Abstract
Ultraviolet (UV) light plays a key role in surficial theories of the origin of life, and numerous studies have focused on constraining the atmospheric transmission of UV radiation on early Earth. However, the UV transmission of the natural waters in which origins-of-life chemistry (prebiotic chemistry) is postulated to have occurred is poorly constrained. In this work, we combine laboratory and literature-derived absorption spectra of potential aqueous-phase prebiotic UV absorbers with literature estimates of their concentrations on early Earth to constrain the prebiotic UV environment in marine and terrestrial natural waters, and we consider the implications for prebiotic chemistry. We find that prebiotic freshwaters were largely transparent in the UV, contrary to assumptions in some models of prebiotic chemistry. Some waters, such as high-salinity waters like carbonate lakes, may be deficient in shortwave (≤220 nm) UV flux. More dramatically, ferrous waters can be strongly UV-shielded, particularly if the Fe2+ forms highly UV-absorbent species such as
1. Introduction
Akey challenge for origin-of-life studies is to constrain the range of environmental conditions on early Earth under which life arose. Knowledge of these environmental conditions informs development of theories of the origin of life and enables assessment of the plausibility and probability of postulated prebiotic chemistries (Pace, 1991; Cleaves, 2013; Barge, 2018; Lyons et al., 2020). Consequently, considerable work has been done to constrain the range of physicochemical conditions available in prebiotic environments (Sleep, 2018; Sasselov et al., 2020).
An important prebiotic environmental factor, particularly for surficial prebiotic chemistry, is the ultraviolet (UV) irradiation environment. UV photons can destroy nascent biomolecules, motivating a search for mechanisms to protect prebiotically important molecules from UV irradiation (Sagan, 1973; Holm, 1992; Cleaves and Miller, 1998), but UV light has also been suggested useful to the origin of life as a source of chemical free energy and selectivity (Deamer and Weber, 2010; Pascal, 2012; Beckstead et al., 2016). Indeed, UV light has been experimentally shown to drive a range of prebiotic chemistries (Ferris and Orgel, 1966; Sagan and Khare, 1971; Flores et al., 1977; Pestunova et al., 2005; Bonfio et al., 2017; Mariani et al., 2018; Xu et al., 2020). Surficial prebiotic chemistries, therefore, broadly fall into two classes: those for which UV light is strictly destructive and must be mitigated or avoided, and those for which UV light is essential and must be sought (Ranjan and Sasselov, 2016; Todd et al., 2020). The self-consistent availability of UV radiation is consequently a crucial part of the assessment of the plausibility of proposed prebiotic chemistries.
The pivotal roles that UV radiation plays in diverse prebiotic chemistries have motivated increasingly sophisticated estimates of the prebiotic UV environment, with particular emphasis on atmospheric transmission. Models generally predict abundant steady-state UV at wavelengths
However, prebiotic chemistry is generally proposed to occur in aqueous reservoirs such as ponds or oceans, which may host UV-blocking compounds of geological origin (Martin et al., 2008; McCollom, 2013; Patel et al., 2015; Benner et al., 2019; Becker et al., 2019). To date, estimates of UV transmission in prebiotic natural waters are generally based on pure water or modern pond water (Cockell, 2000; Ranjan and Sasselov, 2016; Pearce et al., 2017).
Neither modern pond water nor pure water is likely representative of prebiotic waters. The UV-opacity of modern pond waters is largely driven by biogenic dissolved carbon species (Morris et al., 1995; Markager and Vincent, 2000; Laurion et al., 2000). In the limit of low biological productivity, as expected in a prebiotic world, natural waters can be clear down to 300 nm; their transmission from 200 to 300 nm is unconstrained but has been extrapolated to be similarly low (Smith and Baker, 1981; Morel et al., 2007).
At the other extreme, although pure water is clear in the near-UV (Quickenden and Irvin, 1980), the waters in which prebiotic chemistry occurred must by definition have been impure, since they must have contained, at minimum, the feedstocks for that chemistry, and likely other geogenic constituents as well (e.g., Toner and Catling, 2020). Absorption due to these constituents may influence UV transmission. The UV transmission of prebiotic waters likely lies, therefore, between the extremes of high transparency and high opacity represented by the previously utilized proxies of pure water and modern pond water.
In the present study, we constrain wavelength-dependent UV transmission in prebiotic waters. We focus on absorption due to a subset of UV-active species that have been proposed to be present in prebiotic waters. We derive molar absorptivities of these compounds from a combination of literature reports and our own measurements. We draw on literature proposals for the concentrations of these species in representative prebiotic waters. We do not attempt self-consistent geochemical modeling of these waters; such work is important but requires improved measurements of the relevant reaction kinetics under prebiotically relevant conditions, which is beyond the scope of the present study. Our work is similar in spirit to the work of Cockell (2000, 2002), who drew on literature proposals for the composition of the early atmosphere to constrain its UV transmission, without attempting to model its self-consistent photochemistry. Their simple initial analysis guided early work (e.g., Pierce et al., 2005; Gómez et al., 2007) and motivated more atmospherically sophisticated follow-up (e.g., Rugheimer et al., 2015), which came to largely similar conclusions. Similarly, we hope our simple study will guide prebiotic chemistry, while motivating more geochemically sophisticated follow-up work.
2. Background
Relatively few studies have constrained the UV transmission of prebiotic natural waters. A notable exception is the work of Cleaves and Miller (1998), who considered potential “sunscreens” for the prebiotic ocean. Cleaves and Miller (1998) measured the UV absorptivities of salts, hydrogen cyanide (HCN) and spark discharge-derived polymers, HS−, and marine Fe2+. Based on a combination of calculated and literature estimates of the concentrations of these species, Cleaves and Miller (1998) concluded that, in favorable circumstances, HS−, marine Fe2+, or spark discharge polymer could have extinguished UV in the surface layer of the prebiotic ocean. The conditions required for accumulation of a spark discharge polymer to high concentrations in the prebiotic ocean would have included the emission of prebiotic volcanic carbon as CH4, its conversion with unity efficiency to a spark discharge polymer, and its efficient deposition to the ocean. However, more recent modeling studies have indicated that volcanogenic carbon is emitted even on early Earth-like planets as CO2 and not CH4 and the primary fate of abiotic CH4 should be oxidation to CO2 (Kasting, 2014). Such models suggest that the conditions required for accumulation of optically relevant concentrations of spark discharge polymers are unlikely to be met in the steady state, although high hydrocarbon concentrations may be transiently possible after large impacts (Genda et al., 2017; Benner et al., 2019; Zahnle et al., 2020). Similarly, recent studies suggest that the early ocean was ferrugious (Fe2+-rich) and would have titrated out HS− as pyrite. Along with the low solubility of HS−, [HS−] was most likely very low in most natural waters on early Earth (Walker and Brimblecombe, 1985; Poulton and Canfield, 2011; Ranjan et al., 2018). However, elevated [Fe2+] was a possibility for the early ocean (Konhauser et al., 2017).
3. Methods
3.1. Calculating aqueous UV attenuation
We approximate the transmission of UV radiation in homogenous (well-mixed), nonscattering aqueous solutions at low concentrations and low light intensities by the Beer-Lambert law (IUPAC, 1997):
where I 0 is the incident irradiance, I is the transmitted irradiance, T is the fraction of transmitted radiation, ɛi is the molar decadic absorption coefficient for the ith component of the solution, ci is the concentration of the ith component of the solution, a = ɛc is the linear decadic absorption coefficient, and d is the path length. This approach considers only absorption and neglects scattering. This is a reasonable approximation because in the 200–300 nm wavelength range on which we focus, the single-scattering albedo of liquid water ω 0 << 1, that is, absorption dominates scattering (Appendix Fig. A1; Quickenden and Irvin, 1980; Kröckel and Schmidt, 2014). We follow previous workers in assuming that ɛi does not vary as a function of pH for the simple inorganic molecules we consider, that is, that changes in the absorbance of solutions of these molecules with pH are due to changes in speciation or complexation and not intrinsic changes to ɛi (Braterman et al., 1983; Anbar and Holland, 1992; Nie et al., 2017; Tabata et al., 2021). We propagate the uncertainties on ɛi under the assumption that they are independent and normally distributed (Bevington and Robinson, 2003).
3.2. Molar decadic absorption coefficients for potential prebiotic absorbers
We consider absorption due to halide anions (Cl−, Br−, I−), ferrous iron species (Fe2+,
We draw on both the literature and our own measurements for the molar decadic absorption coefficients of potential prebiotic aqueous-phase absorbers. We use our own measurements when available because our measurements typically feature broader wavelength coverage than those in the literature, and they include estimates of uncertainty and are collected by using uniform techniques (Appendix A1.1). For the ferrous iron species, we check that the dominant ion speciation does not vary due to pH drift over the course of our dilutions (Appendix A1.1.4). Our measurement techniques are potentially inaccurate for weak acids and bases because we do not control pH, which may drift during dilution steps and affect speciation. For such species, we rely instead on literature data (Appendix A1.3). Table 1 summarizes the sources of the molar decadic absorption coefficients used in this work.
Molar Decadic Absorption Coefficients Used in this Work
3.3. Abundances of potential prebiotic absorbers in fiducial prebiotic waters
Constraining the impact of potential absorbers on prebiotic aqueous transmission requires estimates of their abundances in natural waters on early Earth. Natural waters are diverse, and we cannot hope to explore this full diversity. Instead, we focus on fiducial waters motivated by proposed origin-of-life scenarios. We focus primarily on shallow terrestrial waters, for example, ponds and lakes. Such waters are of interest for prebiotic chemistry because of their propensity for wet–dry cycles, their capability to accumulate atmospherically delivered feedstocks more efficiently than the oceans, and their potentially diverse palette of environmental conditions (Patel et al., 2015; Deamer and Damer, 2017; Pearce et al., 2017; Becker et al., 2018; Rimmer and Shorttle, 2019; Ranjan et al., 2019; Toner and Catling, 2020). We specifically consider freshwater lakes, carbonate lakes, and ferrocyanide lakes. We also consider the early ocean, to offer a basis of comparison for the terrestrial waters. Our oceanic calculations may also be relevant to origin-of-life scenarios that invoke shallow waters at the land–ocean interface (Commeyras et al., 2002; Lathe, 2005; Bywater and Conde-Frieboes, 2005), but they are not relevant to deep-sea origin-of-life scenarios (e.g., Corliss et al., 1981; Sojo et al., 2016) for which water alone is enough to extinguish UV. The composition of these waters is uncertain, and so we draw on the literature to construct high and low transmission endmember cases to bound their potential UV transmission. Our construction of these endmember cases is summarized in Table 2 and detailed in Sections 3.3.1–3.3.4.
Estimated Range of Concentrations of Potential Prebiotic Absorbers in Prebiotic Waters
3.3.1. Ocean
For halide species, we scale the composition of the modern oceans. We consider a salinity range of 0.5–2 × modern, motivated by theoretical arguments and isotopic evidence (Knauth, 2005, 1998; Marty et al., 2018). On modern Earth, seawater halide concentrations are [Cl−] = 0.6 M, [Br−] = 0.9 mM, and [I−] = 0.5 μM (Channer et al., 1997; ASTM, 2013). Cl− and Br− covary in natural waters, leading us to fix their prebiotic ratios to the modern value (Hanley and Koga, 2018). De Ronde et al. (1997) used fluid inclusions to infer high [I−] at 3.2 Ga, but the interpretation of these samples is strongly contested (Lowe and Byerly, 2003; Knauth, 2005; Farber et al., 2015). We, therefore, fix our prebiotic [I−]/[Cl−] to the modern value as well and assume these ions to covary in the prebiotic ocean.
Estimates of ferrous iron concentrations in the surficial prebiotic ocean vary significantly. Halevy et al. (2017) estimated [Fe2+] < 10−9 M at the ocean surface, largely driven by the assumption of efficient photooxidation. More recent work has reported this process to be less efficient and estimated [Fe2+] = 10−4 M at the ocean surface (Konhauser et al., 2007; Halevy and Bachan, 2017; Konhauser et al., 2017). Fe2+ is predicted to be the main ferrous species at circumneutral pH for solutions with oceanic Cl− and
We take the bracketing range of oceanic [
3.3.2. Freshwater lakes
Terrestrial waters are typically dilute, with an ionic strength of order 10−3 M (Lerman et al., 1995). To represent a dilute endmember scenario for natural waters, we consider freshwater lakes with riverine composition (i.e., not evaporatively concentrated). Modern surface freshwater systems average [Cl−] = 0.2 mM, and the same is proposed for Archean and prebiotic river waters (Graedel and Keene, 1996; Hao et al., 2017). Mean [Br−]/[Cl−]≈1 − 2 × 10−3 in modern terrestrial waters (Edmunds, 1996; Magazinovic et al., 2004), which suggests mean [Br− = 0.1 − 0.2 μM].
Mean iodide concentrations in modern terrestrial waters are reported as 40 nM (range: 0.1 nM − 0.6 μM) (Fuge and Johnson, 1986), and it is not clear that I− is generally correlated with Cl− as Br− seems to be (e.g., Worden, 1996). We, therefore, consider both mean-I (40 nM) and high-I (0.6 μM) compositions. Estimates of [Fe2+] in riverine waters on early Earth span 0.1 μM–0.1 mM (Halevy et al., 2017; Hao et al., 2017), and we consider this range. Hao et al. (2017) predicted Archean river water to have a pH ≤6.34, for which Fe2+ should mainly present as
We take the bracketing range of [
3.3.3. Closed-basin carbonate lakes
Closed-basin carbonate lakes have been proposed as venues for prebiotic chemistry, because the elevated carbonate concentrations in these lakes suppress [Ca2+], permitting the accumulation of phosphate to prebiotically relevant concentrations (Toner and Catling, 2020). For an endmember, we consider a closed basin carbonate lake with 10−2 mol/kg phosphorus, which corresponds to the upper edge of phosphorus concentrations in the sample of closed-basin carbonate lakes reported by Toner and Catling (2020).
Cl− and Br− behave conservatively with P in carbonate lakes, with [Cl−] = 0.1–6 mol/kg and [Br−] = 10−3–10−2 mol/kg for [P] = 10−2 mol/kg (Toner and Catling, 2020). Projecting from Toner and Catling (2020), we simplify the calculation by approximating molarity as molality; we contend this approximation to suffice for the order-of-magnitude estimates we seek. Iodine is proposed to be delivered to surface waters via rainfall, and so we might expect it to be evaporatively concentrated in closed-basin lakes; however, we located no reports of evaporative iodide concentrations in composition studies of such lakes (Friedman et al., 1976; Eugster and Jones, 1979 ; Fuge and Johnson, 1986; Mochizuki et al., 2018; Toner and Catling, 2020; Hirst, 2013). We, therefore, consider a bracketing range of [I−] equal to the freshwater lake scenario. We take
3.3.4. Ferrous lakes
Ferrocyanide lakes have been proposed to form when ferrocyanide salt deposits are irrigated by neutral water (Toner and Catling, 2019; Sasselov et al., 2020). Ferrocyanide is an extremely potent UV absorber, and so we consider transmission in such ferrocyanide lakes. Notably, ferrocyanide has been invoked in UV-dependent prebiotic chemistries and thus provides an opportunity to examine their geochemical self-consistency (Xu et al., 2018; Mariani et al., 2018; Rimmer et al., 2018). We approximated the composition of a ferrocyanide lake to be that of a freshwater lake, but with the Fe2+ present as
4. Results
4.1. Prebiotic ocean
The prebiotic ocean efficiently attenuated shortwave UV radiation but may have admitted longer-wavelength UV radiation to depths of meters (Fig. 1). In the low-absorption endmember, the absorption is dominated by the halides, especially Br− at shorter wavelengths and I− at longer wavelengths. In the high-absorption endmember scenario, absorption is dominated by Br− at shorter wavelengths and
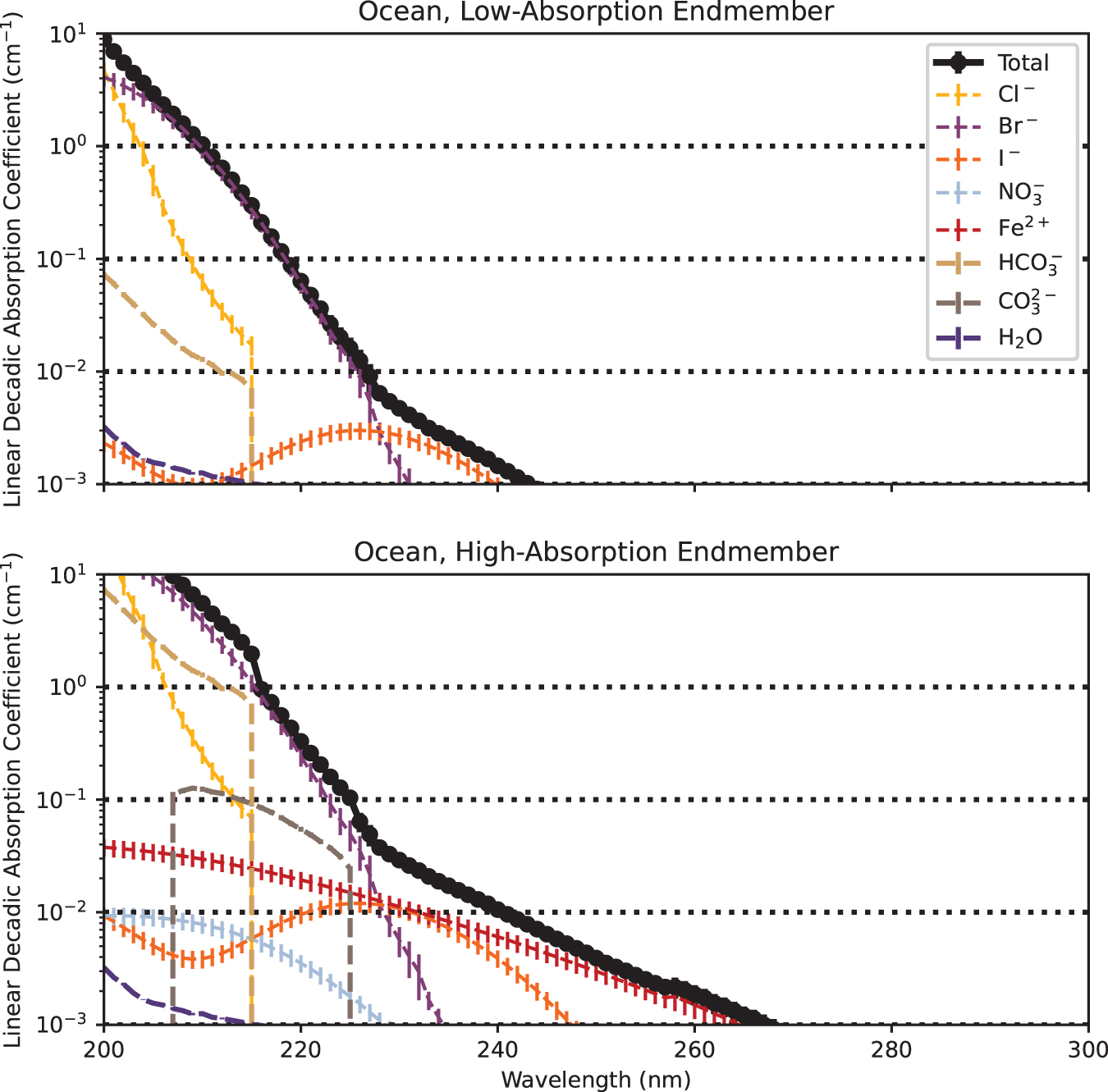
Simulated linear decadic absorption coefficients of the prebiotic ocean and its component solutes, for low-absorption and high-absorption endmember cases. Not all solutes are visible in each case, because the linear decadic absorption coefficients of some solutes fall below the lower limit of the y-axis across the wavelength space plotted here. The shortest-wavelength photons are extinct in the surface layers of the ocean, but the ∼260 nm radiation responsible for nucleotide degradation can penetrate to a depth of meters. Color images are available online.
4.2. Prebiotic terrestrial waters
4.2.1. Freshwater lakes
The transparency of shallow freshwater lakes depends strongly on the abundances of Fe2+ and S(IV) species (sulfite, bisulfite). In the low-absorption endmember scenario, freshwater lakes are essentially transparent in the UV, with depths of meters required for non-negligible attenuation of UV across most of the UV (Fig. 2). However, in the high-absorption endmember scenario, shortwave UV is shielded due to sulfite, with secondary shielding from nitrate and
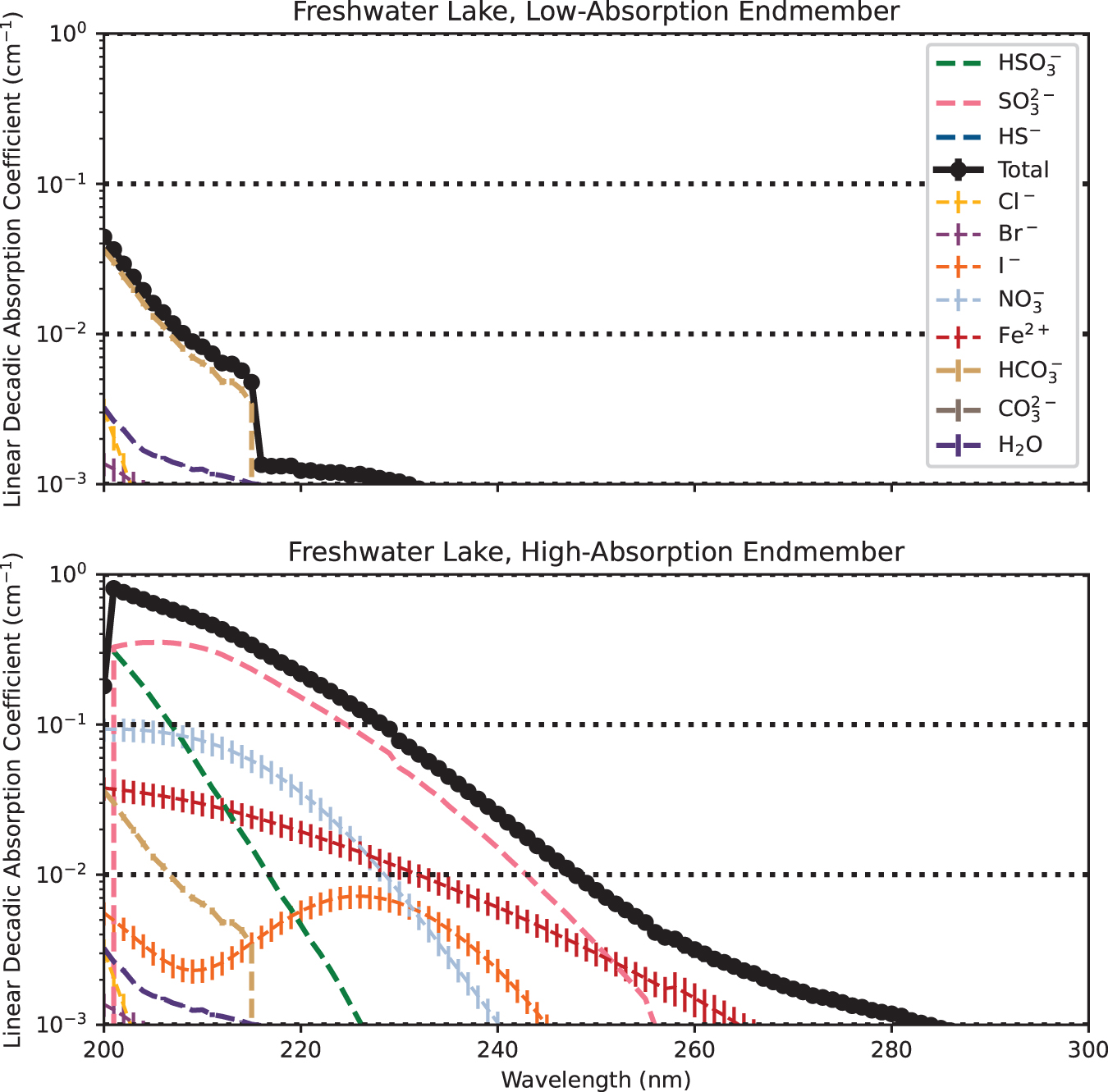
Simulated linear decadic absorption coefficients of the prebiotic freshwater lake and its component solutes, for low-absorption and high-absorption endmember cases. Not all solutes are visible in each case, because the linear decadic absorption coefficients of some solutes fall below the lower limit of the y-axis across the wavelength space plotted here. The freshwater lake may have been largely transparent to UV, and even in the high-absorption endmember would have been largely transparent to UV at the longer wavelengths (∼260 nm) relevant to nucleotide photolysis. UV, ultraviolet. Color images are available online.
4.2.2. Closed-basin carbonate lakes
Closed-basin carbonate lakes can be more UV-opaque compared with the prebiotic ocean (Fig. 3). Elevated [Br−] robustly limits absorption at short wavelengths; in even the low-absorption endmember scenario, ≤220 nm radiation is efficiently extinquished (i.e., is in the optically thick regime) for depths of >3.2 ± 0.6 cm. Radiation at 260 nm is available for depths of ≤1.04 ± 0.07 m even in the high-absorption endmember scenario, but it is depleted at depths of a few meters, due to nitrate absorption with contributions from sulfite.
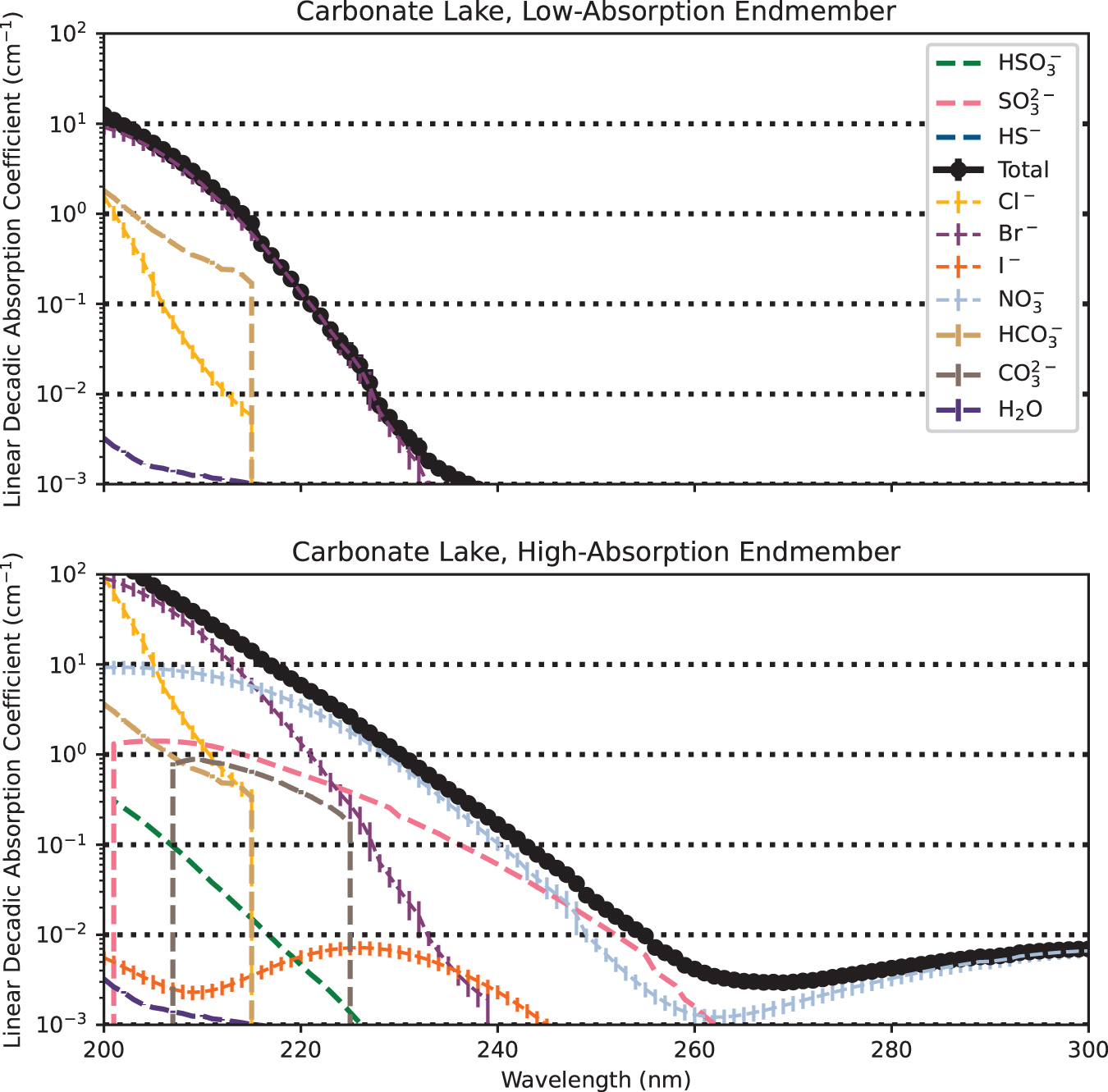
Simulated linear decadic absorption coefficients of the prebiotic carbonate lake and its component solutes, for low-absorption and high-absorption endmember scenario. Not all solutes are visible in each case, because the linear decadic absorption coefficients of some solutes fall below the lower limit of the y-axis across the wavelength space plotted here. The carbonate lake robustly extincts shortwave UV in even the low-absorption endmember scenario, driven by Br−. In the high-absorption endmember case, longer wavelength UV would have been available throughout shallow lakes (d < 1 m), but it would have been extinct at depths of a few meters. Color images are available online.
4.3. Ferrocyanide lakes
Ferrocyanide lakes are may have been low-UV environments (Fig. 4). Ferrocyanide is a stronger and crucially broader UV absorber than Fe2+ and can attenuate UV across the near-UV range relevant to prebiotic chemistry. Attenuation due to ferrocyanide is minimal in the low-absorption endmember scenario. However, for the high-absorption endmember, the ferrocyanide lake becomes optically thick by d = 11 ± 3 cm across 200–300 nm.
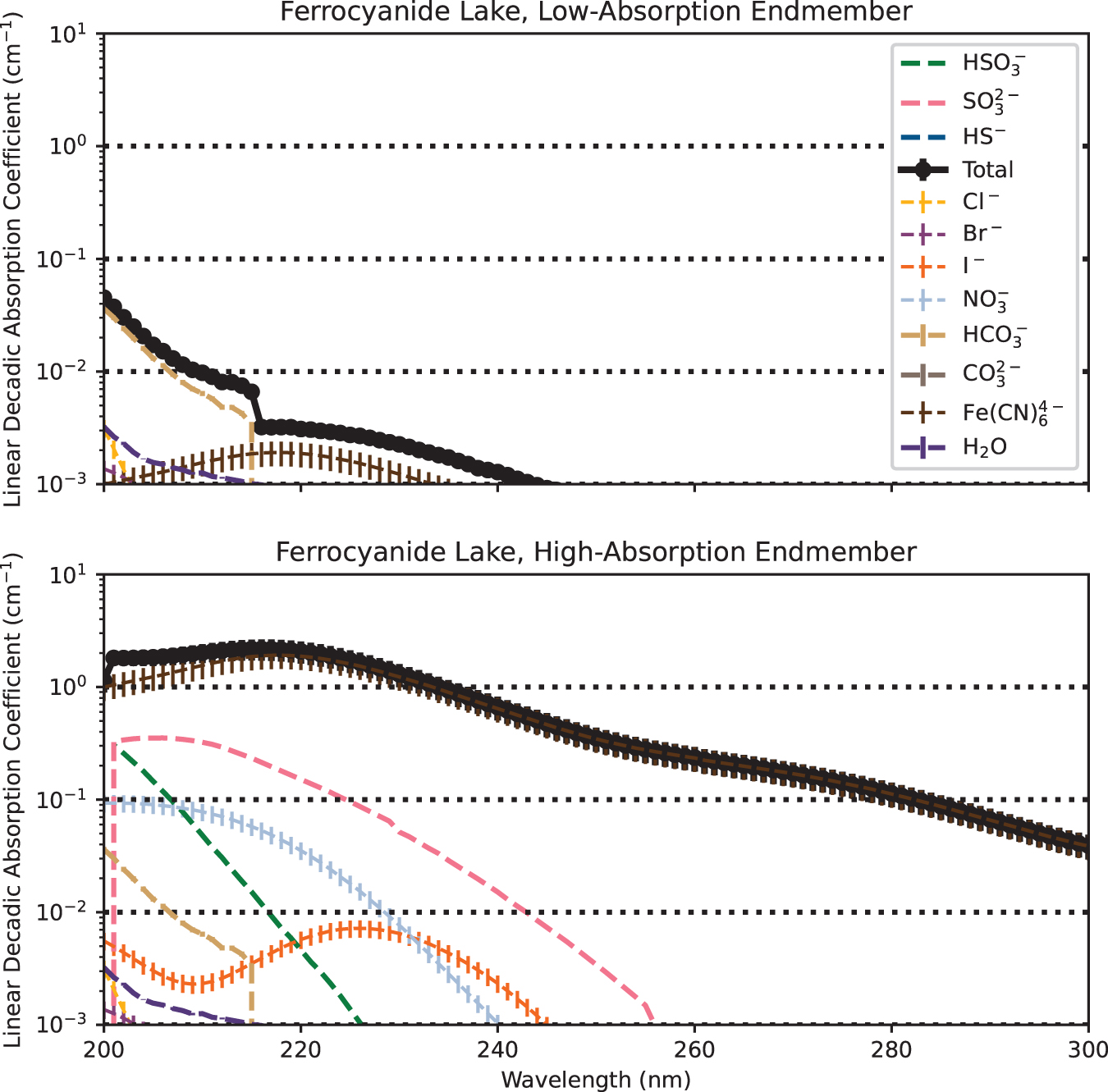
Simulated linear decadic absorption coefficients of the prebiotic ferrocyanide lake and its component solutes, for low-absorption and high-absorption endmember scenarios. Not all solutes are visible in each case, because the linear decadic absorption coefficients of some solutes fall below the lower limit of the y-axis across the wavelength space plotted here. Ferrocyanide is an effective sunscreen, and lakes hosting more than dilute ferrocyanide (
Photochemical derivatives of ferrocyanide, such as nitroprusside and ferricyanide, are similarly potent UV absorbers, which suggests that ferrocyanide-rich lakes will remain UV-poor even if some of the ferrocyanide is photochemically processed into these forms (Strizhakov et al., 2014; Xu et al., 2018; Mariani et al., 2018; Ross et al., 2018). Ferrocyanide and derived compounds may, therefore, have been strong “sunscreens” in select lacustrine environments on early Earth, if at elevated concentrations as postulated by some authors and invoked by others (Xu et al., 2018; Toner and Catling, 2019; Sasselov et al., 2020).
5. Discussion
Ranjan and Sasselov (2016) argued that UV radiation down to wavelengths of ∼204 nm would have been available for aqueous prebiotic chemistry on early Earth, motivated by the transmission of the atmosphere and pure water. However, prebiotic waters were likely not pure. In this article, we re-examine the conclusion of Ranjan and Sasselov (2016) by considering potential UV absorbers that might have been present in prebiotic waters.
5.1. Terrestrial freshwater systems may have been UV-transparent
Prebiotic terrestrial freshwaters may have been largely transparent in the UV. In the low-absorption endmember scenario for terrestrial freshwater lakes, shallow lakes could have been transparent down to depths of meters across most of the >200 nm wavelength range admitted by the prebiotic atmosphere, meaning that UV radiation may have been a pervasive aspect of the surficial prebiotic milieu.
Some investigators have assumed prebiotic pond waters to be UV-opaque based on extrapolation of modern natural waters; however, the opacity of modern terrestrial waters in the UV is generally due to biogenic dissolved organic compounds (e.g., Morris et al., 1995; Markager and Vincent, 2000; Laurion et al., 2000; Pearce et al., 2017). Indeed, even on modern Earth, waters with low biological productivity are transparent to a depth of ≥3 m down to the 300 nm threshold to which transmission has been characterized (Smith and Baker, 1981; Morel et al., 2007). Prebiotic fresh waters may have been similarly transparent, unless abiotic processes were comparably efficient as modern productive ecosystems in generating organics (e.g., potentially in the immediate aftermath of a large impact; Zahnle et al., 2020).
5.2. Shortwave UV was likely attenuated in diverse prebiotic waters
Although prebiotic natural waters in general were not necessarily opaque across the UV (200–300 nm), shortwave UV (≤220 nm) would have been attenuated in diverse prebiotic waters. Numerous prebiotic absorbers strongly attenuate shortwave UV, even if they absorb longwave UV only weakly. In particular, in high-salinity waters such as the prebiotic ocean and carbonate lakes, the halide anions, especially Br−, efficiently attenuate shortwave UV. In even the low-absorption endmembers, λ ≤ 220 nm radiation is restricted to depths d ≤ 7 ± 1 cm in the prebiotic ocean, and d ≤ 3.2 ± 0.6 cm in the carbonate lake scenario (Fig. 5).
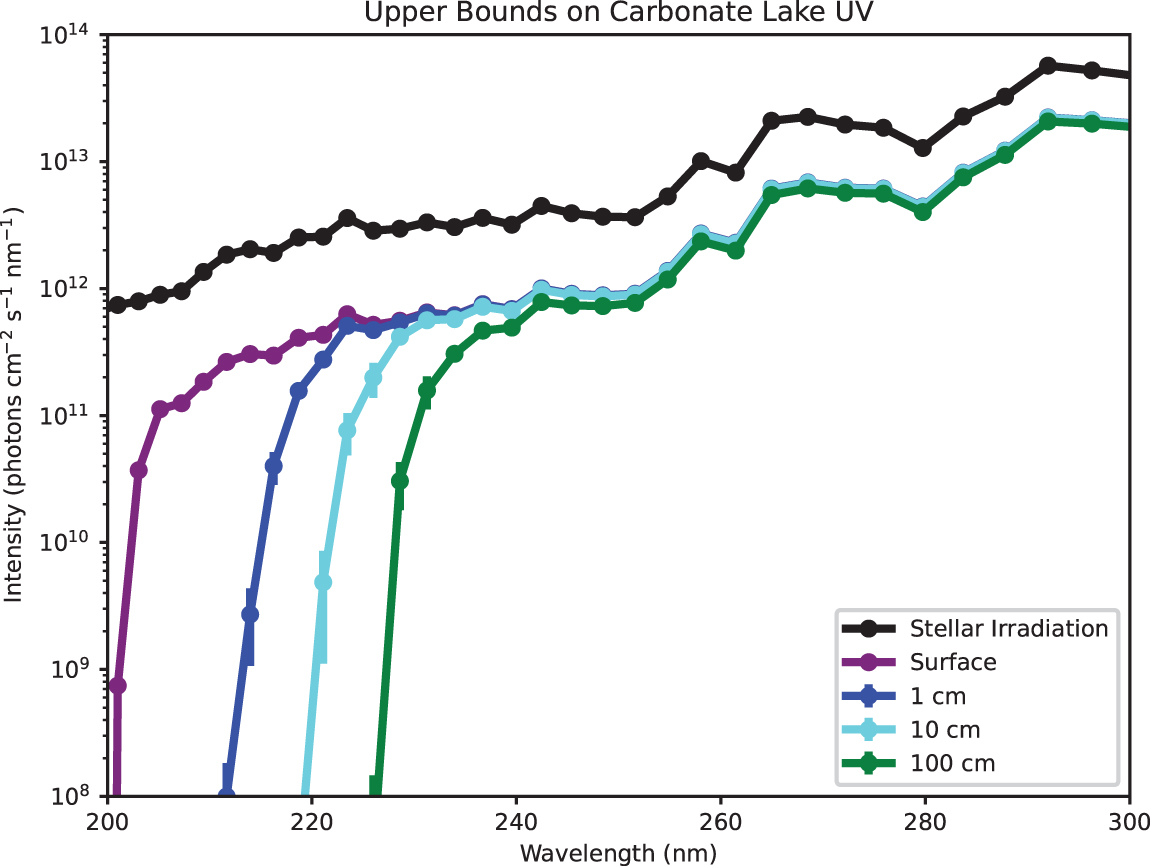
Estimate of actinic UV flux as a function of depth in the low-absorption endmember of the carbonate lake scenario, calculated by attenuating the surface actinic flux of Ranjan and Sasselov (2017) (their “surface radiance”) by Beer-Lambert law, assuming a slant angle of 60°, atmospheric composition from Rugheimer et al. (2015), and stellar irradiation of 3.9 Ga Sun from Claire et al. (2012). In even the low-absorption endmember, attenuation of <220 nm irradiation is significant. Color images are available online.
Shortwave UV may have been even more strongly attenuated in some waters. For example, in the high-absorption endmember for the carbonate lake scenario, nitrate absorption restricts λ ≤ 230 nm UV to d ≤ 0.43 ± 0.06 cm. Such nitrate concentrations would only have been available in shallow closed-basin lakes with large drainage ratios, and only if atmospheric
5.3. Broadband UV was attenuated in some prebiotic waters
While diverse prebiotically plausible absorbers are capable of attenuating shortwave UV, fewer prebiotic absorbers are capable of broadband UV attenuation, including the longer-wavelength UV photons that dominated the early Sun's UV output and hence prebiotic photoprocesses such as nucleobase photolysis. One prebiotic proposed family of broadband UV absorbers are compounds derived from ferrous iron. In particular, ferrocyanide is a strong, broad UV absorber. In the high-absorption endmember scenario for ferrocyanide lakes, ferrocyanide would have suppressed (optical depth >1) ≤260 nm radiation for d ≥ 1.8 ± 0.5 cm, and ≤300 nm radiation for d ≥ 11 ± 3 cm (Fig. 6). Photochemical derivatives of ferrocyanide, such as ferricyanide and nitroprusside, are similarly effective broadband UV screens (Appendix Fig. A12). Ferrocyanide lakes and their derivative waters, if extant, would have been low-UV environments at depth on early Earth.
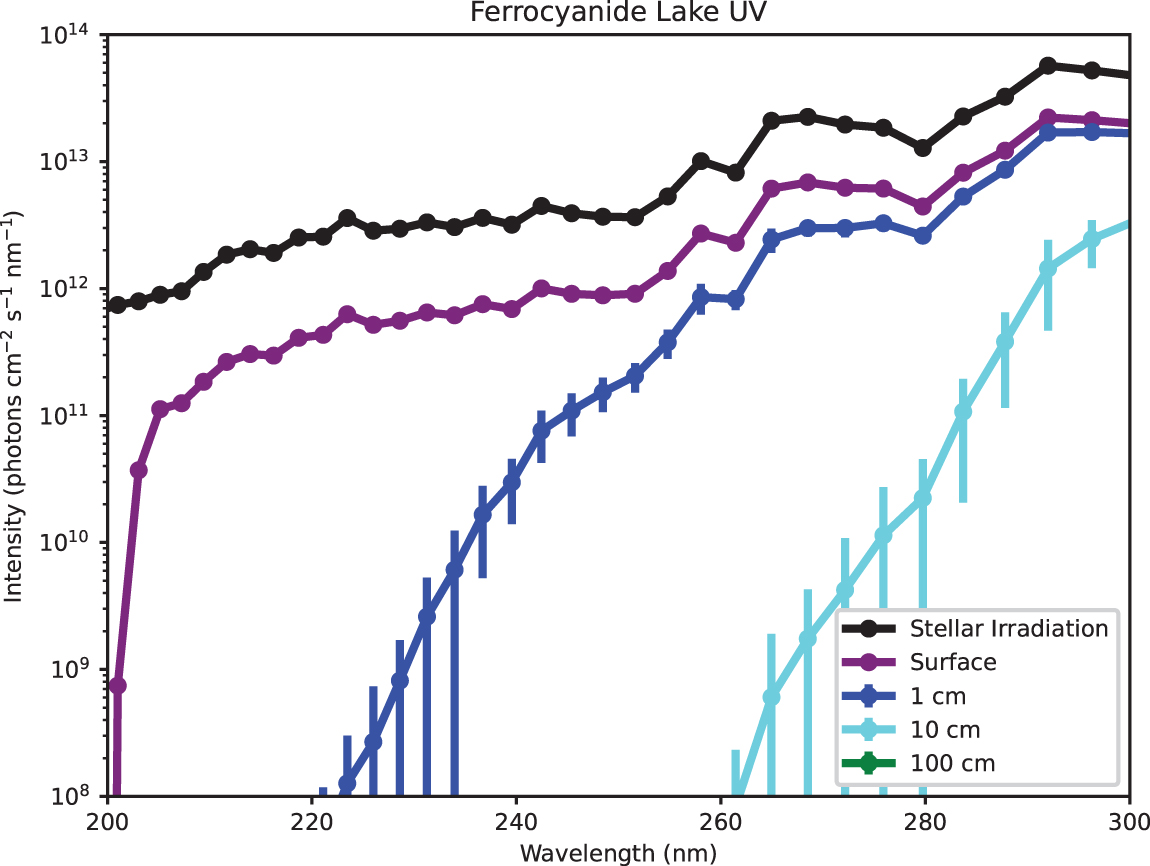
Estimate of actinic UV flux as a function of depth in the high-absorption endmember of the ferrocyanide lake scenario, calculated by attenuating the surface actinic flux of Ranjan and Sasselov (2017) (their “surface radiance”) by Beer-Lambert law, assuming a slant angle of 60°, atmospheric composition from Rugheimer et al. (2015), and stellar irradiation of 3.9 Ga Sun from Claire et al. (2012). In the high-absorption endmember, broadband UV is extincted at shallow depths. Color images are available online.
The existence and prevalence of ferrocyanide lakes on early Earth is uncertain. In particular, even if ferrocyanide lakes form as proposed in the work of Toner and Catling (2019), ferrocyanide undergoes photoaquation under irradiation by ≤400 nm radiation (Ašpergěr, 1952). Toner and Catling (2019) argued that rapid back-reaction stabilizes ferrocyanide against this photodecomposition, based on the experimental study of Ašpergěr (1952). However, the measurements of Ašpergěr (1952) were not conducted in prebiotically representative conditions. In particular, their UV irradiation was not representative of prebiotic UV irradiation, and the ferrocyanide concentrations used in their study were extremely high (≥50 mM). It is not clear whether ferrocyanide lakes would remain stable under more prebiotically representative conditions; detailed measurements and modeling of ferrocyanide photochemistry in prebiotically relevant conditions would be required. Alternately, the detection of remnants of ferrocyanide-rich environments on Mars (e.g., ferrocyanide salt deposits) might inform our understanding of the prevalence of such systems on early Earth (Sasselov et al., 2020; Mojarro et al., 2021).
In addition to ferrocyanide, other ferrous iron compounds such as FeSO4 and FeCl2 may also have acted as prebiotic “sunscreens,” due to their broadband UV absorption; however, much higher concentrations of these compounds would be required compared with ferrocyanide due to their much lower molar absorption in the UV (Appendix Fig. A12). In addition, their formation is not thermodynamically favored relative to Fe2+ under conditions relevant to natural waters on modern Earth (King, 1998), and they may not have been favored on early Earth either; modeling under prebiotic Earth conditions will be required to definitively rule on this question. Finally, the abundance of Fe2+ itself in natural waters on early Earth is still under debate (Konhauser et al., 2017; Halevy et al., 2017; Hao et al., 2017). Detailed, focused modeling of Fe2+ abundances and speciation in prebiotic waters on early Earth will also be required to estimate the prevalence of Fe2+-derived “sunscreens” such as ferrocyanide in natural waters on early Earth.
We do not recover the finding of Cleaves and Miller (1998) that Fe2+ in isolation would be an effective broadband sunscreen at concentrations that correspond to the prebiotic ocean. We report a molar absorbance for the Fe2+ ion (as Fe(BF4)2; Section A1.2.4) at 260 nm (15 ± 4 M
−1 cm−1) that is approximately consistent with the findings of Fontana et al. (2007) and Heinrich and Seward (1990) but two orders of magnitude lower than Cleaves and Miller (1998) (1630 M
−1 cm−1). The molar absorbances of FeCl2, FeSO4, and FeOH+ at 260 nm are also significantly below that reported by Cleaves and Miller (1998) (Anbar and Holland, 1992; Fontana et al., 2007). We speculate that one possible explanation might be the formation of Fe2+ complexes in the study of Cleaves and Miller (1998). At a slightly basic pH, characteristic of the modern ocean conditions simulated by Cleaves and Miller (1998), Fe2+ may have complexed with OH− and/or
5.4. Implications for prebiotic chemistry
Of the species considered in this study, we identify no ubiquitous absorber capable of attenuating broadband UV (200–300 nm) in prebiotic natural waters. Indeed, prebiotic freshwaters may have been essentially transparent in the UV. Prebiotic chemistries that are adversely affected by UV irradiation (UV-avoidant) must either be demonstrated to be so productive as to outpace UV degradation under prebiotic conditions or to invoke a UV-shielded prebiotic milieu (e.g., the organic-haze shielded aftermath of a large impact; Benner et al., 2019). UV light in the ∼240–300 nm wavelength range was not significantly attenuated by the geologically derived solutes considered in this study in most terrestrial prebiotic waters, meaning that low-pressure mercury lamps with primary emission at 254 nm remain reasonable proxies for UV irradiation in initial simulations of prebiotic chemistry. However, follow-up studies (e.g., characterization of action spectra) are required to verify whether pathways discovered with such sources could have functioned in realistic natural environments on prebiotic Earth (Ranjan and Sasselov, 2016; Todd et al., 2018; Rimmer et al., 2021).
Diverse prebiotic absorbers are capable of attenuating shortwave UV (≤220 nm). The general prebiotic importance of removal of shortwave UV (≤220 nm) should not be overstated, because most of the young Sun's UV flux was delivered at wavelengths >220 nm (Claire et al., 2012). In particular, shallow saline waters do not shield the canonical nucleobases, nucleosides, and nucleotides whose photolysis is expected to be dominated by the longwave (∼260 nm) bands (Voet et al., 1963; Todd et al., 2020). Similarly, the photolysis of 2-aminothiazole and the photoconversion of cytidine to uridine are not inhibited in saline waters (Todd et al., 2019, 2020). However, the photolyses of 2-aminooxazole and 2-aminoimidazole, intermediates with proposed roles in prebiotic nucleotide synthesis and activation (Powner et al., 2009; Patel et al., 2015; Li et al., 2017), are dominated by <230 nm radiation under early Earth conditions, and their photodestruction lifetimes are modestly enhanced in highly saline waters. Todd et al. (2019) estimated half-lives of these molecules to photodestruction to be ≈7, 26, and 99 h on early Earth; the half-lives of these molecules to direct photodestruction may be enhanced by up to a factor of a few in carbonate lakes by halide photoabsorption (Appendix A2). Saline systems have been disfavored as venues for prebiotic chemistry, on the argument that high salt concentrations generally inhibit lipid membrane formation (e.g., Deamer and Damer, 2017). However, experimental work suggests that some amphiphile mixtures can form stable vesicles at oceanic salinity or higher, and indeed that some families of amphiphiles require salinity for vesicle formation (Namani and Deamer, 2008; Maurer and Nguyen, 2016; Xu et al., 2017; Maurer, 2017). We therefore argue that it is premature to dismiss saline waters as venues for prebiotic chemistry. Indeed, they may be favorable environments for the accumulation of molecules whose concentrations on early Earth are limited by ≤220 nm photolysis.
Waters rich in ferrous iron compounds such as ferrocyanide were low-UV (UV-shielded) environments. Such waters may have been favorable environments for UV-avoidant prebiotic chemistries. For example, Pearce et al. (2017) proposed meteoritically delivered nucleobases in prebiotic ponds under the assumption that prebiotic pond water was UV-opaque and could shield meteoritic nucleobases from photolysis. In a 1 m-deep lake with a composition that corresponds to the high-absorption endmember scenario for ferrocyanide lakes, the lifetimes of the nucleobases to direct photolysis would be enhanced by approximately two orders of magnitude, which would make such waters potential candidates for the meteoritic delivery scenario (Appendix A2). By the same token, ferrous-rich lakes may be poor environments for UV-dependent prebiotic pathways. For example, the photoisomerization of diaminomaleonitrile (DAMN) to 5-aminoimidazole-4-carbonitrile (AICN), an intermediate for purine nucleobase synthesis, requires irradiation by ≲300 nm photons, which could be strongly attenuated in ferrocyanide-rich lakes (Ferris and Orgel, 1966; Cleaves, 2012; Boulanger et al., 2013; Yadav et al., 2020). Similarly, the UV-driven deamination of cytidine to form uridine, which is driven by 200–300 nm photons, could be inhibited in ferrocyanide-rich waters (Powner et al., 2009; Todd et al., 2020). Solvated electron production by UV photoirradiation of the ferrocyanide–sulfite system has been proposed to efficiently drive HCN homologation, but it is unknown whether ferrocyanide or sulfite (or potentially both) is the photoactive agent. If it is solely sulfite, it is unknown whether the chemistry can function with the dilute, optically thin ferrocyanide required for sulfite photolysis (Xu et al., 2018; Ritson et al., 2018; Green et al., 2021). We encourage further investigation into these questions to better understand the prebiotic potential for this chemistry.
In our discussion so far, we have focused on the implications of absorption of UV radiation by prebiotic solutes for direct photochemical processes such as excitation or photolysis of biomolecules. However, the UV energy absorbed by prebiotic absorbers must be dissipated, and although it may be dissipated thermally, it may also be dissipated through the formation of high-energy species that can participate in further chemistry. For example, the halides, sulfite/sulfide, and ferrocyanide generate solvated electrons (
5.5. Caveats and limitations
We have not considered FeOH+ as a prebiotic sunscreen. FeOH+ is a strong and broad UV absorber. Crucially, its absorption at the longer UV wavelengths where the Sun emits substantially more photons means that FeOH+ is projected to have played a key role in Fe2+ photooxidation and subsequent deposition (Braterman et al., 1983; Anbar and Holland, 1992; Tabata et al., 2021). Because of its ability to absorb longer-wavelength UV (300–450 nm), the total photoabsorption of FeOH+ is comparable to that of Fe2+ at circumneutral pH, despite much lower concentrations (Nie et al., 2017). However, its absorption at longer wavelengths does not aid its potential as a sunscreen at shorter wavelengths (200–300 nm). At these wavelengths, the potential of FeOH+ as a sunscreen is limited due to its low solubility at basic pH due to Fe(OH)2 precipitation. This low solubility means that [FeOH+] is low, implying that large depths are required for it to significantly attenuate UV (i.e., bring the optical depth to unity). The solubility product of Fe(OH)2 is K
sp = 4.9 × 10−17, and Keq
= 3.02 × 10−10 for the reaction
In the present study, we considered only a few of the vast diversity of natural waters. Other natural waters may have different absorption properties. For example, hot springs host high concentrations of HS− and are driven by continuous supply of H2S from below (Kaasalainen and Stef'ansson, 2011). HS− is a strong and broad absorber and, similar to the ferrous iron compounds, attenuates the longer-wavelength UV radiation that dominates solar UV output; some hot springs could, therefore, have been UV-opaque. Similarly, I− is a strong and broad UV absorber; in waters rich in iodine, for example, mineral springs, I− may provide significant UV attenuation (Fuge and Johnson, 1986).
We reiterate that the estimates of UV transmittance presented here are upper bounds. The longwave absorption of
In this work, we have constructed the bracketing low- and high-absorptivity compositional endmembers for various prebiotic waters based on literature estimates. We have not attempted self-consistent geochemical modeling of these waters to estimate their composition, in part because the relevant kinetics have not been characterized under prebiotically relevant conditions (e.g., Ranjan et al., 2018, 2019). Similarly, our work treats UV absorbers as static and neglects further photochemical transformations triggered by their absorption of solar UV. Our work is, therefore, a first approximation and subject to revision by future models that are capable of investigating the self-consistent photochemistry and geochemistry of prebiotic natural waters.
6. Conclusions
Prebiotic freshwaters may have been essentially transparent in the UV; UV-avoidant surficial prebiotic chemistries must invoke a UV “sunscreen” agent. Shortwave (≤220) UV may have been attenuated in diverse prebiotic waters, with the most important absorbers being Br− in saline waters and potentially sulfite in shallow lakes and nitrate in shallow closed-basin lakes. Better constraints on prebiotic
The generally widespread availability of ∼240–300 nm radiation means that low-pressure mercury lamps remain suitable for initial studies of prebiotic chemistry, though more realistic irradiation is required to verify the plausibility of pathways discovered under mercury lamp irradiation.
Some natural waters may have been largely opaque in the UV. In particular, Fe2+-derived compounds are effective broadband “sunscreens” if present at high concentrations. Ferrocyanide is an especially potent UV absorber, and ferrocyanide lake waters would have been low-UV environments that are candidate waters for UV-avoidant origin-of-life scenarios such as the meteoritic delivery hypothesis. On the other hand, such waters may be poor environments for UV-dependent reactions such as the photodeamination of cytidine to uridine. In addition to ferrocyanide, other ferrous iron compounds such as FeSO4 may serve as sunscreens in shallow lakes if present at proportionately higher concentrations to compensate for their lower molar absorptivities relative to ferrocyanide and to thermodynamically favor their complexation. Fe2+ has been suggested to be widespread on early Earth, but the abundance and speciation of Fe2+ compounds on early Earth remain uncertain; we highlight detailed modeling and/or geochemical constraints on ferrous iron concentrations and speciation in prebiotic natural (especially terrestrial) waters as a priority for UV-sensitive prebiotic chemistry.
UV light can trigger photochemistry even if attenuated by an absorber, through photochemical transformations of that absorber. In particular, irradiation of a wide range of prebiotic absorbers, such as the halides, sulfite/sulfide, and ferrocyanide, is predicted to generate
Footnotes
Author Contributions
S.R. conceived and led the study; G.G.L and C.L.K. performed the measurements; C.L.K. synthesized data into molar decadic absorptivities and estimated errors; S.R. and A.H. performed modeling and literature data extraction for comparison; and S.R., D.D.S., and Z.R.T. explored prebiotic implications.
Acknowledgments
The authors are grateful to J. Cleaves, B. Pearce, J. Toner, J. Birkmann, Y. Beyad, J. Sutherland, J. Krissansen-Totton, S. Kadoya, L. Barge, and P. Rimmer for answers to questions and/or for discussions related to this article. The authors further thank P. Rimmer and N. Green for comments on a draft of this article, S. Kadoya for sharing the raw model outputs from Kadoya et al. (2020), and Y. Beyad for sharing the sulfite and bisulfite absorption spectra from Beyad et al. (![]() ).
).
The authors thank two anonymous referees whose feedback substantially improved this article. The authors thank the Simons Collaboration on the Origin of Life and the Harvard Origins of Life Initiative for nurturing many fruitful conversations related to this article.
The raw data derived from the experiments, processed data reported in this article, and scripts used to make the plots reported in this article can be accessed on GitHub via
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This work was supported in part by grants from the Simons Foundation (495062, Ranjan) (3290360, Sasselov) via SCOL. Z.R.T. acknowledges support from the NASA Hubble Fellowship Program, award# HST:HF2-51471.
Abbreviations Used
Appendix A1. Molar Decadic Absorption Coefficients
Associate Editor: Christopher McKay
