Abstract
Significance:
Magnetic resonance imaging (MRI) with hyperpolarized (HP) 13C-labeled redox-sensitive metabolic tracers can provide noninvasive functional imaging biomarkers, reflecting tissue redox state, oxidative stress, and inflammation, among others. The capability to use endogenous metabolites as 13C-enriched imaging tracers without structural modification makes HP 13C MRI a promising tool to evaluate redox state in patients with various diseases.
Recent Advances:
Recent studies have demonstrated the feasibility of in vivo metabolic imaging of 13C-labeled tracers polarized by parahydrogen-induced polarization techniques, which offer a cost-effective alternative to the more widely used dissolution dynamic nuclear polarization-based hyperpolarizers.
Critical Issues:
Although the fluxes of many metabolic pathways reflect the change in tissue redox state, they are not functionally specific. In the present review, we summarize recent challenges in the development of specific 13C metabolic tracers for biomarkers of redox state, including that for detecting reactive oxygen species.
Future Directions:
Applications of HP 13C metabolic MRI to evaluate redox state have only just begun to be investigated. The possibility to gain a comprehensive understanding of the correlations between tissue redox potential and metabolism under different pathological conditions by using HP 13C MRI is promoting its interest in the clinical arena, along with its noninvasive biomarkers to evaluate the extent of disease and treatment response.
Introduction
Over the past several decades, numerous studies have revealed that cellular and tissue redox state shifts from the homeostatic balance in a range of pathological conditions, including cancer, stroke, inflammation, diabetes, and viral infections, among others (14). Changes in redox environment can induce increased oxidative stress and affect gene expression that regulates the energy metabolism of cells. Many enzymes require redox sensitive cofactors such as NAD+/NADH or FAD/FADH2 for their enzymatic activity; as such, the tissue redox state can be estimated by measuring the concentration ratio of a specific redox couple of metabolites (62), whose reversible metabolic reaction is catalyzed by such redox-sensitive enzymes.
The concentration ratio of lactate-to-pyruvate has been traditionally used to estimate the redox potential of cells and tissues. For example, the difference in concentration ratio of the lactate/pyruvate in arterial and coronary vein blood revealed that the redox potential of heart muscle was ∼20 mV more negative under anoxia compared with under normoxic conditions (24), where the pyruvate-to-lactate metabolic flux is related to the cytosolic NAD+/NADH couple as a result of the rapid equilibrium of the lactate dehydrogenase activity.
The development of the dissolution dynamic nuclear polarization (d-DNP) hyperpolarization technique, by which the magnetic resonance imaging (MRI) signal obtainable from 13C-labeled metabolic tracers is enhanced by more than 10,000 times, has enabled the realization of noninvasive monitoring of the flux of a particular metabolic reaction in real time (4, 22). Hyperpolarized (HP) 13C-labeled pyruvate—the most widely studied HP 13C MRI tracer to date—is taken up by cells and undergoes the same metabolism as endogenous pyruvate. As such, 13C labeling of pyruvate in the C-1 position allows the study of pyruvate-lactate metabolism (and in turn, the Warburg effect in cancer), whereas labeling in the C-2 position permits further metabolic pathways in the citric acid cycle to be probed.
Metabolic MRI of HP [1-13C] pyruvate has revealed that the conversion rate of pyruvate to lactate depends on the tissue microenvironment in living creatures, including humans (51), and this metabolic conversion efficiency can be used as a noninvasive biomarker of the extent of the disease and its response to treatments (43, 44). More than 60 different 13C tracers for HP 13C MRI studies have already been reported in in vitro and in vivo preclinical applications (31), some which are specific to tissue redox state. In the present review, we summarize the recent progress in HP 13C MRI techniques, focused on the metabolic tracers that hold most promise for the detection of the redox state in vivo.
HP 13C MRI for Redox-Sensitive Metabolic Imaging
13C is a nuclear magnetic resonance (NMR)-sensitive isotope of carbon that has a gyromagnetic ratio ∼4 × lower than that of 1H, and around 1.1% natural abundance. Thanks to its extensive chemical shift range (∼200 ppm, compared with ∼10 ppm for 1H), 13C NMR presents an attractive method for interrogation of metabolism in vivo (58). Further, detection at the Larmor frequency of 13C yields low background signal and a high contrast. Nevertheless, the sensitivity is relatively poor, and it remains challenging to obtain high-quality spectroscopy or imaging data even after injection of 13C-enriched metabolic probes (8).
Hyperpolarization offers a means to circumvent this sensitivity limitation. By biasing the relative populations of the ground-state nuclear energy levels in the presence of a magnetic field, a four to five order of magnitude boost in the attainable magnetic resonance (MR) signal can be realized. The two most common methods employed for hyperpolarization of 13C nuclei are d-DNP (4) and parahydrogen-induced polarization (PHIP) (7, 56). d-DNP relies on the fact that the electron gyromagnetic ratio is ∼660 times greater than that of 1H, which leads to a near-unity polarization under extreme conditions. On cooling a sample mixed with glassing agent and free radicals to ∼1 K in the presence of a strong magnetic field (several Tesla), microwave irradiation is used to induce polarization transfer from the free electrons to 13C nuclei in the solid state (Fig. 1). The sample is then rapidly dissolved and transported to the MRI system for delivery into the subject.
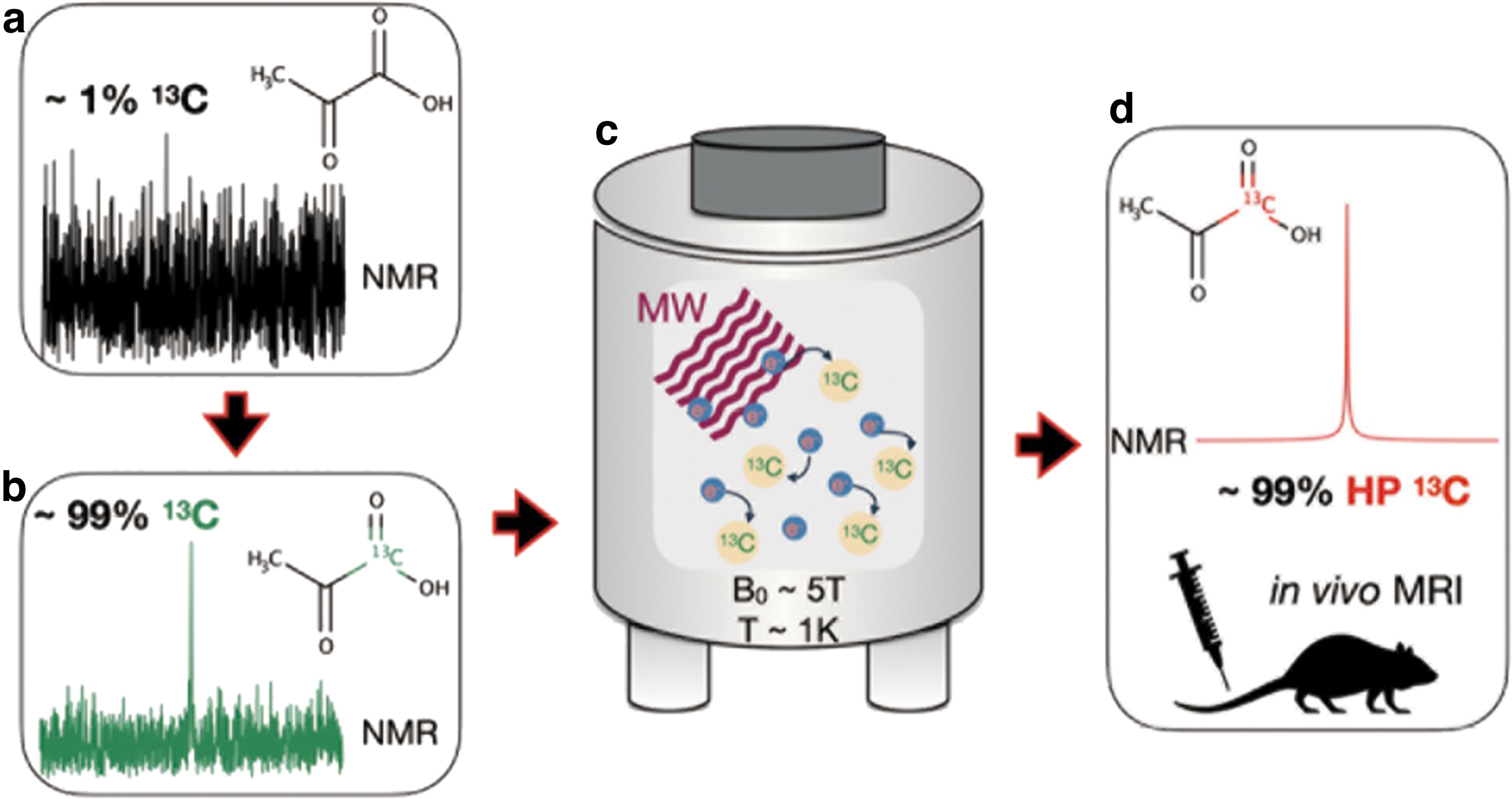
On the other hand, PHIP utilizes the innate spin order of parahydrogen (a spin isomer of the hydrogen molecule) to induce 13C nuclear polarization by irreversible, direct addition of parahydrogen to the molecule of interest (18) or reversible interaction with an intermediary (1). In the following, we will focus on applications of d-DNP that are pertinent to the assessment of redox state; for further information on the differences between d-DNP and PHIP, and the 13C MRI probes that can be accessed with PHIP, we refer the readers to our recent review article (66).
Laboratory set-up requirements
To establish HP 13C MRI technology in a new laboratory, the following requirements must be met:
Polarization device: A number of commercial and open-source 13C DNP and PHIP (13) polarizer apparatus are available for the production of a wide variety of HP 13C molecules for in vitro and preclinical in vivo metabolic imaging experiments. At present, only one sterile polarizer approved for clinical HP 13C metabolic MRI exists: the GE Healthcare SpinLab device (5). The MR system is capable of multi-nuclear MRI; that is, such that the detection of nuclei other than 1H is enabled. Multi-nuclear functionality is typically available on high-field NMR systems and most preclinical MRI systems by default, whereas multi-nuclear clinical MRI scanners remain less common, and MR vendor support must be sought to install hardware and software for this purpose. MR coils for 13C (or other nuclei of choice): Radiofrequency coils for detection of MR signal at Larmor frequencies other than that of 1H nuclei are not routinely supplied by MR vendors, and therefore they must be purchased separately or custom built on site. Optimal pathway for tracer delivery from polarizer to subject: The polarization of HP 13C molecules tends to decay over the order of tens of seconds, and thus it is usually necessary to situate the polarizer close to the MRI scanner for rapid sample transport and injection, and/or magnetically shield the sample during the transport process (46). In the case of human use, such pathways must be sterile.
Tracer characteristics
In the following section, we review the most common 13C tracers used for metabolic MRI applications. The biochemistry of such imaging tracers is a highly active area of research, and a full review is beyond the scope of this article [we refer the readers to Refs. (32, 66)]. When designing an HP 13C tracer for in vivo metabolic MRI applications, it is important to consider the following requirements (61):
Biological safety/compatibility: HP 13C tracers are typically chemically identical to endogenous molecules (aside from 13C-labeling) or are otherwise biologically inert.
Metabolically active: Many HP 13C tracers exhibit metabolic activity without the requirement of co-factors. (13C-urea is an exception and exhibits no metabolic activity.)
Chemical shift sensitivity: By virtue of the extensive chemical shift range exhibited by the 13C nucleus, different metabolites exhibit distinct chemical shift values; this critical feature allows selective interrogation of MR signal dynamics to quantify metabolic behavior. Nevertheless, it must be ensured that the chemical shift difference between reactant and product of the metabolic reaction of interest is detectable (ideal case: approximately few parts per million on conventional MRI systems).
Sufficiently long polarization lifetime: The MR signal enhancement conferred by d-DNP or other hyperpolarization methods is nonpermanent, and the 13C nuclear polarization decays to that of the thermal equilibrium value on a timescale of the spin-lattice relaxation time (T1); typically, of the order of several seconds and governed by 13C-labeling position, molecular weight, and detection field strength among other factors. For example, the most commonly used HP 13C metabolic imaging probe to date—[1-13C] pyruvate—exhibits a T1 of around 65 s at 3 T (9). Novel probes must have a sufficient T1 to ensure detectable signal remains after polarization and delivery to the subject in the MR scanner.
Absorption, distribution, metabolism, and excretion (ADME): The tracer must be delivered to the target organ within the polarization lifetime (iv), and the associated signal should be free of influence (or otherwise decouplable) from that of neighboring tissues. In the HP 13C MRI experiments, it is typical to wait 20–30 s after intravenous injection of HP 13C tracer before MRI scans for the tracer to well distribute into peripheral regions. As most tracers are endogenous and/or involved in metabolic processes, excretion is not usually a concern, though it should be considered for new exogenous tracers. HP 13C dynamic NMR/MRI can be used to study ADME properties of new tracers; and importantly, perfusion to the organ of interest is monitored dynamically by using MR spectroscopy before any imaging acquisition to determine the most appropriate acquisition timing.
Acquisition techniques
The most basic acquisition method used to probe metabolic activity with HP 13C tracers is dynamic MR spectroscopy, wherein the MR signal kinetics determines the relative metabolic flux. Spectroscopy provides a global measurement of the metabolic activity within a certain region (e.g., the whole body, or a slice of the anatomy), whereas MR spectroscopic imaging (MRSI) extends this approach to generate distinct images of the MR signal detected from each metabolite. MRSI comprises a class of methods, including chemical shift imaging (22), echo planar spectroscopic imaging (35), and IDEAL spectral decomposition methods (72) (in order of increasing acquisition efficiency at the expense of spectral information), and it is performed either dynamically, or at a single time-point that is often chosen to coincide with the peak MR signal of the metabolic product of interest (e.g., as predetermined from dynamic MR spectroscopy).
Quantification methods
The MR signal ratios of 13C in different metabolites are commonly used as simplistic yet quantitative biomarkers of metabolic activity (see Table 1 for examples). This approach is effective for analysis of single time-point spectroscopy or imaging data; for example, lactate-to-pyruvate ratio as a marker of the Warburg effect in cancer detection and staging (3). Further quantitative information can be gleaned by acquiring MR spectra dynamically and fitting the time dependence of the MR signals with kinetic models of the chemical exchange process (25). This enables the derivation of parameters such as the forward and backward metabolic reaction rates (e.g., kPL and kLP for pyruvate–lactate conversion), in addition to the spin-lattice relaxation rates; for example, reduced kPL flux postablation and chemotherapy in a patient with high-grade prostate cancer as a marker of treatment response (2).
Currently Available 13C Magnetic Resonance Imaging Tracers That Can Be Used to Assess Metabolic Processes, Including Aspects of Redox State
δ: NMR chemical shift in parts per million; ref: literature reference(s).
Pyruvate can also be 13C-labeled in the 2 position.
βOHB, β-hydroxybutyrate; AA, ascorbic acid; AcAc, acetoacetate; DHA, dehydroascorbic acid; HP, hyperpolarized; MRI, magnetic resonance imaging; NMR, nuclear magnetic resonance.
HP 13C MRI Tracers for Imaging Redox State
A nonexhaustive list of 13C metabolic imaging probes that can be polarized by d-DNP is presented in Table 1, along with their associated biomarkers and biomedical uses [for a more comprehensive review, see, e.g., Refs. (31, 66)]. The most widely studied HP 13C metabolic MRI tracer to date is [1-13C] pyruvate; its conversion to HP [1-13C] lactate and other metabolites (including [1-13C] alanine and [1-13C] bicarbonate) are key components of cellular metabolism. In particular, increased glycolytic pyruvate–lactate metabolism is a hallmark of cancer cells (the so-called Warburg effect); as such, the relative production of HP [1-13C] lactate to HP [1-13C] pyruvate provides a sensitive measure of cancer state (Fig. 2a, b). This metabolic information is distinct (downstream), and complementary to that of 18F fluorodeoxyglucose-positron emission tomography (FDG-PET), which is used clinically to probe the first step of the glycolysis metabolic pathway.
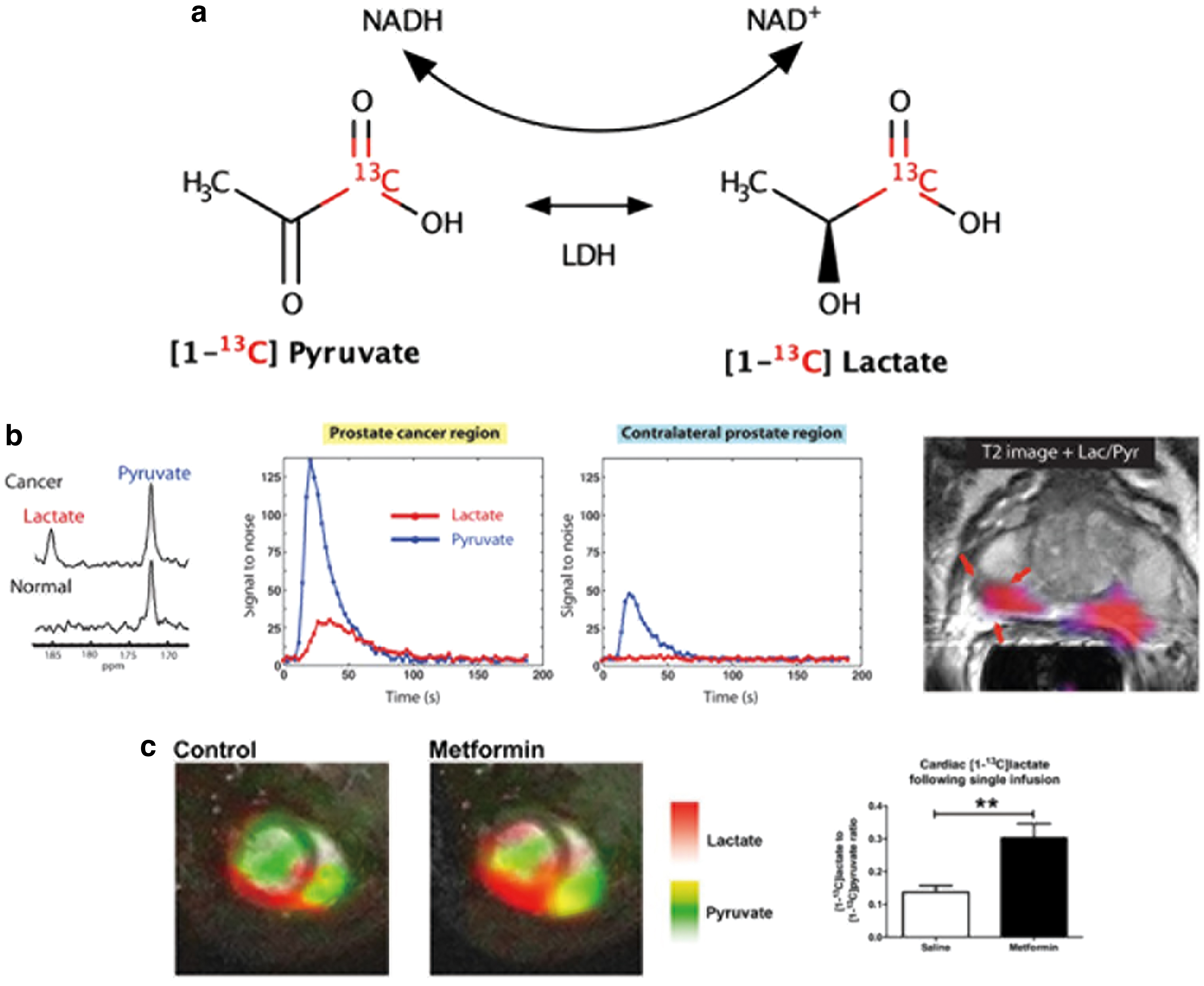
Numerous preclinical studies have reported elevated HP [1-13C] lactate flux in a wide range of cancer models (3, 22, 30). In particular, HP [1-13C] pyruvate MRI is well suited to the assessment of new and existing cancer therapies (16, 54, 59, 75), thanks to its high sensitivity, safety, and nonionizing properties. With the advent of sterile, high-throughput d-DNP polarizer systems (5), in vivo human studies of HP [1-13C] pyruvate to HP [1-13C] lactate metabolism in prostate (2, 51), pancreatic (68), and breast cancer (21), and the healthy and unhealthy heart (15, 57) and brain (23, 48) have been realized, and clinical trials are underway at several sites across the globe. Though most commonly labeled with 13C in the 1 position, pyruvate can also be 13C-labeled in the 2 position, which enables further interrogation of downstream TCA cycle metabolism (12, 26) that cannot be probed with [1-13C] pyruvate.
Beyond pyruvate, HP [1,4-13C2] fumarate has been developed as a sensitive tracer of cellular necrosis; exogenously delivered fumarate cannot easily permeate into healthy cells, and thus production of HP [1-13C], [4-13C] malate only occurs when the cellular plasma membrane integrity is damaged by necrotic processes (20). This selective HP 13C malate production has been utilized to study necrotic processes in myocardial infarction (47) and diabetic nephropathy (37) among other conditions, and it offers a sensitive means to interrogate response to treatment of tumors (19, 74). HP 13C Bicarbonate (H13CO3 −) and its conversion to 13C carbon dioxide can be used to map pH in vivo, for example, its use has been reported in murine tumors (19), ischemic rat hearts (63), and ventilator-induced lung injury (17). [1-13C] Urea is readily polarizable by using d-DNP (4), and, as one of the few probes in common use that exhibits no metabolic activity, it finds application as a probe of perfusion; for example, in imaging of blood flow (70) and assessment of renal function (52). Further, it can be co-polarized with [1-13C] pyruvate for simultaneous assessment of metabolism and perfusion (36).
The most studied HP 13C-labeled tracers for assessment of redox state in vivo are the reduced and oxidized forms of vitamin C, namely HP [1-13C] dehydroascorbic acid (DHA) and [1-13C] ascorbic acid (AA) (6, 28) (Figs. 3 and 4). Both of these molecules are polarizable to acceptable levels of sensitivity by d-DNP and offer different insights into redox processes; HP [1-13C] AA is sensitive to extracellular reactive oxygen species (ROS), whereas HP [1-13C] DHA reflects intracellular redox state, highlighted by the lack of extracellular oxidation of HP [1-13C] AA and rapid intracellular reduction of HP [1-13C] DHA in murine lymphoma tumors (6) (Fig. 3b). As such, subsequent studies have focused on the application of HP [1-13C] DHA and its reduction to AA, a reaction that can be mediated by glutathione- (GSH) or NADPH-dependent mechanisms (in turn, NADPH-dependent reduction of oxidized glutathione to GSH maintains levels of the latter) (28). For example, after introduction of HP [1-13C] DHA into the healthy rat brain, high levels of HP [1-13C] AA were observed, highlighting the brain's reductive properties (Fig. 4a) (28).
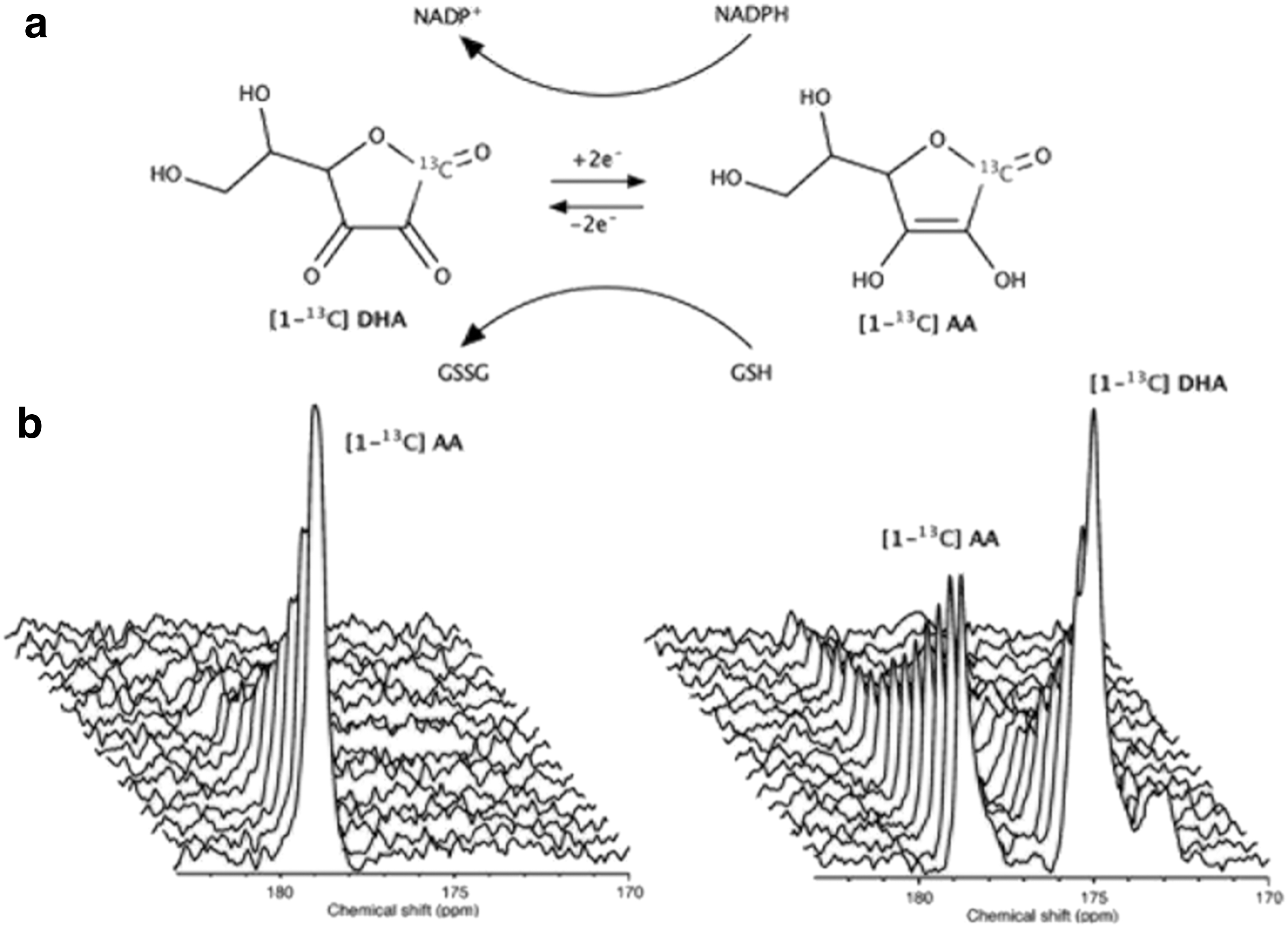
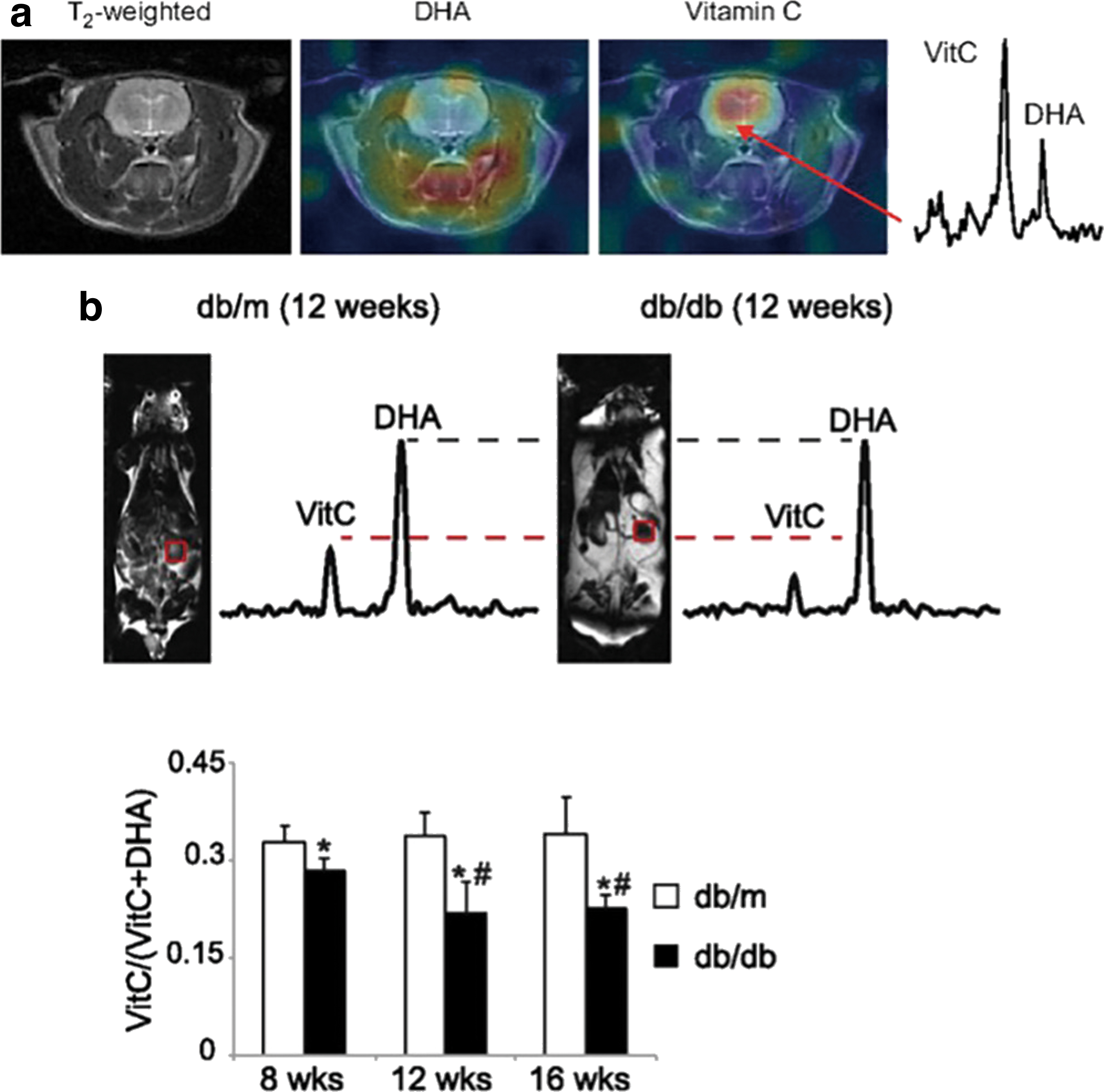
The upregulation of glutathione in cancer cells to counteract oxidative stress has been studied by using HP [1-13C] DHA in transgenic adenocarcinoma of the mouse prostate (TRAMP) tumors, wherein a dramatic increase in reduction of HP [1-13C] DHA in tumors compared with surrounding tissue was confirmed to predominantly reflect increased glutathione (28, 29). Similarly, in a murine model of type 2 diabetes and diabetic nephropathy, wherein overproduction of ROS leads to oxidative stress, a decrease was observed in the reduction of HP [1-13C] DHA in diabetic kidneys, indicative of reduced GSH concentration (32) (Fig. 4b). The sensitivity of HP [1-13C] DHA is yet to be directly compared with that of alternative MRI redox probes such as nitroxide radicals (27), and it may find application as part of a suite of imaging assessments, including such nitroxides and/or HP [1-13C] pyruvate (43); the possibility of co-polarization with HP [1-13C] pyruvate has yet to be explored.
Beyond DHA-AA metabolism, several other HP 13C-labeled tracers have been studied as potential indicators of redox state. For example, Lewis et al. reported that an increase in [1-13C] pyruvate to [1-13C] lactate conversion in the heart on metformin treatment was accompanied by an increase in heart and liver cytosolic redox state, indicating that pyruvate–lactate metabolism is capable of detecting cytoplasmic redox state (Fig. 2c) (39). However, pyruvate–lactate conversion as a measurement of cellular redox state is constrained by [1-13C] pyruvate signal from the vasculature and extra-cellular lactate production; to this end, HP [1-13C] alanine presents an alternative potential tracer of intracellular redox state, where conversion to [1-13C] pyruvate and [1-13C] lactate is indicative of cytosolic [NAD+]/[NADH] levels (55).
More recently, HP [1,3-13C2] Acetoacetate and its reduction to [1,3-13C2] β-hydroxybutyrate have been utilized to directly interrogate mitochondrial redox state in the rat kidney (71) and isolated, perfused rat hearts (10). This conversion is catalyzed by the mitochondrial enzyme β-hydroxybutyrate dehydrogenase and is indicative of the redox state of the NAD(H) system. Metformin treatment was confirmed to increase renal production of HP [1,3-13C2] β-hydroxybutyrate in rats (71), and similar increased production was observed in ischemic rat hearts and those exposed to a complex I inhibitor, when compared with control hearts (10). It is also worth noting that HP 13C-benzoylformic acid has been investigated as a potential tracer with sensitivity to the concentration of hydrogen peroxide (41)—a key metabolite in cellular redox processes; however, in vivo application is yet to be reported. HP d-[1,2,3,4,5,6,6-13C6] glucose-d7 has been utilized to quantify in-cell free cytosolic [NAD+]/[NADH] ratios (11), however in vivo application is constrained by the prohibitively low T1 relaxation time, especially when the molecule is not fully deuterated.
Applications of HP 13C MRI in Various Diseases
Shifts in metabolic flux are a characteristic accompaniment of all pathological conditions, some of which are strongly coupled with tissue redox state especially when the catalytic enzyme requires cofactors such as NAD(H) and can be useful biomarkers to evaluate the extent of the disease and its response to treatments. In this section, we further discuss specific medical conditions for which HP 13C MRI holds particular promise.
Monitoring cancer treatment response
The tumor microenvironment is characterized by chronic and transient hypoxia (45). Upregulation of hypoxia inducible factor signaling evokes a metabolic shift from oxidative phosphorylation (OXPHOS) to glycolysis and generation of a highly reducing redox state in tumors (27). As pyruvate stands at the key branching point of glucose metabolism, HP [1-13C] pyruvate MRI can provide unique information on the OXPHOS-to-glycolysis metabolic shift. As described in the earlier subsections, the feasibility of detection, malignancy assessment, and staging of tumors has been demonstrated in several cancerous organs in humans (21, 51).
Recent research in the field has focused on the monitoring of cancer treatment response. The characteristic aerobic glycolysis in tumors (Warburg effect) is mediated by the activation of mammalian target of rapamycin (mTOR) signaling and cellular Abelson kinase ABL1 regulated oxidative stress in fumarate hydratase-deficient tumor. Inhibition of ABL1 phosphorylation by Vandetanib and resultant alteration of glycolytic tumor metabolism can be readily observed by metabolic MRI of HP [1-13C] pyruvate (65). Further, cancerous cell death is a ubiquitous indicator of tumor treatment response. Monitoring of necrotic cell death using HP [1,4-13C2] fumarate has been reported for treatment-induced cancerous cell death by chemotherapy and photoimmunotherapy (20, 33).
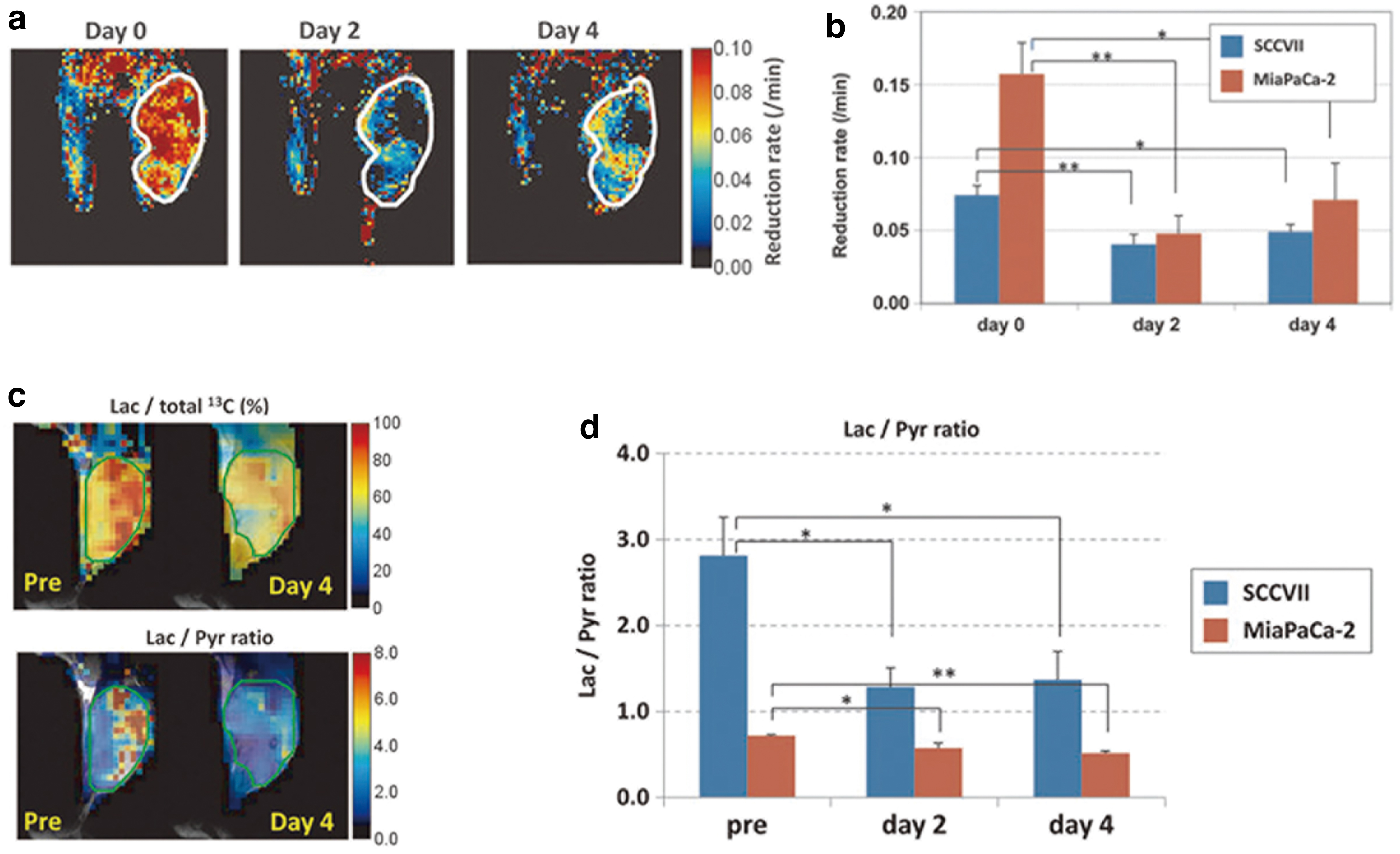
Direct evidence of a correlation between tissue redox state change and metabolism in response to anticancer treatment was investigated with antiangiogenic agent sunitinib treatment of murine tumor xenografts (43). The reduction rate of a redox-sensitive MRI contrast agent 3-carbamoyl-PROXYL was significantly decreased, indicating an oxidative shift in tissue redox potential, 2–4 days after sunitinib treatment during the time known as the vascular re-normalization window (Fig. 5a, b). Consistently, HP [1-13C] pyruvate MRI showed that pyruvate-to-lactate metabolic flux decreased during the same time period, 2–4 days after sunitinib administration (Fig. 5c, d). There is an increasing body of evidence to suggest that metabolic MRI of HP 13C-labeled tracers can provide noninvasive imaging biomarkers of tissue redox state in response to cancer treatments.
Potential applications in heart diseases
The heart pumps 5–6 L of blood every minute and requires a large amount of energy to generate the mechanical force. To fulfill this energy demand, cardiomyocytes are densely packed with mitochondria and consume a large amount of oxygen for OXPHOS. This makes the heart especially susceptible to hypoxia, oxidative stress, and changes in the mitochondrial influx of substrates such as pyruvate. In the following, we consider three specific heart disorders for which HP 13C MRI could have a clinical impact.
Ischemic heart disease is caused by stenosis or occlusion of coronary arteries, leading to hypoxia and dysfunction of the downstream myocardium. Historically, ischemic heart disease was diagnosed by evaluating the degree of coronary stenosis. However, recent evidence suggests that the degree of stenosis does not necessarily correlate with the degree of tissue hypoxia, and thus direct measurement of tissue hypoxia is becoming increasingly important for making treatment decisions (42, 64). The heart mainly uses fatty acid as the substrate for OXPHOS, but during hypoxia, OXPHOS ceases, and glycolysis becomes the primary mode of ATP production. For this reason, FDG-PET imaging, which visualizes the extent of glucose utilization, has been used for detecting cardiac ischemia. However, since glucose can also be oxidized by OXPHOS, 18FDG-PET lacks specificity to glycolysis.
In contrast, HP [1-13C] pyruvate MRI can provide unique information as to whether glucose is being used for OXPHOS or glycolysis by tracking the fate of pyruvate. The HP [1-13C] pyruvate MRI-derived lactate-to-pyruvate ratio within a tissue reflects the NAD+/NADH ratio and the level of oxidative stress, which builds up during ischemia and thus holds promise for detecting cardiac ischemia. In preclinical cardiac ischemia models, an increase in pyruvate-to-lactate conversion in vivo has been reported (49). Further, the technique has recently been proven feasible in humans with a sufficient spatial resolution (Fig. 6a) (15), and further clinical application is anticipated.
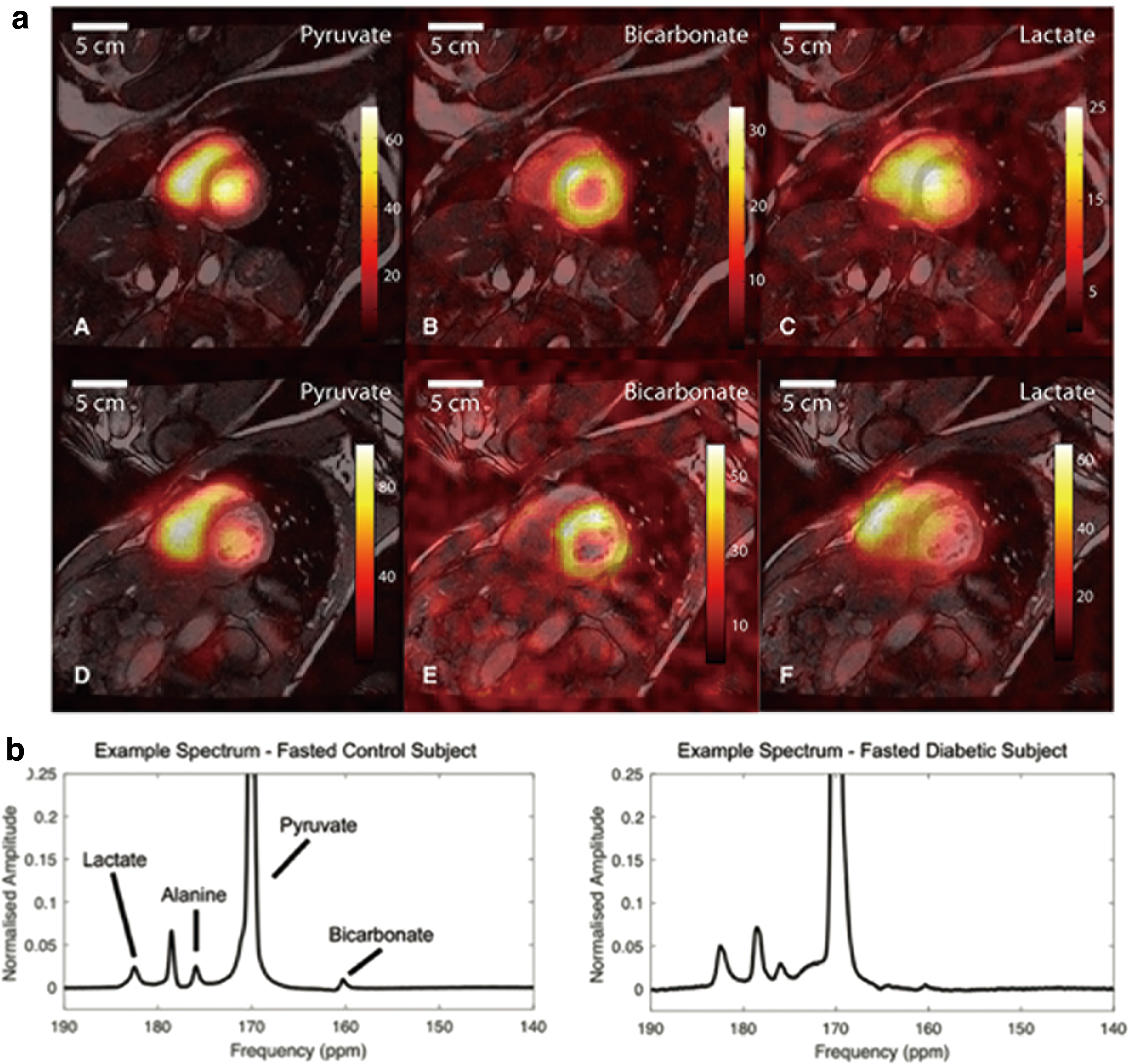
Diabetic cardiomyopathy occurs independently of cardiac ischemia in diabetic patients. It is characterized by structural and functional alterations of the heart, such as hypertrophy, fibrosis, diastolic dysfunction, and systolic dysfunction. Although its molecular mechanisms are still under investigation, decreased pyruvate oxidization, excessive fatty oxidization, and increased oxidative stress have been reported as prominent metabolic features of the diabetic heart (53). In a rodent model of diabetic cardiomyopathy, HP [1-13C] pyruvate MRI revealed a decrease in pyruvate oxidation, and treatment was successfully performed by upregulating pyruvate dehydrogenase activity (Fig. 6b) (38). Recently, a similar decrease in cardiac pyruvate oxidation was observed by using HP [1-13C] pyruvate MRI in human diabetic patients with only mild insulin resistance (57), suggesting that the technique is sufficiently sensitive to detect the earliest changes in cardiac metabolism in diabetes. Response to metformin, an anti-diabetic drug and a complex I inhibitor that ameliorates oxidative stress in the heart, can also be monitored with HP [1-13C] pyruvate MRI (39). We anticipate further application of the technique to aid diagnoses, guide treatment, and provide novel insights on the molecular basis of diabetic cardiomyopathy.
Inflammatory heart diseases, such as sarcoidosis and myocarditis, are characterized by excessive inflammation of the myocardium and resulting tissue necrosis. Radioisotope imaging, namely Gallium-67 scintigraphy and 18FDG-PET, are currently used clinically for diagnoses and evaluating response to immunosuppressive therapy. However, periodic appraisal with a short interval, which is especially useful for evaluating treatment response, is difficult due to the ionizing radiation exposure. As HP [1-13C] pyruvate can detect tissue inflammation and HP [1,4-13C2] fumarate can detect cardiac tissue necrosis (40, 47), we anticipate that a combined metabolic MRI protocol with these safe, nonionizing HP tracers could provide a powerful alternative to the current radioisotope-based imaging for treatment optimization.
Inflammation and infections
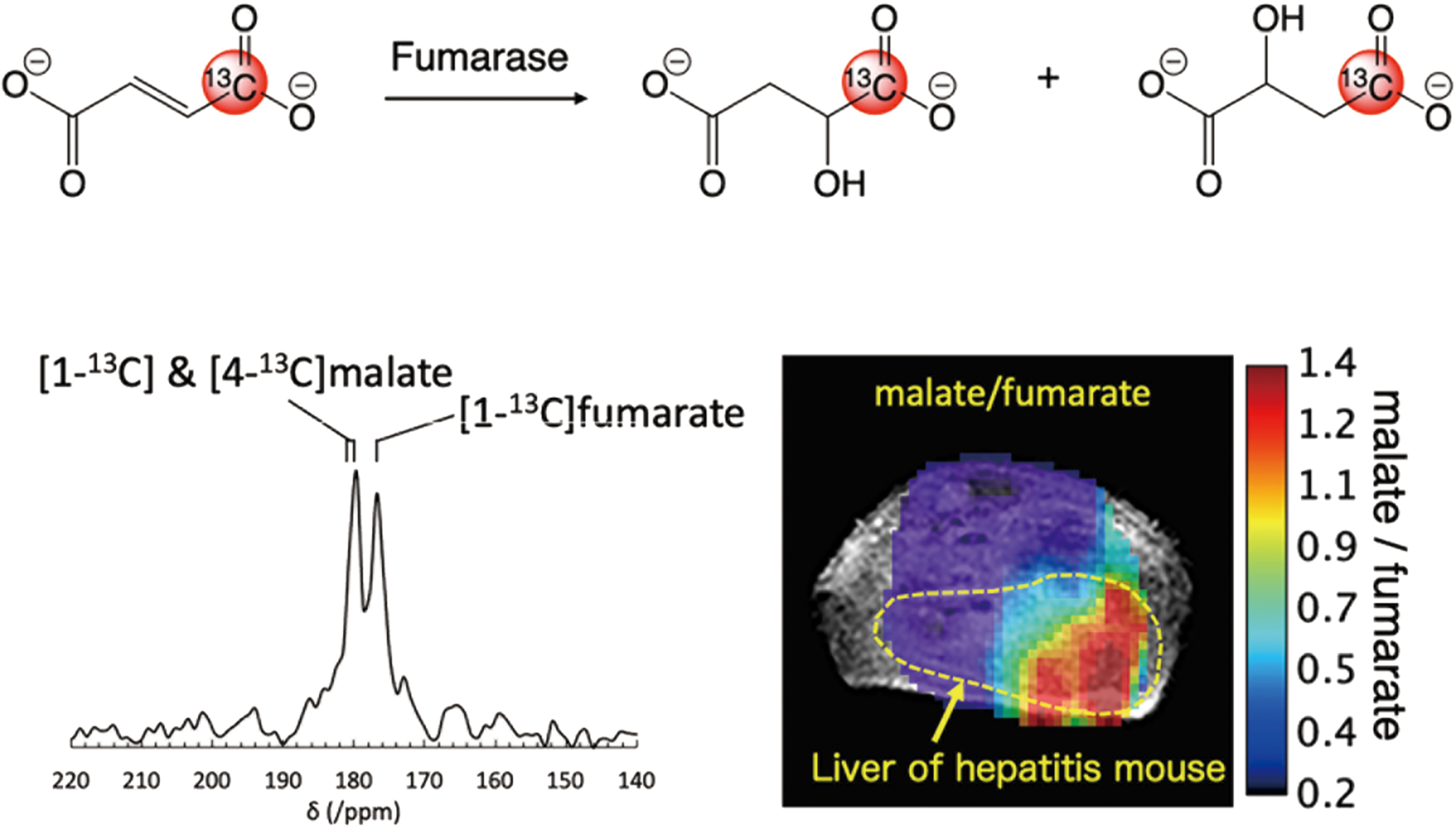
Excess generation of ROS in inflammatory diseases diminishes the antioxidant capacity of the cell and can induce redox-dependent alterations to metabolism. Wilson et al. reported that the diminished liver antioxidant capacity in a mouse model of nonalcoholic steatohepatitis can be estimated from the reduction rate of HP [1-13C] DHA to AA (73). As discussed earlier, this conversion is known to occur in a GSH-dependent manner, catalyzed by redox-regulating enzymes, including glutaredoxin and glutathione transferases. The severity of hepatitis can be also evaluated by the extent of cell death using HP 13C fumarate. We recently reported a feasibility study of in vivo necrotic cell death imaging in an acetaminophen-induced hepatitis model using HP [1-13C] fumarate, which was prepared by PHIP, a relatively low-cost alternative polarization technique to d-DNP, with a trans-selective hydrogenation catalyst (67). Localization of the necrotic cell death biomarkers [1-13C] and [4-13C] malate was observed only at the liver region of hepatitis mice when compared with healthy mice (Fig. 7).
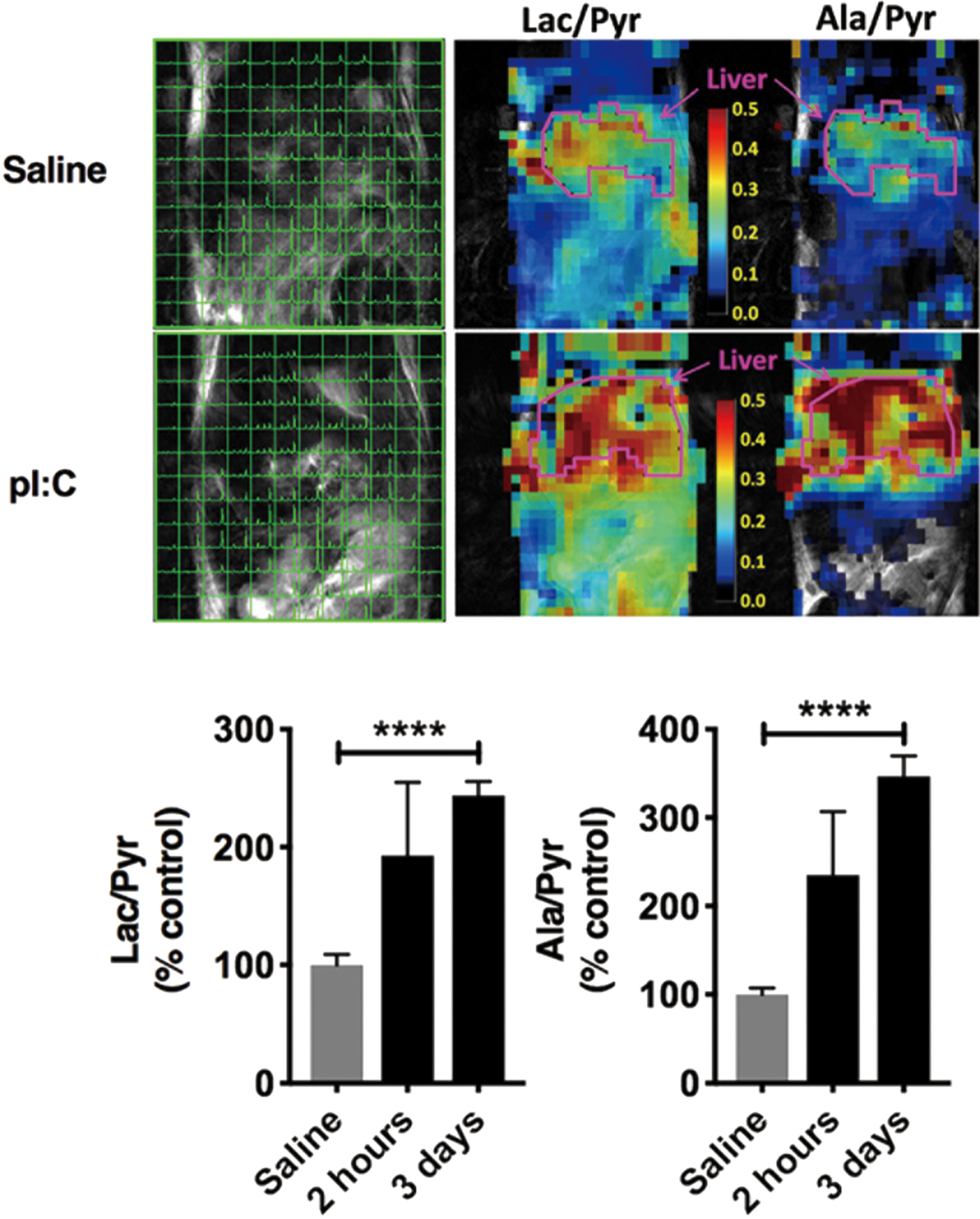
The human coronavirus-2 (HCoV-2), also known as severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2), is an enveloped positive-sense and single-stranded RNA virus causative of coronavirus disease (COVID-19) and led to the present global pandemic. The excess activation of the immune system due to viral infection and resultant acute burst of oxidative stress lead to a cytokine storm in COVID-19 patients, similar to Influenza viral infection, and which may lead to multiple organ failure and, ultimately, death. SARS-CoV-2, similar to most of the other viruses, requires host cells to provide the building blocks for viral replication, and it achieves this by rewiring host cell metabolism, including an increase of glucose carbon entry into the TCA cycle via pyruvate carboxylase expression, and a decrease in oxidative glutamine metabolism (50). The mTORC1, one of two protein complexes of mTOR, may be involved in the viral infection-induced metabolic alteration. Tarasenko et al. recently reported that HP 13C MRI of pyruvate metabolism can visualize the mitochondrial metabolic alterations induced by injection with polyinosinic:polycytidylic acid (poly I:C), which chemically mimics RNA virus infection, and that the metabolic alteration was suppressed in mTOR knockout mice (Fig. 8) (69). These reports suggest that HP 13C metabolic MRI of redox-sensitive 13C tracers can provide a useful clinical tool to investigate the underlying mechanism of redox alteration in inflammation and infection, and so establish strategies for therapeutic intervention.
Future prospects
HP 13C MRI, wherein more than 10,000 times enhancement of the MRI signal of 13C-labeled redox-sensitive metabolic tracers can be realized, has enabled real-time, noninvasive imaging of metabolism in vivo and opened new research avenues for the field of redox biology. Although more than 60 13C metabolic tracers have been reported and attempts have been made at developing specific tracers for detecting ROS (60), to date only a few metabolic tracers have been established for monitoring tissue redox state. As such, future development of new redox-sensitive metabolic tracers and a comprehensive understanding of the correlation between metabolic alterations and the redox state are essential. Recent progress in cost-effective parahydrogen-induced polarization techniques, in addition to conventional d-DNP type methods, may provide a relatively easier entry point for researchers in the redox field to begin HP 13C MRI studies, and additional avenues for novel HP 13C tracer development.
Footnotes
Acknowledgments
The authors acknowledge funding support from AMED (Grant No. JP20hm0102061) and the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant No. 20H006540).
Authors' Contributions
N.T. and T.S. contributed to the writing of the “Potential applications in heart diseases” section in this review article. All other sections were written by N.J.S., H.H., and S.M.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research was supported by AMED (Grant No. JP20hm0102061) and the Japan Society for the Promotion of Science (JSPS) KAKENHI (Grant No. 20H006540) to Norihiko Takeda and Shingo Matsumoto.
