Abstract
Introduction
A
The proteasome is one of the two constituents of the ubiquitin-proteasome system (UPS, the other constituent being the ubiquitin system) responsible for the degradation of a pleiad of both normal and abnormal (in any way) proteins. The main proteasome core is called 20S complex, a barrel-shaped structure with seven different α- and seven different β-subunits, arranged in an α1–7β1–7β1–7α1–7 order. The complex carries the peptidylglutamyl peptide hydrolyzing (PGPH), trypsin-like (T-L), and chymotrypsin-like (CT-L) proteolytic activities hosted by β1, β2, and β5 catalytic subunits, respectively, with CT-L being the major one. Upon attachment of one or two 19S regulatory complexes at the end of the 20S core, the 26S and the 30S complexes are produced to exert ATP- and ubiquitin-dependent degradation (59).
Proteasome activation is feasible in multicellular organisms exerting antiaging and antiaggregation activities. However, in all studies, activation has been succeeded via genetic overexpression of various proteasome subunits. This is the first report of the use of 18α-glycyrrhetinic acid, a diet-derived compound with proteasome-activating properties serving as prolongevity and antiaggregation factor in a multicellular organism. Dietary available active compounds may be supplied through normal diet from young ages, thus having the potential to block the initial stages of misfolded/damaged/oxidized protein accumulation (for aging) or oligomer/aggregate formation (for Alzheimer's disease) before the senescent phenotype or the disease is established.
With the exception of the yeast regulatory particle non-ATPase protein (RPN-4) that acts as the main proteasome transcription factor (38), proteasome subunit expression regulation in higher organisms, including humans, is not entirely elucidated. Several other factors have been implicated in the transcription of proteasome subunits under specific conditions, with skinhead-1 (SKN-1, in Caenorhabditis elegans)/nuclear factor (erythroid-derived 2)-like 2 (Nrf2, in mammals) being one of them (26, 34). More specifically, SKN-1/Nrf2 is implicated in the induction of genes involved in electrophile detoxification and antioxidation, thus regulating the cellular response to oxidative stress (56). Upon proteasome inhibition, SKN-1 translocates to the nucleus to induce proteasome subunit expression (26, 35). Likewise, most of the mammalian proteasome genes are regulated by Nrf2 in response to antioxidants such as 3H-1,2-dithiole-3-thione (D3T) (34). We have previously shown that SKN-1 is involved in proteasome activation in C. elegans following pbs-5 overexpression (8). Moreover, treatment of human fibroblasts with 18α-glycyrrhetinic acid (18α-GA) triterpenoid promotes Nrf2 activation, leading to proteasome activation, and enhanced cellular stress resistance and life span (27). Therefore, compound-mediated targeting of Nrf2 is a plausible strategy to achieve proteasome upregulation.
Proteasome levels and/or function have been documented to decline with aging in vitro and in vivo (12), as well as during several age- and aggregation-related diseases, including AD (47). On the other hand, proteasome activation has been suggested as a potential antiaging (9) and retardation strategy against the progression of age-related diseases (16). Lately, proteasome activation was achieved in multicellular organisms through genetic manipulation of either 20S or 19S subunits with positive outcomes on aging, stress resistance, and aggregation-related neuropathies (8, 55, 58). Given the restraints of genetic manipulation in humans, identification of proteasome-activating compounds is needed to promote proteasome enhancement ideally through human normal diet.
In this study, we sought to investigate whether compound-mediated proteasome activation is feasible in a multicellular model system using C. elegans. We provide evidence that treatment with 18α-GA [a previously identified proteasome activator in cell culture; (27)] induces proteasome enhancement leading to an SKN-1- and proteasome activation-dependent life span extension. We also demonstrate that 18α-GA treatment affects AD progression in a proteasome activation-dependent manner in various AD nematode models and in cells of nervous origin. In total, our results demonstrate the potential use of 18α-GA as a prolongevity and antiaggregation compound.
Results
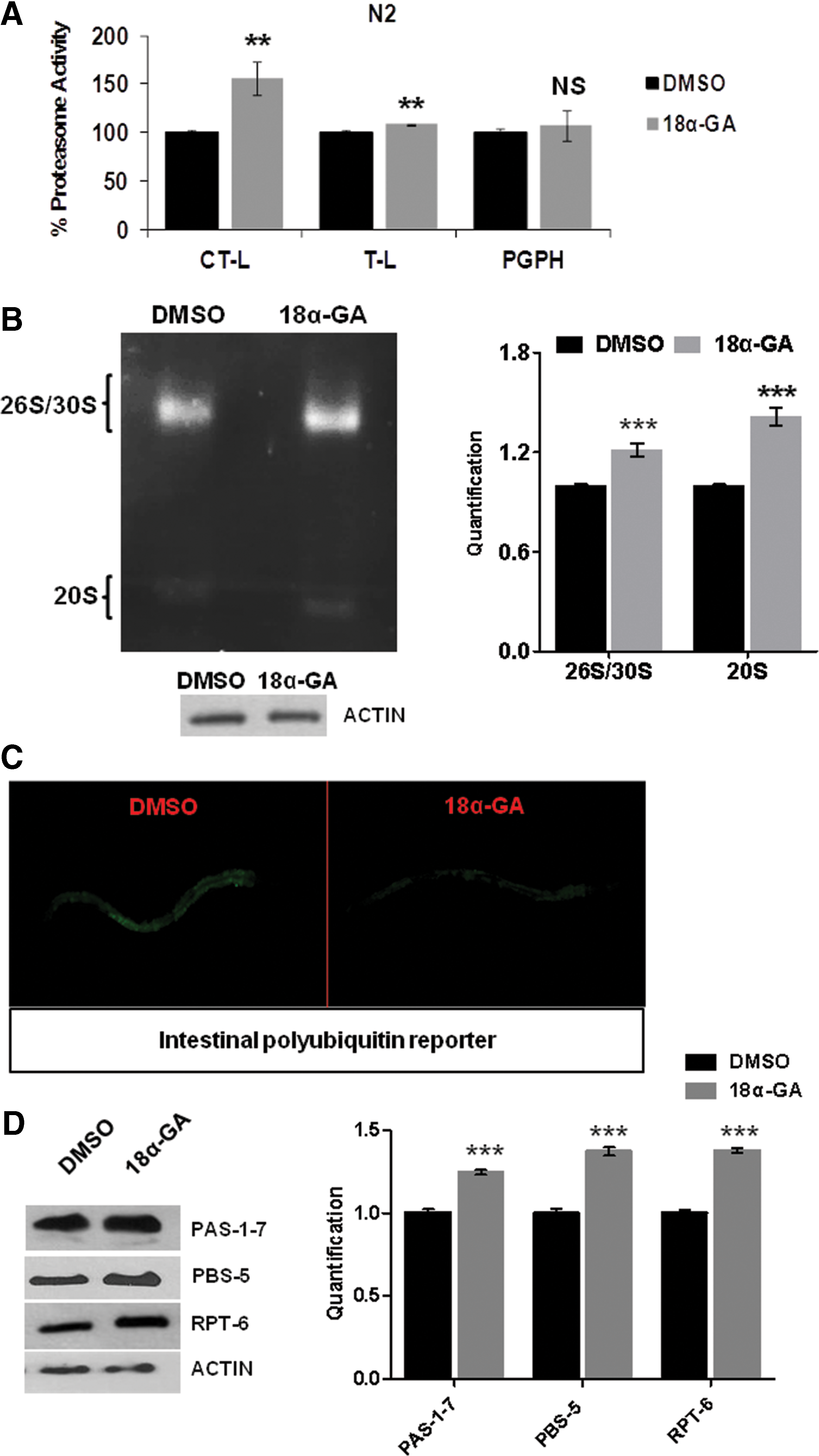
18α-GA confers proteasome activation in C. elegans
We have previously shown that 18α-GA treatment of human fibroblasts induces proteasome activation and results in extended cellular life span (27). We therefore aimed to examine whether this is an in vitro cell-specific effect or whether it may be exerted in a multicellular organism, namely C. elegans. Wild-type (wt) animals were treated with various concentrations of 18α-GA; CT-L and T-L proteasome activities were significantly enhanced (Fig. 1A). In-gel activity assay verified the enhancement of both 20S and 26S/30S proteasome activities (Fig. 1B). By taking advantage of a fluorescent in vivo polyubiquitin reporter system that responds to changes in proteasome activity (40), we also observed induced UPS activity in vivo (Fig. 1C). Protein expression analysis of PAS-1-7, PBS-5, and RPT-6 subunits revealed increased levels (Fig. 1D), correlating with the detected enhanced activity. Collectively, our results show that 18α-GA promotes proteasome activation in the context of a multicellular organism.
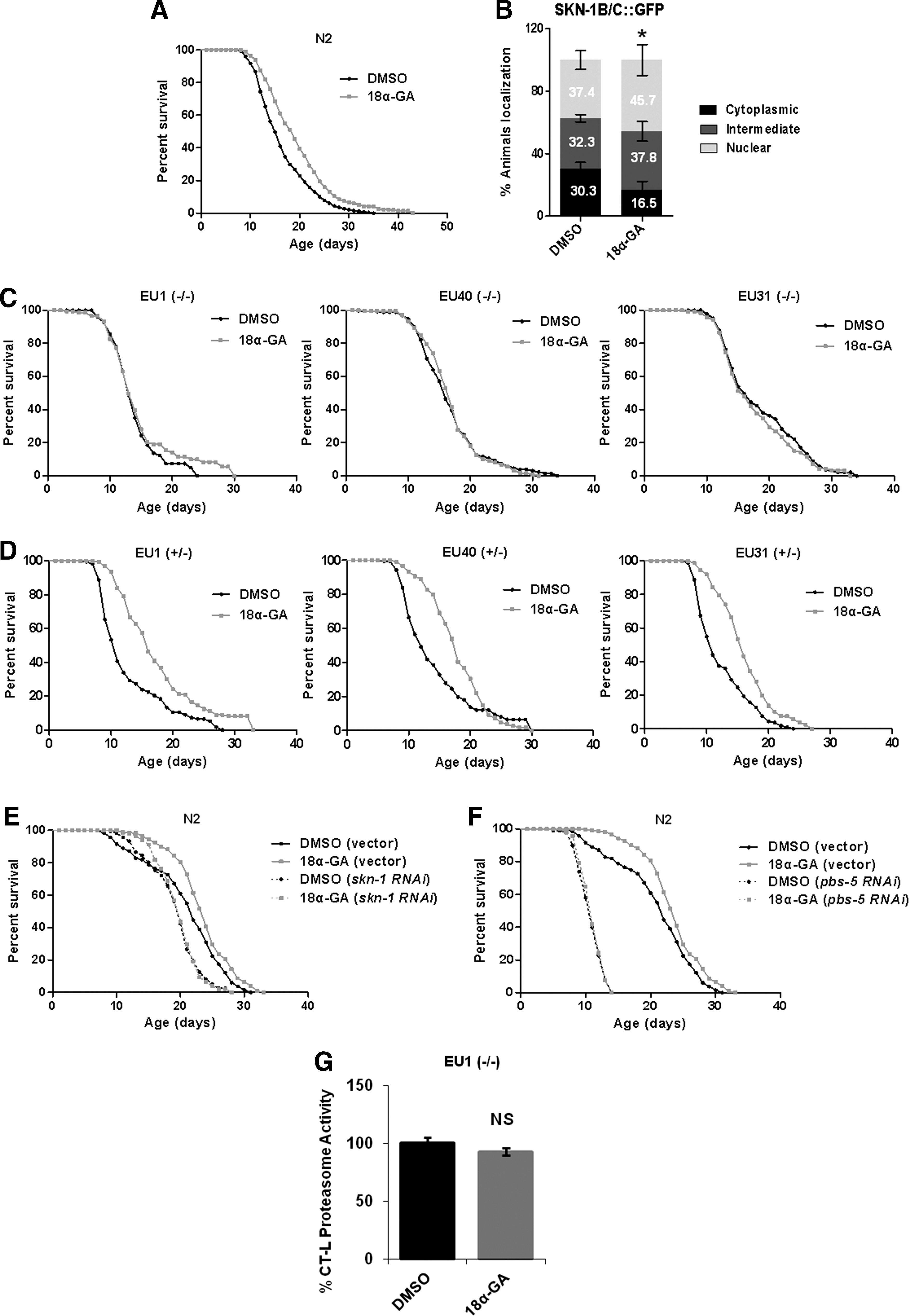
18α-GA promotes life span extension in a SKN-1- and proteasome activation-dependent manner
Since compound-mediated proteasome enhancement is a desired goal in the proteasome and antiaging field, we sought to investigate whether the observed 18α-GA-mediated proteasome activation promotes life span extension in C. elegans. Significant life span enhancement was revealed with the most effective concentration being 20 μg/ml (Fig. 2A and Supplementary Fig. S1A, B; Supplementary Data are available online at
All assays were performed at 20°C unless noted otherwise.
Number of offspring per worm.
Duration of postembryonic development (hours from egg hatching to L4 stage).
Pumps in 15 s at day 1 of adulthood.
Duration of defecation cycle in seconds.
Percentage of animals becoming dauer larva at 27°C.
p < 0.001.
18α-GA, 18α-glycyrrhetinic acid; DMSO, dimethyl sulfoxide.
The transcription factor, SKN-1, has been shown to be essential for life span regulation (3, 56) and has been correlated with the proteasome (26, 35, 45). Having shown that 18α-GA treatment activates Nrf2 in human primary cells (27), we tested whether it also affects SKN-1. Use of a reporter fusion of SKN-1 (skn-1B/C::GFP) revealed its nuclear translocation in the intestinal cells of 18α-GA-treated animals (Fig. 2B). Immunoblot analysis of the activated form of p38 MAPK revealed p38 MAPK kinase phosphorylation following 18α-GA treatment (Supplementary Fig. S2), leading to SKN-1 activation (25). We then employed three different skn-1 mutant strains [EU1, EU31, and EU40, bearing premature stop codon mutations; (56)] to reveal the potential SKN-1 dependence of the observed life span extension; 18α-GA failed to extend the life span of all three homozygous skn-1 mutants (Fig. 2C), thus verifying SKN-1 implication. Interestingly, lifetime 18α-GA treatment of heterozygous skn-1 mutants resulted in extended life span, suggesting that even only one normal skn-1 allele is sufficient for the life span-extending action of the compound (Fig. 2D). In support, knockdown of skn-1 (Fig. 2E; compare dimethylsulfoxide [DMSO] [skn-1 RNA interference [RNAi] with 18α-GA [skn-1 RNAi]) or pbs-5 (Fig. 2F; compare DMSO [pbs-5 RNAi] with 18α-GA [pbs-5 RNAi]) by RNAi abolished this extension. Accordingly, CT-L proteasome activity in EU1 mutant was not enhanced following 18α-GA treatment (Fig. 2G). Given that DAF-16/FOXO transcription factor is also a major aging regulator in C. elegans (30), we have also investigated whether this extension is DAF-16 dependent; 18α-GA did not extend the life span of daf-16 mutants, thus suggesting DAF-16 dependence (Supplementary Fig. 1C). In total, our results indicate that 18α-GA treatment promotes life span extension in a skn-1- and proteasome activation-dependent manner.
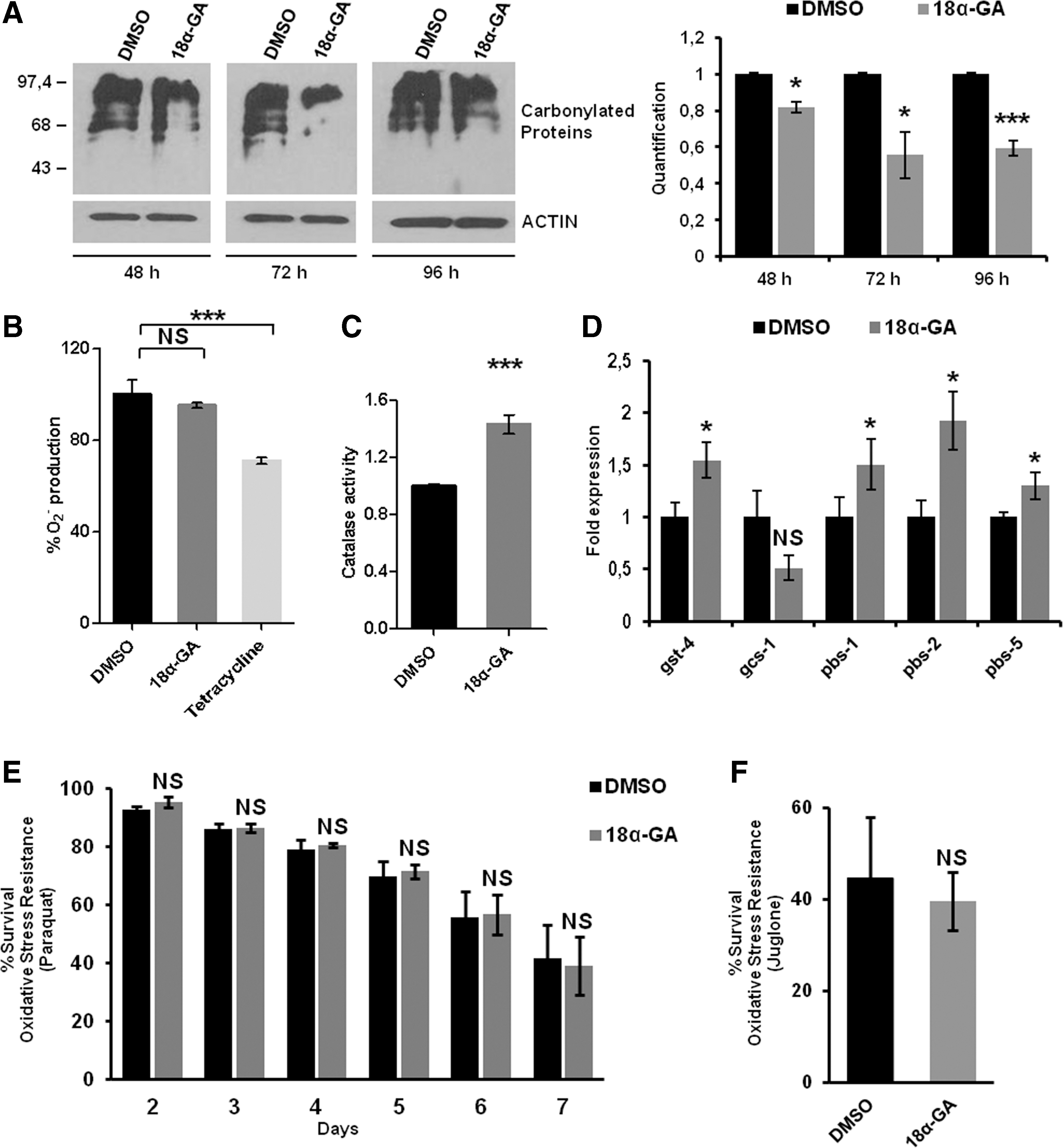
Oxidatively modified proteins are removed by the proteasome (49). Treatment with 18α-GA for 48, 72, and 96 h resulted in reduction of the carbonylated proteins by 18%, 44%, and 40%, respectively (Fig. 3A).These decreased levels may be due either to the increased proteasome activity levels or to the antioxidant activity of 18α-GA. When superoxide dismutase (SOD) activity was evaluated, we did not detect any alterations in the intracellular superoxide anion (O2 −) levels in contrast to the positive control (tetracycline; Fig. 3B). In accordance, monitoring of the reporter p sod-3GFP following 18α-GA treatment did not reveal significant alterations, thus verifying the lack of sod-3 induction (data not shown). In contrast, catalase activity was found to be increased in 18α-GA-treated animals (Fig. 3C). Furthermore, the RNA expression levels of various SKN-1 target genes (glutathione S-transferase [gst-4] and γ-glutamylcysteine synthetase [gcs-1]) were altered in opposite ways following 18α-GA treatment, whereas all proteasome subunits tested were induced (Fig. 3D). Finally, we did not detect oxidative stress resistance of wt animals in paraquat (a generator of O2 −; Fig. 3E) or juglone (that produces damaging adducts with proteins; Fig. 3F). Our data suggest that the positive outcomes of 18α-GA may not be solely related to its antioxidant activity per se and may advocate for the link between the observed lower amounts of oxidized proteins and the increased levels of proteasome activity.
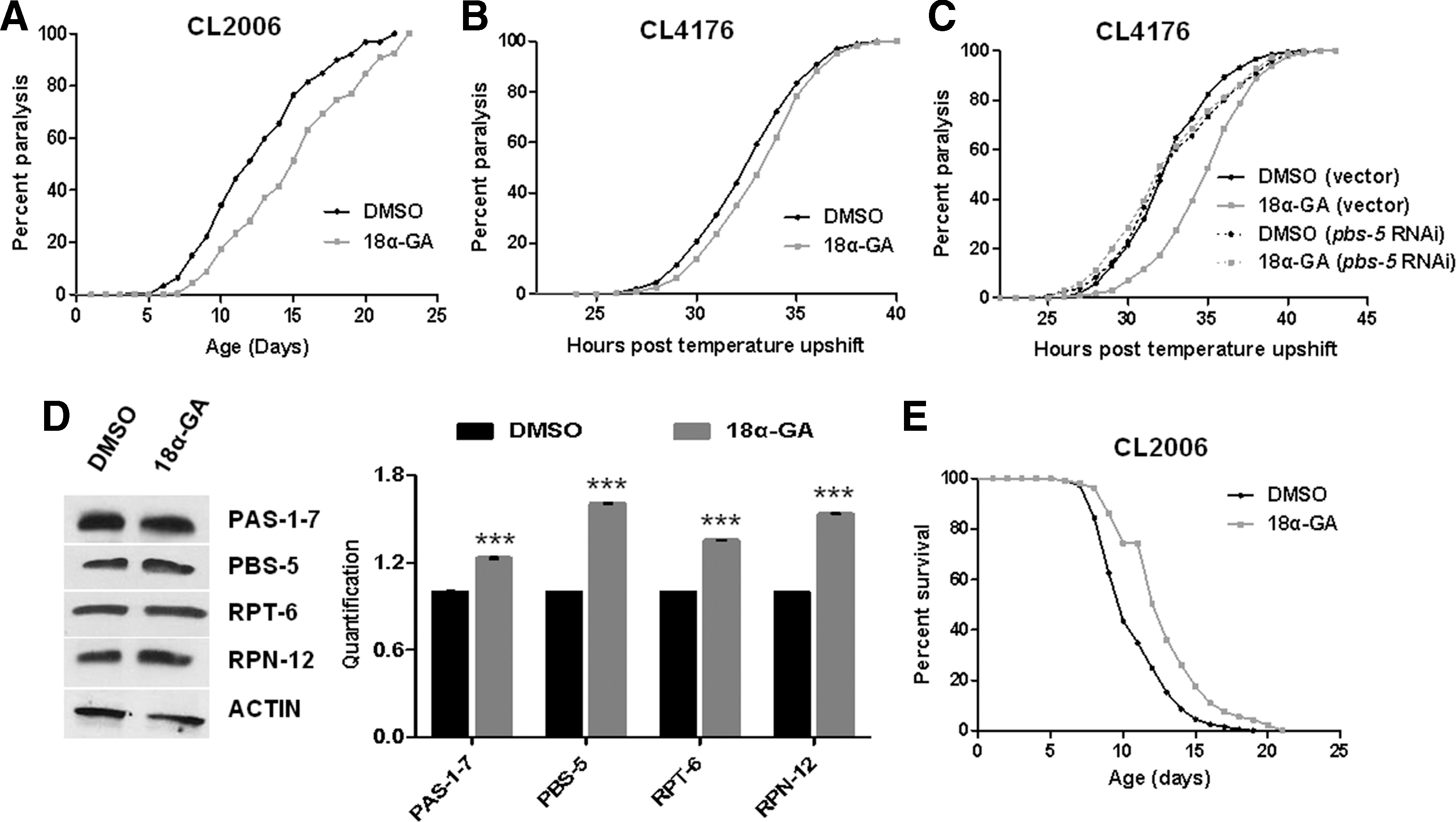
18α-GA-mediated proteasome activation delays aggregation-related pathologies
Aging represents a crucial risk factor for aggregation-related disorders (12, 43). Moreover, proteasome inhibition and diminished proteasome activities have been monitored in AD patients (29, 57). We therefore investigated the effects of 18α-GA-mediated proteasome activation on AD progression by taking advantage of three different nematode AD models. More specifically, paralysis assays in CL2006 strain [constitutive expression of human Aβ1–42 in body wall muscle cells with adult-onset paralysis accompanied by β-amyloid formation; (36)] revealed a significant delay of paralysis upon 18α-GA treatment (Fig. 4A). Similar results were obtained in CL4176 strain [temperature-sensitive expression of human Aβ1–42 resulting in β-amyloid formation; (20); Fig. 4B]. More importantly, combined treatment with pbs-5 RNAi and 18α-GA abolished the delayed paralysis (Fig. 4C), thus advocating for the dependence of the decelerated paralysis rates on proteasome activation. Protein expression analysis of various 19S and 20S proteasome subunits further verified the 18α-GA-mediated enhancement of proteasome levels (Fig. 4D) similarly to the results in wt animals (Fig. 1D). Finally, lifetime 18α-GA treatment resulted in significant life span extension of CL2006 animals (where age-dependent aggregation and paralysis occur; Fig. 4E). Collectively, our results suggest that 18α-GA promotes deceleration of the AD paralysis phenotype in a proteasome activation-dependent manner.
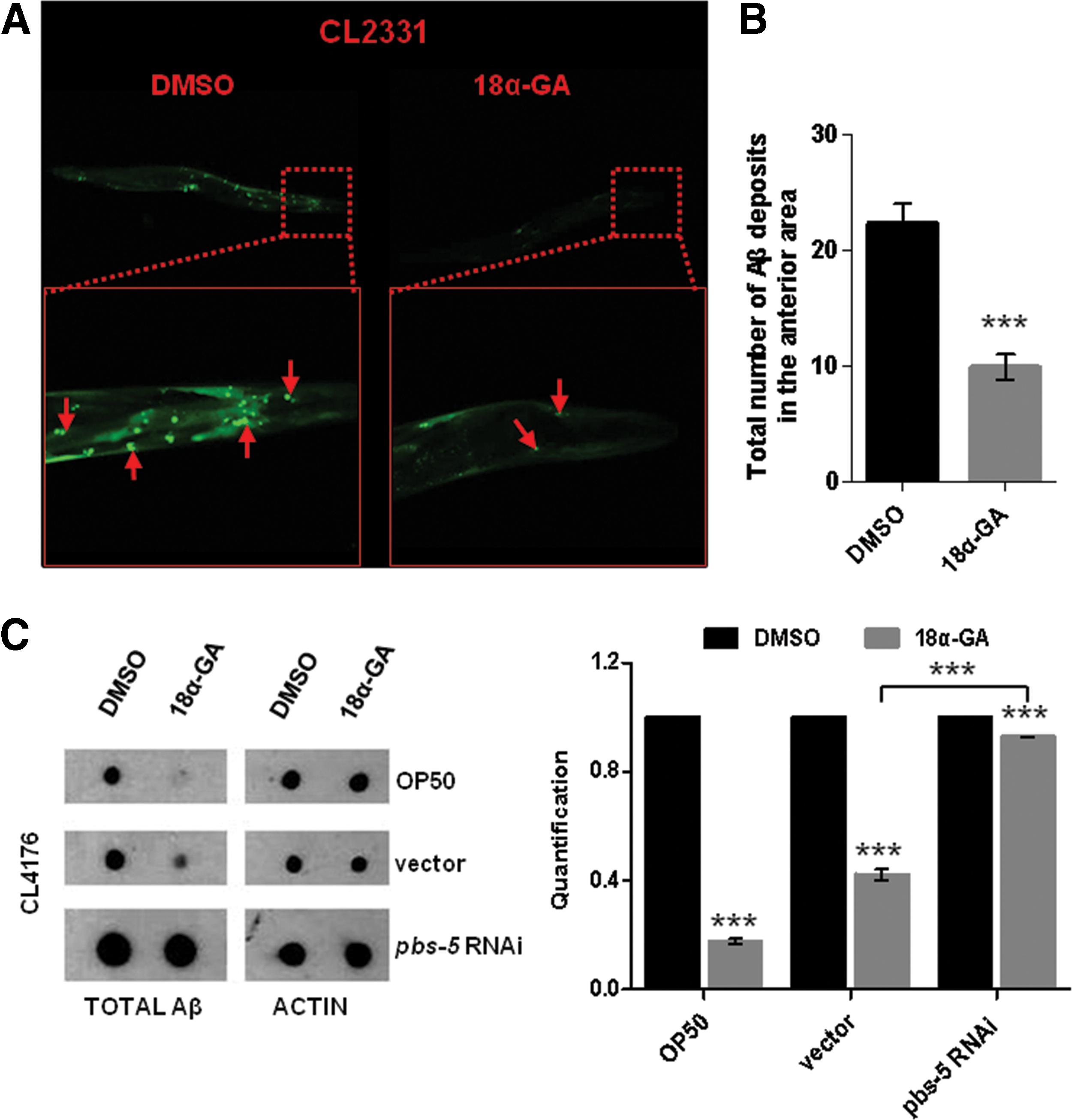
To evaluate the Aβ aggregation state in vivo, we took advantage of CL2331 strain (temperature-sensitive expression of human Aβ3–42 conjugated with GFP in the body wall muscle cells); 18α-GA treatment confers a significant reduction of Aβ deposits (Fig. 5A; red panels and arrows and Fig. 5B). We did not observe any differences in the GFP production upon 18α-GA or DMSO treatment in the CL2179 control strain (data not shown). We have also revealed significant reduction of total Aβ levels in CL4176 animals upon 18α-GA treatment (Fig. 5C upper and middle panels), while pbs-5 knockdown drastically reduced this positive outcome (Fig. 5C compare middle and lower panels). We suggest that these decreased levels are due to the 18α-GA-mediated enhanced proteasome function.
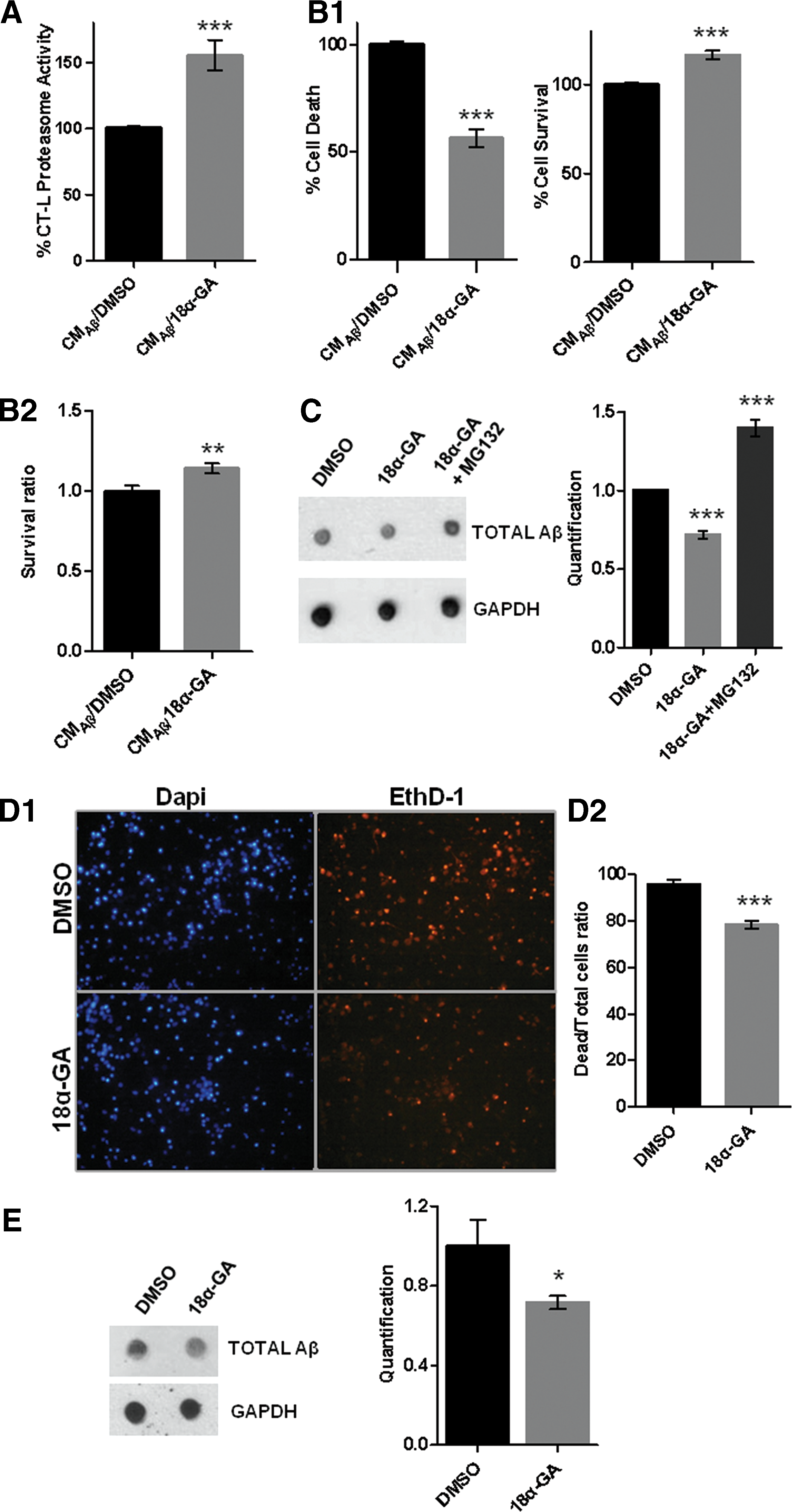
Finally, we sought to evaluate whether 18α-GA treatment may protect neuronal cells against Aβ toxicity. We took advantage of SH-SY5Y human neuroblastoma cells that were exposed to 7PA2-conditioned medium (CM, CMAβ; contains Aβ) in the presence of 18α-GA or DMSO. We initially verified that 18α-GA induces proteasome activation in those cells and conditions (Fig. 6A). Parallel treatment of SH-SY5Y exposed to CMAβ with 18α-GA led to a significantly reduced cell death and increased cell survival compared with the control cultures (Fig. 6B1, B2). Treatment with CMcontrol (no Aβ production) did not reveal any differences (data not shown). Dot blot analysis revealed reduced levels of total Aβ upon 18α-GA treatment, whereas proteasome inhibition by MG132 abolished this reduction (Fig. 6C). The protective role of 18α-GA against Aβ toxicity was also verified in primary murine cortical neurons (Fig. 6D1–E). In conclusion, 18α-GA-mediated proteasome activation appears to be protective against Aβ proteotoxicity in nematodes and neuronal cells.
Discussion
Continuously growing evidence indicates that proteasome activation is a key mediator of enhanced cellular and organismal healthspan and life span. In this study, we show that compound-mediated proteasome activation is possible in C. elegans promoting the extension of its life span in a SKN-1-dependent proteasome enhancement manner. We further show that this activation exerts beneficial effects on AD progression in relevant nematode models and in human and murine cells of nervous origin exposed to human Aβ peptide. This report reveals the potential antiaging and antiaggregation effects of a diet-derived compound.
Proteasomes are central players that need to be functional to preserve proteostasis (37). Given that proteasome function declines upon aging and senescence (12 –14), proteasome activation has immerged as a promising approach for delaying aging and age-related diseases (16). Genetic activation has been achieved in cells, uni- and multicellular organisms (16), but the new trend is the identification of proteasome-activating compounds. Various compounds have been identified so far, while several among them have been examined in the context of aging or age-related diseases (16). Few of them promote proteasome activation in multicellular organisms, including quercetin (21), catechin-enriched green tea extract (18), plant extracts from blackberries, hibiscus, elderberries, or jiaogulan (22) in nematodes and sulforaphane and D3T in mice (33, 34). In this study, we propose the triterpenoid, 18α-GA, as such compound. It is a pentacyclic triterpene glycoside considered as the most important bioactive compound derived from Glycyrrhiza radix (61). Although various properties have been attributed to 18α-GA, including not only anti-inflammatory (41) and anticancer/proliferative (54) but also proproliferative on primary cells (31), we are the first to show its effects on organismal aging as a consequence of SKN-1-mediated proteasome activation.
Our data indicate that 18α-GA treatment results in proteasome activation-dependent life span extension in wt nematodes. The observed dependence is in agreement with studies in human (10, 15, 27) and yeast cells (7, 32) and in multicellular organisms (8, 46, 55, 58). It is, however, noteworthy that with the exception of quercetin (10) and 18α-GA (27) in all other mentioned studies, proteasome activation was achieved through genetic manipulation. The crucial interdependence between enhanced proteasome function and prolongevity effects is also supported by reports in centenarians and naked mole rats, both known for their elongated life span and their increased proteasome function (11, 48, 53).
The lower detected levels of carbonylated proteins [widely used as markers of aging; (1)] advocate for the prolongevity effects of 18α-GA. Both increased proteolysis rates due to proteasome activation and antioxidation may contribute to this reduction. Our data regarding the antioxidant properties of 18α-GA reveal a complex regulation since genes of the same pathway such as gcs-1 and gst-4 (both known SKN-1 targets) or enzymatic activities of the same biochemical pathway such as SOD and catalase are altered in different ways. Those results coincide with recent studies showing increased complexity in the regulation of phase 2 detoxification systems and stress resistance in animals that involves additional SKN-1 independent regulators (17, 44). Therefore, the lack of stress resistance to oxidative stressors might be explained under this prism; maybe just SKN-1 induction is not enough for stress resistance under harsh oxidative stress.
SKN-1 possesses a central role in longevity and oxidative stress resistance (3, 56), thus implying that life span extension is anticipated upon its activation. Our data show that the 18α-GA-mediated proteasome activation is SKN-1 dependent in accordance with the Nrf2 dependence of the same compound in human cells (27). We show that SKN-1 activation occurs through the p38 MAPK pathway (25). Likewise, at least partial SKN-1 dependence has been suggested upon pbs-5 overexpression-mediated proteasome activation (8). Nevertheless, our current study specifies proteasome activation as the main executer for the observed extended life span; the 18α-GA prolongevity effects are lost if proteasome activation is impeded, despite SKN-1 activation.
We show that 18α-GA-mediated proteasome enhancement confers protection against proteotoxic stress not only in AD nematode models but also in neuronal cells treated with Aβ peptide, probably through proteasomal degradation of (at least some) Aβ species. In support, genetically mediated proteasome activation was shown to protect against polyglutamine- and Aβ-induced toxicity (8, 55, 58). Accordingly, several compounds have been shown to improve proteotoxic stress resistance in a proteasome-dependent manner. For example, quercetin supplementation delays the Aβ-induced paralysis in an AD nematode model dependently on enhanced proteasome degradation (52). Likewise, a proteasome-dependent antiamyloidogenic activity has been attributed to resveratrol (39, 51), ganoderic acid DM (6), thioflavin-T (2), and rolipram (42). Oleuropein aglycone was found to delay paralysis through Aβ deposit reduction in an AD nematode model (19) and to exert antiaggregation and neuroprotective activity in an AD mouse model (24) and in rats coinjected with Aβ42 (24). Proteasome function was not tested, but one cannot rule out the possibility of increased proteasomal Aβ aggregate clearance since oleuropein was previously shown to induce proteasome activation (28). Analogous positive outcomes have been unveiled for several compounds (mainly nutrient derived) in models for other aggregation-related diseases (16), thus pinpointing their potential against aggregation-related pathology.
Our results may adequately coincide with the concept of hormesis; depending on the dose, a substance can be cytoprotective or detrimental for cellular life span. Indeed, we have applied concentrations where absent (Supplementary Fig. S1A), significant (Fig. 2A), or still significant, but less pronounced (compared with the optimum concentration; Supplementary Fig. S1B), life span extension was observed. It would not thus be surprising to have a detrimental effect on life span in a higher 18α-GA concentration. Under the auspice of hormesis, the vitagene system has been suggested as a potential mediator to increase neuroprotection and longevity via a hormetic response (4, 5). Cytoprotective heat shock protein 70 (Hsp70), heme oxygenase-1 (HO-1), thioredoxin reductase (Trx), and sirtuins are included in the repertoire of vitagenes. The antioxidant response element induction of HO-1 and Trx-1 subsequently involves Nrf2 in the vitagene network. Although some phytochemicals may be beneficial solely due to their direct antioxidant properties, the observed neuroprotection may be also linked to the activation of various hormetic redox-sensitive intracellular cascades, such as the Nrf2 pathway. Our reported positive outcomes of 18α-GA-mediated SKN-1/Nrf2 induction can be interpreted under the prism of the vitagene network, while proteasome genes may constitute additional members of this system.
Proteasome activation has been shown to be feasible through manipulation of various UPS components. Given the link between proteasome and longevity, aging, and aggregation-related diseases, its (preferably controlled) activation seems to be an attractive preventive/therapeutic approach. Nevertheless, increased caution should be attributed due to the potential relationship of proteasome activation with cancer risk. Given the manipulation restraints in humans, compound-mediated activation might be the key solution. Diet constituents/active compounds may offer an extra advantage since they are supplied through normal diet from young ages. Consequently, they may block the initial stages of misfolded/damaged/oxidized protein accumulation (for aging) or oligomer/aggregate formation (for AD) before the senescent phenotype is established or before the disease is even triggered, thus being potentially preventive. Moreover, dose-dependent activation may be applicable. Future studies should focus on the identification of such compounds and the delineation of their exact mode of action.
Materials and Methods
Nematode strains and growth conditions
Standard procedures were followed for C. elegans strain growth and maintenance. All strains were maintained at 20°C, with the exception of CL4176, CL2331, and CL2179 that were maintained at 16°C. The following strains were used: N2 [wt Bristol isolate], EU1: skn-1(zu67) IV/nT1[unc-?(n754) let-?](IV;V), EU31: skn-1(zu135) IV/nT1[unc-?(n754) let-?](IV;V), EU40: skn-1(zu129) IV/nT1[unc-?(n754) let-?](IV;V), CL4176: dvIs27 [pAF29(myo-3/A-Beta1-42/letUTR) + pRF4(rol-6(su1006))], CL2006: dvIs2[pCL12(unc-54/human Abeta peptide 1–42 minigene) + pRF4], CL2331: dvIs37 [myo-3p::GFP::A-Beta (3 –42) + rol-6(su1006)], CL2179: dvIs179 [myo-3p::GFP::3′ UTR(long) + rol-6(su1006)], LD1: ldIs7 [skn-1B/C::GFP + pRF4(rol-6(su1006))], polyubiquitin reporter strain: N2 [Pvha-6::UIM2-ZsProSensor], and CF1553: muIs84[pAD76(sod-3::GFP)].
18α-GA treatment
18α-GA (Sigma-Aldrich, ≥95% purity) was dissolved as stock solution of 10 mg/ml in DMSO and stored at −20°C. Nematodes were exposed to 20 μg/ml 18α-GA or DMSO (control cultures), unless otherwise indicated. Plates containing 18α-GA were refreshed every 2 days in life span assays.
Antibodies
The following antibodies were used in this study: RPN-12 (Enzo Life Sciences; BML-PW8815-0100), RPT-6 (BML-PW8320-0100), PAS-1-7 (BML-PW8195-0100), 6E10 (BioLegend; SIG-39320), actin (MP Biomedicals; 0869100 clone C4), and GAPDH (Santa Cruz, sc-25778). PBS-5 is a home-customized antibody kindly provided by Prof. Osiewacz.
Life span assay
Synchronized L4 or young adult animals (80–125 animals per condition) were transferred to fresh nematode growth medium (NGM) plates containing the relative concentration of 18α-GA or DMSO and day 1 of adulthood was set as t = 0. The animals were transferred to fresh plates every 2–3 days and examined every day for touch-provoked movement and pharyngeal pumping until death. Each survival assay was repeated at least twice. Survival curves were created using the product-limit method by Kaplan and Meier. The percentage of nematodes remaining alive is plotted against animal age. The log-rank (Mantel–Cox) test was used to evaluate differences between survival curves and to determine p values for all independent data. N in life span figures is the number of animals that died over the total number of animals used (the number of animals that died plus the number of censored animals due to internally hatched eggs, extruded gonad, or desiccation due to crawling off the plates). Median life span values are expressed as mean ± standard error of the mean (SEM).
Paralysis assay
Synchronized L4 larvae CL2006 animals (100–120 animals per condition) were transferred to NGM plates containing either 18α-GA or DMSO at 20°C. Synchronized CL4176 animals (150–300 animals per condition) were transferred to NGM plates containing either 18α-GA or DMSO at 16°C for 48 h before temperature upshift to 25°C for transgene induction. Scoring of paralyzed animals was initiated at day 1 of adulthood for CL2006 strain and 24 h after temperature upshift for CL4176 strain. Each paralysis assay was repeated at least thrice. Nematodes were scored as paralyzed if they exhibited halos of cleared bacteria around their heads or failed to undergo half end body wave propagation upon prodding. Animals that died were excluded. The log-rank (Mantel–Cox) test was used to evaluate differences between paralysis curves and to determine p values for all independent data. N in paralysis figures is the number of animals that were paralyzed over the total number of animals used (the number of paralyzed animals plus the number of dead and censored animals). Median paralysis values are expressed as mean ± SEM.
RNA isolation and real-time polymerase chain reaction analysis
RNA was extracted from synchronized animals after 72 h of exposure to 18α-GA or DMSO and analyzed by standard methods. Primers are summarized in Table 2. Data were analyzed using the comparative 2−ΔΔCt method and are presented as the x-fold difference in mRNA transcript abundance in 18α-GA-treated animals, normalized to the endogenous pmp-3 gene, relative to DMSO-treated animals.
gcs-1, γ-glutamylcysteine synthetase; gst-4, glutathione S-transferase.
RNA interference
The pbs-5 and skn-1 RNAi constructs are from the Ahringer library (23) (Source Bioscience Life Sciences). Synchronized young adult animals (100–120 animals per condition) were transferred to NGM plates containing 2 mM isopropyl-b-
Phenotypic analysis
For all assays, N2 animals were allowed to lay eggs for 15–20 min on NGM plates containing either 18α-GA or DMSO.
Developmental timing
The progeny were frequently observed to record the needed time to reach the L4 larval stage from egg hatching. The experiment was repeated four times.
Pharyngeal pumping
At day 1 of adulthood, the pharyngeal pumping rate was measured. At least 10 animals per condition were examined.
Defecation assay
At day 1 of adulthood, the defecation rate (period in seconds from defecation to defecation) was measured. At least 20 animals per condition were examined.
Dauer formation
The progeny were kept at 27°C and the number of animals at the dauer larval stage over the total number of animals was scored 72 h later. The experiment was repeated six times.
Fecundity assay
Single N2 L4 larvae were transferred on NGM plates containing either 18α-GA or DMSO. Each animal was transferred every day to a fresh NGM plate containing 18α-GA or DMSO. Progeny of each animal were scored at L2–L3 larval stage. At least 10 animals per condition were examined.
Oxidative stress resistance assays
Synchronized L4 larvae N2 animals were exposed to 18α-GA or DMSO until day 5 of adulthood. They were then transferred to NGM plates seeded with UV-killed OP50 Escherichia coli containing 2 mM paraquat (Sigma-Aldrich) and survival was assessed from day 2 to 7 of paraquat exposure. Synchronized N2 animals were exposed to 18α-GA or DMSO until day 1 of adulthood and were then exposed to freshly made 80 mM juglone (Sigma-Aldrich) for 45 min at 20°C and survival was assessed following a 24-h recovery.
Superoxide anion production assay
N2 animals were allowed to lay eggs for 2 h on NGM plates containing 18α-GA, DMSO, or 50 μM tetracycline (used as positive control). After 96 h, the animals were collected into 1% Tween 20 in M9 and subjected to sonication; 0.6 μM phorbol myristate acetate and 0.6 mg/ml nitro tetrazolium blue (Sigma-Aldrich) were added, followed by incubation at 37°C for 30 min. Sample absorbance was measured at 560 nm using the Safire2 Multidetection Microplate Reader (Tecan). The protein content was determined using a Bio-Rad protein assay (Bio-Rad Laboratories). Results are calculated as absorbance units/mg of protein and expressed as % of control, that is, the percentage of O2 − produced with respect to DMSO-treated animals.
Catalase activity assay
N2 animals were allowed to lay eggs for 15–20 min on NGM plates containing 18α-GA or DMSO. After 96 h, the animals were collected into M/15 phosphate buffer, pH 7.0, and subjected to sonication. The protein content was determined using a Bio-Rad protein assay; 1 ml of substrate solution (12.5 mM H2O2) was added in 40 μg total protein. The cuvette was immediately scanned in a spectrophotometer at λ = 240 nm, and the time interval between the optical density from 0.450 to 0.400 was measured. H2O2-free phosphate buffer was used as control. Catalase activity was calculated based on the rate of H2O2 decomposition, which is proportional to the absorbance reduction at λ = 240 nm, using the equation k = 2.3/Δt × log (0.450/0.400), where k = catalase units and 2.3 = factor to convert from ln to log. Results are calculated as absorbance units/mg of protein and expressed as fold catalase activity of control with respect to DMSO-treated animals.
Proteasome peptidase assay
Animals exposed to 18α-GA or DMSO for 96 h were sonicated in proteasome activity lysis buffer (1 M Tris-HCl, pH 7.6, 100 mM ATP, 3 M KCl, 0.1 M ethylenediaminetetraacetic acid, 1 M dithiothreitol, 0.2% Nonidet P-40, 10% glycerol, 10 mg/ml aprotinin, and 10 mM phenylmethylsulfonyl fluoride). CT-L, T-L, and PGPH proteasome activities were assayed with the hydrolysis of Suc-LLVY-AMC, Boc-LRR-AMC, and Z-LLE-AMC fluorogenic peptides (UBPBio), respectively, for 30 min at 37°C. Specific proteasome activity was determined as the difference between the total activity of protein extracts and the remaining activity in the presence of 20 μM lactacystin (Enzo Life Sciences). Fluorescence was measured using a VersaFluor™ fluorescence spectrophotometer (Bio-Rad Laboratories).
Proteasome activity in intestinal polyubiquitin reporter animals
Synchronized transgenic L4 larvae expressing polyubiquitin reporter in the intestine in wt background were exposed to 18α-GA or DMSO until day 1 of adulthood. At indicated time points, they were mounted on a 2% agarose pad on glass slides and immobilized using 0.1 mM levamisole. Imaging was performed using a Zeiss AxioImager.Z1 upright epifluorescence microscope (Carl Zeiss Microscopy GmbH). Images of whole worms were acquired with 10 × 0.45 numerical aperture.
Native gel electrophoresis and in-gel proteasome assay
N2 animals were allowed to lay eggs for 2 h on NGM plates containing either 18α-GA or DMSO. The progeny were exposed to 18α-GA or DMSO until the young adult stage and were sonicated in proteasome activity lysis buffer (see above). Forty micrograms of protein was used for native gel electrophoresis of proteasome complexes. CT-L activity was assayed following gel incubation in 50 mM Tris, pH 7.4, 5 mM MgCl2, 1 mM ATP, and 300 μM proteasome substrate (Suc-LLVY-AMC) for 30 min at 37°C. Proteasome bands were visualized under UV. Part of the lysates was subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and actin was used as a loading control. Each blot was repeated at least twice.
Immunoblot and dot blot analysis and protein carbonyl group detection
N2 animals were allowed to lay eggs for 2 h on NGM plates containing either 18α-GA or DMSO. The progeny were exposed to 18α-GA or DMSO until the young adult stage (for CL4176 strain and N2) and day 6 of adulthood (for CL2006 strain) and were boiled in nonreducing Laemmli buffer. SDS-PAGE and immunoblotting were performed as described previously (11). For dot blot analysis, 5–10 μg of protein lysates were spotted onto 0.2 μm nitrocellulose membranes after soaking into 80°C heated tris-buffered saline. Immunoblotting was performed using the 6E10 antibody that recognizes total Aβ species. Detection of carbonyl groups into proteins (oxidized proteins) was performed with the Oxyblot protein oxidation detection kit (Millipore Corporation). Each blot was repeated at least twice. Blots were developed with chemiluminescence by using the Clarity™ Western ECL substrate (Bio-Rad Laboratories). Actin was used as a loading control.
Confocal analysis
Animals were mounted on 2% agarose pads on glass slides, anesthetized with 10 mM levamisole, and observed at room temperature (RT) using a Leica TCS SPE confocal laser scanning microscope (Leica Lasertechnik GmbH). The LAS AF software was used for image acquisition. At least 20 animals/condition in three independent experiments were processed, unless otherwise indicated. For skn-1B/C::GFP localization, synchronized LD1 animals expressing the p skn1 SKN-1b/c::GFP reporter fusion exposed to 18α-GA or DMSO until day 2 of adulthood were observed. Nematodes were classified into three groups (nuclear, cytoplasmic, intermediate), according to the extent of compartmental GFP distribution in the intestinal cells. For Aβ3–42 deposit measurements, synchronized CL2331 and CL2179 (control strain) animals exposed to 18α-GA or DMSO and grown at 20°C (to induce aggregation) until day 2 of adulthood were collected. Images of whole worms and focused images in the anterior area of nematodes were acquired with 10 × 0.45 and 20 × 0.70 numerical aperture, respectively. For GFP levels, synchronized CF1553 animals exposed to 18α-GA or DMSO until day 1 of adulthood were observed. GFP levels were examined by using a Zeiss AxioImager.Z1 upright epifluorescence microscope. Images of whole worms were acquired with 10 × 0.45 numerical aperture.
Cell culture
The human dopaminergic neuroblastoma cell line, SH-SY5Y, was maintained in RPMI supplemented with 10% heat-inactivated fetal bovine serum and 2 mM glutamine. Murine neurons were isolated from E16.5 C57BL6 embryos, plated onto poly-
CM preparation
CHO and 7PA2 cells were grown to ∼90% confluency, washed with phosphate-buffered saline (PBS), and incubated in serum-free DMEM for ∼16 h. The CM was collected and centrifuged to clear cell debris.
Cell death/viability assay
Cell death/viability of SH-SY5Y cultures was assessed through scoring of (a) detached cells in the supernatant from the CM-treated SH-SY5Y cells (cell death analysis) and (b) attached CM-treated SH-SY5Y cells (cell viability) after 2 h of incubation in triplicates using a Coulter Z2 counter. Cell death of cortical neurons was assessed through labeling of necrotic cells. Neurons treated as described above for 1 h were stained with 0.2 μM ethidium homodimer (nuclei labeling of dead or dying cells, Thermofisher Scientific) and 1 μg/ml 4′,6-diamidino-2-phenylindole (nuclei labeling of dead and alive cells; Vector Laboratories) for 20 min at 37°C. Cultures were then fixed in 4% paraformaldehyde for 20 min and washed with PBS. Dye uptake was analyzed using a Nikon ECLIPSE TE200 inverted fluorescent microscope (Nikon). Digital images were captured with an Olympus SP-510U2 camera (Olympus). Images were processed using the ImageJ 1.48v.
Survival ratio through crystal violet staining
SH-SY5Y cells treated as described above were fixed in 4% paraformaldehyde for 20 min and then stained with 0.2% crystal violet in distilled water. Cells were washed with water, air-dried, and the dye was eluted with 30% acetic acid. Viability was assessed by measuring dye absorbance at 595 nm using the Safire2 Multidetection Microplate Reader.
Statistical analysis
Statistical analysis and graphs were produced using Prism (GraphPad Software, Inc.) and Microsoft Office 2003 Excel (Microsoft Corporation) software packages. Data in all assays (including blot quantification) are depicted as the average of three independent experiments (unless otherwise indicated). Error bars denote ± SEM. Student's t-test was used for comparisons. Asterisks denote p-values as follows: *p < 0.05, **p < 0.01, ***p < 0.001, not significant. Quantification of the ratio of each protein to actin/GAPDH (using the ImageJ software) and normalization to control appear next to each representative native gel/blot.
Footnotes
Acknowledgments
The authors are grateful to Prof. Osiewacz and Dr. Kletsas for antibodies; to Dr. Bano for reagents; to Dr. Vekrellis and his group for the isolation of cortical neurons and reagents; and to Dr. Link for CL2331 and CL2179 strains. Nematode strains used in this study were provided by the Caenorhabditis Genetics Center, supported by the U.S. National Institutes of Health National Center for Research Resources. This work was funded by two Research Funding Programs: Thales GenAge [QALHS AP:10479/3.7.12 MIS380228] (to E.S.G. and N.C.) and MAESTRO (to N.C.) cofinanced by the European Union (European Social Fund) and Greek national funds through the Operational Program, Education and Lifelong Learning, of the National Strategic Reference Framework (NSRF), a European Union (266486 NU-AGE) grant (to E.S.G.), an IKYDA 2012 fellowship, an Empirikion Foundation Scientific Project and a Scientific Project funded by John S. Latsis Public Benefit Foundation (to N.C.), and by the Academy of Finland (259797 to C.I.H.). The present publication is supported by the COST Actions PROTEOSTASIS BM1307 and GENiE BM1408, supported by COST (European Cooperation in Science and Technology).
Author Disclosure Statement
No competing financial interests exist.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
