Abstract
Target lithologies and post-impact hydrothermal mineral assemblages in a new 1.3 km deep core from the peak ring of the Chicxulub impact crater indicate sulfate reduction was a potential energy source for a microbial ecosystem (Kring et al., 2020). That sulfate was metabolized is confirmed here by microscopic pyrite framboids with δ34S values of -5 to -35 ‰ and ΔSsulfate-sulfide values between pyrite and source sulfate of 25 to 54 ‰, which are indicative of biologic fractionation rather than inorganic fractionation processes. These data indicate the Chicxulub impact crater and its hydrothermal system hosted a subsurface microbial community in porous permeable niches within the crater's peak ring.
1. Introduction
The ∼180
Such systems would have been particularly prevalent during the Hadean when Earth was being bombarded by late accreting asteroids and comets. Some of the largest of those impacts should have vaporized seas (Sleep et al., 1989), making conditions untenable for life at the surface. Based on observations at Chicxulub (Kring and Boynton, 1992), the idea emerged that those same impact events produced vast subsurface hydrothermal systems that were potential crucibles for prebiotic chemistry and habitats for the early evolution of life (Kring 2000a, 2000b, 2003), that is, the impact origin of life hypothesis. The end of that period of impact bombardment coincided with what may be the earliest evidence of life (e.g., Mojzsis and Harrison, 2000; cf. Whitehouse and Fedo, 2007), although it was not certain (and remains uncertain) whether life truly emerged at that time or was of a type capable of surviving the bombardment (Maher and Stevenson, 1988; Chyba, 1993). Analyses of ribosomal RNA made in the same decade indicated the earliest organisms on Earth were thermophilic (Woese et al., 1990; Pace 1991, 1997). It seemed plausible that life originated in an impact crater (Kring, 2000a, 2000b, 2003).
To test that concept, studies of Chicxulub, our best proxy of large Hadean impact craters, continued. Chicxulub is the only large peak-ring basin that is still intact and provides an opportunity to study the remnants of an impact-generated hydrothermal system, from depth up to and including the venting surface environment, similar to those that may have existed earlier in Earth's history. Thermal evolution models of that and other hydrothermal systems indicated they were long-lived and produced significant volumes of porous, permeable rock suitable for thermophilic organisms (Abramov and Kring, 2004, 2005, 2007). The International Continental Scientific Drilling Program (ICDP) drilled into Chicxulub in 2001–2002, recovering core in the moat between the crater rim and peak ring. Traces of a hydrothermal system were found in ∼100 m of impactites (Ames et al., 2004; Hecht et al., 2004; Kring et al., 2004; Lüders and Rickers, 2004; Rowe et al., 2004; Zürcher and Kring, 2004; Zurcher et al., 2005; Nelson et al., 2012), augmenting the findings from the borehole that provided evidence of the structure's impact origin (Kring and Boynton, 1992) and producing an evolutionary sequence of the hydrothermal system with time and depth in that portion of the crater (Zürcher and Kring, 2004). Those two boreholes, Yucatán-6 (Y-6) and Yaxcopoil-1 (Yax-1), are located ∼10 and ∼25 km beyond the peak ring (Fig. 1), respectively, indicating hydrothermal activity was not a local phenomenon but rather part of an extensive system. Furthermore, it became increasingly clear (Cockell, 2006) that that type of system had the potential to host life.
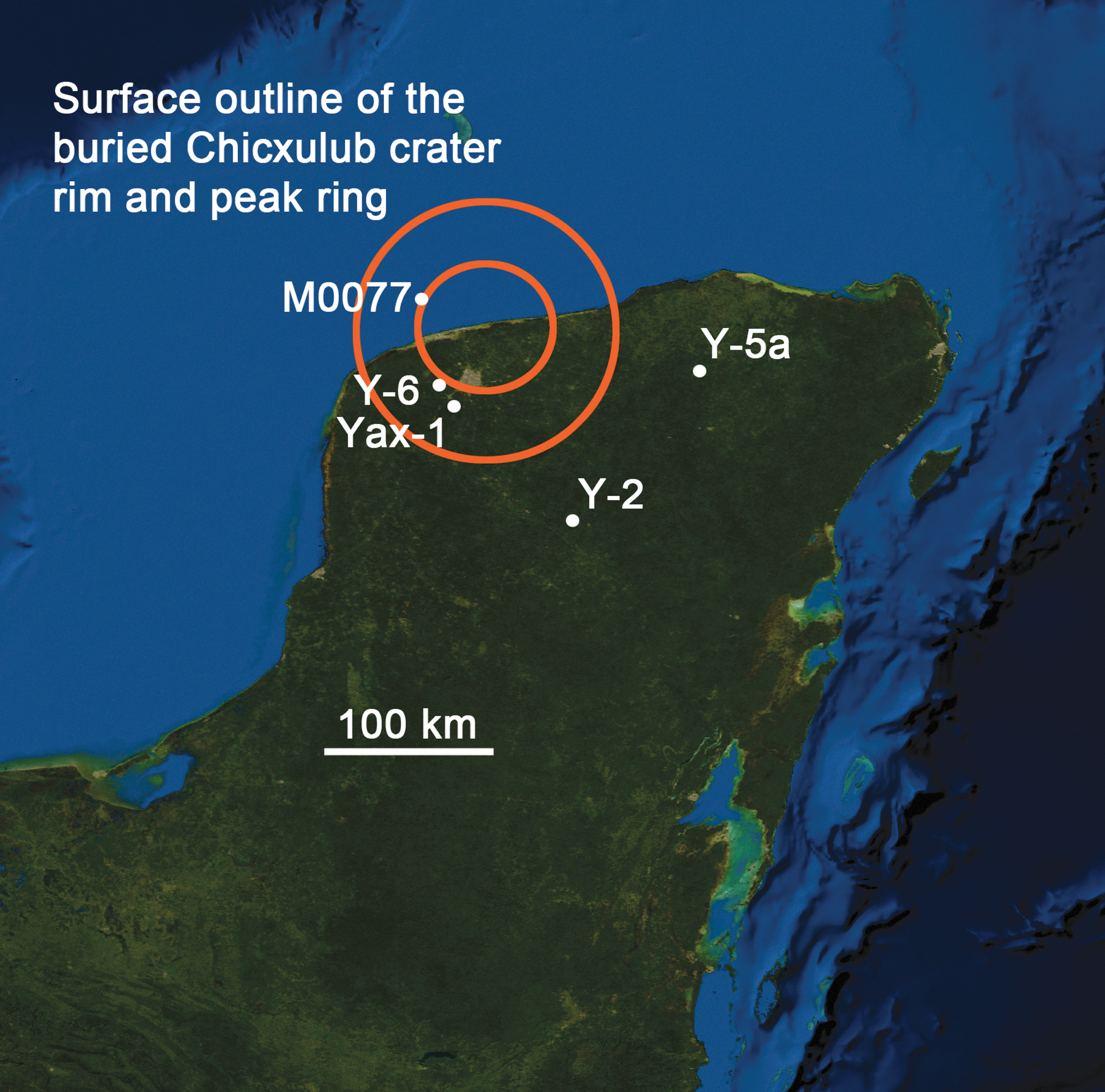
Location of the Chicxulub peak-ring crater beneath the northern edge of the Yucatán Peninsula, México. Samples analyzed in the current study were recovered from the International Ocean Discovery Program and International Continental Scientific Drilling Program Expedition 364 Site M0077 borehole. Other samples described in the paper were recovered from boreholes Yucatán-2 (Y-2), Yucatán-5a (Y-5a), Yucatán-6 (Y-6), and Yaxcopoil-1 (Yax-1). Background: NASA image produced by MODIS satellite observations in October 2004.
To explore the hydrothermal system further, in 2016 the International Ocean Discovery Program (IODP) and ICDP sponsored Expedition 364, drilling a borehole into the peak ring of the Chicxulub crater where model results (Abramov and Kring, 2007) suggested hydrothermal activity may have been extensive. The expedition's petrologic analyses of the recovered core verified hydrothermal activity occurred with initially high temperatures in excess of 300°C (Kring et al., 2017a, 2020). As the system cooled, high-temperature hydrothermal assemblages were crosscut by veins of, and pervasively overprinted by, lower-temperature mineral assemblages that precipitated as temperatures decreased from ∼250°C to an ambient post-impact temperature of ∼25°C in the upper peak ring breccias (Abramov and Kring, 2007), before the peak ring was buried to its current depth of ∼617 m below the seafloor (Morgan et al., 2016). One of the lower-temperature mineral assemblages is composed of analcime and dachiardite-Na (hereafter dachiardite), sometimes with heulandite and calcite, and FeS2 framboids.
An important goal of Expedition 364 was to assess the biological potential of that hydrothermal system. Petrologic examination of the core (Kring et al., 2020; Simpson et al., 2020) indicated there were sharp redox gradients that may have been suitable for microbial reduction and oxidation processes. A potential by-product of microbial activity is sulfide, such as FeS2 framboids (e.g., Kohn et al., 1998; Popa et al., 2004; MacLean et al., 2008; cf. Wilkin and Barnes, 1997). Here we examine the sulfur isotopic signature of sulfide minerals that exist in lower-temperature and, thus, biologically compatible mineral assemblages, to determine whether sulfate reduction was a biologically mitigated process (e.g., as in Canfield and Thamdrup, 1994; Kohn et al., 1998; Popa et al., 2004; Shen and Buick, 2004; Sim et al., 2011).
2. Methods
2.1. Petrography
Rock textures, crosscutting relationships, and initial mineral identification were made with a petrographic microscope at the Lunar and Planetary Institute. Electron microprobe analyses of phase chemical compositions and additional Raman spectroscopy were made at the Johnson Space Center. The procedures are the same as those used for other core samples (Kring et al., 2020, where they are described in detail).
2.2. Measuring δ34S and δ13C
Microscale sulfur and carbon stable isotope measurements were performed with a CAMECA IMS1280 large-geometry ion microprobe at the Swedish Museum of Natural History (NordSIMS facility). Operation of the instrument in multi-collector mode, with the secondary magnet field regulated to high stability by using an NMR field sensor, was common to both methods. Instrument settings for δ34S in pyrite broadly follow those described in the work of Whitehouse (2013). A primary beam of 133Cs+ ions with 20 kV incident energy and beam current of either ∼500 pA for the framboidal samples or ∼1 nA for the massive pyrites was critically (Gaussian) focused with, respectively, a 5 or 10 μm raster applied during analysis to homogenize the beam profile. Secondary ion signals of 32S and 34S were measured by using two Faraday cups operating at a common mass resolution (M/ΔM) of 2460. Analyses of δ13C in calcite used similar primary beam tuning, but with a slightly larger beam current (2.5 nA) and a 10 μm raster. Simultaneous detection of 12C and 13C signals was made in a Faraday cup at M/ΔM of 2460 and an ion counting electron multiplier at M/ΔM of 4000, sufficient to resolve 13C from 12C1H. For both methods, a normal incidence electron gun was used for charge compensation. All presputter, beam centering, and data acquisition steps were automated in the run definition. Sample measurements were bracketed by reference pyrite (S0302A, δ34SV-CDT = 0.0 ‰) and calcite (S0161; δ13CV-PDB = -2.0 ‰) measurements to correct for instrumental mass bias (both reference materials kindly provided by R. Stern, University of Alberta; V-CDT and V-PDB refer to the reference values for Vienna Canyon Diablo Troilite and Vienna Pee Dee Belemnite, respectively). Within-run uncertainties were propagated together with the standard deviation obtained from the relevant reference materials during the analytical session to yield the overall reported uncertainty (Table 1).
Isotope Data for Sulfide and Coexisting Calcite Crystals
Calcite did not coexist with pyrite in 63-2-69.5-72. A table with standard values and other analytical values is in a permanently curated data repository (
3. Results
Samples of the Chicxulub peak ring were recovered from 617.33 meters below the seafloor (mbsf) to 1334.68 mbsf at Site M0077 (Morgan et al., 2016, 2017; Kring et al., 2017b). The uppermost part of the peak ring is composed of ∼104 m of breccia with impact melt fragments (reworked suevite, Unit 2 of the logged core; Morgan et al., 2017). That material overlies ∼25 m of clast-poor and clast-rich impact melt rock (Unit 3). Those impactites cover felsic basement rocks. For the current study, three samples were selected from the porous, permeable breccia sequence, and a fourth sample was selected from the bottom of the core. The deepest sample (0077-297R-1, 93 to 95 cm; 1313.92 mbsf) is a polymict breccia with solidified impact melt fragments (suevite) that occurs within granitoid rocks of the peak-ring, 697 m below the contact between the upper peak-ring lithologies and an overlying siltstone. The interval is hydrothermally altered with secondary garnet (andradite), epidote, pyrite, chalcopyrite, galena, calcite, and clay minerals. This suevite unit does not contain sedimentary clasts, which are evident in upper peak ring suevites (Kring et al., 2017c). The sulfide analyzed here has a rhombohedral habit and occurs along the margin of secondary sparitic calcite (Fig. 2).

Images of sulfide. Regions analyzed for sulfur isotopes in (
The other three samples are from the upper portion of the peak-ring sequence. The deepest of those (0077-63R-2, 69.5 to 72 cm; 685.47 mbsf) is from Subunit 2B, a relatively well-sorted, suevite that occurs 68 m beneath the top of the peak-ring lithologies. The breccia contains fragments of the crystalline basement and overlying carbonate platform sedimentary rocks. The breccia is hydrothermally altered with secondary silica, pyrite, chalcopyrite, other Fe-Co-Ni sulfides, barite, clay minerals, calcite, dachiardite, and analcime. The sample contains both irregular-shaped FeS2 and framboidal FeS2 (Fig. 2), the latter along an open network of channels lined with dachiardite and other zeolites.
Two samples are from Subunit 2A. One of the samples (0077-46R-1, 46 to 52 cm; 635 mbsf) is a suevite that occurs 29.52 m above the bottom of Subunit 2A, which is separated from 2B by an erosional contact. Subunit 2A has a similar matrix and clast content as 2B, but is less coarse than the underlying suevite 2B. The sample interval is hydrothermally altered with secondary silica, pyrite, chalcopyrite, calcite, and clay minerals. Pyrite occurs as framboids concentrated along the walls of a subvertical vent channel (Fig. 2). The sample was ∼18 m beneath the top of the peak-ring lithologies. The other sample from Subunit 2A (0077-40R-2, 105 to 107 cm; 618.72 mbsf) is a relatively well-sorted suevite near the top of Subunit 2A and 1.39 m beneath the top of the peak-ring lithologies. The interval is hydrothermally altered with secondary pyrite, chalcopyrite, calcite, and clay minerals (Kring et al., 2020). The sulfide minerals are isolated and aggregate rhombohedral crystals (Fig. 2). Local cockscomb-textured aggregates suggest those Fe-sulfides may include marcasite (Schmieder et al., 2017a).
The suevite samples contain several sulfide phases: chalcopyrite (CuFeS2), galena (PbS), pentlandite ((Fe,Ni)9S8), pyrite (FeS2), sphalerite (ZnFeS), and villamaninite ((Cu,Ni,Co,Fe)S2) (Kring et al., 2020). Here we focus on those crystals with FeS2 compositions, because they may be a by-product of biogenic processes (e.g., Popa et al., 2004). We refer to the crystals with FeS2 compositions in samples 0077-46R-1 and 0077-63R-2 as pyrite rather than its polymorph marcasite, based on the framboid morphology. Moreover, Raman spectra confirm FeS2 is pyrite in all four samples, including cockscomb-textured crystal aggregates.
The coarse-grained rhombohedral pyrite in 0077-40R-2 and 0077-297R-1 coexists with calcite (CaCO3). Finer-grained pyrite framboids in 0077-46R-1 and 0077-63R-2 coexist with the zeolite minerals analcime (NaAlSi2O6 • H2O), dachiardite (Na10Al10Si38O96 • 25H2O) (Fig. 2), and heulandite ((Na,Ca)2-3Al3(Al,Si)2Si13O36 • 12H2O). Analcime grew as blocky, orthorhombic, equidimensional, trapezohedron crystals and occasionally as sprays of thinner crystals, while dachiardite grew as thin crystal sprays that formed botryoidal masses. The habits of the zeolite crystals indicate they grew into pore spaces within the suevite. They also fill pore spaces in large multi-centimeter-wide swaths through the rock (see Fig. 2 in Kring et al., 2020). Pyrite framboids are enclosed within analcime and dachiardite, indicating that the framboids grew with the zeolites and that the zeolites continued to grow after the framboids precipitated.
Millimeter-sized rhombohedral pyrite crystals at the bottom and top of the core have δ34S values of -1.86 ± 0.05 to -3.10 ± 0.05 ‰ and -9.21 ± 0.05 to -17.23 ± 0.05 ‰, respectively (Fig. 3). Coexisting calcite has δ13C values of 0.30 ± 0.30 to -7.61 ± 0.29 ‰. The 10–20 μm diameter pyrite framboids have very different sulfur isotope values. Framboids ∼68 m beneath the top of the peak ring (sample 0077-63R-2, 69.5 to 72 cm; 685.47 mbsf) have δ34S values of -5 to -22.5 ‰. Framboids ∼18 m beneath the top of the peak ring (sample 0077-46R-1, 46 to 52 cm; 635 mbsf) have δ34S values of -20 to -36 ‰ (Fig. 3). Co-existing calcite has δ13C values of 0.93 ± 0.33 to -7.59 ± 0.30 ‰.
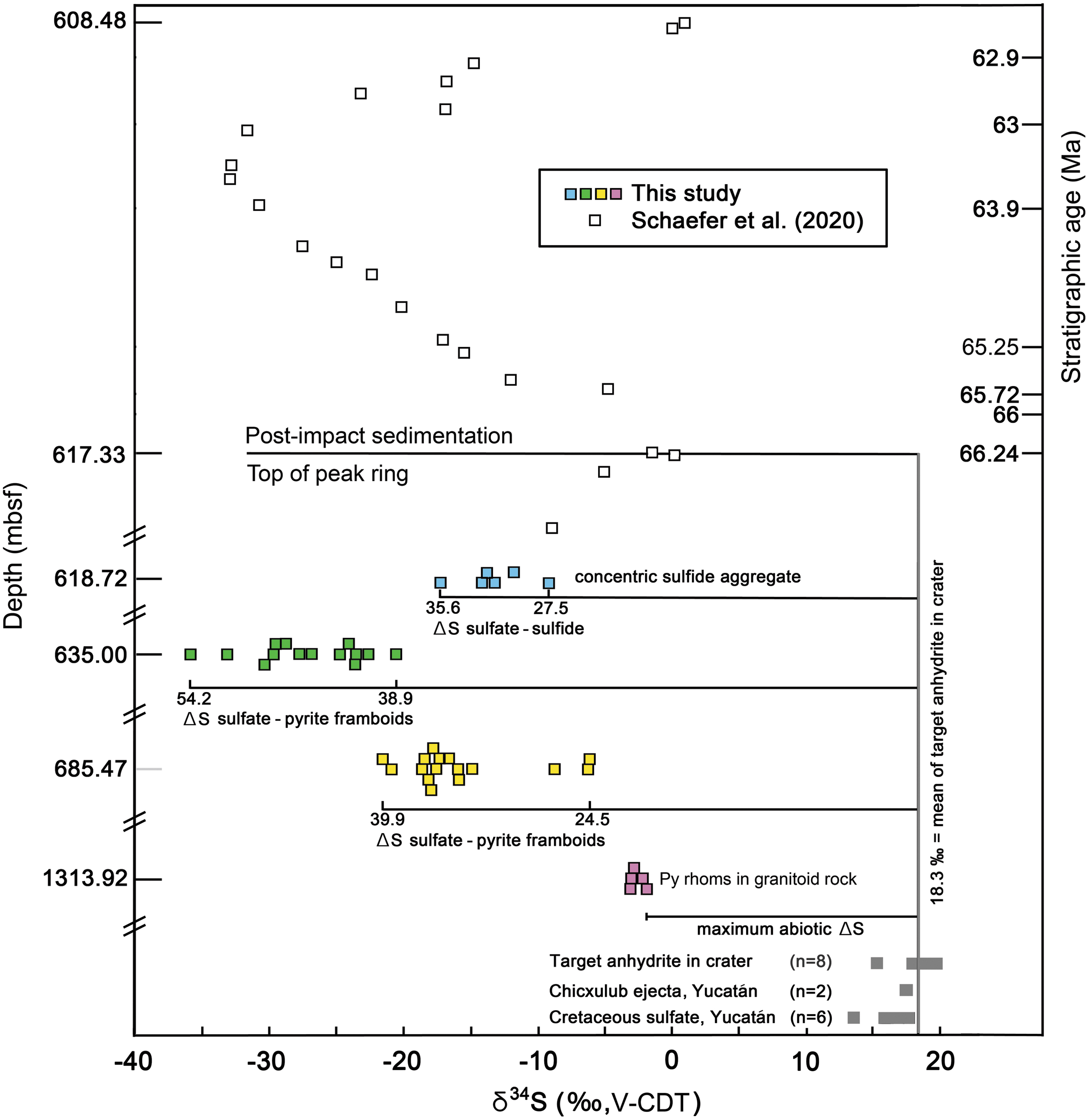
Sulfur isotope compositions for sulfide from four hydrothermally altered core samples recovered by IODP-ICDP Expedition 364 in order of relative depth in the peak ring. Forty-one sulfide analyses were made in suevite within peak ring granitoid rocks (0077-297R-1-93-95, pink squares) and three overlying suevitic breccias (0077-63R-2-69.5-72, yellow squares; 0077-46R-1-46-52, green squares; 0077-40R-2-105-107, blue squares). Analytical uncertainty is smaller than the sizes of symbols. Expedition 364 sulfur isotopes in 22 samples of post-impact sediments (open squares) and their stratigraphic age are from Schaefer et al. (2020). For comparison, analyses of sulfate samples in bedrock of the northern Yucatán (Claypool et al., 1980; Koeberl 1993), in Chicxulub ejecta on the Yucatán (Koeberl 1993), and in two previous core samples (Y-6 and Yax-1) recovered from the crater (Strauss and Deutsch, 2003) are provided (gray squares). Sulfide isotope compositions (‰, V-CDT) of the pyrite framboids are extremely fractionated from those of target sulfate compositions.
4. Discussion
The ∼180 km diameter Chicxulub impact crater was produced by an impacting asteroid with ∼100 million megatons of kinetic energy, which is ∼6 million times more energetic than the largest nuclear explosion test conducted by the United States (e.g., Kring, 1993). The impact event uplifted relatively warm crustal rocks from a depth of 8–10 km (Morgan et al., 2016) and produced 104 to 105 km3 of impact melt (Kring, 1995), heating subsurface water and producing a crater-wide hydrothermal system. Soon after the crater was excavated, it was flooded by the sea, covering the peak ring in the location of the Expedition 364 borehole by as much as 600 m of water (Gulick et al., 2019). The pressure at the top of any venting hydrothermal system at that depth was ∼6000 kPa (880 psi or 60 atm), assuming a seawater density of 1.03 g/cm3. The system would have been biologically sterile during the high-temperature phase, but as temperatures approached 100°C, porous, permeable niches in the impactites had the potential to host microbial life. Conditions (∼50°C to 120°C) suitable for thermophilic life may have persisted for ≥2 million years (Kring et al., 2020), initially in the outer regions of the crater, far from the central melt sheet, and then in areas closer to the crater center as the melt sheet cooled (see Fig. 3 of Abramov and Kring, 2007, for an evolutionary sequence with 25°C to 100°C temperature contours in time steps at 4 thousand, 20 thousand, 200 thousand, and 2 million years after impact).
Water feeding the system was dominated by groundwater (Zurcher et al., 2005; Abramov and Kring, 2007) flowing through a carbonate platform sequence with sulfate strata and through impact breccias that contained fragments of sulfate rocks. Those waters, entering the hydrothermal system, were circulated upward carrying dissolved sulfate and partially vented at the seafloor-seawater interface. Although the bulk of the water was a basinal brine (Zurcher et al., 2005), some drawdown of sulfate-bearing seawater was possible (Abramov and Kring, 2007), producing mixing zones.
Sulfate (anhydrite) clasts (Fig. 4) were observed in the Y-6 polymict impact breccias (Hildebrand et al., 1991; Kring et al., 1991; Kring and Boynton, 1992; Sharpton et al., 1996; Claeys et al., 2003), ∼65 km SSE of Site M0077. Anhydrite was also found in bedrock beneath the Yax-1 impactites, although no clasts were observed in the impact breccias (e.g., Kring et al., 2004), ∼80 km SSE of Site M0077. It is not yet clear if sulfate was heterogeneously distributed in the target rock sequence or if the variation observed in impactites is a product of impact excavation, deposition, and modification processes (Kring, 2005). That sulfate was incorporated into hydrothermal fluids is explicitly shown by anhydrite (CaSO4) veins in Y-6 core samples (Kring and Boynton, 1992; Schmieder et al. 2019), anhydrite veins in Yax-1 core samples (Zurcher et al., 2005), secondary anhydrite in Y-6 samples (Kring and Boynton, 1992), and secondary barite (BaSO4) in an M0077 core sample with pyrite framboids (0077-63R-2) and in samples ∼3, 12, and 32 m deeper in the core (Kring et al., 2020). Sulfate was initially incorporated into hydrothermal fluids at temperatures ≥270°C based on Ti-in-quartz geothermometry of quartz–Ti-magnetite–anhydrite veins (Schmieder et al., 2019), whereas anhydrite also precipitated later at lower temperatures in voids within the breccia (Fig. 4; see also Claeys et al., 2003), particularly within cavities of once-glassy melt fragments that were altered to smectite.

Images of sulfate (anhydrite). (
Bedrock sulfate in northern Yucatán has δ34S values of 13.7–17.7 ‰ (Claypool et al., 1980; Koeberl, 1993). Sulfate clasts in Chicxulub ejecta deposited on the Yucatán peninsula, recovered in boreholes Yucatán-2 and Yucatán-5a, have δ34S values of 17.5 and 17.6 ‰ (Koeberl, 1993). Sulfate clasts recovered in two previous boreholes into the crater (Y-6 and Yax-1) have δ34S values of 18.0–19.8 ‰ (Strauss and Deutsch, 2003). The mean of samples from within the crater is 18.3 ‰, representing the groundwater reservoir and overlying seawater values. The maximum abiogenic ΔSsulfate-sulfide is ∼20 ‰ (Machel, 2001; Popa et al., 2004), which is about that seen in rhombohedral pyrite at the base of the borehole (Fig. 3). An abiogenic origin for the rhombohedral pyrite is also consistent with the absence of an unambiguous biogenic carbon isotope signature in the associated calcite.
In contrast, the δ34S values of the pyrite framboids in the suevite are much lower, producing ΔSsulfate-sulfide of ∼25 to 54 ‰, greatly exceeding abiogenic fractionation. Such large ΔSsulfate-sulfide values are signatures of microbial reduction of sulfate (e.g., Popa et al., 2004). Similar sulfate-sulfide fractionation was observed in the smaller, ∼24 km diameter Haughton crater (Canada) where sulfate also occurs in the target bedrock (Parnell et al., 2010) and ∼40 km diameter Rochechouart crater (France) where sulfate is inferred to have been extracted from seawater (Simpson et al., 2017). We also note that pyrite framboids, albeit without sulfur isotope data, were observed in hydrothermally-altered rocks of the ∼52 km diameter Siljan crater (Sweden) in association with carbonaceous matter with biogenic-like and microbial-like features (Hode et al., 2009).
While samples 0077-46R-1 and 0077-63R-2 were initially 18 and 68 m beneath the seafloor, respectively, by the time the hydrothermal system cooled to temperatures suitable for thermophilic microbes, 1–10 m of marine sediment blanketed the peak ring, depending on the cooling rate of the hydrothermal system (Kring et al., 2020). Thus, sulfate reduction to produce framboidal pyrite was occurring to depths of 20–80 m below seafloor vents.
Textures indicate sulfate reduction occurred in the hydrothermal system, rather than under normal, low bottom water, shallow sedimentary temperatures after the hydrothermal system ceased operating. The pyrite framboids in 0077-46R-1 occur along the zoned walls of a vent channel, and the pyrite framboids in 0077-63R-2 occur along the zoned walls of a network of channels lined with hydrothermal dachiardite. The framboids are not distributed homogeneously or diffusively through the rock as a consequence of equilibrium pore water precipitation beneath a seawater-sediment interface. Nor are the framboids distributed in horizontal layers reflective of a kinetically controlled process parallel to the seawater-sediment interface.
As noted above, pyrite framboids grew amid the zeolites dachiardite and analcime, sometimes in association with heulandite and calcite. In natural hydrothermal environments, analcime and heulandite are generally produced at temperatures between 25°C and 100°C (Iijima, 1980), although they and dachiardite are stable at higher temperatures (Coombs et al., 1959; Liou, 1971; Ueda et al. 1980; Bargar and Beeson, 1984; Bargar et al., 1987). Heulandite may follow analcime precipitation as temperatures fall below 100–120°C (Mehegan et al., 1982; Utada 2001). At Yellowstone, heulandite was observed at hydrothermal temperatures of ∼40°C and ∼80°C at depths of 17 and 30 m (Bargar and Beeson, 1984). Pyrite framboids in association with heulandite, analcime, and calcite (Prol-Ledesma et al. 2002) have precipitated from submarine vents with fluid temperatures of 85–87°C (Núñez-Cornú et al. 2000; Alfonso et al., 2005). Elsewhere, analcime and heulandite, with pyrite, calcite, and other phases, may have been a product of hydrothermal alteration between 60°C and 70°C (Keith and Staples, 1985). Because the pyrite framboids in those samples and in our sample 0077-46R-1 are associated with calcite, we note that clumped isotope analyses of calcite deposited immediately on top of the breccias analyzed here record hydrothermal temperatures of 70°C (Bralower et al., 2020).
Modern strains of thermophilic sulfate-reducing bacteria exist at those temperatures at hydrothermal vents and hot springs. For example, Thermodesulfobacterium hydrogeniphilum (50–80°C; Jeanthon et al. 2002) and Desulfobacteriales (40°C and 70°C; Dhillon et al. 2003) were isolated from hydrothermal vents in the Gulf of California's Guaymas Basin, with sulfate reduction peaking between 60°C and 90°C (Kallmeyer and Boetius, 2004). Thermodesulfobacterium geofontis (70–90°C; Hamilton-Brehm et al., 2013) and Thermodesulfovibrio yellowstonii (40–70°C; Henry et al., 1994) were isolated from a thermal vent and pool, respectively, in Yellowstone National Park. Other examples associated with hydrothermal vents, subsurface geothermal waters, and hot oil field water include Thermodesulfatator altanticus (55°C and 75°C; Alain et al. 2010), Desulfothermus okinawensis (35–60°C; Nunoura et al. 2007), Desulfotomaculum and Thermanaeromanas (50–80°C; Kaksonen et al. 2006), and Thermodesulforhabdus norvegicus (44–74°C; Beeder et al., 1995). Sulfate reduction is a metabolic path utilized by archaea, too. For example, Archaeoglobus fulgidus (peak sulfate reduction rate at 82–84°C; Mitchell et al., 2009; see also Stetter et al., 1987, and Khelifi et al., 2010, 2014) was isolated from an active seafloor vent in Italy; and Archaeoglobus profundus (growing at temperatures up to 90°C; Burggraf et al., 1990) was isolated from a submarine hydrothermal vent in the Guaymas Basin.
The observed biological sulfur isotope fractionation in our Chicxulub samples required an energy source, or electron donors, for sulfate reduction (e.g., Smith and Klug, 1981; Jeanthon et al., 2002; Alazard et al., 2003; Kallmeyer and Boetius, 2004; Finke et al., 2007; Liamleam and Annachhatre, 2007; Cao et al., 2014). Potential electron donors in the Chicxulub system are hydrocarbons that are observed in a porous and permeable impact-brecciated zone within target carbonate beneath hydrothermally altered impactites in the Yax-1 core (Kring et al., 2004). Mobilization and transport of those hydrocarbons are indicated by hydrocarbon-bearing fractures and hydrocarbon-filled pores observed at the base of the Yax-1 impactite sequence and an isotopic signature of hydrocarbons detected in an overlying unit of impact breccias (Zurcher et al., 2005). Hydrocarbons were also detected in fluid inclusions in those upper peak-ring breccias and, when the hydrocarbons were entrained in hydrothermal fluids during the initial higher-temperature phase of the system, appear to have cracked to form ethane and propane (Lüders and Rickers, 2004). Hydrogen may have also been available when impact glass and other Chicxulub crater lithologies were altered (Christou and Bach, 2019), including mafic intrusions in the uplifted peak ring of the crater that contain decomposed olivine (Gulick et al., 2017; Schmieder et al., 2017b). Carbon sources may have included CO2 from dissolving carbonate and, at the top of the sequence, potentially wood and charcoal deposited at the top of the impactite sequence in the Expedition 364 core (Bralower et al., 2020).
Calcite carbon isotope values (δ13C) in sample 0077-46R-1 are 0.93 ± 0.33 to -7.59 ± 0.30 ‰. If hydrocarbons were the electron donor, the observed values imply carbonate precipitation was not contemporaneous with pyrite formation or, perhaps more likely, that the isotopes in the calcite are dominated by the limestone and limestone-derived carbonate beneath and throughout the breccia sequence, which have δ13C values of 0–2.9 ‰ (Lüders et al., 2003) and may have averaged 2 ‰ (Zurcher et al. 2005). A bulk δ13C value of -7.2 ‰ in a Yaxcopoil-1 impact breccia was likewise interpreted to reflect mixing of isotopic contributions from a methane (CH4)-bearing fluid and the carbonate reservoir of Chicxulub (Zurcher et al., 2005). Isotopic mixing of that type is observed in a modern hydrothermal sulfate-reducing system in the Gulf of California's Guaymas Basin, in that case producing intermediate δ13C values of -11.7 ± 1.6 ‰ from organic carbon values of -21.4 ‰ that mixed with marine limestone values of 0 ‰ (Peter and Shanks, 1992). We note that it is also possible that hydrocarbons, while present, were partially to wholly supplanted by other electron donors, such as H2, which would not have driven δ13C to strongly negative values. Some sulfate-reducing thermophiles utilize electrons from mixtures of hydrocarbons and hydrogen (Beeder et al., 1994; Henry et al., 1994; Hamilton-Brehm et al., 2013).
If we momentarily expand our view from the peak-ring location of the samples described here to the crater-wide hydrothermal system, we note that fluid chemistry will not be uniform, either in space or time. At a slightly greater distance from the crater center, in the Yaxcopoil-1 borehole, fluids were derived from basinal brines, with hydrocarbons, and had neutral to alkaline pH (Zurcher et al., 2005). Conditions were not, however, static, as the hydrothermal system cooled through phases of Ca-Na metasomatism, K metasomatism, clay precipitation (e.g., by the alteration of impact-generated glasses), and diagenesis (Zürcher and Kring, 2004). Interior to the peak ring, we envision a separate stream of fluids emanating from a ∼3 km thick central melt sheet with a grossly andesitic composition produced by bulk melting of the crust (Kring and Boynton, 1992). The melt sheet would have thermally consumed and contact-metamorphosed bounding crystalline bedrock, releasing additional fluids that may have been carried upward along the inner walls of the peak ring and possibly through fractures created in uplifted target rock by the impact event. Near the top of the hydrothermal system, whether that be near the peak ring or over the central melt sheet, seawater may have been drawn down and created thermal and chemical mixing zones. Models of fluid flow and heat transport in the Chicxulub system that were calculated by using the computer code HYDROTHERM (Abramov and Kring, 2007) indicate the relative proportions of fluids from different regions of the crater, delivered to any one place in the crater, changed with time. Thus, boreholes drilled elsewhere in the crater may reveal other hydrothermal alteration patterns and metabolic strategies.
It is important to note that a diverse array of microbes was identified in enrichment experiments and with DNA extraction techniques within the site M0077 impactites (Cockell et al., 2019). The organisms occur at depths where temperatures exceed 45°C (Gulick et al., 2017) and are, thus, thermophilic. We cannot completely rule out the possibility that the sulfide framboids analyzed here were produced by this modern assemblage of organisms, but we favor their origin in the original, post-impact hydrothermal system for the following reasons.
First, the pyrite framboids are enclosed within analcime and dachiardite, not on the surfaces of existing fractures and pore spaces. Second, the framboids occur along the margins of a clay-rich vent channel produced soon after impact (e.g., Fig. S5 of Kring et al., 2020). Third, and perhaps more telling, evidence of sulfate reducers exists in sediments deposited on top of the peak ring within 4 million years of impact (Fig. 3). An excursion of δ34S to -33 ‰ occurs in Paleocene sediments deposited on the peak ring ∼2.5 million years after the impact (Schaefer et al., 2020), about the same time a thermal evolution model (Abramov and Kring, 2007) indicates thermophilic temperatures should be prevalent in the hydrothermal system. That sulfur isotope signature may reflect sulfate reduction in the water column above the submerged peak ring (Schaefer et al., 2020). It is also possible the sulfide carrying that fractionated isotopic signature was vented from the hydrothermal system onto the seafloor and, thus, may be reflective of a subsurface biome. In either case, sulfate-reducing organisms existed in the crater ∼66 to 63 million years ago. Fourth, we also note that it is easier to introduce organisms to the system when it is only a few meters beneath the seafloor, rather than at some later time when the suevite is getting buried progressively deeper by post-impact sediments, which are now 617 m thick. For these reasons, we suggest the pyrite framboids capture sulfate reduction that occurred in the crater soon after its formation. This implies that sulfate reducers found in the core today are living remnants of a 66 million-year-old microbial colony that emerged after the Chicxulub impact event.
Sulfate reduction is a process utilized by microbial organisms since at least the Paleoarchean, when it produced strongly depleted δ34S in pyrite embedded in barite from North Pole, Australia (Shen et al., 2001). Likewise, Paleoarchean microbial sulfate reduction produced strongly depleted δ34S in pyrite now found in the Barberton Greenstone Belt, South Africa (Roerdink et al., 2013), possibly due to drawdown of seawater sulfate (Roerdink et al., 2016). Thermophilic sulfate-reducing organisms occur deep within the bacterial domain (e.g., Henry et al., 1994). Nonetheless, Hadean environmental chemistry (e.g., Kasting, 2005; Russell and Arndt, 2005; Zahnle et al., 2007, 2020; Arndt and Nisbet, 2012) may have precluded the availability of sulfate as an energy source. Yet hydrothermal systems in which other metabolic reactions are utilized remain an attractive site for the early evolution of life (e.g., Woese et al., 1990; Reysenbach and Shock, 2002; Schwartzman and Lineweaver, 2004; Ciccarelli et al., 2006), where high temperatures facilitate key reactions (Stockbridge et al., 2010; Wolfenden et al., 2015), either at oceanic ridges and continental volcanoes (e.g., Shock and Schulte, 1998; Nisbet and Sleep, 2001; Martin et al., 2008; Shibuya et al., 2016), continental hot springs (Deamer et al., 2019; Damer and Deamer, 2020), or through carbonaceous sedimentary layers (Westall et al., 2018). Here we suggest that hydrothermal systems were being produced by a global distribution of impact cratering events, affecting all crustal lithologies and thus providing many of the same attributes that made previously described hydrothermal environments attractive. Serpentinization, for example, which produces H2 as an energy source, may have reasonably occurred in impact craters that penetrated mafic and ultramafic crust (Schulte et al., 2006).
5. Conclusions
Sulfur isotope analyses of pyrite framboids in impact breccia from the Chicxulub crater indicate thermophilic colonies of sulfate-reducing organisms inhabited the porous, permeable rock beneath the floor of the crater and fed on sulfate transported through the rock via an impact-generated hydrothermal system. Similar sulfur isotope signatures in overlying sediments (Schaefer et al., 2020) imply sulfate-reducing organisms persisted for at least 2.5 million years after impact, potentially in both the subsurface and in the water column above the crater floor. Thermophilic sulfate-reducing organisms that currently occur in the same rocks, now buried hundreds of meters beneath the seafloor, may be the living remnants of that ∼66 to 63 million-year-old colony of microorganisms.
Studies of Chicxulub have, thus, demonstrated that large peak-ring and multi-ring basins have porous, permeable subsurface environments; that such impact craters host vast subsurface hydrothermal systems; and that those systems can host microbial ecosystems. In the case of 66 Ma Chicxulub, sulfur isotope fractionation, indicative of microbial sulfate reduction, occurred, which is similar to the metabolic pathway used as long ago as 3.52 Ga in the Paleoarchean. In the Hadean, other metabolic reactions may have dominated the energy yields required by life, but, we suggest, those reactions may have occurred in the same type of porous, permeable impact-generated hydrothermal system that existed in the Chicxulub crater.
Footnotes
Acknowledgments
This research utilized samples provided by the International Ocean Discovery Program and International Continental Scientific Drilling Program. The research was supported by the National Science Foundation (grant OCE-1736826). The authors thank their expedition colleagues and MARUM staff for core recovery, core logging, and first-generation studies of core material, which made this second-generation study possible. We thank the LPI library staff, which provided additional help when normal access to previously published research was curtailed by the COVID-19 pandemic. This is LPI Contribution No. 2545. LPI is operated by USRA under a cooperative agreement with the Science Mission Directorate of the National Aeronautics and Space Administration. The NordSIMS facility is supported by Swedish Research Council infrastructure grant 2017-00671; this is NordSIMS contribution 655. We thank three anonymous reviewers, Associate Editor Tim Lyons, Editor-in-Chief Sherry L. Cady, and journal staff for their help to sharpen the science in the paper and for doing so in the midst of a health emergency.
Abbreviations Used
Associate Editor: Tim Lyons
