Abstract
Professor Valerian Kagan (PhD, 1972, MV Lomonosov Moscow State University; DSci, 1981, USSR, Academy of Sciences, Moscow) is recognized as a Redox Pioneer because he has published 4 articles in the field of redox biology that have been cited >1000 times and 138 articles in this field have been cited between 100 and 924 times. The central and most important impact of Dr. Kagan's research is in the field of redox lipidomics—a term coined for the first time by Dr. Kagan in 2004—and consequently the definition of signaling pathways by oxidatively modified phospholipids; this acquires further significance considering that oxygenated phospholipids play multifunctional roles as essential signals coordinating metabolism and physiology. Some examples are the selective oxidation of cardiolipin (CL) by a cytochrome c peroxidase activity leading to the activation of the intrinsic apoptotic pathway; the hydroperoxy-arachidonoyl/adrenoyl phosphatidylethanolamine (PE) species, driven by 15-lipoxygenases (15-LOX), as death signals leading to ferroptotic cell death; the regulation of ferroptosis by iNOS/NO• in pro-inflammatory conditions by a novel mechanism (realized via interactions of 15-LOX reaction intermediates formed from arachidonoyl phosphatidylethanolamine [PE] species) and Ca2+-independent phospholipase A2 (iPLA2β; via elimination of peroxidized PE); the involvement of oxygenated (phospho)lipids in immunosuppression by myeloid cells in the tumor microenvironment; hydrolysis of peroxidized CL by Ca2+-independent phospholipase A2 (iPLA2γ) leading to pro- and anti-inflammatory signals and lipid mediators. Kagan continues his investigations to decipher the roles of enzyme-linked oxygenated phospholipids. Antioxid. Redox Signal. 36, 813–823.

“The search for truth is more precious than its possession.”
–––Albert Einstein
“There is no such thing as boring science: Mother Nature discloses no secrets to idle minds”
–––Valerian E. Kagan
Background, Training, and Development
V
Moscow
At the dawn of the 20th century, social injustice, unrest, and uprisings by workers, peasants, and soldiers—driven by the Marxist ideas that “Proletarians have nothing to lose but their
The two most prominent names (and the Nobel Prize award) in this field were Sir Cyril Hinshelwood and Nikolay Semenov. Semenov demonstrated that during chemical reactions, atoms and molecules regroup and form new constellations. When molecules formed during a reaction readily react with molecules present from the beginning, a chain reaction can occur. Chain reactions commonly proceed via the intermediate formation of free radicals. Elementary steps that generate more free radicals than they consume are called branching reactions. Semenov demonstrated that such reactions formed by far the biggest part of gas phase reactions (56).
Semenov founded the Institute of Chemical Physics in Moscow and, jointly with his many followers and students, demonstrated the importance of chain reactions in the liquid phase. One of them, N. Emanuel, showed that paraffins and cycloparaffins are all products of reaction formed due to the transformation of the primary intermediates, hydroperoxides (9). Because of the low decomposition energy in the O-O bond (30–40 kcal), the hydroperoxides split relatively easily and at the same time formed reactive free radicals. This type of chain reactions was called “degenerate branching.” Later, N. Emanuel transferred the ideas about degenerate branching reactions of liquid phase organic oxidations into the field of biology and suggested that these reactions may occur after the exposure to ionizing radiation and also in cancer (10).
These frontier chemical ideas strongly affected a young student just admitted to the Program in Biophysics at the MV Lomonosov Moscow State University (1963), Valerian Kagan. The Chair of the Biophysics Department, Dr. B. Tarusov, was a big enthusiast and “propagator” of the free radical concepts in biology, particularly in radiobiology. As Valerian was reading the Russian and Western publications in these fields, these exciting new concepts resonated in his mind with the older work by a famous Russian biochemist, Alexei Bach, who proposed the peroxide theory of respiratory processes.
As an MS student (1967–1968) working on his research project at the Institute of Chemical Physics in Emanuel's department of kinetics of chemical and biological processes, V. Kagan and his mentor, E. Neyfakh, attempted to detect degenerate branching chain reactions and their intermediates, lipid hydroperoxides, in lipid bilayers of cells and tissues.
The analytical study of this type has not been done before. After trying several titration protocols, Valerian chose iodometric titration of hydroperoxides. The sensitivity of this reaction using standard colorimetric protocols was not sufficient. After many attempts, the protocol was changed to a far more sensitive amperometric titration, with the dead-stop registration of the endpoint. With many control experiments performed, this yielded the first results demonstrating the presence of hydroperoxy-lipids in tissues of normal animals and their elevated levels in organs of tumor-bearing mice (50, 51).
The sensitivity of amperometric titration was insufficient in detecting hydroperoxy-intermediates in subcellular organelles and in different classes of phospholipids. One method at this time was polarography invented by Heyrovsky and Kuta (18). The method used two electrodes, one of which is a dropping mercury electrode. The technique was implemented in the laboratory of physical chemistry of biomembranes at MV Lomonosov Moscow State University where V. Kagan (1970–1971) was a junior researcher. The sensitivity and specificity of the polarographic technique were remarkable and, indeed, resulted in the quantitative measurements of lipid hydroperoxides in different classes of phospholipids isolated from different cells and their organelles (5, 12).
This work continued for more than a decade and resulted in several discoveries on the fascinating roles of lipid hydroperoxides in normal physiology and injuries. Among them were discovered mechanisms of high-light intensity damage of the retina photoreceptors and understanding of the molecular mechanisms of peroxidation induced Ca2+-permeability and modifications of Ca2+-ATPase in sarcoplasmic reticulum membranes of skeletal muscles (22, 58).
In 1972, Kagan graduated with his PhD degree in biophysics and biochemistry from the MV Lomonosov University and continued his work there. In 1982, he obtained his DSci degree from the USSR Academy of Sciences.
The research topics in free radical biology and antioxidants were becoming very popular in the USSR and were studied in several academic laboratories, including Institute of Chemical Physics, MV Lomonosov University, and NI Pirogov 2nd Medical University in Moscow. In 1983, the direction of research on the role of free radical lipid peroxidation in health and disease in these three institutions and several leading scientists, including Valerian Kagan, were awarded the prestigious and coveted “State Prize of the USSR for Science.” While in Moscow and Sofia, Valerian Kagan published 184 articles, a few examples in Refs. (23, 25, 37 –39, 44, 45).
Sofia
Despite his scientific excellence Kagan had great difficulty travelling to scientific meetings beyond the Soviet bloc. However, Valerian Kagan was invited to organize a new laboratory (Biomembrane Stabilization) at the Institute of Physiology in Sofia (in the Bulgarian Academy of Sciences). He selected a group of young enthusiastic students and junior researchers attracted by the ideas of understanding the mechanisms of free radical reactions and their antioxidant regulation. Dr. Kagan's former restrictions of contacts, visits, and joint research with the colleagues in the West were lifted when he moved to Bulgaria. In 1985, Dr. Kagan initiated joint projects with Professor Quinn at King's College, London.
This joint work established that antioxidant propensities of vitamin E and Coenzyme Q are inherently linked to their nonantioxidant functions in stabilizing biomembranes from natural chaotropic agents—free fatty acids and lysophospholipids. From the discussions with the colleagues from Western Europe, America, and Asia, Kagan realized that the work of the Russian scientists remained largely unknown to them. Therefore, he decided to summarize the major results in a book “Lipid Peroxidation in Biomembranes” published in 1988 (CRC Press, Boca Raton, FL).
Berkeley
Attendance at international meetings and discussions of topics of joint interest, as is so often the case, led Valerian Kagan to visit and eventually move to Lester Packer's Lab at the University of California at Berkeley and Lawrence Berkeley National Lab in 1988.
The Berkeley period was very important for Kagan's career and exposure to international rather than East European science. His work on vitamin E, published in Methods Enzymology, received a huge recognition boost (31, 70). By developing and applying different electron paramagnetic resonance (EPR) protocols, he was able to uncover enzymatic mechanisms of vitamin E recycling from its phenoxyl radicals and uncovering mitochondrial electron-transport chain reactions with α-tocopherol and the requirement of ubiquinol (47, 48) and microsomal membranes (53). By that time, the reduction of the vitamin E radical by ascorbate has been established by Trevor Slater's laboratory (52).
Pittsburgh
After a few years of very productive work at Berkeley, Kagan was offered an academic position at the University of Pittsburgh. He and his family moved to Pittsburgh. On a more personal level, adjusting to the open yet highly competitive environment in Berkeley and dealing with family, schools and all the work that is entailed, for Dr. Kagan to get the opportunity to be a professor at the University of Pittsburgh after just 3 years as an immigrant was remarkable. Alas once again he had to start from scratch, building his laboratory recruiting good graduate students and postdocs and paying for them. This time he had learned much about academic science in the United States and of course he had a huge knowledge of lipid and lipid peroxides.
In Pittsburgh, it was not long before he obtained NIH grants and he developed his laboratory and the Center for Free Radical and Antioxidant Health. Given his appointment in the Department of Environmental Health Sciences, Dr. Kagan extended his research and EPR measurements and initially focused on the detection of phenoxyl radicals of several relevant phenolic compounds, including phenol, etoposide, and pentamethylchromanol. These studies were among the first that reported on the identification of EPR detectable signals from living cells, particularly in cells with high levels of expression of myeloperoxidase (e.g., human myelogenous leukemia cells) (28, 29). In later years his research has focused on deciphering of oxidized phospholipid signaling in cells as explained in greater detail hereunder.
Description of Key Finding 1
Vitamin E recycling
While in Sofia and Berkeley, Dr. Kagan's work was focused on studies of two major natural antioxidants, vitamin E and Coenzyme Q. One of the major goals was to decipher nature's strategies in creating these effective membrane protectors. Working with different types of natural and synthetic biomembranes and a number of homologues of these compounds, particularly those with the variable chain lengths, Kagan's group (that included Drs. E. Serbinova, D. Stoyanovsky, and R. Bakalova in Sofia) formulated principles of optimization of these molecules leading to the realization of the maximal radical scavenging activity of their chromanol and benzoquinol groups with the preservation of the necessary membrane stabilizing propensities.
Through productive interactions with Drs. R. Mehlhorn and J. Maguire in Berkeley, Kagan established that members of the vitamin E family, tocopherols and tocotrienols, act as antioxidant “harvesting” centers of biomembranes through their ability to accept the reducing equivalents from the mitochondrial and endoplasmic reticulum electron carriers, including coenzyme Q (57, 67). Study on vitamin E extended to inhibition of smooth muscle cell proliferation by tocopherols and tocotrienols (11), recycling of vitamin E in human low-density lipoproteins (33), ultraviolet-light-induced formation of vitamin E radicals in skin (41), intermembrane transfer and antioxidant activity of α-tocopherol in liposomes (24), and binding of iron to α-tocopherol in liposomes (64).
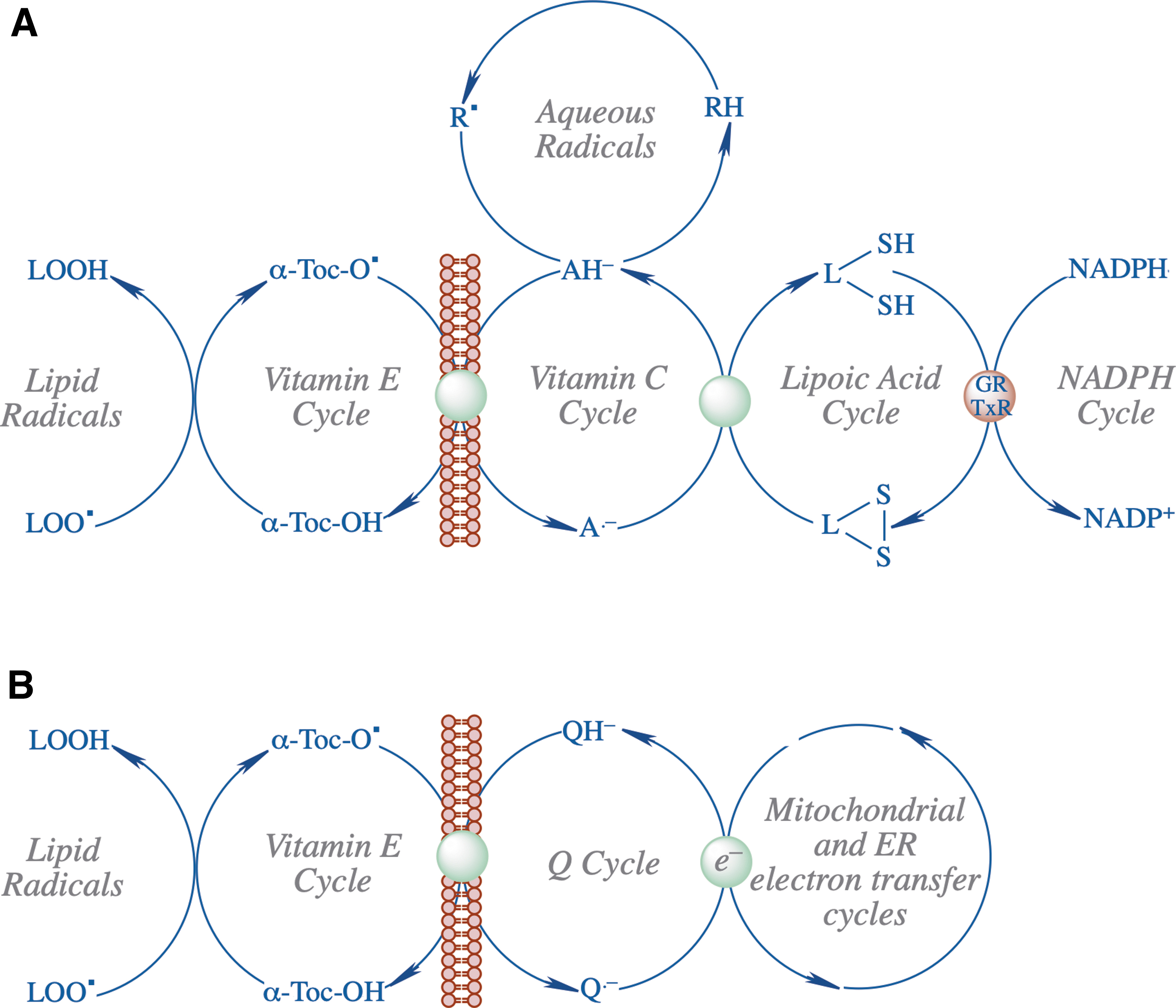
In addition to vitamin E, Kagan was also involved in determining the antioxidant properties of ubiquinone and ubiquinol (34, 35) and comparison of the radical scavenging activity of carotenoids with α-tocopherol (71). A notable finding related to vitamin E tocopheroxyl radical was its interaction with the ascorbate/dihydrolipoate system and the demonstration of the remarkable efficiency of dihydrolipoate in recycling vitamin E in biomembranes and lipoproteins (36) (Fig. 1A) as well as the recovery of the vitamin E radical by ubiquinone (Q cycle) (47) (Fig. 1B).
Description of Key Finding 2
Redox lipidomics and signaling by oxidatively modified phospholipids in programmed cell death
The central and most important impact of Dr. Kagan's research is in the field of redox lipidomics and signaling by oxidatively modified phospholipids. In fact, the term redox (or oxidative) lipidomics was coined by Kagan and has been widely used since then (26, 32). Paradoxically, these studies have stemmed from the disappointment in the understanding of the role and significance of free radical reactions in normal physiology and disease pathogenesis. One of the most popular ideas of that time proposed that free radicals randomly formed in nonenzymatic chemical reactions in cells, tissues, and biofluids are damaging and toxic and their levels are strictly controlled by antioxidants, a group of water-soluble and lipid-soluble molecules readily donating an electron to a free radical (19).
Among endogenous molecules of this type a lot of work has been done with vitamins, particularly vitamin E and vitamin C, as well as with thiols (61). Two other popular groups of chemicals with the electron-donating capacity included phenols and aromatic amines. Simple in vitro studies in model biochemical systems, isolated organelles, cells, and experiments in animals were all indicative that overwhelming free radical reactions and antioxidant deficiency may be causative to the pathogenesis of several major diseases, including neurodegenerative and cardiovascular diseases, cancer, and aging (16).
One of the big attractions was the simplicity and testability of the concept: oxidative stress and insufficiency of antioxidants as the cause of cell and tissue injury and death leading to aberrant redox metabolism and disease conditions. If so, and uncontrolled free radicals are the major drivers—then antioxidants represent a wonderful panacea potentially realizable into therapeutic strategies against several major disease conditions. This clarity and simplicity of the concept has generated intellectual enthusiasm, which, along with the apparent success of animal studies, has led to the initiation of several large-scale antioxidant clinical trials. The results of these expensive studies turned to be less than encouraging and suggested that the concept required a serious reconsideration.
Since the early 1950s, it has been known that several enzymatic pathways catalyzed by different isoforms of lipoxygenases (LOX), cyclooxygenases (COX) and cytochrome P450s can oxidize free polyunsaturated fatty acid (PUFA) to yield specific oxidation products that can act as lipid mediators—both intra- and extracellular signals (15). Effective formation of the PUFA precursors for these oxygenation reactions is achieved via the hydrolysis of PUFA-phospholipids by highly specific phospholipases A2 (PLA2). In the esterified form, oxidized PUFA-phospholipids are not readily utilized by the majority of types of PLA2 with a few exceptions, such as platelet activating factor or representatives of PLA2G6 family (Ca2+-independent PLA2β and PLA2ɤ).
These ideas prompted Dr. Kagan to study enzymatic reactions of lipid peroxidation. A prerequisite for this was the development and use of new protocols of high-resolution and ultrasensitive liquid chromatography–mass spectrometry (LC–MS) protocols. This was essential because of the very low abundance and extremely high diversity of peroxidized phospholipids. Given the focus of cell biology on programmed cell death at that time (in the mid-2000s) the choice was made to explore signaling by oxidized phospholipids and their role in select programs of cell death (Fig. 2). This has led to a success—oxidized cardiolipins (CL) were identified as required intermediates of an intrinsic apoptotic program (26). It was further established that CL oxidation was accomplished by the “reassignment” of cytochrome c as a peroxidase (40).
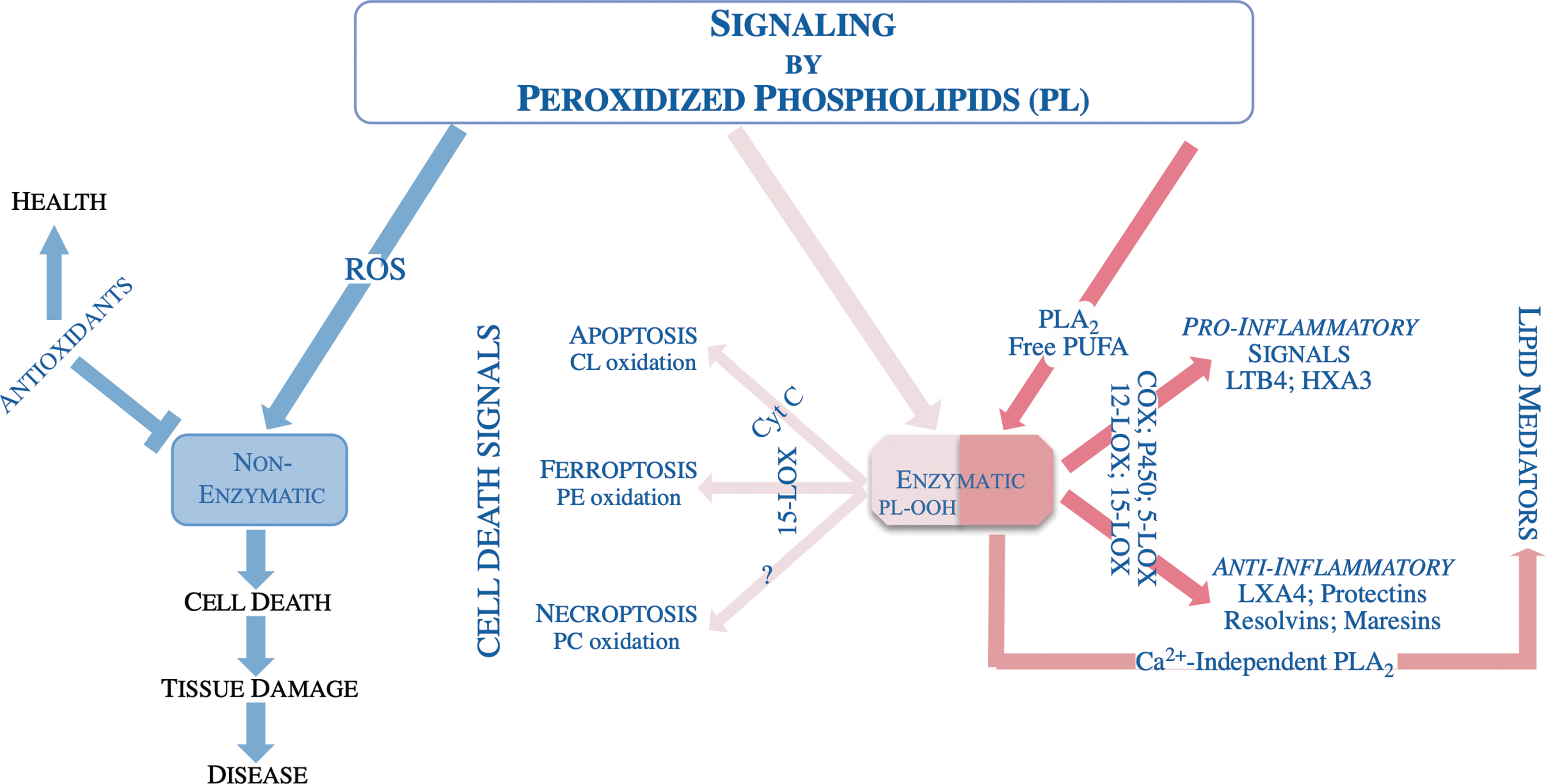
It was shown that upon arrival of the apoptotic stimulus the asymmetric distribution of the CL in mitochondria changes with more CL appearing in the outer side of the inner membrane facilitating a complex formation between CL and cytochrome c. Binding of cytochrome c with CL changes its normally hexacoordinated structure where the coordination bond between heme iron and Met 80 is disrupted allowing available H2O2 or hydroperoxy-PUFA access to the Fe-porphyrin catalytic site of a modified cytochrome c-CL complex (8, 42, 46). The formation of the cytochrome c/CL complex causes a strong negative shift of its redox potential (by ∼400 mV) such that cytochrome c can no longer act as an electron acceptor in the respiratory chain resulting in increased superoxide formation and their dismutation to H2O2 (7).
The latter can be used as a source of oxidizing equivalents for a peroxidase reaction. Liberation of CL's oxidized acyl chains through the action of phospholipases (e.g., Ca2+-independent phospholipase A2 [iPLA2γ]) yields a suite of bioactivate oxylipins (73). These studies—revealing the CL and cytochrome c interactions and the peroxidase function of the complex data—paved the way for design and synthesis of small molecule antiapoptotic regulators with therapeutic potential in a number of disease states where oxidative stress and apoptosis play a pathogenic role (6, 20, 21).
In a subsequent series of studies, it was established that selective peroxidation of yet another phospholipid phosphatidylethanolamine (PE) was a component of the ferroptotic cell death program (30). Among the thousands of possible oxidizable phospholipids arachidonoyl- and adrenoyl-containing precursors were identified as the substrates and their hydroperoxy-derivates as the products leading to ferroptotic death. The specificity of the oxidation products implied that an enzymatic mechanism played a role in initiating ferroptosis.
It was initially thought that 15-LOX is the enzyme responsible for PE oxidation during ferroptosis. However, 15-LOX normally oxidized free arachidonic acid as a preferred substrate. It was shown that PE binding protein 1 (PEBP1) forms a complex with 15-LOX and changes its substrate competence to generate hydroperoxy-PE (3, 77). This has led to yet another discovery of “theft ferroptosis” in which cell death is caused by a Gram-negative bacterium Pseudomonas aeruginosa expressing 15-LOX as a virulence factor; notably bacterial 15-LOX does not require PEBP1 for generating the pro-ferroptotic signals (13, 14).
Further studies shed light on the regulation of ferroptosis by NO•-driven nitroxygenation of arachidonoyl-PE intermediates and hydrolysis of hydroperoxy-PE by Ca2+-independent phospholipase A2 (iPLA2β) (43, 66). Elucidation of the mechanisms of PE peroxidation as drivers of the ferroptotic death program is now leading to the design of new classes of specific small molecule inhibitors as well as better understanding of the mechanism of action of known antiferroptotic agents (4). These briefly described studies took ∼15 years to accomplish. One can imagine there is still huge amount of work to decipher many other roles of oxygenated phospholipids.
Description of Key Finding 3
Role of oxygenated phospholipids on immunosuppression
Another area of signaling by oxygenated phospholipids focuses on immunosuppression by immune cells in the tumor microenvironment (17, 49, 72, 75, 76). In collaboration with Dr. Gabrilovich, a cancer immunologist, Dr. Kagan has shown that oxygenated PUFA triacylglycerols and free PUFA generated by myeloid-derived suppressor cells are taken up by dendritic cells where they are deposited in lipid droplets. This process disrupts cross-presentation of tumor antigens to T cells by dendritic cells (55, 74, 75). One of the latest discoveries is related to the awakening of dormant tumor cells by neutrophil metabolites of polyunsaturated plasmalogens (54) (Fig. 3).
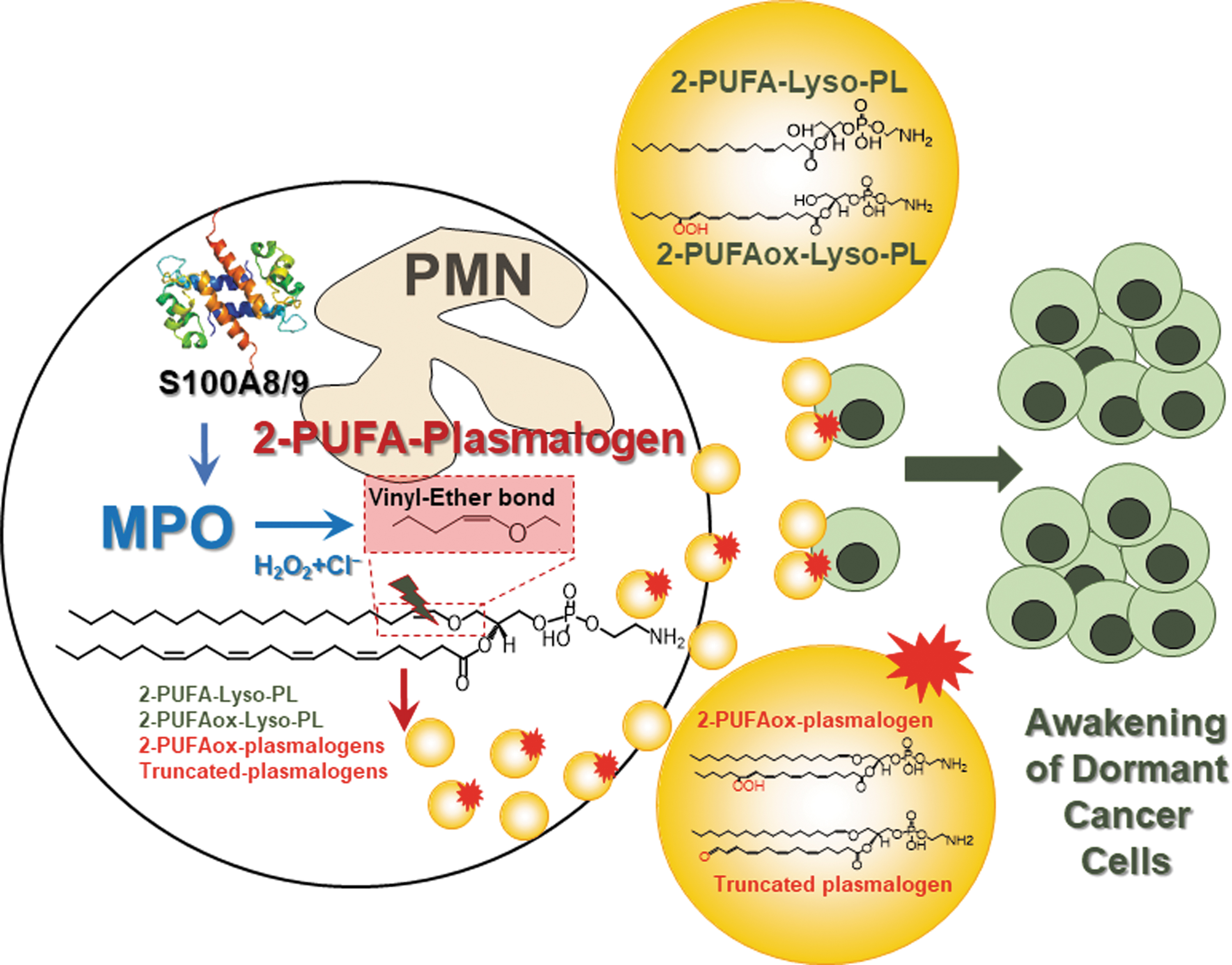
Awakening and reactivation of dormant tumor cells result in tumor recurrence years after seemingly successful treatment of primary tumor. It was shown that stress hormones cause release of proinflammatory S100 proteins leading to activation of myeloperoxidase in neutrophils. Myeloperoxidase-derived oxidized lipids are then released by neutrophils resulting in upregulation of fibroblast growth factor pathway in dormant tumor cells. Thus, depicting neutrophils are a tumor's alarm clock and reactivation of dormant tumor cells is mediated by modified lipids.
Other Important Findings
In addition to LC–MS-based redox lipidomics approach Dr. Kagan has made important contributions to spatial biology of phospholipids and their oxidation products using different imaging mass spectrometry techniques, including matrix-assisted laser desorption ionization (1, 62, 63, 65, 68, 69). These studies advanced the mass spectrometry imaging of phospholipids by the development of new matrices for improved spatial resolution (65) and by the development of methods for label-free mapping of CL spatial distribution in cells and tissues for the first time by eliminating ion suppressive effects of more abundant phospholipids such as phosphatidylcholine (1, 65, 68, 69).
Latest achievement in this area was the subcellular mapping of low abundance ferroptotic oxidized PE signals in cells and tissues in disease conditions (63). These studies expand our understanding of signaling roles of different phospholipids in the context of their production in different intracellular compartments and intercellular communications.
A separate important new research development for Dr. Kagan was in the field of nanotoxicology (2005 to present). After the pioneering demonstration of the unusual early onset of fibrosis in response to the exposure of mice to carbonaceous nanomaterials (e.g., single-walled carbon nanotubes) by Dr. Shvedova et al. (59), Kagan's Lab discovered that redox machinery of innate immune cells (myeloperoxidase in neutrophils, iNOS/NADPH oxidase in macrophages) effectively biodegraded nanomaterials (2008 to present) (2, 27, 60). This series of high-impact article have gathered thousands of citations and opened an entirely new direction of research in nanotoxicology, which at the time of this writing is represented by hundreds of new publications per year (Supplementary Table S1).
Current Position and Adjunct Appointments
Dr. Kagan continues his leadership role as a Professor and Vice Chair of Research at the Department of Environmental and Occupational Health and Director of Center for Free Radical and Antioxidant Health at the University of Pittsburgh. He also holds appointments as Professor at the Departments of Chemistry, Pharmacology and Chemical Biology and Radiation Oncology at the University of Pittsburgh.
In addition to these positions, Dr. Kagan has had adjunct appointments as Visiting Professor in the Division of Life Sciences, King's College, London, United Kingdom; Adjunct Foreign Professor in the Institute of Environmental Medicine, Karolinska Institute, Stockholm, Sweden; Foreign Professor in Taipei Medical University, Taiwan, Foreign Professor in Russian State Medical University, Moscow, Russia; Foreign Professor in MV Lomonosov Moscow State University, Moscow, Russia; Fulbright Visiting Chair in Environmental Sciences, McMaster University, Hamilton, Canada; Visiting Professor at the University of Tel-Aviv, Israel; Head, Laboratory of Navigational Redox Lipidomics, IM Sechenov State Medical University, Moscow, Russia; Professor-Consultant, Institute for Regenerative Medicine, IM Sechenov State Medical University, Moscow, Russia.
Future Look
One can imagine there is still a huge amount of work to decipher other roles of enzyme-linked oxygenated phospholipids. As Dr. Kagan, who is in his mid-70s, is full of creative energy and we are sure that many new discoveries will still be forthcoming. In the meantime, his published work has attracted significant attention garnering him awards from the Society for Free Radical Research Europe and the Oxygen Club of California and has resulted in many high-profile publications yielding Dr. Kagan's Hirsh-factor of 109 and hi-factor of 468.
In conversations with Dr. Kagan we have heard many times that his way of scientific thinking and conceptualizing things have originated from many discussions he has had with colleagues in the field of redox biology. Having come from Eastern Europe many things had to be rethought and reanalyzed, both directly by a scientific and organizational nature. In closing we would like to also mention the theatrical and dramatic style of Dr. Kagan in giving seminars and invited talks perhaps taking roots from his very early career in acting as Sergei Karenin in Lev Tolstoy's Anna Karenina in one of the most famous Russian theatrical organizations, the Moscow Art Theatre (founded by Konstantin Stanislavsky and Vladimir Nemirovich-Danchenko in 1898). We wish him continued success on the stage of science.
Footnotes
Acknowledgments
Dr. Kagan writes, “I have been involved in research for over 60 years, since the time that I started as a Lab technician and slowly transitioning to an independent researcher and lab director. This is a very long way and clearly there were many failures and more rare moments of success. While I am fully responsible for the former, many colleagues helped me to navigate through numerous doubts towards understanding the mechanisms of biomedical phenomena and the scientific truth.
Richard Feynman noted: ‘Religion is a culture of faith; Science is a culture of doubt.’ In this very short Acknowledgement section, I cannot mention even a small fraction of wonderful people with whom, through discussions and sometimes joint research, I adopted the culture of the scientific doubt. I would like to mention at least several names of those who affected my research: Lester Packer, Bruce Ames, Lars Ernster, Sten Orrenius, Sergey E. Severin, Vladimir P. Skulachev, Yuri A. Vladimirov, Mikhail Sitkovsky, Etsuo Niki, Felix Meerson, Barry Halliwell, Peter Quinn, Catherine Pasquier, and Dmitry Gabrilovich.
Having come from Eastern Europe many things of a scientific and organizational nature had to be rethought and re-analyzed. I would like to express my appreciation to all of my colleagues, postdoctoral fellows, and students who made invaluable contributions to my research programs throughout my scientific career at the MV Lomonosov State University, Bulgarian Academy of Sciences, University of California Berkeley and University of Pittsburgh among them, are Drs. Vladimir and Yulia Tyurin with whom I've been working for more than 30 years.
I wanted also to thank three excellent scientists, Drs. Hülya Bayır, Enrique Cadenas and late John Maguire who nominated me and worked hard on the Redox Pioneer commentary. I am thankful but also humbled by this nomination because as a scientist I realize how little our work has explained and I perceive this as a credit and an encouragement to work harder.”
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The authors wish to acknowledge the University of Pittsburgh Medical Canter Endowed Chair, National Institute of Health grants AI156923, NS061817 and NS076511.
Supplementary Material
Supplementary Table S1
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
