Abstract
Significance:
Seed germination and seedling establishment are characterized by changes in the intracellular redox state modulated by accelerated production of nitric oxide (NO) and reactive oxygen species (ROS). Redox regulation and enhanced accumulation of NO and ROS, approaching excessively high levels during seed imbibition, are critically important for breaking endodormancy and inducing germination.
Recent Advances:
Upon depletion of oxygen under the seed coat, NO is produced anaerobically in the reductive pathway associated mainly with mitochondria, and it participates in the energy metabolism of the seed until radicle protrusion. NO turnover involves nitrate reduction to nitrite in the cytosol, nitrite reduction to NO in mitochondria, and NO oxygenation in the cytosol in the reaction involving the hypoxically induced class 1 phytoglobin. In postgerminative degradation of seed tissues, NO and ROS are involved in redox signaling via post-translational modification of proteins and mediation of phytohormonal responses.
Critical Issues:
The crosstalk between the cellular redox potential, NO, ROS, and phytohormones integrates major physiological processes related to seed germination. Intensive accumulation of NO and ROS during imbibition is critically important for breaking seed dormancy. Upon oxygen depletion, NO and other nitrous oxides (NOx) are produced anaerobically and support energy metabolism prior to radicle protrusion.
Future Directions:
The turnover of NOx and ROS is determined by the intracellular redox balance, and it self-controls redox and energy levels upon germination. The particular details, regulation of this process, and its physiological significance remain to be established. Antioxid. Redox Signal. 42, 442–461.
Bioenergetics of Germinating Seeds
The initial plant growth stage is seed germination that occupies the time period starting from water uptake by dry seed and ending with the protrusion of embryonic tissues from the seed coat (Bewley, 1997). This period depends on the optimal environmental factors and physiological conditions within the seed. Germination is followed by stages of postgerminative growth and development, which are initially dependent on the mobilization of storage reserves and represent a direct continuation of the germination stage. Rapid resumption of metabolic and respiratory activities following seed imbibition is directly dependent on ATP synthesis that supports high energetic demands of germinating seeds and early (preautotrophic) stages of seedling growth (Logan et al., 2001).
Seed germination is characterized by a fast consumption of internal oxygen that results in the limitation of respiratory capacity (Bewley, 1997). Few species of higher plants are even able to germinate and grow under strict anoxia. These include the Echinochloa (barnyard grass) species, rice (Oryza sativa), and the African legume Erythina caffra (Kennedy et al., 1992; Magneschi and Perata, 2009). Seeds of most species require oxygen for germination, which is then rapidly depleted upon imbibition until radicle protrusion. In some cases, for example, in barley, germination under hypoxic conditions is prevented due to the induction of secondary dormancy (Gómez-Álvarez et al., 2023; Hoang et al., 2013). The physiological consequence of oxygen limitation is the suppression of mitochondrial respiration that results in a high redox potential and depleted energy status of germinating seeds (Rolletschek et al., 2004; Weber et al., 2005). However, despite low internal oxygen levels, seed ATP levels and energy charge generally do not decrease and even become elevated (Benamar et al., 2003; Duff et al., 1998; Logan et al., 2001). This suggests that switching to the mechanisms for energy generation alternative to aerobic respiration could be in effect. One such mechanism is glycolysis (Kennedy et al., 1992), which has a limited ability to support high energy demands due to low ATP outcome. Another possibility is the operation of mitochondria in the absence of oxygen using nitrite as a terminal electron acceptor (Igamberdiev and Hill, 2004). These two main mechanisms work together to meet the energy demands of the germinating seed (Nie et al., 2022).
The decreasing internal oxygen levels in seeds during imbibition affect intracellular redox potential and impose reductive stress on cells, which triggers the formation of reactive oxygen species (ROS) and reactive nitrogen species (RNS) (Gonzali et al., 2015; Xiao and Loscalzo, 2020) (Fig. 1). The crosstalk between ROS, RNS, phytohormones, and expressed proteins is the central point for regulation of major physiological processes related to seed germination (Bykova et al., 2015; Ciacka et al., 2022). Rapid ROS accumulation approaching dangerous levels in seed tissues during imbibition is critically important condition for breaking dormancy. Upon oxygen depletion, NO and other RNS including different nitrous oxides (NOx) are produced anaerobically. ROS and RNS participate in post-translational modification of proteins, in the mediation of hormonal responses, as well as in other signaling events essential for the regulation and integration of cellular processes in germinating seeds such as fatty acid nitration (Vollár et al., 2020) and degradation of individual proteins via the N-degron pathway (Manrique-Gil et al., 2021).
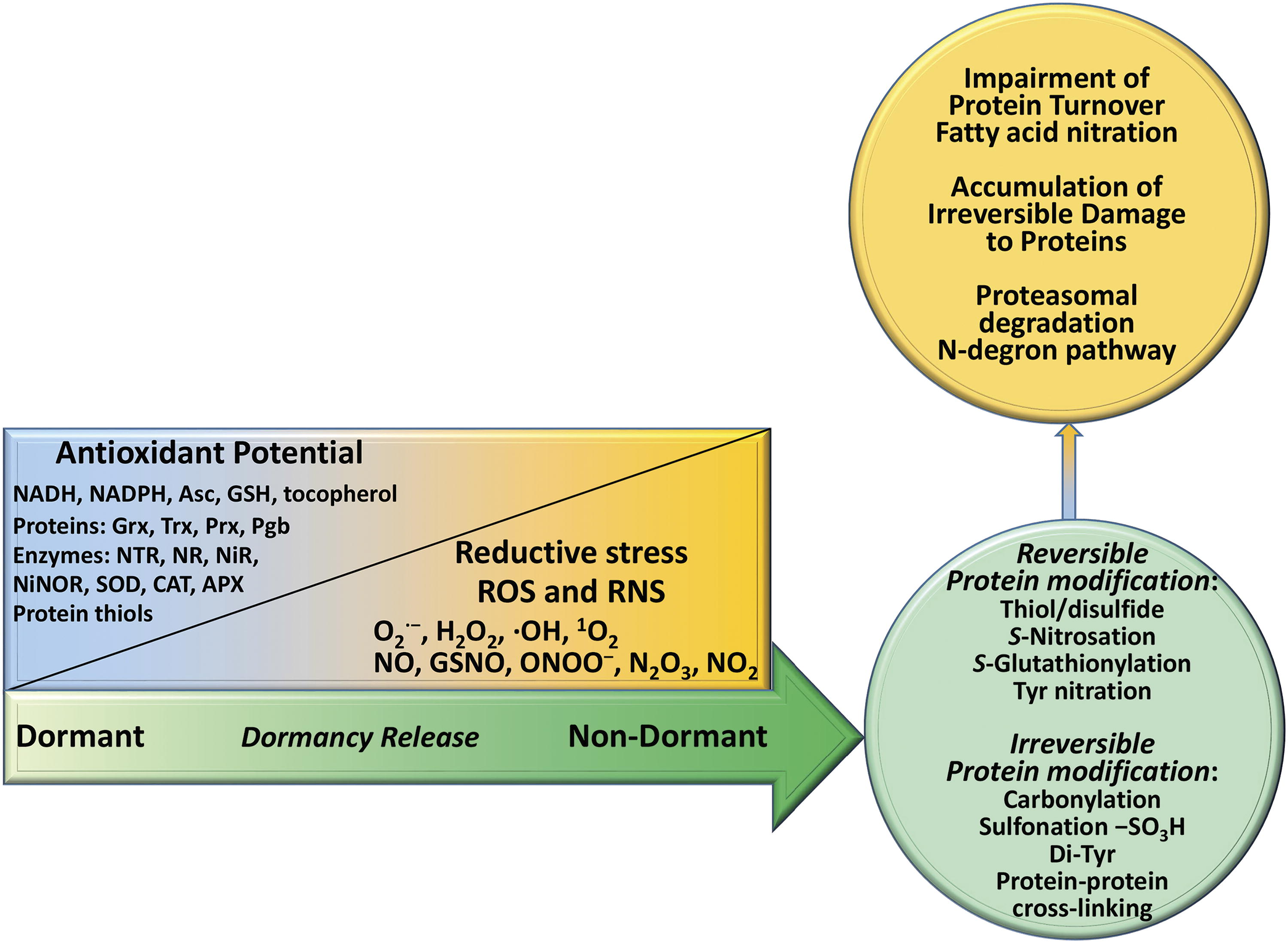
Alterations in the redox potential and ROS/RNS levels are tightly coordinated in cells (Bykova and Igamberdiev, 2016; Igamberdiev and Bykova, 2018) and collectively control seed germination and dormancy (Bykova et al., 2015). This occurs via the complex networks of ROS/RNS-mediated redox signaling pathways and phytohormones that regulate the expression of the key genes participating in cell proliferation and differentiation. The activation and deactivation cycles involving H2O2 and NO contribute to the spatiotemporal organization of seed germination events. Prominent ROS species H2O2 and O2 •−, together with NO and their derivatives (such as peroxynitrite ONOO−, NO2, and N2O3), are key redox signaling agents generated by the dozens of enzymes and redox proteins, including the plasma membrane NADPH oxidase (respiratory burst oxidase homolog, Rboh) and the mitochondrial electron transport chain (Møller, 2001; Sies and Jones, 2020).
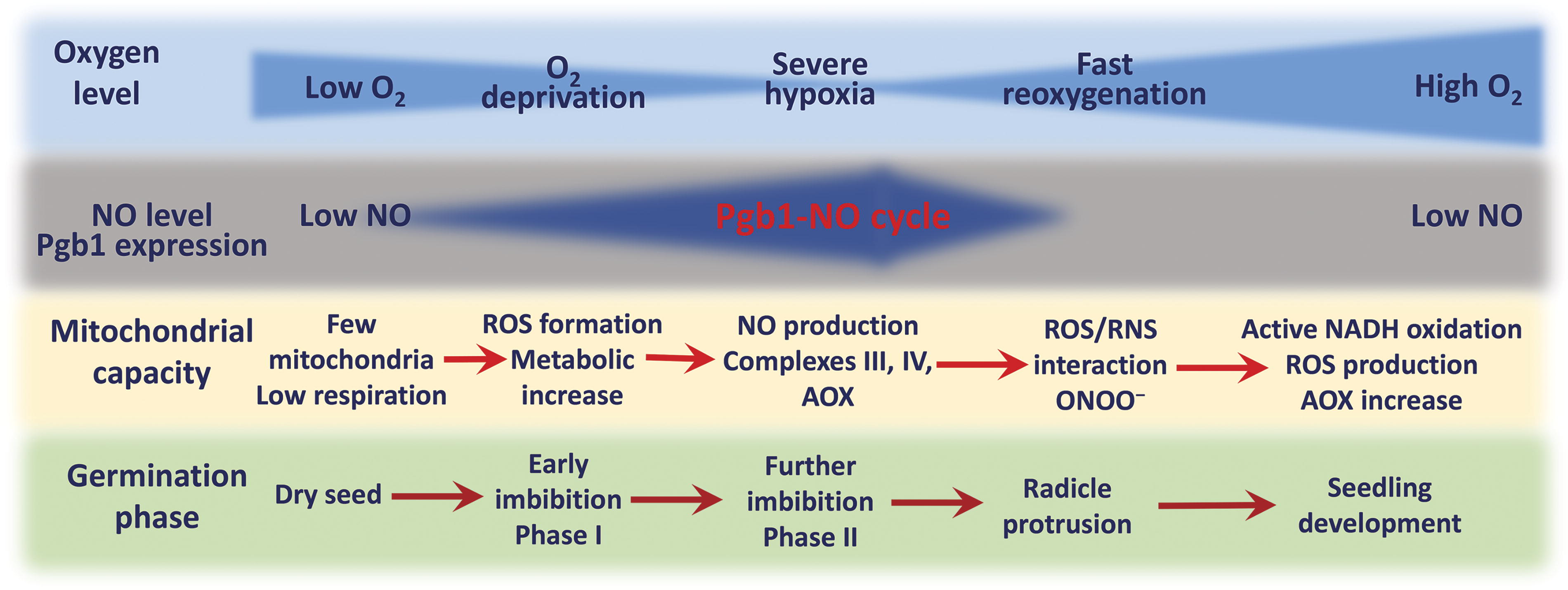
ATP synthesis starts as soon as the seeds are rehydrated, and it continues regardless of the limited oxygen supply condition that persists until the rupture of testa (Duff et al., 1998; Paszkiewicz et al., 2017). Figure 2 shows changes in the levels of oxygen and NO, phytoglobin (Pgb) expression, and development of the mitochondrial capacity in seeds during germination. The adenylate status of cells is balanced with the levels of reduced and oxidized pyridine nucleotides through the regulated uncoupling of the pyridine nucleotide pool from proton translocation, which supports oxidative phosphorylation and ATP production in mitochondria (Igamberdiev and Bykova, 2023), and through the oxidation of substrates noncoupled to NAD+ reduction in peroxisomes (Igamberdiev and Lea, 2002). The set of adenylates and pyridine nucleotides constitutes a generalized cell energy status that determines the rates of major metabolic fluxes. As a result, fluxes of energy and information become organized spatially and temporally, providing physiological conditions for self-maintenance of metabolism (Igamberdiev and Kleczkowski, 2019). In germinating seeds, their energy status via redox regulation of major signal transduction events controls endodormancy, promotes germination, and establishes phytohormone profiles essential for triggering major developmental processes. In this review, we discuss how changes in the intracellular redox state associated with limited oxygen availability and active metabolism result in the production and crosstalk of NO and ROS. We will argue that their production integrates the metabolism and energetics of germinating seeds, alleviates dormancy, and plays a significant role in seedling development.
ROS in Seed Dormancy Alleviation and Germination
Upon imbibition, the immediate activation of metabolism dynamically alters the intracellular redox state and leads to the generation of ROS, causing oxidative stress that targets proteins, membranes, and nucleic acids. The available oxygen is rapidly consumed due to the low permeability of the seed coat, which turns the conditions inside the seed to highly hypoxic. Under these conditions, respiration is suppressed and the bioenergetics of seeds become mainly anaerobic. Later, after radicle protrusion, oxygen supply becomes restored, and oxygen availability stimulates aerobic respiration in mitochondria and flavin-dependent oxidation in peroxisomes and other compartments. Therefore, in regard to oxygen availability, we can distinguish between three phases: (1) early oxygen consumption during the first hours after imbibition resulting in ROS production; (2) the following anaerobic stage before the radicle protrusion characterized by fermentation and NO production in anaerobic reduction of nitrate and nitrite; and (3) aerobic metabolism after the radicle protrusion and cellular reoxygenation, involving fully developed mitochondria and peroxisomes.
In the first phase, ROS are accumulated, leading to breaking dormancy in seeds with low levels of endodormancy. Seeds of plant species from different taxonomic groups actively produce ROS such as H2O2, O2 •−, hydroxyl radicals, and RNS such as nitric oxide (NO) upon imbibition (Oracz et al., 2009; Wojtyla et al., 2016). ROS are generated by NADPH oxidase (Rboh) and by electron leakage from the components of electron transport chain (ETC), representing an oxidative burst during rehydration (Colville and Kranner, 2010; Møller et al., 2007). The antioxidant systems in seeds are activated immediately upon rehydration (De Gara et al., 1997; Müller et al., 2009). The ability of seeds to germinate is related to the accumulation of a critical level of H2O2, which is produced at a higher level or scavenged at a lower rate in nondormant than in dormant seeds upon imbibition (Bailly et al., 2008; Oracz et al., 2007). Lower catalase activity in nondormant seeds results in a higher H2O2 level (Leymarie et al., 2012).
The balance between ROS-producing and ROS-scavenging systems plays a primary role in the induction of dormancy alleviation (Oracz et al., 2009). The nature of this effect can be associated with H2O2 interaction with specific phytohormones and mediation of the interplay between phytohormones (Bailly et al., 2008; Finkelstein et al., 2008). In Arabidopsis, at low H2O2 levels, seeds remain dormant, while the elevation of H2O2 in the radicle triggers gibberellic acid (GA) signaling activation. The increase in H2O2 takes place first in the cytoplasm, then in the nucleus, and finally in the cell wall, and it inversely correlates with the decrease in catalase activity as compared to dormant seeds (Leymarie et al., 2012). In barley (Hordeum vulgare L.), H2O2 treatment upregulated the expression of GA-induced gene HvExpA11, inhibited the expression of HvGA2ox3 involved in GA catabolism, and enhanced the expression of HvGA20ox1 implicated in GA synthesis. Treatment with abscisic acid (ABA) upregulated catalase activity through the transcriptional activation of HvCAT2 gene (Bahin et al., 2011). In sunflower (Helianthus annuus L.) embryos, a marked increase in the generation of H2O2 and O2 •− in the embryonic axes was observed upon HCN treatment that resulted in germination. The effect was due to the inhibition of catalase and SOD as well as activation of NADPH oxidase (Oracz et al., 2009). When sunflower seeds gain the ability to germinate during dry after-ripening, they accumulate high amounts of H2O2 and exhibit a diminished detoxification ability through catalase, which was evident from the decrease in CATA1 transcript (Bouteau et al., 2007). The role of catalase expression in regulating H2O2 levels during dormancy and in the course of dormancy breakage in grapevines (Vitis vinifera L.) was demonstrated by Perez and Lira (2005). One of the possible targets of H2O2 is ABA catabolism, which is upregulated in this process and requires NO accumulation. Furthermore, H2O2 is also involved in the upregulation of GA biosynthesis (Liu et al., 2010). The involvement of H2O2-mediated redox-dependent signaling in dormancy release and germination was demonstrated for several C4 grasses, where the inhibition of NADPH oxidase by diphenyleneiodonium suppressed germination (Sarath et al., 2007). The stimulation of endogenous production of NO by H2O2, and the abolition of the H2O2-responsive induction of seed germination by the application of NO scavengers provide evidence for the crosstalk between H2O2 and NO.
The antioxidant-rich environment in fruits protects the developing plant embryos from the damage caused by ROS and prevents early germination. After the fruit maturation and seed dispersal, a prolonged exposure to oxygen and moisture enables the seeds to produce the ROS needed for seed germination (Grover, 2013). Apple (Malus domestica Borkh.) seeds exhibit a biphasic pattern of variation in H2O2 concentration in the embryos during cold stratification. The H2O2 concentration increased markedly upon imbibition, while NO production by the embryos was low during the entire period of stratification, but increased significantly when seeds started to germinate. This indicates that the increase in H2O2 levels and protein carbonylation are important in seed dormancy alleviation, while NO becomes necessary for germination (Dębska et al., 2013). A key role of H2O2 in the coordination of pea seed germination, where it acted as a priming factor involving specific changes at the proteome, transcriptome, and hormonal levels, was demonstrated; in particular, the activation of one of MAP kinase genes by H2O2 and carbonylation of storage proteins were implicated in dormancy alleviation (Barba-Espin et al., 2011).
Upon imbibition, drastic changes in the potential of redox-active metabolites have a profound effect on the protein thiol redox state. The latter becomes significantly elevated upon imbibition and treatment with GA. A higher abundance of antioxidant proteins and enzymes important for redox control, ROS scavenging, and detoxification in imbibed dormant as compared to nondormant wheat seeds indicates that more intensive ROS scavenging allows for dormancy maintenance (Bykova et al., 2011a; Bykova et al., 2011b). These conclusions can be explained by the concept of “oxidative window” in dormancy alleviation (Bailly et al., 2008), that is, the range of intracellular ROS concentrations necessary to break seed dormancy. The release of dormancy is achieved in the after-ripening process under the conditions when ROS accumulation modifies redox status and breaks dormancy control mechanisms by altering protein functions through oxidative modifications (Bykova et al., 2011a; Bykova et al., 2011b; Bykova and Rampitsch, 2013). As cysteine (Cys) often has catalytic or structural roles in proteins, such modifications profoundly influence protein function (Wouters et al., 2010). This is schematically presented in Figure 1, showing the interconnection between ROS accumulation, dormancy release, and reduction/oxidation of Cys residues in proteins.
The redox signaling via biochemical communication between the ROS-driven carbonylation and RNS-driven nitrosation was demonstrated in a number of recent redox proteomics studies with photosynthetic and nonphotosynthetic plant tissues and in response to abiotic stresses, indicating their importance in redox homeostasis (Bykova et al., 2023; Bykova and Rampitsch, 2013; Lounifi et al., 2013; Sandalio et al., 2023). Protein carbonylation is a permanent marker of oxidative stress, and it has been implicated in the physiological transitions of seeds from dormancy to high germination potential and aging (Lounifi et al., 2013). This irreversible protein modification was shown to be concomitantly increased with ROS accumulation and involved in breaking dormancy and/or the loss of germination ability due to aging. The specificity of protein carbonylation in developing and germinating Arabidopsis seeds reflected selective targeting of proteins with certain functional roles (Job et al., 2005; Lounifi et al., 2013). A number of factors affect the carbonylation process including RKPT-rich and surface-exposed protein regions, and a tendency for clustering of carbonylated sites (Rao and Møller, 2011).
A combination of protein-intrinsic properties and ROS detoxification capacity impacts oxidative stress resistance (Chang et al., 2020). The multistage Cys oxidation resulting in irreversible oxidative damage and tyrosine nitration by peroxynitrite are likely to take place during transition between the state in which ROS prevails and the state in which RNS prevails. S-nitrosation of Rboh was shown to decrease enzymatic activity and follow a high level of NO production during a biotic stress response, thus limiting ROS generation (Yun et al., 2011). The NADPH oxidase AtrbohB was shown to play a role in ROS production during after-ripening in Arabidopsis seeds (Müller et al., 2009). The role of RNS becomes prevailing when oxygen in the seed covered by the coat is depleted and NO starts to generate anaerobic nitrite reduction. We discuss below the mechanisms for the maintenance of redox balance and energy level in hypoxic germinating seeds via NO turnover.
NO and RNS in Seed Germination
Following the discovery of the roles of a gaseous free radical NO in mammalian systems, NO involvement in various cellular functions in plants has been demonstrated, in particular those related to signaling in different physiological processes (Lamattina et al., 2003). One of the proposed roles was the function of NO as an endogenous dormancy-breaking agent (Bailly, 2023; Bethke et al., 2004a; Bethke et al., 2004b; Ciacka et al., 2022; Li et al., 2022). Further studies suggested that the role of NO in dormancy breakage might be a consequence of the inhibitory effect on the electron transport proteins, followed by the stimulation of ROS production (Hancock et al., 2011). The endogenous NO generation starts shortly (in a few hours) after seed imbibition upon the depletion of internal oxygen. It coincides with the induction of Pgb, which participates in NO scavenging (Duff et al., 1998; Igamberdiev and Hill, 2004). Seed germination is a unique stage in the lifecycle of plant, where both the signaling and metabolic roles of NO as well as the expression of Pgb are spatially and temporally integrated. The events leading to the formation of NO in germinating seeds are presented in Figure 3.
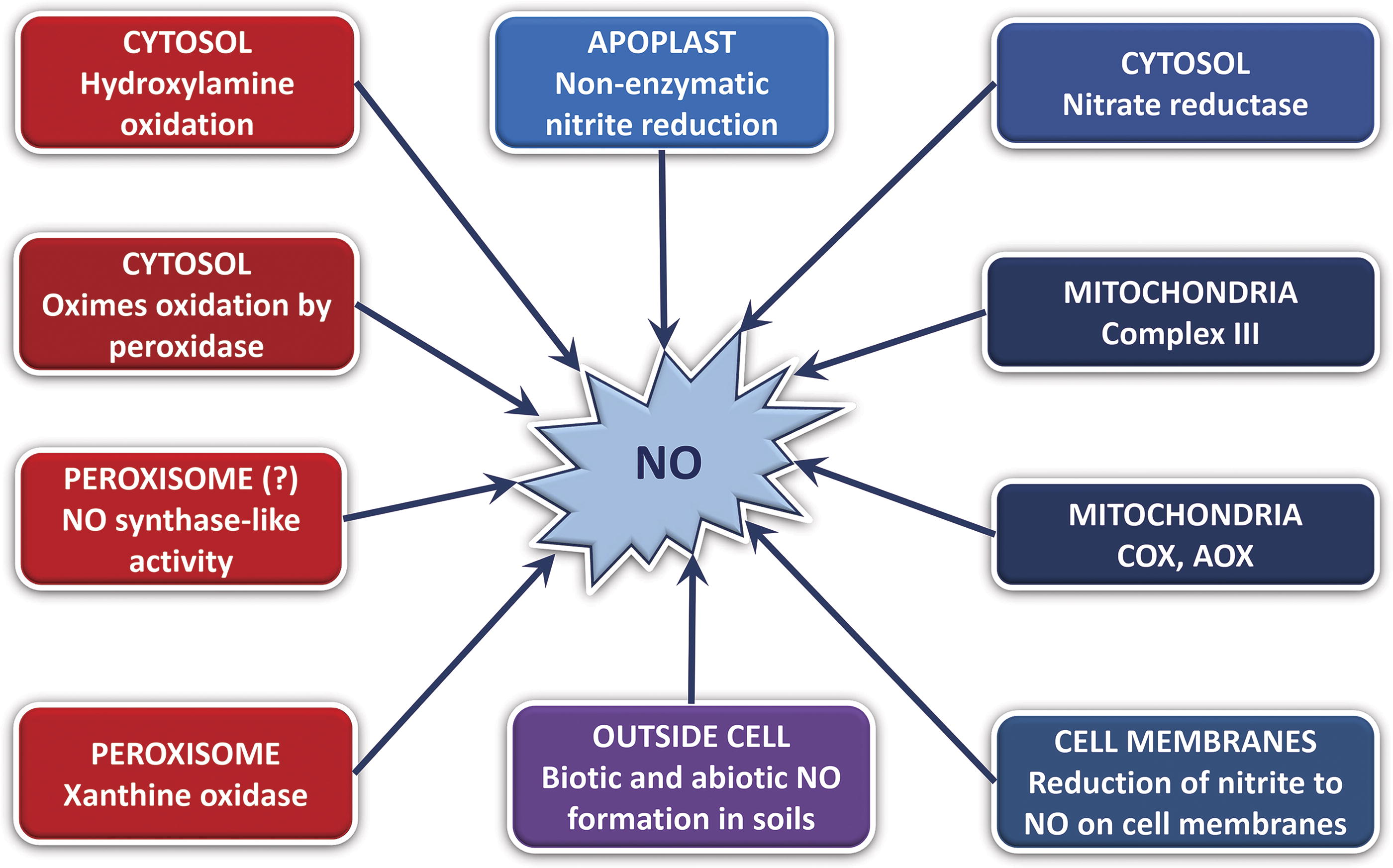
Several reductive and oxidative pathways can generate NO, while the prevailing mechanism is represented by the reductive reaction of nitrite conversion (Gupta and Igamberdiev, 2011). This reaction is readily performed by mitochondria under the conditions of strong limitations of oxygen (Planchet et al., 2005), but the similar mechanism operates in other subcellular compartments with the involvement of redox proteins containing hemes or molybdocofactors (Igamberdiev et al., 2010). These reactions include the side reaction of nitrate reductase with nitrite (Rockel et al., 2002), and the reaction of putative plasma membrane-bound nitrite: NO reductase (Stöhr et al., 2001). Nitrate reductase in the joint operation with the molybdenum-containing amidoxime reducing component (ARC) is able to produce NO from nitrite in Chlamydomonas even at high concentrations of nitrate (Chamizo-Ampudia et al., 2016), however, in Arabidopsis, ARC proteins do not participate in NO production (Maiber et al., 2022).
Nitrate reductase plays a crucial role in NO turnover by supplying nitrite for NO production and also participating by itself in NO generation from nitrite in the side reaction (Rockel et al., 2002). As a result, NO production is enhanced upon germination in nitrate-containing soils. There are many indications that nitrate contributes to seed viability and energy state. Mitochondria in the presence of nitrate retain their ultrastructure and viability (Vartapetian et al., 2003). Nitrate accelerated testa rupture of hedge mustard seeds, modified expression pattern of the cell wall remodeling mannanase proteins (Carrillo-Barral et al., 2013), and induced expression of key genes belonging to the metabolism and signaling of ABA. Apart from the enzymatic reductive pathways of NO synthesis, under the acidic (pH < 6) conditions, nitrite in the form of nitrous acid (HNO2) spontaneously generates NO (Bethke et al., 2004a). Although the low pH requirement for this reaction hinders cytoplasmic operation, this nonenzymatic mechanism remains significant in germinating seeds in the apoplast compartment, which was demonstrated for barley aleurone layers using Isotope Ration Mass Spectrometry (IRMS) detection of NO (Bethke et al., 2004a).
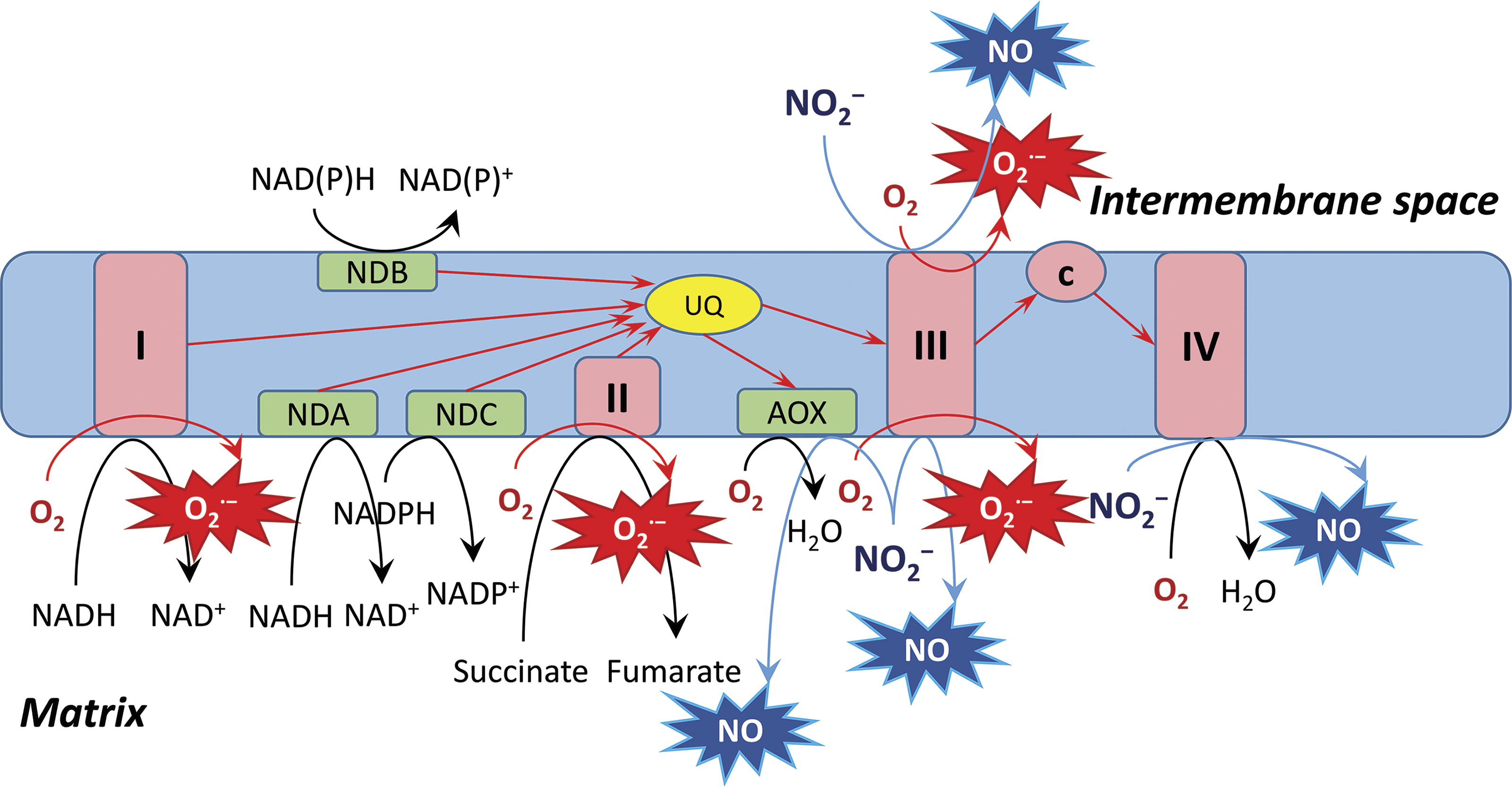
The quantification of NO production by different cell compartments and structures was performed in studies on animal cells. Evidence was provided that mitochondria represent an important NO source due to active electron transport, although other cell membranes were also involved (Feelisch et al., 2008). While NO is generated due to the one-electron leakage from electron transport components such as the mitochondrial Complex III, the formation of this essential diatomic molecule at the sites of cytochrome c oxidase (Gupta and Igamberdiev, 2011) and alternative oxidase (AOX) (Jayawardhane et al., 2020) occurs due to the replacement of oxygen as terminal electron acceptor by nitrite. The leakage of electrons either to oxygen with the formation of O2 •− or to nitrite with the formation of NO is an unavoidable consequence of active electron flux, which increases under the conditions of high intracellular redox potential. The balance between O2 •− and NO formation depends on the redox potential, concentration of oxygen, involvement of noncoupled pathways of electron transport, and its inhibition by the already formed ROS and RNS (Ma et al., 2016a; Ma et al., 2017). The reactions following the formation of O2 •− and NO result in the generation of other ROS and RNS. These include H2O2 produced enzymatically by SOD via dismutation of O2 •−, peroxynitrite (ONOO−) readily generated via interaction of NO with O2 •−, nitrogen dioxide (NO2) formed in the reaction of NO with peroxynitrite, and dinitrogen trioxide (N2O3) generated in the reaction of NO2 with NO (Espey et al., 2002). The nitrogen-derived low molecular oxides are defined as NOx. Their interaction with glutathione and proteins results in the formation of GSNO as well as S-nitrosated and tyrosine-nitrated proteins (Jahnová et al., 2019).
In addition to the reductive pathways of RNS formation, NO can be formed in the oxidative pathways (Fig. 4), the most known of which is the NO synthase reaction. Among other oxidative reactions of NO formation, hydroxylamine oxidation results in ROS production (Rümer et al., 2009). Recently, it was demonstrated that NO can be formed by peroxidases from oximes, such as indole-3-acetaldoxime, a precursor to indole-3-acetic acid (López-Gómez et al., 2024). A putative NO synthase activity was demonstrated in plants (Barroso et al., 1999; Corpas et al., 2009), but the protein was not characterized in sufficient detail, and the gene was not identified (Fröhlich and Durner, 2011). NO synthase was identified in phototrophic prokaryotes Synechococcus (Correa-Aragunde et al., 2018) and in the charophyte alga Klebsormidium nitens (Chatelain et al., 2021), and its sequence was detected in the genomes of green algae (Foresi et al., 2010), but not in higher plants. Oxidative pathways cannot operate with physiologically significant intensity during seed germination due to the limitations in oxygen availability, but after breaking seed testa, they may supply low quantities of NO for signaling functions.
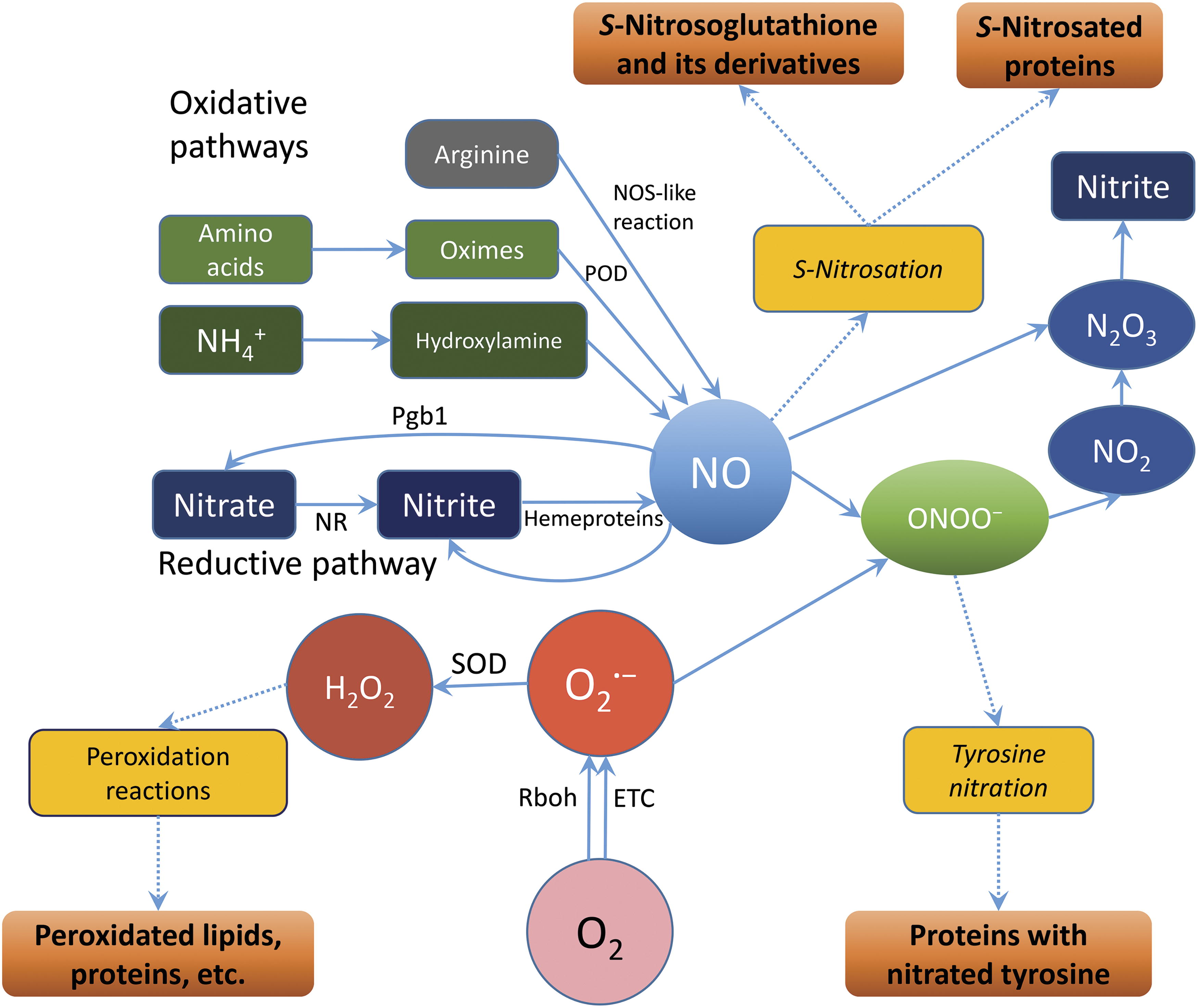
NO acts metabolically to provide oxidation of reducing equivalents and contribute to ATP synthesis (Stoimenova et al., 2007), and it also acts as a signaling agent, where the effect of NO is mediated via the interaction with phytohormones. Evidence has been presented that blue light inhibits germination in dormant wheat (Triticum aestivum L.) grain and that jasmonate and NO counteract this effect by reducing dormancy via interdependent control of ABA level (Jacobsen et al., 2013). The stimulation of ethylene biosynthesis by NO was demonstrated in the course of dormancy breaking in apple embryos (Gniazdowska et al., 2007). In Arabidopsis seeds, the crosstalk between NO, H2O2, and ethylene was demonstrated in dormancy breaking (Lin et al., 2013). Several reports provide evidence for overlapping functions of ROS, NO (mediated by peroxynitrite), UV radiation, light (via phytochrome), GA, ABA, and jasmonate in seed germination events (Andryka-Dudek et al., 2019) and plant development (Beligni and Lamattina, 2000). Collectively, these studies imply that the stimulatory roles of NO, if phytohormone-mediated, are more likely to occur through the GA, as compared to ABA, signal transduction pathways. Additional roles for NO in seed germination include the stimulatory effect of NO on α or β-amylase activity (Beligni et al., 2002; Zhang et al., 2005), and the potential for NO to improve the redox status of seeds (Beligni et al., 2002). Further studies may reveal that NO is capable of circumventing the classical ABA-GA antagonism by preferentially stimulating β-amylase activity, which, in comparison to α amylase, is unresponsive to GA (Zhang et al., 2005).
While NO directly reacts with O2 •− three times faster than SOD, it does not react with H2O2 at physiological pH as shown in Escherichia coli (Brunelli et al., 1995). However, NO can keep H2O2 formation under control, in particular, via activation of the key H2O2 scavenging enzyme ascorbate peroxidase (APX) 1 through S-nitrosation during auxin-dependent root development and abiotic stress (Begara-Morales et al., 2013; Begara-Morales et al., 2019a; Correa-Aragunde et al., 2013). On the contrary, it was shown that S-nitrosation inhibits the enzyme activities of catalase, glycolate oxidase, and two enzymes of ascorbate-glutathione cycle, monodehydroascorbate reductase (MDHAR) and dehydroascorbate reductase (Begara-Morales et al., 2019a). Several mitochondrial enzymes are S-nitrosated, including the subunits of glycine decarboxylase, serine hydroxymethyltransferase, lipoamide dehydrogenase (Møller et al., 2020), the mitochondrial isoform of NAD-malate dehydrogenase (Romero-Puertas et al., 2008), and subunits of Complex I (Burwell et al., 2006). By affecting these enzymes, S-nitrosation modulates the redox potential in mitochondria and influences H2O2 production. González-Gordo et al. (2023) have recently established that one of the molecular forms of lipoxygenase is modulated by NO as well as by hydrogen sulfide (H2S).
Tyrosine nitration was demonstrated to cause strong inhibition of NADP-isocitrate dehydrogenase in pea roots (Begara-Morales et al., 2013), and NADP-malic enzyme 2 (NADP-ME2) in Arabidopsis seedlings (Begara-Morales et al., 2019b). Hence, tyrosine nitration is involved in the regulation of NADPH levels, a key reducing equivalent for biosynthetic processes, antioxidant pathways, and modulator of intracellular redox balance. In Arabidopsis, the activity of mitochondrial Mn-SOD1 was strongly inhibited, whereas the activities of peroxisomal Cu/Zn-SOD3 and chloroplastic Fe-SOD3 were partially inhibited due to tyrosine nitration by peroxynitrite (Holzmeister et al., 2015). Tyrosine nitration was reported for several other enzymes with no information on the modulation of their activity (Begara-Morales et al., 2013). The effects of RNS and ROS on FeS clusters is a well-known phenomenon affecting aconitase and several complexes of the mitochondrial ETC (Hausladen and Fridovich, 1994). The diverse effects of ROS and RNS on plant cell proteins reveal a complex phenomenon of ROS and RNS action, which includes their interaction and leads to either the inhibition of certain enzymes or the activation of others.
Another redox molecule that is involved in redox regulation of seed germination and dormancy is H2S. Existing data suggest that H2S acts either upstream or downstream of the NO signaling cascade, depending on the processes such as stomatal closure or response to abiotic stress, respectively (Corpas et al., 2019). Thus, H2S usually acts together with NO and modifies NO action. It can be directly involved in the regulation of redox state of thiols in proteins (González-Gordo et al., 2020). The physiological activity of a significant subset of cell proteins is modified by the redox state of regulatory thiols. The cellular redox homeostasis depends on the balance between oxidation of thiols through oxygen and ROS, and reduction by thiol-disulfide transfer reactions (Ströher and Dietz, 2006).
Phytoglobin-NO Cycle in Germinating Seeds
Hemoglobin in plants was discovered by Kubo (1939) as a symbiotic leghemoglobin, whose function is related to establishing low oxygen concentration for nitrogen fixation in the nodules of legume plants. It took half a century to discover other Pgbs, which initially were found in the roots of non-nodulating species (Bogusz et al., 1988). Pgb mRNA is undetectable in dry barley seeds, appearing within 2 h imbibition in embryo, aleurone, root, and to a lesser extent in coleoptile (Anderson et al., 1996; Duff et al., 1998). Further studies revealed that Pgbs belong to three separate classes that differ in their phylogenetic origin and physical properties including the affinity for oxygen and other gaseous ligands (Gupta et al., 2011). Dicots contain all three classes, while monocots have the classes 1 and 3. The Pgb initially found in seeds belongs to class 1, however, seeds contain also Pgb2 (in dicots) and Pgb3 (in dicots and monocots). The most established role in plants was determined for Pgb1, which is related to the scavenging and turnover of NO at low oxygen concentrations (Igamberdiev and Hill, 2004). The expression of the Pgb1 gene after seed imbibition, which is accompanied by the drastic decrease in the available oxygen (Benamar et al., 2008), therefore reflects Pgb1 participation in maintaining the energy and redox status within embryo and aleurone layer. In the postgermination period, Pgb1 supports the energy state in oxygen-deprived roots of young seedlings. The ATP levels in barley embryos reach a plateau within 2 h imbibition, which coincides with the increase of the Pgb1 transcripts (Duff et al., 1998).
The sequence of reactions involved in NO turnover defined as the Pgb-NO cycle (Igamberdiev et al., 2005; Igamberdiev and Hill, 2004), is shown in Figure 5. While NO may originate from different processes depicted in Figure 4, the reductive formation from nitrite via nitrate represents the most powerful source of NO in plants, especially under oxygen deficiency (Gupta and Igamberdiev, 2011). In mitochondria, nitrite can be used as an alternative electron acceptor at the levels of Complex IV, Complex III, and AOX (Jayawardhane et al., 2020; Vishwakarma et al., 2018) (Fig. 6). The NO turnover in the Pgb-NO cycle consists of several steps, which include NAD(P)H oxidation, and leads to the maintenance of redox potential in reductive environments. The mitochondrial NAD(P)H oxidation, following the electron transport to nitrite in the absence of oxygen may be coupled to ATP synthesis, with rates comparable to glycolysis (Stoimenova et al., 2007).
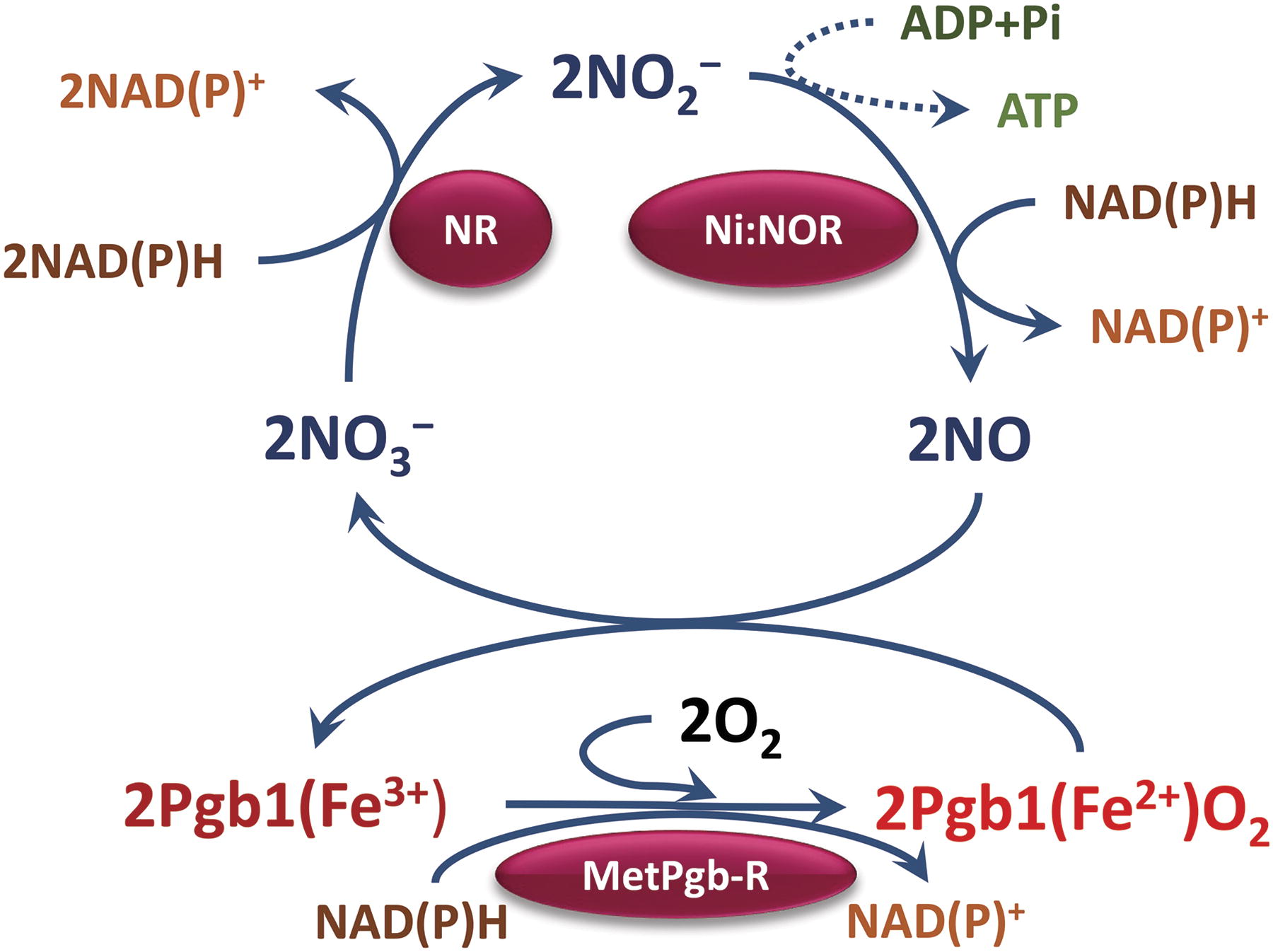
The operation of Pgb-NO cycle results in the diversion of metabolism from fermentation. For germinating barley seeds, it was shown that Pgb and fermentation are regulated reciprocally, with Pgb1 and Pgb3 being induced by GA and glycolytic fermentation by ABA (Nie et al., 2022). On the contrary, the Pgb-NO cycle and the glycolytic fermentation can work together with the Pgb-NO cycle oxidizing the glycolytic NADH. When the glycolytic NADH is oxidized in the nitrate and nitrite: NO reductase reactions, the pool of pyruvate produced at the end of glycolysis can be further converted into alanine, which is one of most accumulated compounds under hypoxic conditions (Fan et al., 1988). Muench and Good (1994) demonstrated that alanine accumulation is caused by the hypoxic induction of alanine aminotransferase. Moreover, Pandey et al. (2019) observed an increased incorporation of 13C into soluble alanine in germinating seeds of chickpea upon treatment with an NO donor. These studies provide evidence that the carbon and the nitrogen metabolic pathways in the bioenergetic system of hypoxic cells cooperate toward the support of energetic needs under oxygen deficiency, in particular, in germinated seeds. This cooperation was more pronounced in hypoxia-tolerant plants (Jayawardhane et al., 2021).
In the Pgb-NO cycle, the NO produced from nitrite is oxygenated to nitrate with oxygen tightly bound to Pgb1, which, in this process, turns into the ferric Pgb(Fe3+) state. For the continuous operation of the Pgb-NO cycle, Pgb1(Fe2+) must be regenerated (Fig. 5). An enzyme possessing ferric Pgb1 (metPgb1) reductase activity is needed to catalyze this reaction (Igamberdiev et al., 2006; Igamberdiev et al., 2010). Recently, it was demonstrated that the ferric form of human cytoglobins 1 and 2 is reduced by the system of cytochrome b5 and cytochrome b5 reductase that provides electrons for NO dioxygenation (Amdahl et al., 2019). It was further shown that changes in the levels of cellular cytochrome b5 and cytochrome b5 reductase protein modulate the process of NO decay (Ilangovan et al., 2021). The same mechanism exists for the neuroprotective neuroglobin, which exhibits an NO dioxygenase reaction in a close-fitting ternary complex with membrane-bound NADH-cytochrome b5 oxidoreductase and cytochrome b5 (Gardner, 2023).
The nature of plant methemoglobin reductase is less clear, although for leghemoglobin such activity was revealed a long time ago (Topunov et al., 1980). It was demonstrated that in barley roots, the reduction of ferric Pgb1 is facilitated by MDHAR (Igamberdiev et al., 2006). Since MDHAR is reactive with cytochrome b5 and has common properties with NADH-cytochrome b5 reductase (Hossain and Asada, 1985), it is possible that the reaction in situ is mediated by cytochrome b5 or another redox protein in a similar manner to the reaction demonstrated for cytoglobin and neuroglobin. In regard to the reduction of metPgb, it is important that cytochrome b561 : ascorbate-mediated transmembrane electron transport was established (Asard et al., 2013), and the corresponding redox mechanism was characterized (Lu et al., 2014). Gradogna et al. (2023) demonstrated that the tonoplast cytochrome b561 is a transmembrane ascorbate-dependent MDHAR. Free reduced flavins may reduce ferric Pgb in the nucleus (Sainz et al., 2013). Jokipii-Lukkari et al. (2016) showed the metPgb1 reductase activity of the cytosol-located ferredoxin NADP+ oxidoreductase. It lacks the plastid presequence and binds to Pgb1 to interact as NO dioxygenase.
Seeds contain all necessary components for the operation of the Pgb-NO cycle during seed germination. The involvement of the cycle in the maintenance of the redox and energy balance in germinating seeds may explain how, at early stages of germination, high metabolic rates and ATP concentrations are achieved in the hypoxic environment of the seed. In particular, the Pgb-NO cycle may be the main mechanism providing energy for the anaerobic germination of rice seeds, with AOX producing a significant part of NO (Kumari et al., 2022).
The Crosstalk Between Redox State, Phytohormones, and ROS
The increase in redox state under the hypoxic conditions during early imbibition prior to seed coat rupture facilitates ROS generation via Rboh and other electron transport processes. The effects of phytohormones that depend on redox potential and ROS generation mediate redox control of seed germination. Dormancy phenotype-associated interaction of signaling pathways in the embryo during early imbibition of seeds is illustrated in Figure 7. The signal transduction sequence of events revealed through functional genomic and proteomic studies of seeds results in dormancy release and induction of germination. Major events of phytohormone signaling are regulated by the redox state of the cell via changing the level of NADPH. The latter catalyzes the formation of O2 •− by Rbohs, which leads to the production of H2O2 by SOD. The level of extracellular H2O2 is regulated by antioxidant enzymes such as peroxidases (Prxs) and by aquaporins (AQPs) (Bienert et al., 2007) that facilitate the entering of H2O2 into plant cells via plasma membrane and propagation of the signal through the cytoplasm. Phospholipase D (PLDα) generates an essential lipid second messenger, phosphatidic acid (PA), which is involved in the alleviation of ABA-induced inhibition of seed germination and seedling growth as a mediator in ABA signaling network (Katagiri et al., 2005). A direct interaction of PA with Rboh stimulates its activity and promotes ROS production (Zhang et al., 2009), whereas NO-modulated S-nitrosation abolishes the ability of Rboh to generate O2 •− (Yun et al., 2011). Moreover, PLDα was shown to be activated by H2O2 (Jakubowicz et al., 2010; Yamaguchi et al., 2004).
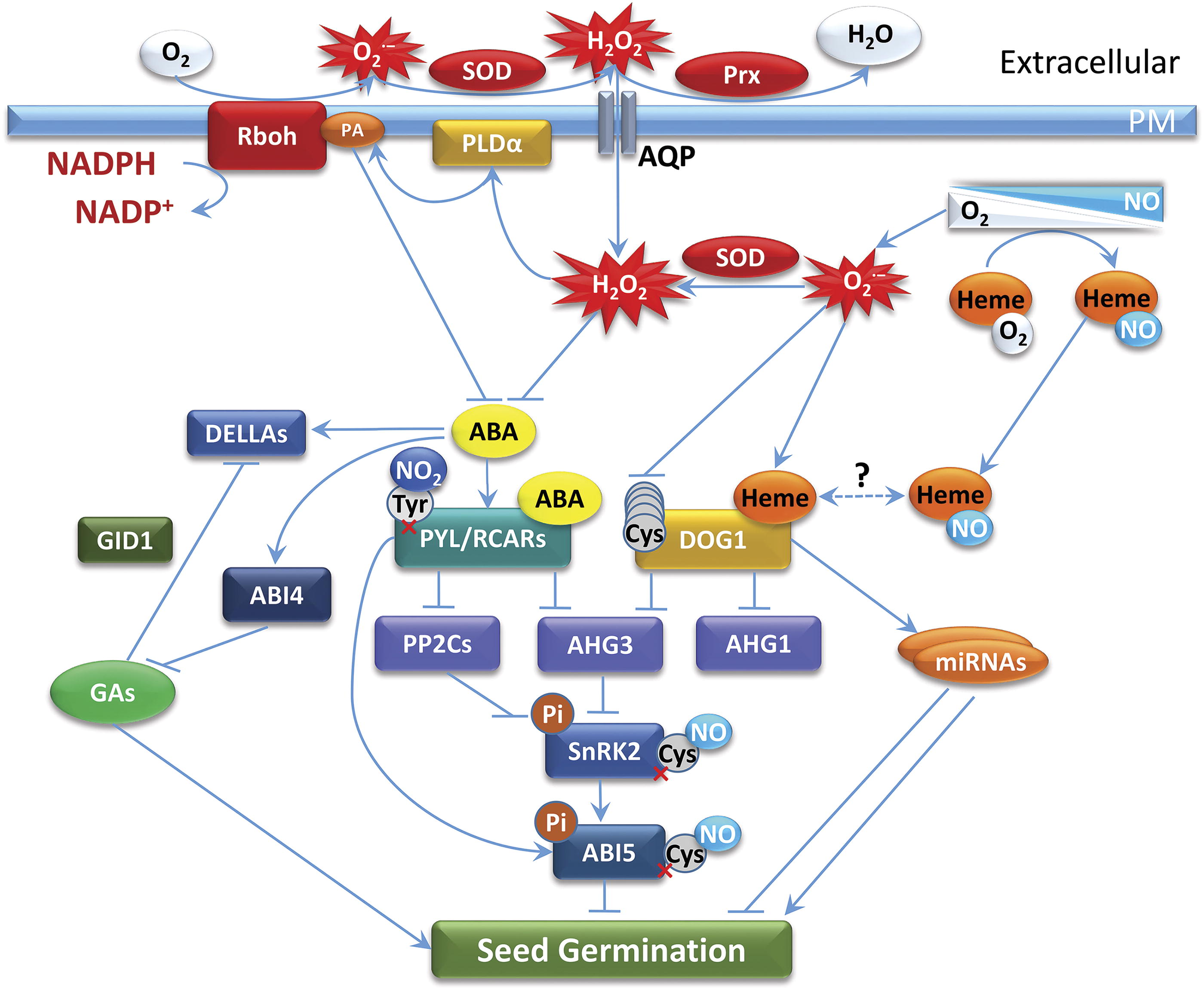
Since H2O2, NO, phytohormones, and microRNAs (miRNAs) (Willmann et al., 2011) are central to directing seed maturation program to the establishment of seed dormancy and repression of this program after germination, it is essential to understand the interplay between ROS, RNS, phytohormones, and miRNA in seed cells. The signaling and interaction of ABA, GA, and auxin phytohormones that control seed dormancy and germination were recently shown to be mediated by miRNAs in seeds from different species (Bai et al., 2017; Tognacca and Botto, 2021). The DELAY OF GERMINATION1 (DOG1) heme-binding protein, which is a master regulator of primary seed dormancy (Nishimura et al., 2018; Nonogaki, 2019), influences the levels of miRNA through the control of miRNA processing (Huo et al., 2016). The depth of dormancy established during seed maturation is controlled by DOG1, which is subject to extensive transcriptional and post-transcriptional regulation by alternative splicing, antisense transcripts, alternative polyadenylation, and epigenetic regulation of chromatin structure (Tognacca and Botto, 2021, and references therein). Oxidative processes and redox post-translational modifications of DOG1 protein were suggested to result in inactive DOG1 during the after-ripening mediated dormancy loss (Nakabayashi et al., 2012; Née et al., 2017). The crosstalk between ABA- and DOG1-dependent pathways for dormancy control (Fig. 7) takes place via interaction with downstream target Protein Phosphatase type 2CA (PP2CA), encoded by ABA HYPERSENSITIVE GERMINATION3 (AHG3). The interaction of DOG1 bound to AHG1 with heme-bound DOG1 represents a unique regulatory system that results in a positive regulation of seed dormancy independently of Pyrabactin Resistance, Pyrabactin Resistance-Like, and the Regulatory Component of ABA Receptors (PYR/PYL/RCAR) (Nishimura et al., 2018). This alternative ABA-independent regulatory pathway has an important capacity for the integration of environmental signals and physiological redox conditions to counteract ABA-dependent regulation (Nishimura et al., 2018). Similar to the class 1 Pgb, the heme in DOG1 heme-protein complex exhibited characteristics of a typical hexacoordinate state and, therefore, is capable of reversible ligand binding (Nishimura et al., 2018). The heme-bound DOG1 form possesses the ability to sense the redox changes in ROS and NO through heme-dependent effects. Under hypoxic conditions with high NO to oxygen ratio (Gupta et al., 2011), the formation of heme-NO adduct could allow for heme-bound DOG1 function to be regulated based on different affinities for heme versus heme-NO and the availability of free heme. The presence of multiple Cys residues in DOG1 protein sequence can facilitate redox sensing via ROS- and RNS-driven post-translational redox modifications (Nakabayashi et al., 2012). The inhibition of ABA accumulation by H2O2 (Bailly et al., 2008) leads to changes in the miRNA-dependent systems integrated by the interaction with DOG1 (Huo et al., 2016).
DELLA proteins positively regulate the expression of several proteins involved in dormancy control (Cao et al., 2006). The GA receptor GA INSENSITIVE DWARF1 (GID1) inhibits the function of DELLA proteins, thus releasing dormancy (Gazara et al., 2018; Ge and Steber, 2018). Phosphorylation by Sucrose Nonfermenting-1-Related Protein Kinase 2 (SnRK2) activates transcription factor ABSCISIC ACID INSENSITIVE 5 (ABI5) and directs the expression of the genes involved in the promotion of dormancy. The ABI4 transcription factor inhibits GA synthesis and thus promotes seed dormancy (Shu et al., 2016). Signaling via NO-induced post-translational protein modifications was shown to counteract ABA signaling processes upon the induction of seed germination. S-nitrosation inactivated SnRK2 and rendered ABI5 susceptible to protein degradation (Albertos et al., 2015; Wang et al., 2015), whereas RNS-triggered tyrosine nitration resulted in ubiquitin-mediated proteolysis of PYR/PYL/RCAR ABA receptors (Castillo et al., 2015). Moreover, transcriptional control by the N-end rule pathway contributed to NO-dependent alterations in the accumulation of ABI5 (Albertos et al., 2015).
ABA and H2O2 have an antagonistic mode of action, but GA and H2O2 have a synergistic effect in the control of the germination process (Ishibashi et al., 2015). The presented scheme (Fig. 7) shows the complex mechanism of interaction of redox potential, ROS, and phytohormones in inducing seed germination. Elevation of redox potential results in the formation of ROS that trigger seed germination, as it was suggested in the concept of “oxidative window” (Bailly et al., 2008; see also Dębska et al., 2013), and in the changes in turnover rates of glutathione and ascorbate redox pairs. It also establishes the “nitrosative door” for seed dormancy alleviation (Krasuska et al., 2015) and initiates phytohormone interactions at the level of promoters of corresponding genes regulating the germination process (Nonogaki, 2019). Thus, the role of redox state changes in seed germination is revealed at the levels of ROS and RNS production, turnover of redox couples, and promoter activation via changing ABA, GA, and ethylene levels.
The Crosstalk Between Redox State, Phytohormones, and RNS
In Section 4, we discussed the role of the Pgb-NO cycle in NO turnover and through this in the regulation of the redox state of cells. This function itself is an important mechanism for balancing the redox state and energy level in seeds. The Pgb-NO cycle operates with the involvement of the cytosol and mitochondria and represents an alternative to fermentation pathways that keeps redox potential under control (Igamberdiev and Hill, 2004) and provides a limited amount of ATP (Stoimenova et al., 2007).
Already in early studies of phytoglobins, it was revealed that a significant part of the pool of class 1 Pgb is located in the nucleus (Seregélyes et al., 2000). This means that NO scavenging and turnover involve not only the cytosolic compartment but also the nuclear compartment of cells. Further studies confirmed the presence of Pgb1 in the nucleus and cytosol (Hebelstrup et al., 2008), as well as in chloroplasts (Rubio et al., 2019), while Pgb2 is likely located exclusively in the cytosol (Godee et al., 2017). It was shown that the promoters of several genes are under the control of NO (Xu et al., 2020), and thus the presence of Pgb1 in the nucleus regulates NO concentration in their vicinity. Expression of Pgb1 results in drastic changes in the profiles of gene expression as well as of nuclear proteins. This leads to modifications in signal transduction, in particular, via a dramatic reduction in the number of kinases induced, but also through the turnover of other signaling molecules such as phytohormones, polyamines, and Ca2+ (Andrzejczak et al., 2020a). Upon Pgb1 overexpression, essential changes in the levels of histones have been observed, indicating that chromatin restructuring plays a role in the conditions of oxygen deficiency, and these changes are under the control of NO production and scavenging (Andrzejczak et al., 2020b).
The observed changes upon overexpression and downregulation of Pgb1 are associated with hormone profiles in plants and, in particular, in germinating seeds. Rubio et al. (2019) showed that plants overexpressing Pgb1 show higher constitutive expression of the ABA-responsive genes Responsive to ABA (RAB18), Responsive to Dehydration (RD29A), and Highly ABA-Induced 2 (HAI2), and are more tolerant to dehydration. The authors conclude that Pgb1 through the modulation of NO levels interacts with ABA, which mediates several essential physiological processes including the response to desiccation. Hartman et al. (2019) demonstrated ethylene-mediated NO depletion in the process of preadaptation of plants to the conditions of hypoxia. The formation of ethylene as a hypoxic response leads to the induction of Pgb1. The crosstalk between Pgb and ethylene was demonstrated in numerous studies, for example., in maize cell cultures by Manac’h-Little et al. (2005), in tobacco plants (Zafari et al., 2022), and in maize embryos (Kapoor et al., 2018). In relation to seed germination, the crosstalk between ethylene and NO is discussed in the review of Manrique-Gil et al. (2021).
NO can stimulate seed germination in the light via its regulation of the phytochrome system. Upon the illumination by red light, phytochrome B moves from the cytoplasm to the nucleus and promotes expression of the 26S proteasome pathway leading to phytochrome interacting factor 1 (PIF1) degradation (Leon and Lozano-Juste, 2011). NO accumulation under hypoxic conditions under seed coat during seed germination results in inhibiting PIF1 transcription and promoting the interaction of PIF1 with the factor LONG HYPOCOTYL IN FAR-RED (HFR1), which further reduces the transcriptional activity of PIF1, leading to its weakened inhibitory effect on seed germination. SOMNUS gene encoding a CCCH-type zinc finger protein negatively regulating light-dependent seed germination, represents a direct target gene downstream of PIF1. It affects the expression levels of the genes responsible for seed germination in the ABA and GA signaling pathways (Zhang et al., 2023). The transcription factors that bind to both EXPANSIN (EXPA) promoter fragments and DELLA GA-response regulators are targeted by NO and the phytochrome interacting TF PIL5. This molecular interaction network therefore directly links the perception of an external environmental signal (light) and internal hormonal signals (GA and NO) with downstream germination-driving EXPA gene expression (Xu et al., 2020).
It has been established that NO induces ABA catabolism and inhibits ABA synthesis. NO accumulation leads to the increase of GA biosynthesis and the suppression of ABA biosynthesis in aleurone layers of Arabidopsis seeds (Bethke et al., 2007). This regulation may take place at the level of regulation of the enzyme nine-cis-epoxycarotenoid dioxygenase (NCED) catalyzing the rate-limiting step of ABA biosynthesis (Wang et al., 2020; Xu and Cai, 2017). In the pathway of ABA catabolism, NO upregulates the expression of ABA 8′-hydroxylase encoded by CYP707A2 and thus induces the degradation of ABA (Liu et al., 2009). Another study shows that NO in potato tubers promotes the expression of the ABA catabolic gene StCYP707A1 and inhibits the ABA biosynthesis-related gene StNCED1 resulting in the reduction of ABA content and alteration of the ABA-GA balance (Wang et al., 2020).
While NO production leads to the suppression of ABA accumulation during seed germination, it increases GA biosynthesis by inducing the expression of gibberellin 3-oxidase genes (GA3ox1 and GA3ox2) (Bethke et al., 2007). NO also stimulates ethylene production, which promotes seed germination. The mechanism of NO effect on ethylene biosynthesis may include the regulation of expression of the genes encoding the enzymes of ethylene biosynthesis 1-aminocyclopropane-1-carboxylic acid (ACC) synthase and ACC oxidase and of the ethylene responsive factors (ERFs) (Sami et al., 2019). Ethylene biosynthesis genes encoding ACC synthase, ACC oxidase, and ERFs are induced during oxygen deficiency and correlate with NO levels (Zafari et al., 2022). The induction of ACC synthase and ACC oxidase by NO was observed in hypoxic roots during aerenchyma formation (Wany et al., 2017). A similar mechanism was suggested in the regulation of somatic embryogenesis (Mira et al., 2015). The removal of dormancy in apple seeds is also related to the stimulation of ACC oxidase activity by NO leading to enhanced ethylene emission (Gniazdowska et al, 2010). NO decreased the sensitivity of dormant caryopses to exogenous ABA and decreased the embryos’ ABA content before germination was completed (Kępczyński et al., 2023). Thus, seed germination involves the crosstalk between ABA, GA, ethylene, and NO (Arc et al., 2013). Figure 8 shows how NO regulates seed germination by integrating multiple signaling pathways involving ABA, GA, and ethylene.
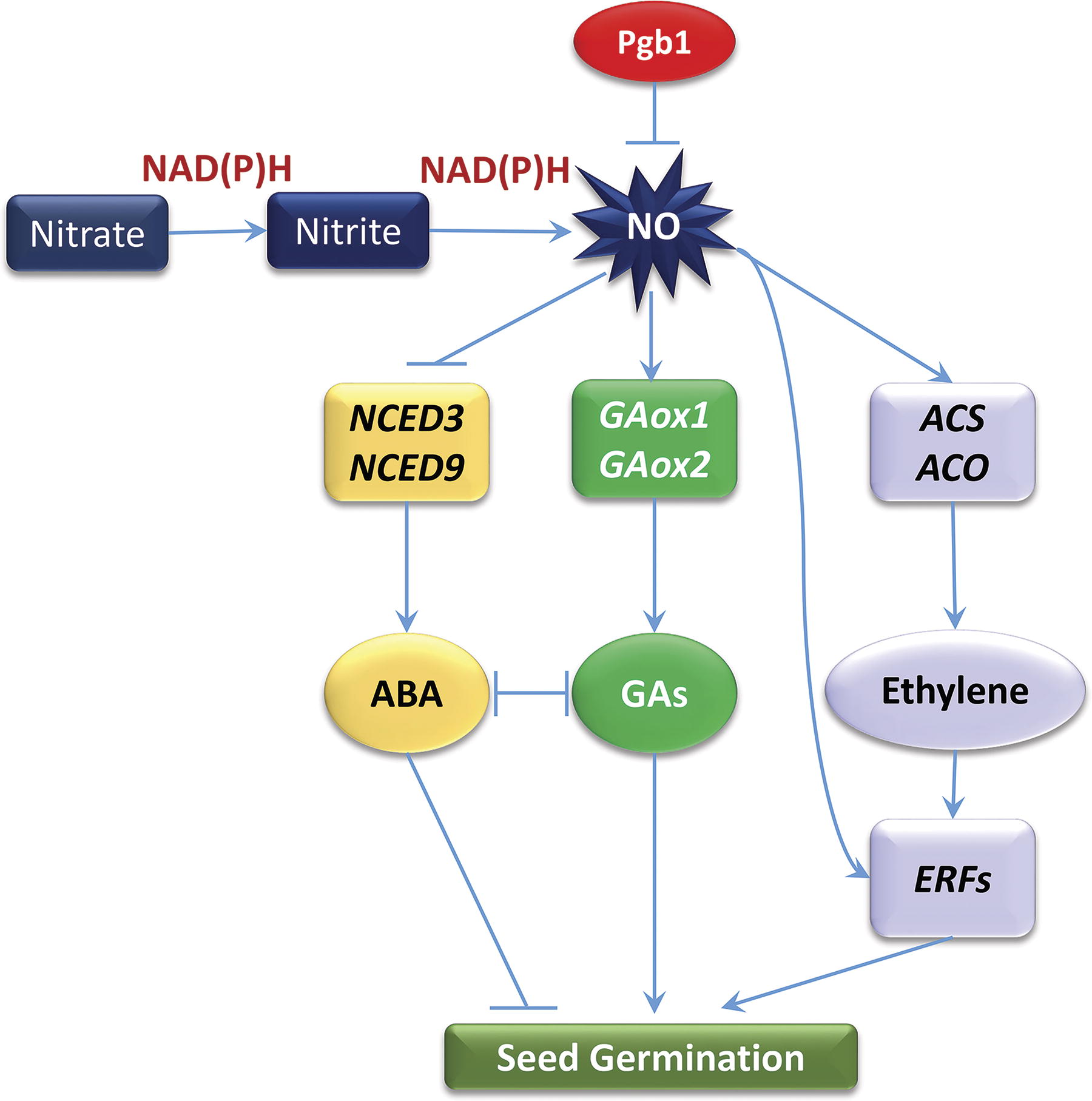
The increased levels of NO can also stimulate auxin biosynthesis via NO-dependent repression of the basic helix-loop-helix (bHLH) transcription factor MYC2 gene in plants differentially expressing Pgb2 (Elhiti et al., 2013). NO also inhibits the expression of Phytochrome Interacting Factor 4 (PIF4), which is established in the plant cultures differentially expressing Pgb2 (Mira et al., 2023). The PIF4 inhibition leads to the induction of several auxin biosynthetic (CYP79B2, AMI1, and YUCCA 1, 2, and 6) and response (ARF5, 8, and 16) genes. Auxin responses, which are mediated by ARF10 and 17, are regulated through NO, in a PIF4-independent fashion (Mira et al., 2023). This is essential in the seed germination processes that are light-regulated via phytochrome.
A close link between NO-mediated PTMs and the proteasomal degradation of proteins via ubiquitylation has been demonstrated (Pande et al., 2022). In some cases, ubiquitin-mediated proteasomal degradation of target proteins is accompanied by a NO-mediated post-translational modification on them, while in other cases NO-mediated PTMs can regulate the ubiquitylation of the components of ubiquitin-mediated proteasomal machinery for promoting their activity (Mata-Pérez et al., 2023). Another pathway that links NO signaling with the ubiquitin-mediated degradation of proteins is the N-degron pathway. It plays an important role in dormancy release and seed germination (Manrique-Gil et al., 2021). The proteolytic N-degron pathway is essential for ethylene-mediated reprogramming of seed proteomes during germination (Wang et al., 2021). Phytohormone signaling plays a major role in inducing the processes of ubiquitinylation (Černý et al., 2016), leading to the regulation of vital physiological processes such as seed germination.
ROS/RNS in Aerobic Metabolism after Radicle Protrusion
The breakage of the seed coat and radicle protrusion initiates the postgerminative growth. It results in a drastic increase in oxygen supply, which strongly enhances oxygen-dependent processes such as mitochondrial respiration and peroxisomal oxidation. The latter provides the mobilization of stored fats via β-oxidation followed by the conversion of acetyl-CoA to succinate in the glyoxylate cycle and the metabolism of succinate to carbohydrates in gluconeogenesis (Igamberdiev and Lea, 2002). The autonomy of seed metabolism, which is a key factor in higher plant evolution, is provided by the acquisition of peroxisome-specific proteins (Palma et al., 2009). Mutant analysis demonstrates key roles for glyoxysomes (specialized peroxisomes) in plant reproduction, seed development and germination, and postgerminative seedling establishment (Ma et al., 2016b).
After radicle protrusion, the production of NO in the reductive pathways is ceased due to the availability of oxygen, which also correlates with a decrease in Pgb expression (Duff et al., 1998) (Fig. 2). NO may be produced in oxidative pathways, in particular in glyoxysomes, and participate in signaling functions, however, we do not have sufficient information on the role of NO during this stage of plant development (Corpas et al., 2013). On the contrary, the production of H2O2 in flavin-dependent oxidation of fatty acids and other compounds increases drastically, and the role of ROS at this stage again becomes prevailing over RNS. Peroxynitrite is actively formed in the interaction between NO and O2 •− (Corpas et al., 2019). Peroxisome becomes a major source of ROS and RNS after radicle protrusion (Corpas et al., 2013).
Catalase, together with APX and other peroxidases, keeps H2O2 at physiological levels (Eising et al., 1990). Catalase is regulated post-translationally by NO and H2S (Palma et al., 2020). When the storage compounds in seeds are consumed, ROS may play a primary role in the processes related to programmed cell death (PCD), resulting in the degradation of seed tissues after fulfilling their functions (Bailly et al., 2008). Activation of nucleases and PCD are associated with the function of peroxisomes during germination (Fath et al., 1999). The formation of ROS facilitates PCD in aleurone through the signaling pathways involving GA, while ABA suppresses PCD (Fath et al., 2002).
The metabolic interaction between plastids, mitochondria, and peroxisomes provides the fine tuning of metabolism to optimize seedling growth (Wrobel et al., 2023). Peroxisomes actively oxidize NADH formed in the glyoxylate cycle via the MDHAR system (Bunkelmann and Trelease, 1996). NADPH is generated in peroxisomes by NADP-isocitrate dehydrogenase and glucose-6-phosphate dehydrogenase isoforms (Corpas and Barroso, 2018). These enzymes are regulated via post-translational modifications caused by NO and H2S (Corpas et al., 2019). Cao et al. (2022) showed that the deficiency of NADP-isocitrate dehydrogenase in seed peroxisomes impairs metabolic processes and leads to suppression of growth. This may occur via the inability to support the redox potential and impairment of ROS signaling.
Conclusions and Perspectives
Changes in redox potential during seed germination starting from the imbibition govern major events such as dormancy release and growth and differentiation of tissues. The increase in redox state result in the intensive production of ROS (O2 •−, H2O2, etc.) and RNS (NO and its derivatives). ROS and RNS accumulation leads to the modulation in phytohormone signaling pathways, in particular via regulation of the proteins involved in the synthesis of ABA, GA, and ethylene, as well as of their degradation. This regulation takes place at the transcriptional, translational, and post-translational levels. Intensive ROS accumulation during imbibition is a critically important condition for breaking dormancy. It is achieved via O2 •− production by Rboh and several electron transport proteins, while O2 •− is converted by SOD to H2O2 having a much longer half-life than O2 •− and representing the main ROS regulating seed germination. While the intensive accumulation of NO and ROS during imbibition is a critically important condition for breaking seed dormancy, their role in supporting energy metabolism and controlling the intracellular redox balance in germinating seeds requires further investigation.
Footnotes
Authors’ Contributions
N.V.B. and A.U.I. wrote the article and prepared the figures.
Author Disclosure Statement
The authors have no conflicts of interest.
Funding Information
The research leading to these results has received funding from the
