Abstract
The pathogenesis and progression of hypoxic–ischemic encephalopathy (HIE), a major cause of severe neurological disability and mortality in the perinatal period, are shaped by the interplay of multiple processes, including inflammation, oxidative stress, and excitotoxicity. We conducted a longitudinal study to determine biomarkers of oxidative stress and inflammation in noninvasive urine samples of newborns with moderate/severe HIE (N = 51), employing liquid chromatography–mass spectrometry. We noted that levels of several biomarkers of oxidative stress increased over time, demonstrating the ongoing propagation of oxidative injury. Prostaglandins, in contrast, showed a decreasing trend in their concentration profiles over time, which probably reflects their mediation in pathogenic mechanisms, including the inflammatory response. Statistically significant differences in the levels of oxidative stress of neonates with distinct brain lesion patterns, as detected with magnetic resonance imaging (MRI), were observed, revealing an increase of lipid peroxidation biomarkers in newborns with cerebral lesions (MRI score of 1 compared with scores of 0 and 2). Moreover, a gender-dependent study showed no statistically significant differences in biomarker concentrations between male and female infants. Our observation leads to the hypothesis that monitoring of noninvasive lipid peroxidation biomarkers could aid in diagnosis and prediction of long-term outcomes as a complementary tool to standard exploration. Antioxid. Redox Signal. 35, 1467–1475.
Color images are available online.
Innovation
Oxidative stress and inflammation are known to be deeply involved in the pathogenesis of cerebral lesions in newborns after a hypoxic–ischemic insult. However, the determination of specific related biomarkers for diagnosis and prediction of long-term outcomes as a complementary tool to standard evaluation is not yet exploited in newborn critical care. Our findings demonstrate the feasibility and potential usefulness of continuous monitoring of biomarkers in noninvasive biofluids from newborns with hypoxic–ischemic encephalopathy undergoing therapeutic hypothermia as oxidative stress and inflammation biomarkers may provide clinically relevant additional information on the patient's status and progress.
Introduction
Approximately 10
After a hypoxic–ischemic insult, brain cells initially undergo impaired oxidative metabolism, followed by reperfusion injury that leads to death of neuronal cells. Neuronal death is triggered by a cascade of events, including reactive oxygen species (ROS) generation. Generation of ROS is involved in physiological reactions during the fetal-to-neonatal transition, and it has been demonstrated that ROS generation upregulates the expression of specific genes whose end products are beneficial for postnatal adaptation. However, triggered by hypoxia and ischemia, ROS production rapidly increases and overwhelms antioxidant defenses and ultimately leads to development of specific conditions. Hence, the beneficial and toxic effects of O2 administration for newborn therapy need to balance to prevent cellular and tissue damage. Both, hypoxia and reoxygenation generate excessive amounts of ROS and, subsequently, oxidative stress.
Several studies illustrate molecular and metabolomic changes associated with asphyxia, duration of hypoxia, and resuscitation with different oxygen concentrations. At high concentrations, ROS modify or degenerate a range of macromolecules, such as lipids, proteins, and polysaccharides, and can damage DNA and RNA and lead to a cascading inflammatory response. The neonatal brain with a high concentration of polyunsaturated fatty acids (PUFAs), high rate of oxygen consumption, low concentration of antioxidants, and high levels of iron is especially vulnerable to excessive amounts of ROS. Inflammation is considered an important contributor in the pathophysiology of cerebral hypoxic–ischemic (HI) injury. The initial inflammatory response to HI produces a secondary neuronal injury, followed by an anti-inflammatory response toward resolution of inflammation.
The state-of-the-art reference method for early characterization of structural damage of the gray matter (i.e., the basal ganglia, thalamus, and cortex) and, to a lesser extent, infarction in white matter is magnetic resonance imaging (MRI) acquired within 5–7 days after birth. The prognostic ability of MRI obtained in the neonatal period to determine neurodevelopmental outcomes in childhood has been demonstrated (8). Currently, literature providing evidence of the relationship between oxidative stress biomarkers and the degree of brain damage is scant and based on the comparison of HIE versus healthy control infants and determination in blood samples. In preterm infants, a reference range for lipid peroxidation by-products in urine samples was established, and preterm babies who later developed bronchopulmonary dysplasia, a disease associated with oxidative stress, showed significantly higher urinary elimination of isofurans in analytical determination performed during the first days after birth (5).
Access to an early and accurate biomarker-guided diagnosis of neonatal HIE could be a powerful ally to reduce neonatal mortality, morbidity, and degree of disability, through a personalized adjustment of treatment options. In the present study, we monitored urinary oxidative biomarker levels in a cohort of term newborns with moderate or severe HIE before, during, and after therapeutic hypothermia (TH). We compared oxidative stress levels between neonates with different degrees of brain injury versus normal MRI outcomes following a standardized scoring system (8) and carried out a gender-dependent study of the detected oxidative stress biomarkers.
Results
A subcohort of 51 newborns from the HYPOTOP trial recruiting infants with moderate or severe HIE qualifying for TH was included in this study. Relevant clinical and demographic characteristics are summarized in Table 1. A total of 22 patients (43%) had normal (score 0) outcomes and 29 patients (57%) had pathologic (score 1, 2, or 3) MRI outcomes. There was no statistically significant difference among both groups (i.e., normal and pathologic MRI outcomes) regarding gestational age, gender, and biometric characteristics (Table 1). From this population, 24 (47%) patients were males and 27 (53%) females, and there was no statistically significant difference between recorded clinical parameters between genders.
Clinical and Demographic Data of Newborns Included in the Study
Data are expressed as mean ± SD for parametric variables, number of cases (percentages) for categorical variables, and median (IQR 0.25%–0.75%) for nonparametric variables. To compare between the two groups, Student's t-test, chi-square test, and Wilcoxon rank-sum test (α = 0.05) were employed, respectively. For four newborns, no information on the degree of injury was available.
HIE, hypoxic–ischemic encephalopathy; IQR, interquartile range; MRI, magnetic resonance imaging; SD, standard deviation.
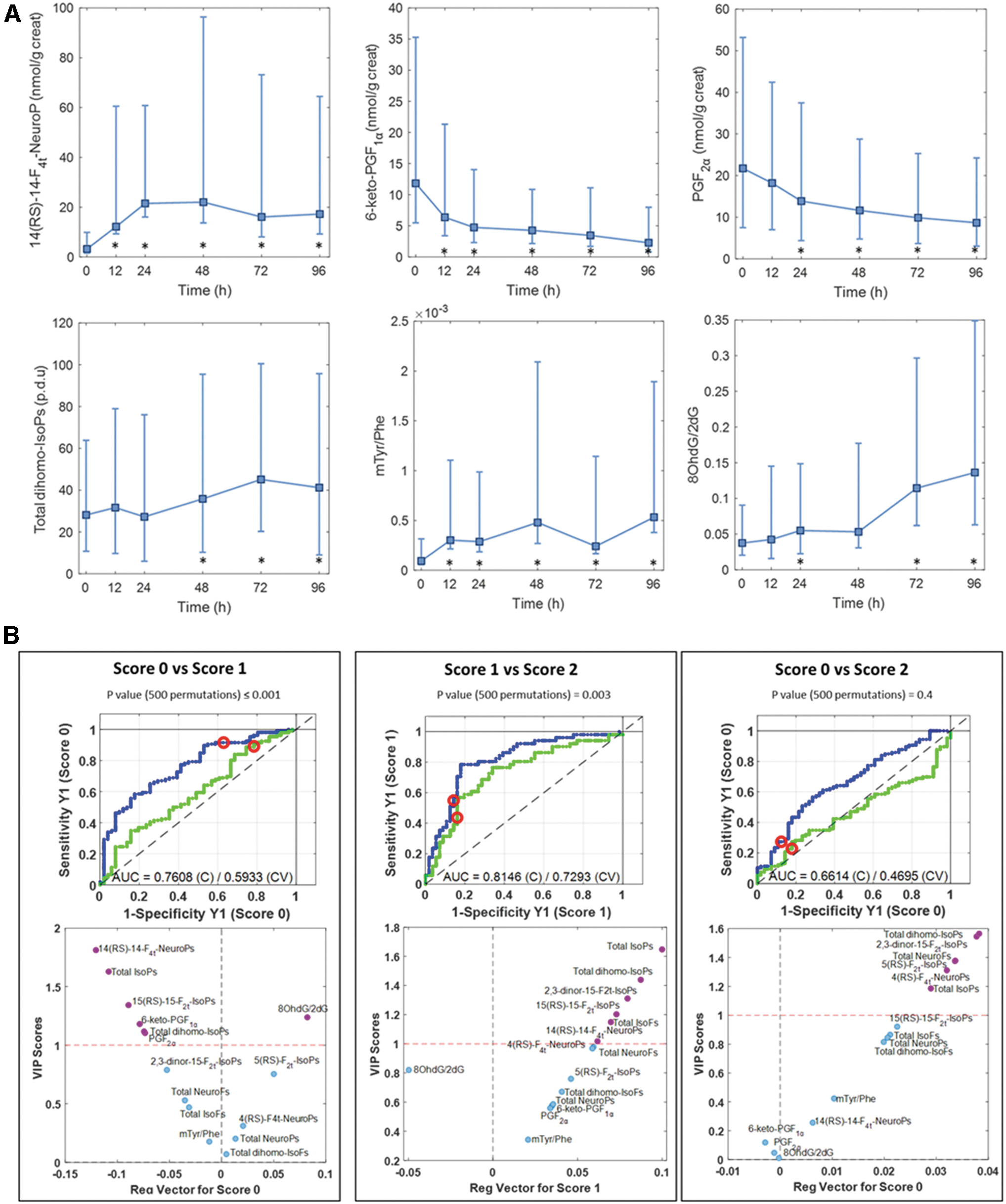
A total of 234 urine samples were collected before initiation of TH (N = 28) and at 12 (N = 37), 24 (N = 41), 48 (N = 47), 72 (N = 42), and 96 h (N = 39) after initiation of TH. Serial collection of urine samples enabled the longitudinal assessment of the evolution of urinary oxidative stress biomarkers before and during cooling and upon rewarming. Of the measured lipid peroxidation compounds, five resolved isoprostanoids (IsoPs) and total intensities of six relative ultra-performance liquid chromatography–tandem mass spectrometry (UPLC-MS/MS) signals characteristic of lipid peroxidation products (i.e., total parameters) were detected (i.e., levels above the limit of quantification [LOQ]) in 50% of samples. Two of five measured prostaglandins (PGs) were detected, in addition to one biomarker of oxidative damage to DNA and one of four biomarkers of oxidative damage to proteins. Median value and interquartile range of each determined parameter as well as the detection frequency are shown in Table 2. No significant trend in the levels of 5(RS)-5-F2t-IsoP, 15(RS)-15-F2t-IsoP, 2,3-dinor-15-F2t-IsoP, and 4(RS)-4-F4t-NeuroP, as well as total IsoPs, IsoFs, NeuroPs, NeuroFs, and dihomo-IsoFs, was observed during the study period. However, levels of 14(RS)-14-F4t-NeuroP, 6-keto-PGF1α, PGF2α, sum of 7-series dihomo-IsoPs, m-Tyr/Phe, and 8OHdG/2dG presented significant changes compared with the initial levels (Fig. 1A). The levels of biomarkers of oxidative stress, including IsoPs, and biomarkers of damage to DNA and proteins increased over time, while PG levels decreased.
Acquisition Parameters and Main Figures of Merit of the Liquid Chromatography–Tandem Mass Spectrometry Methods
Median and IQR of detected analytes in urine samples.
CE, collision energy; IsoFs, isofuranoids; IsoPs, isoprostanoids; LOQ, limit of quantification; NeuroFs, neurofuranoids; NeuroPs, neuroprostanoids; Phe, phenylalanine; p-Tyr, para-tyrosine; RT, retention time.
Urinary biomarker levels of 5(RS)-5-F2t-IsoP, 2,3-dinor-15-F2t-IsoP, 15(RS)-15-F2t-IsoP, 4(RS)-4-F4t-NeuroP, 14(RS)-14-F4t-NeuroP, 6-keto-PGF1α, and PGF2α, as well as total IsoPs, IsoFs, NeuroPs, NeuroFs, dihomo-IsoPs, dihomo-IsoFs, m-Tyr/Phe, and 8OHdG/2dG, were compared between infants with MRI scores of 0 (no injury), 1 (1A: minimal, and 1B: extensive cerebral lesions), and 2 (2A: basal ganglia thalamic, anterior or posterior limb of internal capsule, and watershed infarction, and 2B: 2A with cerebral lesions) (8). Only two infants of this cohort had severe brain injury (score 3) and hence this subgroup was excluded from statistical analysis. Thus, three binary partial least squares discriminant analysis (PLSDA) models were built to assess metabolic differences between patients with MRI scores of 0, 1, and 2. Receiver operating characteristic (ROC) curves based on the results obtained by leave-one-patient-out cross-validation were calculated (Fig. 1B, top). Biomarker levels found in infants with an MRI score of 1 were significantly different from the other two groups (i.e., with MRI scores of 0 and 2), as confirmed by permutation testing (p ≤ 0.003), while no statistically significant difference was found between infants with MRI scores of 0 and 2. Variable importance in projection scores versus regression vectors (Fig. 1B, bottom) were used to measure the influence of each metabolite on the PLSDA models. Several determined biomarkers showed characteristic patterns in samples from newborns belonging to the MRI score 1 group, that is, levels of 14(RS)-14-F4t-NeuroP, total IsoPs, 15(RS)-15-F2t-IsoP, and total dihomo-IsoPs were higher in infants with an MRI score of 1 than score of 0 or 2 (Fig. 1B bottom, left, and middle).
Finally, levels of oxidative stress biomarkers in male versus female infants were assessed in the search for sex-specific associations. However, no statistically significant differences were found (data not shown).
Discussion
This is the first study to examine the relationship between a large panel of oxidative stress and inflammatory biomarkers and brain injury in infants with moderate/severe HIE. Our data demonstrate that specific indicators for oxidative stress-mediated damage and inflammation can be longitudinally monitored in newborns with HIE, thanks to the use of noninvasively collected urine samples. The results (Fig. 1A) reflect an increase of a specific NeuroP isomer [i.e., 14(RS)-14-F4t-NeuroP] as well as total dihomo-IsoPs over the study period, revealing protracted oxidative stress that persisted during TH and the rewarming period. NeuroPs are lipid mediators generated by nonenzymatic lipid peroxidation of docosahexaenoic acid (DHA), a PUFA highly abundant in brain tissue, and have been suggested as markers of oxidative damage to brain tissue, urine, plasma, or cerebrospinal fluid (9). Dihomo-IsoPs, derived from radical attack on adrenic acid (AdA), are considered biomarkers of cerebral white matter injury. Likewise, increasing levels of biomarkers of damage to proteins (m-Tyr/Phe) and DNA (8OHdG/2dG) were observed. The spontaneous oxidation of Phe in the presence of hydroxyl free radicals produces m-Tyr and o-Tyr that are stable molecules and their concentrations are useful markers of oxidative protein damage. Protein oxidation has been associated with neuronal dysfunction in vivo and may be involved in the pathology of neurodegenerative disorders (1). 8OHdG, produced by oxidation of the nucleotide 2dG, is excreted in urine and is commonly used as biomarker of oxidative damage to DNA (7). Oxidative stress is strongly related to acute brain damage in children, and 8OHdG is a useful marker of brain damage (3).
Simultaneously, we studied inflammatory biomarkers in HIE. Decreasing levels of biomarkers of PGs (i.e., 6-keto-PGF1α and PGF2α) were observed. PGs are produced by the enzymatic peroxidation of AA and they play a key role in the inflammatory response. Inflammation is a response to injury and it is an intrinsically beneficial event that leads to removal of offending factors and restoration of tissue structure and physiological function. PGs, particularly PGs E and F2, lead to cerebral vasodilation and their concentrations increase in response to cerebral ischemia (4). More specifically, 6-keto-PGF1α, a stable metabolite of prostacyclin also known as prostaglandin I2 (PGI2), is a vasodilative and is associated with brain perfusion and cerebral infarction. It is probably the most important PG due to its effect on sensory neurons (2). The decrease of PGs with time observed in the present study might reflect a progressive return to normal levels after the hypoxic insult that occurred before/during delivery.
Access to biomarkers that (early and accurately) diagnose oxidative stress and response could potentially aid in the diagnosis and improve prognosis of newborns with HIE. In this regard, we compared infants with pathologic and normal MRI outcomes by using standardized scores discerning between injury patterns. Our data provide evidence of oxidative stress biomarker changes in urine samples of newborns with HIE depending on their lesion type. Specifically, biomarker patterns in urine samples from newborns with an MRI score of 1 were significantly different from newborns with scores of 0 and 2, while no difference between the latter subgroups was found. Infants belonging to subgroups with MRI scores of 0 and 2 presented lower values of the DHA-derived NeuroP [14(RS)-14-F4t-NeuroP], AA-derived IsoPs [total IsoPs and 15(RS)-15-F2t-IsoP], and AdA-derived dihomo-IsoPs (total dihomo-IsoPs) in comparison with infants with an MRI score of 1. Thus, lipid peroxidation biomarker profiles linked to oxidative damage of different PUFAs, which are all highly abundant in the brain, change according to specific injury patterns (i.e., MRI score 1).
Previous studies established male sex as a risk factor for unfavorable outcome of HIE during the perinatal period regardless of lesion types. Some authors found notable differences between microglia and neuroimmune signaling in male and female brains and suggested that these differences play a relevant role. However, in this study, we did not observe consistent and significant alterations between male and female infants associated with oxidative stress levels.
In summary, this is the first study presenting detailed insights on the evolution of a broad panel of oxidative stress and inflammation biomarkers in newborns with moderate/severe HIE undergoing TH. We identified a set of biomarkers with constantly increasing concentrations that demonstrate the propagation of oxidative injury throughout the whole study period, including the secondary phase of HIE. PGs, in contrast, showed a decreasing tendency, which reflects inflammatory response. We found statistically significant differences in newborns with different patterns of pathologic versus normal HIE outcomes, but not between male and female infants.
Notes
Study approval and population
The study was approved by the Ethics Committee for Biomedical Research of the Health Research Institute La Fe (Valencia, Spain) and registered under the acronym HYPOTOP (EudraCT 2011-005696-17). The HYPOTOP trial is a randomized, controlled, multicenter, double-blinded clinical trial for assessing the efficacy of topiramate versus placebo in newborns with HIE undergoing TH. All methods were performed in accordance with relevant guidelines and regulations, and informed consent was obtained from legal representatives of enrolled infants. For a detailed description of the HYPOTOP trial, including the study design and the established inclusion and exclusion criteria, the reader is referred to the literature (6). In this study, a subcohort of 51 newborns enrolled in the HYPOTOP trial was included.
Samples were collected following a standard operating procedure to avoid bias, systematic errors, and flaws related to the experimental design. Urine samples were collected before initiation of TH (within 6 h of delivery) and at 12, 24, 48, 72, and 96 h after initiation of TH. Urine samples were aliquoted into dry microcentrifuge tubes and stored immediately at −80°C until analysis.
Magnetic resonance imaging
MRI was carried out using different high-field magnet systems between days 4 and 8 after birth. Three-dimensional gradient-echo T1-weighted MR images, axial fast spin-echo T2-weighted MR images, and diffusion-weighted images were acquired. The interpretation of MRI results was carried out by an experienced, blinded pediatric neuroradiologist by rating the extent of the injury in the posterior limb of the internal capsule, basal ganglia, thalamus, white matter, and cortex and using a standardized score according to the National Institute of Child Health and Human Development (NICHD) Neonatal Research Network (NRN), discerning between injury patterns as described by Shankaran et al. (8).
Standards and reagents
LC-MS grade solvents (methanol CH3OH, ethanol, heptane, and acetonitrile CH3CN) were purchased from J.T. Baker (Phillipsburg, NJ). Formic acid (HCOOH, 98%) and ethyl acetate (analytical grade) were from Panreac (Barcelona, Spain). Ultrapure H2O was generated using a Milli-Q system from Merck Millipore (Darmstadt, Germany).
For lipid biomarker determination, commercially available IsoPs, 2,3-dinor-15-F2t-IsoP (CAS No. 221664-05-7) (16290), 15-keto-15-E2t-IsoP (Item No. 14390; CAS No. 914804-63-0) (14390), 15-keto-15-F2t-IsoP (CAS No. 191919-01-4) (16390), 15-E2t-IsoP (CAS No. 27415-25-4) (14350), and 15-F2t-IsoP (CAS No. 27415-26-5) (16350), and PGs, PGE2 (CAS No. 363-24-6) (14010), PGF2α (CAS No. 551-11-1) (16010), 11β-PGF2α (CAS No. 38432-87-0) (16520), 6-keto-PGF1α (CAS No. 58962-34-8) (15210), and dihomo-PG (1α,1β-dihomo-PGF2α [CAS No. 57944-39-5] [16050]), were purchased from Cayman Chemical Company (Ann Arbor, MI), with purities ≥95%, as well as deuterated internal standards (ISs), PGF2α-D4 (CAS No. 34210-11-2) (316010), 10-epi-10-F4t-NeuroP-D4, and 15-F2t-IsoP-D4 (CAS No. 211105-40-7) (316350), with purities ≥98% and ≥99% incorporation of the deuterated form (D1–D4) and <1% D0. Other analytical standards employed were F2-IsoP [5(RS)-5-F2t-IsoP]; F4-NeuroPs [4(RS)-4-F4t-NeuroP, 10-epi-10-F4t-NeuroP, 10-F4t-NeuroP, and 14(RS)-14-F4t-NeuroP]; F4-NeuroF [4(RS)-ST-Δ5-8-NeuroF]; F2-dihomo-IsoPs [17-F2t-dihomo-IsoP and ent-7(RS)-7-F2t-dihomo-IsoP]; and F2-dihomo-IsoFs [17(RS)-10-epi-SC-Δ15-11-dihomo-IsoF and 7(RS)-ST-Δ8-11-dihomo-IsoF] synthesized at the Institut des Biomolecules Max Mousseron (Montpellier, France) with purities ≥99%, and all physical data reported in those published articles confirmed their structures.
Standards of o-Tyr (CAS No. 2370-61-8) (93851), m-Tyr (CAS No. 775-06-4) (T3629), Phe (CAS No. 63-91-2) (78019), 3NO2-Tyr (CAS No. 621-44-3) (N7389), 3Cl-Tyr (CAS No. 7423-93-0) (512443), p-Tyr (CAS No. 556-03-6) (145726), 8OHdG (CAS No. 88847-89-6) (H5653), and 2dG (CAS No. 312693-72-4) (D-7145) (>96% w/w purity) were obtained from Sigma-Aldrich (St. Louis, MO). The ISs, Tyr-D2 (CAS No. 30811-19-9) (DLM-449-1), 2dG-13C15N2 (CNLM-6836-SL), and 8OHdG-13C15N2, were purchased from Cambridge Isotope Laboratories and Phe-d5 from CDN Isotopes (Pointe-Claire, Canada).
Isoprostanoid determination
Ten microliters of β-glucuronidase solution (10 mg/mL) was added to 600 μL of urine samples previously thawed on ice and homogenized and then incubated at 37°C for 90 min. After incubation, 300 μL of IS solution (100 nM) in H2O (0.1% v/v HCOOH, pH 3):CH3OH (85:15 v/v) was added, homogenized, and centrifuged at 16,000 g for 10 min at 4°C. For clean up and preconcentration, solid-phase extraction (SPE) using Discovery® DSC-18 SPE 96-well plates (Sigma-Aldrich) was carried out. SPE cartridges were conditioned with 1 mL of CH3OH, followed by rinsing with 1 mL of H2O (0.3% v/v HCOOH, pH 3):CH3OH (95:5 v/v). The diluted samples were loaded onto the SPE plate, followed by washing with 500 μL of H2O and 500 μL of heptane. Cartridges were dried in room air and sample extracts were eluted with 4 × 100 μL of ethyl acetate, which were subsequently evaporated using a miVac centrifugal vacuum concentrator from Genevac Ltd. (Ipswich, United Kingdom). Sample extracts were dissolved in 60 μL of H2O (0.1% v/v HCOOH, pH 3):CH3OH (85:15 v/v) before UPLC-MS/MS analysis.
Samples were analyzed using an Acquity Xevo TQS system from Waters (Milford, MA) operating in negative electrospray ionization (ESI−) mode using a Waters BEH C18 reversed-phase column (2.1 × 100 mm, 1.7 μm) and a binary H2O (0.1% v/v formic acid):CH3CN (0.1% v/v formic acid) gradient.
The UPLC-MS/MS method was based on our previous validated methods for quantification of lipid peroxidation biomarkers (7). The measurement parameters used for the studied biomarkers are summarized in Table 2. Biomarker concentrations were normalized to creatinine. To quantify creatinine concentration, a Urinary Creatinine Detection Kit (Arbor Assays™; Ann Arbor, MI) was employed. Dilution at 1:4 (v/v) of samples with H2O and a 96-well plate microreader (Halo LED 96; Dynamica Scientific Ltd., Newport Pagnell, United Kingdom) were used, measuring absorbance at 490 nm.
Biomarkers of proteins and DNA oxidation
Determination of biomarkers of oxidative damage to DNA and proteins was performed following a previously validated UPLC-MS/MS method (7). Fifty microliters of IS solution (1 μM) in H2O (0.1% v/v HCOOH) was added to 50 μL of centrifuged urine sample. For UPLC-MS/MS analysis, samples were loaded on 96-well plates and analyzed randomly using an Acquity Xevo TQ system from Waters operating in the positive electrospray ionization (ESI+) mode. Separation was performed using an Acquity UPLC BEH C18 reversed-phase column (2.1 × 50 mm, 1.7 μm) from Waters and a CH3OH (0.05% v/v formic acid):H2O (0.05% v/v formic acid) binary gradient. Parameters used for measurement of different metabolites are summarized in Table 2. For comparison between samples, the following metabolite ratios were determined: m-Tyr/Phe, o-Tyr/Phe, 3Cl-Tyr/p-Tyr, 3NO2-Tyr/p-Tyr, and 8OHdG/2dG.
Statistical analysis
UPLC-MS/MS data were acquired and processed using MassLynx 4.1 and QuanLynx 4.1 (Waters), respectively. Values below the LOQ were replaced by ½ LOQ. Further data analysis was carried out in MATLAB R2019b (MathWorks, Inc., Natick, MA) using in-built as well as in-house written scripts and functions and the PLS Toolbox 8.0 (Eigenvector Research, Inc., Wenatchee). PLSDA models of autoscaled data and leave-one-patient-out cross-validation were built. ROC curves were based on PLSDA models. Significance of models was tested by permutation testing (500 permutations). Group comparisons of demographic and clinical characteristics were carried out using Student's t-test for unequal variances, chi-square test, or Wilcoxon rank-sum test for parametric, categorical, and nonparametric variables, respectively. p Values <0.05 were considered statistically significant.
Footnotes
Authors' Contributions
All authors have made substantial contributions to the conception or design of the work or acquisition, analysis, or interpretation of data for the work. All authors were involved in drafting the work or its critical revision for important intellectual content. All authors gave their final approval of the version to be published. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Acknowledgments
The authors express their gratitude to the parents and their newborns who participated in the study. Technical assistance of the Servicio de Soporte a la Investigación Experimental (SCSIE) at the University of Valencia (Spain) is acknowledged.
Authors are thankful to the members of the HYPOTOP Study Group: Isabel Izquierdo, Ana Gimeno, María Gormaz, Raquel Escrig, María Cernada, Marta Aguar, and Ester Torres (Division of Neonatology, University and Polytechnic Hospital La Fe, Valencia, Spain); Isabel Benavente-Fernández (Division of Neonatology, University Hospital Puerta del Mar, Cádiz, Spain); Eva Valverde and Malaika Cordeiro (Division of Neonatology, University Hospital La Paz, Madrid, Spain); Dorotea Blanco (Division of Neonatology, University Hospital Gregorio Marañón, Madrid, Spain); Hector Boix (Department of Neonatology, University Hospital Vall d'Hebrón, Barcelona, Spain); Fernando Cabañas (Division of Neonatology, University Hospital Quirónsalud Madrid, Madrid, Spain); Mercedes Chaffanel (Division of Neonatology, Regional University Hospital Málaga, Málaga, Spain); Belén Fernández-Colomer (Division of Neonatology, Central University Hospital of Asturias, Oviedo, Spain); Jose Ramón Fernández-Lorenzo (Division of Neonatology, University Hospital Complex of Vigo, Vigo, Spain); Begoña Loureiro (Division of Neonatology, University Hospital Cruces, Bilbao, Spain); Maria Teresa Moral-Pumarega (Division of Neonatology, University Hospital 12 de Octubre, Madrid, Spain); Antonio Pavón (Division of Neonatology, University Hospital Virgen del Rocío, Sevilla, Spain); and Inés Tofé (Division of Neonatology, University Hospital Reina Sofía, Córdoba, Spain).
Author Disclosure Statement
The authors declare no conflict of interest.
Funding Information
This research was funded by Instituto de Salud Carlos III [Grant Nos. CP16/00034, CD19/00037, PI17/00127, and PI20/00964] (Co-funded by European Regional Development Fund (ERDF); “A way to make Europe”).
