Abstract
Significance:
Oxygenated polyunsaturated lipids are known to play multi-functional roles as essential signals coordinating metabolism and physiology. Among them are well-studied eicosanoids and docosanoids that are generated via phospholipase A2 hydrolysis of membrane phospholipids and subsequent oxygenation of free polyunsaturated fatty acids (PUFA) by cyclooxygenases and lipoxygenases.
Recent Advances:
There is an emerging understanding that oxygenated PUFA-phospholipids also represent a rich signaling language with yet-to-be-deciphered details of the execution machinery—oxygenating enzymes, regulators, and receptors. Both free and esterified oxygenated PUFA signals are generated in cells, and their cross-talk and inter-conversion through the de-acylation/re-acylation reactions is not sufficiently explored.
Critical Issues:
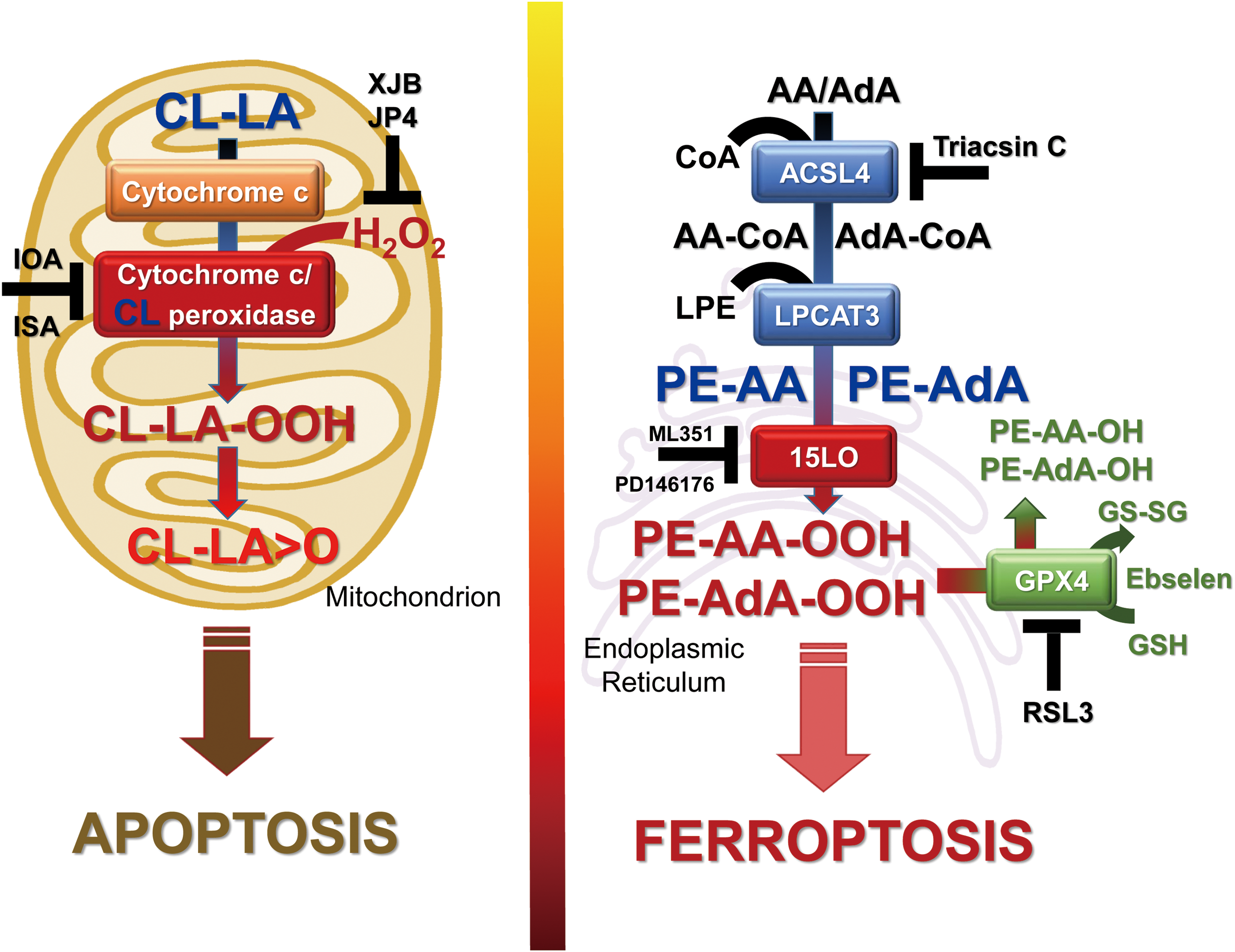
Here, we review recent data related to oxygenated phospholipids as important damage signals that trigger programmed cell death pathways to eliminate irreparably injured cells and preserve the health of multicellular environments. We discuss the mechanisms underlying the trans-membrane redistribution and generation of oxygenated cardiolipins in mitochondria by cytochrome c as pro-apoptotic signals. We also consider the role of oxygenated phosphatidylethanolamines as proximate pro-ferroptotic signals.
Future Directions:
We highlight the importance of sequential processes of phospholipid oxygenation and signaling in disease contexts as opportunities to use their regulatory mechanisms for the identification of new therapeutic targets.
I. Introduction
A
A. Evolution of concepts in oxidative lipidomics
Lipid molecules represent one of the major constituents of cells and tissues that are soluble in nonpolar organic solvents. One of the major classes of lipids are phospholipids, which have an amphiphilic nature—a combination of a polar water-soluble head with a phosphate moiety and a water-insoluble hydrophobic tail (most commonly two 14–20 carbons long fatty acid chains) linked by glycerol—that allows them to form biomembranes. The two most abundant types of phospholipid molecules in membranes are phosphatidylcholine and phosphatidylethanolamine (PE) complemented with several minor constituents such as phosphatidylserine (PS), phosphatidic acid, phosphatidylglycerol, and diphosphatidylglycerols (or cardiolipins, CL).
Self-assembly of amphiphilic (phospho)lipid molecules into bilayers and semipermeable membrane encapsulation of replicating macromolecules was essential for life from the very early stages of its emergence on the surface of our planet (111). Under anaerobic conditions and high temperatures during that period, saturated and monounsaturated lipids were optimal for building and maintaining the semi-fluid state of membranes in single-cell organisms (35). However, with planet cooling, the appearance of molecular oxygen, and multicellularity, the importance of polyunsaturated lipids surpassed a structural building block role and now includes vital functions as communication molecules required for coordination of multiple processes into spatiotemporally integrated and optimized forms of life (58).
Based on fossil records, there was a major change in the propagation of life that abruptly occurred ∼600 million years ago, when the oxygen content in the atmosphere became sufficient for the maintenance of aerobic life (21). All land-based mammals had access to sufficient nutrient resources judged by their large body mass and fast growth rates they attained (99). Archaeological data indicate that sufficiency of food enriched in long-chain polyunsaturated fatty acids (LC-PUFA) was essential for sustaining the large size, complexity of the modern human brain. LC-PUFA were also essential for multi-generational brain development, thus contributing to the advent of Homo sapiens (60). Notably, contemporary evolutionary models emphasize that proximity to marine, lacustrine, and riverine sources of food and utilization of littoral resources with high levels of LC-PUFA by Homo during the period 20–200 kya was critically important for the development of the human brain (13). Today, dietary essential long-chain fatty acid residues (C20, C22) with four, five, and six double bonds remain the most limiting nutrients for brain lipids and neural growth (20, 22 –24, 59). The reasons for the critical dependence of brain development and functions on these LC-PUFA still remain unknown. However, a popular concept that PUFA lipids are essential for the maintenance of biomembranes fluidity is generally accepted, in spite of the fact that oleic acid (C18:1) residues are sufficient for keeping membranes fluid enough.
One of the important chemical propensities of polyunsaturated lipids is their susceptibility toward oxygenation (39). Enzymatically, these reactions are catalyzed by a host of mono- and di-oxygenases yielding myriads of highly diversified and versatile functions. Among these functions, signaling by oxygenated derivatives of free PUFA has attracted much attention as regulators of intracellular metabolism and intercellular communications (86). Numerous functions of octadecanoids, eicosanoids, docosapentanoids, and docosahexanoids mediated by their specialized receptors have been identified in health and disease (27, 109, 115). Different phospholipids with esterified PUFA-FA residues undergo hydrolysis by phospholipases A—a rate-limiting enzymatic process that releases PUFA for subsequent oxygenation steps catalyzed by one of several enzymes such as cyclooxygenase, lipoxygenases (LO), and cytochromes P450 (49, 78, 114, 124, 160). These concepts peacefully accommodate current knowledge about important lipid mediators and their precursors PUFA-phospholipids.
For a long time, oxygenation of PUFA residues of phospholipids has been predominantly associated with structural organization of biomembranes, mostly in the context of oxidative damage (165, 166). Initiated by the interest to radiation-induced injury and free radicals produced during radiolysis of water (8, 142), the chemical concepts of liquid phase oxidation have been transferred to biology and gave birth to new interdisciplinary fields of research—free radical biology and medicine. For more than five decades, the concepts of free radical-induced damage of membranes caused by poorly controlled random free radical oxidation of polyunsaturated phospholipids dominated the field and triggered the high interest to small-molecule chemical scavengers of radicals, that is, antioxidants (28, 112, 149). A simple and readily accepted concept of oxidative stress—a disbalance of endogenous antioxidant/prooxidant systems in favor of the latter—encouraged numerous, initially optimistic, studies aimed at the compensation of endogenous antioxidant deficiency in essentially all major human diseases and in aging (55, 139, 140). Disappointingly, the enthusiasm associated with the use of antioxidants did not withstand the rigorous scrutiny of multiple clinical trials, which consistently demonstrated the lack of therapeutic efficacy of exogenous natural or chemically designed antioxidants (107, 108, 147, 150, 167).
B. LC-MS-based oxidative phospholipidomics: a technological breakthrough
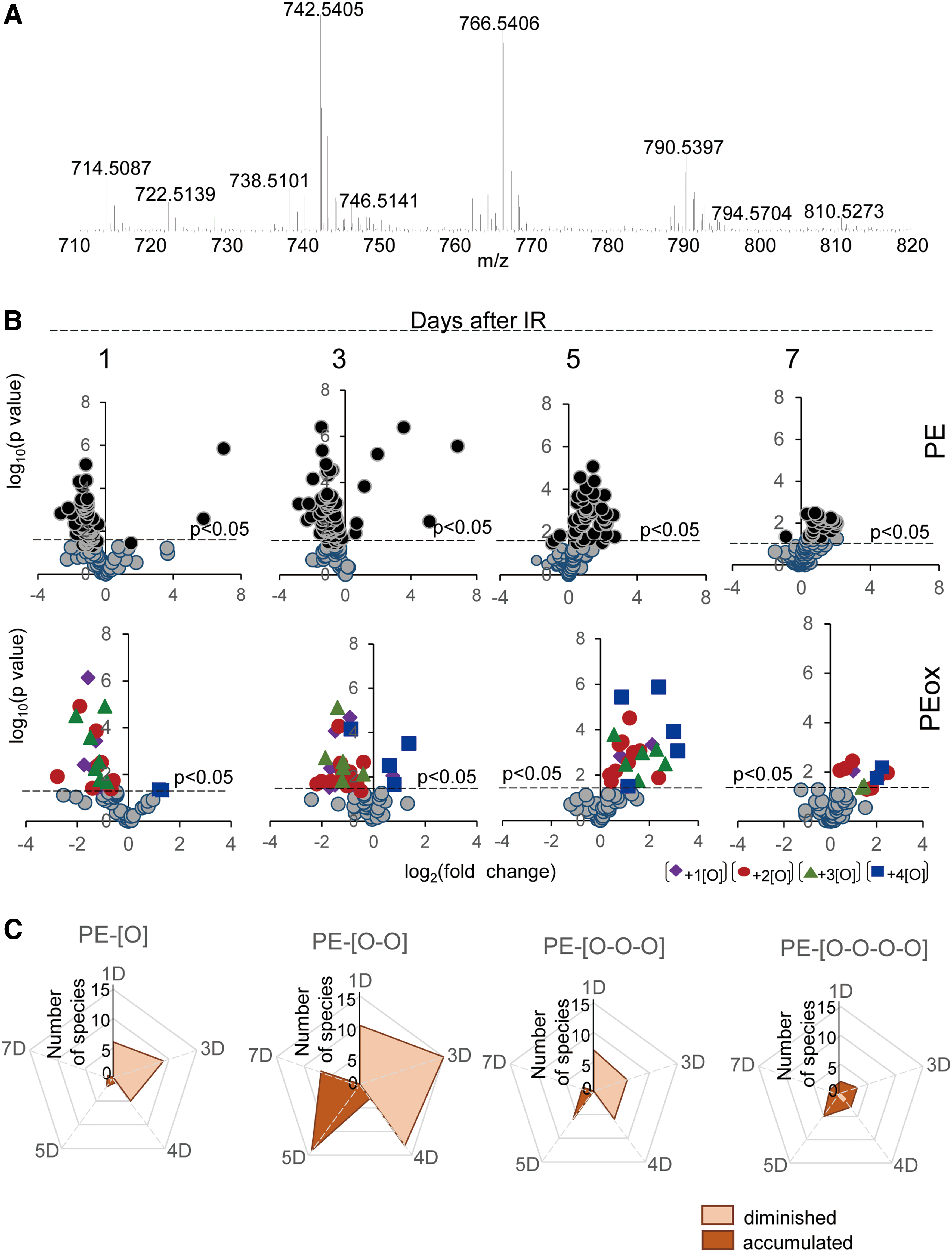
The new wave of interest in oxidative reactions of polyunsaturated phospholipids paralleled the advancement of small-molecule mass spectrometry (MS), associated with mild ionization, particularly electrospray, technologies (38). The discovery of a remarkably rich and diverse (phospho)lipidome and multiple signaling functions of phospholipids inspired research in the field of MS-based oxidative phospholipidomics (76, 121, 145). The enormous diversity of nonoxidized lipids is sufficient to constitute a significant component of metabolomics. Oxidative modification of PUFA lipids increases the number of their molecular species by at least an order of magnitude, thus making lipids dominant in the metabolome. This is mainly due to positional distribution of several oxygen-containing functionalities—hydroperoxy-, hydroxy-, oxo-, and epoxy—after oxygenation of bis-allylic carbon atoms at different positions in the PUFA residues (27) as well as oxidative truncation of the hydroperoxy-derivatives yielding the products with a shortened side chain (Fig. 1).
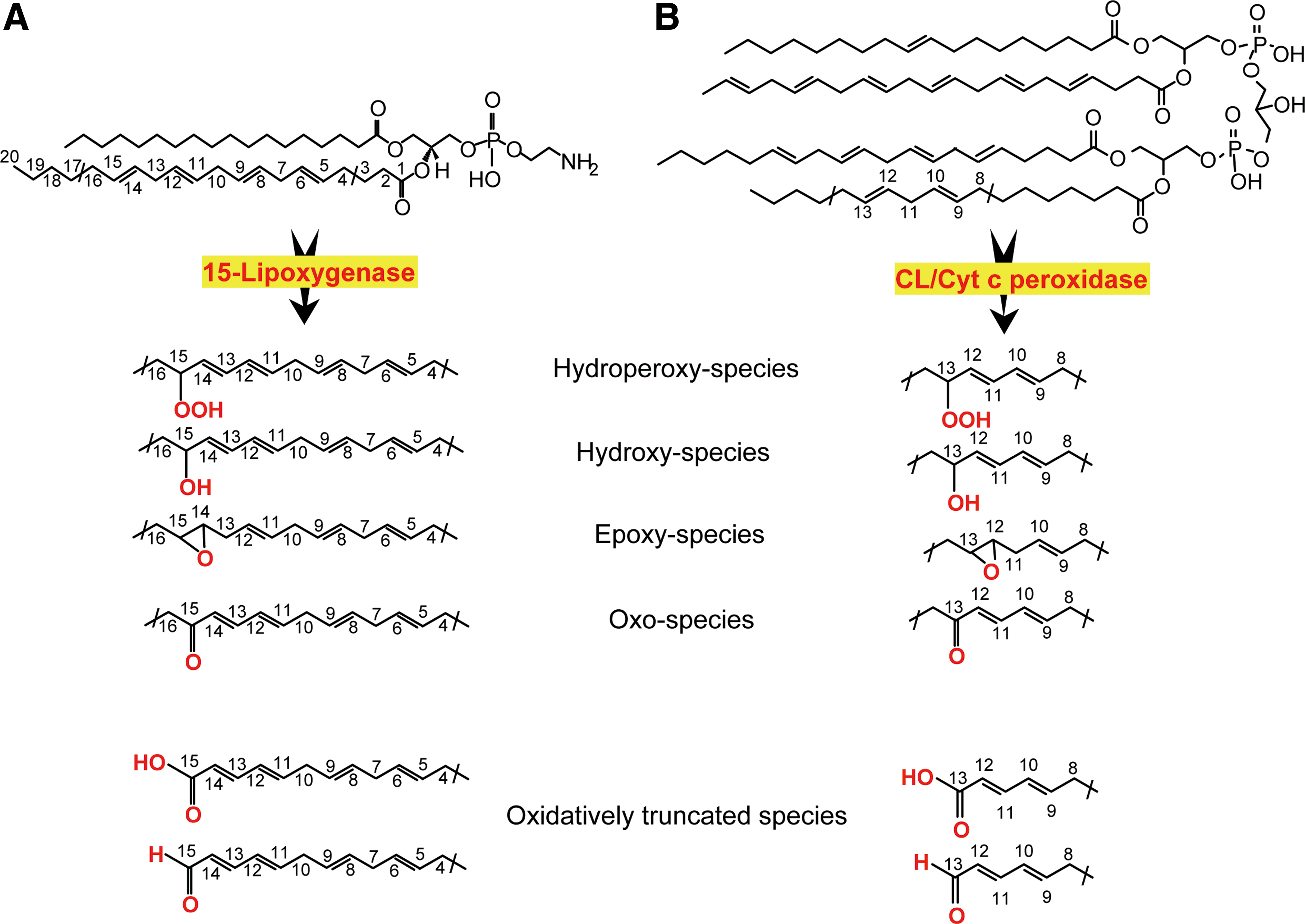
E
In spite of the huge variety of the oxygenated (phospho)lipids in terms of the molecular speciation, their absolute amounts are usually very low, on the order of 0.1–3.0 mol% of total nonoxidized phospholipids. Thus, identification and quantitative characterization of LC-MS-based protocols of phospholipid oxidation products is challenging, particularly in a nontargeted approach aimed at maximizing detection of all diversified species for subsequent identification of those that are associated with a particular function of interest. In spite of significant technical difficulties, partly also caused by the lack of standards for the majority of individual oxidized phospholipids, their semi-quantitative comparative analysis permits obtaining valuable and biologically interpretable data. The analytical task becomes much easier when targeted quantification of a partial list (“inclusion list”) of oxygenated phospholipids is required. Finally, it should be mentioned that many of the oxygenated phospholipid species are highly electrophilic and can readily form adducts by reacting with nucleophilic sites of proteins (33, 159). These adducts may represent the most functionally important products whose formation is responsible for the signaling effects. Thus, the new direction of research and technology advancement demands the development of oxidative proteo-lipidomics (17, 101, 144). Obviously, technological difficulties of this endeavor may be even greater due to multiplicity and even a greater variety of covalent complexes of myriads of electrophilic phospholipid oxidation products with thousands of target proteins.
II. Different Signaling Pathways for Nonoxidized and Oxidized CLs in Mitochondria
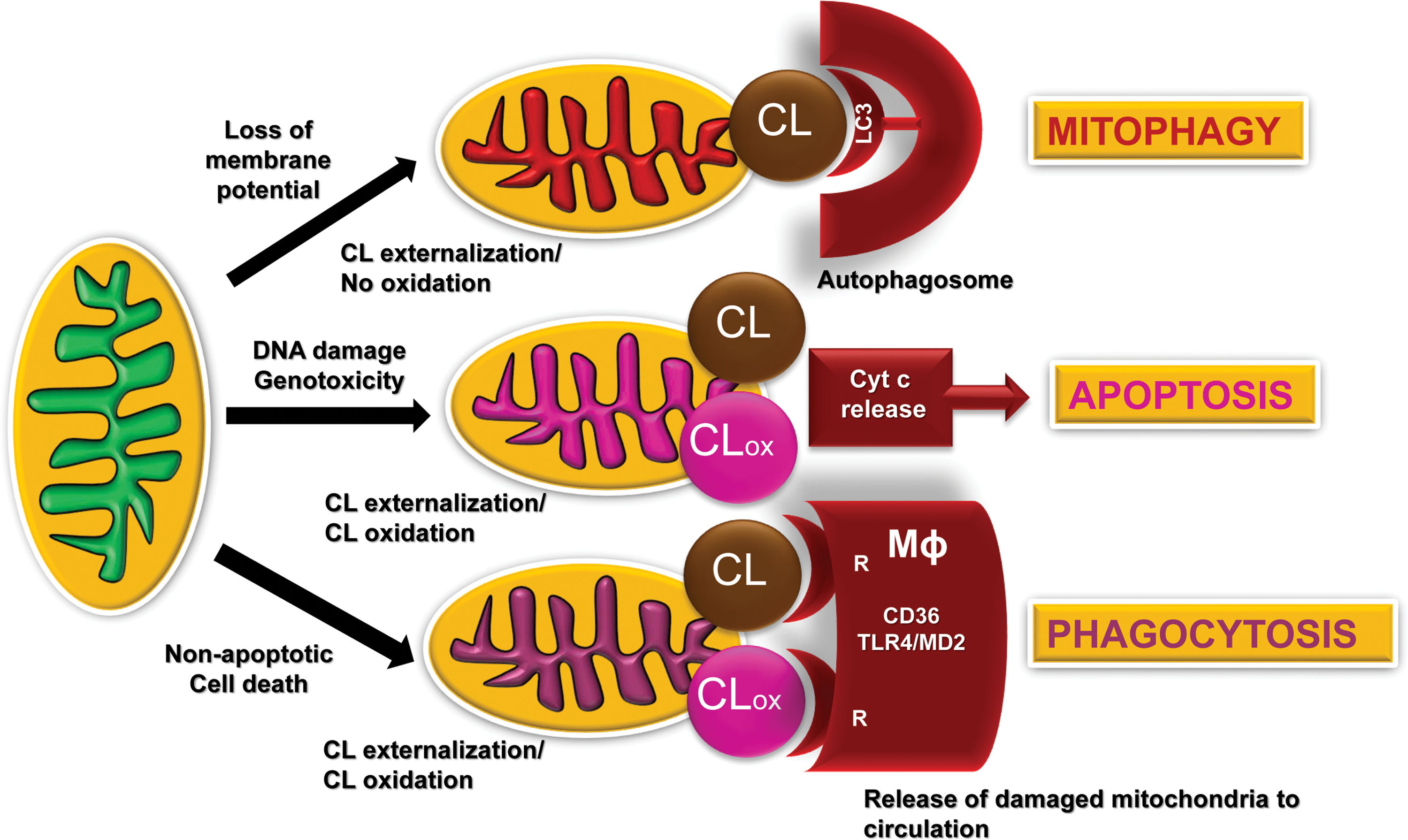
The presence of life-sustaining atmospheric oxygen content in the atmosphere for the maintenance of aerobic life based on fossil records occurred ∼600 million years ago. This created a new universal intracellular instrument organelle, mitochondria, and caused significant changes in the forms of life (21). As a thermodynamically open system of eukaryotic cells—from protozoan to mammals—mitochondria produce and exchange energy, metabolites, and information. They are filled with the machinery for oxidative metabolic reactions coupled with electrochemical generation of membrane potential and leading to the production and preservation of energy in the form of ATP (18, 36, 143). (18, 36, 143). In addition to being the major powerhouse of cells, mitochondria also act as a regulatory hub and are involved in the coordination of multiple intra- and extracellular processes and in controlling cell death (41). Interestingly, many regulatory functions of mitochondria are associated with their unique and asymmetrically distributed phospholipid, CL (122) (Fig. 2).
Chemically, CLs represent an unusual dimeric molecule with an anionic doubly charged (with two phosphate groups) polar head and a highly hydrophobic body with four fatty acyls linked via glycerol moieties. In contrast to other phospholipids, CLs are synthesized exclusively in mitochondria (56, 98,128) by CL synthase on the matrix side of the inner mitochondrial membranes (IMM) (131) and can undergo subsequent postsynthetic remodeling (129, 130). The remodeling process is believed to generate homo-acylated—with four identical fatty acid residues, most commonly C18:2—CL species in some tissues (heart, skeletal muscles, liver) (56, 113, 123, 132), in contrast to highly diversified hetero-acylated CLs in most tissues, particularly in the brain (12, 81, 154). Overall, the majority of CL species in mammalian cells contain one or more PUFA and, hence, are readily oxidizable (92, 125).
III. Externalization of Nonoxidized CL Signals Elimination of Damaged Mitochondria
Electron transport and other redox functions of the mitochondrial respiratory chain and enzymatic machinery are conducive of one-electron transfer reactions, including the reduction of molecular oxygen and thus generating superoxide anion-radicals and H2O2 (91). Discoordination of these reactions may lead to excessive production of reactive oxygen species (ROS), potentially leading to injury of the affected mitochondria, and other intra- and extracellular targets. To avoid the calamitous consequences of these redox misfortunes, a mitochondria-specific type of autophagy, mitophagy, is usually activated to eliminate the dysfunctional organelles, thus representing a pro-survival mechanism (51, 174). Notably, mitochondrial membrane depolarization caused by the excessive production of ROS is not the only trigger of mitophagy. In particular, superoxide anion radicals, one electron reduction product of oxygen, are not effective modifiers of mitochondrial membranes. However, their dismutation product—formed spontaneously or catalytically wih the participation of Mn-SOD—H2O2 may act as a source of oxidizing equivalents for the peroxidase function. In this regard, cytochrome c (cyt c)/CL complex may play a role of a very potent membrane modifier that is specific for CL oxidation (77, 79).
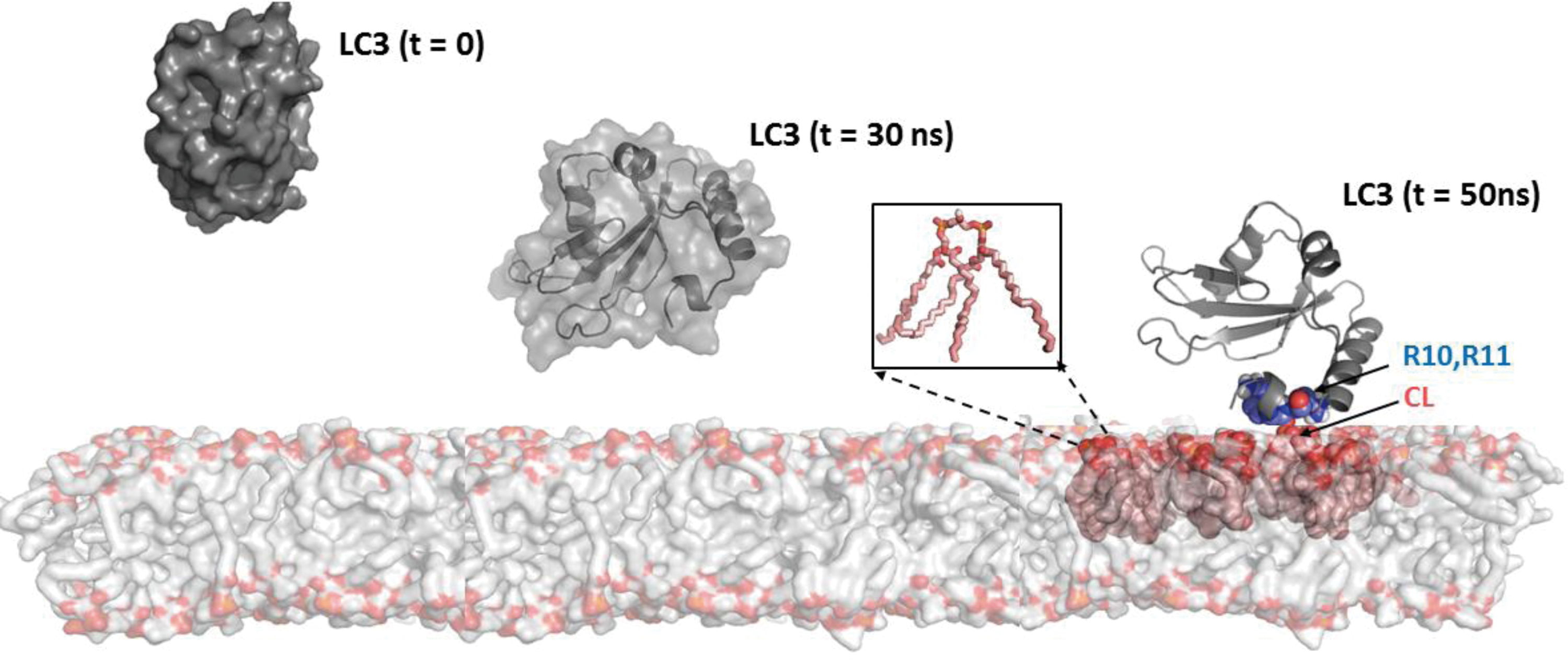
Oxidation of CL is associated with the loss of membrane potential in mitochondria (141). For example, treatment of cells with protonophoric uncouplers, such as carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP), also initiates mitophagy of depolarized mitochondria (16). Importantly in the context of this work, these pro-mitophageal stimuli cause a collapse of mitochondrial asymmetry of CL confinement to the IMM and trigger its redistribution to the outer mitochondrial membrane (OMM) and externalization to the surface whereby the unusual phospholipid is recognized by microtubule-associated protein 1 light chain 3 (LC3) (Fig. 2), one of the major components of the autophageal equipment of cells (16). The recognized signal with the attached cargo stimulates the inclusion of the damaged mitochondria in the autophagosome for further digestion and elimination. Binding with LC3 is mostly driven by electrostatic interactions of positively charged basic patches of the protein, particularly N-terminal residues R10 and R11, with two CL's negatively charged phosphate groups (16). Although the presence of all four acyls in CL is essential for its binding to LC3, the overall mitophagy process does not display any preference for individual fatty acid residues. The mitophageal signaling by externalized CL is not associated with the oxidation of the PUFA residues in the phospholipid (15, 16, 72).
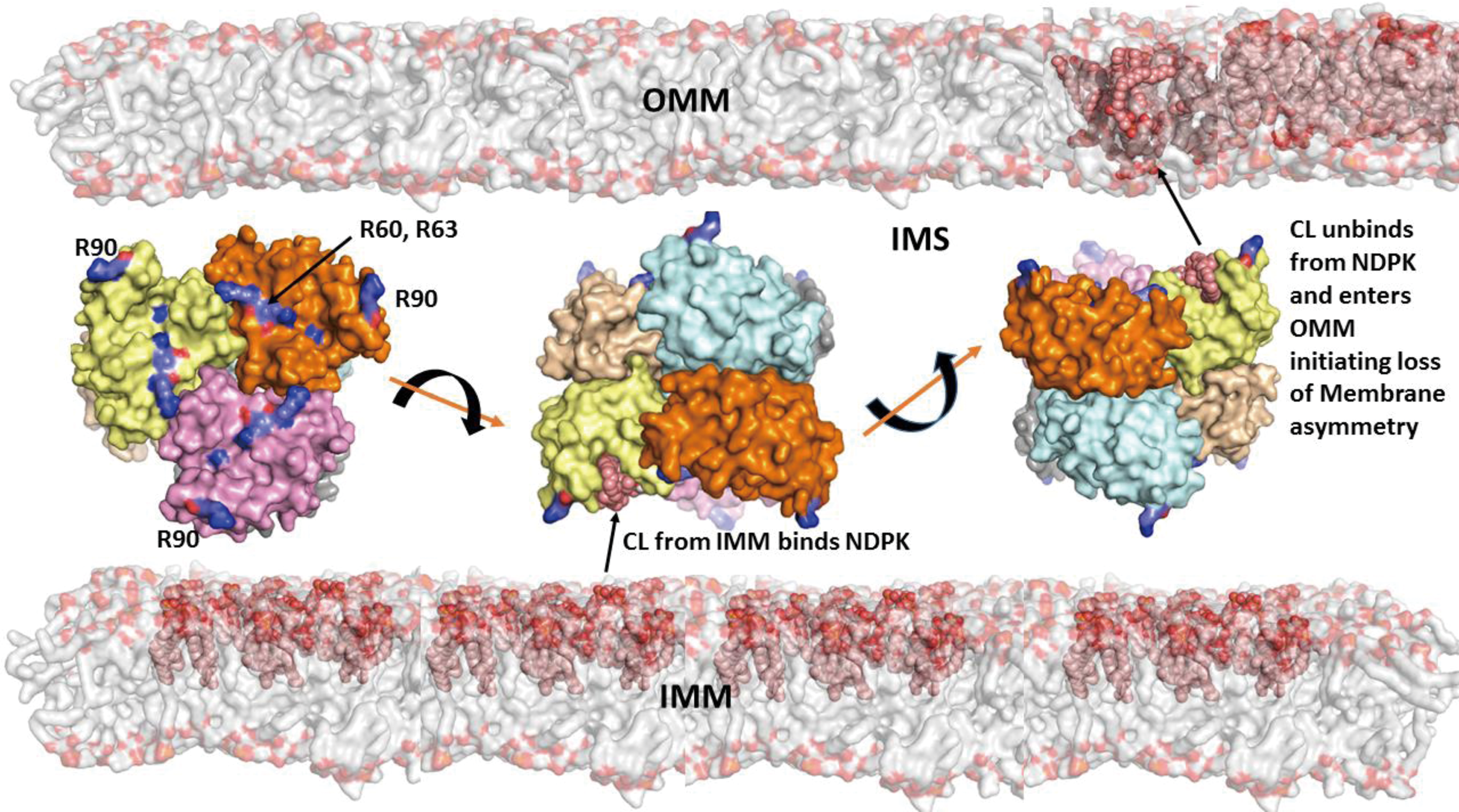
The central event in mitophageal signaling by CLs is the collapse of its membrane asymmetry and redistribution from the IMM to the OMM. The complete network of enzymatic mechanisms fulfilling this function remains enigmatic. Given that CLs are synthesized on the inner leaflet of the IMM and interact with LC3 after their appearance on the outer leaflet of the OMM, one can envision the necessity of three discrete translocations: within the IMM, between the IMM and OMM, and within the OMM (Fig. 3). As CL molecules include a bulky hydrophobic body and a doubly charged head, the travel through and in between two membrane surfaces is associated with significant energetic expenditures.
Currently, only one of the proteins involved in the CL transfer between the IMM and OMM has been identified—a mitochondrial isoform of nucleoside-diphosphate kinase D (NDPK-D) (Fig. 4) (74, 134). A well-known function of the enzyme is the transfer of phosphate groups between different nucleosides such as ATP>GDP, thus providing a pool of available GTP for mitochondrial GTPases (e.g., OPA1) (134). NDPK-D has been known to bind and transfer CLs between donor and acceptor liposomes (87). More detailed studies including LC-MS-based identification of molecular species of CLs in the IMM and on the mitochondrial surface revealed engagement of the hexameric protein in the process of CL externalization (74, 133). The CL-binding site includes R90 as the mutated protein product R90A lacked the binding and transporting capacity (74). Computational assessments of ligand binding energies suggest that the affinity of NDPK-D for CL is relatively low. This may be an important mechanism to preclude “excessive” sensitivity to fluctuations of free CL content in the IMM. The CL-dependent NDPK-D-mediated pathway, thus, may be an effective pro-survival factor, and in a number of episodes injury alternative signaling machineries may be triggered.
Several distinct receptors participating in the mitophageal process have been characterized (50). Among them are a ubiquitin-binding domain that localizes them to Parkin-ubiquitinylated mitochondria, including p62/SQSTM1, NBR1, and optineurin whereby Parkin binds AMBRA1 to localize it at the OMM. Another group of mitophagy receptors operates by engaging Bnip3, its homologue Bnip3L/Nix, FUNDC1, and Bcl2L13 (50). These mitophagy receptors have transmembrane domains that constitutively localize to the OMM. It is possible that CL externalization may be involved, at least in part, in realization of these alternative processes for maintenance of mitochondrial and cell health. In lieu of this, it is tempting to speculate that the recruitment and redistribution of some of these molecules into mitochondria (e.g., PINK1) may be associated with the presence of externalized CL on the mitochondrial surface. Finally, it is worth noting that CL externalization in mammalian mitochondria may reflect a more ancient mechanism that is also employed by prokaryotes. Indeed, in interactions of gram-negative bacteria with the target/host cells, bacteria externalize their CLs from the inner membranes to the surface (25).
IV. CL Oxidation Signals Apoptosis
Successful and effective elimination of damaged mitochondria via mitophagy may be sufficient for the protection of the injured cell from the risk of continuously spreading of damage. However, the failure of the cell to cope with the accumulating damage and (geno)toxicity via activation of repair processes, including DNA repair, necessitates the next elimination step—initiation of apoptosis, commonly via activation of p53-dependent mechanisms operating via mitochondria-dependent pathways (10, 85). This “sacrificial” behavior is particularly important for the protection of the entire cell community from possible release of dangerous factors, leading to uncontrolled spreading of the toxic impact.
When mitochondrial CLs are no longer confined to the IMM and have re-distributed into the OMM, they have essentially unlimited access to the intermembrane space components, including a mobile electron carrier, the small hemoprotein cyt c (126). Usually, the latter shuttles electrons between respiratory complexes III and IV, a function to which it is ideally adapted due to full occupancy of its coordination bonds (54, 169). Indeed, hexa-coordinated arrangement of the heme-iron in cyt c—with tetra-coordination by the heme and two axial ligands, Met80 and His18—precludes its unwanted capacities for a peroxidase-like catalysis.
Readily available CLs bind cyt c and change its coordination state by loosening heme-Fe-S bonding with Met80, thereby permitting the access for the pro-oxidant small molecules, such as H2O2, to act as a source of oxidizing equivalents and induce peroxidase competence of cyt c (6, 11, 100, 105). Radical migration from the porphyrine ring of the heme, typical for the peroxidase catalysis mechanism, occurs also in the cyt c/CL complexes whereby the formation of oxo-ferryl species (Fe4+ = O) takes place at the expense of the electron withdrawal predominantly from its proximity to the heme-Fe, Tyr67 (77, 80). The targets for the enzymatic oxidative attack by the peroxidase activity of cyt c/CL complex are PUFA residues of CLs (77). Accumulation of oxygenated forms of CL (CLox) in the OMM facilitates the release of cyt c from mitochondria into the cytosol, defining a point of no return in the apoptotic program (Fig. 5, left panel). Given recent evidence that formation of cyt c/CL complexes is important for the OMM permeabilization and the release of the hemoprotein from mitochondria into the cytosol (82), it is possible that CL oxidation facilitates this process.
The participation and essentiality of selective CL oxidation as a required mitochondrial stage of intrinsic apoptotic program has been demonstrated for many different types of wild-type cells. As expected, genetic manipulations causing deficiency of either CL-synthase as well as the entire cyt c or mutation of its Tyr67 residue suppressed the apoptotic responses (5, 9, 62, 95, 97). Moreover, in several in vivo models with characteristic massive apoptotic death, such as hyperoxia in mice, inhalation exposure to single-walled carbon nanotube in mice, total body irradiation (TBI) in mice, and traumatic brain injury in “pediatric” rats—the accumulation of CL oxidation products has been confirmed by quantitative LC-MS-based oxidative lipidomics analysis (12, 65, 153, 156, 158).
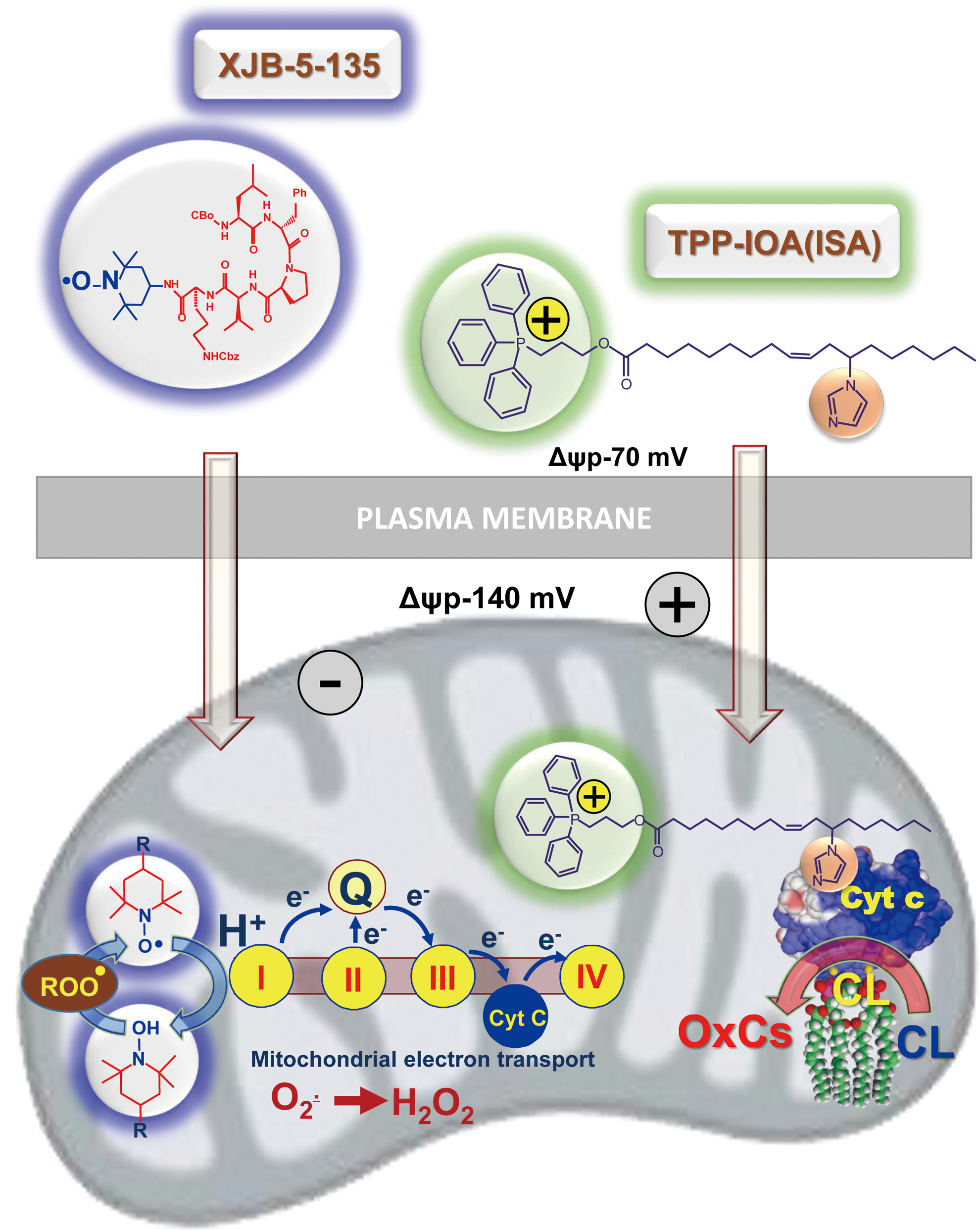
The importance of CLox accumulation as a pathogenic mechanism has been confirmed by the protective effects of several mitochondria-targeted inhibitors of CL oxidation (7, 37, 66, 69, 119, 148). Among them, a series of newly developed electron scavengers (XJB-5-125, XJB-5-131, JP4-039) capable of preventing the formation of H2O2 as a source of oxidizing equivalents for the cyt c/CL catalyzed reactions were very effective (Fig. 6) (37, 66, 119). Another group of effective regulators includes imidazole-substituted fatty acids whereby strong liganding of the imidazole group with the heme-Fe prevented its further participation in the catalysis of the peroxidase CL oxidation (Fig. 6) and protected against radiation-induced apoptosis in vivo (7).
As an enzymatic oxidation reaction, cyt c catalyzed oxidation of PUFA CLs is expected to display specificity toward different molecular species of phospholipids. Assessments of the specificity are complicated by the multiplicity of the products formed concomitantly in several reactions of CL oxidation during the same incubation, even in relatively simple biochemical systems (Fig. 7).
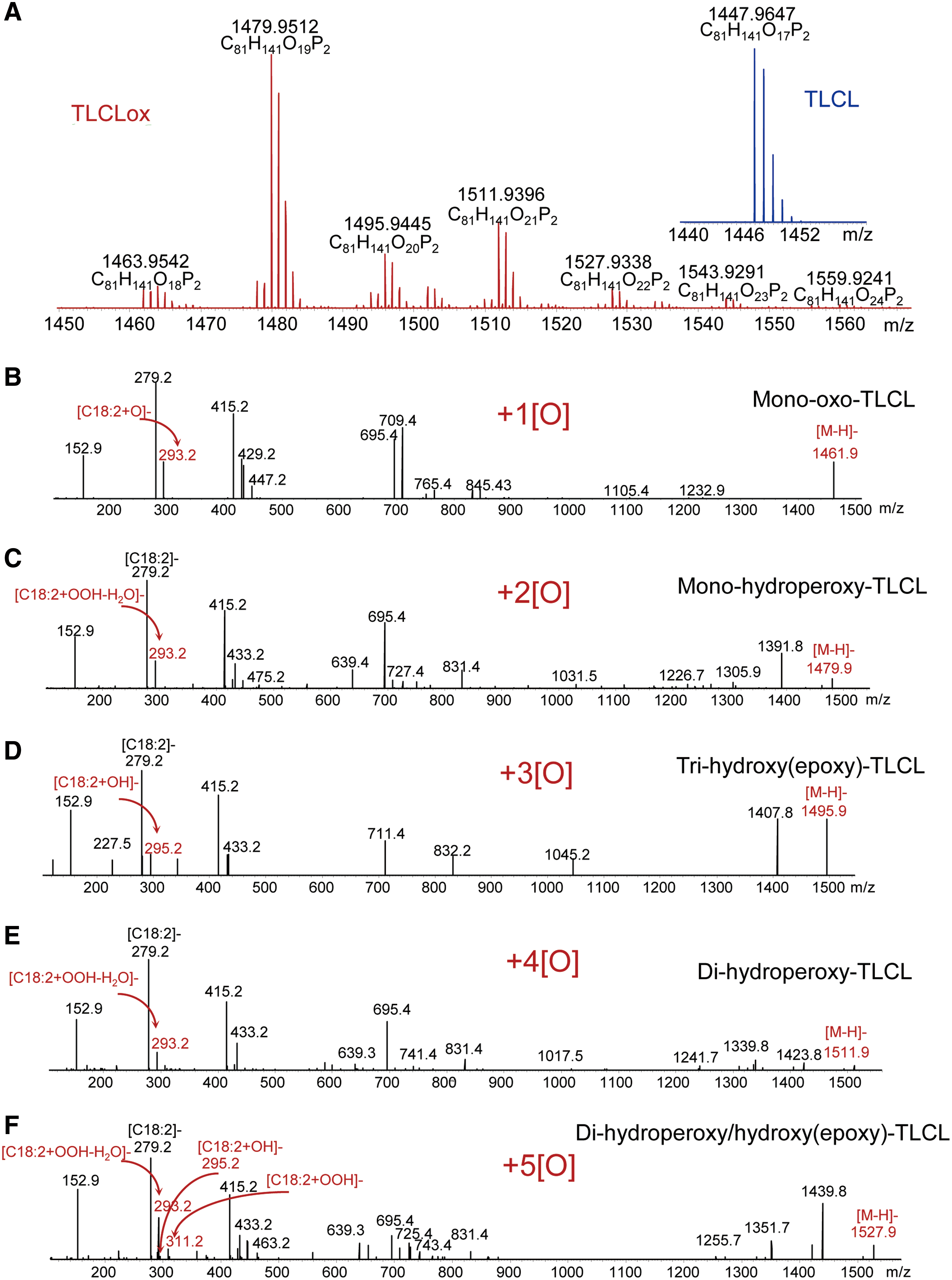
The evaluation of the CL oxidation specificity is difficult in cultured cells and even more complex in animal tissues, where a heterogeneous cell population such as inflammatory cells and professional phagocytes affect specific pro-oxidant features of the microenvironment and also clear injured and dying cells. Amazingly, in spite of all these potentially strong confounding factors, features of remarkable selectivity in the molecular speciation of CL oxidation products can be revealed. For example, after traumatic brain injury, oxidation products in hetero-acylated CL species containing C22:6, C20:4, and C18:2 residues were detected. The rules of random chemical oxidation predict that the most polyunsaturated FA residues should be preferred oxidation substrates. Instead, C18:2 residues were found to undergo preferential oxidation within the same CL molecule whereby C20:4 and C22:6 species remained nonoxidized (96).
Similarly, in the lung of animals exposed to hyperoxia, γ-irradiation, as well as inhaled carbon nanotubes, C18:2 residues were the major targets of oxidation whereas more chemically susceptible polyunsaturated residues displayed lower levels of oxidative modification (153, 156, 157). Overall, the available results lead to a conclusion that strict control of enzymatic processes rather than stochastic chemical reactions governs apoptosis-associated CL oxidation.
Among the numerous oxidation products, oxygenated CLs with one to eight added oxygen atoms are commonly detectable during LC-MS-based oxidative lipidomics analysis (65, 155). Aside from the enzymologic issues related to the mechanisms of cyt c catalyzed reactions, the important question is the relevance of the individual products to the execution of the apoptotic program. Indeed, although only ∼10 molecular species of CLox are found in the brain of normal rats, the elevated levels and more than 150 new oxygenated CLox species can be detected after brain trauma (65). Which of these multiple and diversified species of CLox are directly involved in the execution of an apoptotic program? Bioinformatics combined with the newly developed opto-lipidomics approach identified four molecular species of CLox that were persistently associated and common to several cases of apoptosis, when induced in different types of cells and tissues by different stimuli (96). All four of them contained mono-oxygenated C18:2 species, whose structural identity remains to be confirmed. Assuming that the involvement of CLox in the release of cyt c from mitochondria into the cytosol may be mediated by their interactions with one or more proteins in the OMM, one can speculate that electrophilic oxo- or epoxy-derivatives are likely candidates. Further oxidative lipidomics work, particularly based on the application of the new generation of more sensitive high mass accuracy instrumentation (e.g., a Tribrid™ Fusion™ Lumos™ mass spectrometer; ThermoFisher Scientific), may help in resolving these important questions.
V. PS Oxidation Signals Phagocytic Clearance of Apoptotic Cells
Our previous work has shown that execution of an apoptotic program in a variety of cell lines by different intrinsic pro-apoptotic stimuli resulted in selective oxidation of PS in plasma membrane [reviewed in Refs. (70, 71, 73)]. Further, we demonstrated that PS oxidation is important for its externalization on the cell surface. We also showed that PS species with docosahexaenoic fatty acid in sn-2 position were oxidized during apoptosis (152, 156). Notably, oxygenated PS molecules exposed on the surface of apoptotic cells are better recognized by professional phagocytes, which results in a more effective engulfment and digestion of apoptotic cells. Thus, not only PS (127, 136) but also oxidized species of PS act as universal “eat-me” signals (151).
VI. Oxidized PEs Signal Ferroptosis
The broad regulatory functions of oxygenated polyunsaturated lipids also include involvement in impaired metabolism and disease. In these situations, oxygenated polyunsaturated lipids potentially represent a source of endogenously generated hydrophobic electrophilic carbonyl compounds—aldehydes, ketones, and epoxides—capable of covalently reacting with essential nucleophilic sites in proteins, causing their post-translational modifications that can act as diversified signals or initiators of pathophysiological events (138, 161, 163). These secondary reactive lipid electrophiles are generated from the common hydroperoxy-precursors, the primary molecular products of lipid peroxidation reactions. Therefore, control of the hydroperoxy-lipids is operated by the key intracellular redox regulatory system—thiols—and their discoordination leads to ferroptosis, a nonapoptotic, iron-dependent form of regulated cell death (43).
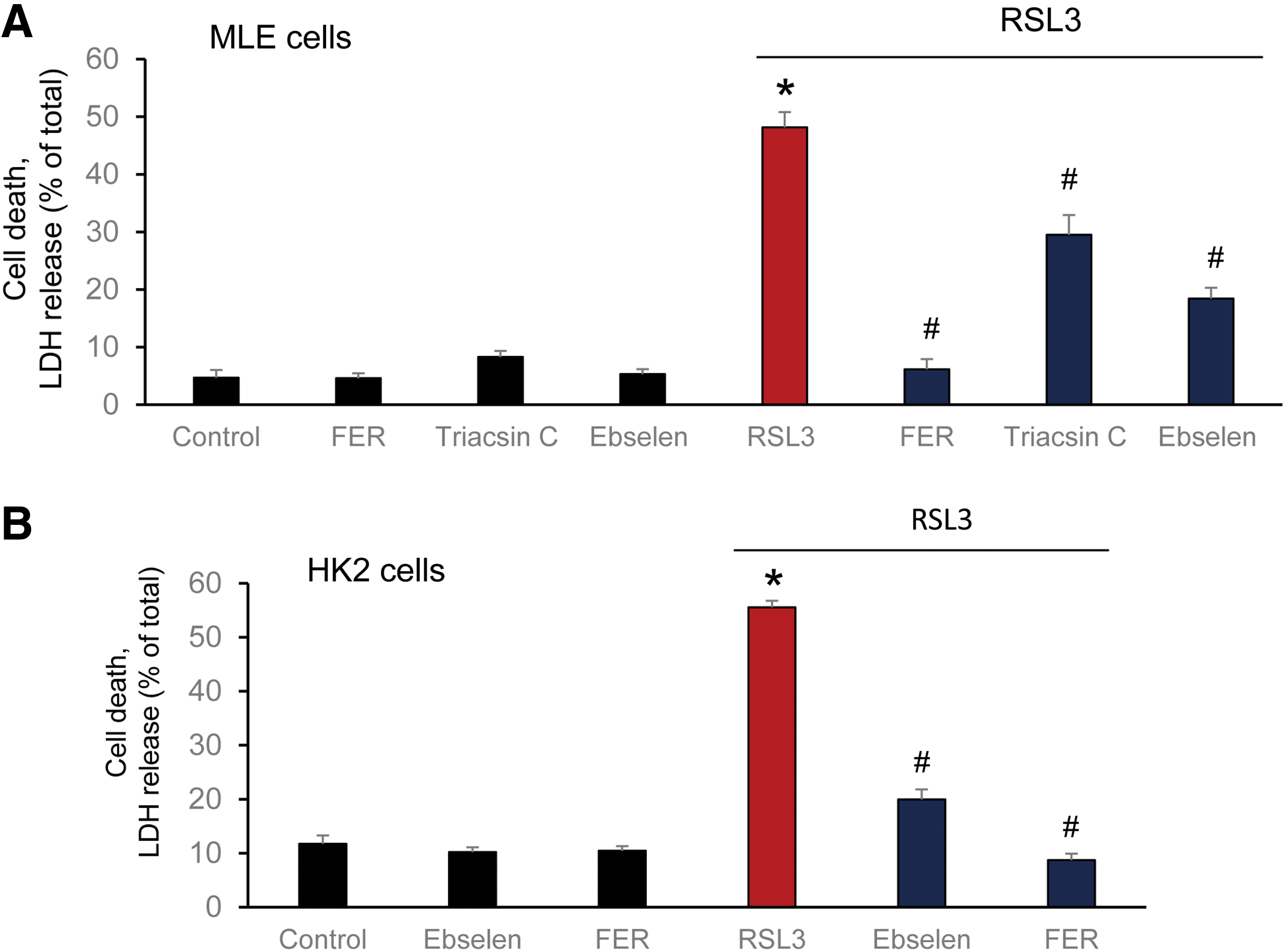
Several nonspecific fluorescence-based assays are commonly used to reveal the possible connection of ferroptosis with lipid peroxidation (40, 94). Although they all clearly indicate that the process of lipid peroxidation is somehow involved in the execution of ferroptotic program, the real acting products—their identity, structure, and amounts—until recently remained unknown (29, 173). Several lipid-related specific features of ferroptosis have been discovered: (i) deficiency/inactivation of the phospholipid hydroperoxides (PL-OOH) metabolizing enzyme, GPX4 (40, 173); (ii) sensitivity to iron chelators (29); (iii) rescue by LC-PUFA (arachidonic acid residue) specific acyl-CoA synthase (ACSL4) (30) and lysophospholipid acyl transferase 3 (LPCAT3) (30, 32); and (iv) suppression by vitamin E and other lipophilic antioxidants (29, 40). The sensitivity of the ferroptotic outcomes to the earlier mentioned factors has been established on a significant number of different genetically (30, 32) and pharmacologically manipulated cancer (29) and normal cell lines (Fig. 8). These facts not only clearly indicate that the process of lipid peroxidation is central and specific to the execution of ferroptosis (29, 172, 173) but also suggest that representatives of one or more classes of phospholipids are the proximate pro-ferroptotic agents.
Deciphering the nature of the proximate oxygenated lipid signals came from the use of the combination of LC-MS-based global oxidative phospholipidomics with reverse genetics, bioinformatics, and systems biology (75). It was established that only one class of phospholipids, PE, undergoes oxidation in the ER-associated compartments with the specificity toward two fatty acyls—arachidonoyl (AA) and adrenoyl (AdA). Suppression of AA or AdA esterification into PE by knocking out the expression of acyl-CoA synthase 4 (specific for AA) or inhibition of this enzymatic activity by Triacsin C led to strong protections against ferroptosis, thus confirming that blockade of synthesis of AA-/AdA–PE precursors represented a specific anti-ferroptotic rescue pathway (32).
It was further found that LO, particularly 15-LO, were mostly responsible for the generation of doubly and triply oxygenated PE species, which act as death signals. Notably, the 15-LO-mediated production of oxygenated PE derivatives occurred through attack on the phospholipid molecules rather than on free AA or AdA, with subsequent re-esterification of the hydroperoxy-fatty acids. Creation of GPX4 deficiency achieved through either genetic deletion or chemical inactivation (e.g., by RSL3) or depletion of GSH (e.g., by Erastin) as a source of reducing equivalents (29, 173) was absolutely necessary for ferroptosis induction, thus providing further evidence for the primary role of AA-OOH-PE and AdA-OOH-PE in the process (Fig. 5, right panel).
Thus, two metabolic pathways—lipid oxidation by 15-LO and reduction of the oxygenation products by GPX4—are tightly coordinated to generate vitally important oxygenated lipid signals with multiple physiological functions (64, 88, 135, 137). Dis-engagement of this system, particularly through the ablation of GPX4, results in the disturbed regulation of the production of electrophilic lipid mediators, thus necessitating the elimination of the impaired cell through the ferroptotic death mechanism.
In line with the complex multistage mechanism of the generation of ferroptotic signals, many small-molecule regulators can be used to control this pathway: (i) LO inhibitors (baicalein, ML351, PD146176), (ii) iron chelators preventing integration of Fe into LO; (iii) regulators of PUFA-PE biosynthesis and remodeling (e.g., inhibitors of ACSL4 like Triacsin C and LPCAT3 like thimerosal) (44); (iv) mimics of glutathione peroxidases, including GPX4 (e.g., Ebselen) (146); and (v) precursors of GSH (like N-acetyl-cysteine, NAC) (4). Interestingly, members of the vitamin E family—tocopherols and particularly tocotrienols—turned to be very effective in inhibiting LO as the generator of death signals, thus pointing to a new physiological role of vitamin E (75). Clearly, each of these regulators can be only effective if it compensates for a specific metabolic pathway deficiency in the biosynthesis of ferroptotic death signals.
Implicit in the name of this type of cell death, ferroptosis is an iron-dependent process. The exact role of iron, however, in the overall chain of reactions is still unclear. Although LO enzymes are involved in the generation of essential lipid peroxidation-derived death signals, it is conceivable that they are not unique as either enzymatic or nonenzymatic mechanisms that can trigger ferroptosis (31, 40, 75, 172, 176). There is a common opinion—although poorly substantiated by experimental data—that free iron (that can be scavenged by high-affinity chelators) may be mostly responsible for the pro-ferroptotic lipid peroxidation (176). It is also presumed that Fenton/Haber-Weiss chemistry (84, 175) leading to hydroxyl radical formation—rather than strictly regulated enzymatic peroxidation—is mostly accountable for the generation and emanation of hydroperoxy-lipids. Although the participation of nonenzymatic mechanisms in the production of these products cannot be fully excluded, the stochastic nature of these chemical reactions makes it very unlikely and essentially incompatible with the high specificity of the major pro-ferroptotic signals—15-hydroperoxy-AA/AdA-PE. Based on the partial identification of the pro-ferroptotic signals such as 15-hydroperoxy-AA-PE (15-HpETE-PE), it is likely that 15-LO, including both of its isoforms—15-LO1 and 15-LO2 (75)—define the specificity of lipid-driven death pathways in different tissues. It is also possible that other Fe-containing proteins, including hemoproteins and other representatives of the LO family, may act as important contributors to tissue-specific mechanisms of ferroptosis. Therefore, it is interesting to consider the relationships of LO-generated products released from different types of diverse cell types as regulators of several oxygenated lipid mediators in the context of their possible putative involvement in ferroptosis in vivo.
VII. Signaling by Free and Esterified Oxygenated Lipids in Acute Radiation Injury as an Inflammatory Disease: Sequential Engagement of Different Programmed Cell Death Pathways
TBI causes radiolysis of water—the most abundant component of our body in cells, extracellular compartments, and biological fluids (57). The reactive intermediates of this process—reducing and oxidizing radicals—randomly attack different components of cells although with different biological consequences largely defined by the uniqueness of modified molecules (89). Although aqueous radicals can directly interact with proteins, lipids, and carbohydrates, these reactions do not represent totally irreparable events and may be effectively repaired through standard catabolic and metabolic processes. In contrast, accumulation of single- and double-strand breaks in DNA beyond the capacity of the repair systems generates “danger” signals (117, 118). These danger signals produced in hematopoetic cells of bone marrow and epithelial cells of the small intestine—occurring early after the radiation exposure—trigger the mitochondrial mechanisms of intrinsic apoptosis, thus leading to elimination of cells—carriers of genotoxic materials (117, 118).
These initial events—although happening on a relatively small scale—are remarkably important. This is because the elimination of the primary sensitive cells directly injured by radiation awakens very strong responses of the innate immune system whose capacity to kill the neighboring cells within the microenvironment exceeds the chemical modifying potency of the initial radiation-induced intermediates of water radiolysis. Massive production of potent oxidants by activated neutrophils (predominantly hypochlorous acid) and macrophages (mostly peroxynitrite and hydroxyl radicals) causes substantial collateral damage by injurious modification of innocent by-stander cells. Engagement of these secondary target cells magnifies the innate immune response and, hence, propagates the initial direct radiation effect, thus initiating a vicious cycle of massive and delayed cell death (90, 103). Notably, the secondary death pathways are not limited to apoptosis but include other types of programmed death, such as necroptosis and ferroptosis (61).
Another enhancing cascade, breach of the intestinal epithelial barrier, designates the transition from “sterile” to infectious inflammation, ultimately leading to sepsis and multiple organ failure and thus worsening the overall prognosis and decreasing survival (46, 48). In spite of the calamitous nature of radiation inflammatory disease, its sequential development with characteristic phases and dominating cell death pathways gives an encouraging promise for multiple therapeutic approaches within the newly developed concept of precision medicine—accurately timed application of specific regulators targeting selective pathogenic mechanisms and pathways. Of course, the application of the principles of precision medicine necessitates the profound understanding of the mechanisms involved, particularly specific signals regulating recruitment, activation, and guidance of inflammatory cells to maximize their effectiveness in eliminating radiation-induced damage with minimal effects of “friendly fire” (170).
Among the major regulators of immune responses are different eicosanoids and docosanoids, as well as cytokines (14, 26, 27, 67, 171). Although experimental and clinical applications of cytokine analysis have been substantially improved over the past decade, the complexity of eiocosanoid and docosanoid signaling systems is much greater and lacks the necessary level of understanding. This particularly relates to esterification/de-esterification processes that may act via entirely different mechanisms and receptor systems (3, 83).
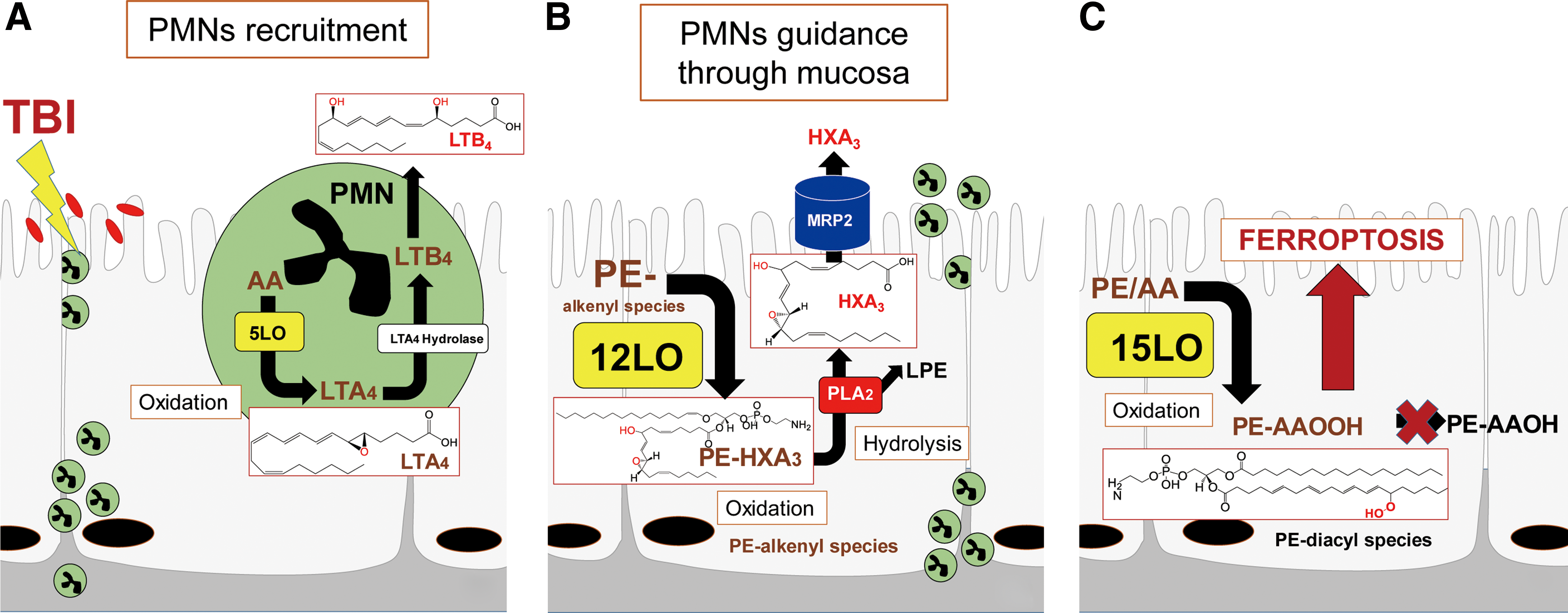
LC-MS analysis can provide unique information on the production of the most essential lipid mediators in free and esterified forms. This relates to the eicosanoids leukotriene B4 (LTB4) and hepoxilin A3 (HXA3)—the major regulators of neutrophil recruitment and penetration through the epithelium/mucosal layers when encountering bacterial pathogens (104, 106).
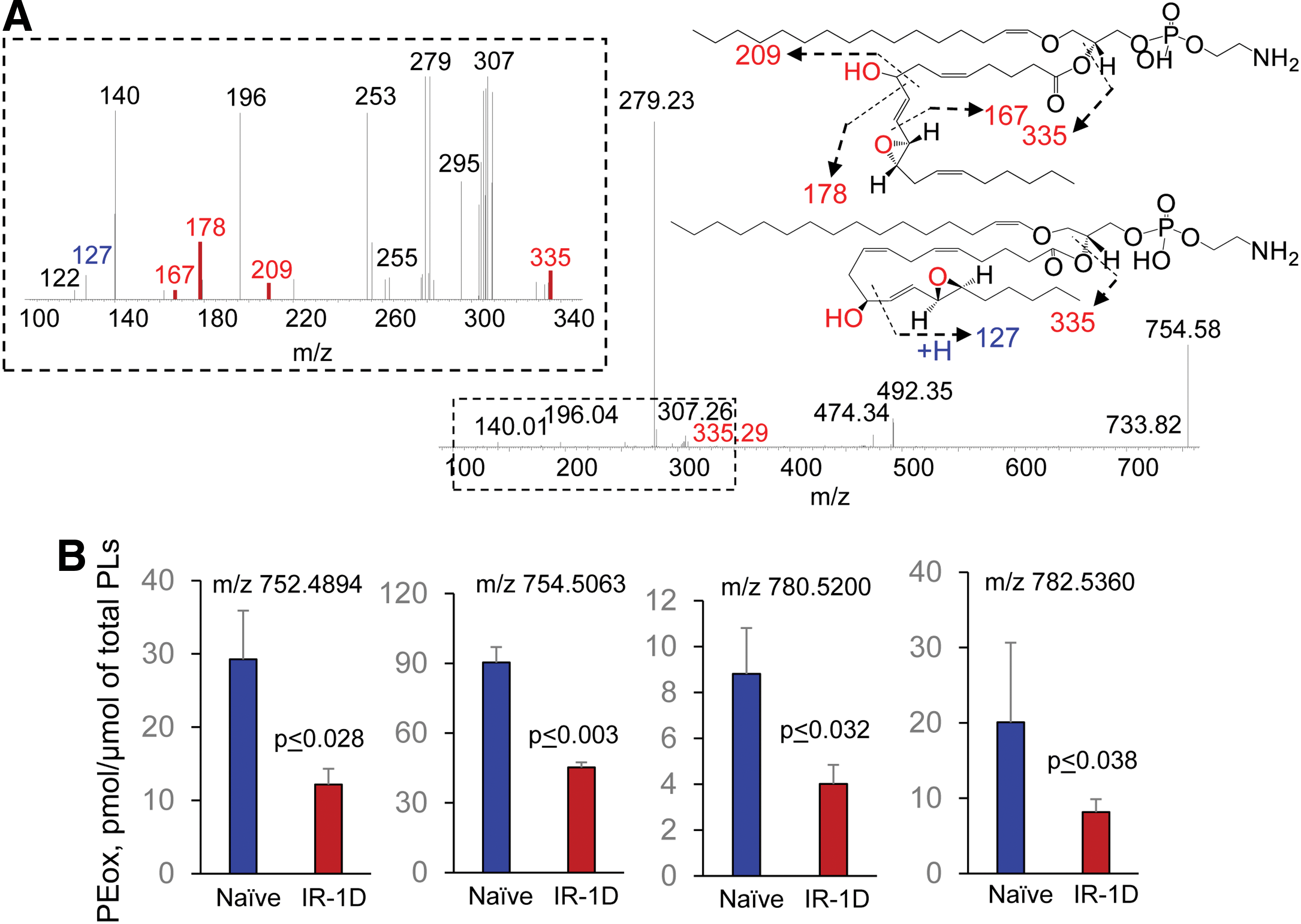
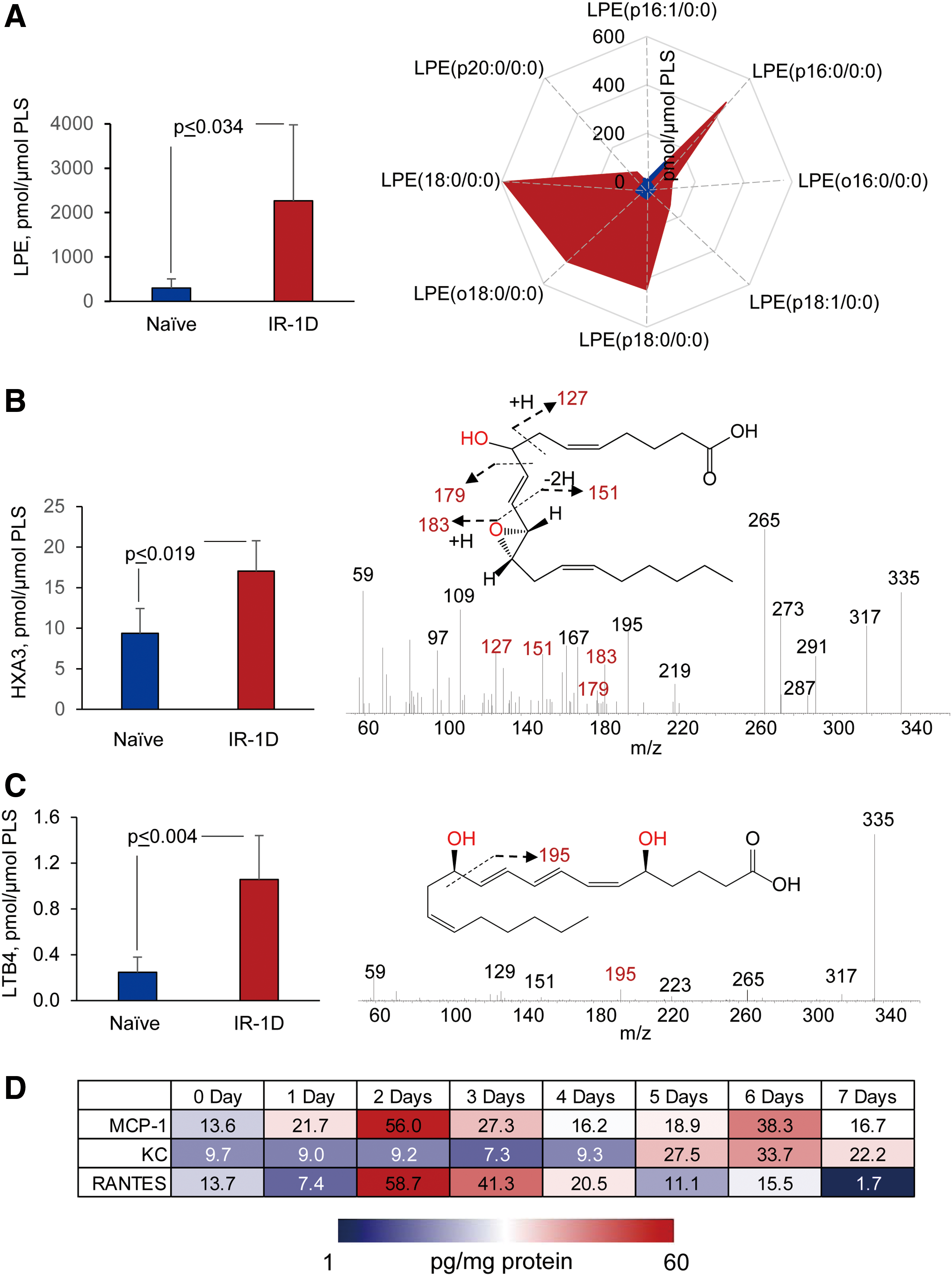
To illustrate some of the new opportunities offered by the LC-MS-based oxidative lipidomics in linking the prototypical radiation-induced responses with the production of esterified and nonesterified oxygenated lipid signals, we will consider the effects of a lethal dose of TBI (9.5 Gy) on damage to the ileum. The initial massive epithelial apoptosis is followed by the accumulation of activated neutrophils, where their dysregulated migration through the epithelium and “discharge” of reactive oxidizing intermediates leads to disintegration and breach of the epithelial barrier (Figs. 9 and 10). Phospholipidomic assessments of nonoxidized PE metabolites revealed 115 molecular species of PE based on accurate mass analysis (Fig. 11 and Table 1). In the ileum, high levels of oxidizable PUFA acyls—linoleic (C18:2), arachidonic (C20:4), eicosapentaenoic (C20:5), adrenic (C22:4), docosapentaenoic (C22:5), and docosahexaenoic (C22:6)—are esterified into diversified PE species (Table 1). Accordingly, MS/MS analysis identified di-oxygenated signals of PE with HXA3 esterified into sn-2 position (PEp16:0/HXA3; PEp18:0/HXA3) (Fig. 12A). Early after TBI (day 1), a significant decrease of doubly-oxygenated PE species (Fig. 12B) was accompanied by the accumulation of both lyso-PE (LPE) (Fig. 13A) and free HXA3 (Fig. 13B). This suggests the involvement of 12-LO in the radiation-induced responses whereby a direct attack on AA-PE substrate and oxygenation of the phospholipid is likely followed by hydrolysis of the oxygenated product to yield free HXA3 and lyso-PE. Further, a fourfold accumulation of LTB4—an eicosanoid that is synthetized by 5-LO in neutrophils and is involved in inflammatory response—was also observed (Fig. 13C).
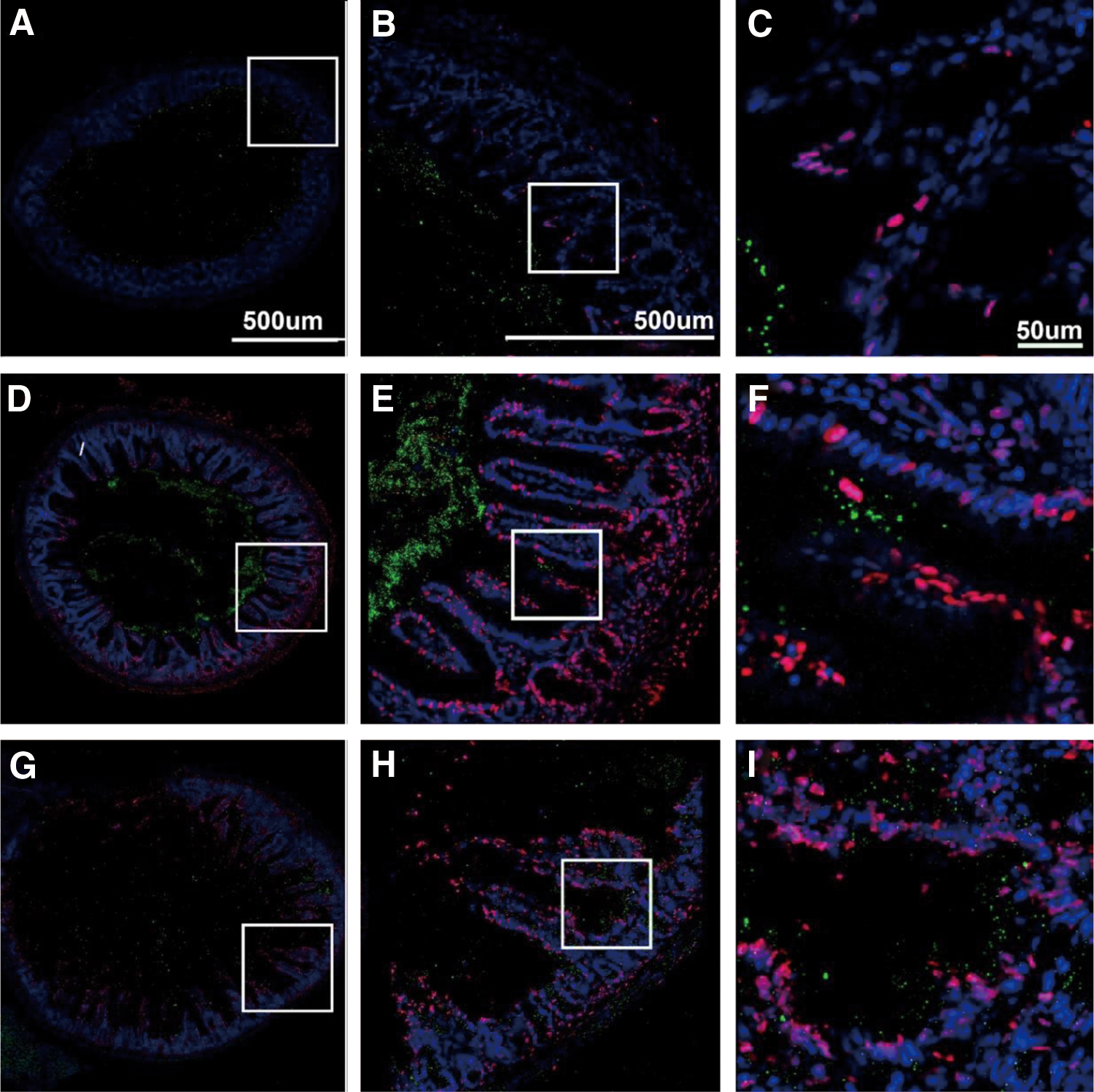

PE, phosphatidylethanolamine.
Radiation-induced lipid oxygenation responses were also accompanied by typical changes in the levels of cytokines and chemokines participating in the recruitment and regulation of neutrophils (Fig. 13D). Thus, radiation-triggered responses include a series of sequential reactions associated with the recruitment and guidance of neutrophils to and within the inflammation site (Fig. 9). The detailed characterization of PE oxidation products as potential sources of important lipid mediators—along with the detailed time course of cytokine formation—may be important for the development of mechanism-based targeted and “personalized” therapeutic approaches.
Concomitantly with these processes of phospholipid oxygenation and hydrolysis—leading to the release of “free” signaling lipid mediators—accumulation of esterified oxygenated phospholipids associated with different types of programmed cell death occurred. Cyt c catalyzed oxidation of CL and 15-LO catalyzed oxidation of AA-PE were also triggered, resulting in the production of pro-apoptotic and pro-ferroptotic signals, respectively (156, 157) (Fig. 14). Fragmentation patterns of PE species from the ileum showed that 18 molecular species contained arachidonic acid (20:4)—potential precursors of ferroptotic death signals. Robustly increased levels of PEox were detected at 5 days postirradiation (Fig. 14). Overall, 27 oxygenated PEs were revealed by oxidative phosholipidomics, of which doubly oxygenated PEs represented the most abundant derivatives, in terms of both the number of species (Fig. 11) and their contents (Fig. 14A). Structural analysis revealed 14 doubly oxygenated species of PE versus 2 mono-, 6 tri-, and 5 four- oxygenated PE species. Among doubly oxygenated PE species, PE containing doubly oxygenated arachidonic acid (PE38:4-OOH), a ferroptotic death signal, predominantly accumulated in the ileum on day 5 after irradiation (Fig. 14B).
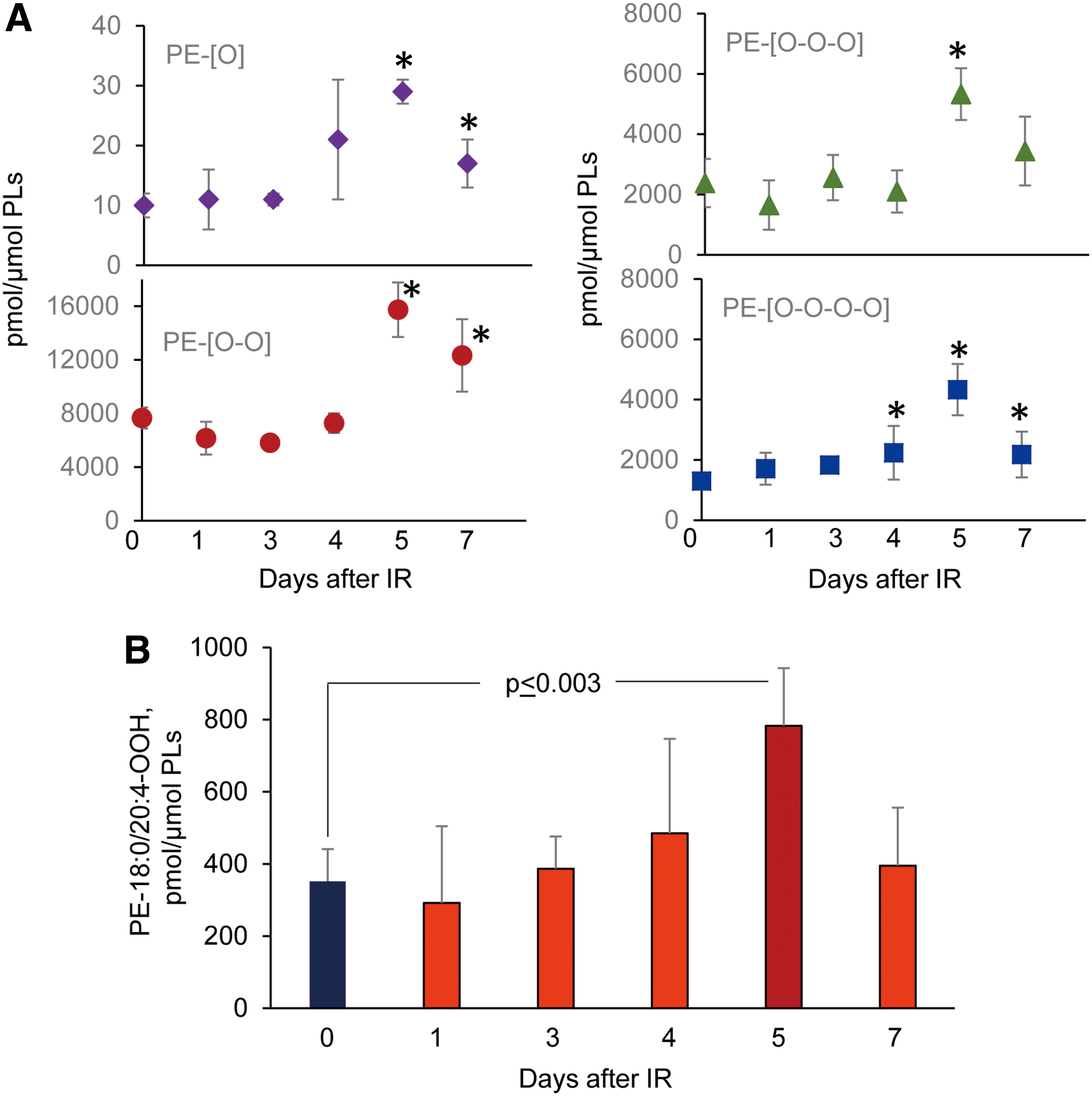
Overall, these data illustrate the complexity of lipid mediator metabolism and lipid mediator signaling triggered by TBI in a radiosensitive tissue (Fig. 15). Further insights into the generation and steady-state levels of these products may lead to identification of reliable indicators of γ-irradiation-induced responses in vivo as important targets for the discovery and development of radioprotectors and radiosensitizers.
VIII. Concluding Remarks
Aerobic multicellular life has enormously benefited from the emergence of PUFA lipids, due to at least two remarkable new propensities: (i) adaptive capacities to fine-tune lipid bilayer modifications to maximize the effectiveness of the membrane proteins, and (ii) elaborate an extraordinary rich language including millions of naive and oxidatively modified lipid molecules as words-signals. With regards to oxygenated, such as octadecanoids, eicosanoids, docosapenta- and docosahexanoids, numerous meaningful lipid mediators and their receptors controlling major functions of cells and tissues have been identified. In contrast, oxygenated PUFA esterified into phospholipid molecules have been mostly associated with oxidative stress, damage, and disease conditions and only recently emerged as important regulators of normal physiological processes. Of course, free PUFA and PUFA phospholipids may be closely linked via effective inter-conversions by reacylation and hydrolysis enzymatic reactions, respectively, as has been recently demonstrated for oxidized forms of mitochondrial oxygenated CLs (155). However, the most common pathways for the production of free PUFA lipid mediators are realized via initial hydrolysis of PUFA phospholipids by phospholipases A2 followed by subsequent enzymatic oxygenation reactions (110). Both direct oxygenation of PUFA phospholipids and esterification of oxygenated-free PUFA into phospholipids have been described as metabolic pathways generating meaningful signals (3, 52, 53). The significance and importance of these two different forms of oxygenated lipids in multiple specific functions are the subjects of active research.
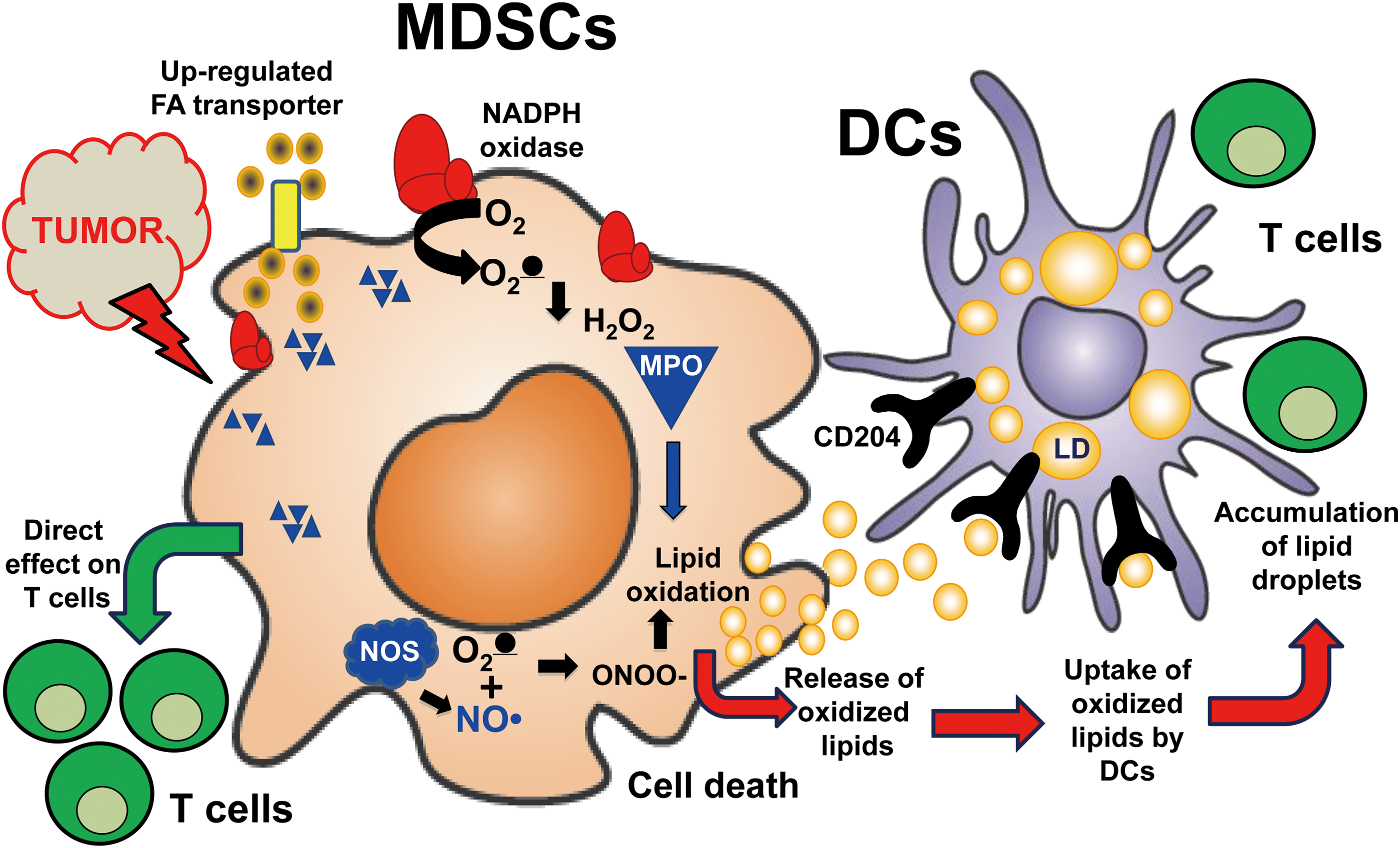
Although oxygenated phospholipids have been considered important sources of lipid mediators, the signaling by oxygenated PUFA-containing neutral lipids, particularly of triacyl and diacyl-glycerols, is an emerging new direction of oxidative lipidomics research (2, 63, 102, 116). Notably, aberrant oxidative metabolism of fatty acids commonly results in the accumulation of lipid droplets that may contain highly oxidizable PUFA-containing neutral lipids (102). The disturbances in neutral lipid metabolism may occur in many diversified conditions with their specific pathophysiological significance and mechanisms. For example, the profiles and activity of placental enzymes, receptors, and transporters defining the balance between free and esterified PUFAs determine the extent of maternal lipid transfer to the fetus strongly affecting the fetal fat accretion (45). In cancer, dysregulated lipid metabolism may be accountable for the distorted surveillance mechanisms and suppressed functions of immune cells over the growing tumor (42, 164). Recently, the important role of oxygenated neutral lipids—PUFA triacylglycerols and free PUFA—in the inhibited processing and cross-presentation of tumor antigens to T cells by dendritic cells and the complex relationships of the latter with myeloid-derived suppressor cells has been documented (120) (Figs. 16 and 17).
Uninterrupted and effective exchange of information between different types of cells in the microenvironment is integral to the maintenance of normal tissue homeostasis. The danger signals that are generated by the multicellular communities inform the irreparable injury of a particular cell, and they lead to its elimination through one of multiple specific programmed cell death pathways—in line with the principle presented in the title of this work: “Only a life lived for others is worth living.” More than a dozen of these programs and the respective types of cell death have been identified and associated with specific features of cellular injury (19, 34, 47, 68, 93, 162, 168). Here, we focused on two pathways—apoptosis and ferroptosis—and demonstrated a very high specificity of phospholipid signaling by oxygenated species of CLs and PEs, respectively. Although the identification of these oxygenated lipid signals is an important step achieved through oxidative lipidomics, the molecular mechanisms of their action leading to specific features of cell membrane damage and penultimate executions of apoptotic and ferroptotic programs are still unclear.
A quantitative comparison of the lost PUFA PLs and detectable steady-state concentrations of PL oxidation products suggests that the latter represent only a fraction of the former. What is the fate of the “disappeared” oxygenated PLs? One of the possible explanations is that they form adducts, with the proteins, as has been discovered for electrophilic-free oxygenated PUFA products. For the electrophiles formed from free PUFA, some of the protein targets have been identified by LC-MS-based redox proteomics (2, 17). To the best of our knowledge, this has not been accomplished for the electrophiles produced from oxidatively modified (truncated) PLs. It is likely that one of the major reasons is their remarkable diversification minimizing hopes for success. Identification of only three oxygenated CLs and four oxygenated PEs as biomarkers of apoptotic and ferroptotic death signals, respectively, narrows the search for the potential modified proteins—the targets of covalent modification by the oxidatively truncated electrophiles derived from these CLox and PEox precursors.
Although the number of potential nucleophile-rich targets may be quite large, the knowledge obtained through global oxidative lipidomics combined with the specific conditions for triggering these two programs of cell demise creates optimistic prognosis for the next move toward oxidative lipo-proteomic identification of the ultimate executors of cell death programs.
Footnotes
Acknowledgments
This work was supported by NIH (P01HL114453, U19AI068021, NS076511, NS061817, CA165065, ES020693), and HFSP-RGP0013/2014, March of Dimes Pennsylvania, University of Pittsburgh Vascular Medicine Institute, the Barth Syndrome Foundation, Inc., and the Barth Syndrome Foundation of Canada.
