associated with increased mortality (Ren et al., 2020), has also been detected in astronauts. Besides the cold-sore-causing HSV-1 (Crucian et al., 2016a), the reactivation of other herpesviruses such as EBV (Payne et al., 1999), human cytomegalovirus (HCMV; Vuong et al., 2000), and VZV has been observed in astronauts before with mild symptoms (Mehta et al., 2004). Though serious consequences have not yet been observed in astronauts, this might be due to the currently short duration of human missions. However, planned long-term missions carry the danger of astronauts developing severe symptoms stemming from latent viral infections. This is especially dangerous due to the limited resources available in space missions to isolate and treat the affected individuals.
5.2. Protecting astronauts from virus infections
In addition to infection by reactivation of latent viruses, there is also a possibility of virus infection on space missions that increases the risk of outbreaks in modules and habitats. Due to the limited possibility of identifying the cause of infection in space, it is challenging to recognize viral infections in addition to treating them. So, how can astronauts protect themselves from viral infections? Currently, most herpesvirus infections cannot be prevented through vaccines, with the exception of VZV, the causative agent of chickenpox and zoster (Papaloukas et al., 2014). A balanced diet that supports a healthy metabolism, boosting the immune response, like probiotics or foods rich in vitamins, minerals, or amino acids could in theory support the fitness of the immune system, though the research in this area is still ongoing (Perdigon et al., 1995; Mora et al., 2008; Crucian et al., 2018). Physical exercise has been found to significantly contribute to the reduced reactivation of viruses in astronauts on the ISS (Agha et al., 2020). A more drastic approach would consist of using immunostimulant drugs such as bacille Calmette–Guérin (BCG), levamisole, isoprinosine, or others (Bascones-Martinez et al., 2014). Anti-herpes products like acyclovir can be used to treat herpesviruses, but these have shown toxicity with prolonged use (WHO, 2013). Current journeys to space are limited in time, but in the case of longer journeys to Mars or further, latent viruses could have a greater impact.
Currently, the standard safety procedure of human spaceflight is the preflight astronauts' quarantine and disinfection of cabins and equipment. However, we must consider the risk that some viral infections might go unnoticed during the quarantine period and cause significant harm once in the space station. Development of optimized methods for virus detection and calculating the impact of the space environment on virus spread will help address this problem and provide the basis for the development of improved protocols to control eventual outbreaks in space.
6. Conclusion and Outlook
Viruses are a diverse biological group that is part of microbial communities in human-inhabited space modules. As such, they can influence astronauts' well-being and may pose a health threat to the crew. Intensive research is required in the field of space virology to improve the current knowledge on the dynamics caused by space stress. The effect of extreme g-forces or microgravity on viruses is underrepresented. It would be, for instance, interesting to test the influence of microgravity on viral spread throughout the body. Also, studies showed that hypergravity encourages the proliferation of healthy cells (Ciofani et al., 2012; Genchi et al., 2016), while the effect on viruses or infected and immune cells is not known. Research of viral UV-stability could be used to develop postflight UV-based sterilization of spacecraft cabins as an easy and efficient method for viral elimination. It would be especially interesting to further investigate the inactivation of human pathogenic viruses by UV radiation in various levels of RH. Developing new antimicrobial materials is another promising method for limiting viral spread during space travel.
Since the plans of future space missions tend to be more prolonged, preflight isolation and a healthy immune system might not be enough to protect astronauts against some viruses due to the overwhelming conditions during space travel. Hence, developing new methods for the detection and treatment of viral infections in space is a relevant topic.
Footnotes
Acknowledgments
We thank Christine Moissl-Eichinger and her entire team for the fruitful discussion and generous exchange of scientific data. We express our gratitude to Melanie Brinkmann and Satish Mehta for their critical reviews and valuable suggestions. We acknowledge Kevin McAlpin's help with the English revision of the manuscript.
Funding
The Aerospace Microbiology research group received funding from the following DLR grants: ISS LIFE (Programm RF-FuW, Teilprogramm: 475), AEROMASK, NGT-BIT (DLR V&E: Next Generation Train), and simplAIR (Programm LF (simplAIR ME / KoPa33), Teilbereich: 251). M.C. was supported by the DLR/DAAD Research Fellowship Doctoral Studies in Germany, 2017 (57370122) and B.P. by an ERASMUS+ fellowship (2019-1-HR01-KA103-060250). These results will be included in the PhD thesis of Bruno Pavletić.
Abbreviations Used
Associate Editor: Lewis Dartnell
