Abstract
Significance:
The incidence of cardiovascular events (CVEs) increases with age, representing the main cause of death in an elderly population. Aging is associated with overproduction of reactive oxygen species (ROS), which may affect clotting and platelet activation, and impair endothelial function, thus predisposing elderly patients to thrombotic complications.
Recent Advances:
There is increasing evidence to suggest that aging is associated with an imbalance between oxidative stress and antioxidant status. Thus, upregulation of ROS-producing enzymes such as nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and myeloperoxidase, along with downregulation of antioxidant enzymes, such as superoxide dismutase and glutathione peroxidase, occurs during aging. This imbalance may predispose to thrombosis by enhancing platelet and clotting activation and eliciting endothelial dysfunction. Recently, gut-derived products, such as trimethylamine N-oxide (TMAO) and lipopolysaccharide, are emerging as novel atherosclerotic risk factors, and gut microbiota composition has been shown to change by aging, and may concur with the increased cardiovascular risk in the elderly.
Critical Issues:
Antioxidant treatment is ineffective in patients at risk or with cardiovascular disease. Further, anti-thrombotic treatment seems to work less in the elderly population.
Future Directions:
Interventional trials with antioxidants targeting enzymes implicated in aging-related atherothrombosis are warranted to explore whether modulation of redox status is effective in lowering CVEs in the elderly. Antioxid. Redox Signal. 27, 1083–1124.
Table of Contents
I. Introduction
C
Progressive development of the atherosclerotic process during aging and its late thrombotic complications are two key phases contributing to artery occlusion in coronary and cerebral districts. Among factors predisposing to MI and stroke, platelets play a pivotal role as they contribute to acute artery occlusion occurring at the site of plaque rupture/erosion by adhering to the exposed sub-endothelium. Then, they concur to thrombus growth via platelet aggregation propagation (75). Clotting system activation and endothelial dysfunction are other important mechanisms implicated in the occurrence of CVD, as they represent a backbone for both artery and venous thrombosis (76). Experimental and clinical studies documented a progressive increase of platelet function, clotting activation, and endothelial dysfunction in the elderly population, supporting their role in the progression of atherothrombosis.
Another age-related process is the imbalance between oxidative stress and antioxidant status. Thus, an increased function of the enzymes responsible for the production of reactive oxygen species (ROS) by aging was reported, along with a parallel decrease of antioxidant pathways, ultimately leading to a pro-oxidant phenotype in elderly subjects. These modifications may also negatively influence platelet and clotting activation and endothelial function, eventually concurring to cardiovascular complications.
In this comprehensive review, we analyzed the mechanisms of atherothrombosis in the aging process focusing on: (i) imbalance between oxidative stress and antioxidant status as factors favoring atherosclerosis and thrombosis in an elderly population, (ii) alterations in platelet and clotting activation and endothelial function in relation to oxidative stress modifications by aging, and (iii) current and future therapeutic antioxidant and anti-thrombotic strategies in the elderly.
II. Oxidative Stress and Aging
The main biological consequence of aging is a functional decline in cells, tissues, and organs functions (319). Under normal conditions, cells can go through a limited number of divisions on reaching the end of their replicative lifespan (137). This phenomenon is defined as replicative senescence (RS) and largely depends on alterations in DNA replication that eventually affect chromosomal stability and genome function (187). The RS is mediated by several signaling cascades that are linked to the activation of tumor suppressing proteins, such as p53/p21, and results in shortening of telomeres (316) that are located at chromosome ends to prevent DNA damage (316). Moreover, cell exposition to different agents damaging DNA, such as ROS, UVA, and UVB, results in reduced mitotic ability and increased signs of senescence. This second phenomenon is termed as stress-induced premature senescence (SIPS) (316).
Thus, the aging process is the result of physiological RS and external SIPS that concur together in accelerating the natural progression of aging (187). In accordance, senescent markers, including Discoidin Domain Receptor family member 1 kinases, senescence-associated β-galactosidase, p53/p21, and telomere dysfunction, were detected in various tissues from elderly individuals (316).
Although a unique comprehensive theory for the initiation and progression of cell senescence and aging is far from being elaborated, several systems, including mitochondrial dysfunction, protein glycation, deregulation of immune system, hormonal changes, gene modifications, dysfunction telomere attrition, and redox stress, have been identified so far (111).
Oxidative damage represents the most well-documented subset among aging-associated damage. Although its impact on cellular function is only one of the proposed mechanisms of senescence, it seems to be an attractive one as it connects several different mechanisms such as modifications in the regulation of gene expression and mitochondrial dysfunction (50, 122).
The theory of ROS overproduction as the main mechanism involved in aging by inducing cumulative damage was first proposed in 1956 (135). According to this hypothesis, an increase in pro-oxidant pathways would promote the aging process, which conversely would be delayed by an improvement in antioxidant defenses.
The validity of this hypothesis was explored in experimental models where the effect of endogenous antioxidant enzymes, such as superoxide dismutase (SOD), catalase, glutathione peroxidase (GPx), and thioredoxin, was investigated. An increase in lifespan was observed in flies overexpressing copper-zinc SOD (CuZnSOD), an enzyme that converts superoxide anion (O2 −) into hydrogen peroxide (H2O2), and catalase, which changes H2O2 into water (262, 363). Moreover, reduced levels of GPx, an enzyme that reduces lipid hydroperoxides to their corresponding alcohols and H2O2 to water, and of thioredoxin that possesses a similar activity, were observed in an animal model of aging (64). Transgenic and knockout mouse models of antioxidants provided conflicting evidence (328). For example, knockout mice models of GPx1 and SOD were associated to reduced lifespan (79, 96), whereas transgenic mice overexpressing SOD and catalase showed an unmodified lifespan (275).
Growing evidence suggests a role for pro-oxidant systems in the aging process. Pro-oxidant enzymes producing ROS, such as nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (Nox), myeloperoxidase (MPO), and uncoupled nitric oxide synthase (NOS), mediate important biological functions by regulating the activity of several intracellular pathways involved in cell growth, apoptosis, survival, metabolism, and migration, all of which are altered during the aging process.
Among ROS, O2 − plays a fundamental role, as it reacts with nitric oxide (NO), thus lowering its activity and/or concentration (199). Reduced NO bioavailability influences migration and proliferation of vascular smooth muscle cells (VSMCs), which is one of the early atherosclerotic changes (199). Moreover, O2 − upregulates the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), which, in turn, induces the production of atherogenic cytokines such as tumor necrosis factor α (TNFα), interleukin 6, monocyte chemoattractant protein (MCP)-1, and adhesion molecules (254).
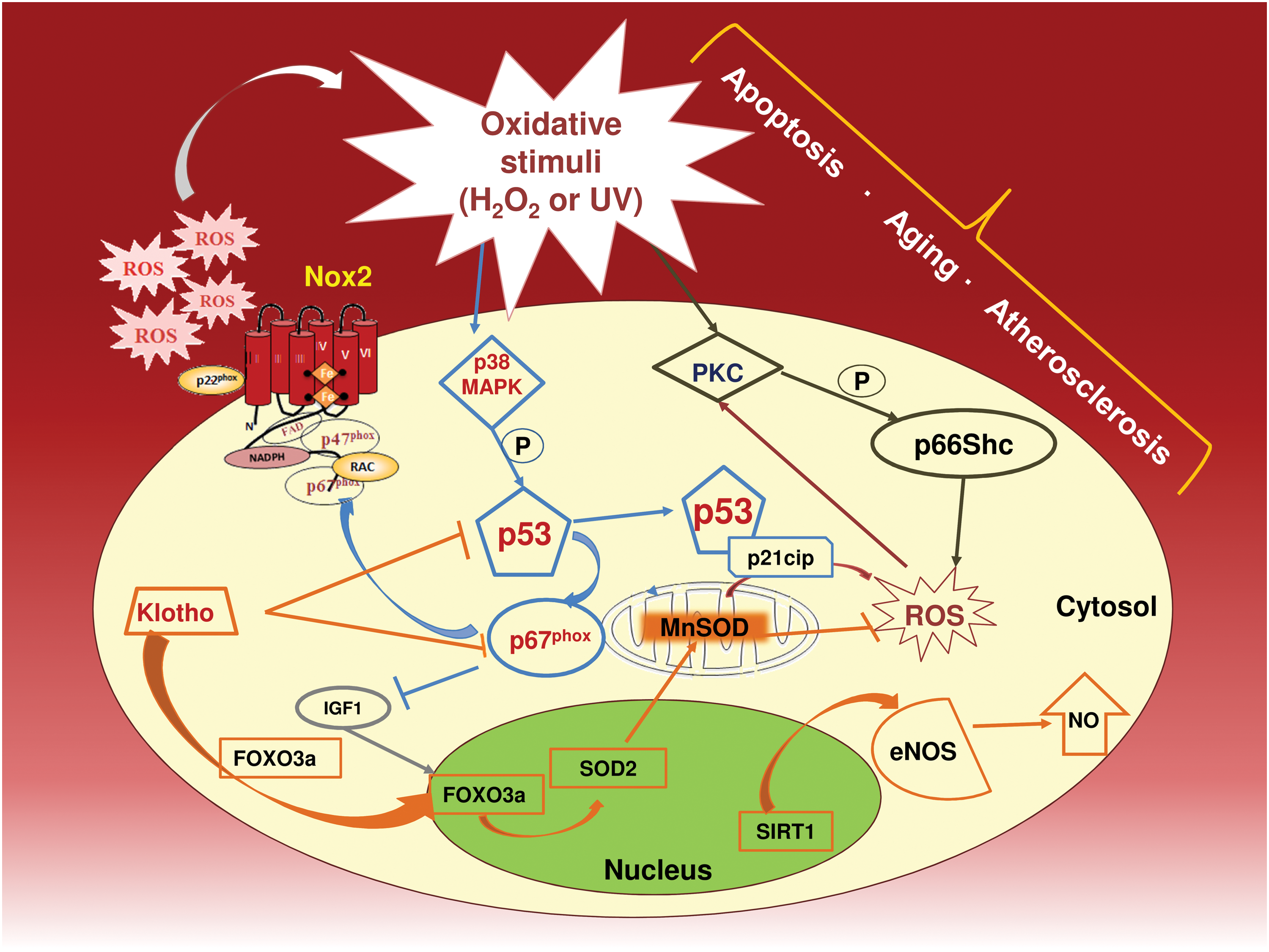
A specific interplay between ROS, gene transcription, and subsequent signal transduction seems to be implicated in aging. Thus, ROS induce phosphorylation of the redox-sensitive transcription factor p53 by activating p38 mitogen-activated protein kinase (MAPK) (34) and Polo-like kinase 3 (409). The p53 factor enhances ROS signaling, presumably via upregulation of the p67phox subunit of the Nox system (154). Once activated, p53 is able to modulate RS by controlling some specific genes involved in cell cycle arrest. The role of p53 in premature aging has been demonstrated by several mouse models where persistent p53 activation promoted senescence or irreversible cell cycle arrest (372). Interestingly, human endothelial cells undergo p53/p21cip-dependent cell cycle arrest via Nox2-derived O2 − production (201).
Other oxidative-linked effectors of longevity, such as Klotho gene, may influence aging-related atherosclerotic process. Thus, Klotho-null mice phenotypes display similarities with premature human aging, including accelerated atherosclerosis (188). Once activated, Klotho protein induces MnSOD and SOD2 biosynthesis, thereby increasing the antioxidant cell defense (176, 303, 412). Moreover, Klotho influences the intracellular signaling pathways involved in oxidative stress responses and aging via inhibition of p53/p21cip (81).
The gene regulator histone deacetylase sirtuin (SIRT) is also implicated in the aging process. SIRT is present in seven isoforms and encompasses a nicotinamide adenine dinucleotide-dependent enzymatic activity associated with aging (92). In particular, the isoform SIRT1 is localized in the nucleus and plays an important role in preserving vascular health; Sirt1-Tg/ApoE2/2 mice showed upregulated endothelial NOS (eNOS) activity and reduced atherosclerotic plaque formation as compared with wild type (421). In support of this, treatment of ApoE2/2 mice with a specific SIRT1 activator resulted in a significant reduction of oxidized low-density lipoprotein (ox-LDL) concentration and plaque formation (350).
ROS are also involved in the activation of mitochondrial-mediated patterns that are implicated in reducing lifespan and accelerating atherosclerosis progression. Thus, ROS elicit mitochondrial adaptor protein p66shc phosphorylation, which, in turn, regulates intracellular pathways that are involved in ROS production and apoptosis (236). In particular, H2O2 induces serine phosphorylation on p66shc through protein kinase C, resulting in increased apoptosis (290). Thus, animals on a chronic high-fat diet disclosed an aortic cumulative early lesion area by ∼21% in wild-type mice and only by 3% in p66shc(−/−) mice. Further, in p66shc(−/−) mice, a significant reduction of systemic and tissue oxidative stress along with a 30% prolongation in lifespan was observed (236, 253).
A synthetic scheme of the mechanisms reported earlier is reported in Figure 1.
III. Atherothrombosis in Elderly: Clinical Studies
Observational studies in the general population or in patients with CVD documented a progressive increased incidence of acute vascular events by aging (90). In the elderly, a higher prevalence of atherosclerotic risk factors, including arterial hypertension, diabetes mellitus (DM), and metabolic syndrome (MetS), might partly account for this phenomenon (50). Of note is that there is no standardized definition of “old population,” as changes occurring by aging are not linear and often depend on several factors, including environment, individual behaviors, social position, and ethnicity. The “WHO World report on ageing and health” states that “by age 60, the major burdens of disability and death arise from age-related losses in hearing, seeing and moving, and non-communicable diseases, including heart disease, stroke, chronic respiratory disorders, cancer and dementia” (26). However, chronological age with a cut-off set at 65 and 75 years is the most used method to define old age, but the need for “multidimensional/functional” definition remains (341).
A. Hypertension
The prevalence of hypertension increases with age, ranging from 7.3% in people aged 18–39 years, to 65.0% among those ≥60 years in the United States (257). Further, about one third of U.S. adults aged ≥80 years are treated with ≥3 classes of antihypertensive medication (247), suggesting a more severe phenotype of hypertension in elderly patients.
B. Diabetes mellitus
The overall prevalence of DM in the United States is progressively increasing from 8.4% (1988–1994) to 12.1% (2005–2010). The corresponding figures for subjects aged ≥65 years are 18.6% and 28.5%, respectively (62). Similarly, in European countries, DM prevalence is 10% and 15–20% in subjects <60 and >70 years, respectively (11, 126).
C. Metabolic syndrome
MetS is characterized by the coexistence of at least three of the following atherosclerotic risk factors: (i) central obesity, (ii) atherogenic dyslipidemia (i.e., low high-density lipoprotein [HDL] and high triglycerides), (iii) hypertension, and (iv) elevated fasting glucose (4). The prevalence of MetS among U.S. men ranges from 20.3% in subjects 20–39 years of age to 51.5% for subjects ≥60 years of age in the NHANES 2003–2006. Among U.S. women, prevalence ranges from 15.6% in subjects 20–39 years of age to 54.4% for those ≥60 years of age (98).
D. Atrial fibrillation
Aging is associated with an increased prevalence and incidence of atrial fibrillation (AF) that is expected to increase in the next years worldwide. Age is not only a predictor of AF but also an established risk factor for CVD and cerebrovascular disease (CD) (266, 267, 343, 386) in patients with paroxysmal or persistent/permanent AF. Thus, by the seventh decade of life, the incidence of athero-thromboembolism in AF patients sharply increases compared with the younger population (205).
E. Peripheral artery disease
Peripheral artery disease (PAD) is an uncommon finding in people <50 years, but its prevalence sharply rises with aging. In the Heinz Nixdorf recall study, which included 4814 subjects, the prevalence of symptomatic/asymptomatic PAD in men aged 45–49 years was 3.0%, rising to 18.2% in those aged 70–75 years. In women, the percentage rose from 2.7% to 10.8% in the same age categories (181). Similarly, in the Framingham Heart Study, the incidence of intermittent claudication rose from 0.4 per 1000 in men aged 35–45 years to 6.0 per 1000 in those aged ≥65 years (170).
F. Interaction between cardiovascular risk factors in the elderly
The presence of multiple risk factors is common in elderly patients (403) and contributes to the aging-related atherothrombosis burden. The prevalence of multimorbidity, defined as ≥2 concomitant chronic diseases, significantly increases by aging and may be detected in >70% of patients ≥75 years (28). For instance, DM is associated with other atherosclerotic risk factors; in particular, hypertension, elevated LDL, and obesity may coexist in 75–85%, 70–80%, and 60–70% of diabetic patients, respectively (152). The cumulative risk of experiencing a cardiovascular outcome in multimorbidity patients overcomes the risk conferred by each condition alone (97). Taking into account the frequent combination of multiple risk factors in the elderly, this could be considered one of the important factors contributing to the high risk of CVD in the elderly (129).
IV. Gut Microbiota and Atherothrombosis
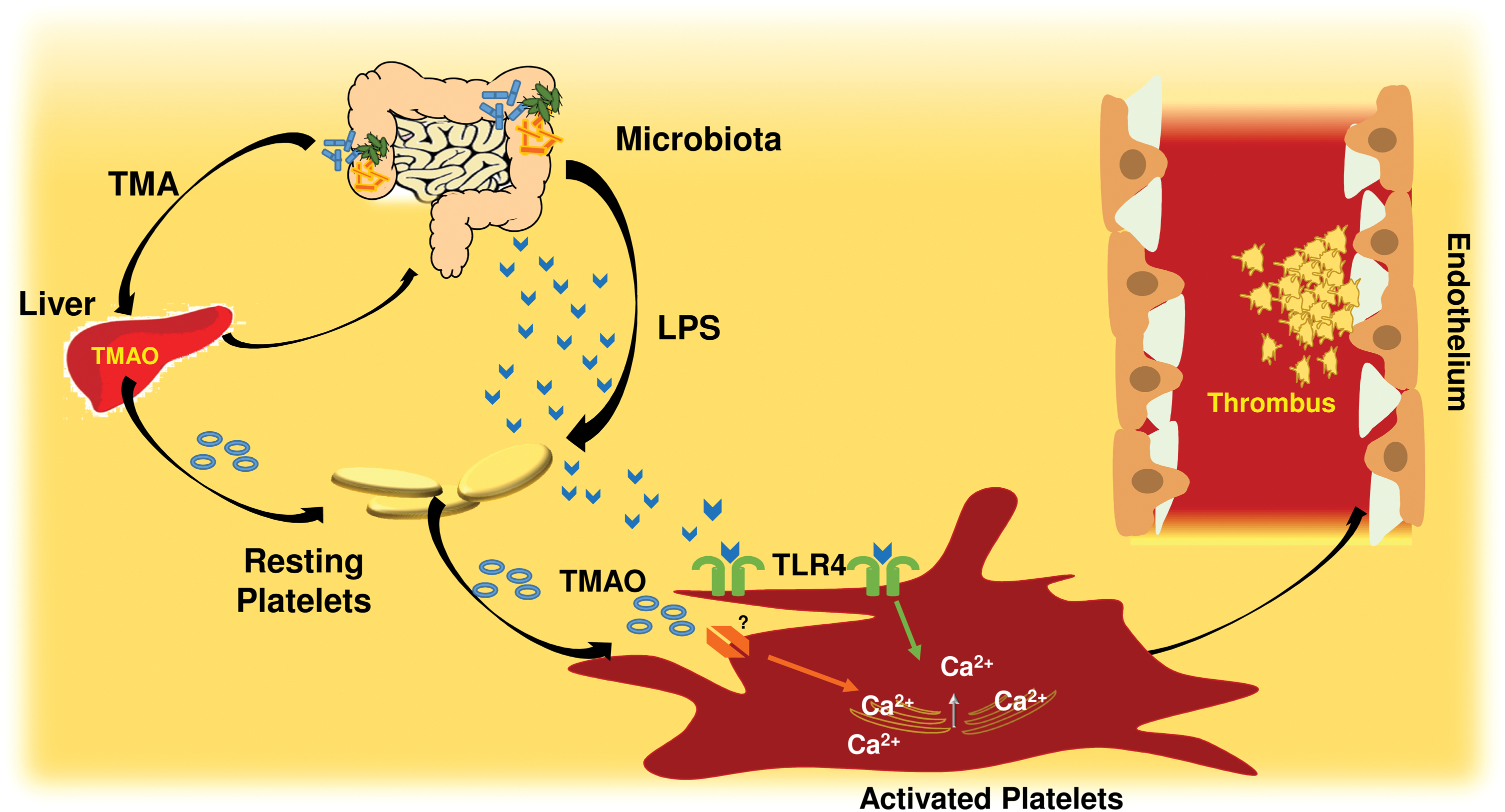
Gut microbiota is emerging as a novel player in the process of atherosclerosis as it is implicated in the development of atherosclerotic risk factors such as diabetes and hypertension (166). However, there is also mounting evidence that products of intestinal microbiota may cross the intestinal barrier, reach the circulatory system, and directly contribute to atherothrombosis. For instance, recent studies on this topic discovered that intestinal microbiota produces trimethylamine N-oxide (TMAO), which may be implicated in the process of atherosclerosis and thrombosis (300, 360).
Other products of gut microbiota such as lipopolysaccharide (LPS) may concur to atherosclerosis, possibly via chronic inflammation and thrombosis (235). About 100 trillion of gut bacteria contribute to an enteric reservoir of >1 g of LPS, which can be found in human circulation from healthy subjects in a range of 15–200 pg/ml (313). A recent prospective study showed that circulating LPS was predictive of cardiovascular events (CVEs) in a population affected by AF (265).
An important but not-yet-solved issue is whether changes of gut microbiota occurring with aging may be potentially implicated in atherothrombosis. Indeed, from newborns to elderly gut, microbiota is characterized by continuous modifications, which alter its metabolism. From a phase of instability and low complexity, which characterizes newborns until the age of 3 years, gut microbiota gets more stable in adults with essentially saccharolytic bacteria activity (317). Conversely, in the elderly population, gut microbiota gets again unstable with an activity shifted toward a prevalent proteolytic metabolism. It is unclear, however, whether these changes are due to different dietary patterns or to intrinsic changes of gut microbiota of the elderly population. Whatever is the mechanism, it would be interesting to appreciate whether in the elderly gut microbiota is more prone to deliver metabolic molecules, such as TMAO or LPS, which are implicated in atherothrombosis (Fig. 2).
V. Atherosclerosis and Oxidative Stress in Animals and Humans
Retention and accumulation of LDL particles within the vessel wall is a critical step of the early atherosclerotic process. In fact, LDL accumulation causes migration of inflammatory cells such as monocytes/macrophages, which then uptake and oxidize LDL via an oxidative stress-mediated mechanism (109). In this regard, kinetic analysis of LDL across the artery wall demonstrated that retention of LDLs in the sub-intima space depends on LDL permeability and on the ability of LDL to leave (efflux) the artery wall (358). Concentration of LDL in the circulation is likely to be an important element contributing to LDL permeability and degradation in the artery wall. Bartels et al. (21) studied this issue in cholesterol-fed, LDL receptor (LDLR)-deficient mice treated with an anti-ApoB antisense oligonucleotide versus mismatch control antisense oligonucleotide for 1–4 weeks before an injection of iodinated LDL particles. Animals treated with an anti-ApoB antisense oligonucleotide showed ∼90% reduction of plasma LDL, which was associated with 50% and 85% reduction of aortic permeability and degradation, respectively, of newly entered LDL particles after 1 week of treatment. Conversely, 4 weeks of treatment were necessary to observe a reduction in foam cell content, plaque size, and aortic LDL pool size. Interestingly, plasma LDL cholesterol lowering was associated with 70% reduction of sub-luminal foam cells and ∼90% reduction of messenger RNA (mRNA) expression of inflammatory genes. The enhanced permeability of LDL into atherosclerotic plaque is almost evident in the elderly population with established atherosclerosis. This was documented by an injection of iodinated autologous LDL in elderly patients 24 h before undergoing endoarterectomy for critical carotid stenosis. The analysis of carotid specimens demonstrated that LDL localized into macrophages and that this phenomenon was prevented by pre-treatment with vitamin E, suggesting that LDL is rapidly uptaken and oxidized by foam cells of atherosclerotic plaque (155).
Chronic deposition and accumulation of LDL causes an injury response, which results in the recruitment of macrophages, dendritic cells, and lymphocytes at the site of atherosclerotic lesion. As for sterile inflammation, this process may undergo resolution with classical tissue repair or, in case of defective resolution, progress to advanced lesion (330). Advanced atherosclerotic plaque is characterized by a central necrotic core, composed by macrophages and VSMC. Impaired clearance of necrotic cells, a process called efferocytosis, causes accumulation of inflammatory material that further exacerbates atherosclerotic lesion, eventually leading to its rupture or erosion (358). Persistent inflammatory stimulus due to continuous accumulation of LDL in the sub-intima coupled with impaired efferocytosis and presence of an inflammatory phenotypes leads to progression of inflammation and the atherosclerotic process (358).
In this context, oxidative stress has an important role as it is a stimulus for further macrophage accumulation and activation, and for the production of oxidant products that perpetuate tissue damage (345). For instance, isoprostanes, which were found in human atherosclerotic plaque (294), induce mitogenesis of VSMCs, proliferation of fibroblasts and endothelial cells, and overexpression of endothelin 1 in mouse aortic endothelial cells (293). Moreover, blockade of thromboxane (Tx) receptor improves the anti-atherogenic effect of Tx inhibition in LDLR-deficient mice (71). Other effects of oxidative stress include expression of adhesion molecules such as vascular cell adhesion molecule (VCAM)-1, intercellular adhesion molecule (ICAM)-1 and E-selectin, and the MCP-1, which promote monocyte adhesion, migration, and accumulation in the sub-endothelium (68) and activation of inflammatory cytokines such as TNFα. In this regard, it is interesting that in p47phox knockout mice, TNFα failed to induce expression of ICAM-1 in coronary microvascular endothelial cells (200). In other studies, ROS elicited expression of VCAM-1, which serves as a scaffold for leukocyte migration and a trigger for endothelial signaling via inducing Nox2 activation (67). Oxidative stress is closely related to activation of pro-oxidant pathways such as Nox, MPO, uncoupled eNOS, and lipoxygenases (LOXs) and is counteracted by antioxidant enzymes, including SOD, catalase, GPx, paraoxonase (PON), and NOS. These endogenous antioxidants protect against athero-genesis by scavenging ROS, facilitating endothelium-dependent vasorelaxation, inhibiting inflammatory cell adhesion to the endothelium, and altering vascular cellular responses, such as VSMC and endothelial cell apoptosis, VSMC proliferation, hypertrophy, and migration (114) (Table 1 and Fig. 3).
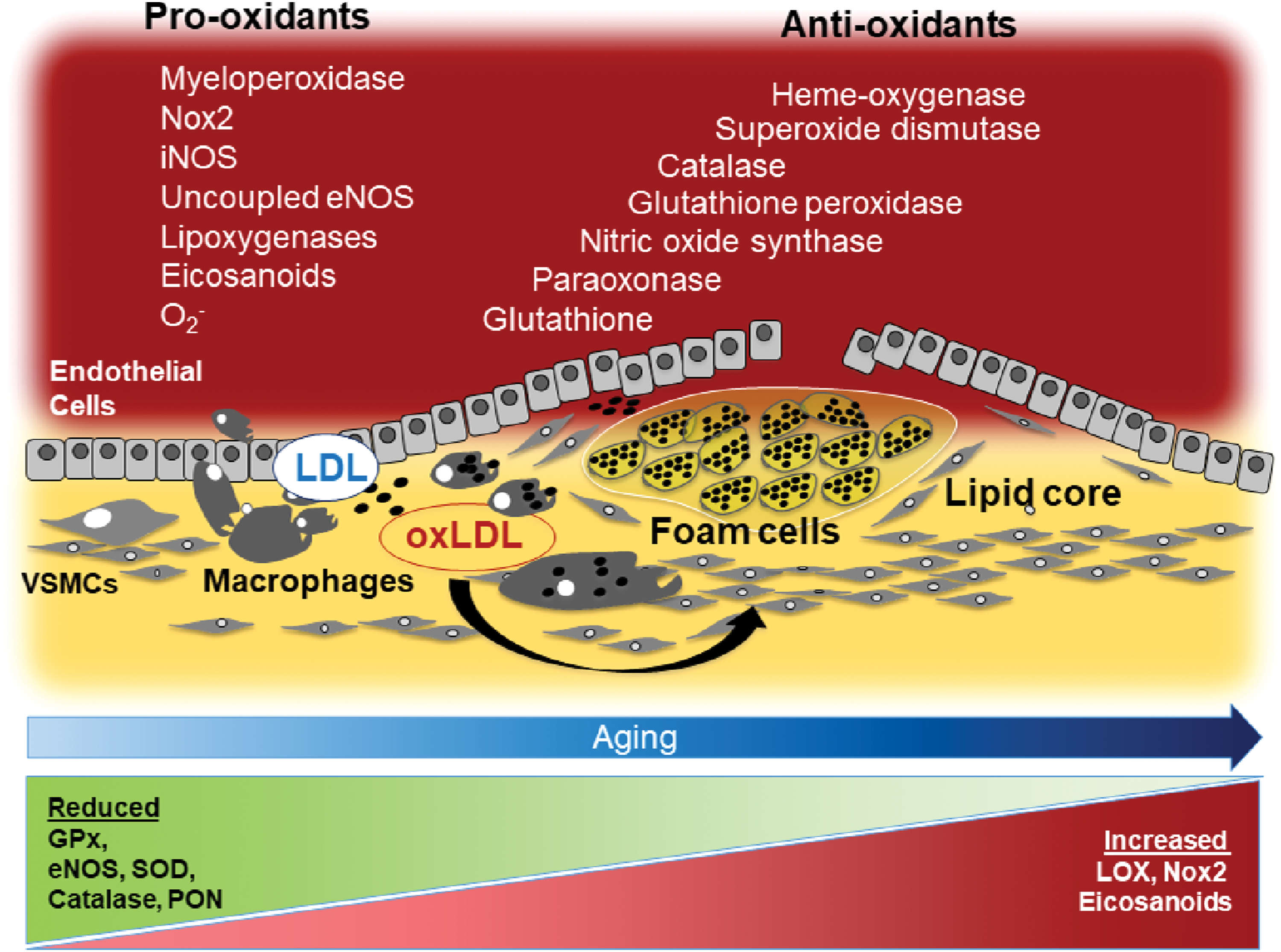
eNOS, endothelial NOS; GPx, glutathione peroxidase; iNOS, inducible NOS; LOX, lipoxygenase; MPO, myeloperoxidase; NADPH, nicotinamide adenine dinucleotide phosphate; nNOS, neuronal NOS; NO, nitric oxide; Nox, NADPH oxidase; PON, paraoxonase; ROS, reactive oxygen species; SOD, superoxide dismutase.
Defective tissue repair with impaired damage resolution is a relevant key element for atherosclerotic progression that is not yet poorly understood (124, 171). Clearance of inflammatory cells and necrotic materials along with vascular regeneration are hampered in atherosclerosis and, more in particular, in aging-related atherosclerosis. Lesion resolution depends on several factors, including formation of (i) bioactive lipids such as lipoxins, resolvins, protectins, and maresins, called specialized pro-resolvin proteins; (ii) proteins such as interleukin 10, transforming growth factor-beta, and annexin A1; and (iii) bioactive gas such as NO, hydrogen sulfide (H2S), and carbon monoxide (357). The presence of cell phenotypes with resolving capacity such as regulatory T cells and resolving-type macrophages is also relevant (357).
An important role in vascular tissue repair seems to be played by mononuclear cells with a specific phenotype, namely progenitor cells (PCs), which originate primarily from bone marrow (BM) and differentiate into hematopoietic stem cells, including endothelial progenitor cells (EPCs). Several age-related modifications of BM have been described (311). Thus, the hematopoietic active BM (red BM) gradually undergoes adipose replacement (yellow BM), becoming less active during the aging process. Also, BM cellularity is strictly correlated with age (123), together with a reduced availability and ability to migrate of EPCs, and possibly other BM cells that are necessary for successful arterial repair in the elderly (218). Thus, in 123 women with nonobstructive coronary artery disease (CAD), the number of circulating PC (CD34+, CD34+/CD133+, and CD34+/CXCR4+) was inversely correlated with age (233).
Indirect evidence of the potential role played by PC in vascular disease has been provided by Hill et al., who showed lower circulating PC in patients at risk of CVD with an inverse correlation between PC and artery dysfunction (144). Other reports in patients at risk of CVD supported these findings and also underscored that PC lowering is more marked in an elderly population with concomitant atherosclerotic risk factors than in apparently healthy subjects in whom such PC decline by aging is not evident (136). Experimental studies are in agreement with this negative interplay between PC and aging as depicted by experiments reporting lower BM-derived PC in aging ApoE−/− mice, along with a positive impact of chronic treatment with BM-derived PC in the atherosclerosis progression (301). However, the clinical relevance of these findings in human atherosclerosis is still speculative and needs to be further investigated. In particular, prospective studies are necessary to assess whether BM-derived PC are actually decreasing by aging independently or not by classic atherosclerotic risk factors, and their potential role to halt atherosclerotic progression.
In contrast with this hypothesis, a recent work showed that clonal hematopoiesis of indeterminate potential (CHIP), which is a common aging-related phenomenon characterized by the presence of an expanded somatic blood-cell clone in people without hematologic disease, which may lead to an increase in circulating myeloid cells, was significantly associated with the risk of coronary events in 4726 participants with coronary heart disease (CHD) and 3529 controls, and with accelerated atherogenesis in a murine model of atherosclerosis (159).
VI. Pro-Oxidant Pathways
A. Myeloperoxidase
MPO is an enzyme belonging to the mammalian heme peroxidase superfamily, which is detectable in neutrophils, monocytes, and macrophages. MPO produces various compounds with pro-oxidant properties, such as hypochlorous acid, chloramine, tyrosyl radicals, and nitrogen dioxide, contributing to oxidative stress by oxidizing LDL and lowering NO activity and biosynthesis (57, 423). Moreover, MPO activation generates eicosanoids and bioactive lipids and atherogenic forms of both LDL and HDL. Indeed, studies using MPO knockout mice demonstrated that this enzyme plays an important role in the formation of products derived from the oxidation of arachidonic acid (AA) that are involved in the inflammatory response (423) and in lipid peroxidation (38, 423).
Although the role of MPO as a pro-oxidant and pro-inflammatory enzyme is well demonstrated, its contribution to atheroma progression is a matter of debate. In fact, in ApoE(−/−) mice, genetic deletion of MPO had no impact on atherosclerotic lesion (37), whereas specimens from human aortic plaques expressed increased MPO (219) and MPO-derived compounds (404). Interestingly, “humanized” MPO/atherosclerosis animal models demonstrated an accelerated plaque development in LDLR(−/−) transgenic mice expressing human MPO-463G/A alleles (58, 396).
Clinical studies performed in patients with CHD (143, 234, 361, 422) or PAD (6, 40) showed a predictive role for elevated MPO. Conversely, few data regarding its role in aging have been reported. Son et al. found that in rats MPO activity increases by 70% and 85% at 18 and 24 months, respectively, compared with the 6-month-old rats (344). MPO was also more elevated in cardiac tissue after 24 h from myocardial injury in old compared with young rats (101). Data from the prospective cohort study Aging and Longevity Study in the Sirente Geographic Area (SIRENTE Study), which included 363 people aged ≥80 years, showed an enhanced mortality risk in individuals in the highest MPO tertile as compared with the lowest one (145).
A recent study in patients with acute MI reported an increased MPO activity along with a reduced glutathione (GSH) concentration in 45 patients aged ≥65 years compared with 34 patients aged <65 years.
B. Lipoxygenases
LOXs are non-heme iron-containing dioxygenases that oxidize polyunsaturated fatty acids released from the cell membrane under inflammatory conditions into hydroperoxy fatty acid derivatives (88). LOXs form a heterogeneous class of lipid peroxidizing enzymes that seem to be involved in the pathogenesis of atherosclerosis (115). LOXs, in particular 5-LOX and 12/15 LOX, were found to be overexpressed in advanced atherosclerotic lesions (17, 120), where they can contribute to leukocyte recruitment through the generation of two classes of AA-derived lipid mediators, namely leukotrienes and lipoxins (141). In particular, 5-LOX catalyzes the transformation of free AA into leukotriene B4, a potent chemo-attractant and leukocyte activator. However, inconclusive data were obtained with respect to the patho-physiological relevance of this leukotriene signaling in atherosclerosis. Thus, leukotriene B4 antagonist was able to decrease monocyte-derived foam cell translocation into the plaque (3), in a double knockout mice model BLT(−/−) and ApoE(−/−), whereas deletion of BLT1, the leukotriene B4 receptor, was associated to reduced lesion formation during the early stages of plaque development, but was not effective at more advanced stages (352).
12/15-LOX catalyzes the oxidation of AA, resulting in the formation of the pro-oxidant 12-/15-hydroxyeicosatetraenoic acids. Moreover, 12/15-LOX oxidizes polyunsaturated acyl chains in phospholipids and cholesteryl esters, two relevant LDL components (27, 134). Experiments in knockout models supported the relevance of 12/15-LOX in the atherosclerotic process. Thus, 12/15-LOX(−/−) mice on a high-fat diet disclosed reduced ox-LDL, plasma, and urinary isoprostanes levels, and atherosclerotic lesion initiation was significantly delayed in the double-knockout mice compared with ApoE(−/−) mice (70). Consistent with this, overexpression of human 15-LOX in the vascular endothelium of LDLR(−/−) mice was associated with increased early atherosclerosis (133).
Although data on animal models are promising, there are only few studies regarding the involvement of LOX in human CVD. Only recently, in a case-cohort study including 57,053 participants aged 50–64 years, polymorphisms of 5-LOX pathway, resulting in LOX5 gain of function, were associated with incident MI (117).
LOXs seem also to be affected by animal and human aging. Thus, 5-LOX mRNA and protein levels were significantly increased in the cerebral nervous system of 25 month- compared with 3 month-old mice (216). Similarly, in humans, expression of the 5-LOX gene and the activity of 5-LOX were increased in elderly subjects, suggesting a role for this enzyme in neurological diseases associated with aging (104).
C. NADPH oxidase
Nox is an enzymatic system composed by several subunits, and different isoforms have been described so far. Nox is responsible for the formation of O2 − (383) that induces LDL oxidation, expression of VCAM-1, MCP-1, and endothelial monocyte adhesion and infiltration (118).
The impact of the different Nox isoforms in human atherosclerosis is still a matter of debate (379). Thus, Nox1 and Nox5 are known to essentially elicit O2 − formation and to contribute to proliferation of humans VSMC (379). Nox4 yields H2O2 formation and only scarcely stimulates O2 − production (379); Nox4 also possesses vasodilating properties via eNOS activation (323). Among ROS generated by Nox2, O2 − and H2O2 seem to play a relevant role in redox signaling of atherogenic processes. These two molecules appear to have different impact on vascular function as O2 − is devoted to controlling endothelial dilatation whereas H2O2 may have not only vasodilation but also pro-inflammatory properties (114). Thus, in a model of carotid ligation, overexpression of the Nox2 subunit p22phox was associated with a marked increase of atheroma and a concomitant overproduction of H2O2 (173). This finding was confirmed in experiments in which the use of an H2O2 scavenger, such as catalase, reduced the formation of atheroma (173).
An experimental study demonstrated that atherosclerotic plaque formation was significantly mitigated in animals treated with apocynin, a molecule that reduces subunit p47phox translocation to the membrane catalytic Nox2 (206, 298). Apocynin dose dependently decreased total monocyte accumulation, platelet adhesion, and atherosclerotic progression (206). The relationship between Nox2 and atherosclerotic lesion progression has been supported by Judkins et al., who studied a double knockout model of accelerated atherosclerosis represented by Nox2(−/y)/ApoE(−/−) mice (168). They found a significantly lower vascular ROS production, increased NO bioavailability, and reduced early lesion development compared with ApoE(−/−) mice (168). Similar results were also obtained in ApoE(−/−)/p47phox(−/−) mice (20). These data were further confirmed by Quesada et al., who found a significant regression of atherosclerotic plaque in mice fed with a high-fat diet and given a specific Nox2 inhibitor (298). Finally, in mice overexpressing endothelial Nox2, an early increase of endothelial activation and macrophage accumulation within the sub-endothelium were observed compared with controls, whereas aged animals showed similar atherosclerotic burden and progression (89).
In human, overexpression of some Nox subunits was associated with a marked increase of atheroma (173). Azumi et al. demonstrated that p22phox subunit was overexpressed in the vessel wall of atherosclerotic coronary arteries using coronary sections from autoptic specimens (15). This observation was confirmed by Guzik et al. (131), who found enhanced superoxide production in coronary arteries from patients with CHD in association with upregulation of p22phox and Nox2, suggesting that both these subunits contribute to oxidative stress in human coronary atherosclerotic lesions (131).
Experiments performed in subjects affected by chronic granulomatous disease (CGD), which is characterized by hereditary deficiency of Nox2 subunits, allowed to investigate the role of this Nox in human atherosclerosis. The most common form of CGD is related to Nox2 hereditary deficiency, but other subunits such as p47phox may be lacking. To investigate the role of Nox2 in the physiology and pathophysiology of cardiovascular system, we developed an immuno-assay that measures the extra membrane peptide of the enzyme released in the medium on Nox2 activation, called soluble Nox2-derived peptide (sNox2-dp) (278). Blood analysis demonstrated that ∼90% of sNox2-dp stems from activation of leukocytes, lymphocytes/monocytes, and platelets (278).
The interplay between Nox and atherosclerosis was also investigated in female carriers of Nox2 deficiency (384). The study showed a significant reduction of the carotid intima-media thickness (IMT), which is another surrogate marker of atherosclerosis, in carriers compared with controls (215). Using a more sophisticated diagnostic approach, that is, magnetic resonance imaging and computed tomography, Sibley et al. (337) supported these preliminary reports by demonstrating that CGD patients, compared with control subjects, had a 22% lower internal carotid artery wall volume with a similar reduction detected in both the p47phox− and gp91phox-deficient subtypes (337).
The relationship between Nox2 and aging has been investigated in ApoE(−/−) mice, which showed an upregulation of Nox2 in atherosclerotic plaque compared with controls; Nox2 was upregulated in endothelial cells and macrophages of atherosclerotic lesion and associated with elevated ROS levels. Of particular interest was the fact that upregulation of Nox2 and elevation of ROS were age dependent, with a significant increase from 12 to 19 weeks (168).
The relationship between aging and Nox2 has been prospectively investigated in AF, who were followed up for about 40 months; Nox2 activity increased coincidentally, with aging-related GPx3 downregulation suggesting an imbalance between oxidant and antioxidant status (268); and the imbalance between Nox2 and GPx3 was significantly evident at the age of 75 years, coincidentally with an abrupt risk of CVEs.
VII. Oxidative Products
A. F2-isoprostanes
F2-isoprostanes are a family of eicosanoids with pro-atherogenic and pro-thrombotic properties. The eicosanoid 8-iso-prostaglandin F2α (8-iso-PGF2α) derives from the nonenzymatic oxidation of AA, and it is involved in the late phase of platelet activation and thrombus growth (280). The production of 8-iso-PGF2α is partly a result of activation of Nox2 (53), as shown by impaired formation of 8-iso-PGF2α in CGD patients (384). Urinary excretion of 8-iso-PGF2α is a validated and accepted reliable biomarker of in vivo oxidative stress, which has been investigated in healthy subjects and in patients at risk or with CVD (53). Increased values of urinary 8-iso-PGF2α (23) and serum sNox2-dp levels have been detected in subjects with CHD, hypertension, type 2 DM, MetS, AF, and PAD. Plasma or urinary excretion of 8-iso-PGF2α has been extensively investigated in patients with acute or chronic CAD and, with very few exceptions, the levels have been found to be elevated in patients compared with controls (Table 2). Production of 8-iso-PGF2α was more pronounced in patients with acute versus stable CHD and correlated with the number of affected vessels, suggesting a relationship between oxidative stress and coronary atherosclerotic burden (65, 392).
8-iso-PGF2α, 8-iso-prostaglandin F2α; CAD, coronary artery disease; CHD, coronary heart disease; NS, not significant; SD, standard deviation.
The predictive role of 8-iso-PGF2α levels has been tested in a nested case-cohort study including 141 CHD cases, 109 stroke cases, and 142 controls (306). The highest quartile of urinary 8-iso-PGF2α compared with the lowest ones had an odds ratio of 1.8 to develop fatal CVD in a median 10-year follow-up.
LeLeiko et al. (197) measured serum F2-isoprostanes in 108 patients presenting with acute coronary syndrome (ACS) and in 101 control patients. ACS patients had higher serum F2-isoprostanes compared with controls and were predictive of cardiovascular recurrences.
More recently, in 1002 anticoagulated elderly AF patients followed up for about 3 years, median levels of urinary 8-iso-PGF2α (160 vs. 100 pg/mg creatinine, p < 0.001) and sNox2-dp (13 vs. 9 pg/ml, p < 0.001) were significantly higher in patients with CVEs compared with those without CVEs during the follow-up (284).
The relationship between F2-isoprostanes and aging has been scarcely investigated. Apparently, the production of 8-iso-PGF2α increases with age as a significant positive correlation was found between age and 8-iso-PGF2α (247). Even if this would be consistent with an overproduction of isoprostanes in the elderly, further studies are necessary to support this single finding.
B. Oxidized LDL
ox-LDLs play a pivotal role in the process of atherosclerosis. On reaching the sub-endothelium, LDL undergo oxidation via ROS produced by pro-oxidant enzymes such as Nox2, MPO, or xanthine-oxidase and are taken up by macrophages via scavenger receptors, thus promoting the formation of foam cells (351). Ox-LDL have been evaluated in several studies, including patients with different degrees of CHD. A cross-sectional study (148), which included 63 patients with ACS, 35 nontransplanted patients with angiographic stable angina, 28 heart transplant patients with post-transplant CAD, 79 heart transplant patients without CAD, and 65 control subjects, showed that ox-LDL levels were significantly higher in patients with CAD compared with those without. The association between ox-LDL levels and CAD severity was also corroborated in patients with ACS, who displayed higher levels of ox-LDL compared with patients with stable CAD. Prospective studies investigating the prognostic role of ox-LDL provided positive (147, 163, 302, 334, 370, 406) and negative (2, 36, 177, 402) results.
Holvoet et al. (147) studied 3033 subjects, 1147 with and 1886 without MetS, and found elevated values of ox-LDL in patients with MetS (1.45 ± 0.82 mg/dl) compared with those without MetS (1.23 ± 0.67 mg/dl, p < 0.0001). ox-LDL levels were associated with an increased rate of MI during the follow-up (relative risk 2.25 for the highest quintile). The incidence of CHD events was registered in a 6-year follow-up among 18,140 men from the HPFS (Health Professionals Follow-up Study) and in an 8 year follow-up among 32,826 women from the Nurses' Health Study. Subjects experiencing CHD showed higher values of ox-LDL compared with controls in both men and women. The highest quintile of ox-LDL was significantly associated with an increased risk of CHD in a multivariate model (406). In the prospective case-control study in which 44,725 men and women from the World Health Organization Multinational MONICA (Monitoring of Trends and Determinants in Cardiovascular Disease) project and the Västerbotten Intervention Program were enrolled, there was no difference in the levels of IgG, IgA, and IgM autoantibodies against copper-ox-LDL or malonaldehyde-LDL between patients experiencing CD and controls (2). In the Framingham offspring study, IgG antibodies to ox-LDL were measured in 1192 men and 1427 women who were followed for 8 years for the occurrence of CHD and CVEs (402). Although IgG were significantly correlated with age in both men and women, no association with events was found. Finally, in 36 patients undergoing carotid endoarterectomy, ox-LDL levels were higher in comparison with 20 controls and were significantly higher in unstable versus stable carotid plaques (338).
Different methodology used in the earlier reported clinical studies may account for these conflicting results. Indeed, some studies used antibodies against ox-LDL, whereas some others made a direct measurement of ox-LDL, with or without adjustment for lipid profile. A more reliable methodology to measure ox-LDL is likely necessary to further investigate the role of ox-LDL in atherothrombosis.
VIII. Atherosclerosis and Antioxidant Status
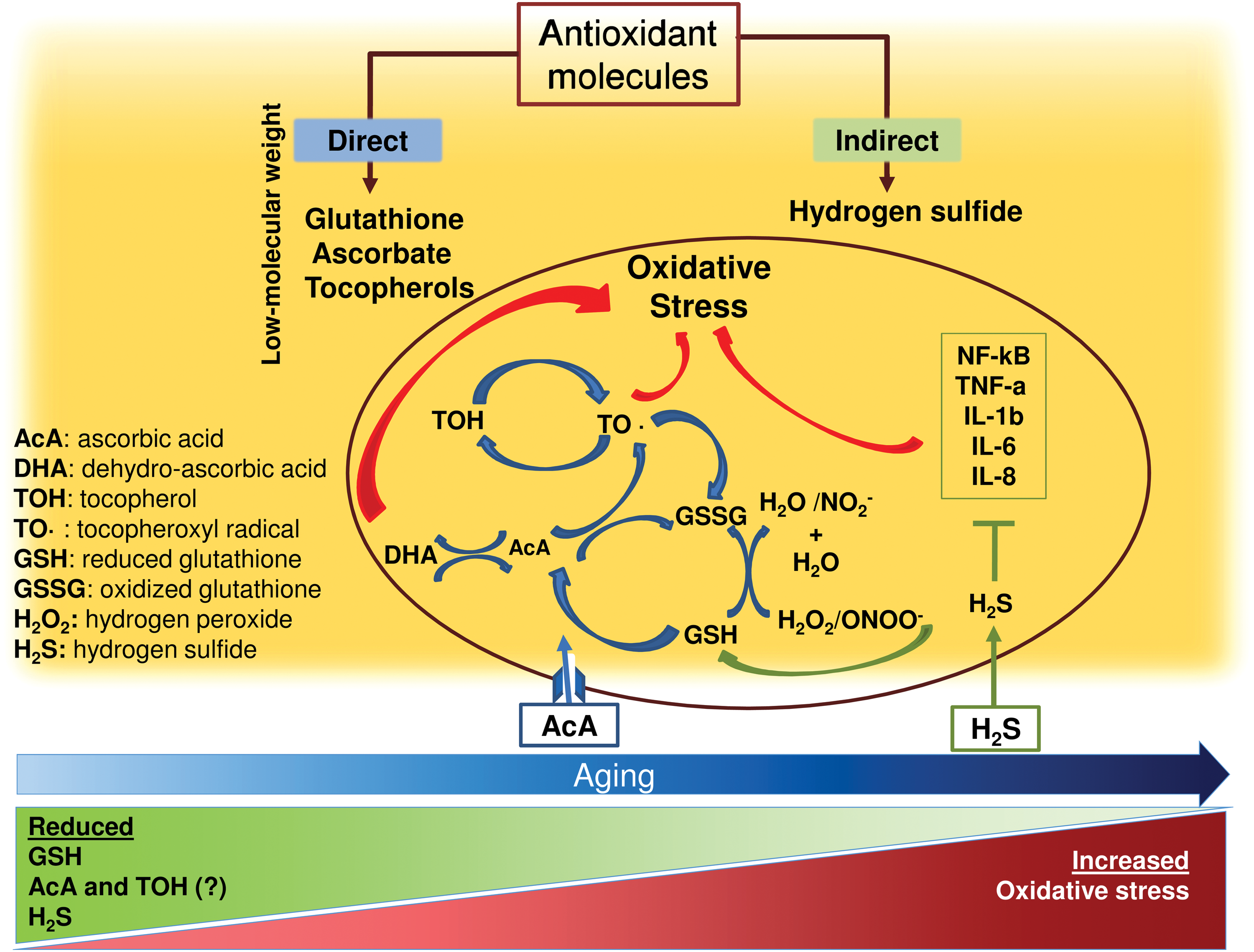
Cellular protection against oxidative products is provided by a complex network of antioxidant systems, which can be classified according to their activity (i.e., enzymatic and nonenzymatic antioxidants) or to their behavior into redox reactions (i.e., direct and indirect antioxidants) (Fig. 4) (87, 295). However, it should be noted that this classification does not reflect the more complicated functional interplay existing between direct and indirect antioxidants. For instance, some antioxidants are both direct and indirect and can be referred to as “bifunctional.” Moreover, cytoprotective proteins may participate in the synthesis/regeneration of direct antioxidants, which are, in turn, required for the catalytic functions of cytoprotective proteins (87, 295).
B. Antioxidant enzymes
1. NO synthase
The NOS family generates NO from conversion of
The iNOS isoform is synthesized in response to pro-inflammatory agonists such as cytokines and, differently from eNOS and nNOS, seems to play a pro-atherogenic role. Thus, iNOS activation leads to a sustained production of NO, which corresponds to ∼100-fold the amount produced by constitutive eNOS. Large quantities of NO could combine with O2 − to form peroxynitrite, an adduct with enhanced oxidizing capability. iNOS is detectable in human atherosclerotic lesions (46), where it contributes to peroxynitrite formation and, eventually, LDL oxidation. In fact, LDL isolated from aortic atherosclerotic intima had 90-fold higher levels of peroxynitrite compared with healthy subjects. The pro-atherogenic role of iNOS was documented in ApoE(−/−)/iNOS(−/−) mice, which showed a significantly reduced atherosclerotic lesion area and lipid peroxidation compared with ApoE(−/−) mice (182, 242).
The activity of NOS profile seems to be modified in the elderly. Thus, a recent study, evaluating age-related progressive organ dysfunction in 338 healthy subjects (age ranging from 3 to 92 years), demonstrated that eNOS protein level was significantly decreased in elderly people as compared with adults (174). However, the impact of this change on atherosclerosis progression and CVEs is still unknown.
2. Superoxide dismutase
SOD family encompasses three isoforms, namely SOD1, SOD2, and SOD3, which are cytoplasmic, mitochondrial, and extracellular isoforms, respectively. All isoforms seem to protect against atherosclerosis. Thus, overexpression of SOD1 and catalase or catalase alone decreased plasma and aortic F2-isoprostane levels and retarded atherosclerotic lesion development in ApoE(−/−) mice (413). On the contrary, SOD2 deficiency induced mitochondrial DNA damage and accelerated atherosclerosis in ApoE(−/−) mice (18). SOD1 and SOD2 deficiency resulted in VSMC hyperplasia and hypertrophy mediated by different kinases (217). Previous studies showed that SOD1(−/−) mice are prone to develop various aging-related pathologies, concomitantly with an increase of oxidative damage in organs (398). In accordance with this, SOD1 and SOD2 prevented DNA damage during aging in experimental models. Of note, in aging mice, damage related to SOD downregulation was counteracted by atorvastatin treatment, resulting in delayed age-related cardiac alterations (i.e., cardiac hypertrophy) and inhibition of inflammatory cytokines (132). The relationship between SOD activity and aging was also investigated in humans. In lymphocytes from healthy subjects, a progressive decrease across decades of age of antioxidant enzyme activity such as SOD, catalase, and GPx has been reported (119). Further, in samples of human brain tissues, a significant decrease in SOD and GPx activities with increasing age was detected, suggesting a role for impaired antioxidant status in the onset of neurologic disorders such as Parkinson and Alzheimer diseases (377). SOD3 seems to be involved in protecting LDL particles from oxidation. Thus, in rabbit as well as human endothelial cells, recombinant SOD3 decreases LDL oxidation (196, 359). In patients with CAD, low levels of plasma SOD3 were independently associated with a history of MI (394).
3. Glutathione peroxidase
Even if there is a growing body of evidence to suggest that GPx has anti-atherosclerotic property (308), differences among the eight isoforms belonging to the GPx family do exist. However, all GPx isoforms share the ability to detoxify peroxides, with GSH acting as an electron donor in the reduction reaction, producing GSH disulfide (110). GPx1 is prevalently localized in red blood cells, GPx2 is in the gastrointestinal tract, and GPx3 is the only circulating isoform. GPx4 exists as a cytosolic, mitochondrial, and nuclear isoform derived from alternative splicing of a single gene. In human, GPx4 acts as a phospholipid hydroperoxidase protecting cells from lipid peroxidation. It also plays a role in primary T cell response to infections by protecting T cells from ferroptosis, a cell death secondary to an iron-dependent accumulation of lipid ROS. Finally, GPx4 serves as a structural function in mature sperm cells. The isoforms 5–8 are less characterized.
GPx1 and GPx4 play a role in preventing atherosclerosis progression, and GPx1 seems to also possess antithrombotic properties (308). Thus, in ApoE(−/−) GPx1-deficient mice, increased aortic ROS, decreased NO, and increased atherosclerosis burden were observed (365). In accordance with experimental studies, GPx1 activity is either decreased or absent in human carotid atherosclerotic lesions, and its absence is associated with lesion severity (195). In another human study that investigated platelet activity in two brothers with cerebral vein thrombosis, Freedman et al. found a decreased activity of GPx3 along with an increased platelet reactivity compared with controls (113). The addition of exogenous GPx restored NO-mediated platelet function. Further, overexpression of GPx4 reduced aortic F2-isoprostane levels, lowered IL1-induced VCAM-1 expression, and delayed lesion progression in experimental animal models (19, 130).
The relationship between GPx isoforms and CVEs has been investigated in few studies. In a cohort of 636 patients with suspected CHD, an inverse relationship between GPx1 activity and risk of CVEs during a follow-up of 4.7 years was reported (33). Further, a recent prospective cohort study that included 909 AF patients reported a significantly lower value of GPx3 and SOD in patients experiencing a CVE in a mean follow-up of 43.4 months (268). The same study also reported that GPx3 was the antioxidant enzyme that progressively decreased by aging, with an abrupt reduction at age of 70 years. Of note, such a reduction was associated with an overproduction of TxB2, suggesting an interplay between impaired detoxification of ROS and platelet activation (268).
4. Paraoxonase
PON is an antioxidant enzyme that exists in three isoforms (PON1, PON2, PON3) and possesses atheroprotective effects. The antioxidant activity of PON1 has been detected in vitro, where it inhibited LDL and HDL oxidation (12, 13), and in PON1-knockout mice, which displayed higher levels of oxidized phospholipids and increased aortic atherosclerosis compared with wild type (331, 332). Two common polymorphisms of PON1 (i.e., 55 and 192) were shown to be associated with human atherosclerosis and heart disease.
PON2 attenuated triglyceride accumulation into macrophages and foam cell formation via the inhibition of redox-sensitive microsomal diacylglycerol acyltransferase 1 (310). PON2-deficient ApoE(−/−) mice developed enhanced mitochondrial oxidative stress and exacerbated atherosclerosis when fed with chow and Western diet (85).
Similar to PON1, PON-3 prevents oxidative modification of LDL; humanized PON3 transgenic mice fed with an atherogenic diet exhibited decreased atherosclerotic lesion areas compared with nontransgenic animals (333).
Serum PON1 concentration decreased in the atherosclerotic ApoE(−/−) mice during aging and was inversely correlated to the development of atherosclerotic lesions (13). This finding was corroborated by a human study demonstrating that PON1 concentration was lower in old (60–89 years) compared with young (20–30 years) subjects (63). Similarly, Milochevitch and Khalil found that plasma PON1 was inversely correlated to age in a group of subjects ranging 26–77 years (238). Increased age was also negatively correlated with PON activities, specifically with the PON1-192 variant. In the same study, higher expression of this polymorphism was found in centenaries, suggesting a role for PON1 in successful aging (223).
5. Heme-oxygenase
Heme-oxygenase (HO) is a microsomal enzymatic system involved in the degradation of heme (186, 248, 368). The HO system regulates heme-protein levels and protects cells from the deleterious heme-induced iron-dependent reactions, leading to ROS generation and lipid peroxidation (186, 417). HO reduces cellular free heme by splitting the tetrapyrrole heme ring to biliverdin, free ferrous iron, and carbon monoxide. Biliverdin is subsequently metabolized to the antioxidant molecule bilirubin by biliverdin reductase (248). The mechanism by which bilirubin reacts with ROS is not completely understood, although its hydrophobic tetrapyrrole structure has been reported to inhibit the activation of Nox (189). Recently, bilirubin was reported to suppress atherosclerotic plaque formation in LDLR-deficient mice by disrupting endothelial VCAM-1 and ICAM-1-mediated leukocyte migration through its ROS-scavenging property (389).
Of the two HO isoforms described, HO-1 is involved in the atherosclerotic process whereas HO-2 seems to play a protective role against anoxic processes at neuronal and cardiac level (186, 248). HO-1 has a broad spectrum of inducers such as ROS, cytokines, PGs, and endotoxin (84). HO-1 is highly expressed in macrophages, especially in those contained in the intra-plaque hemorrhage where it plays an athero-protective role. Thus, HO-1+ macrophage population is significantly expanded in atherosclerotic plaques in the heme-rich regions (261). When HO-1-deficient macrophages are treated with ox-LDL, generation of ROS is amplified and the secretion of pro-inflammatory IL-6, MCP-1, and the IL-8 homologue is increased (261). Thus, HO-1 expression in macrophages makes them resistant to foam cell formation and pro-inflammatory mediators that exacerbate atherogenesis and plaque progression (150).
The importance of HO-1 in vascular biology was highlighted in a child with HO-1 deficiency, in whom endothelial cells were more susceptible to oxidative insults and heme-mediated LDL oxidation (410). Further, Exner et al. carried out a cohort study to evaluate the relationship between HO-1 gene promoter polymorphisms associated to higher HO-1 expression and the risk for arterial restenosis after percutaneous transluminal angioplasty (100). Patients with short (<25 GT) dinucleotide repeats in the HO-1 gene promoter had significantly less restenosis than patients with longer (≥25 GT) dinucleotide repeats (100). This finding was corroborated in a large cohort of 812 subjects from the Bruneck Study, which described an inverse association between low HO-1 and atherosclerotic progression along with a trend toward higher levels of oxidized phospholipids on ApoB-100 (272). The behavior of this antioxidant enzyme in the elderly population is still unknown.
B. Direct antioxidants
Several low-molecular-weight compounds belong to direct antioxidants such as ascorbate, GSH, and tocopherols. They are all involved in redox reactions by scavenging ROS and nitrogen species. Direct antioxidants have peculiar features such as being redox active, consumed or chemically modified on antioxidant reaction, and needing reloading or regeneration.
1. Glutathione
The non-protein thiol GSH (
GSH levels may vary significantly among different organs, with some of them being responsible for GSH synthesis, whereas some others are essentially exporters. In human tissues, GSH levels range from 0.1 to 10 mm, being most concentrated in the liver (up to 10 mm) (258).
Although cells have relatively high concentrations of GSH, an increase in synthesis of GSH is clearly part of the adaptive response to oxidative stress (367). Several evidences support the role of GSH in atherosclerotic progression. The GSH content in mice aorta is reduced before and during lesion development (30), and GSH-dependent expression of GPx1 is reduced in ApoE-deficient mice treated with an atherogenic diet (356). Modulation of GSH levels by treating ApoE-deficient mice with
A few data associated GSH levels in plasma or red blood cells with CHD. In a case-control study, patients with angiographically proven CAD or awaiting coronary artery bypass graft (CABG) presented significantly lower red blood cell GSH levels compared with controls (41, 127). More recently, an analysis among 134 CVD cases and 435 age- and sex-matched healthy control subjects from the Hisayama Study demonstrated that plasma GSH concentrations were lower in all CVD cases than in controls, particularly in patients affected by severe cerebral infarction (335). Data on the role of GSH in preventing CVD stem from genetic studies involved the GCLC and GCLM units of GCL, as GSH biosynthesis is paralleled by GCL gene expression. Thus, the CT polymorphism of the GCLM gene was associated with an increased risk of MI (251). Similarly, the 129T polymorphism of the GCLC gene was associated to reduced levels of GSH and endothelial dysfunction in MI patients compared with controls (178). These data are in contrast with a recent study that did not show significant difference in plasma GSH comparing PAD patients with controls (339).
Two prospective studies investigated whether GSH predicts CVD. GSH appeared independently and inversely associated with recurrent atherothrombotic events in a cohort of 375 survivors admitted for an ACS, followed for a median duration of 2.7 years (108). Similarly, in a sample from the Hunter Community Study free of CVEs, 5-year absolute cardiovascular risk score calculated using the Framingham Risk Equation was independently and inversely associated to plasma GSH levels (220).
GSH concentration appears to decline with advancing age. Thus, in healthy subjects aged 19–85 years, plasma ratio between reduced and oxidized GSH in plasma did not change before 45 years, but later on GSH was oxidized at a nearly linear rate of 0.7 mV/year, suggesting that after 45 years GSH declines rapidly (165). Although these data could be questioned as plasma GSH accounts for only 1% of total GSH, similar results were obtained while studying GSH from other sources such as erythrocytes and lymphocytes. Thus, in healthy individuals, GSH levels were significantly lower in the erythrocytes of “old” (60–79 years) compared with “mature” group (40–59 years) and with the young reference group (20–39 years), which expressed the highest GSH content (193). Similarly, human lymphocytes display a progressive decrease of GSH by advancing age, with 60–80 year-old subjects having about 50% less of GSH lymphocyte content compared with 20–40 year ones (375).
The decline of GSH by aging may also have an impact on extracellular levels of other antioxidants, such as alpha tocopherol and ascorbic acid, as GSH is involved in their restoration cycle. Thus, cycles of regeneration of these three antioxidants are strictly correlated (374). For example, in human erythrocytes, ascorbate regeneration from dehydroascorbate is largely dependent on GSH (232), and oxidized vitamin E radical is reduced by both GSH and ascorbic acid, giving formation to the ascorbyl radical (374).
Of note, the bio-availability of cysteine appears to be the rate-limiting factor for synthesis of GSH, which is a sulfur-containing metabolite. The reduced dietary protein intake occurring by aging could be an additional detrimental factor concurring to age-related GSH decrease (153, 255). Food, such as garlic, onions, and brussels sprouts, contain significant amounts of sulfur, and could be considered as part of a nutritional approach for elderly patients.
2. Vitamin E
Natural vitamin E consists of a family of eight different compounds, four tocopherols and four tocotrienols. All tocopherols and tocotrienols are potent antioxidants with lipoperoxyl radical-scavenging activities, which consist of donating hydrogen atoms from the phenol group on the chromanol ring. Animal studies provided evidence of a role for α-tocopherol in modulating arterial lipid content, lipid oxidation, and eventually plaque evolution. Verlangieri and Bush (378) reported a 35% inhibition of atherosclerotic lesion in cholesterol-fed macaques supplemented with α-tocopherol over a 3-year period of follow-up (378). In rabbits with established experimental atherosclerosis undergoing angioplasty, a reduced restenosis and IMT after angioplasty was observed after α-tocopherol supplementation (190). Similarly, α-tocopherol supplementation inhibited aortic intimal thickening as well as lipid peroxidation in a chicken model (342). Further, in ApoE(−/−) mice, α-tocopherol supplementation was able to reduce aortic lesions in 4- to 10-week-old ApoE(−/−) mice, when fatty streaks are absent or very sparse (274). Similar results were obtained in older animals with established aortic lesion (273). Finally, diet α-tocopherol intake led to a reduced deposition of cholesterol in arterial plaques from rabbits fed with atherogenic diet (325), and ex-vivo copper mediated oxidative modification of LDL in the same animal model (401). α-tocopherol also possesses anti-inflammatory effect as depicted by its capacity of slowing cell migration into the lesion site. Thus, in aortic lesions of ApoE(−/−) mice treated with α-tocopherol, a reduced level of MCP-1 was observed (274). In addition, α-tocopherol can affect the atherothrombotic evolution by modulating platelet activation; thus, intravenously administered tocotrienols induced a fourfold increase in α-tocopherol concentration and inhibited acute platelet-mediated thrombus and agonist-induced platelet aggregation in stenotic canine coronary arteries (299).
The relationship between aging and α-tocopherol concentration is controversial. Serum and visceral levels of α-tocopherol were found to be decreased by aging in rats (169), whereas another study failed to confirm this relationship (252). In accordance with this last finding, decreased levels of ascorbic acid and GSH, but no differences of α-tocopherol by aging, have been reported (292).
3. Vitamin C
Ascorbic acid (vitamin C) is a hydrophilic vitamin that interferes with several mechanisms involved in the atherosclerotic process mostly by preventing NO degradation (399) and LDL oxidation (161). Ascorbic acid may preserve NO by several mechanisms, including direct reduction of nitrite to NO, release of NO from nitrosothiols, scavenging O2 − (231), and sparing endothelial cell-derived NO by recycling tetrahydrobiopterin (139, 231). Ascorbic acid plays a protective role against atherosclerotic burden by decreasing endothelial cells' ability to oxidase LDL (225). This effect is also achieved by influencing α-tocopherol metabolism as depicted by in vitro experiments where ascorbic acid restored the reduced form of α-tocopherol in human monocyte/macrophages, ultimately preventing LDL oxidation (161). Combined deficiency of vitamin E and C increased lipid peroxidation, doubled plaque size, and increased plaque macrophage content by two- to threefold in ApoE(−/−) mice (16). The progression of the atherosclerotic plaque was also higher in ApoE(−/−) and gulonolactone oxidase(−/−) mice that prevented animals from synthesizing their own vitamin C (16). Ascorbic acid concentration seems to be affected by the aging process. In animals, serum vitamin C content was inversely related to age (252). Similarly, in humans, a progressive decrease in serum concentration of ascorbic acid by aging has been described (292).
C. Indirect antioxidants
Indirect antioxidants include small-molecule inducers of cytoprotective proteins such as H2S and polyphenols. Differently from direct ones, indirect antioxidants may be redox active and can also activate the NF-E2-related factor 2 (Nrf2)/antioxidant-responsive element (ARE) pathway, resulting in transcriptional induction of a battery of cytoprotective proteins (also known as phase 2 enzymes); the latter act catalytically, are not consumed in their antioxidant action, have longer half-lives, and catalyze several chemical reactions, ultimately leading to detoxification (87, 295).
1. Hydrogen sulfide
H2S is a “gaseous signaling molecule,” which freely diffuses through cell membranes, targeting proteins, enzymes, transcription factors, and membrane ion channels (295). In mammals, cysteine and methionine, the only two sulfur-containing amino acids, represent the major source of H2S (295), which is generated on the activity of four main enzymes, that is, cystathionine γ-lyase, cystathionine β-synthase, cysteine aminotransferase, and 3-mercaptopyruvate sulfurtransferase (222) [reviewed in detail in Caliendo et al. (48)]. H2S has important functions in cardiovascular and immune systems (418). In CVD, H2S acts by (i) promoting endothelial cells' proliferation and migration, (ii) favoring vasodilatation (via interaction or less with NO), and (iii) as an anti-inflammatory molecule by the NF-κB pathway. At immune level, H2S seems to be involved in the regulation of T cells (418).
Recent evidence indicated, in fact, that H2S inhibits LDL oxidation via scavenging hypochlorite and H2O2 and inhibiting MPO activity (191). H2S can modulate atherosclerotic plaque evolution by suppressing monocytes' adhesion to activated endothelium and reducing plaque volume, as demonstrated in ApoE(−/−) mice treated with H2S donor sodium hydrosulfide (395). Consistent with this, cystathionine γ-lyase-knockout mice displayed decreased endogenous H2S level, elevated plasma levels of LDL, increased oxidative stress and adhesion molecule expression, and early fatty streak lesions in the aortic root (221).
H2S is also demonstrated to interfere with the atherosclerotic process by diminishing the tissue injury induced by LPS-mediated neutrophil activation via induction of apoptosis and/or scavenging neutrophil-derived hypochlorite (400). H2S also acts by downregulating several pro-atherogenic cytokines, including NF-κB, TNFα, IL-1b, IL-6, and IL-8, as demonstrated by in vitro studies (295).
Decreased levels of H2S have been found in different CVD, such as hypertension, heart failure, and type 2 diabetes [see also Wallace et al. (390)].
Aging is associated to reduced levels of H2S in animal models, but data on humans are lacking (42).
2. Polyphenols
Polyphenols are a class of natural, synthetic, and semisynthetic substances characterized by large multiples of phenol units. Polyphenols, in the form of flavonoids, are broadly classified into (i) anthocyanins, namely cyanidin, delphinidin, malvidin contained in raspberry; (ii) flavanols, namely catechin, epicatechin contained in cocoa, red wine, green tea; (iii) flavonols, namely quercetin, fisetin contained in onion, blueberry, red apple, many fruits and vegetables; (iv) flavanones, namely hesperidin; and (v) flavones luteolin in citrus fruits (369).
In addition, resveratrol (3,5,4′-trihydroxy-trans-stilbene) is a non-flavonoid phytoalexin polyphenol mostly contained in plants and red wine [reviewed in detail in Xia et al. (408)].
Antioxidant ability of polyphenols to scavenge free radicals has not been fully elucidated (146). However, polyphenols exert an indirect antioxidant activity by inhibiting Nox, thus increasing NO availability (211). Moreover, polyphenols seem to exert a cytoprotective action; for instance, at neuronal level, polyphenols may be neuroprotective by modulation of the Nrf2/ARE pathway, which regulates phase II antioxidant responses, triggering the simultaneous expression of numerous protective enzymes and scavengers including HO-1 (321). Polyphenols may also have a cardioprotective effect due to their uncoupling properties. Endogenous uncoupling is a physiologic mechanism to lower mitochondrial ROS production, and it is regulated by specific inducible uncoupling proteins. According to the “uncoupling to survive” theory, this mechanism could be important in minimizing oxidative damage to DNA and in slowing aging (35). Thus, “mild mitochondrial uncoupling” has been proposed as a mechanism to reduce upstream ROS production; it has been suggested that “mild mitochondrial uncoupling” could be a more effective therapeutic strategy than removing ROS once they are formed (69).
IX. Clotting Activation
The risk of thrombosis in the artery and venous circulation typically increases with aging, and it is responsible for an enhanced incidence of CVD and venous thromboembolism in the elderly population (180). Activation of the clotting system is relevant for thrombus growth for both arterial and venous thrombosis. As depicted in Figure 5, the clotting system includes several proteases, which are activated as a “cascade” and lead to fibrin formation, which serves for vessel hemostasis in case of injury.
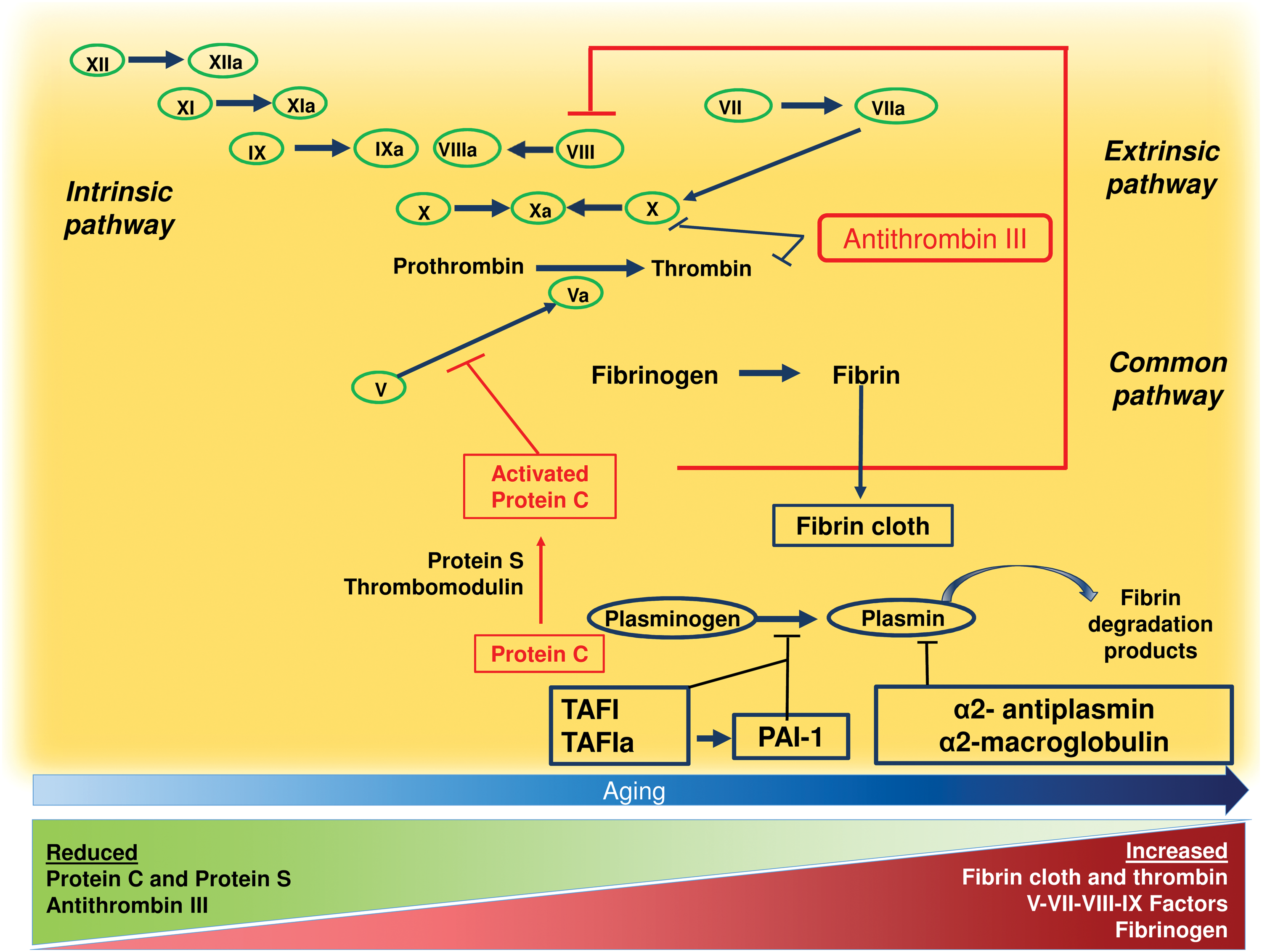
Several studies analyzed modification of the clotting system by aging using different global coagulation assays or markers of clotting activation (1, 103). Analysis of global tests, which were performed prevalently in healthy volunteers, consistently showed a more rapid clotting activation in the elderly compared with the younger population regardless of the assay used. In accordance with these reports, measurement of markers of clotting activation such as the prothrombin fragment F1 + 2 demonstrated a significant increase of thrombin generation in the elderly compared with the younger population (24). This was also evident when analyzing activation of specific clotting factors, such as factor IX and X activation peptides, which were more elevated by aging (103). It should be underscored, however, that the sample size of these studies was small and, hence, it is difficult to adequately appreciate the change of clotting activation across decades of age. Moreover, studies demonstrating that changes of the clotting system by aging are associated with an enhanced thrombotic risk are lacking.
Increased activation of the clotting system by aging might be attributed to changes of coagulation factors, anticoagulants, and the fibrinolytic system. One factor potentially contributing to enhanced clotting activation by aging is the increased concentration of some coagulation factors, such as FV, FVII, FVIII, FIX, and fibrinogen, which, in fact, increase by aging (112). For some of them such as factor VIII and fibrinogen, prospective studies documented an increased risk of thrombosis related to their blood concentration, but it is difficult to draw definite conclusions from these studies as both factor VIII and fibrinogen are acute-phase reactant proteins, which may largely vary depending on the timing of blood sampling. Reduction of natural anticoagulants by aging might be a mechanism accounting for enhanced clotting activation but data regarding this point are unclear and conflicting. Clinical studies investigated whether the activity of some anticoagulants such as antithrombin III (ATIII), protein C, S, and tissue factor (TF) pathway inhibitor may decrease by aging. However, the clinical impact of these reports is unclear as some studies showed no differences in anticoagulant proteins in elderly compared with younger subjects, whereas some others reported an increase of these anticoagulants in the elderly population (112).
Changes in the activity of the fibrinolytic system may also concur to increase the thrombotic risk by aging but also in this case, results are inconclusive. Using global tests of fibrinolysis activation, such as plasmin-alpha2-antiplasmin complex, it was demonstrated that the fibrinolytic system would be activated by aging in 800 elderly subjects free of clinical CVD in the Cardiovascular Health Study and the Honolulu Heart Program cohort (315). Conversely, plasminogen activator inhibitor 1 (PAI-1) has been shown to increase by aging (411) but, as for factor VIII and fibrinogen, PAI-1 is an acute-phase reactant and may not be the expression of an actual inhibition of the fibrinolytic system.
ROS are important promoters of clotting activation as they upregulate TF in leucocytes (47). The role of ROS and, more importantly, the role of Nox in the activation of the clotting system has been investigated in endothelial cells, where TF activation is modulated by ROS formation. Accordingly, an experimental study demonstrated a crucial role for Nox-derived ROS in upregulating endothelial-dependent TF activation, an effect significantly inhibited by cell incubation with antioxidants (140). Further, an experimental study demonstrated that oxidant molecules such as H2O2 produced by activated leucocytes induced thrombomodulin oxidation and eventually impaired protein C activation (140). This effect was prevented by cell incubation with diphenylene iodonium, which is an inhibitor of Nox (140). The effect of antioxidants on thrombus formation has been further investigated in a murine model of genetic deficiency of SOD, where susceptibility of carotid thrombosis was investigated in response to photochemical injury. Thus, animals deficient of SOD displayed faster and larger thrombotic occlusion in artery and venous vessels compared with wild type (77). This effect was dependent on impaired protein C activation and was restored by treatment with antioxidants such as SOD and catalase. Impaired protein C activation was attributed to ROS formation, which determined thrombomodulin oxidation with 40% reduction in thrombomodulin-dependent protein C activation (77).
Finally, oxidative stress may also promote thrombosis by acting on the fibrinolytic system, where ROS can upregulate PAI-1 in endothelial cells, thus favoring the thrombotic process in animals prone to atherosclerosis (355).
The role of oxidative stress as clotting activation promoter has been investigated both in vitro and in vivo with vitamin E, which has been shown to negatively interfere with intrinsic and extrinsic coagulation pathways and to promote fibrinolysis. This vitamin possesses anticoagulant property via inhibiting oxidation of vitamin K, which serves to activate the prothrombinase complex, that is, factors II, VII, IX, and X (263). Further, an in vitro study demonstrated that vitamin E downregulates the monocyte expression of TF, indicating that it can interfere with the extrinsic coagulation pathway (105). Finally, in patients with coronary spastic angina, Miyamoto et al. showed that 400 mg/day vitamin E significantly reduced PAI-1 activity after 1-month treatment (241).
X. Platelet Activation
Platelets have been long recognized as crucial players for primary hemostasis at sites of vascular injury. The activity of platelets at sites of plaque rupture consists of three different phases: platelet adhesion, platelet activation, and platelet recruitment (Fig. 6).
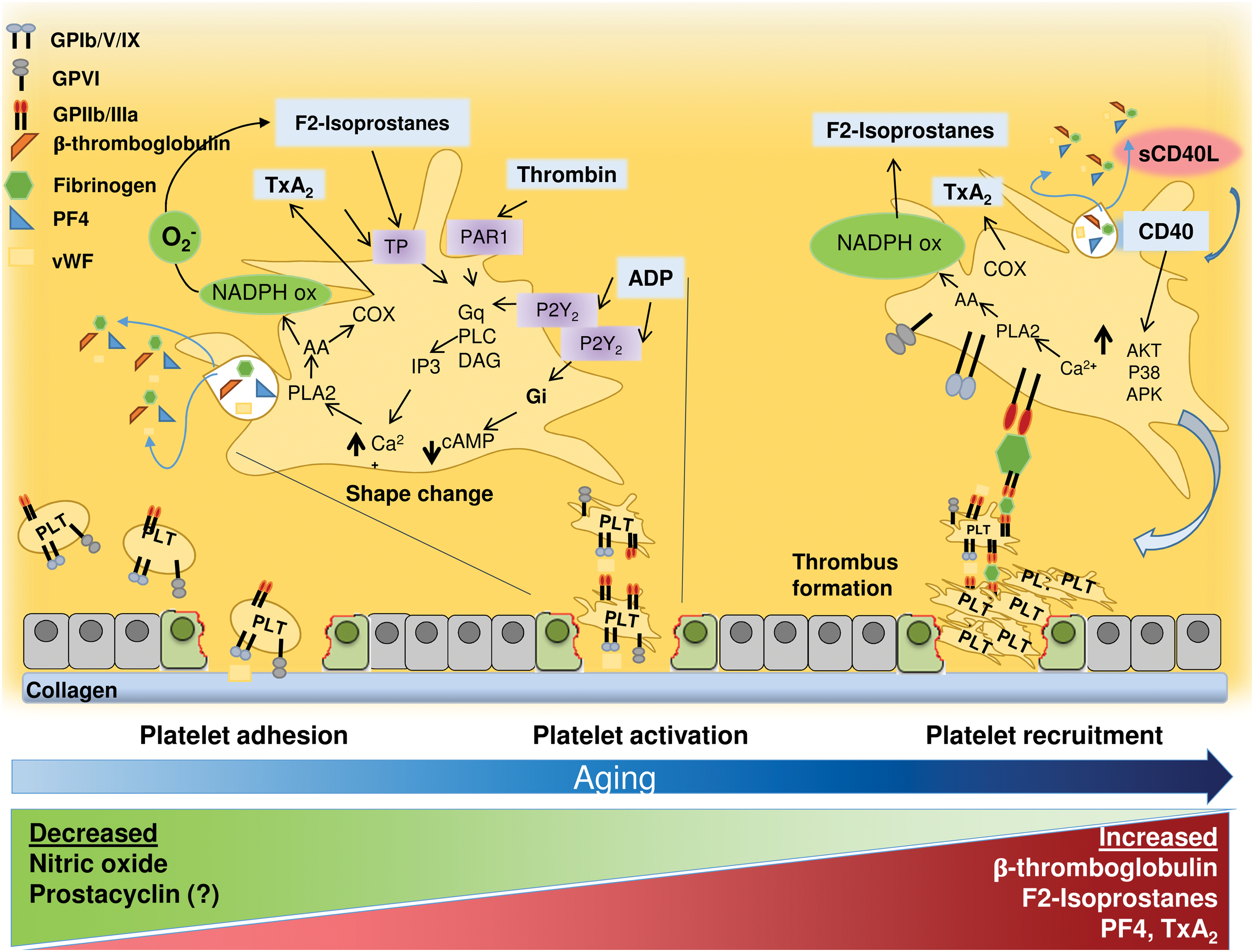
The initial step of primary hemostasis is platelet adhesion to the extracellular matrix. In particular, platelets roll, adhere, and spread on collagen matrix to form an activated platelet monolayer. This process is mediated by platelet receptors that bind extracellular matrix components, such as von Willebrand factor, collagen, fibronectin, thrombospondin, and laminin, which are exposed to blood on vascular injury. Once firmly adherent, platelets spread and release the content of their granules that contain pro-aggregating and pro-inflammatory molecules. Then, interaction of several agonists with the specific receptors expressed on platelets induces the propagation of platelet activation.
Platelet eicosanoids are important mediators of platelet aggregation and propagation. Once produced by adherent platelets, TxA2 amplifies platelet adhesion response by binding to TPα and TPβ receptors (the effects in platelets are mediated primarily through the α form) (260). TxA2 is synthetized from AA through phospholipase A2 (PLA2) and cyclooxygenase (COX)-1, and it can activate phospholipase C. This enzyme induces formation of second messenger inositol triphosphate and diacylglycerol that activate intracellular protein kinase C. The production of inositol triphosphate increases cytosolic levels of calcium. Peroxidation of membrane phospholipids leads to the generation of another eicosanoid family named F2-isoprostanes, which modulate platelet activation via TxA2 receptors (246). F2-isoprostanes are produced from AA by a free radical-mediated mechanism (246) that is likely dependent on Nox2 activation (280). Thus, reduced F2-isoprostanes platelet formation was detected in patients with CGD (280). In this clinical model, the reduced production of platelet F2-isoprostanes was associated to impaired platelet activation that was restored by adding exogenous F2-isoprostanes (280).
The recruitment phase depends on the release of several pro-aggregating substances such as adenosine diphosphate, eicosanoids, or ROS, which spread platelet activation at the site of thrombus growth.
Previous studies consistently showed that platelet activation is enhanced in the elderly population (243). Cross-sectional studies demonstrated an enhanced platelet response to common agonists as documented by a lower concentration needed to aggregate platelets in the elderly compared with the younger population (243). In accordance, Sverdlov et al. demonstrated a progressive increase of platelet sensitivity to common agonists in 204 subjects followed for 4 years (353). Further, proteins released by activated platelets such as Beta-thromboglobulin and platelet factor 4 were elevated in the elderly population compared with the younger one (243). Finally, bleeding time, which explores platelet activation in vivo, was found to be shorter in elderly patients (167). However, a limitation of these studies is that bleeding time reflects not only in vivo platelet activation but also vascular reactivity (112). Urinary excretion of 11-dehydro-TxB2 is among the most reliable tests of platelet activation in vivo. It is the stable metabolite of TxA2, and urinary excretion of 11-dehydro-TxB2 has been used for clinical purpose in patients with atherosclerotic risk factors such as DM, hypertension, and hypercholesterolemia (66, 128, 318), and in patients with acute and chronic CVD. In these clinical settings, urinary 11-dehydro-TxB2 was elevated, suggesting an increased platelet activation in patients at risk or with overt atherosclerosis (93). In a small cross-sectional study performed in 20 healthy subjects, Reilly and FitzGerald found increased urinary excretion of 2,3-donor-TxB2 in elderly (>65 years) compared with younger (<65 years) subjects (304). Differently from previous reports, ex vivo and in vivo tests of platelet function were similar in elderly and young patients. This finding is apparently in contrast with a previous study, which measured serum TxB2 in 177 patients with atherosclerotic risk factors and found no difference according to aging; however, small sample size and number of elderly patients limit data interpretation (5). To address the relationship between platelet activation and aging, we measured 11-dehydro-TxB2 in 833 patients affected by AF, which is associated with several atherosclerotic risk factors and poor vascular outcomes, such as thromboembolic stroke and MI (269). A cross-sectional analysis of life decades demonstrated that urinary 11-dehydro-TxB2 increased by aging with a significant elevation at the age of 74 years (269). Of note, during a follow-up of ∼5 years, patients with elevated TxB2 experienced more CVEs compared with those with lower TxB2. Thus, platelet age-dependent ROS production could represent an important mechanism accounting for atherothrombosis in the elderly.
The relationship between ROS and platelet activation was originally demonstrated by Del Principe et al. (83), who found increased platelet calcium mobilization by adding in vitro exogenous H2O2 to platelets. This finding was later confirmed by another in vitro study where sample treatment with catalase resulted in inhibition of agonist-induced platelet activation and calcium mobilization (286). This finding suggested that also antioxidant compounds or enzymes may modulate platelet function. Thus, direct antioxidants such as vitamin C and E significantly inhibited platelet aggregation (287, 289). Among the enzymatic pathways implicated in platelet ROS formation and aggregation, Nox2 has a prominent role as shown by the anti-platelet effects elicited by inhibition of Nox2 activity, which resulted in impaired production of platelet O2, lower calcium mobilization and GPIIb/IIIa activation, and, eventually, inhibition of platelet aggregation. The role of Nox2 was further investigated (288) in CGD patients, whose platelets displayed impaired platelet activation and thrombus growth (280). Nox2 is able to influence platelet activation through at least three different mechanisms that are relevant in platelet aging, including (i) O2 − dismutation to the more stable and pro-aggregating molecule H2O2, (ii) inhibition of the antiplatelet activity NO by O2 −, and (iii) nonenzymatic transformation of AA into F2-isoprostanes.
Analysis of platelet H2O2, which is implicated in platelet aggregation, via calcium mobilization, COX1 activation, and MTORC1, demonstrated an important role for these oxidant species in aging-related platelet aggregation (286, 414). Thus, prospective analysis in animals demonstrated that platelet H2O2 increases by aging coincidentally with an enhanced risk of thrombosis (78). A key factor for platelet H2O2 overexpression was activation of Nox2, which was, in fact, upregulated in elderly animals. In accordance, animals treated with apocynin, which inhibits p47phox translocation to Nox2, disclosed reduced platelet H2O2 formation and age-related thrombosis (78).
It is still to be determined, however, whether overproduction of platelet ROS by aging is a result of upregulation of enzymes producing ROS or reduction of antioxidant status. Recent studies support this latter hypothesis. As mentioned earlier, vasodilating properties by NO are rapidly lost on its interaction with O2 − to give formation of peroxynitrite (52, 382). Peroxynitrite rapidly induces tyrosine nitration of proteins with the same pattern of tyrosine phosphorylation, thus inducing platelet activation (244) Impaired NO generation by inactivation and/or reduced biosynthesis is associated with enhanced platelet aggregation (280). In a prospective study performed in 204 subjects followed up for 4 years, platelet responsiveness to NO was significantly reduced compared with baseline coincidentally with a progressive increase in plasma asymmetric dimethylarginine, which is an inhibitor of NOS (353). Alternatively, reduced platelet response to NO may be related to an impaired detoxification of intracellular ROS. The relevance of this phenomenon has been documented in previous reports in animals (162) and humans (78, 297) in which the deficiency of GPx was associated with serious thrombotic complications. In support of this, in animals overexpressing GPx1, platelet activation as well as platelet-related thrombosis were significantly inhibited, further reinforcing the hypothesis that impaired H2O2 detoxification favors occurrence of thrombotic process (78). The role of GPx in aging-related platelet activation has been documented by an observational study in patients with AF followed up for ∼40 months; thus, the activity of GPx3 progressively decreased by aging with a significant decline at the age of 75 (268). The decline of GPx3 was coincident with upregulation of Nox2 and enhanced platelet activation, suggesting that impaired H2O2 breakdown could result in enhanced Nox2-derived ROS formation and platelet Tx production (268).
Taken together, these data can lead us to hypothesize that aging is associated with platelet activation via an oxidative-stress-mediated mechanism. Reduction of GPx activity and increase of Nox2 upregulation seem to be important factors promoting platelet-derived thrombus formation. Thus, both may contribute with different mechanisms to enhance platelet eicosanoid formation and lower NO generation via its enhanced degradation or impaired biosynthesis (Fig. 7). Consequently, upregulation of GPx activity and/or downregulation of Nox2 may represent a potentially important tool to reduce aging-related platelet activation and, eventually, thrombosis-related CVD.
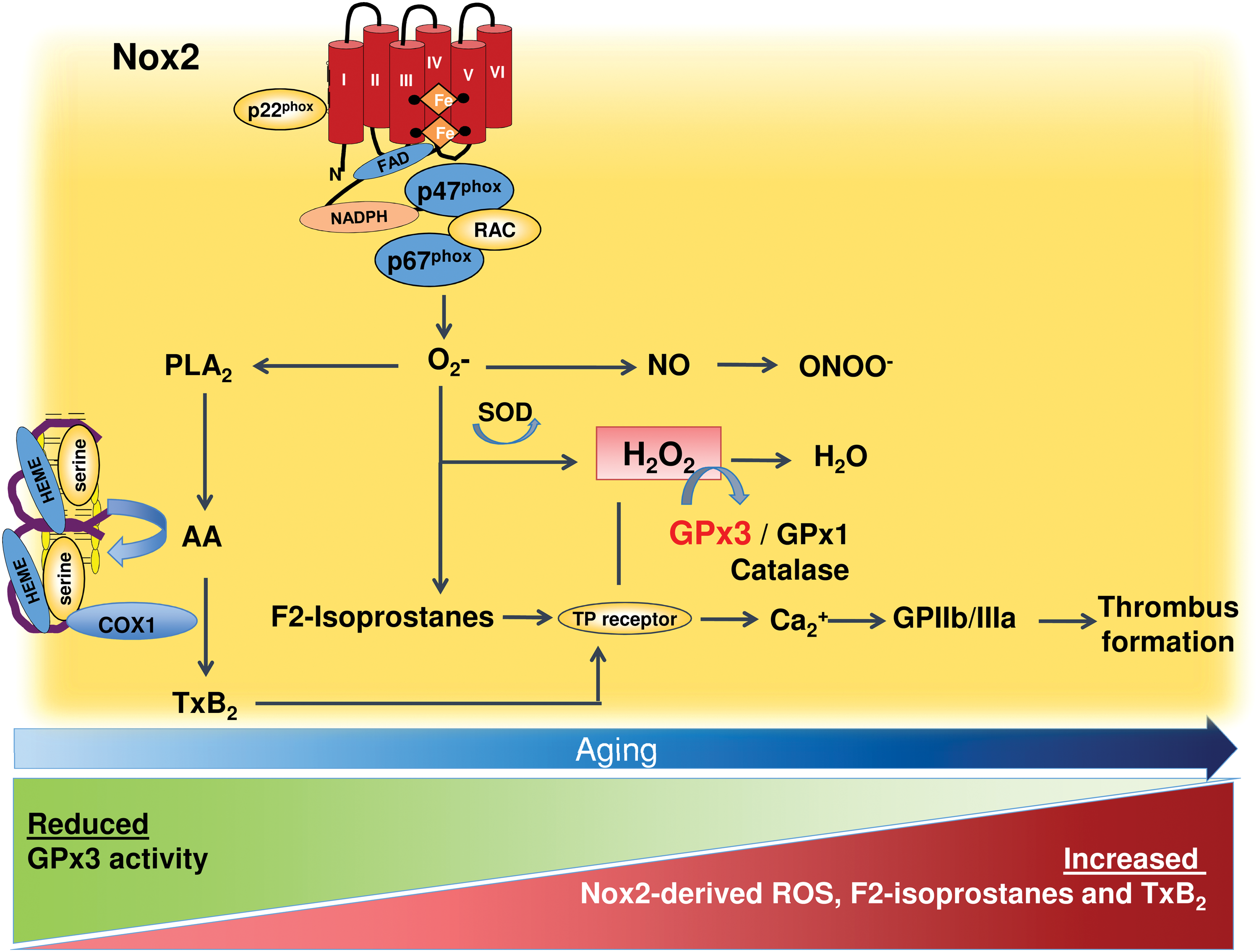
So far, there are few interventional studies investigating the effect of antioxidants on platelet function, and results are not univocal. Jain et al. demonstrated that 100 IU/day of vitamin E significantly reduced blood TxB2 in patients with type 1 DM, suggesting that vitamin E exerts an antiplatelet activity via inhibition of platelet AA metabolism (158). This finding has been confirmed and extended by Davì et al., who administered 600 mg/day of vitamin E to 85 type 2 DM patients for 2 weeks (74); the authors found a significant decrease of urinary excretion of 11-dehydro-TxB2 in patients compared with controls. Conversely, Patrignani et al. reported no effect of vitamin E supplementation on urinary 8-iso-PGF2α or 11-dehydro-TxB2 excretion in 46 moderate cigarette smokers (271).
XI. Endothelial Dysfunction
Endothelium integrity/function has been classically identified as one major determinant factor to maintain vascular homeostasis. Under physiologic conditions, eNOS is responsible for maintaining vasodilation by producing NO; NO activates soluble guanylyl cyclase, which converts guanosine-5′-triphosphate to cyclic guanosine monophosphate and, eventually, decreases smooth muscle tension, causing vasodilatation (116).
The eNOS may be activated by changes in shear forces or by binding of some molecules (such as acetylcholine, adenosine, bradikinine) to membrane receptors, which cause an increase in calcium levels in endothelial cells (240). Increased calcium levels also activate PLA2, which generates AA from membrane-bound phospholipid and, eventually, prostacyclin, a powerful anti-aggregating and vasodilating molecule, via COX2 activation (Fig. 6).
In addition to NO, endothelial cells produce small oxidant vasoactive molecules, such as O2 − (vasoconstrictive) and H2O2 (vasodilating). Laminar shear stress promotes H2O2 generation, which, in turn, activates p38 MAPK and, ultimately, NOS, leading to vasodilation (39). As mentioned earlier, the balance between NO and O2 − is crucial, as an increase in O2 − has a detrimental effect on NO bioactivity/biosynthesis or elicits eNOS uncoupling.
Endothelial cells produce O2 − prevalently via several Nox isoforms such as Nox1, Nox2, Nox4, and Nox5, which may contribute toward modulating arterial dilatation with different mechanisms (43, 179) (Fig. 8). In this regard, we measured flow-mediated dilatation (FMD), which is dependent on endothelial release of NO and is a surrogate marker of atherosclerosis (364), in CGD patients with Nox2 (X-linked) (385) or p47phox hereditary deficiency (209). CGD patients showed enhanced FMD, which, however, was more marked in Nox2-deficient patients, suggesting a relationship between the degree of ROS formation and artery vasoconstriction (209, 385). The mechanism accounting for enhanced artery dilatation was attributed to heightened NO generation, which was also suggested to account for enhanced vasodilation detected in knockout animals for Nox2 (59, 229). The close relationship between endothelial dysfunction and Nox2 has been studied in several conditions such as dyslipidemia, obesity, smoking (208), hypertension, MetS, DM, PAD (207), and obstructive sleep apnea (82). These classic cardiovascular risk factors provoke endothelial dysfunction since childhood, and the coexistence of Nox2 upregulation suggests this enzyme as a potential trigger (210, 214, 226). Indeed, children with hypercholesterolemia, obesity, or obstructive sleep apnea displayed Nox2 upregulation coincidentally with a reduced FMD (210).
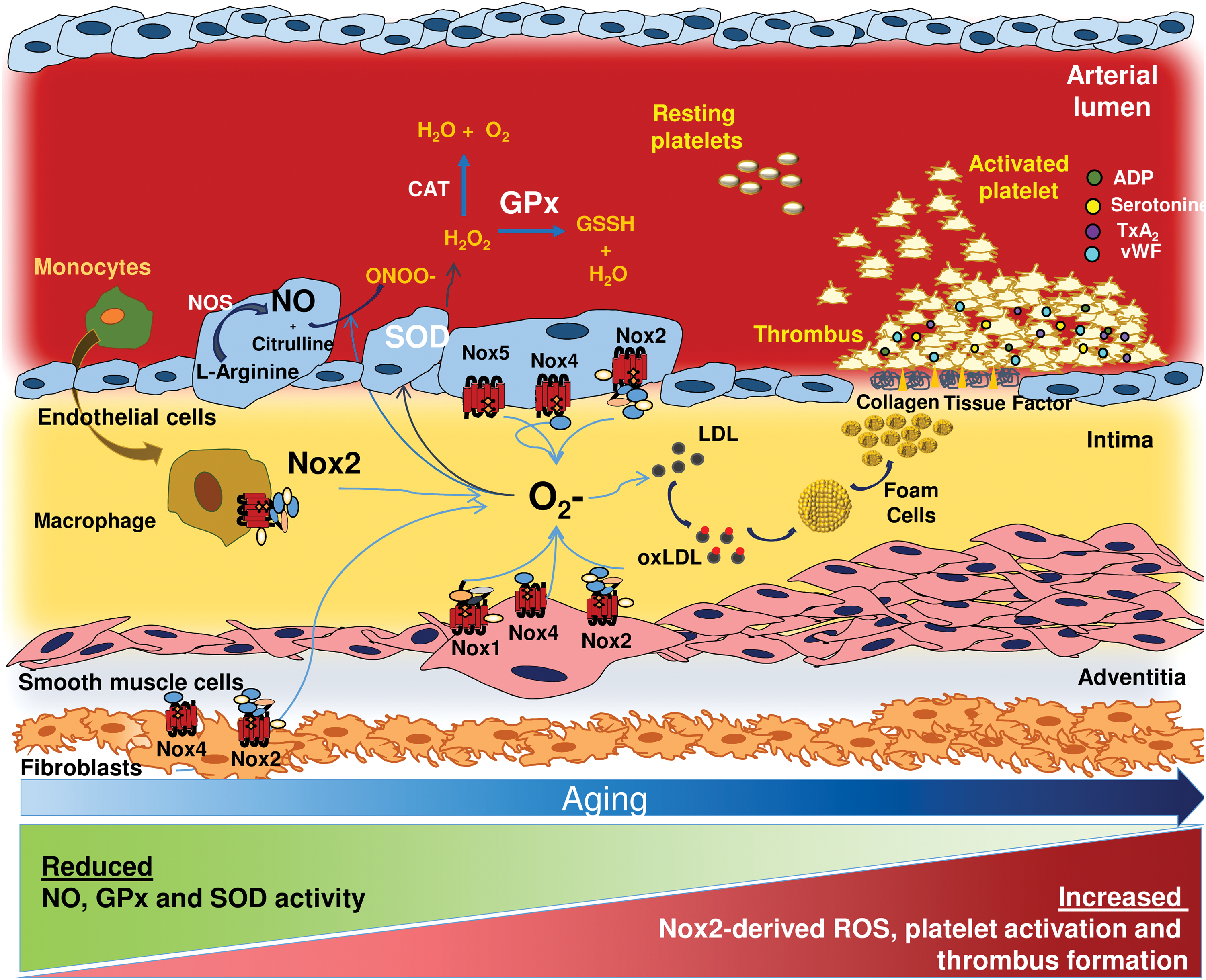
Nox4 plays a peculiar role as this isoform produces H2O2 rather than O2 −; hence, differently from Nox2, Nox4 has vasodilating property via eNOS activation (323). Animal studies showed that genetic deficiency of Nox4 was associated with endothelial dysfunction and increased atherosclerosis burden (194).
Other Nox isoforms, such as Nox1 and Nox5, may concur to vasodilation. Nox 1 is mainly located in VSMC where it enhances production of O2 −, causing eNOS uncoupling and consequent impaired vasorelaxation (86). The effect of Nox1 on endothelial function was evaluated by studies in mice with genetic deficiency of Nox1 that showed an increased vasodilatory response to acetylcholine (229). In endothelial cells, Nox5 directly produces O2 − in response to an intracellular increase of calcium levels, without requiring the activation of other subunits (91). The biological role of Nox5 in vasodilation remains unclear, but O2 − produced by Nox5 contributes to determining endothelial dysfunction by inhibition of extracellular NO (245) and, paradoxically, activates eNOS, increasing NO generation (420).
Natural antioxidants such as SOD, catalase, and GPx serve to counteract oxidative stress in the endothelium (114). In particular, SOD3 prevents inactivation of NO by O2 − in the extracellular space; SOD1 preserves NO levels within the endothelium; and SOD2 protects against oxidative stress-mediated mitochondrial dysfunction (114).
Aging is characterized by a progressive arterial loss of function and structural changes, which may strongly concur to atherothrombosis. Thus, several studies showed that, compared with young healthy adults, artery dilation is impaired in elderly people (60, 256, 326). Classic cardiovascular risk factors can accelerate the age-related modifications of the artery, favoring the onset and progression of atherosclerosis and CVD (250). A large cross-sectional study conducted in 5314 Japanese adults showed that endothelial function decreases with age decades (20–70 years) in close relationship with cumulative cardiovascular risk factors (227). In particular, the study found that age, body mass index, systolic blood pressure, DM, smoking and baseline brachial artery diameter were independent predictors of FMD (206).
Functional impairment of age-related artery dilatation may be related to an imbalance between ROS formation and detoxification, which eventually results in vascular oxidative stress. For instance, as Nox2 is upregulated by aging, it would be arguably an implication of this enzyme in favoring artery dysfunction. Experimental studies are in support of this as evidenced in cholesterol-fed animals knockout for Nox2, which displayed enhanced artery dilation compared with wild type (237). Conversely, antioxidant status seems to be implicated in age-related endothelial dysfunction. Thus, Barton et al. (22) found that plasma endothelin-1 levels increased whereas plasma SOD activity decreased with aging coincidentally with impaired endothelium-dependent artery dilatation and NO release by rat aortic endothelial cells (22). Interestingly, they also found that SOD activity and relaxations to acetylcholine were preserved in femoral arteries, indicating a heterogeneity in endothelial function and oxidative stress related to size and sites of the arteries (22). Further support to the pivotal role of SOD in aging was provided by animal studies showing that SOD mimetics restored endothelial function in elderly mice (198, 362). Endothelial dysfunction by aging can also be dependent on GPx regulation (138) as shown in animals knockout for GPx, which displayed impaired NO bioavailability in elderly compared with younger animals and, eventually, vascular dysfunction (259).
Others factors potentially contributing to endothelial dysfunction by aging are related to a link among short length of telomeres, senescence, and endothelial dysfunction (415). The exact mechanism through which telomeres would determine endothelial dysfunction and CVD remains unclear. However, it was suggested that cardiovascular risk factors may accelerate telomere shortening through increased oxidative stress (415). Also, the human abdominal aorta (14) displays typical alterations of senescent cells such as age-dependent telomere erosion and consequent reduction of telomere length (14).
Doppler ultrasonography is a first-level examination to assess the atherosclerotic burden; one surrogate marker of atherosclerosis is the carotid artery IMT, which was directly correlated with the occurrence of CVEs, such as stroke and MI (215). There is evidence that carotid IMT increases by aging (228, 373, 393) as reported by the Bogalusa study, which compared the progression of IMT in childhood and adults with cardiovascular risk factors (202). Autoptic studies showed that aortic IMT increased with aging even in populations at a low risk of atherosclerosis (387) and, in accordance with this, the Baltimore Longitudinal Study on Aging demonstrated a two- to threefold carotid IMT increase from 20 to 90 years of age (106, 192). An increase of aging-related carotid IMT in the general population was also confirmed by other studies that estimated a rate of IMT growing >0.01 mm/year (102, 149, 366).
There is evidence that an imbalance between ROS formation and detoxification (10, 416) is associated with IMT increase. Thus, a recent nested case-control study by Yoon et al. showed that IMT was associated with oxidative stress markers such as urinary 8-hydroxy-20-deoxyquuanosine, malondialdehyde, and 8-iso-PGF2α (416). Further, Ashfaq et al. (10) reported that the ratio between reduced/oxidized form of GSH was an independent predictor of IMT. Upregulation of Nox2 could be another potential mechanism accounting for a progressive increase of IMT. In patients with CGD, a reduced carotid thickness was detected by ultrasonography and magnetic resonance imaging (337). Further, in children with obesity and hypercholesterolemia or in subjects without clinically overt atherosclerotic disease, Nox2 activation was significantly associated with carotid IMT (210, 419). However, prospective studies are needed to substantiate the role of Nox2 as a predictor of IMT progression.
Aging is also associated with arterial structural changes such as an increase of luminal enlargement and wall stiffness (192). Pulse wave velocity (PWV), the velocity of pulse propagation through the arteries, is considered a noninvasive index of vascular stiffness. Cardiovascular risk factors, such as DM, smoking, and hypertension, increase PWV, particularly in the elderly population (151, 204). The structural changes of the artery wall, which accounts for increased PWV, are probably dependent on an increase of collagen content, reduction of elastin, and calcification by aging. Thus, an impaired synthesis and accelerated degradation of elastin (250), and a higher deposition of collagen fibrils were observed in aged arteries (250). Changes in the content of collagen fibrils and elastin were studied in the human thoracic aorta by cross-link products (250). From 0–19 to 80 years, an increase of about 100% of collagen cross-link product and a decrease of about 50% of elastin cross-link products were observed, confirming a loss of elasticity during aging (250). These changes can be detected in large- and medium-size arteries that become less distensible, with luminal dilatation and being somewhat hypertrophic (320).
Oxidative stress may be implicated in impaired arterial elasticity (270, 407) as it directly correlates with enhanced arterial stiffness in healthy subjects (270, 407). Further, Nox2 deficiency is associated with reduced impairment of age-dependent neovascularization in mice (371).
Taken together, these data suggest that aging is associated with important alterations of endothelial homeostasis (94), which are shifted toward loss of dilatation function and elasticity and enhanced vasoconstriction. Due to the relevant role of oxidative stress in endothelial homeostasis, antioxidants might favorably influence endothelial function. Antoniades et al. demonstrated, for example, that 2-week administration of 2 g vitamin C and 800 IU vitamin E improved forearm blood flow in chronic smokers; this effect seemed to be mediated by an upregulation of NOS (8). Similar effects have been detected by polyphenol administration, which increased FMD (203) via Nox2 downregulation and, eventually, enhanced NO bioavailability (211).
Experimental studies with other antioxidants such as polyphenol supported the potential usefulness of these molecules to counteract age-related artery dysfunction. Thus, a chronic intake of polyphenols protected against aging-induced endothelial dysfunction and decline of physical performance via Nox inhibition and upregulation of eNOS in rats (72, 73). No human study has so far analyzed the effect of polyphenols on aging and endothelial function.
XII. Antiplatelet and Anticoagulant Treatments
The modifications induced by aging in platelet function may have clinical and therapeutic consequences, as the increased platelet activation may result in a lower efficacy of antiplatelet in elderly patients compared with younger ones. The effect of aspirin, which inhibits COX1, thus preventing the formation of TxA2, has been investigated in a meta-analysis of randomized clinical trials (7). Despite an apparent similar efficacy of aspirin in the young (−13%) and elderly (−12%) population, it was speculated that aspirin could be even more effective in the elderly population because the younger population are at a lower absolute risk (7). However, data analysis is not consistent with this interpretation as aspirin efficacy was significant only in patients <65 years (hazard ratio [HR] 0.87, 95% confidence interval [CI] 0.78–0.98) compared with those aged ≥65 years (HR: 0.88, 95% CI 0.77–1.01) (7). This different efficacy seems to be also evident for thienopyridines, which are reversible (ticagrelor) or irreversible (clopidogrel and prasugrel) inhibitors of the platelet receptor P2Y12 (Table 3). The Dual Antiplatelet Therapy (DAPT) trial included 9961 patients who underwent a coronary stent placement and were randomly assigned to continue thienopyridine treatment or to receive a placebo in addition to aspirin. The study showed a lower efficacy of antiplatelet therapy in reducing CVEs and CD in patients <75 (n = 8929) versus ≥75 years (n = 1032) (230). Similarly, the CLARITY–TIMI 28 trial (314), which included 1752 ACS patients with ST-segment elevation, showed that clopidogrel was less effective in 1015 patients ≥65 years than in 2466 patients <65 years (314).
ACS, acute coronary syndrome; CI, confidence interval; FU, follow-up; HR, hazard ratio; MACCE, major adverse cerebrovascular and cardiovascular events; MI, myocardial infarction; RR, relative risk.
Similar results were obtained by three recent studies, which explored the clinical efficacy of ticagrelor (Table 3). The PLATO study (391) included 18,624 patients with ACS without ST segment elevation, to compare the efficacy of ticagrelor versus clopidogrel during a follow-up of 30 days. The study showed a favorable effect of ticagrelor in patients <75 years (n = 15744) in reducing the composite endpoint of CV death, MI, and stroke, which was not significant in those aged ≥75 years (n = 2878).
This different efficacy was also observed in the SOCRATES trial (164), which included 13,199 patients with nonsevere ischemic stroke or high-risk transient ischemic attack treated with ticagrelor versus aspirin in a 90-day follow-up. Patients treated with ticagrelor had significantly lower CVEs, but this positive effect was age related; thus, the trial reported a lower efficacy of ticagrelor in patients >75 years (n = 2995) versus those <65 years (n = 6028). Together, these data seem to indicate a sort of “antiplatelet resistance” in the elderly population, which needs to be further investigated.
In addition to antiplatelet drugs, elderly patients are often treated with oral anticoagulants such as vitamin K antagonists or non-vitamin K oral anticoagulants (NOACs), which inhibit thrombin or factor Xa. The efficacy and safety of NOACs have been investigated in patients with deep venous thrombosis, AF and ACS with an efficacy at least comparable to warfarin, along with a better safety profile. Such beneficial effects seem to be independent from age and may be explained by the fact that NOACs, in particular the Xa inhibitors, possess not only an anticoagulant but also an antiplatelet effect, which may turn useful in patients with upregulation of platelet and clotting activation as detected in the elderly AF population (283).
XIII. Observational and Interventional Trials with Antioxidants in Humans
The efficacy of antioxidants for the prevention of CVEs and atherosclerosis progression was largely investigated. Vitamin E was the most studied molecule among the antioxidant vitamins (vitamins A, C, E, and β-carotene). The strong interest for vitamin E was based on observational studies reporting an inverse association between vitamin E plasma levels, CHD, and incidence of CVEs (380). The WHO/MONICA project found an inverse correlation between CHD mortality and vitamin E plasma levels (121). Conversely, no association was observed between CHD and other vitamins (vitamin A, C, and β-carotene). Similarly, Singh et al. found that in a population of 595 urban Indians, plasma levels of vitamins C and E were inversely related to CAD (340). Finally, blood vitamin E levels predicted CVEs in 1012 elderly AF patients during 27 months of follow-up (51). In addition to vitamin E, low blood vitamin C levels were found to be predictive of cardiovascular mortality in an elderly British population (107).
At variance with this, meta-analyses of interventional trials with vitamin C and E provided equivocal results (249) and, paradoxically, potentially harmful effects as enhanced risk of all-cause mortality (31, 32) or hemorrhagic stroke (322). However, in many trials, a combination of antioxidant vitamins was used on the assumption that more antioxidants could synergically enhance the antioxidant status potentially achievable by a single antioxidant (296). Actually, a combination of antioxidants may negatively influence the activity of a single antioxidant. For instance, a combination of vitamin E with vitamin C was shown to be not effective for cardiovascular prevention in The Physicians' Health Study II (329); this negative effect might depend on the fact that vitamin C may exert a pro-oxidant activity when orally administered to humans, thus impairing vitamin E antioxidant property (291, 296). A harmful effect was even detected by combining vitamin E with beta-carotene, which was, in fact, associated with enhanced risk of total mortality (388). The fact that vitamin E could be useful when given alone is suggested by a more recent meta-analysis, where vitamin E supplementation seemed to be associated to a reduction of MI (212).
Other reasons for inconsistency of interventional trials with antioxidant vitamins rely on methodological issues. Thus, potential bias of the interventional studies with vitamin E could be dependent on the concomitant use of statins, which were given in association with vitamin E in about one out of three patients (388). Thus, an experimental study demonstrated that statins reduce oxidative stress and improve circulating levels of vitamin E, thus minimizing its putative antioxidant benefit (388). Further, interventional trials did not take into account that vitamin E is poorly absorbed if not assumed with meals (156). This issue has never been considered by interventional trials as scarce data regarding vitamin E bioavailability have been reported. In the absence of such information, it cannot be excluded that a poor bioavailability of vitamin E may represent another explanation for its lack of efficacy. Finally, none of the interventional trials measured the antioxidant status at baseline; therefore, it could not be appreciated whether patients included in the studies disclosed low levels of vitamins and actually needed antioxidant supplementation.
Several epidemiological studies showed that a diet rich in polyphenols reduces CVEs and cardiovascular mortality (9, 25, 44, 45). These beneficial effects have been observed particularly with the Mediterranean Diet (Med-Diet), which is rich in polyphenols for its high content of fruit, vegetables, extra-virgin olive oil (EVOO), and moderate red wine consumption. Thus, in the Seven Countries study (172), Ancel Keys described for the first time that people from the Mediterranean area (Greece, Italy) experienced a lower mortality rate for CVD, when compared with populations living in Northern Europe (172). The Lyon Diet Heart Study, which was the first interventional trial that studied the effect of Med-Diet on CVD in patients affected by MI, showed that Med-Diet reduced cardiovascular complications by 50% (80). Recently, the PREDIMED trial investigated the effect of Med-Diet on CVEs (99) by randomizing 7447 people at high vascular risk to Med-Diet supplemented with EVOO, mixed nuts, or control diet (99). Med-Diet reduced the risk of CVD complications by 30% over a follow-up of about 5 years in the two arms supplemented with EVOO or nuts (99). Of note, a subgroup analysis of PREDIMED showed that, compared with control diet, Med-Diet was particularly effective in preventing major CVEs in people >70 years (99). Consistent with this finding, we recently demonstrated that adherence to Med-Diet reduced the risk of CVE in an elderly population affected by AF (264).
Cocoa is another polyphenol-rich nutrient that seems to possess beneficial effects on the cardiovascular system (213). The Iowa Women's Health Study reported an inverse relationship between chocolate intake and CHD mortality in postmenopausal women (239). Accordingly, the European Prospective Investigation into Cancer and Nutrition (45) showed a lower rate of MI and stroke in subjects with the highest chocolate consumption. Moreover, an observational trial in elderly people, the Zutphen Study, showed that cocoa intake was inversely related to cardiovascular mortality (44). Finally, the Stockholm Heart Epidemiology Program (160) reported a reduction of cardiovascular mortality in a population of nondiabetic subjects with previous MI taking a high amount of chocolate. However, due the observational nature of these findings, the effect of polyphenols contained in cocoa on cardiovascular protection needs further evaluation by interventional randomized trials (213).
Another polyphenol largely studied for its antioxidant property is resveratrol, which is mostly contained in red wine. The “French paradox” ascribed to resveratrol the beneficial cardiovascular effects provided by red wine (305). However, this hypothesis was not confirmed in elderly people by the “Invecchiare In Chianti (inChianti) Study” that did not find any effect on mortality risk and CVD in 783 subjects who regularly drink red wine (327). Further investigation on the possible beneficial effect of resveratrol on CVD is, however, needed considering that, as for other antioxidants, interventional trials provided likely inconsistent results for several reasons, including study methodology, dosage, and subjects' selection (29).
Green and black tea are other polyphenol-rich nutrients that are believed to exert cardiovascular protection (346). Epidemiological studies in the elderly showed that tea consumption reduces the risk of CVD and total mortality (142, 347). However, a meta-analysis of 17 studies by Peters et al. did not find an association between tea intake and CVD (277). Large prospective and interventional studies are necessary to evaluate the effect of tea on the cardiovascular system.
XIV. Future Perspectives and Conclusions
Data here reported suggest that platelet and clotting activation along with endothelial dysfunction are typical features of the elderly population, which contribute toward enhancing the risk of cardiac and cerebral ischemic complications. Even if the modern approach to treat or prevent thrombosis with single or dual antiplatelet agents or with NOACs achieved positive results in terms of reduction of CVD, an elevated residual risk still remains. In this context, experimental and clinical data are in favor of the hypothesis that systemic inflammation and oxidative stress tends to worsen with advancing age and can precipitate acute events via activation of the platelet and clotting system. As classic atherosclerotic risk factors are recognized as the most important triggers of systemic inflammation and oxidative stress and, considering that they frequently overlap in the elderly population, careful attention should be given to optimize adherence to anti-atherosclerotic treatments. This issue deserves careful attention as the drug's compliance is drastically reduced in case of multiple treatments as shown; for instance, adherence to aspirin prescription was significantly lowered when patients were treated with >5 pills/day (282).
Gut microbiota is another emergent risk factor that was recently proposed as contributing to systemic inflammation and oxidative stress with at least two different mechanisms: (i) gastrointestinal colonization of bacteria-producing inflammatory molecules; (ii) enhanced gut permeability due to abnormal changes of local circulation with ensuing translocation into systemic circulation of pro-inflammatory and pro-oxidant molecules such as TMAO or LPS. Although changes of gut microbiota occurring in the elderly have been described, no data regarding blood levels of TMAO or LPS in the elderly population have been reported. Due to the relevance of diet and changes of gut composition by advancing age, these data would be of particular interest also in view of developing new approaches to lower the risk of CVD in the elderly population.
Systemic inflammation and oxidative stress could also be counteracted by targeting specific oxidant pathways that are potentially implicated in atherothrombosis. Among them, an intriguing attractive approach is represented by the inhibition of Nox2, which is upregulated and is associated with platelet activation in the elderly (Fig. 9). Thus, experimental studies demonstrated that inhibition of Nox2 results in delayed thrombus growth, suggesting Nox2 as a target to modulate platelet function. A matter of concern is represented, however, by the role played by Nox2 in the innate system, as a complete suppression of Nox2 activity is associated to serious life-threatening infection disease as depicted by clinical history of patients with Nox2 hereditary deficiency (280). It is interesting, however, that in case of 50% reduction of Nox2 activity, as observed in Nox2 deficiency carriers, platelet inhibition was similar to that found in patients with complete Nox2 absence, in the absence of serious infection complications. Thus, it might be possible that 50% Nox2 inhibition would be appropriate to achieve effective platelet inhibition and be clinically safe (55).
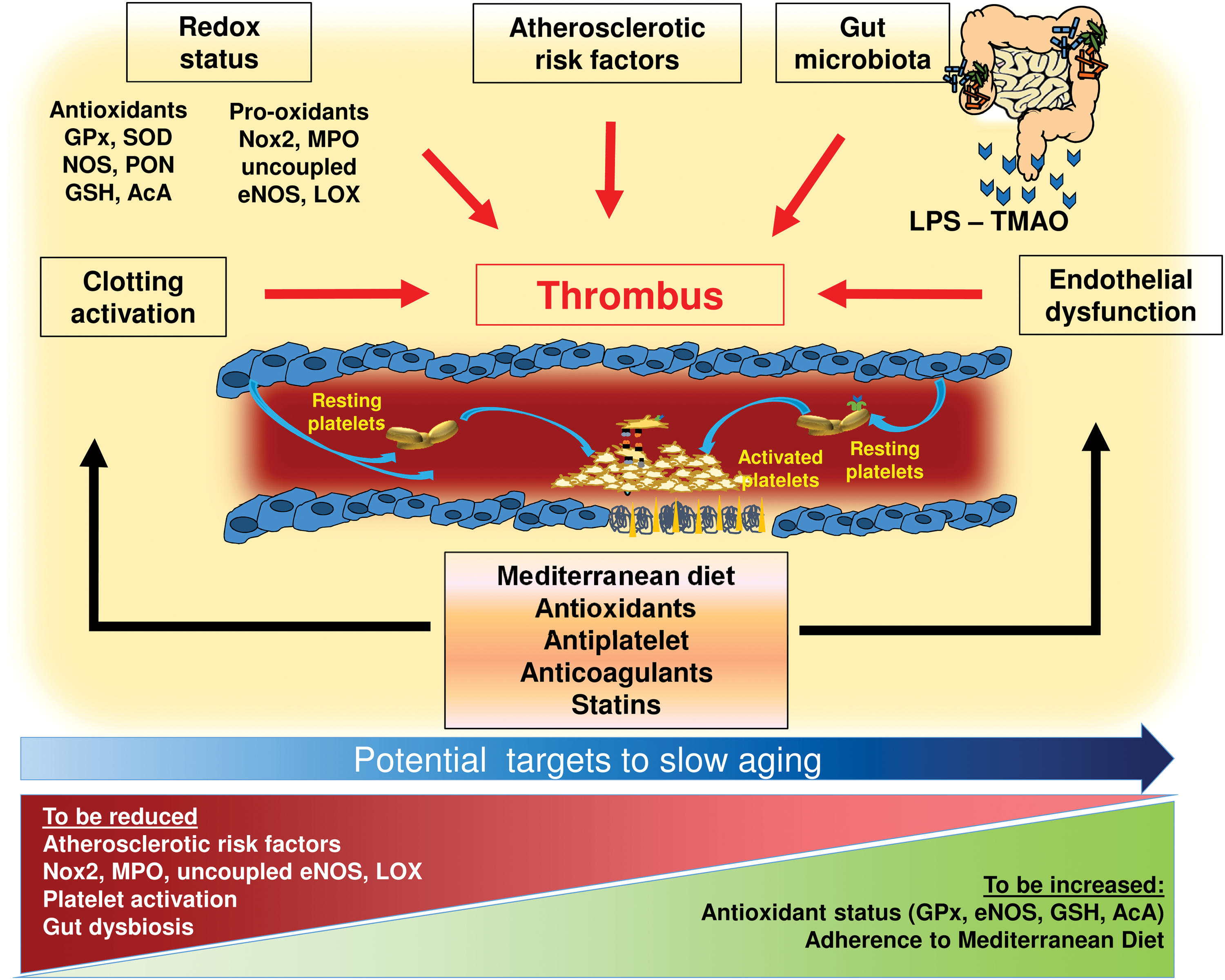
Among drugs modulating platelet function by interfering with Nox2, statins represent a promising candidate as these drugs inhibit the activity of the Nox2 subunit RAC1 (281). Thus, in a randomized study in hypercolesterolemic patients, 40 mg atorvastatin ingestion was associated to immediate downregulation of Nox2, along with inhibition of platelet isoprostanes (281). Similar results were obtained by using rosuvastatin, another powerful lipid-lowering molecule (279). However, prospective studies are needed to investigate whether the protective effects of statins against CVD are also attributable to Nox2 inhibition. Another therapeutic option could be represented by apocynin, a molecule that reduces p47phox translocation to the catalytic site of Nox2. This approach would have less negative impact on the innate immune system as indicated by the more favorable clinical history of patients with hereditary deficiency of p47phox (185). Few studies analyzed the effects of apocynin in humans. In these studies, apocynin was nebulized and administered by inhalation to healthy subjects or asthmatics to examine its effect on ROS levels in exhaled breath condensate (276, 348, 349). In each study, apocynin administration for a few hours was well tolerated, and it effectively reduced exhaled ROS levels (276, 348, 349). Up to now, chronic systemic administration of apocynin in human has never been tested.
Another factor contributing to atherothrombosis in the elderly is the continuous decline of antioxidant status. Theoretically, increasing antioxidant status may represent a novel therapeutic option but, as reported earlier, data on supplementation with antioxidant vitamins are equivocal. However, other potential therapeutic options include supplementation with GSH precursors, which is reduced in the elderly and is predictive of CVEs. In this regard, a potential therapeutic approach is represented by N-acetylcysteine (NAC), a direct precursor to GSH synthesis frequently proposed as a cardio-protective agent (95, 381). This compound is a nutritional supplement that acts by raising the intra-cellular concentration of cysteine/GSH, and by scavenging oxidant species (95). Its pharmacological actions include restoration of cellular antioxidant potential by replenishing GSH depleted by ROS scavenging and inhibition of neutrophil activity and TNFα production (95). Preliminary data suggest a potential role of NAC in preventing postsurgery AF (381), but further data are necessary to explore the effect of NAC on CVEs. An alternative approach would consider upregulation of specific enzymatic pathways that exert antioxidant effects by scavenging intracellular oxidant species (78). In this regard, an experimental study provided interesting data, as GPx1 overexpression resulted in impaired platelet activation and thrombus growth (78). Therefore, upregulating GPx or other antioxidant enzymes such as PON1, NOS, or HO may represent an option to be considered in future.
We have interesting information, however, on the effect of an antioxidant-rich diet such as Med-Diet on oxidative stress and platelet function. Indeed, adherence to Med-Diet was associated with an antioxidant effect in a large prospective cohort study including elderly AF patients, by reduced Nox2 activity and urinary 8-iso-PGF2α levels (264). Of note, adherence to Med-Diet was also associated to impaired production of TxB2, suggesting that downregulating Nox2-derived oxidative stress would result in impaired platelet activation (285). Taking into account that Nox2 upregulation and platelet overactivation are coincident in the elderly population, this diet could be advised particularly in the elderly population. It is still unclear, however, whether such beneficial effects may be attributed to the Med-Diet per se, or to its specific nutrients. EVOO could be effective through several mechanisms, including an anti-diabetic, antioxidant, or antiplatelet effect. Regarding the first point, intake of EVOO has been shown to improve postprandial glycemic control with a mechanism involving upregulation of glucagon-like peptide 1 and insulin secretion (54). Further, we found that EVOO exerts an antioxidant activity by downregulating Nox2-derived oxidative stress and that its intake is associated with impaired platelet activation (56). Thus, identification of EVOO components implicated in this antioxidant effect could lead to discovering new molecules to be used for anti-anti-atherosclerotic purposes.
Finally, considering the reduced availability and ability to migrate of EPCs in elderly with CVD, EPC cell therapy may be a promising approach for the treatment of CVDs. However, trials performed so far provided inconclusive results; this may be dependent on the age-dependent functional impairment in terms of proliferation, migration, and survival capacity of EPCs, which limits the efficacy of autologous EPCs in the treatment of CVDs. To overcome these limitations, several EPC enhancement strategies have been tested in experimental models [reviewed in detail in Rurali et al. (311)]. These approaches aim at enhancing proliferation, migration/homing, and/or differentiation of EPCs. They can be broadly classified into (i) strategies that can improve EPC function, operating at the ischemic tissue levels (namely in vivo conditioning), and (ii) approaches acting directly on EPCs before their in vivo administration (namely ex vivo priming). Despite the potential interest for this approach, clinical studies are needed to support the beneficial effects.
In conclusion, the elderly population is characterized by a pro-thrombotic phenotype that is dependent on platelet and clotting activation and artery dysfunction. An imbalance between ROS formation and antioxidant status occurs by aging and may contribute to this pro-thrombotic status. Targeting specific pathways implicated in ROS formation or counteracting age-related decline of specific antioxidant enzymes would represent a novel approach to assess whether oxidative stress is a mere mirror of vascular disease or is actually implicated in promoting atherosclerosis and thrombosis.
Footnotes
Acknowledgment
The authors would like to thank Dr. Vittoria Cammisotto for assistance in the preparation of figures.
