Abstract
Significance:
Hydrogen sulfide (H2S) has been recognized as the third gaseous transmitter alongside nitric oxide and carbon monoxide. In the past decade, numerous studies have demonstrated an active role of H2S in the context of cancer biology.
Recent Advances:
The three H2S-producing enzymes, namely cystathionine γ-lyase (CSE), cystathionine β-synthase (CBS), and 3-mercaptopyruvate sulfurtransferase (3MST), have been found to be highly expressed in numerous types of cancer. Moreover, inhibition of CBS has shown anti-tumor activity, particularly in colon cancer, ovarian cancer, and breast cancer, whereas the consequence of CSE or 3MST inhibition remains largely unexplored in cancer cells. Intriguingly, H2S donation at high amounts or a long time duration has also been observed to induce cancer cell apoptosis in vitro and in vivo while sparing noncancerous fibroblast cells. Therefore, a bell-shaped model has been proposed to explain the role of H2S in cancer development. Specifically, endogenous H2S or a relatively low level of exogenous H2S may exhibit a pro-cancer effect, whereas exposure to H2S at a higher amount or for a long period may lead to cancer cell death. This indicates that inhibition of H2S biosynthesis and H2S supplementation serve as two distinct ways for cancer treatment. This paradoxical role of H2S has stimulated the enthusiasm for the development of novel CBS inhibitors, H2S donors, and H2S-releasing hybrids.
Critical Issues:
A clear relationship between H2S level and cancer progression remains lacking. The possibility that the altered levels of these byproducts have influenced the cell viability of cancer cells has not been excluded in previous studies when modulating H2S producing enzymes.
Future Directions:
The consequence of CSE or 3MST inhibition in cancer cells need to be examined in the future. Better portrayal of the crosstalk among these gaseous transmitters may not only lead to an in-depth understanding of cancer progression but also shed light on novel strategies for cancer therapy.
Table of Contents
I. Introduction
Hydrogen sulfide (H2S) is a colorless gas characterized with a strong rotten egg smell under standard conditions of temperature and pressure. It has been more than 300 years since the first description of H2S as a poisonous molecule (18). For instance, it has been recorded that heavy exposure to H2S (>500 ppm) causes unconsciousness and death in humans (238). Usually, the intoxication of H2S is ascribed to its strong suppressive effect on several critical enzymes in humans such as cytochrome c oxidase (238), Na+/K+ ATPase (238), carbonic anhydrase (205), and monoamine oxidase (299).
Nonetheless, the physiological importance of H2S is suggested by the fact that mammalian cells are able to actively produce this gaseous molecule (71, 240, 264). This was first demonstrated by Abe and Kimura in 1996 (1) showing that H2S is an endogenous modulator in the central nervous system. Subsequently, H2S has been revealed to participate in the regulation of various physiological and pathological conditions within mammalian systems, including central nervous (1), cardiovascular (89), renal (284), reproductive (293), respiratory (83), and digestive systems (64). It is now well recognized as a third endogenous gaso-transmitter along with nitric oxide (NO) and carbon monoxide (CO).
Intriguingly, very recent evidence has accumulated to show that H2S has a previously unrecognized role in cancer biology. In this review, the roles of H2S in cancer development and the underlying mechanisms will be surveyed. Moreover, our review will also discuss the progress and the therapeutic potential of H2S-based molecules for cancer therapy.
II. Biochemistry of H2S
A. Physical and chemical properties of H2S
Under ambient temperature and pressure, H2S is a colorless and flammable gas with a strong rotten egg smell. Acute exposure to high amounts of H2S (>500 ppm) can lead to human death (238). H2S is readily dissolved in water with a solubility of about 80 mM at 37°C (121). In aqueous solutions, H2S is a weak acid and quickly reaches the equilibrium of H2S/HS−/S2− species. It is estimated that 14% of the sulfide will be H2S and nearly 86% will be HS− in plasma where the pH value is around 7.4 (311). Nevertheless, changes of pH value can readily alter the equilibrium of H2S/HS−/S2− species. Unless elsewhere specified, in this review, we use the term H2S to reflect the sum of the species H2S/HS−/S2− and intermediates formed from these, present at physiological pH (306).
Due to the volatile nature of H2S, it can be easily predicted that the equilibrium will constantly shift to the left, which results in the formation of H2S evading from the solution (H2S ↔ H+ + HS−↔ H+ + S2−). In fact, there are published reports showing that half of H2S can escape from medium in 5 min in tissue culture wells and in an even shorter time in a bubbled tissue bath (62, 101, 208). This may have partially contributed to the miraculous variations on the reported concentration of H2S in tissues, plasma, and certain experiments (84, 249, 311). In addition, it is notable to point out that H2S, as a low-molecular-weight lipophilic gaseous molecule, is freely diffusible across cell membranes without the need of a specific transporter (109, 176).
Sulfur is a multifaceted atom with multiple oxidation states such as −2 in H2S, 0 in S8, +2 in SO, +4 in SO3 2− and +6 in SO4 2−. As clearly seen from the earlier described fact that −2 in H2S is the lowest oxidation state of sulfur, therefore H2S is a reductant and can only be oxidized. As a result, H2S (and/or intermediates derived from it) can readily modify biological pathways and elicit biological effects via the following reactions. The reactions between H2S and the reactive oxygen species (ROS) or reactive nitrogen species (RNS) species have been extensively studied in solutions (306 –308). Nevertheless, Olson et al. (211) recently showed that the reaction of H2S with H2O2 is way too slow to elicit any biological significance in mammalian systems. Therefore, it is unlikely that H2S serves as a direct ROS scavenger in physiological conditions (43). Besides ROS/RNS species, another primary target of H2S in mammalian systems would be biological irons, namely the metal centers on proteins (199).
In recent years, numerous studies revealed a process called persulfidation as the third H2S-mediated interaction by which H2S can react with protein thiol groups and forms protein persulfides that may induce functional changes of target proteins (194). However, one should bear in mind here that H2S reacts with R–SOH rather than free thiols since conversion of R–SH to R–SSH is associated with oxidation rather than chemical reduction of sulfur. Similar with other modifications of protein thiols such as S-nitrosation, S-sulfenylation, and S-glutathionylation, H2S-mediated persulfidation has been found in several proteins and demonstrated to account for the plethora of H2S-mediated signaling transduction (195, 225). Nevertheless, it should be noted that several critical issues are still awaiting resolution in the field (41, 78). For instance, (i) reliable and proper quantification methods are to be developed; (ii) the mechanisms underlying such modification needs to be thoroughly studied; and (iii) it is required to understand the actual impact of persulfidation on cellular functions.
Interestingly, a fourth H2S-mediated interaction was recently reported to be the chemical reduction of protein disulfide bonds (220, 289), whereas physiological relevance of the reaction still awaits further demonstration. For a thorough description of the chemical basis of the interactions mentioned earlier, it is advised to refer a recent review from Filipovic et al. on the chemical foundation of H2S biology (79).
B. Endogenous production of H2S in mammals
The first description of H2S production in life can be retrieved back to 1895 in bacteria (215). One hundred years later, the detection of sulfide was first reported in brain tissues (93) and later in other mammalian systems (107, 124). Mammalian cells rely on both enzymatic and non-enzymatic pathways to generate H2S (Fig. 1).
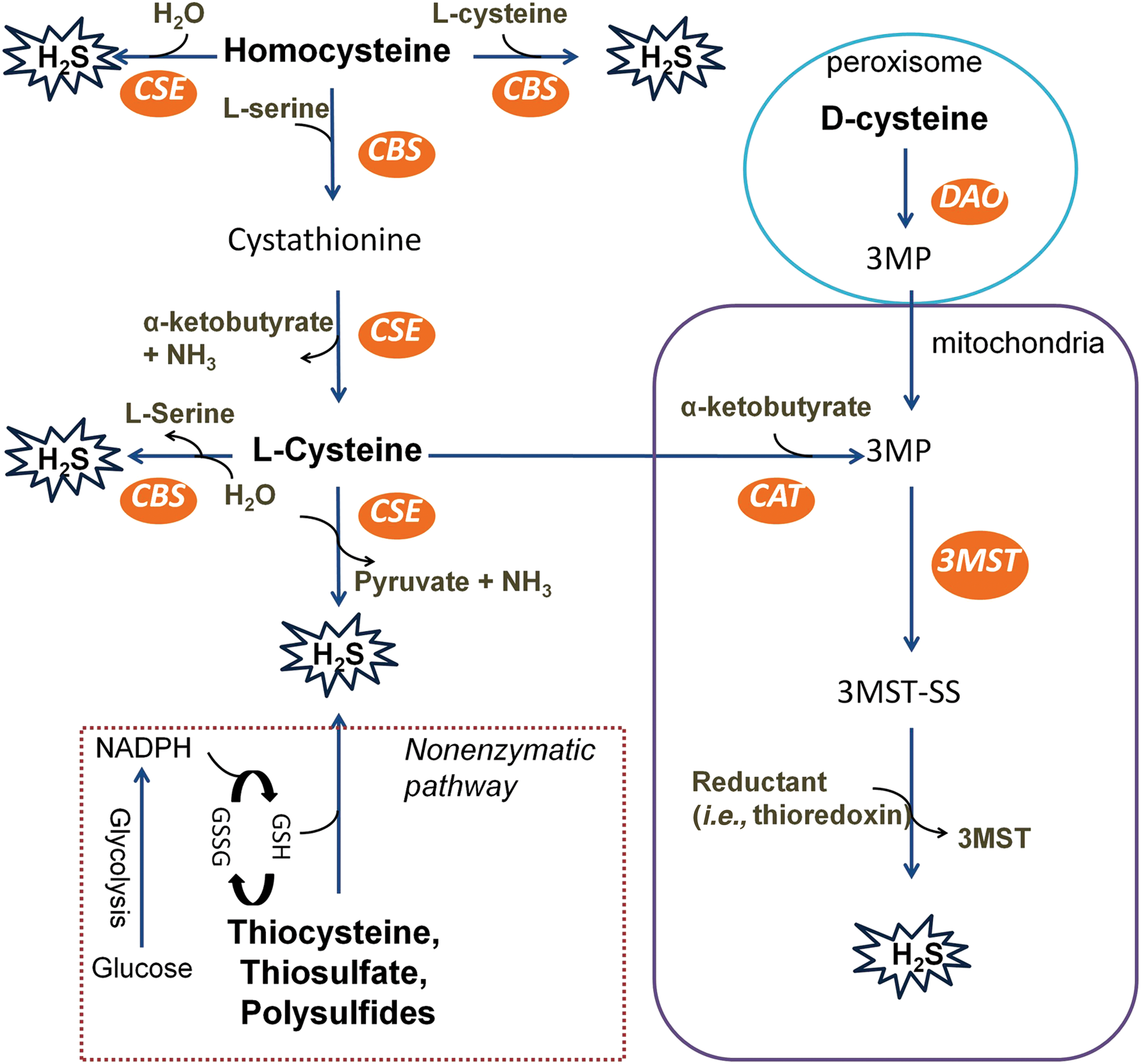
By using an enzymatic system, H2S production can be regulated tightly and precisely in mammalian cells, in which H2S may serve as an endogenous signaling molecule. Among the enzymes, cystathionine β-synthase (CBS) and cystathionine γ-lyase (CSE) are pyridoxal 5′-phosphate (PLP)-dependent enzymes and are located in the cytosol, whereas PLP-independent 3-mercaptopyruvate sulfurtransferase (3MST) mainly resides and generates H2S within mitochondria. Nevertheless, CBS and CSE can translocate into mitochondria when the activity of 3MST is significantly suppressed in oxidative conditions such as hypoxia (82, 276). CBS and CSE also play a role in trans-sulfuration and reverse trans-sulfuration pathways that are responsible for homocysteine metabolism.
Moreover, it is worth pointing out that the mechanisms underlying the regulation of these enzymes such as the expression and activities remain largely unsettled regardless of physiological or pathophysiological conditions. Interestingly, Olson et al. (212) recently showed that H2S may be generated from thioredoxin by catalase using nicotinamide adenine dinucleotide phosphate (NADPH) in hypoxia in vitro but this has yet to be demonstrated in vivo or in mammalian cells. This could be very relevant to solid tumors where O2 is low and NADPH is elevated (23).
1. Enzymatic production of H2S
a. Cystathionine-β-synthase
CBS (EC 4.2.1.22) was first identified in 1969 by Braunstein et al. (28) and was later found to be able to produce H2S mainly through catalyzing
The activity of CBS is found to be mainly regulated by three sites on the protein, including a heme group at the N-terminus, an S-adenosyl-
CBS is regarded as the predominant H2S synthase in the central nervous system, although its expression has also been observed in various types of other organs such as kidney, liver, lymphocytes, uterus, placenta, and pancreas islets (1, 125, 224). Recently, this notion seems to be challenged by a report that brain homogenates of CBS-knockout mice produce a similar level of H2S to that obtained from wide-type mice (253). However, we propose that the conflict may result from compensatory expression of 3MST in the brain when CBS is knocked out, which may help maintain the endogenous level of H2S for neuronal cell survival. As a result, CBS knockout mice-derived brain lysates produce a comparable level of H2S with that obtained from wide-type mice. Therefore, the data perhaps do not necessarily contradict with the existing notion that CBS is predominant for H2S production in the brain, particularly in physiological conditions. In this case, more experiments are needed to test the hypothesis whether such possible compensatory mechanisms among H2S-producing enzymes cause the contradiction.
b. Cystathionine-γ-lyase
CSE (EC 4.4.1.1), similar to CBS, utilizes homocysteine as a substrate to generate H2S along with α-ketobutyrate and ammonia. Alternatively, CSE can also catalyze
The activity of CSE is known to be influenced by intracellular Ca2+ concentration (182, 343). Specifically, low levels of intracellular Ca2+ constitutively induce the production of H2S by CSE whereas CSE activity is suppressed on the rise of intracellular Ca2+ by cell stimulation even in the presence of PLP. Further study has revealed that the regulation is independent of calmodulin as neither calmodulin nor its selective inhibitor W-7 alters the level of H2S derived from purified CSE from the liver (182). Nevertheless, the precise mechanism underlying Ca2+-mediated regulation of CSE activity remains to be defined in future.
In comparison to CBS, CSE distributes more widely in mammalian tissues. It is abundantly expressed in the cardiovascular system and respiratory system (107, 344). CSE may also be the dominant H2S-producing enzyme in tissues such as liver, kidney, uterus, and pancreatic islets (135, 327, 344). A detectable level of CSE mRNA has been reported in the brain (71, 89); however, it seems that the presence of CSE may not be related to H2S production in the organ because CSE inhibitors do not alter the production of H2S in the brain (1).
c. 3-Mercaptopyruvate sulfurtransferase
3MST is the most recently discovered H2S-producing enzyme (253). With this enzyme system (Fig. 1),
Distinct from CBS and CSE, the activity of 3MST appears to be regulated intrinsically by its redox state rather than interactions with other factors. Based on its crystal structure, three redox-sensitive cysteines (Cys154, Cys247, Cys263) have been identified on its catalytic site (196). In line with this, oxidative stress significantly suppresses the activity of 3MST and, therefore, H2S production probably by oxidation of these protein thiols (36, 133). Moreover, modulation of CAT or DAO activity can also apparently affect the generation of H2S by 3MST. For instance, extensive evidence has shown that aspartate, an inhibitor of CAT, largely inhibits H2S biosynthesis and is commonly used as a pharmacological tool to study the function of 3MST-derived H2S (4, 252, 253) whereas one should bear in mind that aspartate is not a selective 3MST inhibitor. Therefore, special caution should be given when studying 3MST-mediated cellular H2S production due to the fact that multiple enzymes are participating in the process, particularly in organs such as the brain and kidney. In addition, increased intracellular Ca2+ has been shown to suppress the activity of CAT and the subsequent H2S synthesis independently of calmodulin (134, 181). However, the precise molecular mechanism underlying this regulation is unknown.
3MST, similar to CBS and CSE, is expressed in many tissues with particularly high activity in cardiac cells, pericentral hepatocytes in the liver, and the proximal tubular epithelium of the kidney (252, 253, 312). Recent evidence shows that 3MST may be crucial for H2S generation in the brain as brain homogenates of CBS-knockout mice produce a similar level of H2S to that of wide-type mice (253). Within mammalian cells, 3MST is mainly located in mitochondria, although a detectable level of 3MST has also been reported in the cytoplasm (134). Nevertheless, it is believed that the occurrence of H2S generation by 3MST is mainly in mitochondria, because the concentration of
2. Non-enzymatic production of H2S
A portion of endogenous H2S is derived from sulfane sulfur via non-enzymatic chemical reduction. In general, the process requires reducing equivalents such as NADPH and nicotinamide adenine dinucleotide (NADH), which are supplied by oxidation of glucose via glycolysis or from phosphogluconate via NADPH oxidase (244). In the presence of such equivalents, reactive sulfur species in persulfides, thiosulfate, and polysulfides can be reduced into H2S and other metabolites (210). Essentially, all the components of this non-enzymatic route are available in mammals including reducible sulfur, suggesting the necessity of this pathway in mammalian systems. In accordance with this, hyperglycemia is demonstrated to promote H2S generation by enhancing this pathway (296).
C. H2S catabolism
Along with the production of endogenous H2S, it has to be disposed very quickly as the accumulation of H2S may result in organ toxicity. In comparison to the biosynthesis of H2S, the catabolism of this gaseous transmitter is not well studied and most evidence was obtained by using exogenous H2S. Therefore, further validation is needed in experimental settings of physiological conditions. Herein, we will discuss three catabolic routes of H2S in mammalian systems, namely oxidation, methylation, and expiration. It is worth mentioning that hemoglobin also interacts with H2S, resulting in the formation of bound sulfur in plasma as sulfhemoglobin (45); however, this may serve as an internal storage of sulfide rather than its catabolism and is, therefore, not included here in this review.
1. Oxidation
The vast majority of H2S is disposed via the kidney in the form of sulfate. In the process of sulfate formation, oxidized H2S combines with another molecule of H2S and forms one molecule of thiosulfate in mitochondria. This step does not require any enzyme and has been demonstrated within isolated rat kidneys and livers (13, 165). H2S can also be oxidized by sulfide quinine oxidoreductase (SQOR) in mitochondria to generate persulfide, which is then catalyzed into thiosulfate by rhodanese (115, 116). Recently, Bostelar et al. (26) discovered that ferric hemoglobin is also able to oxidize sulfide to thiosulfate, whereas the physiological significance of this reaction still awaits further exploration. Subsequently, conversion of thiosulfate into sulfate and/or sulfite occurs in the presence of sulfide-detoxifying enzymes such as rhodanese (228). The produced sulfite is also quickly oxidized into sulfate (152). To this end, sulfate can be excreted via urine as a main product of H2S catabolism.
However, it should be noted that the urinary concentration of sulfate does not serve as an accurate marker for endogenous H2S level; sulfate in urine can also be derived from the direct oxidation of
2. Methylation
Unlike H2S oxidation, the methylation of H2S mainly occurs in the cytosol rather than mitochondria. In the process, H2S is methylated into methanethiol, which can be further methylated into non-toxic dimethylsulfide by thiol S-methyltransferase (305). Compared with sulfide oxidation, the methylation of sulfide is shown to be ∼10,000 times slower in colonic mucosa (152). Therefore, it may dispose minimal amounts of H2S in physiological conditions.
3. Expiration
H2S can also escape from mammalian lungs. The amounts of exhaled H2S can be readily detected along with administration of sodium sulfide (Na2S) (113) or inhibition of endogenous NO synthesis (229). Therefore, it seems that expiration may provide a disposal route of H2S when large amounts of H2S are generated. In support of this notion, the exhalation of H2S is detectable in pathological conditions such as septic shock, hemorrhagic shock, and chronic obstructive pulmonary disease when H2S is generated excessively (110, 189, 263). However, it remains unknown how much H2S is lost through lungs in healthy conditions since there is not enough to pick up for measurement. Considering that the free level of H2S is extremely low in mammalian blood (84, 209), the loss of H2S via this route should be very minimal if there is any.
D. Concentration and quantification methods of H2S in mammals
1. Concentration of H2S in mammals
In spite of definitive evidence showing the presence of H2S in mammalian systems, the exact concentration of H2S in vivo has been in debate for a long time due to conflicting data reported. Using the methylene blue method, initial literatures reported that the concentration of H2S is above 35 μM and 50–160 μM in mammalian plasma and brain, respectively (93, 242, 344, 349). However, later studies have shown that the high concentration of H2S may be caused by the usage of a strong acid in the measurement (114, 311), because H2S is released from acid-labile sulfur in the presence of a strong acid when using the methylene blue method.
With the exclusion of a strong acid, 0.7–3 μM sulfide has been reported in the mammalian plasma utilizing monobromobimane (MBB) method with or without dithiothreitol (111, 249, 282, 315) by more robust analytical techniques such as high-performance liquid chromatography (HPLC). Nevertheless, one should bear in mind that MBB reacts and removes HS− from solution, which may result in the release of more HS− from some proteins ionized by HS− at cationic sites (99).
In contrast, the polarographic sensor method can be used to measure H2S concentration without sequestrating it by which a low nano-molor range of H2S concentration has been observed in rodent plasma (311, 315). In line with this, it has recently been shown that the concentration of H2S is about 15 nM in mouse plasma by a newly developed gas chromatography (GC) method (151). Taken together, the concentration of sulfide is suggested to be in the range of nanomolar to submicromolar in mammalian plasma in spite of controversy.
Although the concentration of H2S in mammalian plasma is demonstrated to be rather low, it may be substantially increased in certain microenvironments. This may be due to three possible mechanisms, including: (i) H2S release from bound sulfur. For example, it has been demonstrated that activation of neurons causes the release of sulfide from the bound sulfur of nearby astrocytes, which stimulates the activity of Na+/HCO3 − cotransporter and alkalinizes the astrocytes (114). It is likely that other tissues or organs embrace a similar mechanism as the brain considering the spread of bound sulfur across the mammalian body. (ii) Upregulation of H2S biosynthesis. It has been shown that tissue H2S biosynthesis is enhanced in conditions such as streptozotocin-induced diabetes (334). In such conditions, it is expected that H2S will be temporarily concentrated in the microenvironment, particularly around H2S-producing enzymes before its diffusion or oxidation. (iii) Suppression of H2S catabolism. During hypoxia, O2 reduction leads to suppression of H2S oxidation and subsequent accumulation of H2S, which has been demonstrated in various tissues/systems such as blood vessels, carotid body, kidney etc. (21, 25, 36).
2. Quantification methods of H2S in mammals
As reviewed earlier, the concentration of H2S does not always remain constant and it may fluctuate in particular conditions. Therefore, the accurate measurement of H2S level is crucial for a precise portrayal of the role of H2S in individual circumstances. Herein, the commonly used quantification methods of H2S will be briefly overviewed with emphasis on their sensitivity and reliability for measuring the level of H2S in biological samples. The readers are also advised to refer to the review by Kevil and coworkers (140) and another review article from Nagy et al. (200) discussing in detail the pitfalls that are associated with sulfide quantification.
a. Methylene blue method
In this method, sulfide is first trapped with a metal (usually zinc acetate). The resultant complex reacts with N,N-dimethyl-p-phenylenediamine and leads to the formation of methylene blue in the presence of Fe3+ under acidic conditions. Subsequently, methylene blue is measured at 670 nm with a spectrophotometer. The methylene blue method was initially developed by Fischer (80) for the quantification of H2S in natural water and thereafter adapted for analyzing non-biological samples such as sewage, food, and gas (6, 117, 231). Ever since the demonstration of H2S as an endogenous signaling molecule, it has been extensively employed to measure H2S concentration in blood, tissues, and other biological specimens perhaps due to its easy protocol for usage (1, 77, 209).
However, it is currently believed that this method is not suitable for these biological samples due to the release of H2S from acid labile sulfide pools under acidic conditions (114, 311), though it may be useful in estimating total sulfide pools. Nevertheless, it may still serve well as an acceptable assay to measure the activity of H2S-producing enzymes with proper control groups. Furthermore, it is essential to make the standard curves for sulfide calculation under exactly the same conditions (i.e., pH) with the sample solutions (200).
b. MBB method
In the assay, MBB derivates H2S into sulfide-diamine product in basic conditions, which is subsequently determined with HPLC equipped with a fluorescence detector. The detection limit of this method is 2 nM at optimum conditions, and the good stability of the sulfide-diamine product allows batch storage and analysis (249). Thus, this method is regarded as a highly useful method for sulfide quantification. Nonetheless, examination of publication revealed that the concentration of H2S measured under pH 9.5 seems to be higher than that under pH 8.0 (249, 315). We speculate that this discrepancy may be caused by the release of H2S from some bound sulfide under basic conditions. Thus, special caution should be given for pH control when employing this method to compare H2S levels among different experimental groups. It is worth mentioning that this method can measure the conversion of sulfide into polysulfide, another potential endogenous signaling molecule, as suggested by Nagy and Winterbourn (201).
c. S2−-sensitive electrodes method
In this method, S2−-sensitive electrodes are used to measure the electronic voltage, which can be further calculated as sulfide concentration. It is able to detect H2S concentration in 1 min with a detection range of 1–100 μM and is now widely used for the quantification of H2S in biological samples (328, 344). Since this method does not require sulfide derivation, it, therefore, allows the kinetic measurement of free sulfide concentration (200). Moreover, a polarographic sulfide sensor was developed to overcome the interference of biomolecules and frequent reconditioning (65), which has shown its feasibility to measure free sulfide levels in in vivo models (62, 65).
d. Polarographic H2S sensor method
This method employs polarographic H2S sensors for the measurement of H2S level with Na2S or sodium hydrosulfide (NaSH) as a standard. The assay is sensitive with a detection limit of nanomolar range under anoxic conditions and is able to provide a real-time measurement of H2S from biological samples (65, 139). Though the method is well documented as a reliable method for the measurement of H2S levels by various studies (139, 209), Whitfield et al. (311) have recently reported that they cannot detect any sulfide in plasma derived from various animal species. Later, it was suggested that the earlier on successful studies had constructed electrodes with a unique silicone polycarbonate blend membrane that is not commercially available any longer (140). Intriguingly, Faccenda et al. (73) recently showed that polydimethylsiloxane membrane is successful in construction of H2S electrodes and after measurement of H2S. However, it can be clearly seen that the sensitivity of the method is apparently affected by the materials of H2S electrodes. In addition, how to preserve the sulfide content for later measurement remains to be settled when using this method.
e. GC method
The method uses an alkylation technique to extract H2S followed by GC measurement of the H2S-derived substance. Shigetoshi et al. (254) first established the method and successfully detected the sulfide level in rat blood exposed to H2S. Recently, Furne et al. (84) have improved the method via combing GC with a chemiluminescence sulfur detector by which the concentration of free H2S in mouse blood is later reported to be around 15 nM (151). Both studies showed that the detection limit of the improved method is at nanomolar range. Though it may be not as user friendly as aforementioned techniques, this method is evidently sensitive and specific. Nevertheless, it is notable that the method may primarily facilitate quantification of the species of H2S alone over other species of sulfide, including HS-.
f. Fluorimetric probes
In the method, the reducing ability of H2S serves as the chemical basis for the synthesis of such probes. Compared with the techniques mentioned earlier, fluorescence probes have tremendous advantages on the sensitive and real-time H2S detection within specific tissues and cellular compartments. Due to this, the development of H2S-sensitive fluorescence probes is one of the most rapid growing areas in the field of H2S biology. As a result, numerous such probes have been reported, such as sulfidefluor-1/2 (SF-1/2), dansylazide azide, sulfide-selective fluorescent probe-1/2, and 7-Azido-4-methylcoumarin (96, 166, 227, 236).
However, they, indeed, have some limitations that should be noted when employing these probes: (i) Some of the probes may have limited sensitivity, and, therefore, long time exposure may be needed to catch the changes of H2S levels; (ii) tissue antofluorescence may disturb the use of some probes that have a similar excitation/emission wavelength (175); (iii) some of the probes may have non-specific interaction with other reducing biological thiols such as cysteine and glutathione (311, 315), which may limit their use, particularly in in vivo settings; and (iv) they actually measure the amount of H2S produced over the duration of exposure as the chemical reactions of H2S with these fluorophores are essentially irreversible (99).
III. H2S-Producing Enzymes in Cancer
H2S is produced in mammalian cells by three major enzymes, including CSE, CBS, and 3MST. Among these enzymes, CSE and CBS are located in the cytosol whereas 3MST resides and generates H2S in mitochondria. Numerous studies have shown altered expression of the enzyme(s) in the progression of various types of cancer cells, which have been reviewed next and summarized in Table 1.
Changes in the Expression of Various Hydrogen Sulfide-Producing Enzymes in Various Types of Cancer
3MST, 3-mercaptopyruvate sulfurtransferase; CBS, cystathionine β-synthase; CSE, cystathionine γ-lyase; H2S, hydrogen sulfide.
A. CBS in cancer
The pro-cancer effect of CBS has been thoroughly demonstrated in colon cancer. Tu et al. first observed the correlation of CpG island methylation in CBS promoter with the occurrence and progression of colon cancer (285), implying a possible role of CBS in the tumorigenesis. After this, the expression level of CBS has been measured in human colon cancer biopsies, colon cancer cell lines, and normal margin mucosa by Szabo et al. (266) and Szabo and Hellmich (267). They found that in comparison to normal tissues the expression levels of CBS rather than those of CSE and 3MST are selectively upregulated in colon cancer biopsies and colon cancer cell lines (266, 267). Intriguingly, the proliferation of colon cancer cell line HCT116 was further accelerated on the activation of CBS with SAM (185) or supplementation of NaSH (although <100 μM) (32). Conversely, genetic silencing or pharmacological inhibition of CBS lessened the proliferation of colon cancer cell lines along with the reduced migration and invasion (266, 267).
These anti-cancer effects of CBS blockage in colon cancer have been further substantiated in several mouse xenograft models (48, 102). Recently, Pagliara et al. (219) reported that 5-fluorouracil treatment induces the downregulation of CBS, which may contribute to the following apoptosis in colon cancer cells. Taken together, these results indicate that CBS is a pro-cancer factor in colon cancer development and its inhibition may serve as a promising strategy for colon cancer therapy.
Accumulating evidence also suggests a pro-cancer role of CBS in human ovarian cancer and breast cancer. The expression of CBS is commonly found in primary serous ovarian carcinoma (24) and also ovarian cancer cell lines (47); however, minimal expression of CBS is detected in the non-malignant ovarian surface epithelial cell line OSE (24). siRNA silencing of CBS abrogates tumor growth (24, 47) and increases the sensitivity of ovarian cancer cell lines to chemotherapeutic drug cisplatin (24). The outcome of CBS silencing might be the disruption of mitochondrial ROS production and adenosine triphosphate (ATP) generation, which may result in the inhibitory effect in ovarian cancer cells (24).
In human breast cancer, a more subtle impact of CBS has been suggested. When CBS is silenced with siRNA, it does not alter breast cancer cell proliferation in in vitro culture but significantly reduces tumor growth in xenograft models (246). Further study has shown that CBS silencing inhibits breast cancer cell growth in the presence of activated microphages (246), suggesting that CBS-derived H2S might protect breast cancer cells from the attack of microphage instead of directly promoting cell growth. The pro-cancer effect of CBS in human breast cancer has been further consolidated by a recently reported association between the 844ins68 polymorphism in the CBS gene and the occurrence of breast cancer (87).
In contrast, shRNA knockdown of CBS has been reported to cause rapid growth of human glioma tumor cells both in vitro and in vivo (270). However, the expression level of other H2S-producing enzyme has not been examined in the study. As recent evidence has shown that 3MST is a crucial enzyme for H2S production in brain cells, particularly when CBS is genetically deleted (253), therefore it is possible that compensatory H2S generated from 3MST may have promoted the growth of glioma cells on CBS silencing. The expression of CBS is downregulated in human hepatocellular carcinoma and associated with high tumor grade and high Edmondson grade (132); however, further studies are required to clarify the role of CBS in the progression of this type of cancer.
B. CSE in cancer
CSE might be the dominant H2S-producing enzyme in peripheral tissues as indicated by experiments with mice lacking CSE (328). Increasing evidence suggests that CSE plays a role in the survival of various types of cancers. In prostate cancer, the abundance of CSE is first found in the LNCaP cell (95), which was later confirmed in other cell lines such as PC-3 (226) and LNcaP-B (342). Overexpression of CSE or H2S supplementation with NaSH (although 50–200 μM) apparently induces cancer cell proliferation (226, 342), suggesting a role of the CSE/H2S pathway in the survival of prostate cancer. A similar effect of CSE has also been observed in melanoma (222) and gastric cancer (123, 338), as either CSE overexpression or H2S donors leads to cell apoptosis in both of the cancer types. However, whether CSE promotes or inhibits cancer cell survival remains to be determined, since none of the studies examined the consequences of CSE inhibition or deletion in these cell lines.
Nevertheless, CSE has been found to support cell survival in hepatocellular carcinoma (221, 332). Yin et al. (332) has shown that the expression of CSE is under the control of the phosphoinositide-3-kinase/protein kinase B (PI3K/AKT) pathway in hepatoma cell lines QGY-7703 and SMMC-7721. CSE knockdown by siRNA significantly inhibits the proliferation of both cell lines, which has been further echoed in HepG2 cells by another group (221). In colon cancer cells, the expression of CSE is under the regulation of the Wnt/β-catenin pathway (74), which is an essential player in this type of cancer. Moreover, either siRNA knockdown or pharmacological inhibition of CSE leads to cell death in SW480 and HCT116 (74), suggesting a possible pro-cancer effect of CSE in colon cancer. This has been further supported by the evidence that NaSH (200 μM) stimulated cancer cell proliferation by activating extracellular signal-regulated kinase (ERK) and AKT pathways in HCT116 and SW480 (32). Recent evidence has also shown that the expression level of CSE is positively associated with urothelial cell carcinoma of the bladder (86). However, the exact role of CSE remains to be investigated in bladder cancer development.
C. 3MST in cancer
3MST is the third enzyme that is responsible for H2S production in mammals. Distinct from aforementioned cytosol resident CSE and CBS, it locates and produces H2S in cellular mitochondria (318). Provided mitochondria can use H2S as an electron donor for ATP production (82, 276), it may be interesting to postulate that 3MST-derived H2S might be a direct source of energy in certain tumors if the O2 supply is sufficient. In accordance with this, high levels of 3MST have been discovered in numerous cancer cell lines or tissues, including astrocytoma cell U373 (120), neuroblastoma cell SH-SY5Y (120, 262), melanoma cell A375 and WM35 (120), and gliomas tissue (316). However, no study has shown the consequence of 3MST modulation in cancer. Therefore, the exact role of 3MST in cancer development still remains uninvestigated but will be an interesting area open to explore in the future.
IV. Dual Role of H2S in Cancer Biology
On the discovery of altered expression of H2S-producing enzymes, the possible role of H2S was extensively studied in cancer cells with genetic or chemical approaches to modulate the level of H2S. Existing evidence tends to show a dual role of H2S in cancer progression (summarized below and in Table 2). Nevertheless, before this, one should bear three facts in mind when interpreting the data, including (i) modulation of H2S-producing enzymes can alter not only the level of H2S but also other byproducts such as pyruvate, glutathione, and
Possible Molecular Mechanisms Relevant to the Dual Role of Hydrogen Sulfide in Cancer Biology
AKT, protein kinase B; ATP, adenosine triphosphate; ERK, extracellular signal-regulated kinase; KATP, ATP-sensitive potassium; MAPK, mitogen-activated protein kinase; MEK1, ERK activator kinase 1; NF-κB, nuclear factor kappa-light-chain enhancer of activated B cells; Nrf2, transcription factor NF-E2-related nuclear factor 2; PI3K, phosphoinositide-3-kinase; STAT3, signal transducer and activator of transcription 3.
A. Cancer-promoting effect of H2S
This section provides a summary of cancer-promoting effects of H2S, including stimulation of bioenergetics, pro-angiogenesis, activation of anti-apoptotic pathways, and acceleration of cell cycle. Moreover, H2S-linked protein persulfidation that is relevant to cancer biology is also overviewed.
1. H2S is a bioenergetic stimulator
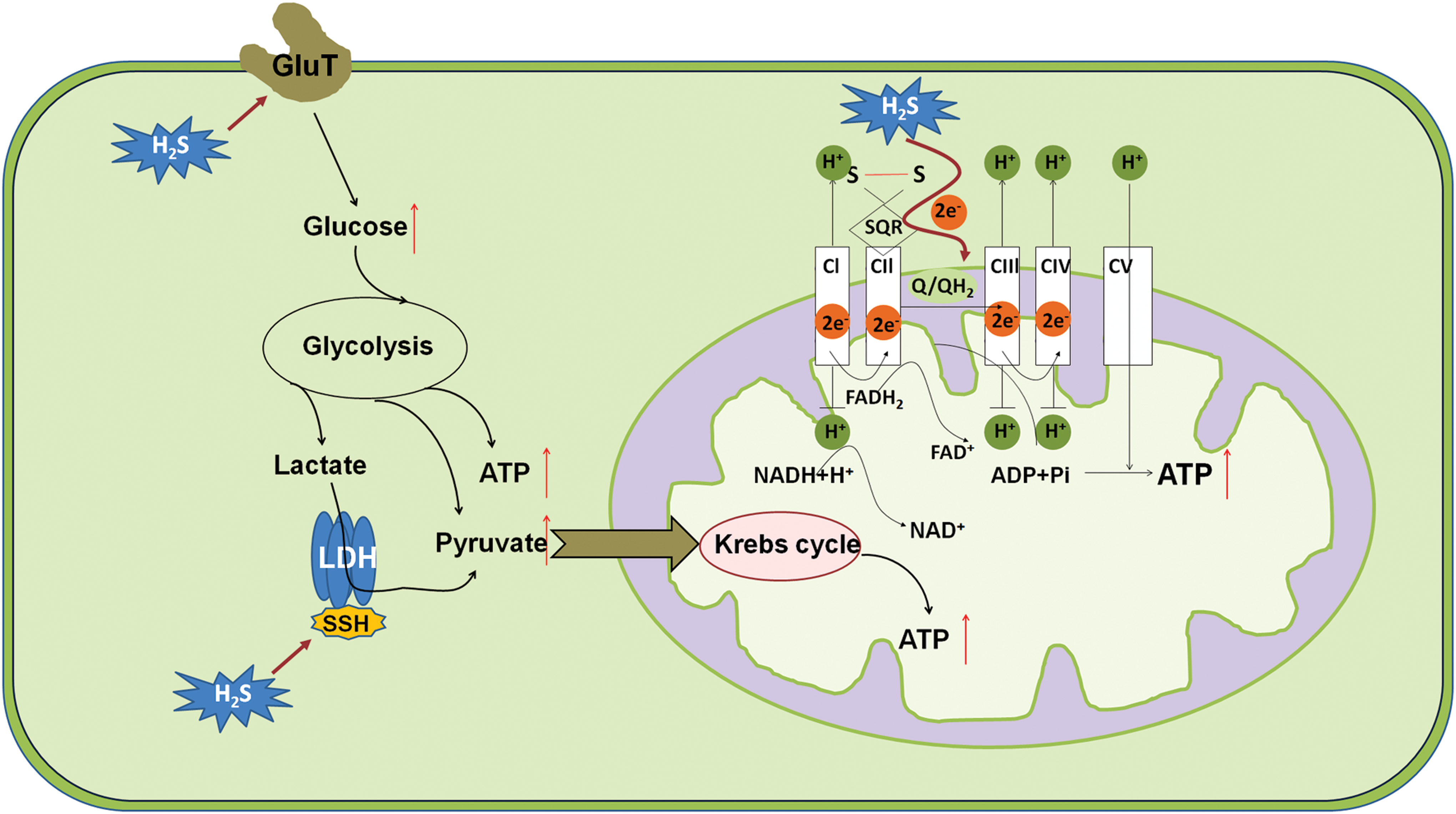
Historically, H2S was well known as a suppressor of the mitochondrial respiratory chain due to its inhibitory effect on cytochrome c oxdiase (56). However, the fact that some bacteria utilize sulfide for energy production has urged to test whether this scenario also exists in mammals (30). Currently, several layers of evidence suggest that H2S at low doses serves as a bioenergetic stimulator in mammalian cells. Goubern et al. (94) have shown a very high affinity for mammalian mitochondria with sulfide, which allows the use of sulfide as an energetic substrate at low micromolar concentrations. Subsequently, the enzyme consuming H2S has been identified to be mitochondrial SQOR, which is an independent parallel electron donor to coenzyme Q, in addition to complexes I and II (145, 186).
Endogenous H2S derived by 3MST plays a pivotal role in supporting the physiological cellular bioenergetic functions, as basal bioenergetic parameters are reduced on siRNA knockdown of 3MST (186, 269). However, they can be enhanced by 3MP (186), the substrate of 3MST. Although stress conditions such as hypoxia were reported to induce the translocation of CSE and CBS into mitochondria to sustain energy production (82), yet the deprivation of O2 should reduce the production of ATP from H2S as O2 serves as the terminal electron acceptor. Furthermore, it has been shown that H2S metabolism is progressively reduced as PO2 falls in isolated mitochondria and this occurs at physiologically relevant PO2 (213). Therefore, H2S-mediated mitochondrial respiration may be only useful to cancer development when O2 supply is still sufficient.
Other than serving as a substrate in mitochondrial respiration, H2S may also stimulate cellular bioenergetics by elevating intra-mitochondrial cAMP levels (188), inducing the persulfidation of mitochondrial ATP synthase (187) and lactate dehydrogenase A (286). Besides mitochondrial respiration, H2S has also been implicated in the regulation of glycolysis, which is well recognized as the main energy source that cancer cells rely on (287). For instance, an inorganic source of H2S (using the salt NaSH) enhanced glucose uptake and glycolysis efficiency in cardiomyocytes likely via stimulating the activity of glucose transporter (GluT). Subsequently, this finding has been paralleled in a panel of cancer cell lines (163).
In line with these, CBS knockdown significantly diminishes the bioenergetic parameters such as oxygen consumption and ATP production in both colon cancer (102, 266) and ovarian cancer cells (24). Nonetheless, the elaboration of whether and how H2S enhances the activity of GluT is still required, particularly in cancer cells. Taken together, H2S stimulates mitochondrial respiration and glycolysis, thereby supporting the energy required by cancer development (Fig. 2).
2. H2S is a pro-angiogenic mediator
Angiogenesis plays a critical role in cancer development given that it promotes new blood vessel formation, which provides tumors with oxygen and nutrients (44). The pro-angiogenic function of H2S was initially found in the late 2000s. NaSH, a commonly used experimental source of H2S, accelerates cell proliferation, migration, and tubule-like structure formation in cultured endothelial cells (31, 223). In addition, exposure of chicken chorioallantoic membrane to NaSH promoted the growth and branching of blood vessels (223). The pro-angiogenic effect of H2S has further been confirmed in rat models (223). Moreover, either pharmacological inhibition or genetic deletion of CSE blocked vascular endothelial growth factor (VEGF)-induced angiogenesis (223, 233), suggesting that H2S may act as as a physiological angiogenic mediator.
Further studies revealed that the signaling pathways underlying the pro-angiogenic effect of H2S may involve the PI3K/AKT pathway, the mitogen-activated protein kinase pathway, and ATP-sensitive potassium (KATP) channels (31, 70, 159, 223). The pro-angiogenic effect of H2S has recently been observed in endothelial cells of tumors. With a well-established model of tumor angiogenesis, namely endothelial cells obtained from breast carcinomas (B-TECs), Pupo et al. (233) found that NaSH (1–10 μM) enhances the migration of B-TECs whereas inhibition of CSE suppresses VEGF-induced migration of B-TECs, suggesting that both exogenous and endogenous H2S promote angiogenesis in breast cancer. In addition, genetic silencing of CBS diminished the neovessel density and tumor growth in rat models of colon cancer (266) and ovarian cancer (24). These results suggest that H2S may be beneficial for tumor growth by promoting angiogenesis, thereby delivering nutrients and oxygen to cancer cells (Fig. 3).
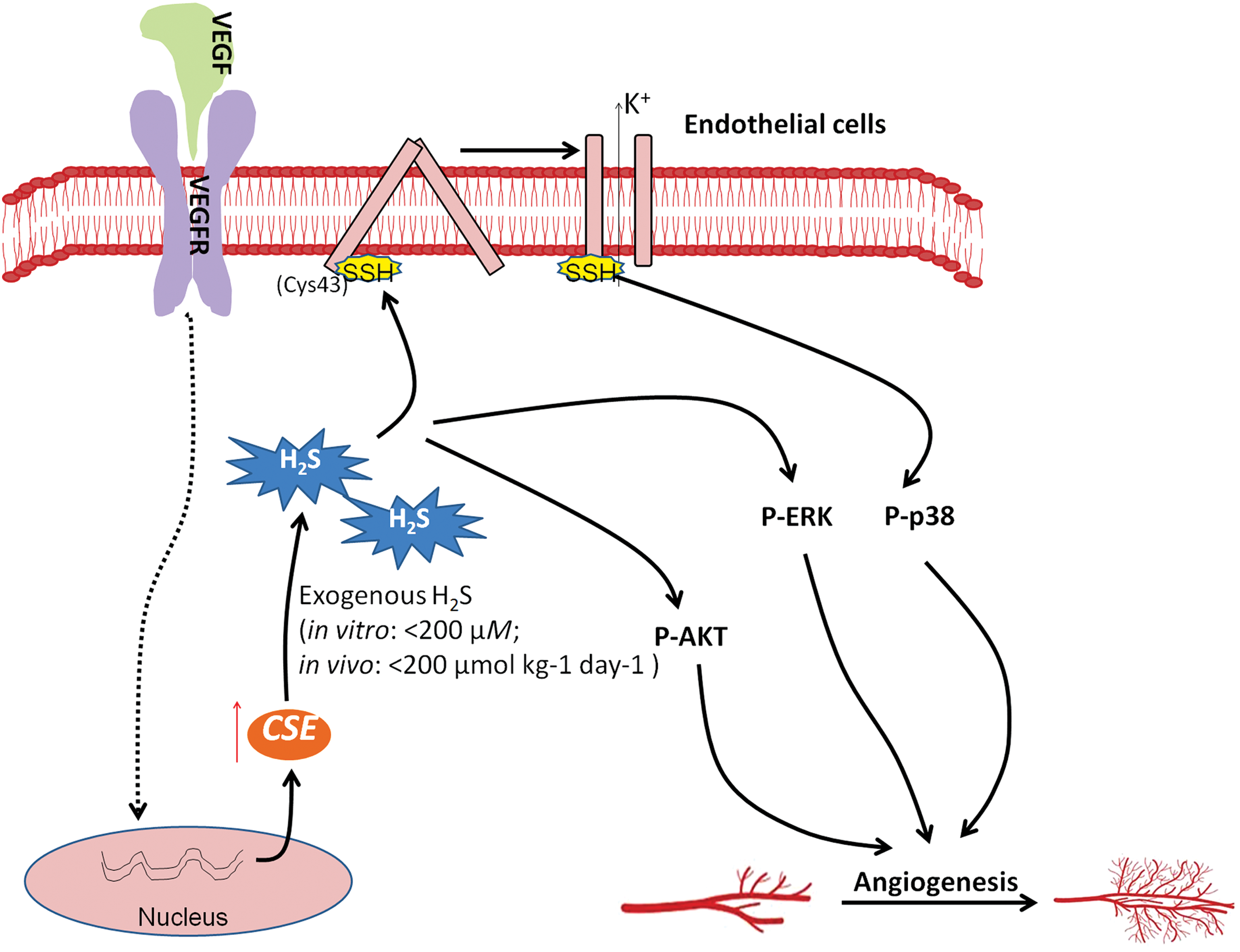
Nevertheless, it is still noteworthy that NaSH at high concentrations or doses (in vitro: >200 μM; in vivo: >200 μmol/kg/day) may also exhibit a suppressive effect on angiogenesis (31, 317), suggesting that the pro-angiogenic activity occurs only with endogenous H2S and/or exposure of low amounts of exogenous H2S in the context of cancer biology.
a. H2S-mediated persulfidation of KATP channel
KATP channel has long been recognized as a potential target of H2S, accounting for its cardiovascular effects such as vasorelaxation, cardioprotection, and hyperpolarization (119, 272, 340, 344). Interestingly, the KATP channel antagonist glibenclamide attenuates H2S-induced p38 phosphorylation and migration of endothelial cells (223), suggesting that H2S–KATP channel–p38 axis may partially mediate the pro-angiogenic effect of H2S.
An elegant study from Mustafa et al. has recently unclosed that H2S-mediated persulfidation activates KATP channel and induces the downstream effects (195). In the study, they have shown that NaSH induces persulfidation of KATP channels in KATP channel overexpressed HEK293 cells. The persulfidation can also be detected in cells overexpressing CSE but not in those lacking CSE, indicating that endogenous H2S induces the persulfidation of the KATP channel. Importantly, the physiological relevance of the persulfidation is further demonstrated by the observation that cholinergic stimulation of mouse aorta increases persulfidation of the KATP channel in wild-type but not CSE-deficient mice. In addition, mutation of cysteine 43 that lies close to the surface of the KATP channel completely abolished the persulfidation and H2S-mediated vasorelaxation, indicating that Cysteine 43 is the principal target of persulfidation in KATP channel.
b. H2S in hypoxia induces angiogenesis
Hypoxia is recognized as a primary physiological regulator of angiogenesis. Tumor hypoxia occurs when a tumor rapidly grows, resulting in limited supply of oxygen. This leads to the induction of the transcriptional factor hypoxia inducible factor 1α (HIF-1α) and subsequent angiogenesis, which helps bring nutrients and oxygen to the tumor. One is advised to refer to Liao and Johnson's article for a detailed description of the process (164). Notably, hypoxia can profoundly evaluate the level of H2S because it inhibits the catabolism of H2S (37) and induces the expression of CSE (294), which is the main H2S producer in endothelial cells. Interestingly, a series of recent studies also provides strong evidence showing that H2S promotes ischemia-induced angiogenesis by augmenting the expression of HIF-1α (25, 168, 268). Therefore, it is highly likely that H2S may partially mediate hypoxia-induced angiogenesis in cancer progression, though direct demonstration of this notion remains lacking.
3. H2S activates anti-apoptotic pathways
Apoptosis is critical for the maintenance of tissue homeostasis in multicellular organism; however, it is suppressed in cancer cells, which affords tumor cells the capability of surviving under various stresses. The anti-apoptotic role of H2S is clearly manifested by several aspects, including (i) mouse embryonic fibroblasts and endothelial cells derived from CSE deficiency mice display accelerated cellular senescence compared with the counterparts from wild-type mice (329, 341); (ii) H2S supplementation reverses CSE deficiency-induced senescence (329, 341); (iii) inhibition of H2S biosynthesis induces apoptotic responses in various types of cells (102, 103); and (iv) H2S shows a protective effect against various apoptotic stimuli (250, 275, 331, 339, 346).
In the context of cancer biology, the anti-apoptotic effect has also been demonstrated in numerous types of cancer cells such as colon cancer (241), hepatomas cancer (345), and neuroblastoma (281). Further studies have revealed the possible underlying mechanisms such as activation of nuclear factor kappa-light-chain enhancer of activated B cells (NF-κB) (245), Keap1-transcription factor NF-E2-related nuclear factor 2 (Nrf2) (329), and ERK activator kinase 1 (MEK1)-ERK pathways (341) mediated by H2S-linked persulfidation (Fig. 4).
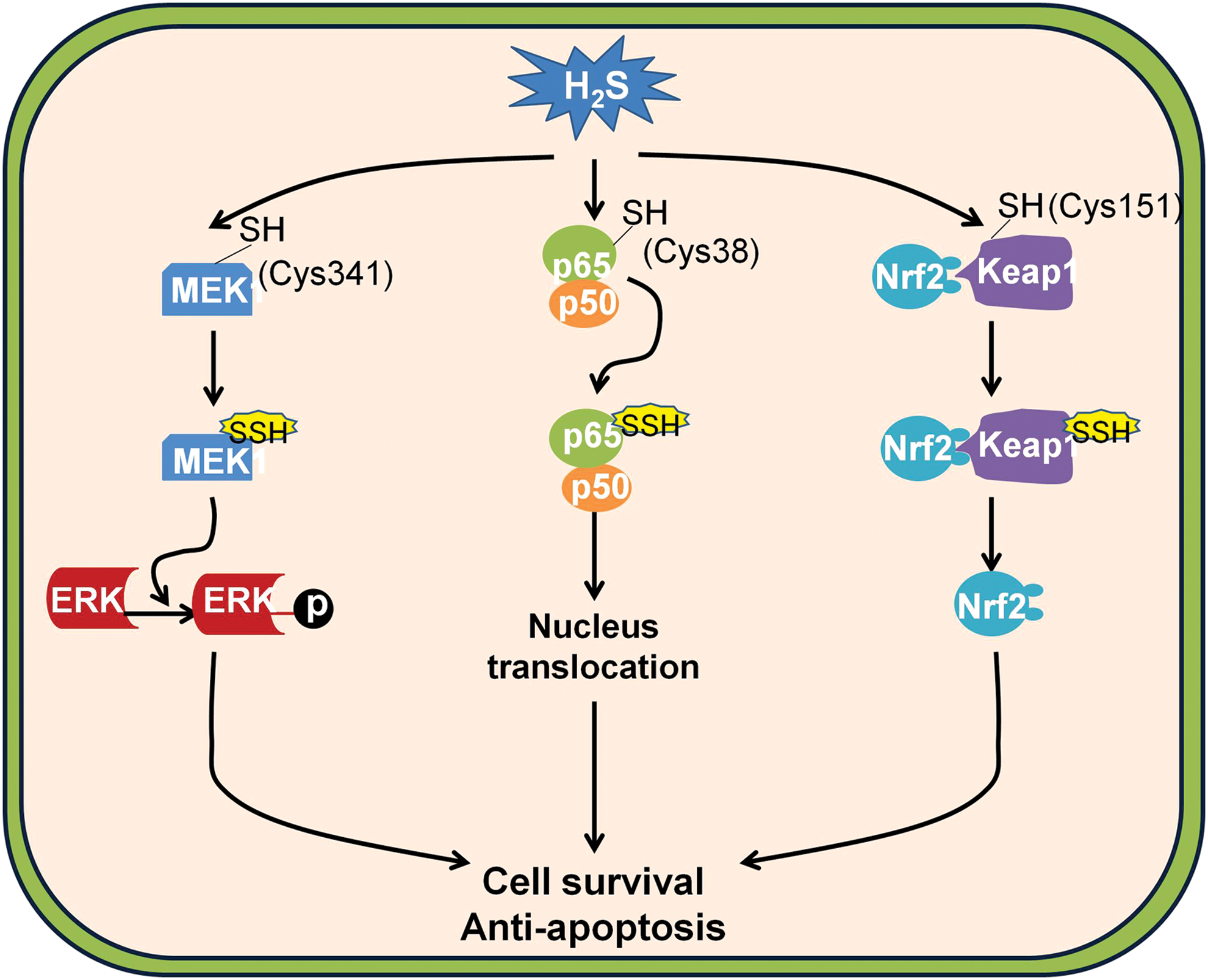
a. H2S-mediated persulfidation of NF-κB p65
The NF-κB pathway mediates its anti-apoptotic function by inducing the expression of several anti-apoptotic proteins such as TNFR-associated factor (TRAF)-1, TRAF-2, caspase-8-c-FLP, and cellular inhibitor of apoptosis (207). The activation of the pathway requires the nucleus translocation of p65 (19), whose lacking induces the death of mice at the embryonic stage due to extensive liver apoptosis (20). Tumor necrosis factor (TNF)α, an activator of the NF-κB pathway, stimulated CSE expression, thereby tripling H2S generation. CSE generated H2S-enhanced DNA binding of p65 and the downstream gene expression, which is absent in mice lacking CSE. As a result, the anti-apoptotic function of the NF-κB pathway is largely diminished in CSE-deleted mice (245).
Sen et al. have recently revealed that H2S promoted the binding of p65 to the promoters of downstream genes by persulfidation cysteine-38 on p65, as the mutation of cysteine-38 abolishes the effect of H2S on p65 persulfidation and NF-κB activation (245). Importantly, the persulfidation is verified by several methods, including biotin switch assay, maleimide assay, and liquid chromatography–mass spectrometry (245). In line with this finding, H2S has long been suggested to be an endogenous mediator of inflammation by augmenting NF-κB pathway activity (154, 310, 336). Similarly, inhibition of CSE may diminish cell proliferation of melanoma cells via suppression of the NF-κB pathway (222). Despite this, one should bear in mind that exogenous H2S may actually be able to suppress the activation of the NF-κB pathway in inflammatory conditions (36, 38, 83, 92, 155).
b. H2S-mediated persulfidation of Keap1
The importance of the Keap1–Nrf2 pathway in tumorigenesis can be evidenced by the finding that somatic mutations of Nrf2 or Keap1 are frequently detected in cancer cells with overactivation of Nrf2-mediated transcription (130), which may afford malignant cells the growth advantage by promoting tumor growth and enhancing cancer chemoresistance (146, 344).
Intriguingly, a recent study from Wang et al. suggested that H2S may be an endogenous stimulator of the Keap1–Nrf2 pathway by persulfidation Keap1 at cysteine-151 (329). In the study, they have shown that in comparison with mouse embryonic fibroblasts from wild-type mice the cells obtained from CSE knockout mice display increased oxidative stress and accelerated cellular senescence, which can be reversed by the supplementation of NaSH (329). In addition, they have shown that NaSH can persulfidate Keap1 and the persulfidation can be fully abolished by the mutation of cysteine-151 of Keap1, but not cysteine-288 (329). This indicates that H2S may persulfidate Keap1 at cysteine 151, which induces the release of Nrf2. Importantly, in CSE wild-type cells, the persulfidation of Keap1 is detectable and can be further enhanced by the H2S-producing substrate cysteine; however, it is absent in CSE knockout cells (329), suggesting that H2S may serve as an endogenous activator of the Keap1/Nrf2 pathway.
By activating the Keap1–Nrf2 pathway, H2S may induce the downstream expression of a battery of cytoprotective genes called vitagens such as glutathione S-transferases, epoxide hydrolase, and ferritin, which may afford cancer cells the adaption for chronic oxidative stress during their progression (33 –35). Nevertheless, how persulfidation of Keap1 triggers the release of Nrf2 remains unclear and further study is, therefore, needed.
c. H2S-mediated persulfidation of MEK1
Somatic mutation is one of the most pervasive characteristics of human tumors (106). To overcome the devastating effect of genetic instability, cancer cells activate a batch of responses, including activation of DNA repair pathways (14, 39). Among these pathways, the overactivation of MEK1-ERK-poly [ADP-ribose] polymerase 1 (PARP1) pathway is commonly detected in a broad spectrum of cancer cells whose inhibition serves as a promising strategy for cancer therapy (300).
An interesting article from Zhao et al. suggested that H2S may facilitate DNA repair by activating the MEK1–ERK–PARP1 pathway (341). In the study, they found that the activity of PARP1 is significantly lower in CSE knockout kidney, liver, and endothelial cells compared with their wild-type counterparts (341). NaSH treatment apparently activates PARP1 and enhances the capability of DNA damage repair, which can be inhibited by two inhibitors of MEK1 (U0126 and PD98059) that can suppress ERK1/2 phosphorylation (341), suggesting the involvement of MEK1 in H2S-stimulated ERK1/2 phosphorylation and PARP1 activity. Moreover, the persulfidation of MEK1 is clearly observed in kidney lysates from wild-type mice, which can be further enhanced by the treatment of NaSH (341). The mutation of cysteine-341 not only fully abolishes NaSH-induced persulfidation of MEK1 but also suppresses the phosphorylation of ERK1/2 (341), indicating that H2S selectively persulfidates MEK1 at cysteine-341 and induces the downstream effects. However, whether cancer cells have higher levels of MEK1 persulfidation is still not clear.
4. H2S accelerates cell cycle
Cancer cells gain the capability of infinitive proliferation through evading cell cycle arrest (100). Recent evidence indicates that H2S is able to accelerate cell cycle in various types of cells such as endothelial cells, cardiomyocytes, and cancer cells (15, 63, 326). For instance, exogenous application of H2S (NaSH, 200–500 μM) reduces the expression of cell cycle regulatory genes such as replication protein A70 and retinoblastoma protein 1 but increases the expression of proliferating cell nuclear antigen and cyclin-dependent kinase 4, thereby promoting cell proliferation in several oral squamous cell carcinoma cell lines (172).
The acceleration effect of H2S on cell cycle has also been found in colon cancer cells (32) and hepatoma cells (221). The signaling mechanism underlying the action might be attributed to the activation of the ERK and AKT pathway (32, 172, 174, 221) because inhibition of either ERK or AKT phosphorylation has been revealed to partially abolish the cell cycle accelerating effect of H2S on squamous cell carcinomas cell lines and colon cancer cell lines (32, 172). Although not directly demonstrated, the underlying mechanisms of H2S-induced ERK activation may be ascribed to the persulfidation of MEK1 as discussed earlier. However, the molecular mechanism underlying H2S-induced AKT phosphorylation remains unclear. Considering the crucial role of AKT in human cancer development (216), the deciphering of this will be of great value.
B. Anti-cancer effect of H2S
In spite of the earlier mentioned evidence that H2S is beneficial for cancer development, recent studies have also disclosed that H2S donation especially at high doses or long time duration leads to cancer cell death without affecting the viability of normal fibroblast cells. In this section, the possible mechanisms underlying the anti-cancer effects of H2S have been reviewed in the context of cancer development (Fig. 5).
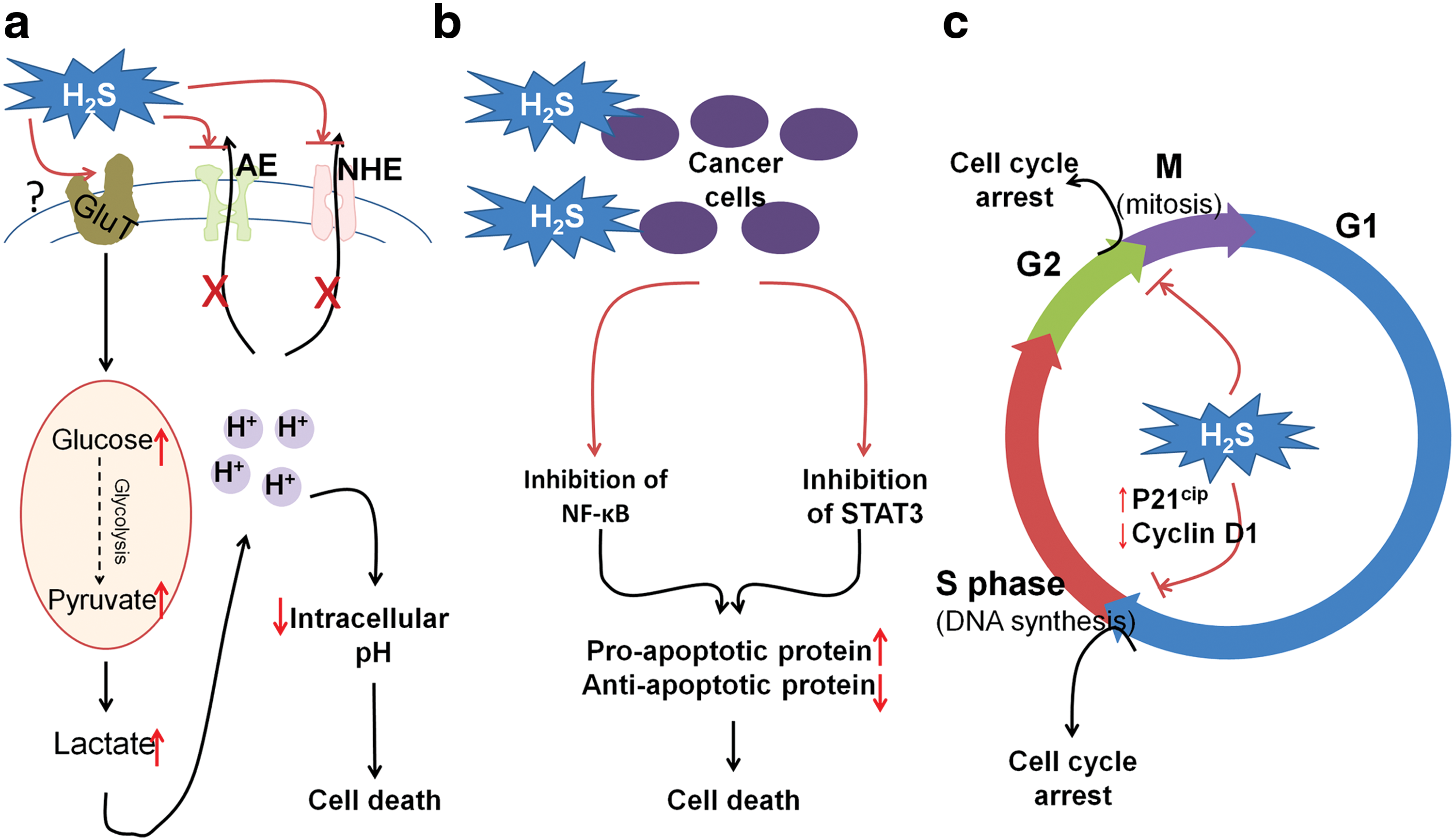
1. H2S donors induce uncontrolled cellular acidification
One hallmark of cancer cells is the utilization of glycolysis as the main pathway for energy production (97). Thus, they tend to have enhanced capacity for glucose uptake and its conversion into lactate. Lactate accumulation results in cellular acidification and stress. However, cancer cells can efficiently export intracellular acid out of cells, which helps form an acidic microenvironment promoting angiogenesis and tumor metastasis (90, 243). Therefore, targeting regulators of intracellular pH is recognized to be a promising strategy for cancer treatment (301).
It was found that a slow-release H2S donor GYY4137 (200–1000 μM) enhances glycolysis of cancer cells by increasing the uptake of glucose, meanwhile it disrupts the exporting of intracellular acid possibly by suppressing the activity of anion exchanger (AE) and sodium/proton exchanger (NHE) (149) (Fig. 5a). Nevertheless, it should not be neglected that the catabolism of H2S to H2SO4 may also contribute to the subsequent intracellular acidification. As a result, this causes uncontrolled intracellular acidification and subsequent cell death in a panel of cancer cell lines (149). ZYJ1122, a control compound for GYY4137 that cannot release H2S as it has no sulfur, had no such effect (149, 150), suggesting that the aforementioned activity of GYY4137 may be exclusively derived from H2S. Intriguingly, GYY4137 exhibits strong anti-tumor effect in mice models (150).
The effect of GYY4137 has also been studied in fibroblast cells such as Wi-38 and MCF10A in their study (149, 150) and GYY4137 did not cause intracellular acidification in these non-cancerous cells. In contrast to this, NaSH (10 μM–1 mM) was found to decrease the intracellular pH of vascular muscle cells by stimulating the activity of Cl−/HCO3 − transporter (148). Thereafter, this effect has been reproduced in primary cultured glia cells, yet not in the neuroblastoma cell line SH-SY5Y (170). Therefore, the controversy among these studies needs to be resolved in the future; for instance, whether the discrepancy can be ascribed to (i) the distinct releasing manner of H2S between NaSH and GYY4137; (ii) the different levels of AE, NHE, and Cl−/HCO3 − among different types of cells; and/or (iii) non-H2S effects derived from GYY4137.
2. H2S suppresses cell survival signaling pathways
Cancer cells disrupt the balance between apoptosis and survival through sustained activation of pro-survival signaling pathways, leading to the constant increase of cancer cell numbers (60). The NF-κB pathway acts as such a signaling pathway and its abnormal activation has been commonly found in various types of cancers, including non-small lung cancer, breast cancer, prostate cancer, etc. (237).
Despite that H2S may stimulate the NF-κB pathway as mentioned earlier by persulfidating the p65 subunit, H2S has been demonstrated to inhibit TNFα and lipopolysaccharides-stimulated NF-κB activation (92, 309). Therefore, it is not surprising to find that constant exposure of cancer cells to H2S (222) or its donating hybrids (127) suppresses NF-κB activity and causes cancer cell apoptosis. However, the molecular target for the inhibitory effect of H2S on NF-κB activity has not been identified. In addition, other pro-survival signaling molecules have been reported to account for the anti-cancer effect of H2S. For instance, GYY4137 causes the apoptosis of hepatocellular carcinoma cell lines by suppression of signal transducer and activator of transcription 3 (STAT3) activation and downregulation of STAT3-mediated downstream proteins such as B cell lymphoma 2, survivin, and VEGF (171). In addition, constant exposure to H2S causes oral cancer cell apoptosis, possibly by reducing the expression of pleckstrin homology-like domain-A1, an apoptotic suppressor in this type of cancer (193). In the future, the target proteins of H2S in cell survival pathways need to be unveiled and studied in detail.
3. H2S induces cell cycle arrest
Dysregulation of cell cycle has been proved to be involved in cancer progression (72). Thus, induction of cell cycle arrest is effective to treat cancer (173). The suppressive effect of H2S on cell cycle transition has been reported by numerous studies (Fig. 5c). S-proargyl-cysteine (SPRC), an H2S donor, causes cell cycle arrest at the G1/S phase in the gastric cancer cell line SGC-7901 and subsequent apoptosis both in vitro and in vivo (123). NaSH (0.4–1 mM) leads to cell cycle arrest at G1/S, possibly by upregulating cyclin-dependent kinase inhibitor p21Cip1 in a panel of colon cancer cell lines (HT-29, SW116, and HCT116) (321).
The inductive effect of GYY4137 on cell cycle arrest has also been proposed in various cancer types (150, 171). For instance, Lu et al. (171) found that GYY4137 suppressed G1/S cell cycle transition by downregulating the expression of cyclin D1 and the tumor growth is inhibited in the subcutaneous HepG2 xenograft model as a result. It was also reported that GYY4137 caused partial G2/M arrest in breast cancer cell line MCF7 whereas the underlying mechanism has not been studied (150). Attractively, H2S seems to specifically induce cell cycle arrest in cancer cells, as neither NaSH nor GYY4137 caused cell cycle arrest in normal fibroblast cells in the aforementioned studies (150, 321). However, the molecular targets of H2S eliciting these effects remain unclear.
Due to its pleiotropic effect on biological processes, there are more potential mechanisms likely underlying H2S-mediated anti-cancer activity. For example, H2S may increase the level of E-Cadherin possessing anti-metastatic effect (108); and H2S has also been found to downregulate the level of histone deacetylases that may result in the epigenetic reactivation of tumor suppressor genes (204). These have been summarized elsewhere in an excellent review by Predmore et al. (232).
C. Bell-shaped model for the dual role of H2S in cancer
The role of H2S in cancer development seems to be controversial. Therefore, an appropriate model system is highly desirable to explain why H2S exerts diametrically opposite effects in cancer cells, in different laboratories. Recently, Hellmich et al. (102) proposed a reasonable bell-shaped model to illustrate the effects of H2S in cancer cells. In the model (Fig. 6), a bell-shaped curve is employed to describe the effects of H2S on cancer cell proliferation. In general, endogenous H2S or a relatively low level of exogenous H2S exhibits pro-cancer effect. In contrast, exposure to H2S with high amounts leads to cancer cell death. This is consolidated by the fact that CBS inhibition not only leads to the inhibition of cell growth but also limits the toxic effect of either 3 mM NaSH or 3 mM GYY4137 in the colon cancer line HCT116 (102).
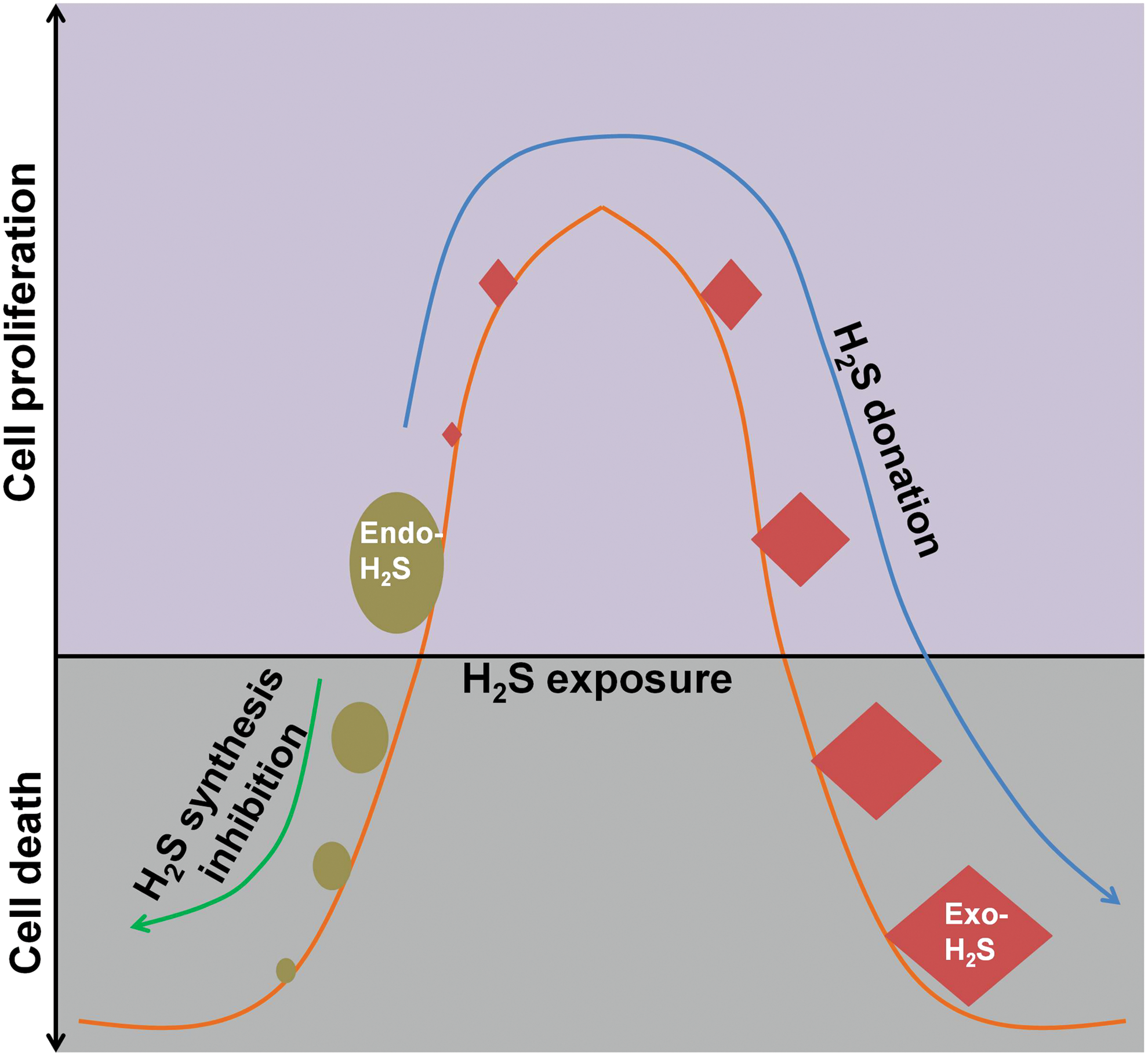
Supportively, Wu et al. (317) recently showed that NaSH (25–100 μM) facilitated vessel formation and tumor growth of hepatocellular carcinomas, both of which were significantly suppressed when the concentration of NaSH was increased to 800 μM. Nonetheless, it is notable that the study relies on a hypothesis that modulating endogenous H2S production is similar to exogenous H2S donation, which may still be plausible considering the unknown intracellular and subcellular H2S concentration after these modulations. Moreover, SAM, a putative allosteric CBS activator, promoted cancer cell proliferation at a lower concentration range (0.1–0.3 mM). However, it inhibited cell proliferation at higher concentrations (1–3 mM) and this effect was partially blocked by CBS silencing (102), suggesting that over-activation of H2S-producing enzymes can also suppress cancer cell proliferation.
Interestingly, this biphasic dose response of H2S in cancer cells well reflects a hormetic response that was recently proposed by Calabrese et al. (34). Specifically, H2S at low concentrations may activate endogenous cellular defense mechanisms that lead to cell death at high concentrations due to its toxic effects. In addition, continuous supplementation of NaSH, (5–20 μM; every 2 h), induced cancer cell death while sparing non-cancerous cells such as MCF10A and Wi-38 after 5 days (150). This effect was well reflected with the slowly releasing H2S donor GYY4137 (42, 150). These results suggest that long time exposure to H2S, even at low doses, can also cause cancer cell death.
Taken together, the bell-shaped models may provide a valuable framework to harmonize the controversies regarding the seemingly controversial role of H2S in cancer development. Meanwhile, it is suggested that H2S biosynthesis inhibition and H2S donation serve as two distinct ways for cancer treatment. Nonetheless, further studies are warranted to precisely define what the exact levels of sulfide exposure are with respect to carcinogenesis in the bell-shaped curve.
V. Crosstalk of H2S with Other Gaso-Transmitters in Cancer Biology
A. Crosstalk of H2S and NO in cancer
There is plenty of evidence showing that H2S and NO actually share some similar actions such as relaxing smooth muscle and interacting with heme groups in protein molecules (271, 290, 296). Intriguingly, these molecules are also demonstrated to crosstalk with each other, even in physiological conditions. Herein, the crosstalk of H2S with NO is reviewed with an emphasis in the context of cancer biology.
1. Endogenous H2S and NO serve as mutual functional effector molecules
The similarities in the function of H2S and NO have urged the investigation whether these two molecules serve as each other's downstream signaling molecule. Up to date, there is adequate evidence to suggest that they may accomplish this by mutually affecting each other's biosynthesis in mammalian cells (Fig. 7). NO is able to induce CSE expression (298, 344) and facilitate the cellular uptake of H2S-producing substrate
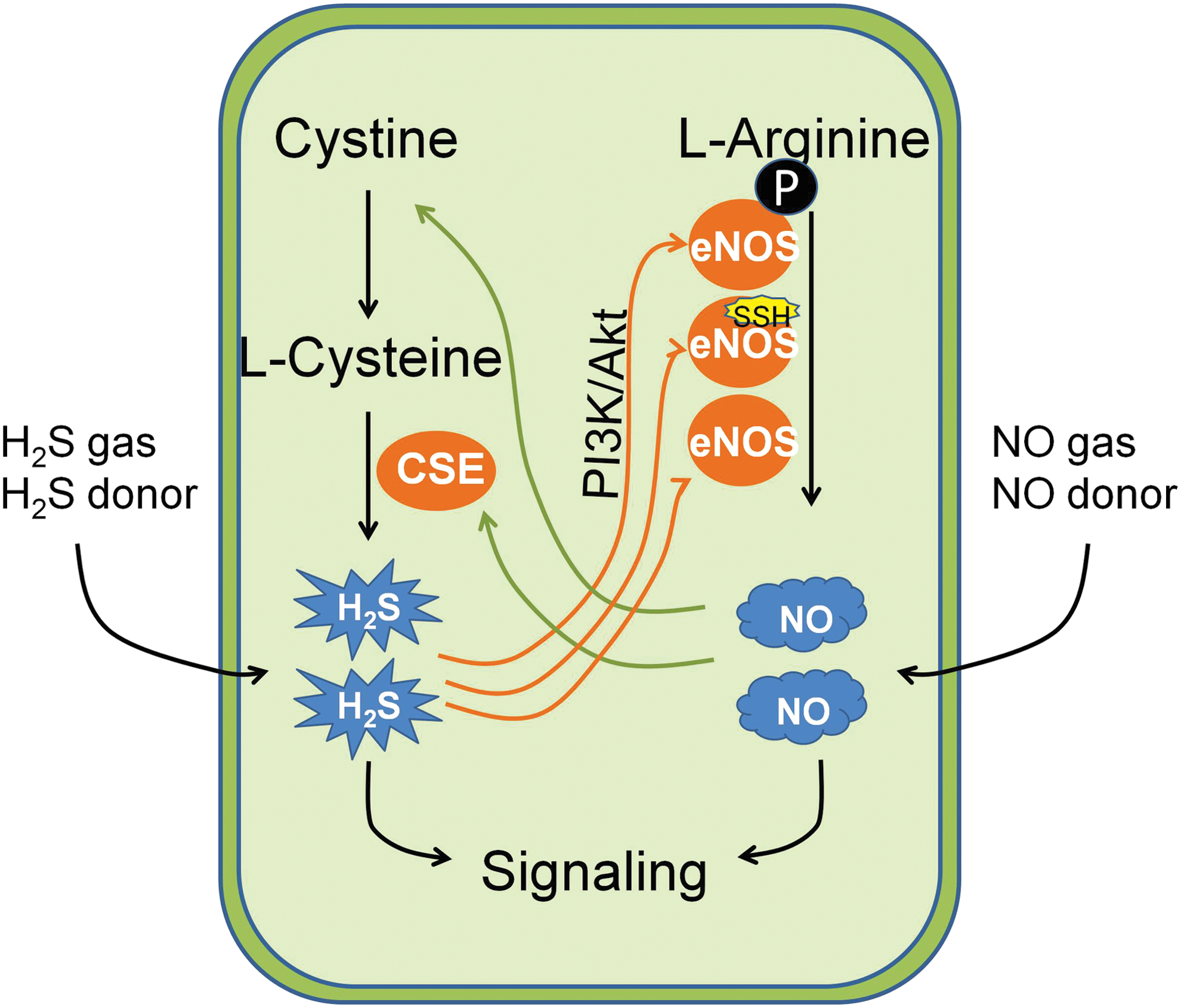
Conversely, H2S also enhances the production of NO. For example, CSE deficiency in mice causes endothelial nitric oxide synthase (eNOS) dysfunction, NO reduction, and aggravated myocardial ischemia/reperfusion injury (137). When H2S is supplemented, NO level increases by the induction of eNOS expression (55, 142, 180). In addition, NO production is also prompted on the persulfidation of eNOS induced by H2S (7, 131). As a result, it was found that suppression of eNOS lessens H2S-mediated vasorelaxation and genetic knockdown of CSE attenuates NO-stimulated angiogenesis (55). In the context of cancer biology, both endogenous H2S and NO are suggested to be beneficial for cancer progression (265, 314). Therefore, it is likely that NO partially mediates the pro-cancer effect of H2S and vice versa. This implies that simultaneous inhibition of H2S and NO biosynthesis may afford synergic effects on the suppression of cancer cell growth, which should be tested in the future.
2. Bioactive products of H2S and NO interaction possess anti-cancer effect
For a long period, H2S and NO signaling pathways have been also observed to be intimately intertwined with mutual potentiation of responses, particularly in the cardiovascular system (69, 198). For instance, Yong et al. (333) have reported that a combination of H2S and NO produces positive inotropic and lusitropic effects likely via formation of a thiol-sensitive molecule that is suggested to be nitroxyl (HNO) in the study. This is further confirmed by studies published by Filipovic et al. (78) and Zhou et al. (347). However, the possible interaction between these two gaseous transmitters remains unexplored in the context of cancer biology.
A recent study from Whiteman et al. (308) suggests that thionitrous acid (HSNO) is spontaneously formed when H2S and NO gases are mixed at room temperature in the presence of metallic surfaces. And a further reaction of HSNO with H2S may lead to the formation of HNO and inorganic polysulfides such as H2S2 (202). Subsequently, Cortese-Krott et al. (59) have identified that the chemical reaction between H2S and NO occurs even in physiological conditions and the key bioactive reaction products of the interaction are S/N-hybrid species, inorganic polysulfides, and HNO (Fig. 8).
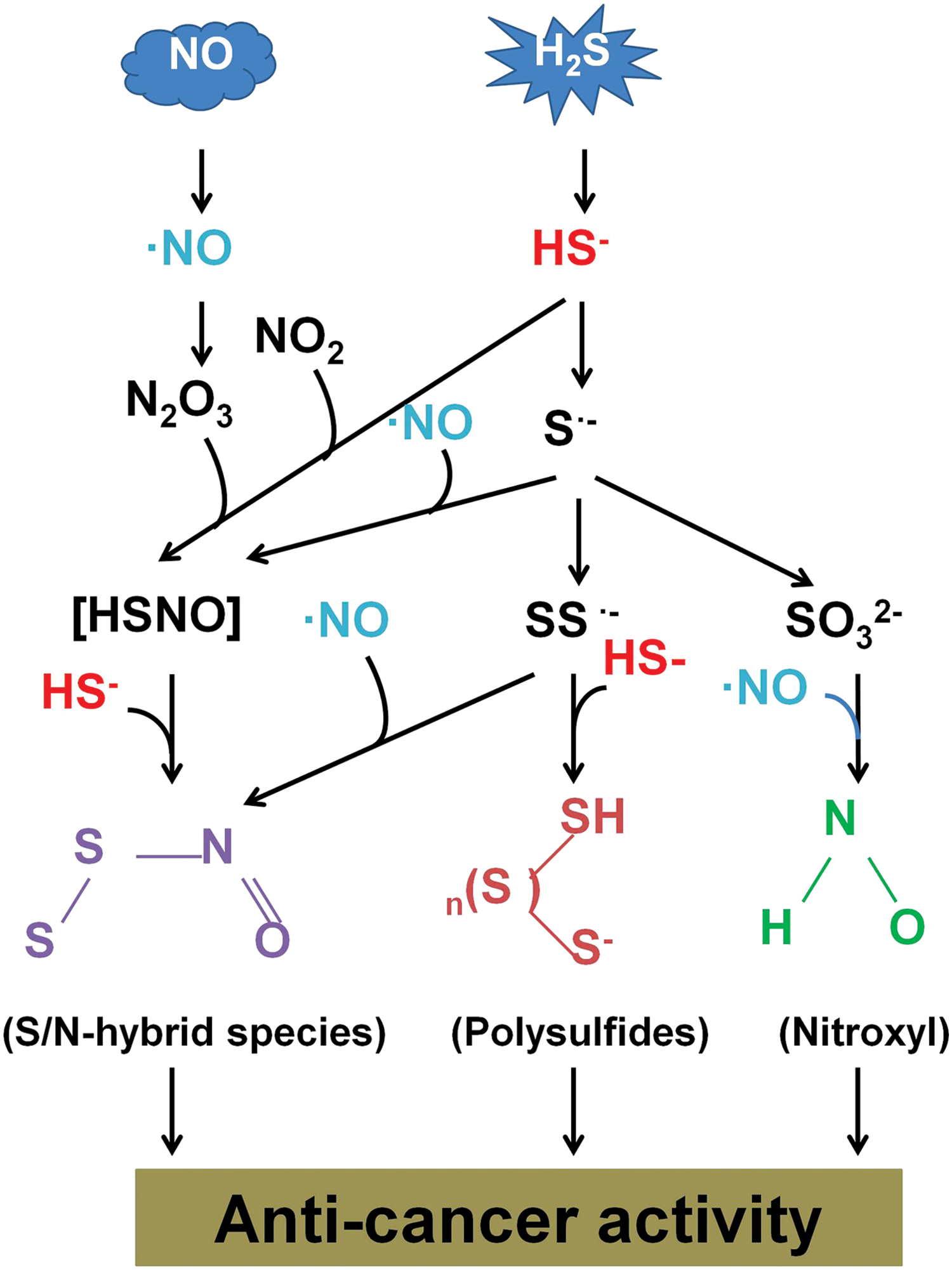
Chemical studies on some of those S/N-hybrid species, however, have strongly questioned the formation of these intermediates in the cells, and more so in vivo. For example, Koppenol and Bounds (141) showed that it would take more than 1 day to generate a maximum of 1 nM of SSNO– from 1 μM H2S based on the values reported by Cortese-Krott et al. (59). Thus, studies are warranted to resolve this conflict in the future. Nonetheless, all these species have been shown to possess anti-tumor effects by previous studies (46, 51, 255a), indicating the potential of a direct interaction between the two gaseous transmitters in cancer biology. Further studies are, therefore, needed to validate the interaction that may lead to the discovery of novel strategies for cancer therapy.
B. Crosstalk of H2S with CO in cancer
The direct reaction between H2S and CO has not yet been reported so far; however, emerging evidence suggests that the axis of CO–CBS–H2S may be important in cancer regulation, which has been discussed next.
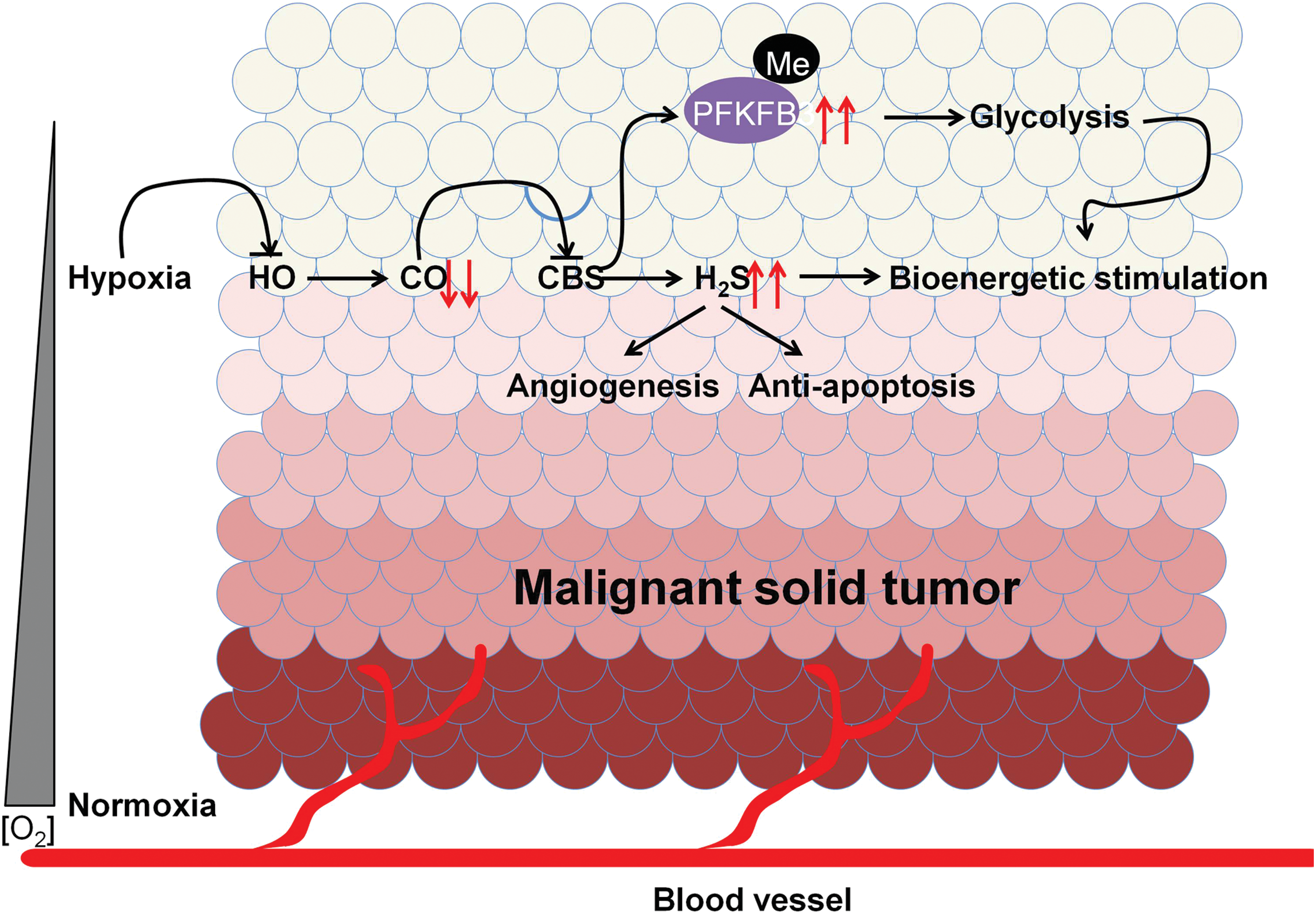
1. CO–CBS–H2S axis in cancer regulation
CO is a gaso-transmitter generated from heme oxygenase (HO) by catalyzing the oxidative degradation of heme, a process highly relying on the availability of cellular O2 (313, 319). So far, there is plenty of evidence suggesting that CO is an intrinsic modulator of H2S biosynthesis by inhibiting CBS activity. For example, an increase of CO level in liver leads to the reduction of H2S concentration and a subsequent increase of bile excretion, both of which are shown to be absent in CBS heterozygous knockout mice (255). In line with this, hypoxia increases CBS-mediated H2S production through inhibition of HO-2 activity and CO biosynthesis, which may contribute to vasodilation of precapillary arterioles (190). It is worth mentioning that hypoxia can also lead to the suppression of H2S oxidation, thereby augmenting H2S signaling (210, 213), which could be very relevant to the development of solid tumors.
Moreover, the inhibitory effect of CO on CBS activity has been ascribed to its irreversible binding to the heme group of CBS (122, 234, 256). Currently, accumulative studies suggest that this regulation may be relevant to cancer development. As a tumor grows, it rapidly outgrows its blood supply, leading to a significantly lower concentration of O2 in tumor tissues compared with that of normal tissues (104, 105). Such a low level of O2 could lead to the reduction of CO biosynthesis and H2S overproduction, which may be supportive for tumor growth through the beneficial effects of H2S in cancer development as reviewed earlier. In accordance with this, the low enzymatic activity of HO-1 is found in moderately differentiated prostate tumors, which correlates with relatively worse clinical outcomes (303).
In addition, exposure to a low level of CO diminishes the rate of glycolysis in cancer cells and sensitizes them to chemotherapy with enhanced growth arrest and apoptosis (303), which are very similar to that after CBS inhibition (266, 267). Moreover, overexpression of HO-1 in cancer cells attenuates the growth of lung carcinoma xenografts in mice (203, 258, 277). However, it is notable that although the reduction of H2S caused by CBS inhibition may be, at least partially, involved in this CO-mediated cancer inhibition, a direct demonstration of such involvement is still lacking.
Other than the decrease of H2S, CBS inhibition by CO has also been found to cause demethylation of proteins such as 6-phosphofructo-2-kinase/fructose-2,6-bis-phosphatase 3 (PFKFB3) (325). The demethylation of PFKFB3 results in the attenuation of glycolysis in cancer cells (261). Therefore, it can be proposed that CBS activation on CO reduction in hypoxic conditions such as a tumor microenvironment may contribute to the maintenance of glycolysis partially by enhancing PFKFB3 methylation. The possible mechanisms underlying CO-CBS-H2S axis in cancer regulation have been illustrated in Figure 9.
VI. The Potential of Targeting H2S for Cancer Treatment
A. Treatment of cancer with H2S biosynthesis inhibitors
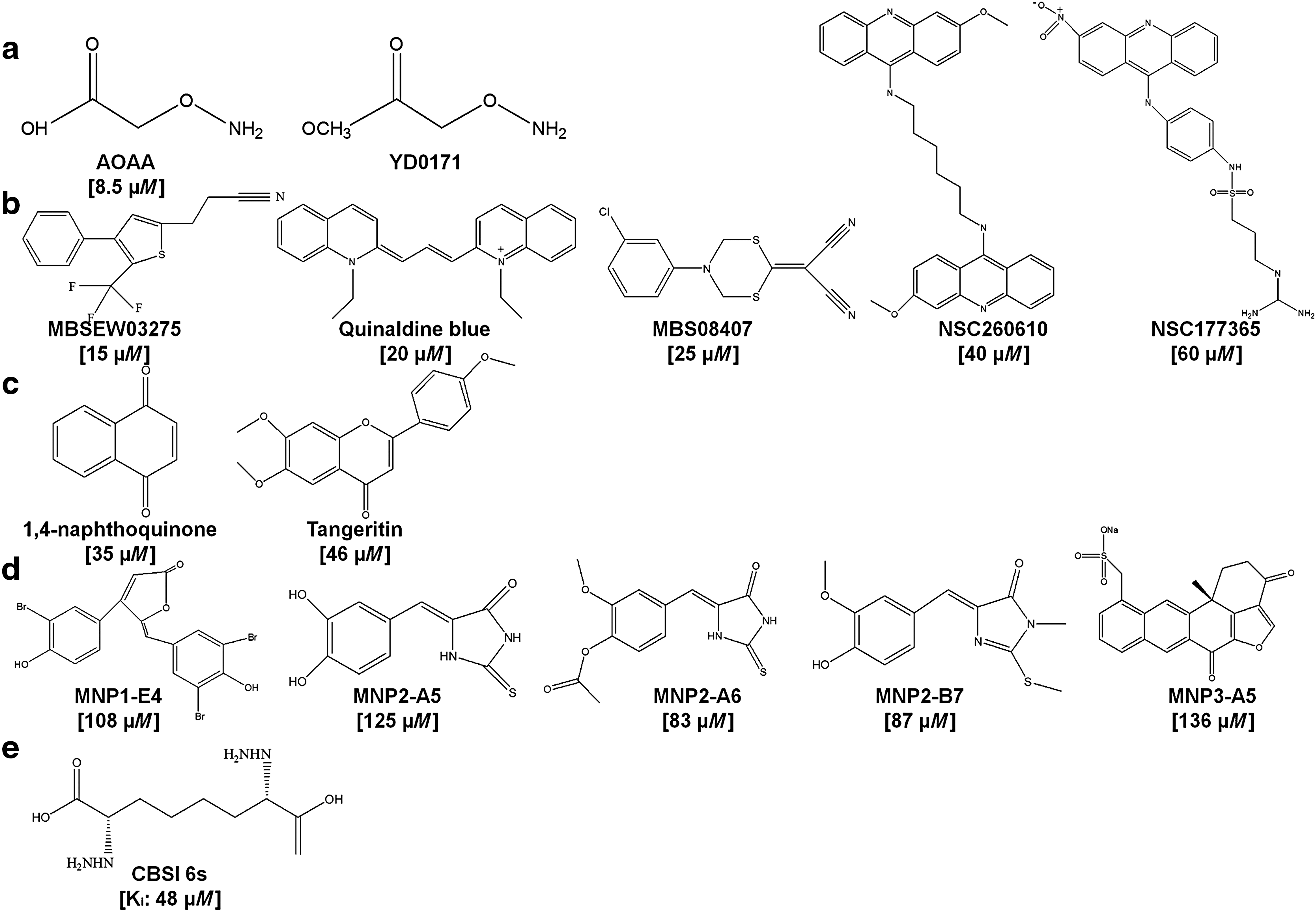
All the three H2S synthesizing enzymes, namely CSE, CBS, and 3MST, might be involved in cancer development, as shown in section III, however, only CBS has been demonstrated as a potential therapeutic target for cancer treatment, particularly in colon cancer (48, 102, 103, 266, 267) and ovarian cancer (24, 47). As a result, the discovery of CBS inhibitors has attracted more attention compared with their counterparts. In this section, we summarize the current CBS inhibitors and their usage, particularly in cancer treatment. The chemical structures of these CBS inhibitors have been illustrated in Figure 10.
1. CBS inhibitor aminooxyacetic acid shows great potential in cancer therapy
Aminooxyacetic acid (AOAA) (Fig. 10a) is a traditional CBS inhibitor and has been extensively used in the field of H2S biology. In colon cancer cell lines, AOAA treatment inhibits H2S production, cell migration, and cell growth, closely mimicking the effect of CBS silencing (266, 267). A similar effect was observed in other cancer cell lines, including ovarian cancer cells (24), breast cancer cells (278), and pancreatic adenocarcinoma cells (259). In vivo, AOAA exhibits remarkable inhibition of HCT116 cell xenografts (267) and the proliferation of patient-derived colon tumor xenografts (102). Moreover, the inhibitory effect on breast cancer has also been shown in a xenograft mouse model (278). However, it is noticed that non-cancerous cells have not been included in most studies mentioned earlier. Therefore, it remains unclear whether AOAA has sufficient therapeutic window for cancer treatment.
Recently, Chao et al. (48) found that addition of a methyl ester group on AOAA enhances the cellular uptake of AOAA and the new molecule YD0171 (Fig. 10a) exerts higher potency on the growth inhibition of colon cancer cell line HCT116 both in vitro and in vivo. In addition, YD0171 reduces tumor growth in the patient-derived tumor xenograft model and causes no systemic toxicity in a 5-day safety study, indicating the value of the molecule for cancer treatment. On top of these, one should bear in mind that the selectivity of AOAA for CBS over CSE or other PLP-dependent enzymes is limited as it targets the PLP cofactor, which is commonly used by this class of enzyme (102). In line with this, a study has indicated that AOAA is three times less potent for CBS than CSE (9). Therefore, selective CBS inhibitors are of high demand.
2. Advances in the development of selective CBS inhibitors
To obtain novel selective CBS inhibitors, Zhou et al. (348) initiated the first high-throughput assay for the identification of potent and selective inhibitors of CBS in 2013. With a tandem-microwell assay, 21,599 agents from various libraries have been screened against recombinant human CBS (hCBS). As a result, 35 compounds have been confirmed as hits with an IC50 of <50 μM for the inhibition of hCBS. Among them, five compounds, namely MBSEW03275, JHU-8555/Quinaldine blue, MBS08407, NSC260610, and NSC177365 (Fig. 10b), have shown a preference to inhibit hCBS over human CSE with MBS08407 and Quinaldine blue as the most selective ones. Interestingly, quinaldine blue is an anti-tumor drug approved by FDA; however, its molecular mechanism is not clear (75). This study implies that quinaldine blue may inhibit tumor growth by selectively suppressing the activity of CBS.
Later, Barrios and colleagues (279) conducted another screen of a small chemical library containing 1900 compounds with an H2S probe-based assay. Eventually, two compounds, namely 1,4-naphthoquinone and tangeritin (Fig. 10c), were yielded that selectively suppress the activity of CBS without affecting that of CSE. Intriguingly, both the compounds have been suggested as potential anti-cancer molecules, especially for colon cancer in other studies (129, 191). Using a library of marine natural products (MICL-240 library), Barrios and colleagues (280) have recently performed another screen to identify novel CBS inhibitors. They identified five hits (IC50 < 150 μM; Fig. 10d) as CBS inhibitors; however, the selectivity of these compounds remains elusive, as the effect of these compounds on CSE has not been tested. Nevertheless, these scaffolds may serve as useful starting points for the synthesis of selective CBS-targeted molecules in the future.
Apart from the earlier mentioned high-throughput strategy, McCune et al. (177) recently reported a novel way to synthesize selective CBS inhibitors. They chose the enzymatic product (
B. Treatment of cancer with H2S donors
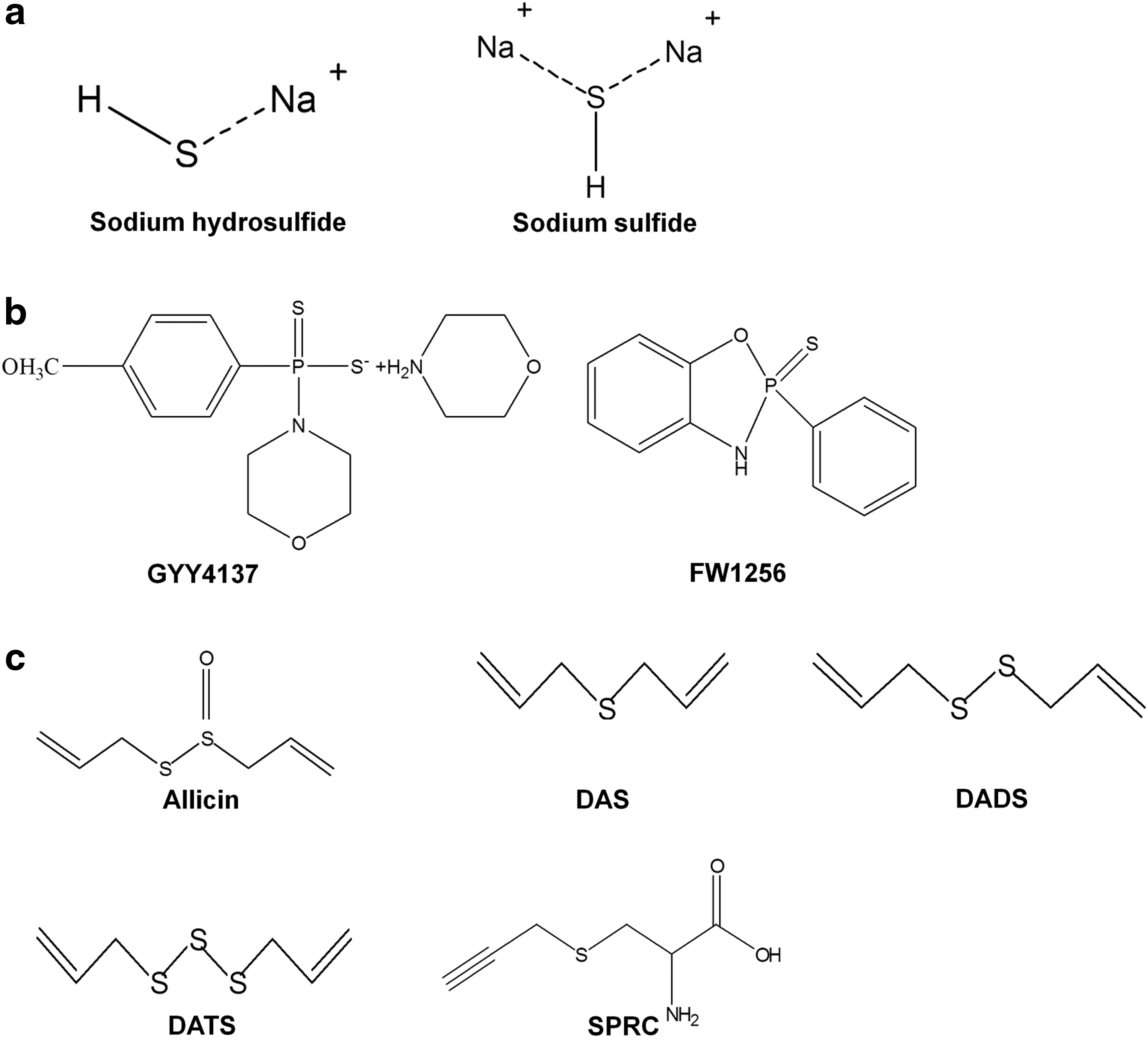
H2S donors with different properties have been developed and tested in various disease models. Herein, we review three categories of H2S donors, including sulfide salts, phosphorodithioate derivatives, and allium sativum extracts and derivatives, with an emphasis on their application in cancer treatment (Fig. 11).
1. Sulfide salts
Sulfide salts (Fig. 11a), namely NaSH and Na2S, are the most commonly used H2S donors in the field of H2S biology. They readily dissolve in physiological solution buffers and water and form HS- and H2S (157). As they do not have any backbone structures, it is believed that the biological effects derived from these molecules are solely from H2S. In the context of cancer biology, the effect of salt sulfides appears to be concentration dependent. Specifically, it promotes cancer cell proliferation at low doses and inhibits cancer cell growth at high concentrations (102). Perhaps due to the distinct effects caused by concentrations, no studies have been performed to show their efficacy for cancer inhibition within in vivo models. In addition, the concentration-dependent effect of these H2S donors has also been observed in non-cancerous cells such as heart cells and smooth muscle cells (15), indicating a possible marginal therapeutic index of these compounds in cancer treatment. The other concern of utilizing sulfide salts is that they release H2S at an uncontrolled manner and may cause acute toxicity to organs.
Taken together, sulfide salts serve as great tool compounds to illustrate H2S-mediated biological effects and the underlying mechanisms whereas the feasibility is rather low to translate them as a therapeutic drug, especially for cancer treatment.
2. Phosphorodithioate derivatives
GYY4137 (Fig. 11b) is the first phosphinodithioate-derived H2S-releasing compound that was synthesized in 2008 (162). It slowly releases H2S in aqueous solution via a two-step process and after intravenous or intraperitoneal injection in animals, closely mirroring the physiological releasing manner of H2S from mammalian cells (5, 162). Therefore, it has been extensively used to study both physiological and therapeutic effects of H2S. The anti-cancer effect of GYY4137 has been demonstrated both in vitro and in vivo (149, 150, 162, 171). Importantly, GYY4137 induces cell apoptosis in cancer cell lines without affecting the cell viability of noncancerous fibroblast cells (149, 150), indicating the potential value of the molecule for cancer treatment. However, examination of published data (149, 150) clearly indicates that the concentrations and/or doses of GYY4137 employed to exert the anti-cancer effect are high (400–1000 μM for in vitro; 100–300 mg/kg for in vivo), indicating its marginal potency. However, H2S production from GYY4137 is inefficient (5), and very little H2S is produced, necessitating its use at high concentrations/doses.
To improve the anti-cancer potency of GYY4137, Feng et al. (76) have recently synthesized a series of compounds based on the structure of GYY4137. Among these compounds, FW1256 (Fig. 11b) has shown superior potency in MCF7 tumor spheroids compared with GYY4137 (IC50: 5.7 μM for FW1256 vs. 368 μM for GYY4137) and caused cancer cell death through apoptosis by activating PARP and caspase-7. At a concentration inhibiting cancer cell proliferation, no toxic effect is observed in noncancerous fibroblast cell Wi-38. Further analysis with a series of these compounds shows a positive correlation between the anti-cancer potency and H2S-releasing rate, which may provide a guide for the future development of such compounds. However, the study did not show the anti-tumor activity of FW1256 in vivo, which needs to be done in the future.
3. Allium sativum extracts and derivatives
Allium sativum (garlic) has been extensively used for numerous diseases, including cardiovascular disease and cancer in traditional Chinese medicine (324). It is abundant with sulfur-containing compounds that can be easily recognized with a smell. Among these compounds, allicin (diallyl thiosulfinate; Fig. 11c) has been extracted and studied thoroughly. Intriguingly, compiling evidence suggests that allicin possesses anticancer effects in various types of cancer both in vitro and in vivo (17, 218, 273). For instance, allicin significantly inhibits cell proliferation of several colon cancer cell lines such as Caco-2, HT-29, and HCT116 (17).
In addition, allicin administration apparently inhibits tumor growth in L5178Y lymphoma-bearing mice (218). Allicin is very unstable and decomposes quickly into several compounds, including diallyl sulfide, diallyl disulfide, diallyl trisulfide, and allyl methyl sulfide (Fig. 11c). Therefore, these individual compounds have also been synthesized and tested in various disease models. The results have shown that the number of sulfur atoms well reflects the efficacy of these molecules not only in cancer inhibition (192) but also in vasodilation (22), which positively correlates with the H2S-releasing capacity. In spite of this, further studies are still needed to investigate whether the effects are derived from other reactive sulfur species or H2S directly, as suggested by a recent study (61).
In addition, SPRC (Fig. 11c), a novel stimulator of endogenous H2S, is a structural analogue of S-allycysteine that is derived from garlic component S-alkyl cysteine sulfoxides. It has been shown to stimulate H2S production continuously in mammalian cells (295). Recently, SPRC was found to cause cell cycle arrest at the G1/S phase in gastric cancer cell line SGC-7901 and subsequent apoptosis both in vitro and in vivo (123). Intriguingly, the anti-cancer effect of SPRC can be blocked by CSE inhibition (123), indicating that the effect can be at least partially attributed to the production of H2S. However, the marginal potency (IC50 ≈ 5 mM) may be an issue to use this compound as cancer therapy, which needs to be improved in the future.
4. H2S-releasing hybrids
a. H2S-releasing non-steroid anti-inflammatory drugs
(1) H2S-releasing non-steroid anti-inflammatory drugs possess anti-cancer activity
The finding that H2S is able to enhance ulcer healing (291) has impelled the synthesis of H2S-releasing non-steroid anti-inflammatory drugs (HS-NSAIDs) to limit NSAIDs caused gastrointestinal side effect. Usually, 5-(4-hydroxyphenyl)-3H-1, 2-dithiole-3-thione (ADT-OH) is covalently conjugated with an NSAID to form a hybrid (128). So far, a number of HS-NSAIDs have been developed, including HS-Sulindac, HS-Ibuprofen, HS-Mesalamine, HS-Naproxen, HS-Diclofenac, HS-Aspirin (HS-ASA), and HS-Indomethacin (127). Most of these drugs have shown less damage to the gastrointestinal tract compared with their respective NSAID (127), which favors the initial hypothesis. Besides inflammation, NSAIDs, in general, also serve as potential chemopreventive agents against cancer as indicated by Kune et al. (144) that subjects taking NSAIDs have a significantly lower incidence of colon cancer.
Recently, four HS-NSAIDs, including HS-Sulindac, HS-Ibuprofen, HS-naproxen, and HS-ASA (Fig. 12), have been tested within 11 cancer cell lines from six tissue origins across from solid tumor to leukemia (50). Remarkably, all the HS-NSAIDs tested exert inhibitory effects on the growth of the 11 cancer cell lines. To achieve the same extent inhibition, the parental NSAID treatment alone requires 38- to 1300-fold higher concentration (50), indicating a synergic effect of H2S and NSAID on cancer inhibition. The anti-tumor effect of HS-NSAIDs was further demonstrated in several in vivo models (50). Taken together, HS-NSAIDs not only increase the therapeutic index but also enhance the anti-tumor activity of NSAIDs, showing their potential value as novel anti-cancer agents. Nonetheless, though existing data showed that some H2S-NSAIDs such as HS-Diclofenac can release H2S in plasma and their effects can be mimicked by an injection of ADT-OH and corresponding NSAID (160), however, whether these compounds release H2S and NSAIDs simultaneously that account for the subsequent anti-cancer effect was not established in many cases.
(2) Mechanisms of action of H2S-NSAIDs in cancer inhibition
Along with the recognition of the remarkably anti-cancer potential of HS-NSAIDs, the mechanisms underlying such effects have also been thoroughly studied, which has been summarized in Figure 13. Chattopadhyay et al. (50) have found that the four HS-NSAIDs (HS-Sulindac, HS-Ibuprofen, HS-naproxen, and HS-ASA) induce apoptosis in HT-29 colon cancer cells accompanied by cell cycle arrest in the G0/G1 phase, which has been further paralleled in breast cancer MDA MB-231 (98). In addition, also using MDA-MB 231 cells, it is suggested by studies from Kashfi et al. that HS-ASA may inhibit cancer cell growth by suppression of the NF-κB signaling pathway via preventing the phosphorylation of the IκB kinases (12, 49). Moreover, it has been shown that HS-ASA concentration dependently results in the cellular ROS formation, which may also contribute to the following cancer cell apoptosis (50, 54). Though the mechanisms underlying HS-ASA-mediated ROS production remain elusive, it is worth mentioning that H2S, in fact, can behave as a pro-oxidant through the interaction with dioxygen to produce sulfur-centered and oxygen-centered free radicals (10, 260). This could induce a redox shift over the redox set points of cancer cells (i.e., redlines) and result in cancer cell death (232).
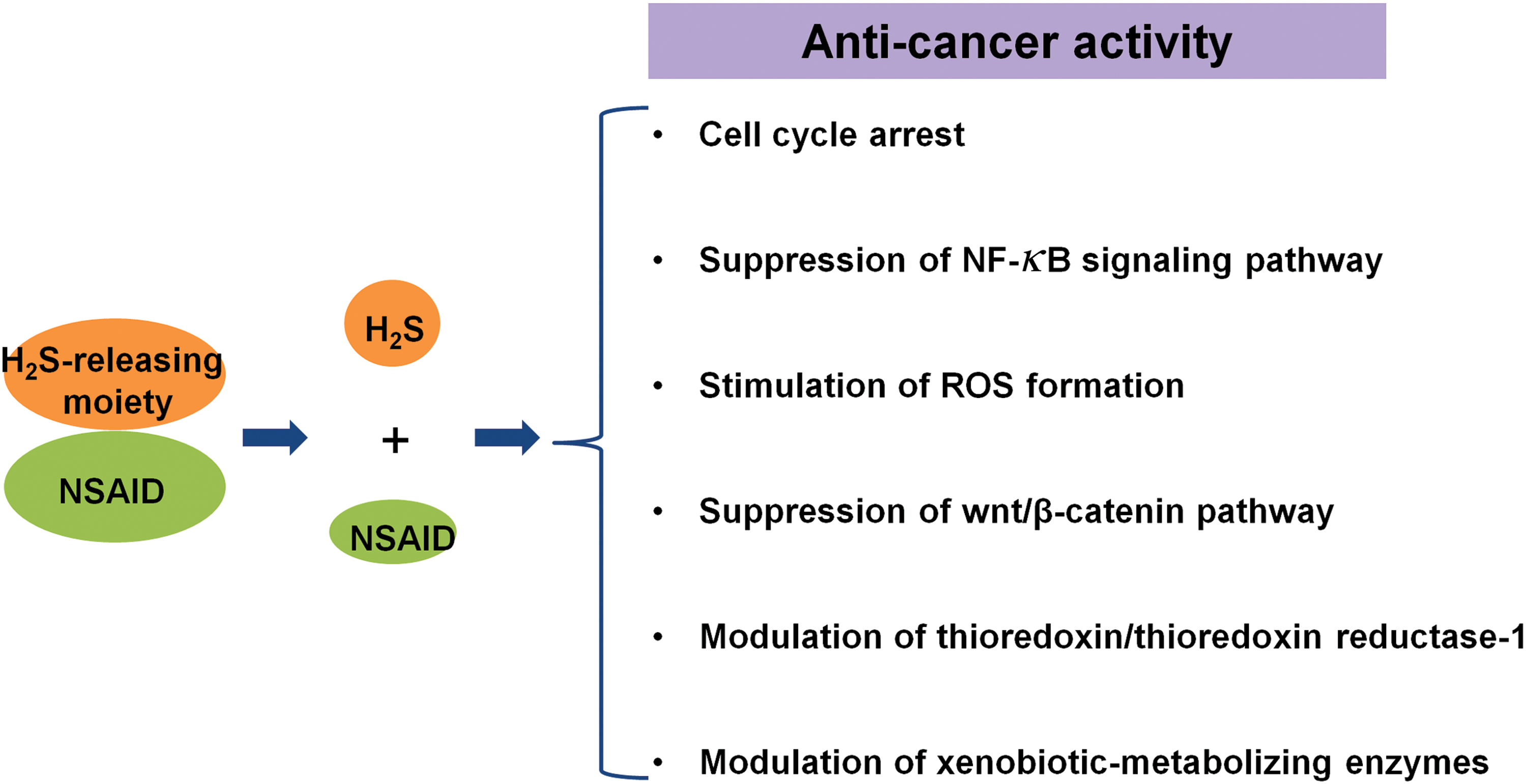
Intriguingly, the suppression of the wnt/β-catenin pathway is suggested to be the mechanism underlying the anti-cancer effect of HS-ASA in T cell acute lymphoblastic leukemia (52, 88). Other than these, modulation of thioredoxin/thioredoxin reductase-1 and/or xenobiotic-metabolizing enzymes may also contribute to the anti-cancer effect of HS-NSAIDs, as suggested by various studies (16, 50). As clearly seen from what has been described earlier, HS-NSAIDs exert their anti-cancer effect through multiple mechanisms. Nevertheless, why HS-NSAIDs possess anti-cancer activity with such a higher potency than their parental drugs is not yet revealed.
Therefore, it would be better to compare the efficacy and mechanism of action among H2S donor, NSAID, H2S donor together with NSAID, and HS-NSAID hybrid to obtain a better understanding of the possible interactions between H2S and NSAIDs in the treatment of cancer.
b. NOSH compounds as anti-cancer agents
Due to the anti-inflammatory effects of exogenous NO and H2S, it is hypothesized that compounds simultaneously donating NO and H2S may achieve higher potency in the suppression of inflammation. This class of compounds is termed NOSH compounds (51, 138). Currently, four such NOSH compounds have been reported, including NOSH-1 [(4-(3-thioxo-3H-1,2-dithiol-5-yl) phenyl 2-((4-(nitrooxy)butanoyl)oxy)benzoate)], NOSH-2 [(4-(nitrooxy)butyl (2-((4-(3-thioxo-3H-1, 2-dithiol-5-yl)phenoxy)carbonyl)phenyl))], NOSH-3 [(4-carbamothioylphenyl 2-((4-(nitrooxy)butanoyl)oxy)benzoate)], and NOSH-4 [(4-(nitrooxy)butyl 2-5-((R)-1,2-dithiolan-3-yl)pentanoyloxy)benzoate)] (Fig. 14), all of which use aspirin as a scaffold coupled with NO and H2S-releasing moieties (138). As expected, these compounds show great potential in inflammatory inhibition (128, 147, 235). Remarkably, all of the four NOSH compounds exhibit extreme effectiveness in the growth inhibition of cancer cells, including colon cancer (HT-29, HCT15, and SW480), breast cancer (MCF7, MDA MD-231, and SKBR3), T cell leukemia (Jurkat), pancreatic cancer (BxPC3 and MIAPaCa-2), prostate cancer (LNCaP), and lung cancer (A549) (138). Among these compounds, NOSH-1 was shown to be the most potent one with IC50 ranging from 48 to 240 nM for inhibition of cancer cell growth at 24 h (138).
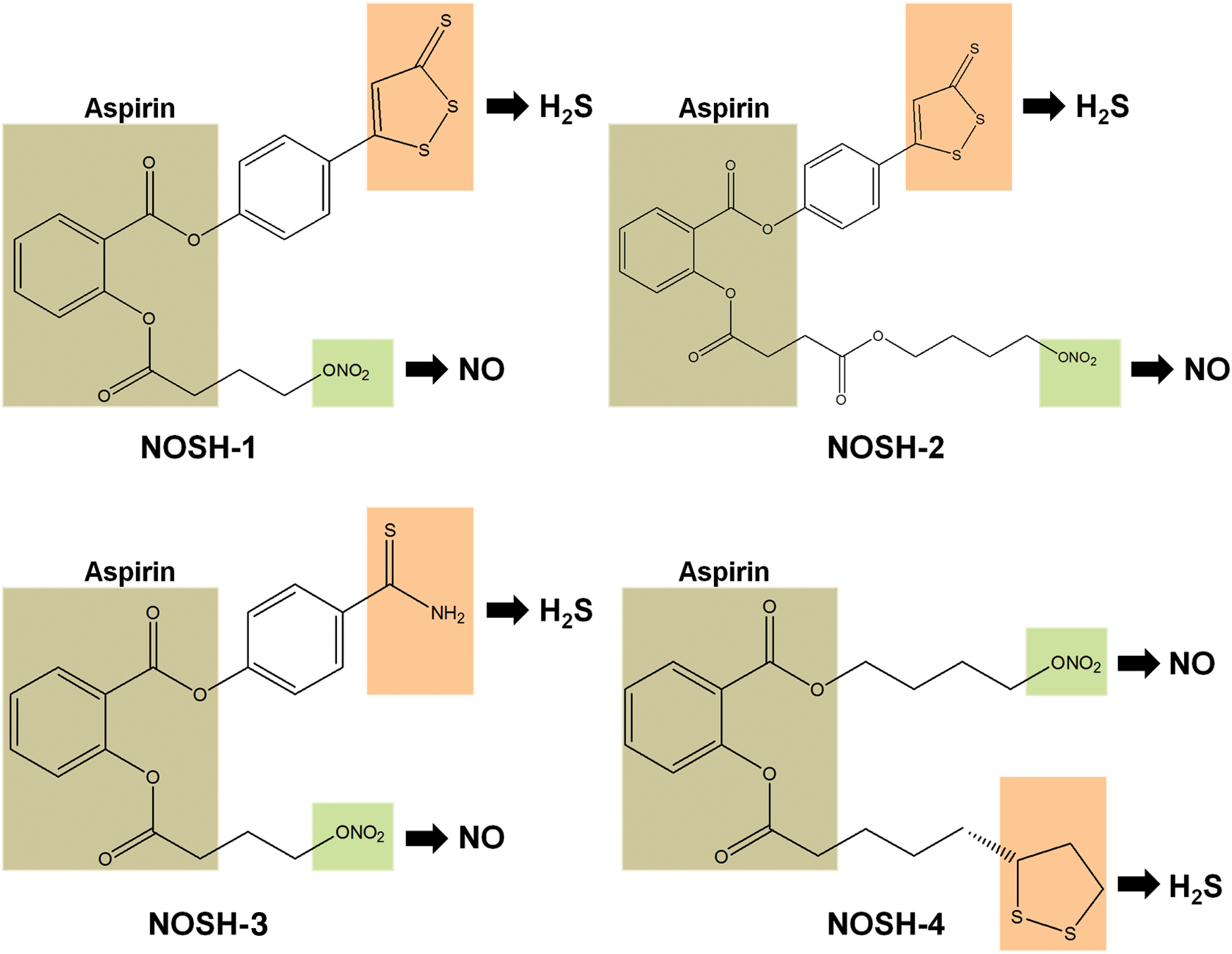
Compared with its parental ASA, the potency for inhibition of cancer cell growth is increased by 100,000-fold, 60,000-fold, 600-fold, and 16,000-fold for NOSH-1, -2, -3, and -4, respectively, in HT-29 colon cancer cells (138). Significantly, the anti-tumor effect of NOSH-1 has been demonstrated in a human colon cancer xenograft model, which shows an 85% reduction in tumor volume after treatment for 18 days (51). Although the underlying mechanisms of NOSH compounds have not well understood yet, it is highly likely that they may include the aforementioned mechanisms of HS-NSAIDs and also the interaction between NO and H2S, which needs to be tested in the future.
C. The therapeutic potential of H2S donation for cisplatin nephrotoxicity
Cisplatin is a widely used chemotherapeutic drug for cancer treatment (335). Due to the remarkable ability to treat various forms of cancers, it has gained the reputation as “the penicillin of cancer” (320). However, its effective usage for cancer therapy has been largely limited because of numerous adverse effects (81). Among these adverse effects, nephrotoxicity is the most prevalent one as evidence shows that more than 30% of patients show symptoms of acute kidney injury after the administration of cisplatin (239). This causes either discontinuation of treatment or severe renal damage. After years of efforts, some success has been made to curtail cisplatin nephrotoxicity. For instance, hydration of patients during cisplatin administration appears to reduce the occurrence after renal dysfunction (11, 57). Despite this, cisplatin nephrotoxicity remains a crucial factor compromising the use and efficacy of cisplatin in the therapy of cancer.
Very different from traditional chemotherapeutic drugs, H2S exhibits both cell protective effects and anti-cancer effects. Interestingly, H2S donation with GYY4137 inhibits cancer cell growth at doses that exhibit protective effects against oxidative stress and/or inflammation-induced cell injuries (57, 150, 155, 304). Therefore, we proposed that H2S donation may be a promising strategy for the treatment of cisplain nephrotoxicity, which is mainly caused by oxidative stress and inflammation in renal proximal tubule cells (8, 217). By this strategy, we speculate that H2S donation may alleviate cisplatin caused renal toxicity and add on more anti-cancer activity when combining with cisplatin in cancer cells. This hypothesis is partially supported by recent proof-of-concept studies that NaSH attenuates cisplatin caused renal toxicity in rat models (3, 67) whereas whether NaSH influences the anti-cancer activity of cisplatin is not studied.
However, a recent report from Liu et al. somewhat disfavors the hypothesis because they have shown that H2S slow-releasing donor GYY4137 aggravates cisplatin-induced renal damage by increasing inflammatory response (167). But several defects in using GYY4137 can be identified after clear examination of the study, such as (i) GYY4137 is prepared and stored in dimethyl sulfoxide, which has been shown to be detrimental and to accelerate GYY4137 decomposition (162, 178, 179); (ii) the study used a rather low dose of GYY4137 (21 mg/kg). It is likely that H2S might be not provided adequately considering the releasing property of H2S by the compound (178, 179); (iii) the chemical analogue of GYY4137, ZYJ1122 (150), was not used in the study, thus the possibility that the chemical backbone of GYY4137 molecule aggravated cisplatin nephrotoxicity cannot be ruled out, particularly at such a low concentration of generated H2S. As such, further studies are warranted to unclose the effects of H2S slowly releasing compounds such as GYY4137 on both cisplatin caused renal toxicity and the anti-cancer activity of cisplatin.
Besides, as discussed earlier, the recently developed H2S-releasing hybrids such as H2S-NSAIDs and NOSH-NSAIDs possess similar effects to those of H2S slowly releasing compounds but with greater potency not only in cellular protection but also in cancer inhibition. Therefore, they may have promising potential in the treatment of cisplatin nephrotoxicity while without influencing its anti-cancer activity even if not potentiating it.
VII. The Novelty and Challenges of H2S-Based Cancer Therapy
A. The novelty of H2S-based cancer therapy
Cancer is one of the main threats to human beings and tremendous efforts have been invested in the treatment of this devastating disease. Traditionally, the discovery of anti-cancer drugs was led by the identification of cytotoxic compounds. For several decades, these drugs similar to alkylating agents and nitrogen mustard have shown effectiveness against various types of tumors. However, they also induce severe adverse effects because they can hardly discriminate cancer cells and normal cells.
The recent advances in molecular sciences and genomics have recognized the genetic basis of tumorigenesis and generated potential new targets. This has led to a change in the paradigm of anticancer drug discovery toward molecularly targeted therapy (27). The paradigm shows success as indicated by the emergence of molecular targeted drugs, including imatinib, gefitinib, and bortezomib. However, several limitations appear in recent years, such as (i) cancer cells can develop resistance to these drugs; (ii) the treatment can be lost as sometimes the target changes; (iii) drugs may be difficult to be developed for some targets; (iv) one drug can hardly abolish tumor growth due to the heterogenecity of tumor populations; and (v) the drug may be not able to adequately penetrate into solid tumors. This is well reflected by the emergence of a changing paradigm by targeting multiple targets using either drug cocktails or multiple-targeted drugs, particularly for complex diseases such as cancer, diabetes, acquired immune deficiency syndrome, etc. (58, 288).
H2S is a potential therapeutic agent with multiple biological targets with unique properties. In contrast to toxic chemotherapeutic drugs, H2S possesses protective effects in multiple organs even in the doses that are able to inhibit tumor growth as shown with the slowly releasing donor GYY4137 (27, 161, 178, 179). Thus, it is likely that the existence of H2S is critical for the maintenance of cellular homeostasis, including both normal cells and cancer cells. This is supported by the evidence that H2S is essential to regulate the homeostasis of redox (322) and thiols (91). Thus, modulation of H2S may disrupt the cellular homeostasis of cancer as a whole, thereby leading to subsequent death. Perhaps due to this, H2S-based therapy has been demonstrated to be effective in multiple cancer types (91, 149, 150).
Importantly, the distinct level of endogenous H2S in cancerous and non-cancerous cells and some other reasons might afford them the different tolerances to H2S supplementation or H2S inhibition. This has been indicated by the demonstrated therapeutic window of the H2S-based approach for cancer therapy (149, 150). In addition, H2S is a small lipophilic molecule that allows it to freely penetrate into the cell membrane of all types and become biologically active (37). This may at least have two implications: (i) H2S may have a significant influence on the tumor microenvironment, which has shown to be important for tumor progression (283); (ii) it may be able to readily infiltrate into solid tumors. Compared with molecularly targeted therapy, H2S is shown to affect multiple targets in cancer cells and as a result may overcome the limitations of molecular targeted drugs mentioned earlier. Therefore, although still in its infancy, H2S-based therapy may represent a novel and unique strategy for cancer treatment.
B. The challenges of H2S-based cancer therapy
Despite the earlier mentioned merits, successful translation of H2S as a therapeutic agent still faces great challenges, particularly for cancer therapy. It is no doubt that H2S-related toxicity is always the first obstacle in the development of H2S-based drugs. Notably, promising progress has been made in the field as clearly seen from the successful transition of H2S donors such as SG-1002 and ABT-346 from phase I into phase II clinical trials (292). However, to achieve the cancer killing effect, higher doses of H2S donors are expected compared with those for other indications such as heart failure and inflammatory diseases. This may narrow the therapeutic window of these H2S donors in the context of cancer therapy, which could be an impediment for their further development.
In addition, most studies, particularly those done with H2S acute releaser NaSH or Na2S, seemed to neglect the toxic effect of these compounds themselves and failed to include normal cells to profile the therapeutic window of H2S donors when studying their anti-cancer activities (103, 317, 318, 330, 338). Therefore, the demonstration of whether and how H2S discriminates cancer cells and normal cells will surely encourage the enthusiasm to develop H2S donors for cancer therapy.
VIII. Future Directions
A. Relationship between H2S level and cancer progression
Although evidence suggests the implication of H2S in cancer development, a clear relationship between H2S level and cancer progression remains lacking. For instance, no reports have shown the level of H2S in the plasma or tumor microenvironment in cancer patients compared with normal subjects till now. Moreover, most studies have ignored the fact that modulation of H2S-producing enzymes can alter not only the level of H2S but also other byproducts such as pyruvate, glutathione, and
B. Test of drug-like H2S donors in cancer
H2S donors such as GYY4137 and SPRC have shown promising anti-tumor effects. However, their marginal potency and as-yet-unknown human toxicity may hinder their use as anti-cancer agents in humans. Recently, a batch of drug-like H2S donors have been developed and tested to be safe in clinical trials. Among them, an orally active H2S donor SG-1002 is undergoing Phase II study for heart failure; ATB-346 is being tested for osteoarthritis; ATB-352 is under the investigation for acute pain; and ATB-350 is being evaluated for thrombosis (292). It is worth mentioning that SG-1002 has been demonstrated to be safe and produced a sustained increase in both plasma H2S and plasma NO levels in humans (230). Interestingly, a promising outcome has been observed for some pediatric cancer patients as described in the patent (patent number: US8771755B2). The detailed information of such H2S donors can be found in Wallace and Wang's recent review (292). Testing of these drug-like H2S donors will not only consolidate the anti-cancer effect of H2S donation but also shed light on the translation of H2S as a therapeutic agent for human cancers.
C. Understand the molecular mechanisms underlying H2S effects
H2S elicits most of its biological effects by induction of protein persulfidation. Among these target proteins, NF-κB and Keap1/Nrf2 are closely related to cancer progression. Interestingly, endogenous H2S or H2S at low levels has been found to stimulate the translocation of p65/p50 into the nucleus (245) whereas high levels of H2S apparently inhibit the activity of NF-κB, particularly at inflammatory conditions (92, 309). This may partially explain the dual role of H2S in cancer development. The revelation and in-depth depiction about the interaction between H2S and its target proteins will surely facilitate our understanding on the role of H2S in cancer biology. On the other hand, protein cysteine persulfides may be generated by CSE in an H2S-independent manner (112), implying that CSE may modulate cancer cell survival even in the absence of changes of H2S levels in cancer cells.
D. Confirm H2S-linked persulfidation of target proteins
As reviewed earlier, several target proteins have been identified to be persulfidated and, therefore, take part in cancer development. However, the reliability of the most commonly method for the identification, namely the biotin switch assay was recently questioned because it seems that the thiol-blocking reagent S-methyl methanethiosulfonate used in the method would not differentiate free thiols and persulfides (78). Therefore, further studies are warranted to confirm H2S-linked persulfidation of target proteins by employing a more reliable method. Recently, two other persulfide detection methods, namely tag switch assay and protein persulfide detection protocol (ProPerDP) were developed by Xian and collegues (337) and Nagy and colleagues (66), respectively. Significantly, the ProPerDP method is to date the only method alkylating sulfhydral group before cell lysis that potentially avoids artificial redox post-translational modifications, including persulfidation, during lysing cells (66), representing a huge merit of this method.
E. Crosstalk of H2S with NO in cancer
For a long period, H2S and NO signaling pathways have been observed to be intimately intertwined with mutual potentiation of responses, particularly in the cardiovascular system (198). However, the crosstalk between these two gaseous transmitters remains largely unexplored in the context of cancer biology. An article from Cortese-Krott et al. has recently identified the key bioactive reaction products of NO/H2S interaction to be S/N-hybrid species, inorganic polysulfides, and nitroxyl in physiological conditions (59). Intriguingly, all these species have been shown to possess anti-tumor effects by previous studies (46, 51, 255a), indicating the potential of the crosstalk between the two gaseous transmitters in cancer biology. However, the validation of such interaction is rarely studied in the field of cancer biology. It is expected that an in-depth understanding of such interaction may not only facilitate our understanding on tumorigenesis but also lead to novel strategies for cancer treatment.
F. Inorganic polysulfide accounts for the anti-cancer effect of H2S?
H2S is a very short-lived molecule; however, numerous studies have shown the long-lasting biological effects of H2S in mammalian systems. Therefore, it is reasonable to speculate whether the formation of certain more stable substance(s) accounts for H2S-derived effects. Kimura and colleagues have recently identified inorganic polysulfides in the solution of NaSH (114). Intriguingly, compared with NaSH, the polysufides (Na2S2, Na2S3, and Na2S4) elicit much stronger effects on the activation of TRPA1 in rat astrocytes (214) and activation of Keap1/Nrf2 in neuronal cells (40, 214). The existence of endogenous inorganic polysulfides has also been demonstrated by the group (136). Moreover, garlic-derived polysufides have long been well known to possess anti-tumor effects (192). Therefore, it will be of great value to evaluate whether inorganic polysulfides are the active molecules derived from H2S and display the anti-tumor effect.
G. A new regulatory circuit of thioredoxin and H2S by controlling persulfidation in cancer?
Thioredoxin, a small redox-regulating protein, has been found to be highly expressed in multiple cancers and plays important roles in cancer cell survival (126). Notably, two separate studies recently showed that thioredoxin acts as depersulfidase and, therefore, may control H2S signaling by reducing protein persulfidation and regenerating H2S (66, 302). Interestingly, H2S was known to upregulate the expression of thioredoxin by activating the Keap 1–Nrf2 pathway (38). Moreover, the fourth interaction mediated by H2S was shown to reduce the disulfide bonds of proteins (220, 289), coincidently, an event leading to thioredoxin activation (126), suggesting a possibility of direct interaction between H2S and thioredoxin. Altogether, this evidence implies a likelihood that thioredoxin and H2S may work coordinately to control persulfidation in cancer cells and therefore their survival and progression.
H. H2S-mediated immune cell regulation in cancer progression and therapy
The crosstalk between immune cells and cancer cells has gained great attention due to the impressive clinical benefits of restoration of tumor-suppressed immune function. Currently, it has been shown that H2S can regulate the physiological function of immune cells. For example, Roberts and colleagues (183, 184) found that H2S is an endogenous potentiator of T cell activation. Recently, Yang et al. (330) showed that H2S deficiency by knocking out CBS led to systemic autoimmune disease due to regulatory T (Treg) cell deficiency, suggesting that H2S takes part in the regulation of Treg-cell-associated immune homeostasis. Despite this, the studies of H2S-mediated immune cell regulation remain lacking in the context of cancer progression and therapy.
IX. Concluding Remarks
On recognition to be the third gaseous transmitter, the image of H2S is expanding in mammalian systems. In the past decade, numerous studies demonstrated complicated roles of H2S in the context of cancer development. Based on the published evidence, bell-shaped models have been proposed to explain the role of H2S in cancer development. Specifically, endogenous H2S or a relatively low level of exogenous H2S may exhibit pro-cancer effects, whereas exposure to H2S for long period or a high amount may lead to cancer cell death. However, one should bear in mind that the consequence of CSE or 3MST inhibition in cancer cell is still largely unexplored, which needs to be examined in the future. Nevertheless, the models, indeed, have been favored by most studies in the field.
Remarkably, H2S is not alone in the regulation of cancer biology, because it intertwines with the other two gaso-transmitters such as NO and CO. Better portrayal of the crosstalk among these gaseous transmitters may not only lead to an in-depth understanding of cancer progression but also shed light on novel strategies for cancer therapy. Recently, the developments of CBS inhibitors and H2S donors have gained enthusiasm because of their potential value as novel anti-cancer therapeutics. More selective CBS inhibitors compared with traditionally used AOAA are now available and some of them have shown anti-tumor activity. Moreover, H2S-releasing hybrids, especially HS-NSAIDs and HSNO compounds, have shown very promising anti-tumor activity with marginal toxicity, indicating their value for cancer therapy. Nonetheless, the development and application of H2S-based therapy for cancer treatment is still in its infancy. In the future, more studies are still warranted to identify H2S-targeted signaling molecules and develop better compounds with higher selectivity and potency.
Footnotes
Acknowledgments
This work was supported by Ministry of Education of Singapore Tier 2 Research grant (MOE2017-T2-2-029), NMRC (CIRG/1363/2013 and CIRG1432/2015), and Medical Research Council UK (MR/M022706/1).
