Abstract
Significance:
Four decades have passed since the first successful human embryo conceived from a fertilization in vitro. Despite all advances, success rates in assisted reproduction techniques still remain unsatisfactory and it is well established that oxidative stress can be one of the major factors causing failure in in vitro fertilization (IVF) techniques.
Recent Advances:
In the past years, researchers have been shown details of the supportive role CCs play along oocyte maturation, development, and fertilization processes. Regarding redox metabolism, it is now evident that the synergism between gamete and somatic CCs is fundamental to further support a healthy embryo, since the oocyte lacks several defense mechanisms that are provided by the CCs.
Critical Issues:
There are many sources of reactive oxygen species (ROS) in the female reproductive tract in vivo that can be exacerbated (or aggravated) by pathological features. While an imbalance between ROS and antioxidants can result in oxidative damage, physiological levels of ROS are essential for oocyte maturation, ovulation, and early embryonic growth where they act as signaling molecules. At the event of an assisted reproduction procedure, the cumulus/oophorus complex is exposed to additional sources of oxidative stress in vitro. The cumulus cells (CCs) play essential roles in protecting the oocytes from oxidative damage.
Future Directions:
More studies are needed to elucidate redox biology in human CCs and oocyte. Also, randomized controlled trials will identify possible benefits of in vivo or in vitro administration of antioxidants for patients seeking IVF procedure.
Introduction
Cumulus cells (CCs) play essential roles in the oocyte's growth and maturation processes. For example, they protect the oocyte from oxidative stress damage (116) and cope with substrates that the oocyte is incapable of metabolizing (7). In the follicle, oocyte and CCs maintain an intense bidirectional communication by metabolite exchange in several biological processes. The direct communication via gap junctions allows the two cell types to exchange small molecules and ions (7,30,75). However, the CC-oocyte communication goes beyond gap junction transfer and involves oocyte-secreted factors that drive the paracrine signaling in CCs, regulating in a loop manner the CC metabolism (29). This bidirectional communication orchestrates the oocyte and follicle growth, maturation, and ovulation processes (29, 94, 95, 111). The CCs are the sensors to follicle and oocyte health, and are capable of modulating the microenvironment in response to specific demands (25).
Reactive oxygen species (ROS) are produced in the healthy follicle during physiological processes and are important for oocyte maturation (10, 32). Despite their essential role, an excessive production of ROS can be detrimental to the follicle, affecting its oocyte maturation (17, 51). As a result of oxidative stress, an exacerbated inflammatory reaction is generated in the oocyte and also in the CCs, inducing an imbalance in growth factor and cytokine production leading to a detrimental effect on reproduction (5). These insults can come from the external environment, provoked by patients' lifestyle, from the inner follicle microenvironment, originating from dysfunctional CCs, or from the oocyte itself, caused by aneuploidies or other defects of the gamete. Either way, those insults can impact on CC's redox metabolic functioning that can, therefore, act as a gatekeeper for oocytes' developmental potential. This review focuses on the supportive role of CCs, on the relationship between redox imbalance and oocyte quality, on the impact of female reproductive pathologies on oxidative stress in the cumulus/oocyte complex (COC), and suggests clinical applications of proposed biomarkers of redox activity. We also make suggestions of antioxidant management in the clinical environment. Oxygen (O2) radicals in reproduction (91), or oxidative stress in the female reproductive tract (2), on female infertility (4, 45), in the oocyte (26), in the oocyte during in vitro maturation (19, 59) and in the sperm (122) are or were earlier discussed elsewhere and are not addressed in this review.
The Intimate Relationship of CCs with Oocytes
In the mammalian ovary, oocytes are contained inside follicles, structures that when matured are composed of somatic granulosa cells and filled with follicular fluid (FF). In the antral follicle, the cumulus oophorus, a specialized subgroup of granulosa cells, are surrounding the gamete (Fig. 1) (22). CCs differentiate from mural granulosa cells by the action of oocyte-secreted factors and ovarian hormones (41). The most inner CC layers, called corona radiata, are in direct contact with the oocyte through transzonal projections. These extensions of the granulosa cells transgress the oocyte's zona pellucida and form specialized junctions with the oolemma (76).
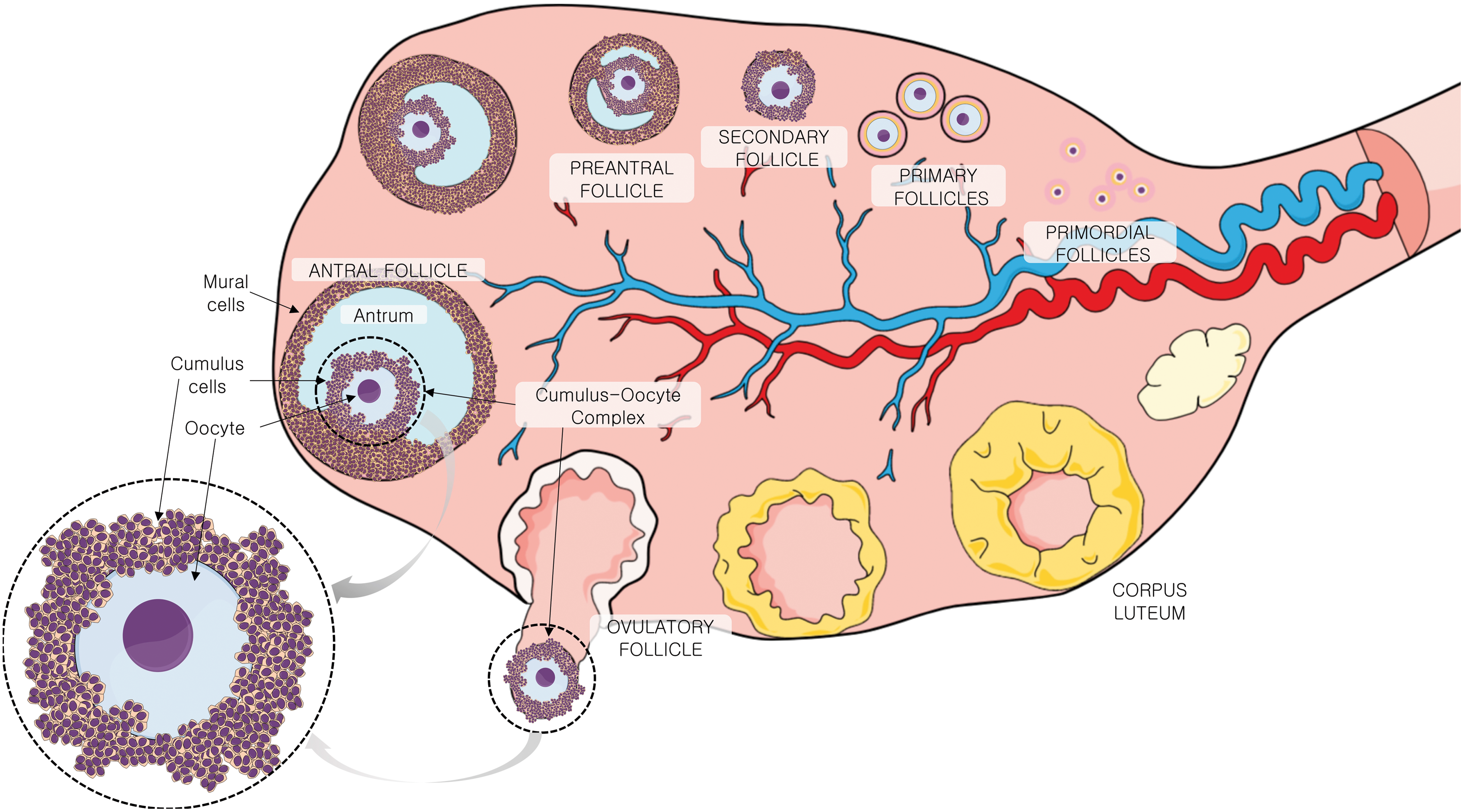
Together, CCs and oocyte form the COC, located inside the antral follicle in contact with FF, COCs are surviving in an avascular compartment. The FF is a plasma-like fluid, originating from the plasma (25) and constituting a source of COC metabolites, small signaling molecules, proteins, ROS, and antioxidants (38). The CCs are the gatekeepers for the oocyte with its surroundings. Thus, the CCs act as a biological barrier that selects and processes the metabolites that oocyte will receive.
Oxidative Phosphorylation Is the Source of Energy and Biosynthesis for the Oocyte
The female reproductive tract is considered a hypoxic environment, with O2 concentration variating between 2% and 8% (80) (Fig. 2), while the antral follicle is exposed to O2 concentrations between 1% and 4% (55).
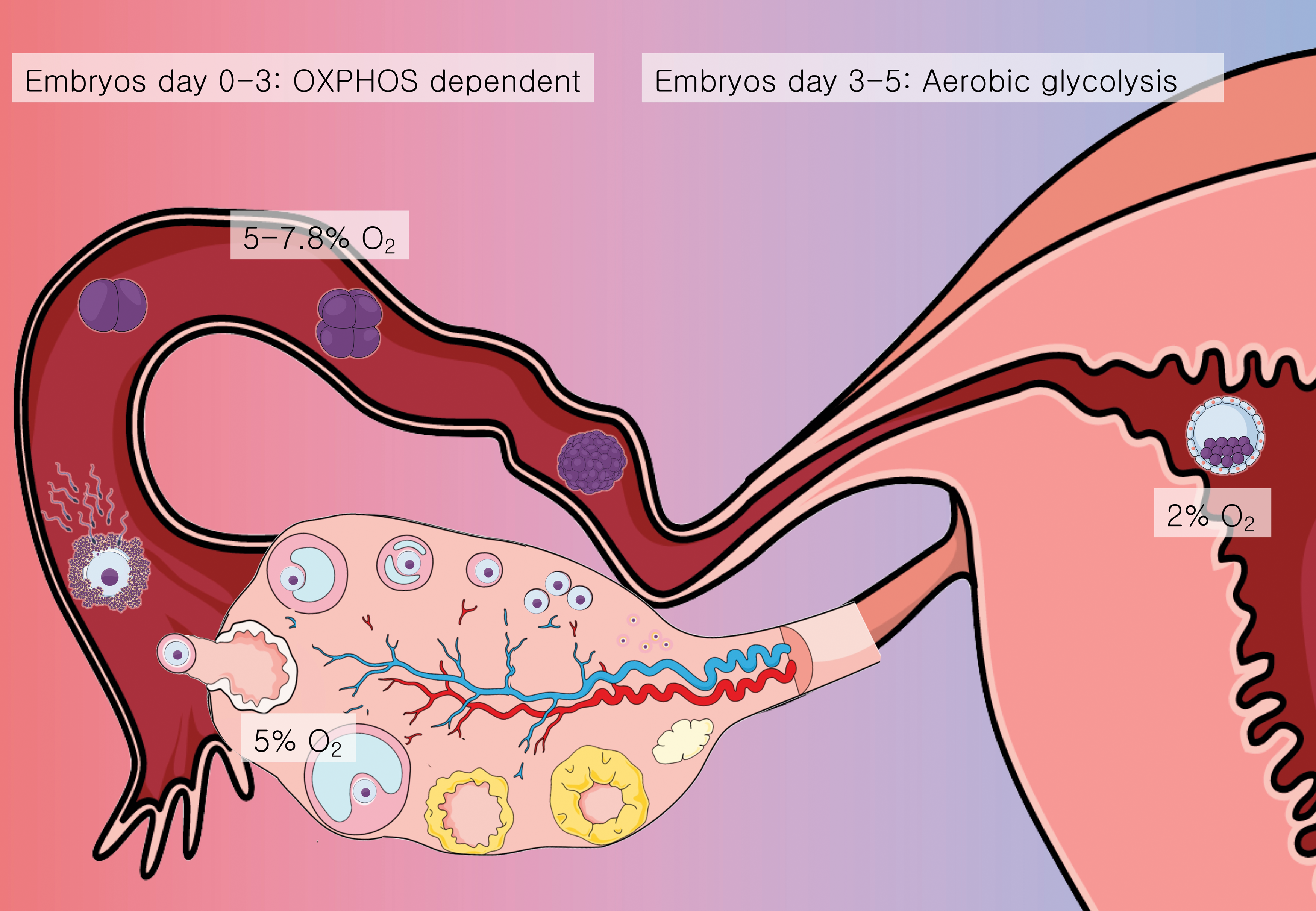
The oocyte does not metabolize glucose, but is highly dependent on oxidative phosphorylation (OXPHOS) to obtain energy. CCs can metabolize glucose captured in the follicle's microenvironment through glycolysis, producing pyruvate that is sent to the oocyte for further processing.
It was recently found by Dunning and colleagues (63) that mural cells and CCs have an abundant amount of hemoglobin, a molecule with high O2 affinity. This finding goes in accordance with the fact that although the oocyte relies on OXPHOS and electron transport chain reaction for energy generation, it is located inside the antral follicle, a hypoxic microenvironment with only 2% O2 available (55). The CC's proximity with oocyte and antrum allows hemoglobin to capture the slightest available O2 to be transferred to the oocyte for OXPHOS. Even though OXPHOS followed by electron transport chain reaction is the most efficient metabolic pathway for energy production in eukaryotic cells, it has a high price: it produces ROS as a by-product (47, 48). Proton leak from OXPHOS in mammalian oocytes can represent up to 37% of mitochondrial respiration, which might indicate a high response to ROS production (106). Although ROS are essential for some biological processes such as signaling molecules, they can react with biomolecules such as lipids and nucleic acids, causing cell damage and oxidative stress, harming the oocyte and lowering its quality (103).
The oocyte does not have the capacity on its own to mobilize all the necessary antioxidant defense mechanisms. This protection is provided by the surrounding CCs (102). Besides O2 rescuing, hemoglobin possesses other functions of extreme relevance in the COC: it functions as an antioxidant molecule, capable of protecting cells from oxidative stress via scavenging reactions with hydrogen peroxide (H2O2) and nitric oxide (NO). These features were discussed elsewhere (63).
The healthy (and young) female reproductive tract provides all the necessary conditions for follicle growth, oocyte maturation, and embryo development, a feature still not equally reproduced in vitro (39). While therapeutically effective, in vitro fertilization (IVF) techniques fail to replicate comparable rates of good-quality oocytes and embryos as observed in healthy individuals.
In Vivo and In Vitro Sources of Oxidative Stress in the COC
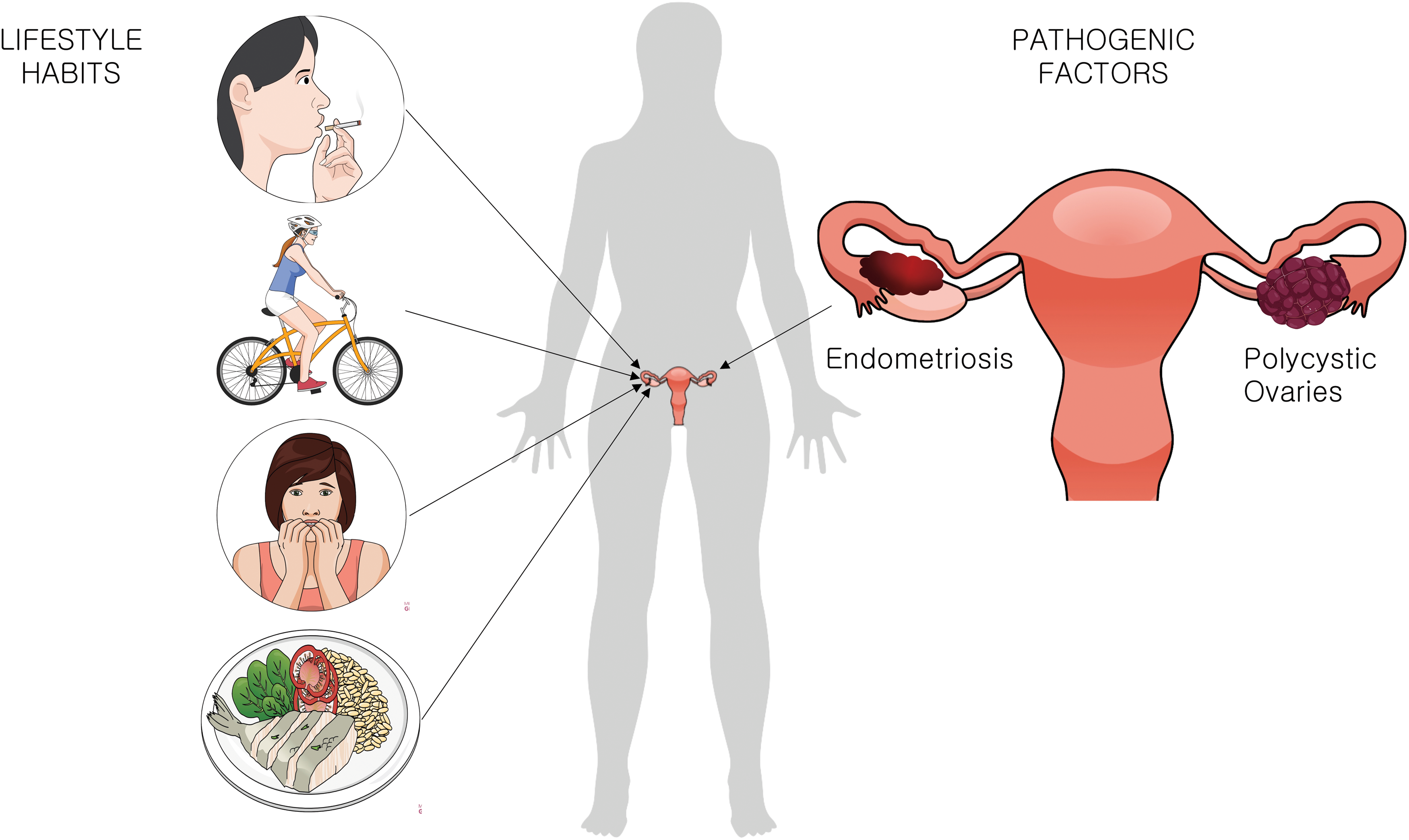
Oxidative stress and redox imbalance is known to play a significant role in infertility (8). There are several possible sources of reactive species that can impact on the COC's health, either in vivo, influenced by the women's lifestyle or physical conditions, or in vitro, during the IVF technique (88). The review by Agarwal et al. summarizes different sources of ROS in vivo and in vitro (3). Smoking (105), exercising regularly (23), the diet (56, 61), stress (86), body mass index (BMI) (43), and pathologies such as endometriosis (53) and polycystic ovary syndrome (PCOS) (97) lower the fertility capacity (2) and impact the functioning of CCs (Fig. 3).
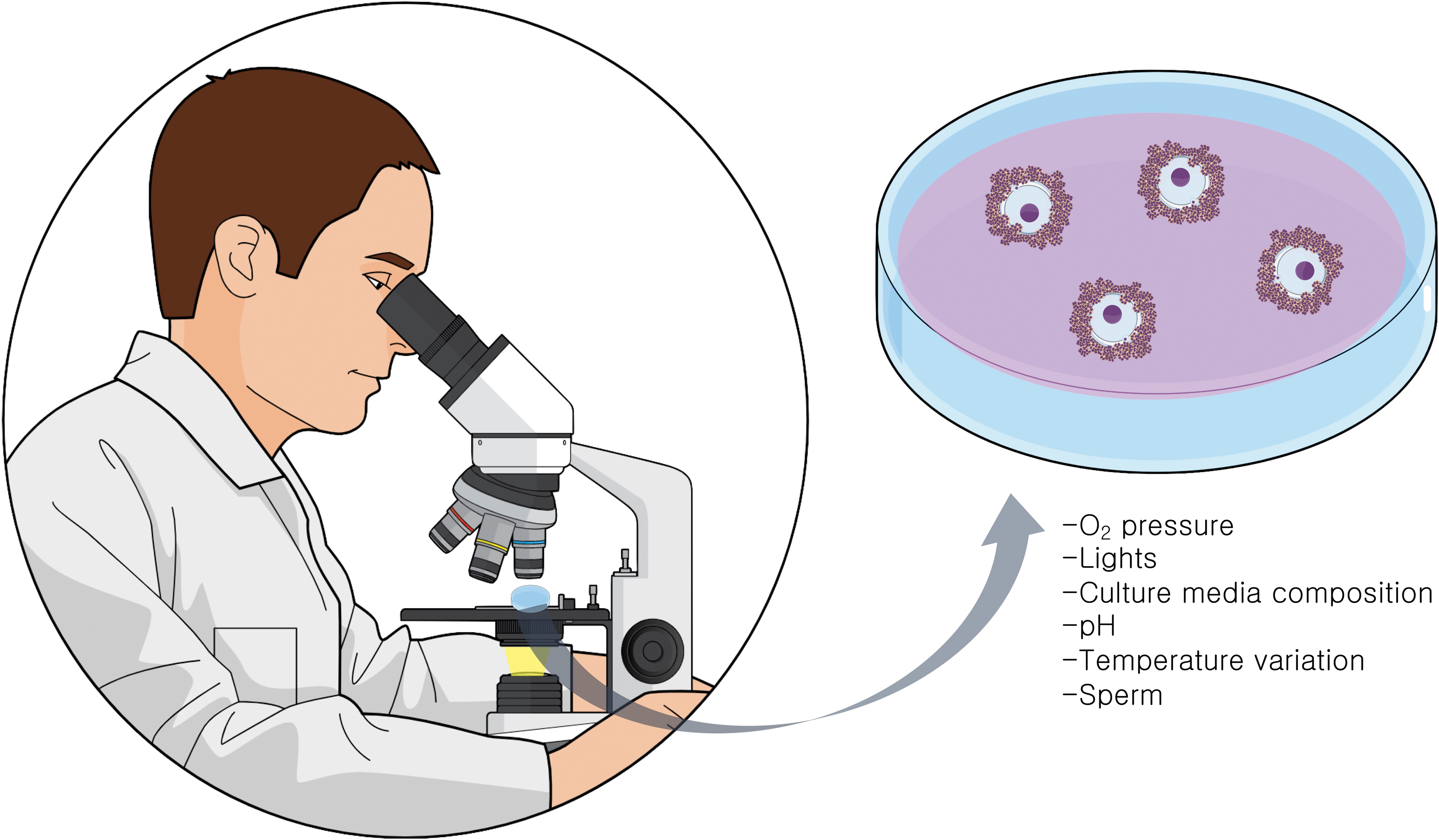
During IVF, the COCs are exposed to several potential sources of oxidative damage (3). In ovarian stimulation procedures, multiple follicles will be induced to mature through the use of gonadotropins. In this process, numerous genes related to inflammation are induced in the ovarian follicles (90). ROS originate from the inflammatory cells, which are attracted by the luteinizing hormone surge, also also by the activation of cytochrome P450 system in the steroidogenic cells of the follicle (133) (Fig. 4). Once COCs are obtained in the culture dish, variations in O2 pressure (45), exposure to visible light wavelengths (79, 107), pH, and different media compositions (68) can generate ROS. At insemination, sperm concentration and quality (96) are additional factors that directly impact on COC oxidative stress levels. It should be noted that the influences of CCs on oocyte metabolism change accordingly to environmental factors such as O2 tension (15). For example, expression levels of some antioxidant enzymes are regulated by hypoxia factors (14).
CC Redox Biology
The FF in direct proximity of the COC can act like an antioxidant buffer, maintaining the redox balance in vivo (38). While some studies found significant correlations of ROS/antioxidant levels in FF and oocyte quality (21), others reported no correlation (21). Because of its origin, it might be possible that the FF composition reflects rather the plasma composition than the COC biological state.
DNA damage caused by oxidative stress in granulosa cells is inversely correlated with fertilization and embryo quality rates (99). CCs are mainly responsible for the oocyte oxidative stress defense (116). When CC antioxidant capacity is low, oocyte quality might be affected (33, 77). The mature metaphase II (MII) oocyte being transcriptionally silent (135), the contribution of CC metabolite production (such as glutathione and melatonin) is even more important at this stage of maturation. Importantly, when metabolizing glucose, the CCs also generate reduced nicotinamide adenine dinucleotide phosphate (NADPH) for biosynthesis by the pentose phosphate pathway (PPP), contributing to the redox balance of the oocyte (46) (Fig. 5), deviating <3% of the already small portion of the glucose it can metabolize to PPP (124). NADPH is essential for reactive species metabolization, since it is necessary for reduced glutathione (GSH) recycling. A perfect functioning of both glycolysis and PPPs in CCs has been shown to be essential for oocyte health in mouse (62).
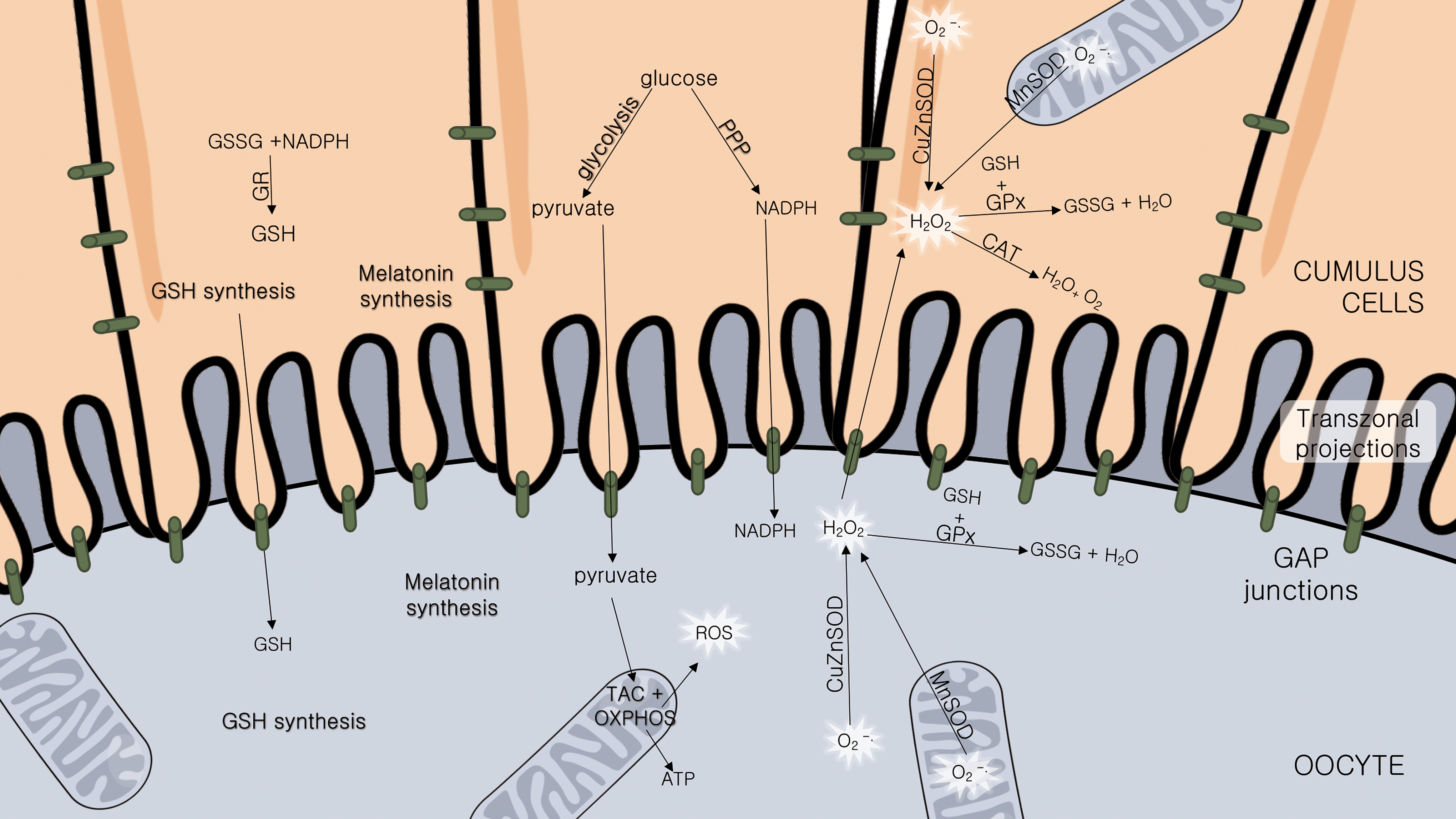
The tripeptide glutathione (GSH), the main intracellular antioxidant, plays an important protective role against oxidative damage of biomolecules and in detoxification processes (72). GSH concentrations have been related to the oocytes' meiotic spindle morphology and fertilization, and posteriorly, in early embryo development (66). It is produced by CCs (69) and oocyte (30), but optimal GSH levels in oocyte are dependent on CC synthesis (40) (Fig. 5). This was demonstrated experimentally; oocytes stripped from their CCs showed a higher cryotolerance when pretreated with an exogenous glutathione donor (121), suggesting a compensatory effect by GSH supplementation in the absence of CCs.
The CCs protect the oocyte also through the expression of several antioxidant enzymes (6). Besides low expression of phosphofructokinase (78), oocytes lack also other enzymes essential for their survival. Instead of being expressed by the gametes, these enzymes are provided by the CCs. Catalase (CAT), for example, is an antioxidant enzyme not expressed in oocytes (77). It metabolizes H2O2 into nonreactive molecules (H2O and O2) and its action is supplied by the CCs (Fig. 5). When exposed to H2O2, oocytes still inside the COC were protected from oxidative stress damage, while oocytes from which CCs have been stripped-off presented a major decrease in viability (33).
The expression of antioxidant enzymes in CCs is correlated with oocyte and embryo quality (43) and even with pregnancy rates and fetal development (70, 74). Glutathione-S-transferases (GSTs) form a large family of enzymes that protect cells from oxidative damage, lipid peroxidation of membranes caused by ROS and toxic compounds (52). Wathlet et al. correlated glutathione S-transferase alpha-3 and -4 and glutathione peroxidase (GPx) 3 in CC expression with pregnancy outcome (129). GPx3 is an extracellular, seleno-containing enzyme that catalyzes the reduction of H2O2 and lipid peroxides using GSH as cofactor (67). Therefore, it acts as an important antioxidant in reproductive biology.
Superoxide dismutase (SOD), the enzyme responsible for metabolizing the reactive superoxide anion (O2 •−) into H2O2 and O2, is expressed in CCs and oocytes, in mitochondria (manganese superoxide dismutase [MnSOD]) and cytoplasm (Cu/ZnSOD). Importantly, its expression and activity level in CCs was positively correlated with successful pregnancy (70).
The inducible form of nitric oxide synthase enzyme generates nitric oxide (NO•). NO• has dual effects in the ovary. It is an essential nonpolar signaling molecule that freely diffuses through membranes. It is involved in oocyte maturation and ovulation, but in a dose-dependent matter can become a source of oxidative insult (120). CCs are the oocyte's NO• source, stimulating oocyte maturation (16). CCs from COCs that fertilize successfully synthesize less iNOS and HO-1, another redox-sensitive gene and important antioxidant enzyme, indicating that COCs with higher antioxidant activity might be reflecting a defense mechanism against an oxidative insult occurring in the follicle and related to a diminished biological capacity of the oocyte (13).
The differences in oxidative damage and ROS levels in oocytes fertilized using the classical IVF technique compared with intracytoplasmic sperm injection (ICSI) suggest that the CCs play a major role in protecting the oocyte from ROS (9). The ICSI technique consists of detaching the CCs from the oocyte to allow the injection of a single sperm through a needle inside the female germ cell (81). ICSI is mainly used in cases of severe male factor infertility, with low sperm fertilization capacity. Although ICSI represents a great advance in those cases, overcoming the difficulties of a natural fertilization, it requires the denudation of the oocyte, increasing significantly its exposure to the environment.
In classic IVF, the CCs are not removed and the oocyte fertilization by the sperm happens in a more natural way. When the biological shield of cumulus-corona is maintained, the oocyte retains a living barrier of antioxidants that protect it from external ROS sources, increased O2 tension, and exaggerated exposure to sperm (45). The improved antioxidant defenses are another element of the synergism between CCs and the gamete.
It is known that the patients' clinical characteristics such as age (70, 71, 117, 118), BMI (83, 93), infertility causes (101, 134), and stimulation protocol applied (82, 130) influence directly the CC biological characteristics. Gene and protein expression patterns, for example, change drastically according to the stimulation protocol applied, even though the success rates can remain similar (37, 49).
Pathological Patterns of Redox Metabolism in Human CCs
The important roles played by CCs to ensure oocyte's health suggest that changes in their structural or physiological composition could be related to infertility (70). CC gene expression and biochemical activity are directly influenced by the oocyte conditions, the follicular environment, and interactions with the ovarian environment. In that manner, the CC pattern might indirectly reflect the biological processes taking place in the oocyte (85).
Several studies analyzed redox compounds in the follicular environment and found significant differences between patient's pathophysiological profiles, such as endometriosis (24, 87), PCOS (125), advanced maternal age (131), and obesity (127). These differences may be a consequence of the patient's overall status/pathology, and not necessarily representative of the oocyte status; however, they need to be taken into account as potential confounders when searching for possible oocyte quality indicators. In PCOS patients, for example, the total antioxidant status in plasma is decreased (34, 126). It was also found that SOD activity in serum and FF was significantly lower; however, there was no correlation with oocyte fertilization capacity, embryo quality, or pregnancy rates (101). The significant alteration in enzyme activity can vary for each pathophysiological condition. In patients with ovarian dysfunction and endometriosis, the opposite pattern has been observed, with copper/zinc superoxide dismutase (CuZnSOD) activity being elevated (70).
Endometriosis is a pelvic inflammatory disease characterized by the occurrence of implants of endometrial tissue outside the uterine cavity, with high levels of oxidative damage in ovarian cells (98). In fact, DNA damage caused by oxidative stress in granulosa cells is higher in patients with endometriosis (99). Erratic patterns of antioxidant enzymes are known to occur in CCs from patients with PCOS and endometriosis (70).
PCOS is an endocrine and metabolic condition that causes a complex imbalance in ovarian function and ovulation process (42). In PCOS patients, CCs have shown to have a higher mitochondrial ROS production and a lower antioxidant capacity, with lower GSH/GSSG (oxidized glutathione), NADH/NAD+, and NADPH/NADP+ (oxidized nicotinamide adenine dinucleotide phosphate) ratios, suggesting enhanced oxidative stress (134). In those patients, a higher GPX3 gene expression in CCs is correlated with blastocyst formation (54).
It is clear that pathologies change the metabolic functioning and redox status of CCs. However, even though these cells are behaving differently than in the optimal physiological state, their gene expression pattern might be presenting an adaptive response to the external insults and not necessarily reflecting a poor oocyte quality. For example, CuZnSOD levels are significantly decreased in female patients with age-related infertility (67). Thus, Matos et al. correlated increased levels of SOD activity in this patient group with successful Assisted Reproduction Technique (ART) outcomes (70). This might indicate that CCs are increasing their defense mechanisms against insults that could harm their oocyte. The authors also observed, however, that in patients diagnosed with endometriosis or ovarian dysfunction, CC SOD activity is significantly increased (70).
Fertility starts to decline in an accelerated matter in women from the second half of the third decade of living (73). Aging of aerobic cells is directly related to oxidative damage caused by ROS (36). Aging is one of the most important factors in oocyte competence. It is known that the aging ovary suffers imbalances of redox metabolism and protein's carbonyl stress (117). Mature oocyte mitochondria are originated from few precursors since embryonic life. In this way, oocytes from women approaching their forty might present aged mitochondria, with higher levels of mitochondrial DNA damage and stress (26). In fact, oocytes from advanced maternal age present lower levels of messenger RNA stores and lower efficiency of DNA repair (50).
Granulosa cells of patients with advanced maternal age present an overexpression of up to 10 times higher of GST teta 1 levels (57, 58). The GST enzyme being of great importance in cellular xenobiotic detoxification, this could be indicative of a compensatory mechanism of CCs in an effort to maintain the oocyte's health. A lower expression of the genes encoding SODs (70) and catalase (119) in CCs and granulosa cells has been reported in relation to mitochondrial swelling and degeneration, as a reflection of the high levels of oxidative stress and mitochondrial dysfunctions (119). CCs from advanced maternal age patients also reveal differences both on messenger RNA and protein expressions involved in OXPHOS, mitochondrial function, and posttranscriptional splicing (71). Mitochondrial respiratory activity dependent on coenzyme Q10 is decreased in granulosa cells from older women (12). In accordance with these results, it was shown that CCs have significantly lower expression of genes involved in coenzyme Q synthesis (11).
These observations make us aware of the fact that different patient characteristics influence directly on CC functioning, with the cells adapting to potential insults. These particular effects must be taken into account in the biological functioning of the COC and in the therapeutic management in the clinics.
Targeting COC Redox Biology in the Clinical Scenario
Two strategies can be adopted while using antioxidants for improving IVF outcomes: oral administration, to alleviate endogenous sources of oxidative stress such as pathologies, and the supplementation of antioxidants in the laboratory, to diminish the impact of oxidative stress caused by the in vitro environment (Fig. 6).
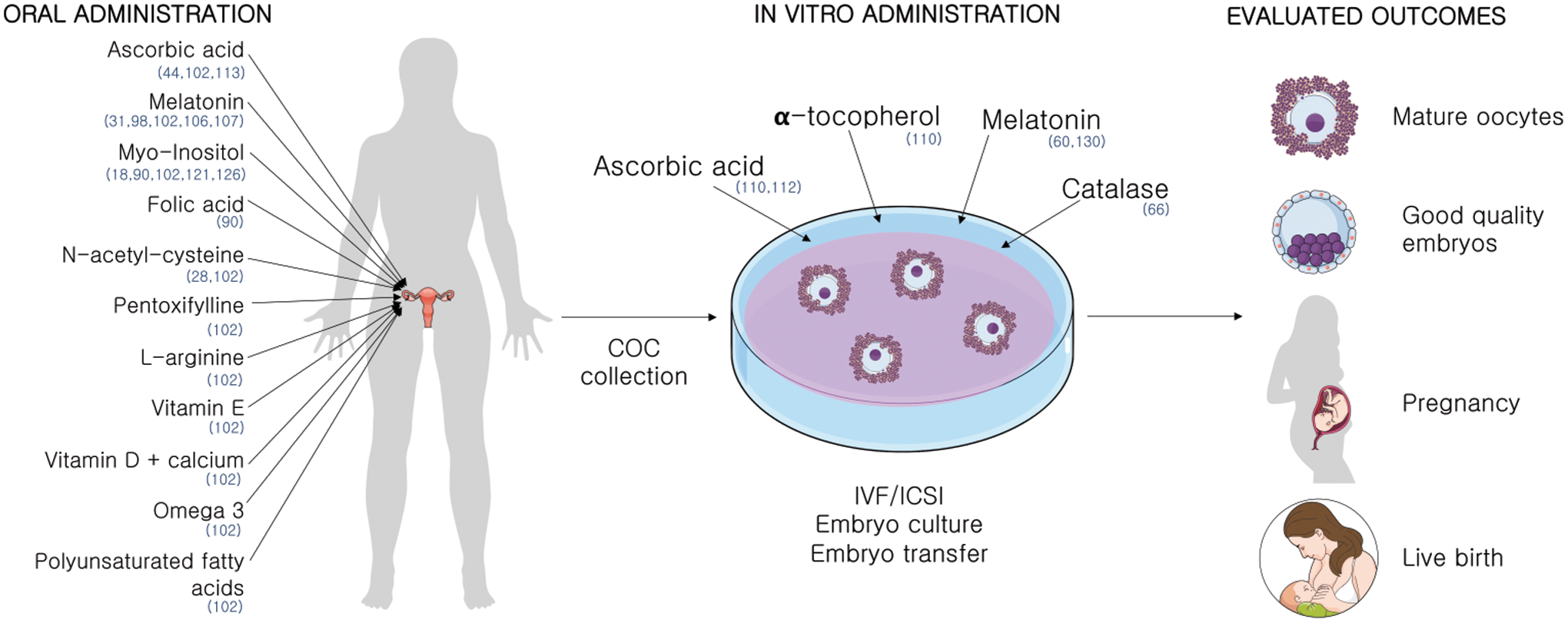
Oral administration of antioxidants
Besides its action as a hormone driving nycthemeral rhythm by the pineal gland, melatonin turned out to possess several local properties (1). FF has higher levels of melatonin than plasma, indicating an important action in the ovary. Its role on preservation of gametes (20) and usage in ART is abundant and reviewed elsewhere (35). It is an extremely efficient antioxidant (35) produced by the COC (27) that plays a major role in the follicles' oxidative defense (89, 109, 110).
Melatonin's scavenger properties have been explored in ART (65). Several researchers have studied the effects of oral supplementation with different outcomes (100, 109). Melatonin administration improved the number of retrieved oocytes, oocyte maturation rates, and embryo development rates (31). It was also shown to improve fertilization rates from patients with previous failed IVF cycles (108).
The combined administration of melatonin and myo-inositol, a precursor of phosphoinosides, essential for oocyte maturation and embryo development, has shown positive results (128). Women with a previous failed IVF cycle received oral administration of melatonin+myo-inositol for 3 months before a new cycle showed improved oocyte maturation, fertilization, embryo number, and quality rates (123). Another study administrating myo-inositol+folic acid compared with myo-inositol+folic acid+melatonin, even though the treatment time frame was shorter, showed that melatonin improves the capability of myo-inositol to improve oocyte maturation and embryo development rates (92). Myo-inositol administration in PCOS patients was also shown to positively impact IVF outcomes (18).
The acetylated form of the amino acid
The administration of ascorbic acid, or vitamin C, in fertility treatments has been showing contradictory results. While some observed a detrimental effect (115), others have not shown any significant differences in implantation and pregnancy rates after its administration (44).
A large meta-analysis on antioxidant oral supplementation analyzing administration of combinations of antioxidants, pentoxifylline, NAC, melatonin,
In vitro administration of antioxidants
The administration of antioxidants on COCs in vitro has also shown some promising results. Supplementation with low concentrations of melatonin (10−2–102 nM) improved maturation rates of denuded oocytes (132). Also, the incubation of oocytes from PCOS patients with melatonin showed positive results in implantation rates (60). However, higher concentrations of melatonin medium supplementation (105–107 nM) were shown to be detrimental (132). The beneficial effects of antioxidant administration in vitro are dose dependent (113).
Besides its controversial effects in oral administration, the addition of ascorbate to IVF medium also did not improve embryo quality in cultures with 5% O2 (114).
Catalase administration decreased the rate of oxidation of COCs compared with nonsupplemented commercial media (68), implying that H2O2 is a major ROS generator in COC in vitro culture. Catalase is absent in oocytes, being physiologically supplied by CCs in vivo.
Even though the study of Tao et al. was not conducted on humans but in porcine, it brings interesting results about the contribution of CCs to the in vitro environment. While denuded porcine oocytes supplemented with α-tocopherol showed a greater rate of progression to the MII stage, the same was not observed supplementing COCs, since oocytes within the complex already have a high spontaneous MII maturation rate, reinforcing the role of CCs in oocyte maturation and protection against oxidative insult. Also, the administration of α-tocopherol and L-ascorbic acid prevented CC DNA fragmentation when cultured at 20% O2, but no effect was seen in cumulus-enclosed oocytes (112).
Just like the influence of pathologies on COC functioning in vivo, it is of major importance to consider the differences of the in vitro environment when analyzing the potential beneficial effects of antioxidant administration. In a study with bovine oocytes, the antioxidant benefits of melatonin administration were even higher when culture was conducted with exposure to a high O2 tension (84).
Conclusion
Alterations in CCs may have several causes and may be responsible for reproductive disadvantage, this being a direct cause, a reflection of a decline in the functional and structural qualities of the oocyte, or a consequence of a detrimental follicular environment that affects both the oocyte and the somatic cells. Either way, studying CCs and their relationship with oocyte quality could guide us toward valuable tools for improving routine IVF rates. Nevertheless, it is paramount to also take into consideration the patient-specific background: clinical characteristics such as age, BMI, pathological features, stimulation protocols, and in vitro environment characteristics such as O2 tension, media composition, and manipulation conditions. Many authors have indicated that all these parameters will cause distinct metabolic and gene expression patterns in COCs, directly affecting the functionality and health of the oocyte and its developmental fate.
Oxidative stress is one of the major causes of poor oocyte quality. In the healthy female tract, along with the physiological environment, the CCs provide the necessary antioxidant defenses the oocyte needs for an optimal development. In vitro conditions stand far from the ideal parameters found in vivo. More physiological culture conditions can be promoted in the clinical environment, by more sophisticated technological features. Minor parameter adjustments to optimize the cultures are within reach to obtain higher success rates in IVF. For example, FF provokes much lower levels of ROS when compared with several commonly used culture media (68). Developing a medium composition that mimics the FF in vivo, and its changes in relation to the temporal variations in hormone concentration would enable the cultured COCs to better support oocyte's metabolic needs. Critically important are also the physical characteristics of the in vivo environment, such as O2 tension and temperature, since both cell types have the ability to adapt their metabolic pathways according to those parameters. Submitting the COCs to suboptimal conditions might result in a major effort of the cells to survive, deviating their energy from biosynthetic routes to damage control process, impairing their potential for normal further development. “Ex ovo Omnia,” the potential of the oocyte in generating all life as expressed by the illustrious 17th century physician and scientist William Harvey, is still true. What has been learned over the last 50 years, thanks to the development of IVF and the access to the so far closed environment of the ovarian follicle, is that the oocyte's unique capacity to generate all cells is only effective by outsourcing critical functions to corona-CCs. The cumulus functions as a Praetorian Guard to preserve oocyte's integrity for further development; hence, these cells constitute an important target for future optimization of oocyte culture.
Future Directions
There is still a lot to unravel about the redox metabolism of CC: how it is regulated, how the external sources affect it, its responses to the environment, and how it modulates the oocyte's health. Our research addresses the redox metabolism pattern in the cumulus/corona/oocyte complexes under several culture conditions and different patient groups. The levels of redox enzymes and molecules are being correlated with embryo development and patients' characteristics. Such studies are relevant to better understand the biology of CCs, and to open new possibilities of future treatments and clinical approaches. For assessing the real effects of antioxidant administration in IVF, RCTs are indispensable.
Footnotes
Funding Information
We thankfully appreciate the Brazilian Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (INCT-TM/CNPq/FAPESP-465458/2014-9) funding, Brazilian Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES-PDSE 47/2017-Seleção 2018) and PRONEX/FAPERGS (16/2551-0000499-4) that made this research feasible. L.v.M. received a fellowship from CNPq and CAPES during this research and F.K. received a fellowship from MCT/CNPq (306439/2014-0).
