Abstract
Significance:
Carbonyl sulfide (COS) is the most prevalent sulfur-containing gas in the Earth's atmosphere, and it plays important roles in the global sulfur cycle. COS has been implicated in origin of life peptide ligation, is the primary energy source for certain bacteria, and has been detected in mammalian systems. Despite this long and intertwined history with terrestrial biology, limited attention has focused on potential roles of COS as a biological mediator.
Recent Advances:
Although bacterial COS production is well documented, definitive sources of mammalian COS production have not been confirmed. Enzymatic COS consumption in mammals, however, is well documented and occurs primarily by carbonic anhydrase (CA)-mediated conversion to hydrogen sulfide (H2S). COS has been detected in ex vivo mammalian tissue culture, as well as in exhaled breath as a potential biomarker for different disease pathologies, including cystic fibrosis and organ rejection. Recently, chemical tools for COS delivery have emerged and are poised to advance future investigations into the role of COS in different biological contexts.
Critical Issues:
Possible roles of COS as an important biomolecule, gasotransmitter, or sulfide transport intermediate remain to be determined. Key advances in both biological and chemical tools for COS research are needed to further investigate these questions.
Future Directions:
Further evaluation of the biological roles of COS and disentangling the chemical biology of COS from that of H2S are needed to further elucidate these interactions. Chemical tools for COS delivery and modulation may provide a first avenue of investigative tools to answer many of these questions. Antioxid. Redox Signal. 28, 1516–1532.
Introduction
S
Despite the significant research on the chemical biology of NO, CO, and H2S, investigations into other potential gasotransmitters, such as sulfur dioxide (SO2), ammonia (NH3), or carbonyl sulfide (COS), remain significantly underdeveloped (Fig. 1) (84, 149). Both SO2 and NH3 are produced enzymatically in mammalian cells and are interconnected with established gasotransmitters, giving credence to the suggestion that they may play significant roles in biology. For example, SO2 can be generated from H2S by NADPH oxidase or from thiosulfate (S2O3 2–) by thiosulfate sulfurtransferase (52, 90). Alternatively, cysteine oxidation by cysteine dioxygenase (CDO) generates cysteine sulfonate, and subsequent transamination by aspartate aminotransferase (AAT) generates β-sulfinylpyruvate, which spontaneously decomposes to extrude pyruvate and SO2 (123, 129). Although still in its infancy, early investigations into possible biological actions of SO2 suggest roles as a vasorelaxant and in providing protection against myocardial ischemia-reperfusion injury (52). Similarly, NH3 is formed in many pathways, including deamination of amino acids, nucleic acids, nucleotides, and nucleosides; as a byproduct of transsulfuration enzymes, including H2S-producing cystathionine γ-lyase (CSE) and CBS; and from urea recycling (149). Once formed, NH3 may not only provide a viable form of nitrogen for DNA and RNA synthesis but also contribute to acid-base buffering capacity. In addition, NH3 has been demonstrated to increase inducible nitric oxide synthase (iNOS) expression in cultured astrocytes (49) and also to increase iNOS and neuronal nitric oxide synthase (nNOS) expression in animal models of hyperammonemia (131).
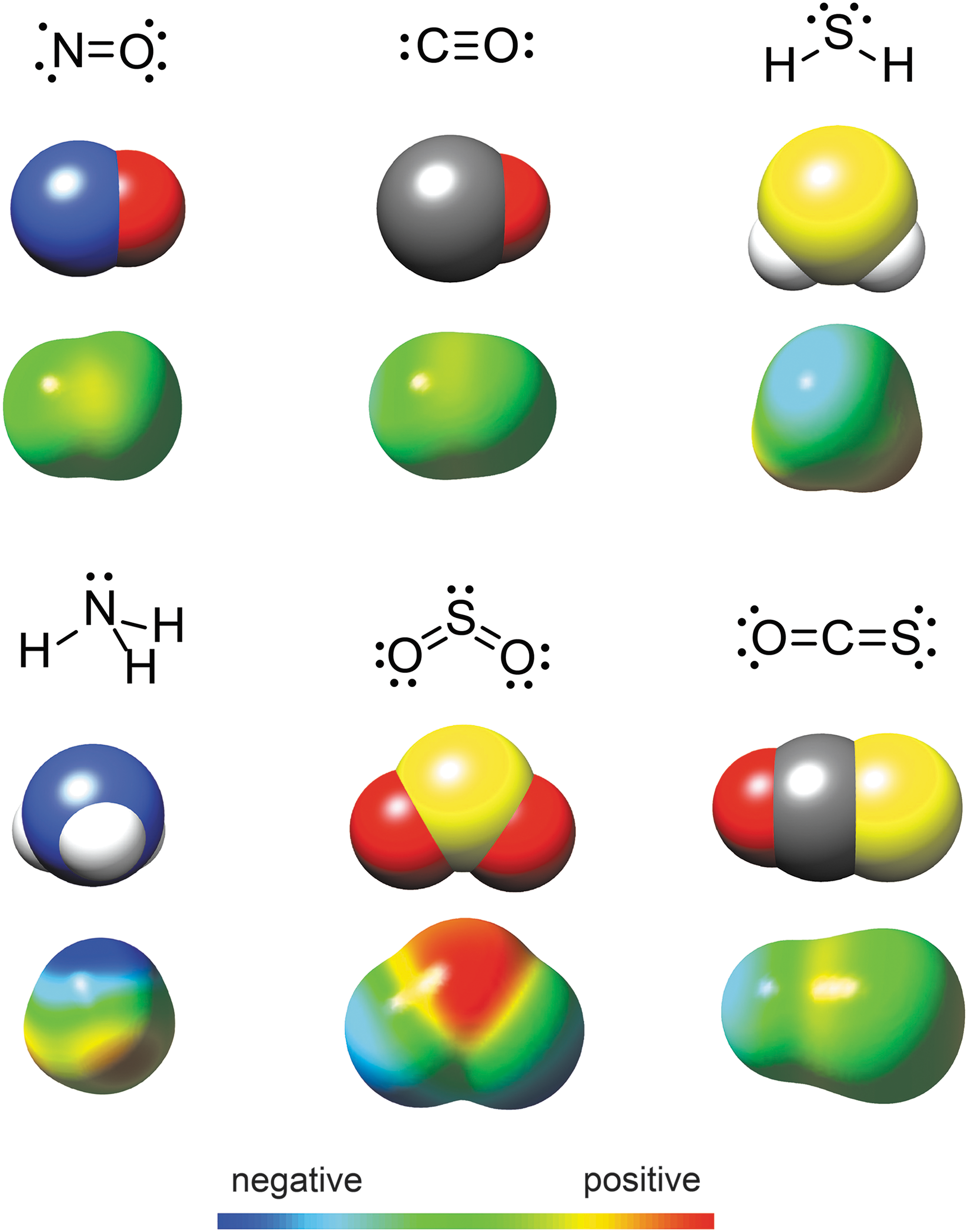
Unlike SO2 or NH3, pathways for enzymatic COS synthesis in mammals have yet to be identified, although COS has been detected in various biological tissues and in exhaled breath, supporting the presence of pathways for endogenous generation (vide infra) (7, 60, 121). Furthermore, COS shares an interconnection with H2S generation through the action of different metalloenzymes, including carbonic anhydrase (CA), which rapidly converts COS to H2S (22, 67, 119). Although historical investigations of terrestrial COS have focused on its atmospheric presence and importance in the global sulfur cycle, contemporary chemical investigations have focused primarily on COS-mediated peptide-bond formation under prebiotic conditions, many of which suggest important roles of COS in origin of life chemical ligation (53, 75). When taken together, the potential role of COS in thermophilic origin of life theories, COS detection in tissues, as well as its cell permeability and moderate water solubility, suggests that COS may play a much more significant role in mammalian chemical biology than initially appreciated. Building on these factors, this review focuses on current knowledge of biological COS formation and consumption, growing evidence that COS may play roles in sulfide transport and disease pathology, emerging chemical tools for investigating COS in biological contexts, and the potential role of COS as a new member of the gasotransmitter family.
Basic Properties
COS (CAS 463-58-1, also referred to as carbon oxysulfide, CO monosulfide, carbon oxide sulfide, OCS) is a colorless and odorless gas in its pure form (41). Initially misidentified as a mixture of carbon dioxide (CO2) and H2S due to its rotten egg smell in an impure state, COS was first characterized by Than in 1867 by the reaction of CO with elemental sulfur vapor in a glowing porcelain tube (Eq. [1]) (8). Although removal of CO impurities was not practical from the initial preparations, experimental modification allowed for generation and purification of COS by acid-mediated hydrolysis of thiocyanate (SCN−) salts (Eq. [2]) (8). This reaction can be used to prepare COS in the laboratory, but the produced gas requires significant purification due to common contamination by gaseous impurities (113). Commercial sources of high-purity COS (generally >97.5%) often contain significant levels of H2S as the main impurity. Although only moderately soluble in water, COS is stable in acidic solution but undergoes base-mediated hydrolysis to generate H2S and CO2 (41). As a moderately lipophilic gas [log Poct(COS) = 0.79 by comparison to log Poct(NO) = 0.70] (57), the dipole moment of COS is more similar to that of NO, CO, and H2S rather than the significantly more polar SO2 and NH3. These properties suggest a sufficient lipid solubility to enable cell membrane permeability and also penetration to the central nervous system. Basic properties of COS, as well as those of H2S, NO, CO, SO2, and NH3 are provided in Table 1.
CO, carbon monoxide; COS, carbonyl sulfide; H2S, hydrogen sulfide; NH3, ammonia; NO, nitric oxide; SO2, sulfur dioxide.
Toxicity and safety
Much like H2S, NO, and CO, COS is a flammable gas that is toxic in high concentrations. COS is a skin, eye, nose, throat, and lung irritant, matching many of the topical toxicity characteristics of H2S. These similarities are likely due to the hydrolysis of COS to form H2S on contact with different mucosal membranes that typically contain CA, thus facilitating H2S generation (vide infra). Similarly, in toxicological investigations of COS, treatment of rats with the CA inhibitor acetazolamide (AAA) reduces measured blood levels of H2S, and also the toxicity of COS, suggesting that at least some of the observed COS toxicity is due to CA-mediated metabolism to H2S (22, 23). Although specific hazardous concentrations of COS are not specified by the US Environmental Protection Agency (EPA), some reports have documented irritation in the upper respiratory tract at concentrations above 20 mg/L, although different toxicity thresholds have also been reported in different investigations (137). Low-to-moderate concentrations are accepted to elicit lachrymatory effects, photophobia, nausea, increased salivation, headache, mental confusion, as well as other characteristics (137). Higher COS concentrations can result in decreased vision, tachycardia, and collapse, and continuous exposure to COS concentrations of 0.1% (v/v) (1000 ppm) can result in death within 2 h due to respiratory paralysis. A limited number of animal studies have investigated acute or chronic COS toxicity, but toxicological investigations of 50 ppm levels of COS in rabbits for a maximum of 7 weeks did not impact the myocardial ultrastructure significantly (54). Similarly, toxicological investigations in rats revealed LC50 of more than 2000 mg/m3 (∼750 ppm), with further investigations classifying COS as a non-carcinogenic, low-toxicity fumigant (150).
Natural sources
COS is the most prevalent sulfur-containing gas in the Earth's atmosphere and is produced by both biological and chemical pathways (Fig. 2) (46, 137). Primary abiotic COS emissions occur from volcanos, hot springs, and oceans, with biotic sources stemming from soils, trees, marshes, plant roots, manure, microorganisms, and biomass burning (see the Biological Roles of COS section for more detailed information) (151). Up to one half of terrestrial COS is generated from secondary production in the global sulfur cycle through oxidation of atmospheric dimethyl sulfide (Me2S) and carbon disulfide (CS2) (151). Anthropogenic sources of COS, which include aluminum production, coal and automobile fuel burning, and industrial desulfurization, contribute minimally to global COS production, but more recent estimates indicate that industry and manufacturing may produce more COS than has been previously reported (74). Measurement of atmospheric COS levels from Antarctic ice cores have shown that COS levels have risen over the past 350 years, which has been attributed to human sources of industrialization (5). Once produced, COS is more stable in the atmosphere than the common sulfur-containing gases Me2S, H2S, and CS2, which results in a prolonged atmospheric lifetime of about 4 years (68). As a result, COS is generally transported from the troposphere to the stratosphere, where it undergoes photodissociation and oxidation to SO2 and sulfate particles, therefore influencing stratospheric ozone concentrations (137, 139). The long atmospheric lifetime, as well as its formation from CS2 in the atmosphere, makes COS the primary sulfur gas in the Earth's atmosphere, with measured concentrations of 0.5 ppb (151).
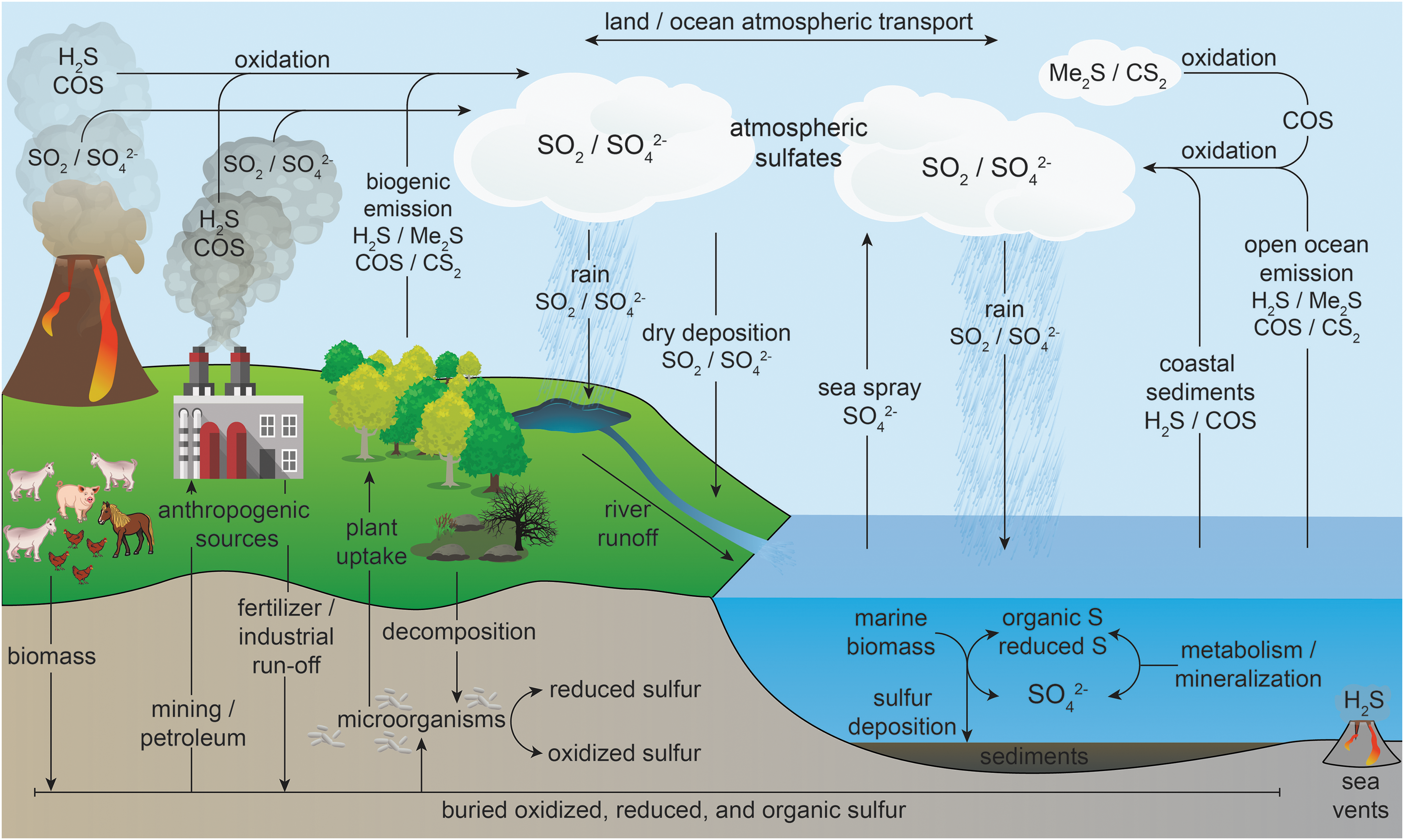
Although oxidation of COS to sulfate occurs in the atmosphere by hydroxyl radical, the major sink of atmospheric COS is uptake by many plants, soils, marine algae, and microbes, which often convert COS to CO2. This conversion is typically mediated by the ubiquitous enzyme CA as well as by other COS-metabolizing enzymes (vide infra). In addition to COS metabolism by CA, some plants are able to convert COS to CS2, as demonstrated by increased CS2 release after COS absorption by moist soils (88). These observations suggest a link between CS2 and COS not only in the atmosphere but also in soils and vegetation.
Biological Roles of COS
Growing evidence supports the importance of biologically relevant reactive sulfur species, such as persulfides, polysulfides, thiosulfate, or other partially oxidized forms of sulfur-based compounds, but surprisingly, little attention has focused on the most-prevalent sulfur-containing gas in the Earth's atmosphere: COS. Reported investigations into the biological roles of COS remain sporadic, and our understanding of the potential physiological roles and pharmacological roles of COS remain underdeveloped. Despite these limited reports, enzymatic COS production and consumption are well documented, and COS generation in cell culture and in exhaled breath in human subjects suggests a broader role for this important gas in diverse biological processes.
Enzymatic consumption of COS
CAs are a family of ubiquitous metalloenzymes that catalyzes the reversible hydration of CO2 to bicarbonate (HCO3 −) (78, 83). CAs comprise five classes found primarily in vertebrates (α-CAs), higher plants and some prokaryotes (β-CAs), archaebacteria (γ-CAs), and diatoms (δ-CAs and ξ-CAs). Although not its natural substrate, CA is also able to readily hydrolyze COS to H2S, providing a broad enzymatic platform for redox-neutral conversion of COS to H2S. The catalytic efficiency (k cat/KM) of CA-mediated COS conversion to H2S is less efficient than the canonical CO2 metabolism (∼8 × 107 M −1 s−1) (66), but it still boasts a high catalytic efficiency of 2.2 × 104 M −1s−1 for bovine CA-II (Table 2) (47). Drawing parallels to CA, other enzymes are also able to catalyze the hydrolysis of COS, even though it is not their natural substrate. For example, CS2 hydrolase, nitrogenase, CO dehydrogenase, and ribulose-1,5-bisphosphate carboxylase oxygenase (RuBisCO) have all been reported to hydrolyze COS (36, 80, 120, 125). Of these enzymes, CS2 hydrolase, initially isolated from acidophilic and thermophilic archaea extremophiles (125), exhibits the greatest catalytic efficiency of enzymatic COS hydrolysis (Table 2). Because the distribution of this enzyme is limited to extreme and sulfur-rich environments, such as sulfotaras, it is unlikely that CS2 hydrolase contributes significantly to global COS consumption. The only known example of an enzyme for which COS is the natural substrate is carbonyl sulfide hydrolase (COSase), which was recently identified, purified from Thiobacillus thioparus strain THI115, and characterized (95, 106). COSase shares both a high sequence homology and a similar Zn(II) active site with the β-CAs and is structurally similar to enzymes in clade D of the β-CA phylogenetic tree (95). Importantly, COSase has a higher efficiency than widely distributed enzymes that are able to catalyze COS hydrolysis, suggesting that it may play an important role in the global consumption of atmospheric COS. Although less efficient than COSase, other metalloenzymes, such as CO dehydrogenase (36), RuBisCO (80), and nitrogenase (120), have been shown to metabolize COS in plants and bacteria with varied levels of catalytic efficiency (Table 2).
CA, carbonic anhydrase; COSase, carbonyl sulfide hydrolase; CS2, carbon disulfide; RuBisCO, ribulose-1,5-bisphosphate carboxylase oxygenase.
Modified from Ogawa et al. (95).
Biological production of COS
SCN− is a common organic anion found in plants, mammals, and natural environments. In plants, SCN− is formed from the hydrolysis of glucosinolates, often found in Cruciferae (Brassicaceae) by glucosidases (37). In mammals, SCN− is commonly found in saliva, blood, and milk, and it derives from the ingestion of glucosinolates, often derived from broccoli, cauliflower, and other cruciferous vegetables, as well as from cyanide (CN−) detoxification by the ubiquitous enzyme rhodanese (27). Although mammalian SCN− degradation primarily occurs through peroxidation by myeloperoxidase and lactoperoxidase (153), at a microbial level, a number of chemoorganotrophic bacteria are able to degrade SCN− as a source of nitrogen and sulfur. Furthermore, chemolithoautotrophic sulfur bacteria, such as T. thioparus, have been identified that utilize SCN− as their primary energy source (64). More specifically, the enzyme thiocyanate hydrolase (SCNase), which catalyzes the hydrolysis of SCN− to COS and NH3 (Eq. [3]), was initially isolated and identified in T. thioparus strain THI115 (4). SCNase consists of three subunits (α [19 kDa], β [23 kDa], and γ [32 kDa]), which share a high sequence homology to bacterial nitrile hydratases, and it maintains an unusual distorted square pyramidal low-spin Co(III) active site (63). SCNase has also been identified as the primary enzyme for initiating SCN− hydrolysis in the sulfur-oxidizing bacterium Thiohalophilus thiocyanoxidans (9).
In addition to SCNase, COS can also be formed from archaeal CS2 hydrolase, which converts CS2 to H2S and CO2, but it proceeds through intermediate generation of COS (125). CS2 hydrolase, which has been isolated from the hyperthermophilic Archaea Acidianus A1-3 that lives in volcanic solfataras, has a structure that is similar to typical β-CAs, but it does not hydrolyze CO2. Instead, this enzyme has evolved a highly hydrophobic tunnel that serves as a filter by blocking the entrance of CO2 into the active site, which is otherwise identical to that of CA (124). Similarly, in mammalian systems, CS2 can be metabolized to COS by the mixed-function oxidase enzyme system. Liver damage, as well as a measureable decrease in the concentration of cytochrome P450, is observed when rats are treated with CS2 (21, 31, 32). This damage has been attributed to the binding of sulfur species that are released during the hydrolysis of CS2 to COS. Once released, the COS is further metabolized to H2S, most likely by hepatic CA. Alternatively, COS can function as a suicide substrate for cytochrome P450, generating CO2 and sulfur species that react with and inhibit the P450 (31).
Abiotic COS generation
Because much of the COS in the atmosphere derives from open waters, a number of studies have investigated possible mechanisms of COS formation, such as the reaction of carbonyl groups of dissolved organic compounds with thiyl radicals (43). Interestingly, and possibly of more direct biological relevance, is the direct reaction of polysulfides with CO to generate COS (61). Inorganic polysulfides play an important role in the global sulfur cycle and such polysulfides, as well as their organic counterparts, are now understood to be of increasing importance in the biological action of H2S (24, 25, 69, 70, 107). By using inorganic polysulfides and CO, both of which are abundant in aquatic systems, mechanistic investigations revealed that the rate of COS generation had a first-order dependence in both CO and the molar sum of polysulfide species in solution (61). Although further investigations are needed to determine whether the reaction kinetics, pH, and temperature dependence make such mechanisms of COS formation viable under physiological conditions, these observations highlight the interconnected role of COS with CO and polysulfides.
COS detection in mammalian systems
Although the precise mechanisms of COS biosynthesis in eukaryotes remain unknown, two primary pathways for COS genesis have been postulated in investigations of COS in the body: metabolism (or impaired metabolism) of sulfur-containing precursors, and direct generation by cohabitating bacteria. COS has also been detected as a metabolite of different sulfur-containing drugs, supporting that abnormal metabolism of sulfur-containing compounds may, in part, contribute to COS generation. One simple example of such is disulfiram (tetraethylthiuram disulfide), which functions as an acetaldehyde dehydrogenase inhibitor and is commonly used for treatment of chronic alcoholism (57). Reduction of the dithiocarbamate disulfide in disulfuram, followed by partial hydrolysis to form diethylthiocarbamate, may provide a path for COS extrusion, although it is also possible that initial CS2 release generates COS as a metabolic byproduct. In addition, metabolism of the commonly used dithiocarbamate pesticides has also been demonstrated to release COS. Although such systems provide convenient examples of COS in biological contexts, the high concentrations of such sulfur-containing molecules significantly simplify COS detection. By contrast, detection of endogenous COS remains significantly more challenging. These challenges arise primarily from the likely low levels of COS as well as the efficient metabolism of COS by CA to form H2S, which has so far necessitated detection of COS from only gaseous biological samples or from the headspace of samples, where COS can be isolated outside of a CA-rich environment. Despite these challenges, observation of COS in mammalian cell culture, ex vivo tissues, and exhaled breath has provided compelling evidence for the importance of COS in biology and implications for COS involvement in various pathologies.
One strategy for COS detection is to monitor the gas content of the headspace over cell culture or ex vivo tissues. A benefit of this approach is that analysis of the headspace is readily accessible through standard GC-MS techniques or by various spectroscopic techniques (26). In one such example, headspace analysis of porcine coronary artery (PCA) and cardiac muscle tissue in vitro demonstrated COS formation by GC-MS analysis (7). In addition, COS was found to induce arterial dilation. Moreover, stimulation of PCA with acetylcholine and calcium ionophore A23187 resulted in increased COS levels, suggesting that muscarinic acetylcholine receptors (mAChRs), which have already been shown to be involved in the production of CO, NO, and H2S leading to vasorelaxation (13, 44, 55, 91, 94, 158), may also be involved in COS genesis. Although a simple example, these experiments may suggest a promising starting point for future COS investigations.
In addition to the detection of COS in the headspace of tissue culture experiments, COS has also been detected in exhaled breath, providing evidence for the role of COS as a potential gaseous biomarker for various disease states. For example, investigations into the presence of sulfur gases in patients with cystic fibrosis (CF) revealed measurable differences between COS levels in inhaled and exhaled breath (60). As a whole, CF patients had a reduced uptake of atmospheric COS by comparison with healthy patients. Furthermore, CF patients with reduced pulmonary function exhibited greater COS levels in exhaled breath than normal patients, with a strong inverse correlation between COS concentration and all four indices of pulmonary function in CF patients, with no correlation observed in normal patients. Prior research has also demonstrated an inverse correlation between pulmonary function and respiratory bacterial load (99, 114, 115), which, when taken together, provide support for the hypothesis that cohabitating bacteria may play an important role in biological COS generation. Consistent with this hypothesis, recent studies have reported that CA distribution and activity may be altered in CF patients (15, 38, 59), suggesting a lower potential for COS metabolism. One hypothesis, which is consistent with the experimental data, is that impairment of CA function/expression in CF patients may reduce COS metabolism in the lungs, which, when coupled with increased bacterial load and COS production in the respiratory tract, may result in higher COS levels observed in exhaled gas. On the basis of these observations, these studies suggest that exhaled COS may provide a potential non-invasive biomarker for bacterial colonization of the respiratory tract of CF patients.
Furthering the potential role of COS as a biomarker for lung pathologies in exhaled breath, COS has also been investigated as a potential marker of acute rejection (AR) after organ transplant (130). AR after lung allograft is a major risk factor for bronchiolitis obliterans (BO), which is one of the primary causes of death in lung transplant patients. Early detection and diagnosis of AR typically requires routine biopsies, which are invasive and associated with pulmonary complications. In a study investigating the efficacy of non-invasive breath testing for AR monitoring, comparisons between healthy, non-rejection patients and AR patients did not provide significant differences in exhaled ethane, isoprene, acetone, or H2S, all of which are potential organ transplant-related biomarkers. By contrast, exhaled COS levels were demonstrated to provide a biomarker for AR, with elevated COS levels observed for AR patients but not in non-rejection patients. Furthermore, individual patient tracking documented examples of COS levels increasing with worsening AR and decreasing with AR resolution. Although the direct COS origin was not identified, the authors hypothesized that abnormal metabolism of sulfur-containing compounds in AR patients may be responsible for the observed increase in exhaled COS.
In addition to direct lung pathologies, COS levels in exhaled breath have also been characterized in various stages of liver disease (121). When compared with patients with normal liver function, patients with hepatocellular injury (grouped to include: alcoholic cirrhosis, autoimmune cirrhosis, cryptogenic cirrhosis, fulminant hepatitis, hepatitis B, hepatitis C, 1-antitrypsin deficiency, and steatohepatitis) exhibited elevated COS levels in exhaled breath. In addition, COS, but not CS2 or Me2S, was found to correlate with the severity of disease, resulting in increasing COS levels in early-, mid-, and end-stage liver disease, potentially providing a diagnostic tool for early detection. By contrast, patients with bile duct injury diseases (grouped to include: CF, primary biliary cirrhosis, sclerosing cholangitis, and biliary obstruction) exhibited reduced COS levels in exhaled breath. These human studies are consistent with previous observations in isolated rat hepatocytes and liver microsomes, which have been observed to generate COS, possibly from CS2 metabolism or metabolism of other sulfur-containing compounds (23). These data are also consistent with previous experiments where COS has been observed in exhaled breath of rats exposed to CS2, suggesting that the observed COS may be due to metabolism of CS2 or, by analogy, the incomplete metabolism of other sulfur-containing compounds (32).
The examples mentioned earlier not only provide key demonstrations of COS generation from metabolic abnormalities or bacterial colonization associated with disease but also provide compelling evidence for the necessity of further exploration into potential metabolic pathways of COS in biology. Although mammalian COS production from natural sources remains poorly understood, one possible route for COS synthesis is SCN− hydrolysis by SCNase, although the presence of this enzyme has yet to be reported in mammals. Given that COS has been detected in biological samples, particularly in non-diseased tissues, elucidation of possible enzymatic pathways for COS production remains an important area of investigation. Importantly, these studies additionally suggest a key role of bacterial colonization in COS generation in mammalian systems. In the cases of exhaled breath analysis, a distinct interplay between the host, mammalian tissues, and bacterial colonization not only suggests a complex landscape in COS generation but also may provide access to a convenient method of detection for a variety of disease states.
COS Chemistry
The most simple reactions of COS, including hydrolysis, oxidation, reduction, and dissociation, have been known for many years and are the main subject of a thorough COS review published in 1957 (41). Since then, COS chemistry has expanded to address environmental concerns associated with COS contaminants in industrial settings, and catalysts for the low-temperature rapid hydrolysis of COS have been developed. Although the most basic chemical reactions are well researched, more complex and biologically relevant COS-related reactions remain less understood, even though contemporary investigations suggest important biological roles for COS stemming from initial thermophilic origin of life chemistry.
Simple reactions of COS
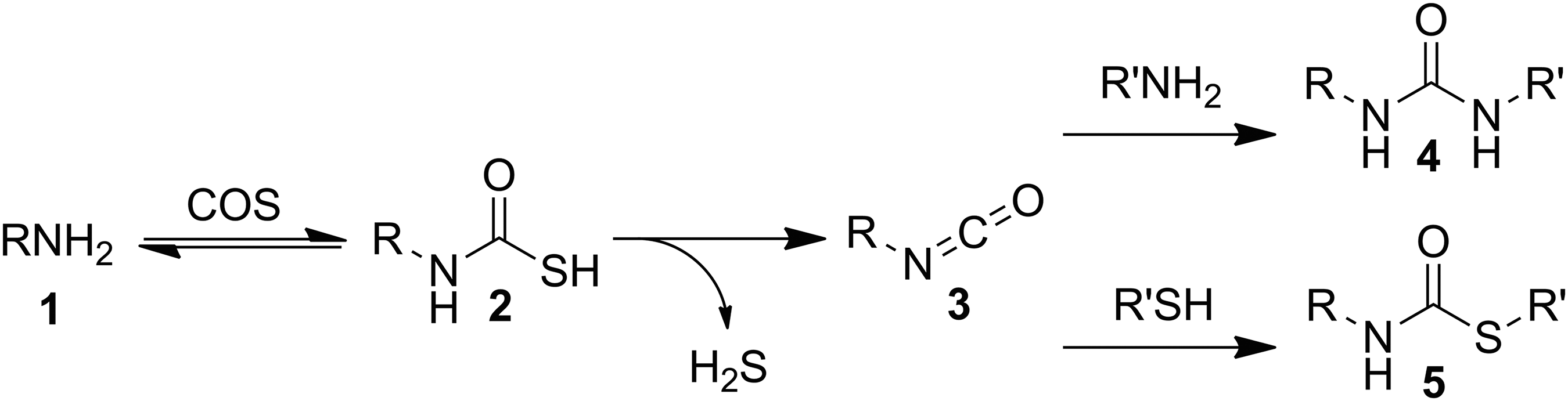
Shortly after the initial synthesis of COS, early reactivity studies demonstrated that COS reacts with primary amines (Fig. 3:
Implications in origins of life and prebiotic bond formation
In addition to the role of COS in laboratory-scale and industrial syntheses, contemporary investigations have focused on the potential role of COS in nascent bond-forming reactions under prebiotic conditions. Perhaps unsurprisingly, the volcanic and geothermal generation of COS, as well as its bond-forming potential with simple nucleophiles, appears consistent with thermophilic origin of life requirements. Much of this work stems from studies by Hirschmann and co-workers in 1971 that focused on peptide formation from 2,5-thiazolidinediones, in which a footnote comments that traces of dipeptides were formed from phenylalanine thiocarbamate—a known reaction product of amines with COS (34).
More than 30 years later, more detailed investigations into origin of life peptide synthesis were performed by mimicking volcanic or hydrothermal environments. Using prebiotic building blocks such as CO, nickel and iron sulfides [(Ni,Fe)S], and a reducing atmosphere of H2S or CH3SH at elevated temperatures, amino acids
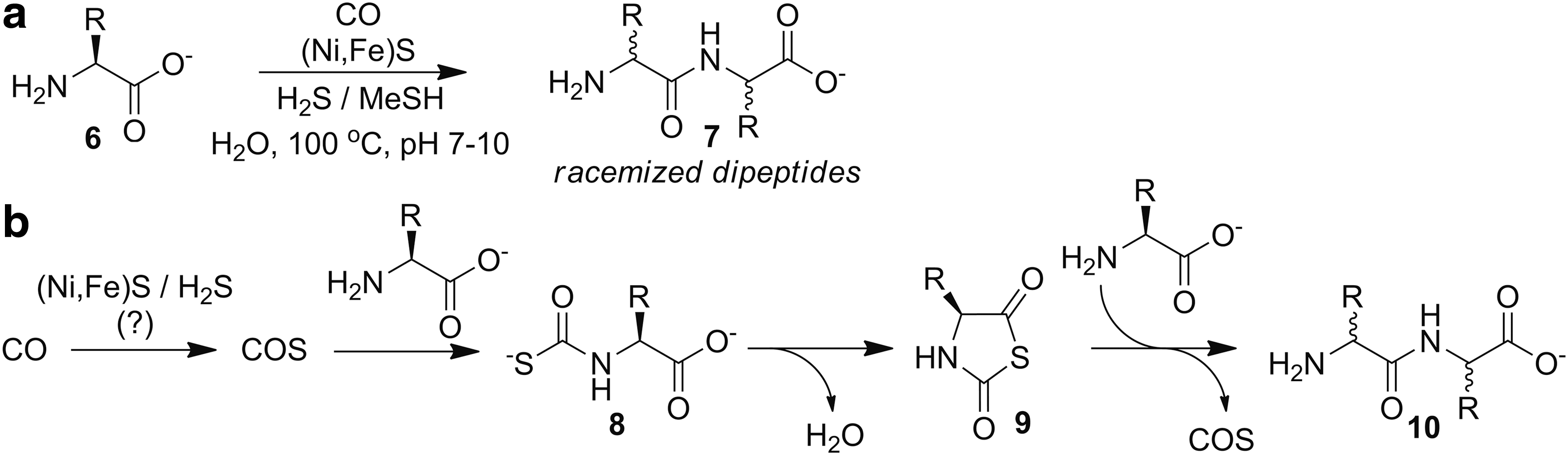
Furthering investigations into the role of COS in primordial amino acid chemistry, an elegant study by Ghadiri and co-workers demonstrated that COS can facilitate the direct formation of small peptides from amino acids in water under mild conditions in the absence of metal sulfides (Fig. 5) (75). Remarkably, even the simplest conditions, such as addition of excess COS gas to an aqueous buffered solution of phenylalanine, resulted in 7% dipeptide formation after 2 days at 25°C. Further investigations revealed quantitative formation of thiocarbamate
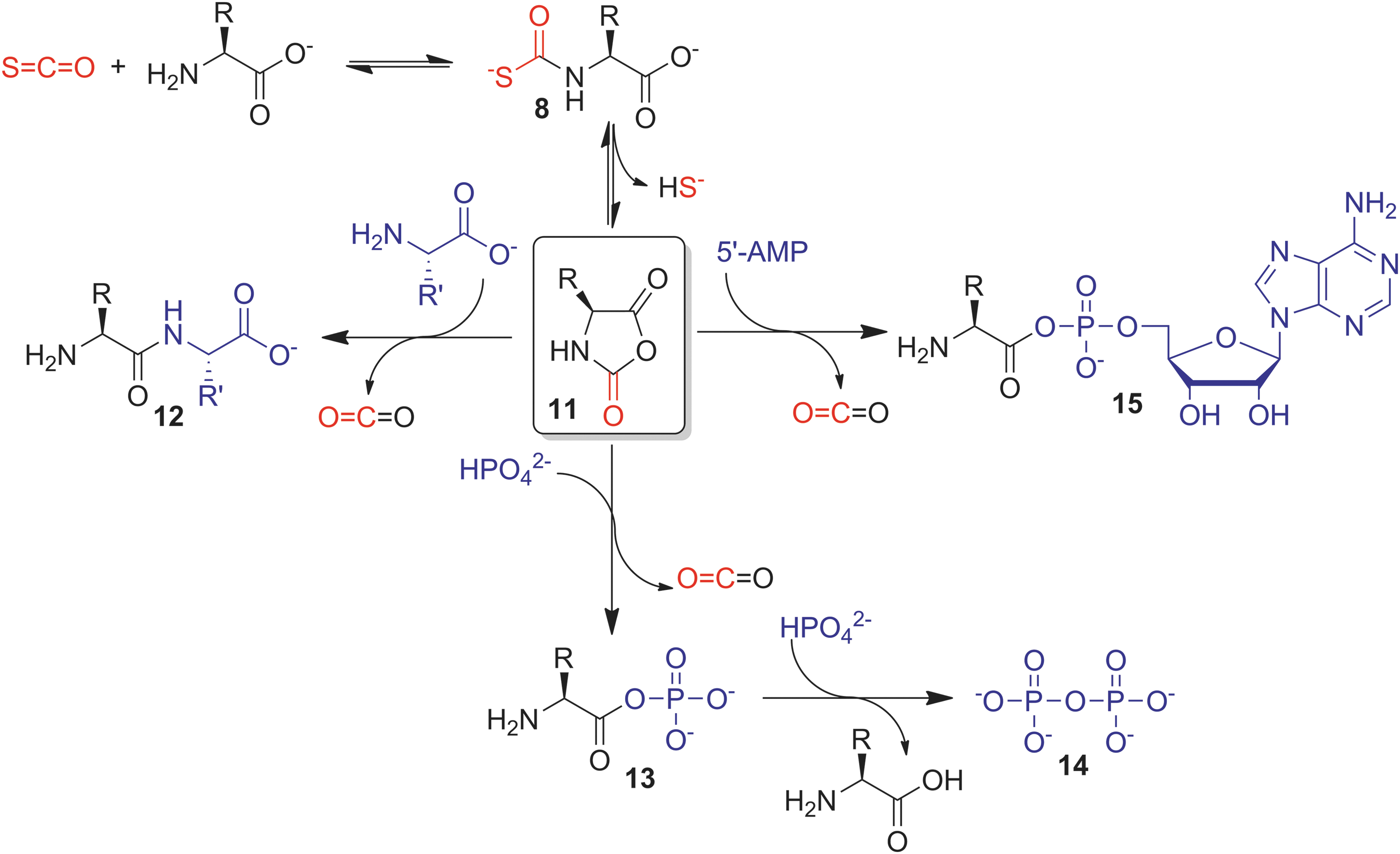
In addition to simple peptide-forming reactions, the activation of amino acids by COS has been demonstrated to be a more general pathway to biologically relevant peptide functionalization. For example, under mild aqueous conditions, COS facilitates the formation of aminoacyl phosphates
Although the atmospheric levels of COS are unlikely to generate suitable concentrations of the required thiocarbamate intermediates to facilitate efficient peptide coupling under global prebiotic conditions, the higher temperatures and COS levels near geothermal locales of COS generation could likely facilitate access to the reaction manifolds that are associated with these important prebiotic bond-forming reactions. More importantly, these studies set the stage for establishing the potential role of COS in biologically relevant reactions and intermediate generation, paving the way for future applications of COS chemistry.
Emerging Tools for COS Investigations
Both chemical and biological tools are needed to expand our understanding of the potential roles of COS in biology. Because direct knockout, overexpression, and blockage of the enzymes that are associated with production of the currently identified gasotransmitters have proved essential in studying the chemical biology of these gases, the definitive identification of COS-producing enzymes or pathways will be equally important. Before such information, downregulation of biological COS will remain a significant challenge. In the interim, one potential strategy to increase COS bioavailability is to shunt pathways that are associated with COS metabolism. For example, because of the wide distribution of CA and its high activity toward COS hydrolysis, CA inhibitors and/or enzymatic knockout could potentially be used to increase COS accumulation. Inhibition of CA by small molecules is a well-researched field (102, 132 –136), and a variety of methods are available for strong inhibition of a variety of CA isoforms. Unfortunately, such inhibition studies would also alter the normal CO2/HCO3 − equilibrium, and thus normal buffering capacities and cellular pH levels, likely leading to complicating effects. In addition, even small changes in pH would also alter the distribution of H2S and HS−, likely leading to confounding results resulting from further entanglement of COS/H2S/HS− chemical biology.
Before insights gleaned from enzymatic regulation, the development of small-molecule chemical tools for COS research may offer an attractive initial platform for expanding our understanding of COS in biology. Chemical tools for detection and delivery of the canonical gasotransmitters NO, CO, and H2S have been invaluable for investigating the multifaceted roles of these important biological molecules (18, 20, 40, 48, 56, 62, 71, 72, 77, 79, 85, 103, 108, 126, 138, 144, 145, 159), suggesting that similar constructs may find utility for COS investigations. Reaction-based fluorescent probe development for COS is likely to remain a significant challenge based on the inherent reactivity of COS. Because COS is a weaker electrophile than CO2, strategies to intercept COS by nucleophilic trapping are likely to be plagued by unwanted side reactivity, leading to significant selectivity challenges. By contrast to reaction-based detection motifs, small molecules that release COS may offer a more attractive first line of tools for investigating COS in different biological contexts.
General strategies to develop COS donors
Because of the structural similarities between COS and CO2, many decarboxylation reactions can be engineered to release COS rather than CO2 by simple replacement of an oxygen atom with a sulfur atom in the parent scaffold. This basic design concept enables structurally diverse COS donors that provide access to both triggered-/active-release donors, such as those that respond to specific biological or biorthogonal stimuli, and slow-/passive-release donors, including those activated by hydrolysis or a reaction with ubiquitous cellular enzymes and nucleophiles. In addition, easily accessible control compounds are available through synthesis of analogous carbamate compounds, which release CO2 rather than COS. Access to such control compounds is instrumental in differentiating the biological effects of the donor scaffolds themselves from the released COS. Recognizing the potential powerful utility of engineered COS release, our lab was the first to harness such motifs to develop COS-releasing small molecules (127). Since this initial report, we have been delighted that other researchers are using related strategies to broaden the palette of COS-releasing motifs that are available for future biological investigations.
The key breakthrough in our initial design was recognizing that self-immolative benzyl carbamates (Fig. 6a) (17, 73), often used as delivery platforms for prodrugs, fluorophores, and other small molecules (2, 12, 117, 122), could be modified to release COS rather than CO2 by exchanging the canonical carbamate linker with a thiocarbamate (Fig. 6b). In a proof-of-concept demonstration of this approach, we established the utility of on-demand COS extrusion as a strategy to access both analyte replacement fluorescent probes and triggered COS/H2S donors (127). The early motivation of this work was to address a major challenge in reaction-based probes for small-molecule analytes, especially RSONS. Activation of these reporter scaffolds results in analyte consumption, thus perturbing homeostasis. Exploitation of the self-immolative decomposition of thiocarbamates to release COS enabled the generation of the first examples of analyte replacement fluorescent probes, which react with (and consume) H2S to produce a turn-on fluorescent response, concomitant with the release one equivalent of caged H2S in the form of COS, thus providing progress toward analyte homeostasis in reaction-based detection systems (Fig. 6c). By using an H2S-reactive azide trigger, which is reduced to an amine after reduction by H2S (50), we established that the subsequent self-immolative cascade reaction extrudes COS, which is quickly hydrolyzed to H2S by CA. Importantly, this donor motif was found to be stable in whole mouse blood before trigger activation, thus highlighting the biological stability of these platforms. Control experiments on thiocarbamate motifs lacking a latent fluorophore demonstrated that triggered reductive cleavage by tris(2-carboxyethyl)phosphine (TCEP) in mouse blood resulted in COS donation with a nearly 50% efficiency and conversion to H2S by CA. In addition, addition of the CA inhibitor AAA abrogated H2S production in vitro, confirming that H2S release is a result of CA-mediated COS hydrolysis. Although this initial report provided an important contribution toward H2S detection technology, the broader impact is providing a viable and highly tunable COS donating strategy for accessing chemical tools for expanding our understanding of the chemical biology of COS.
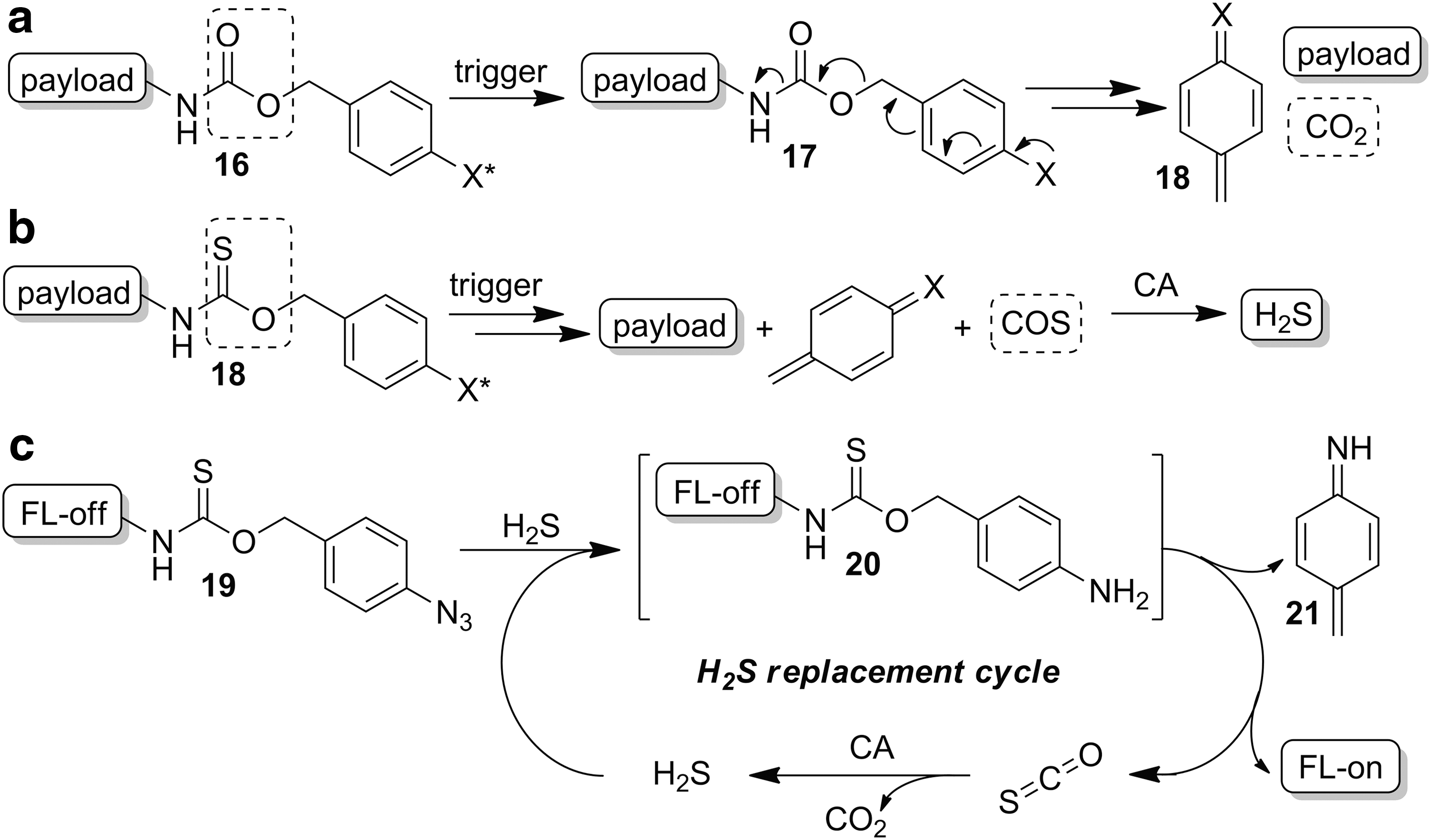
Triggered/active release of COS
Furthering the strategies outlined earlier, simple changes to the general self-immolative thiocarbamate scaffold can provide access to new donor motifs with more specific functions. Notably, the incorporation of protecting groups that selectively respond to specific stimuli and result in on-demand COS release may provide access to highly targeted COS donors with utility in investigating the chemical biology of COS, as well as for site-selective COS/H2S donation. Such triggerable donors can be expected to be useful in therapeutic applications in which COS delivery is targeted to a specific location as well as for studying COS delivery with a high level of temporal control.
Reactive oxygen species triggered COS donors
A powerful application of such responsive donors is the judicious choice of triggering analytes that are associated with contexts in which H2S can exert beneficial action. For example, because H2S has been demonstrated to provide protection against increased oxidative stress, a system in which COS release is triggered by reactive oxygen species (ROS) should not only provide access to actively triggered COS donors but also provide a platform with high pharmacological potential. Using an ROS-cleavable aryl boronate as the protecting group (35, 87), we developed a class of COS donors that respond to increased ROS levels (Fig. 7a) (160). Highlighting the responsive nature of this design platform, COS is released from PeroxyTCM-1 in a dose-dependent manner on the addition of H2O2 as well as other ROS and is quickly converted to H2S by CA.
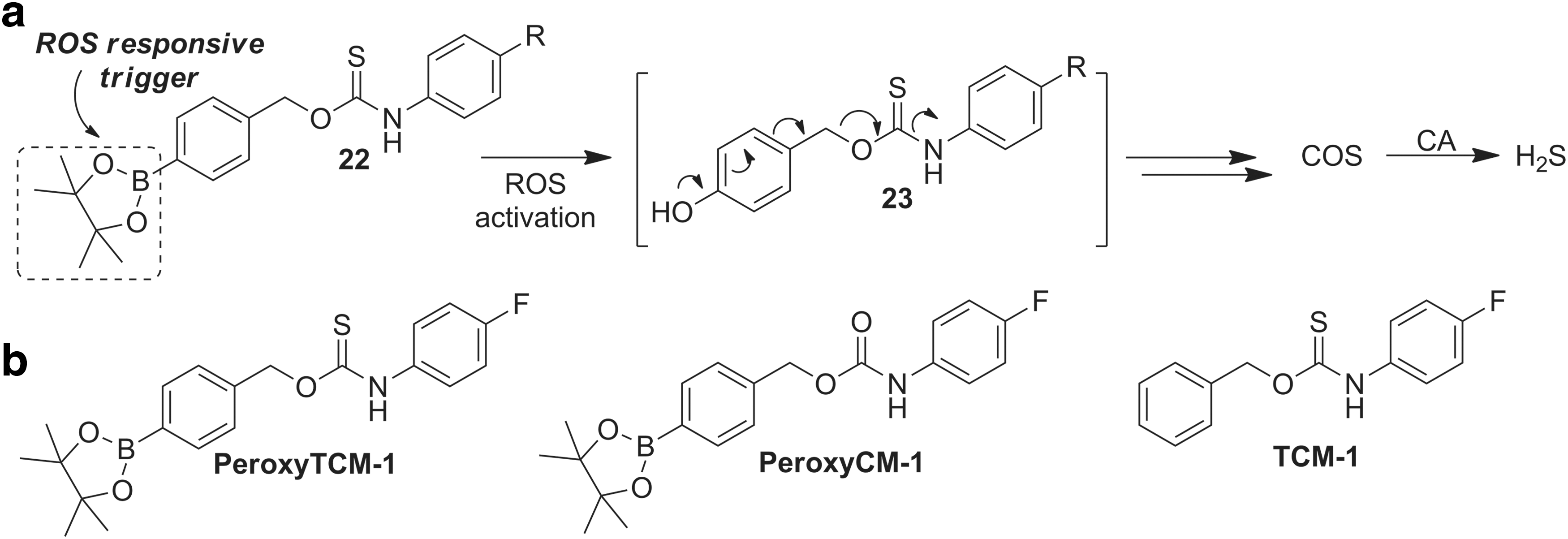
Supporting in vitro investigations, stimulation of endogenous ROS production in Raw 264.7 cells by addition of phorbol 12-myristate 13-acetate (PMA) resulted in COS/H2S release from PeroxyTCM-1, as evidenced by an increase in fluorescence when imaged with the H2S-responsive probe HSN2 (93). Providing early insights into the potential of such donors to impart cellular protection under conditions of increased oxidative stress, PeroxyTCM-1 exhibited a dose-dependent increase in cell viability in HeLa cells treated with exogenous H2O2, which is consistent with ROS protection. Importantly, the use of carbamate control compound PeroxyCM-1 or triggerless TCM-1 (Fig. 7b) did not recapitulate the cytoprotective effects, suggesting that the observed cytoprotection was, indeed, due to the COS/H2S release rather than from the organic scaffold or reaction byproducts. In addition, in vitro investigations also demonstrated that H2O2 can react directly with COS to generate H2S, thus decreasing the need for CA and also highlighting the importance of COS as a potential ROS scavenger.
Bio-orthogonal COS release
COS donors triggered through bio-orthogonal methods have also been reported. One advantage of this approach is that the donor constructs are stable until exposure to a benign external stimulus and release COS without the need for a detrimental cellular trigger. In addition, bio-orthogonal methods allow for the potential of high spatial and temporal resolution, as evidenced by the utility of such strategies for targeted drug delivery (45, 142). For example, incorporation of a COS-releasing thiocarbamate into the trans-cyclooctene reaction partner
Continuous/passive release of COS
A complementary approach to access caged COS donors is to develop continuous release COS donors, which are activated by ubiquitous cellular nucleophiles or enzymes. Such donors would result in continuous, rather than triggered, COS release, thus increasing basal COS levels in an otherwise normal physiological environment. Similar to well-known hydrolytically activated donors for NO and H2S (1, 65), these compounds are likely to contribute to an important class of tools for investigating COS chemical biology.
Nucleophile activation
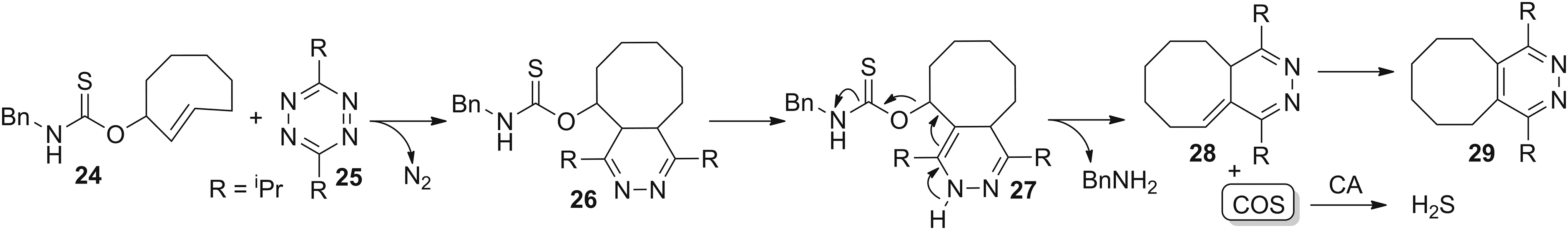
Matson and co-workers recently investigated N-thiocarboxyanhydrides (NTAs, Fig. 9a) as COS-releasing molecules (109). These electrophiles release COS after reaction with nucleophiles, and they are analogous to the thiocarboxyanhydrides proposed by Hirschmann in studies of peptide couplings (
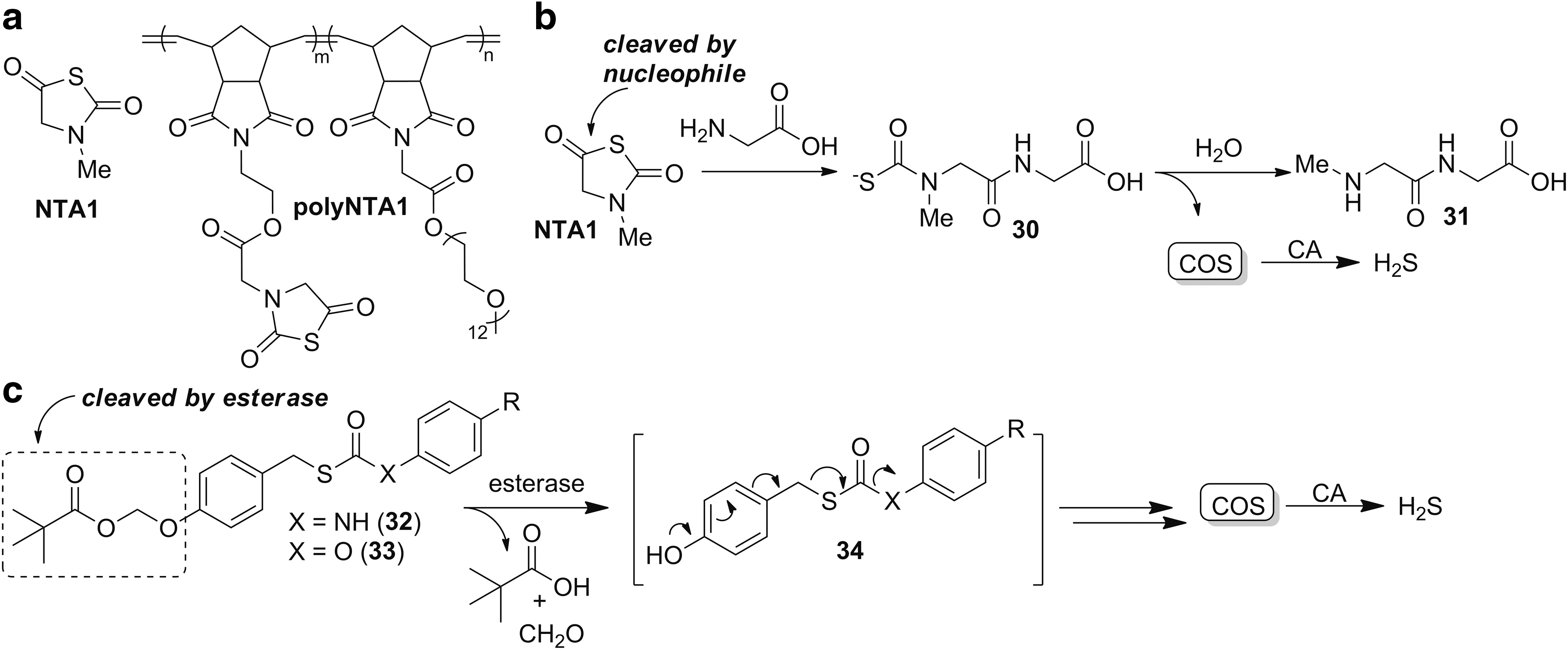
Ubiquitous enzyme activation
Complementing COS donors that function by a reaction with bioavailable nucleophiles, Chakrapani and co-workers recently reported thiocarbamate
Outstanding questions
Although the recent introduction of a variety of COS donor compounds provides simple ways to introduce exogenous COS into biological samples, the direct CA-mediated metabolism of COS to H2S represents a significant challenge in differentiating the biological actions of COS from those associated with H2S. In addition, because COS metabolism by CA generates CO2/HCO3 − in addition to H2S, it is important to consider the total amount of COS metabolized in a system to ensure buffering capacities are not exceeded by these otherwise innocuous products. New insights into the biological roles of COS will likely require thoughtful and careful applications of available H2S and COS donors that are used in concert to investigate specific biomolecular questions. Whether available COS donors function merely as clever sources of biological H2S, or whether the released COS imparts different outcomes in biological contexts remains to be determined. If realized, COS-releasing molecules that provide outcomes distinct from those attributed to available H2S donors will likely play a significant role in assessing and advancing not only the role of COS as a potential gasotransmitter but also its role in potential therapeutic applications that are associated with human health.
Conclusions and Outlook
Key challenges remain in further elucidating the chemical biology of COS, but our current, although limited, understanding of the biological production and consumption of COS suggests that it may play diverse roles. Could COS be poised to be next on the list of established gasotransmitters? This distinction will first require identification of enzymatic COS production in higher organisms and evidence that COS and H2S function independently. It is also possible that COS functions primarily as a source of “caged” H2S that is liberated by CA metabolism. Such a pathway is intriguing because it would provide a source of reduced sulfur that is not ionizable through acid-base equilibria at physiological pH, and that is less susceptible to ambient or enzymatic oxidation through direct action of oxidases or sulfur:quinone oxidoreductase (SQR) (97), thus bypassing interaction with the sulfane-sulfur pool (89). In addition, the neutral state of COS could enable distribution to locales that would otherwise be challenging for H2S/HS− alone. Finally, there is compelling evidence that COS stems from bacterial generation, especially in certain disease pathologies. In these cases, COS could provide a transport mechanism from pathogen to host. Even if eukaryotic COS synthesis is not a major source of endogenous COS, a thorough understanding of the role that COS plays in different diseases will likely be beneficial in early detection and treatment. When viewed more broadly, the absence of well-established metabolic pathways for COS formation in eukaryotic systems paired with the presence of COS-producing pathways from simple and abundant sulfur sources by a variety of bacteria may paint a broader, yet fundamentally underexplored picture of COS functions in sulfur biology and transport.
Footnotes
Acknowledgments
Aspects of work in the authors' lab that focused on H2S/COS delivery are supported, in part, by the NIH (R01GM113030 to M.D.P.), NSF GRFP (DGE-1309047 to A.K.S.), Sloan Foundation (M.D.P.), and Dreyfus Foundation (M.D.P.).
