Abstract
The “RNA first” model for the origin of life holds that RNA emerged spontaneously on early Earth and developed into life through its dual capabilities for genetics and catalysis. The model's central weakness is the difficulty of making its building blocks, in particular, the glycosidic bond joining nucleobases to ribose. Thus, the focus of much of the modern literature on the topic is directed toward solving this difficulty and includes elegant, though indirect, methods for making this bond. Here, we report that the glycosidic bond in canonical pyrimidine and purine ribonucleotides can be formed by direct coupling of cyclic carbohydrate phosphates with free nucleobases, all reported to be available by experimentally supported pathways that might have operated on early Earth.
1. Introduction
Because of its dual capabilities as a genetic molecule and as a catalytic molecule (Kruger et al., 1982; Guerrier-Takada et al., 1983), RNA seems to be an ideal first chemical to support Darwinian evolution as life emerged on early Earth. This “RNA first” hypothesis (Gilbert, 1986) requires, of course, that RNA was synthesized spontaneously on early Earth by abiotic pathways. Accordingly, many groups, using “RNA first” as their working hypothesis, have attempted to combine laboratory chemistry with models for the early geology of Earth and its early atmospheric evolution to yield building blocks for RNA (Orgel, 2004; Harrison et al., 2005; Ruiz-Mirazo et al., 2014).
Models to make various components of the building blocks of RNA have developed over the years. Routes to make the canonical nucleobases have been known since the 1960s, including adenine (Oro, 1961), guanine (Levy et al., 1999), cytosine, and uracil (Robertson and Miller, 1995). Likewise, prebiotic routes to ribose involving borate minerals are proposed (Ricardo et al., 2004; Kim et al., 2011; Furukawa and Kakegawa, 2017). Also, the prebiotic synthesis of enantiomeric enriched carbohydrates (Breslow et al., 2013) and 2-deoxy ribose are reported (Steer et al., 2017).
However, prebiotic routes to nucleosides, which connect the ribose to the nucleobases, have been a prominent missing link in the abiotic synthesis of RNA. Direct glycosidic bond formation was first studied by the Orgel group, which showed that the incubation of ribose with purine nucleobases in the presence of Mg2+ in dry state yielded purine nucleosides, adenosine, and guanosine (Fuller et al., 1972). However, the direct condensation of ribose and these purines also gave unwanted isomers as major products and failed entirely for the pyrimidines, cytosine, and uracil.
This drove Sutherland, Carell, and others in the field to consider indirect approaches. For example, Carell's group reported the direct condensation of formylated aminopyrimidines with ribose to yield a product that, although not a canonical nucleoside, could be further elaborated to yield adenosine and guanosine, without phosphates but here with regiochemical control (Becker et al., 2016). Sutherland and Powner focused on the pyrimidine nucleotides (as 2′,3′-phosphates), forming a precursor of these from fragments of both the nucleobase and the ribose. The product was then elaborated to complete both the sugar and the nucleobase, together with photochemical conversion of the unwanted stereoisomers (Powner et al., 2009; Stairs et al., 2017; Xu et al., 2017). Also, photoanomerization of α-cytidine 2′-phosphate provided a mixture of β-cytidine and uridine 2′-phosphate (Powner and Sutherland, 2008).
Direct syntheses have been reported recently by Nam et al. (2017) and Nam et al. (2018), who sought to phosphorylate ribose followed by further reaction with nucleobases to form nucleosides in microdroplets in acidic condition. Regiochemistry as well as stereochemistry of the nucleosides was not known due to the limitation of their analytical methods, unfortunately. Separately, Saladino et al. (2015) reported that various organics, including nucleosides, are made from formamide by high-energy proton irradiation in the presence of meteorites. High-energy proton fluxes are expected, of course, only in outer space (in meteors or comets). Examination of a small set of carbonaceous chondrites finds none of these materials in easily detectable amounts for delivery to Earth, although ribose and other carbohydrates are made in cosmogenic ice simulants (Meinert et al., 2016).
2. Materials and Methods
2.1. Materials
Ribose 1-phosphate was obtained from Toronto Research Chemicals. Nucleoside starting materials for the chemical synthesis of ribonucleoside 2′-phosphate were obtained from Carbosynth. All other chemicals were obtained from Sigma-Aldrich and TCI and used as received.
2.2. Methods
The reaction of ribose 1,2-cyclic phosphate
Ion exchange high-performance liquid chromatography (HPLC) analysis was done with a DNAPac PA-100 column (4 × 250 mm; Thermo Scientific) on a Waters 2695 separation module equipped with a 996 photodiode array detector. The column was eluted with a gradient of (A) water and (B) 1 M of ammonium bicarbonate. The elution program created a linear gradient started from 100% (by volume) A to 80% A at 15 min with a flow rate of 0.5 mL/min. Peak detection and integration were conducted with the signal at 260 nm. Full UV spectra (210–400 nm) were also obtained. Reverse-phase HPLC analysis was done with a C-18 reversed-phase narrow bore column (3 mm i.d., 150 mm length, 5 μm; SunFire; Waters) on a Waters 2695 separation module equipped with a 996 photodiode array detector. The column was eluted with a gradient of (A) aqueous 25 mM triethylammonium acetate and (B) 100% acetonitrile. The elution program created a linear gradient started from 100% (by volume) A to 85% A at 10 min with a flow rate of 0.5 mL/min. Peak detection and integration were conducted with the signal at 260 nm. Full UV spectra (210–350 nm) were also obtained. Preparative HPLC purification was achieved with an ion exchange column (22 mm i.d., 250 mm length, 5 μm; DNAPac PA-100; Thermo Fisher Scientific) on a Waters Delta 600 module. The column was eluted with a gradient of (A) water and (B) 1 M of ammonium bicarbonate. The elution program created a linear gradient started from 100% A to 80% A : 20% B at 15 min with a flow rate of 10 mL/min. Peak detection was conducted by using the 260 nm absorbance. Nuclear magnetic resonance (NMR) spectra were recorded in deuterium oxide on a Varian Mercury 300 NMR spectrometer. High-resolution mass spectrometry was conducted on Agilent 6220 Time-of-Flight connected with an Agilent 1100 series system that consisted of a G13793 degasser and a G1312B binary pump with electrospray ionization in negative mode.
Ribose 1,2-cyclic phosphate
3. Results
We previously reported prebiotic synthesis of adenosine 2′-phosphate (Kim and Benner, 2017), nicotinamide nucleoside 2′-phosphate (Kim and Benner, 2018), and some noncanonical purine and pyrimidine nucleoside 2′-phosphates (yields range from 15% to 35%) (Kim and Benner, 2017) by the coupling of ribose 1,2-cyclic phosphate
When combined, these chemistries provide a scenario for a prebiotic source for adenylic acid. We therefore asked whether some combination of these chemistries might yield direct synthesis of all four of the canonical nucleotides. We show that they can be synthesized in plausible prebiotic environments (Fig. 1).
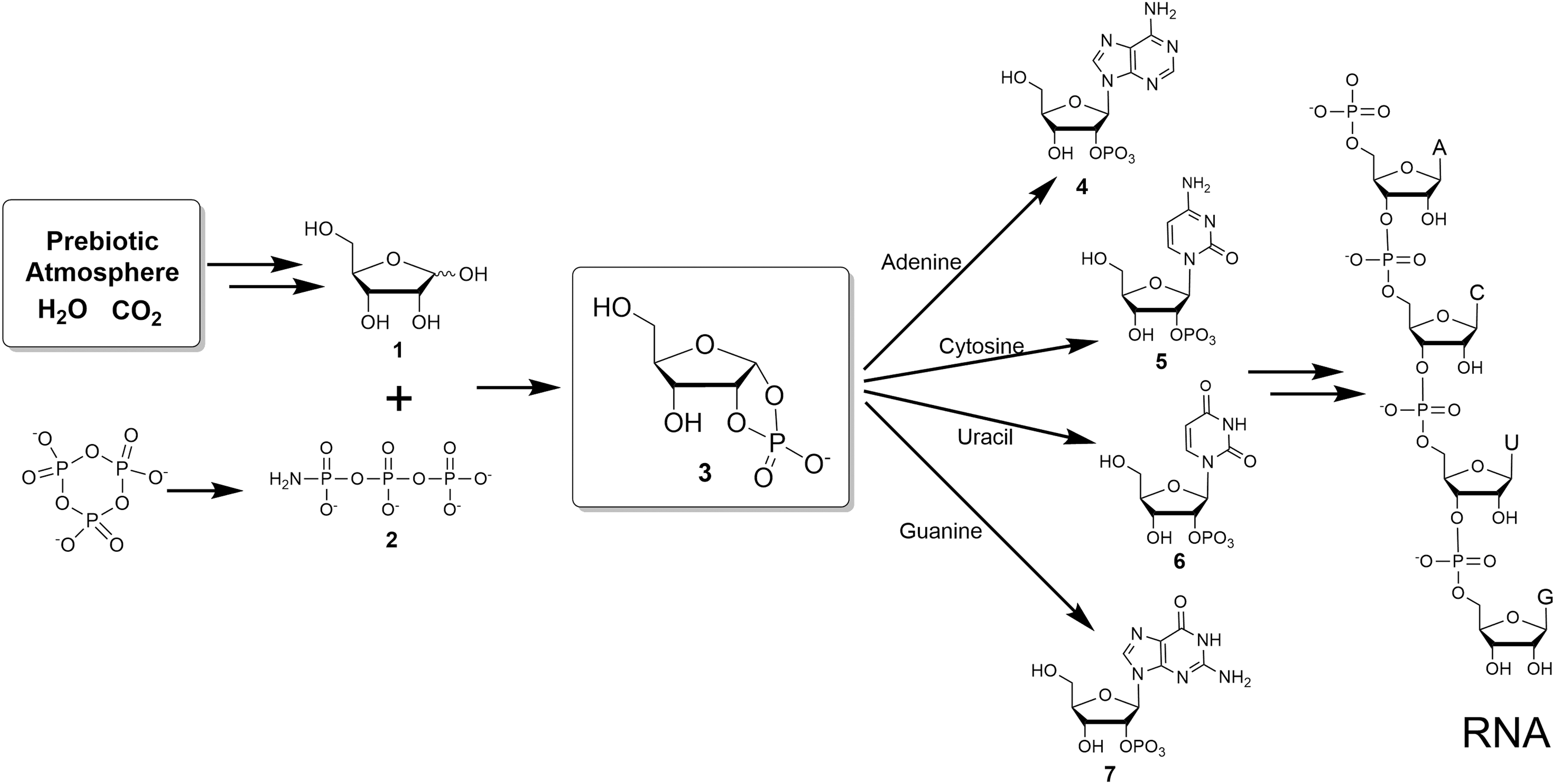
Plausible prebiotic synthetic pathway to RNA. The coupling of ribose 1,2-cyclic phosphate
First, coupling of ribose-1,2-cyclic phosphate
However, the coupling reaction of cytosine and ribose 1,2-cyclic phosphate
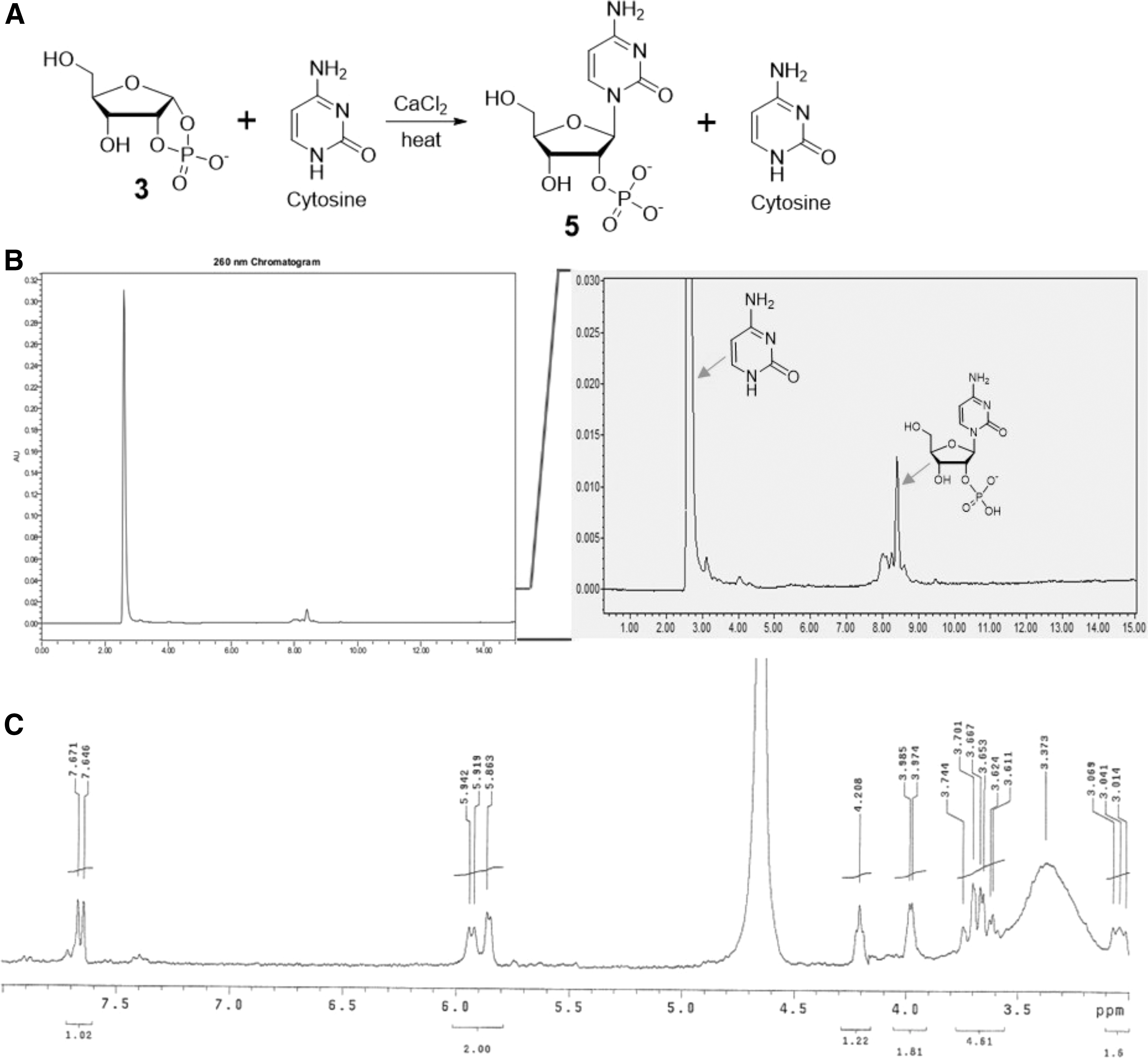
The coupling of ribose 1,2-cyclic phosphate
Formation of Cytidine 2′-Phosphate
A mixture of ribose 1,2-cyclic phosphate
This reaction largely produced the N1-product, with minor materials possibly arising from the reaction of the N4-exocylic amine group. The identity of the product was confirmed to be canonical cytidine 2′-phosphate
Uridine can be made from cytidine by hydrolytic deamination of a cytosine amine group (Powner et al., 2009). However, the direct synthesis of uridine from uracil would also be interesting, as uracil is known in meteorites (Stoks and Schwartz, 1979) and is likely to have been present in relative abundance on a prebiotic Earth, with cytosine, by endogenous synthesis (Robertson and Miller, 1995). The coupling of uracil and ribose 1,2-cyclic phosphate
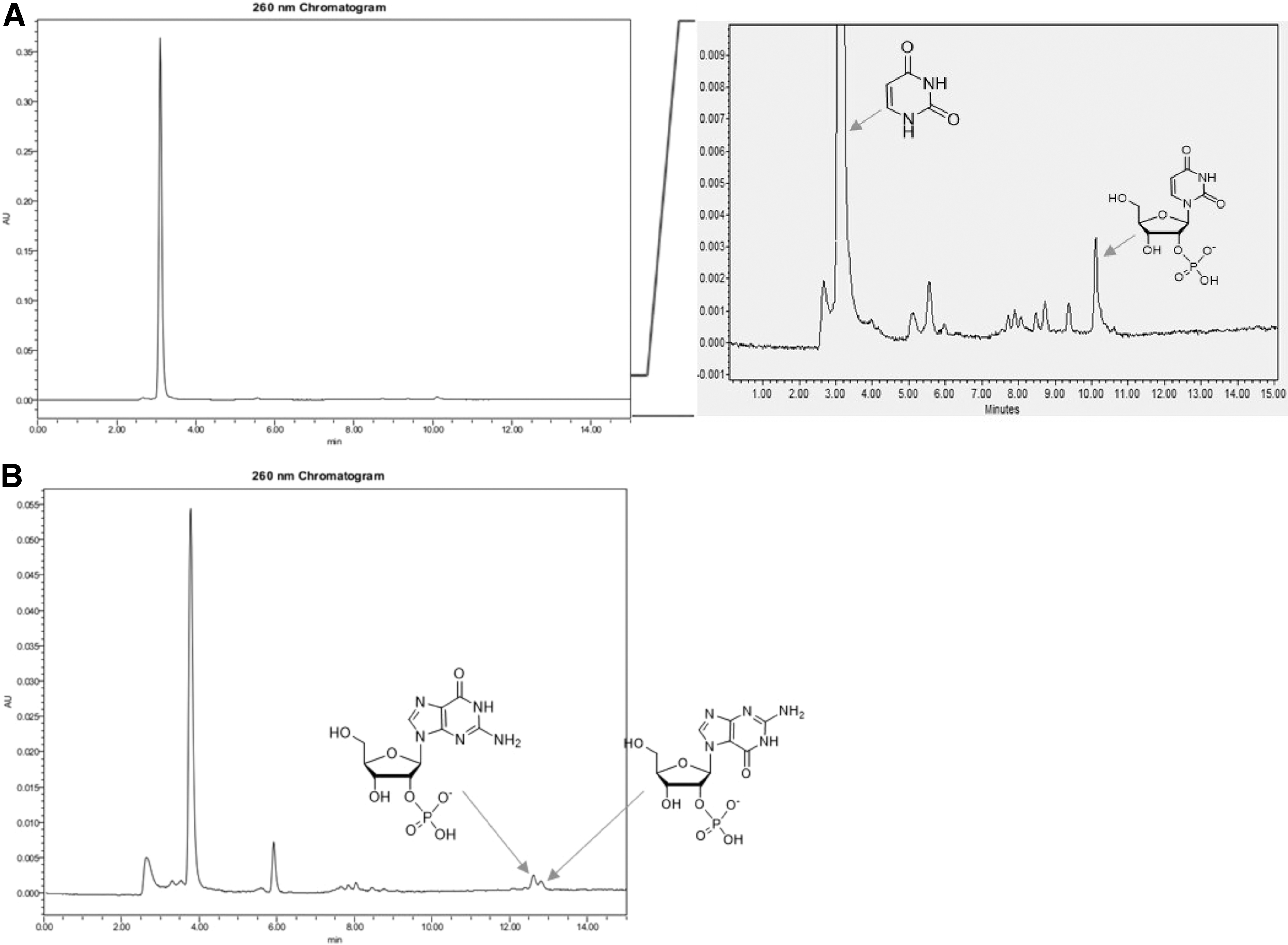
The coupling of ribose 1,2-cyclic phosphate
Interestingly, these reaction conditions also worked for both canonical purines, adenine, and guanine. The adenosine phosphate from this methodology was reported earlier (Kim and Benner, 2017). Thus, the coupling of guanine and ribose 1,2-cyclic phosphate
4. Discussion
These results show that all four of the glycosidic bonds in the four canonical nucleosides for RNA can be made directly from ribose 1,2-cyclic phosphate
One of the glycosidic bond formations presented in this study (in case of cytosine) gave a generally acceptable reaction yield (∼7%), but some of the others (uracil and guanine) gave a very low condensation yield (less than 1%). The limitations of the current study due to the low yield of uracil and guanine can be mitigated by some justifications. In the case of uracil, although uracil gave a low condensation yield with ribose 1,2-cyclic phosphate, uridine can be generated from cytidine by deamination reaction. This means that the higher yield of condensation of cytosine can be translated to the acceptable synthesis of uridine. In the case of guanine, the prebiotic synthesis of guanosine often provided a low reaction yield (Fuller et al., 1972; Becker et al., 2016). This can be attributed to the very low solubility of guanine.
Besides the reaction yield, all the materials in this study can be obtained under prebiotic conditions. Generation of the nucleobases requires classical reactions of hydrogen cyanide, cyanoacetylene, and other species available from the atmosphere, either directly or indirectly by way of formamide. Generation of the ribose requires atmospheric formaldehyde percolating through serpentinizing basalts that contain igneous borate minerals (e.g., tourmalines). The selective prebiotic synthesis of ribose with high yield has not been reported, although the borate mineral shows selective affinity and stabilization of ribose (Ricardo et al., 2004; Furukawa and Kakegawa, 2017). This may be why some investigators do not start with ribose in their prebiotic synthesis of nucleosides (Powner et al., 2009). However, other successful prebiotic syntheses of nucleosides still require ribose (Becker et al., 2016). These circumstances indicate that the high-yield selective ribose synthesis is needed for the successful prebiotic synthesis of RNA. Generation of the amidotriphosphate requires ammonia (from the atmosphere) to react in water with cyclic trimetaphosphate, which is made by strong dehydration of inorganic phosphate.
Footnotes
Acknowledgments
We would like to thank Prof. Steven A. Benner for many discussions and comments on the manuscript. This publication was made possible through the support of a grant from the John Templeton Foundation 54466. The opinions expressed in this publication are those of the author and do not necessarily reflect the views of the John Templeton Foundation. Mass spectrometer facility was available from NIH funding (S10 OD021758-01A1).
Author Disclosure Statement
No competing financial interests exist.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
