Abstract
Introduction
R
The World Health Organization has defined a biomarker as any substance, structure, or process that can be measured in the body or its products and influence or predict the incidence of outcome or disease (192). Markers of oxidative stress often fulfill the first part of the criteria (i.e., they can be measured) and many studies suggest oxidative stress can influence the disease, but to be a clinically relevant biomarker, some additional issues must also be addressed. In summary, a clinically useful biomarker must be able to meet one of the following criteria: (i) show specificity for a certain disease (diagnostic), (ii) have prognostic value, and (iii) correlate with disease activity. This then allows treatment efficacy to be assessed. To be clinically useful, a biomarker must also be reasonably stable, present in an easily accessible tissue, and cost-effective to measure reproducibly on a large scale.
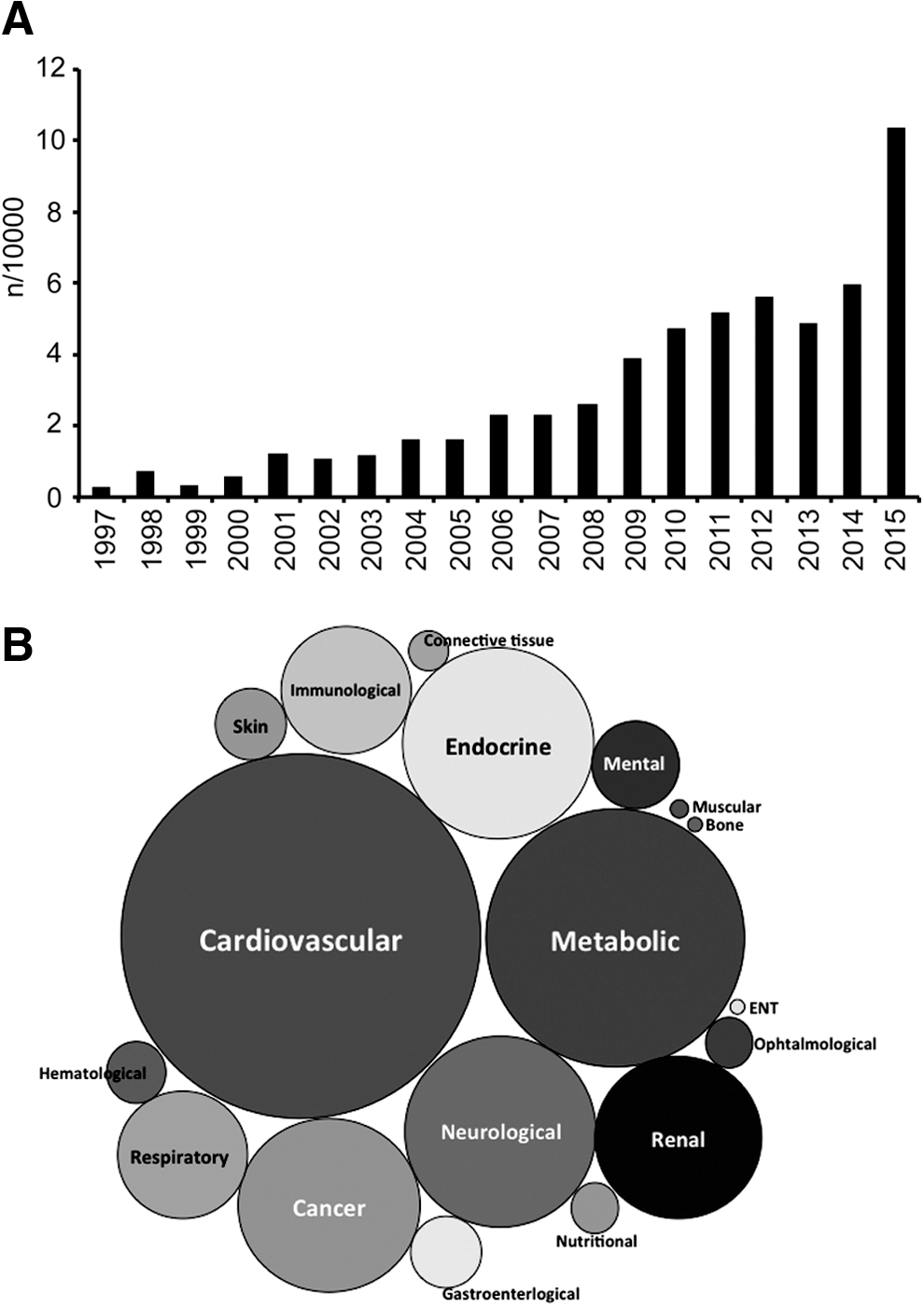
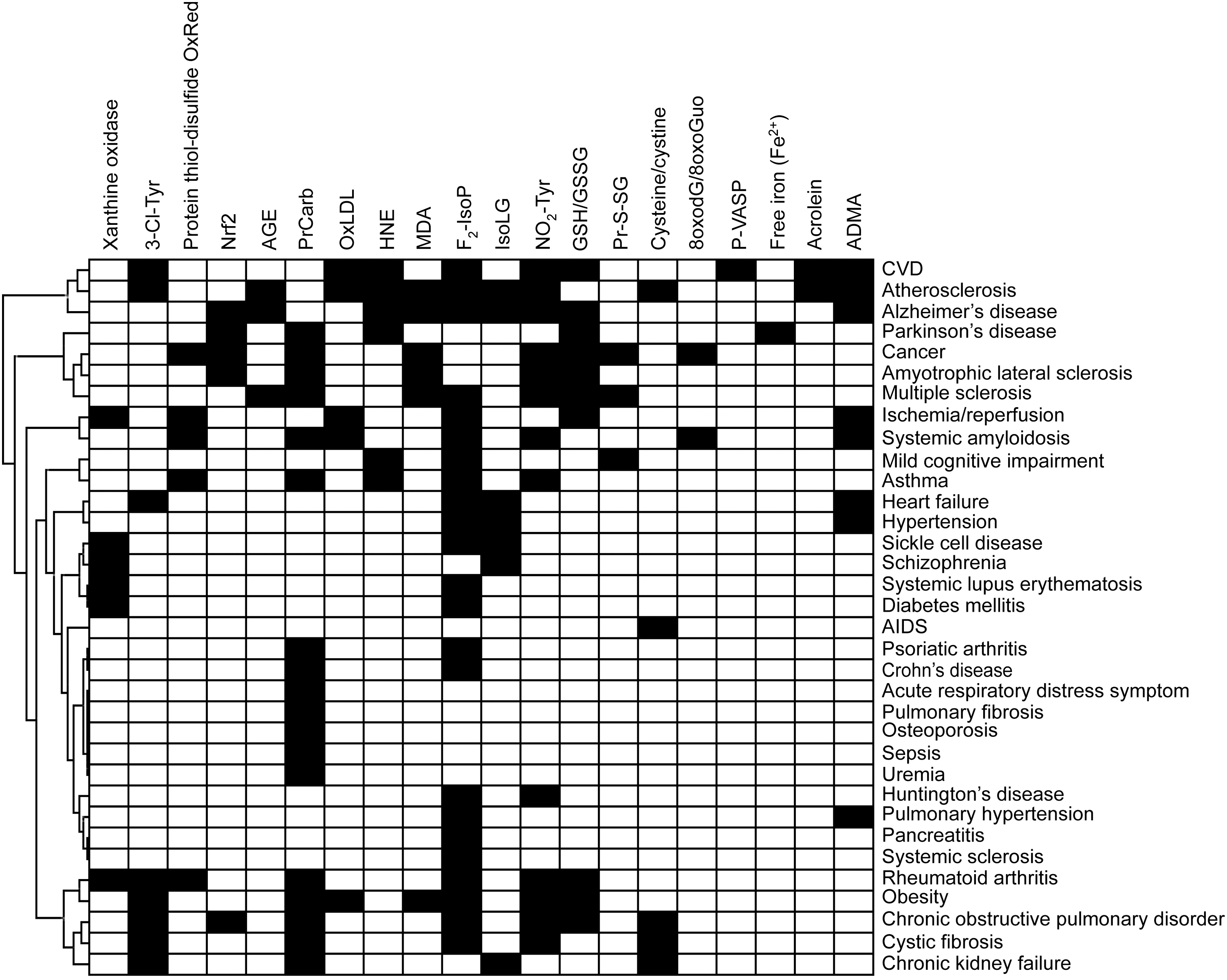
An increasing number of studies are published on markers of oxidative stress in a whole range of human diseases (Fig. 1). While a plethora of markers and methods are used, many of these do not correlate well with each other, do not reflect a state of oxidative stress, or are not specific. In this study, we critically review the current state of oxidative stress biomarkers that are used to assess the redox state of the body or specific tissues and cells in health and disease, with a focus on those that can be realistically applied to the clinic (Fig. 2). This approach excludes by definition several commonly used preclinical and in vitro methods. Visualization of biomarkers measured in various diseases by cluster analysis (Fig. 3) shows that the majority of studies have used ROS-induced modifications as markers of oxidative stress, which will be discussed first. We then focus on biomarkers assessing two important components whose deregulation can result in oxidative stress, ROS generation, and antioxidant defense. We conclude with two functional markers that are downstream of oxidative stress. From a clinical perspective, what matters is which marker is predictive with respect to risk and therapeutic outcome.
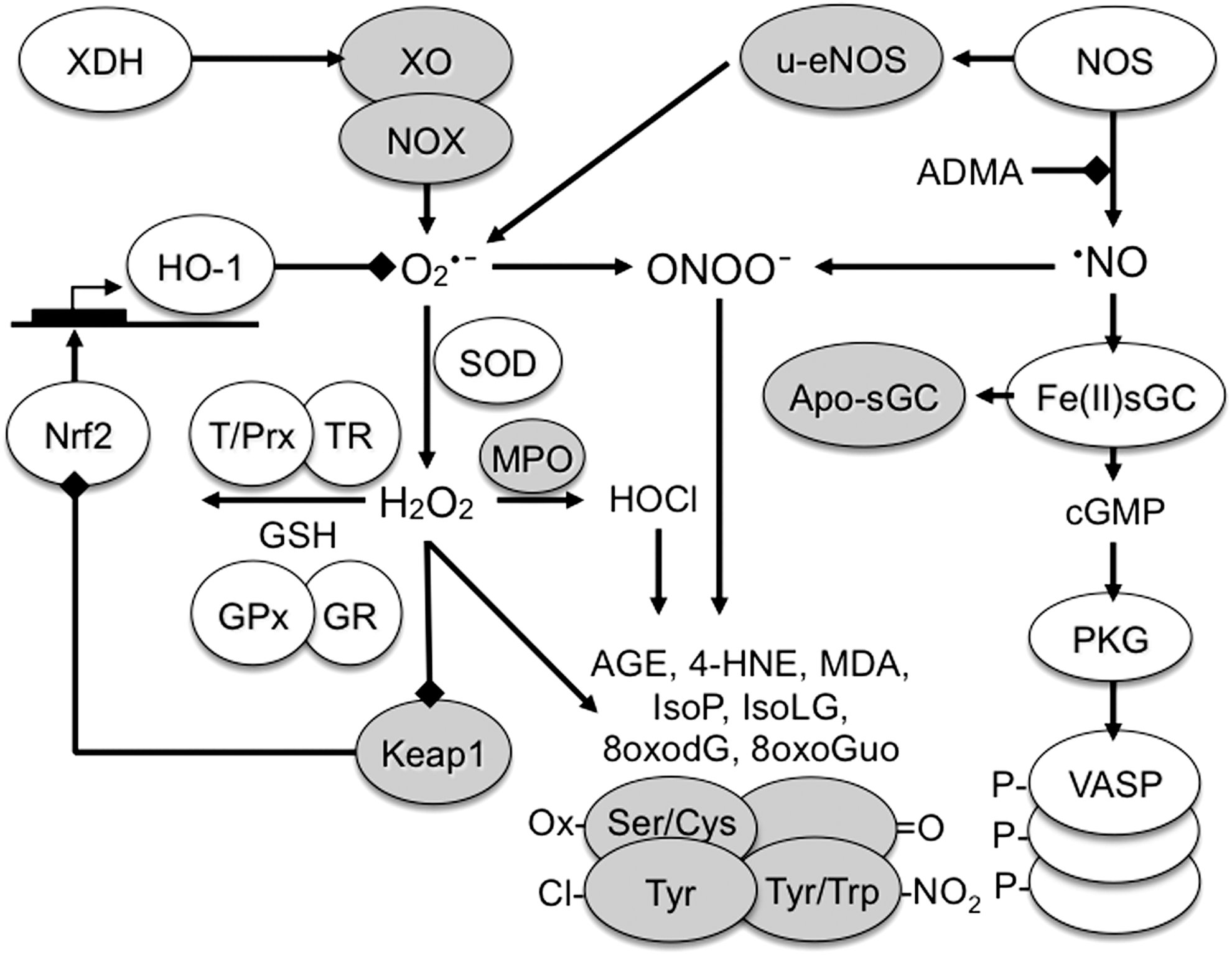
ROS-Induced Modifications
This category includes biomarkers measuring evidence of direct chemical impact of ROS in biological systems. One of the ROS subsets is also described as RNS, for example,
Protein carbonyls and advanced glycation end products
Protein carbonyls are formed through oxidative cleavage of protein backbones. Oxidative deamination of lysine and glutamic acid also results in protein carbonyls (34). Since carbonyls can arise from different mechanisms, their concentration is commonly higher than that of other biomarkers (40).
Carbonyl groups may also be introduced by binding of aldehydic lipid oxidation products to lysine, cysteine, and histidine residues—a reaction termed Michael addition—resulting in advanced lipoxidation end products. Reactions between lysine and arginine residues and carbohydrates—a reaction called glycoxidation—result in advanced glycation end products (AGEs).
AGEs are a group of heterogeneous molecules that arise from the nonenzymatic reaction of reducing sugars with amino groups of lipids, DNA, and especially long-lived proteins. This process occurs during normal metabolism, but is even more pronounced under hyperglycemic, hyperlipidemic, and oxidative stress conditions.
The glycation reaction can be accompanied by an oxidation leading to glycoxidation products. Carboxymethyl valine and pentosidine are among the most prominent AGEs resulting from glycoxidation. Glyoxal, generated from metal-catalyzed oxidation of polyunsaturated fatty acids (PUFAs), forms adducts with lysine (resulting in carboxymethyl lysine [CML]), an advanced lipoxidation product (55). About 90% of CML and pentosidine in blood are bound to proteins (116). Due to their relationship to sugars, AGEs have been linked to diabetes mellitus and other diseases, such as obesity (20), atherosclerosis, renal failure (193), and Alzheimer's disease (172). Due to the different possible formation mechanisms and heterogeneity, numerous glycation products exist, of which only some have been characterized so far.
Protein carbonyls (i.e., having aldehyde and ketone moieties) are usually detected after derivatization with 2,4-dinitrophenylhydrazine (DNP). The resulting carbonyl-2,4-dinitrophenylhydrazine adduct (101) can be detected spectrophotometrically or by specific anti-DNP antibodies with ELISA (24), Western blot (91), immunohisto- and cytochemistry, or by high-performance liquid chromatography (HPLC). The results of the ELISA correlate well with the colorimetric assay (24), whereby the ELISA is more convenient to analyze a larger number of samples within one run and requires significantly less sample volume. Concerning clinical settings, the only methods that seem to be applicable are ELISA (kits are available) and HPLC as they enable high throughput, involve internal/external standards, and comparison of samples under constant conditions.
A number of methods have been reported to measure AGEs based on the use of antibodies for immunohistochemistry, immunoblot, and commercial ELISA, as well as special AGE readers that utilize the autofluorescence properties of AGEs in human skin to assess AGE concentrations. Spectrofluorometry can be applied to diluted plasma or serum samples and a fructosamine assay to detect ketoamines (9). HPLC allows the identification and measurement of specific AGEs such as pentosidine (169) and CML (52). Creatinine glycation products can be measured with stable isotope dilution analysis and liquid chromatography (LC)-MS/MS (97). Due to the structural heterogeneity of AGEs, there is no method that can be especially recommended for measuring specific AGEs in a clinical setting. Noninvasive spectrographic autofluorescence readers can be applied in a clinical setting; however, this should be standardized in terms of using the average of three readings, the same body region, avoiding surrounding light and skin areas with tattoos. Elevated skin autofluorescence has been demonstrated in diabetes, kidney disease, and in patients with arterial stiffness.
In humans, elevated protein carbonyl levels have been reported in numerous conditions, including aging (61), neurodegenerative diseases (62), obesity, diabetes mellitus, age-related macular degeneration (174), human immunodeficiency virus (HIV), anemia, sickle cell disease, newborn bronchopulmonary dysplasia, and hepatocellular carcinoma (Table 1).
References are provided as supplementary material (Supplementary Data are available online at
HIV, human immunodeficiency virus; PC, protein carbonyl.
Protein carbonyls increase with age in healthy women and men (61, 122). With age, AGEs accumulate in the skin and correlate with the glucose exposure dose in patients on peritoneal dialysis (25). In diabetes, ROS are generated through several pathways, and elevated AGE concentrations have been reported. Ischemia/reperfusion is clearly associated with oxidative stress. Following coronary surgery in the reperfused human heart, a 2–3-fold increase in protein carbonyls, as measured by ELISA, was observed in plasma isolated from the venous coronary sinus (130). Protein carbonyls remained increased in blood for up to 18 h and therefore meet one important criterion for being a marker of oxidative stress, which is their stability.
Most methods detect protein carbonyls after derivatization and therefore do not provide a direct measure of these oxidative modifications. While commercial ELISA kits for AGE measurement provide ease of use, many of these do not specify the antibody used, which is just described as polyclonal anti-AGE antibody. This may lead to differences between commercial kits. Nevertheless, protein carbonyls and AGEs have been among the most successful markers of oxidative stress and are associated with disease state and treatment in multiple diseases (Tables 1 and 2).
References are provided as supplementary material.
AGE, advanced glycation end product; BMI, body–mass index; CML, carboxymethyl lysine; LC, liquid chromatography; MS, mass spectrometry; PCOS, polycystic ovary syndrome; SAF, skin autofluorescence; sRAGE, secreted receptor for AGE; T1D, type 1 diabetes; T2D, type 2 diabetes.
Oxidized low-density lipoprotein
The measurement of oxidized low-density lipoprotein (oxLDL) as a biomarker of oxidative stress has its origin in the oxidative modification hypothesis of atherosclerosis (165). However, oxLDL does not chemically define a specific form of modified LDL, molecule, or family of molecules (166). As the potential of oxLDL as a biomarker for cardiovascular disease (CVD) has been the subject of previous reviews (175, 177), we will critically assess the clinical utility of oxLDL as a biomarker.
OxLDL is most commonly measured in plasma or isolated LDL by immunological methods using one of three different antibodies that appear most frequently in the literature: 4E6, DLH3, and E06. The monoclonal antibody, 4E6, binds to aldehyde-modified lysine residues on LDL (73) and is the basis of a commercial method. The monoclonal antibodies, DLH3 and E06, recognize oxidized phosphatidylcholine (82) and phosphorylcholine containing short oxidized or nonoxidized side chains, respectively. Plasma oxLDL has been consistently found elevated in patients with CVD, independent of the assay used. However, conflicting results have been reported in studies on the association of oxLDL with atherosclerosis severity and the usefulness of oxLDL for CVD prediction. Contrasting results, depending on the assay, have been reported for plasma oxLDL following pharmacological intervention with statins (175, 177). In addition to CVD, plasma oxLDLs are increased in patients with insulin resistance, diabetes, and obesity (175).
A general limitation of the single most commonly used 4E6-based assay is that native LDL is also detected. As a result, the concentrations of oxLDLs determined closely reflect the concentrations of LDL cholesterol, and the predictive value of the assays is dependent on the levels of apoB (194). This casts serious doubt over the usefulness of oxLDL as a measure of oxidative stress and its clinical utility to predict cardiovascular and associated diseases above that of LDL cholesterol. Another general problem is that results obtained with different antibodies/methods cannot be compared and often do not correlate with each other, which is inconsistent with oxLDL being a quantitative measure of oxidative stress or representing a meaningful tool to predict CVD. The DLH13-based method was developed for isolated LDL, which limits its clinical utility because LDL isolation is time-consuming and isolated LDL is prone to ex vivo oxidation when stored at 4°C or after coating on plates. An extension of this assay to plasma has been developed commercially, but its utility is questionable because plasma and isolated LDL data do not match (81). A major problem with E06-based methods to determine oxLDL is that contrary to the common notion (175, 177), the monoclonal antibody is not specific for oxidized (phospho)lipids (51) and most of the recognized antigens in plasma reside in lipoproteins other than LDLs (178).
Given the limitations summarized above, oxLDL is unlikely a specific measure of oxidative stress. This is consistent with the majority of human plasma F2-isoprostanes (135a) and cholesterylester hydroperoxides (16) being associated with high-density lipoproteins (HDLs) rather than LDLs.
Lipid oxidation products
PUFAs, in particular linoleic and arachidonic acid (AA), are important targets of lipid peroxidation. Reaction of ROS, in particular hydroxyl and peroxyl radicals, with bisallylic hydrogen of PUFAs initiates the autocatalytic chain reaction of lipid peroxidation (Reactions 1–3) during which lipid peroxyl radicals act as chain-carrying radicals and lipid hydroperoxides are formed as the primary end products.
The chain reaction of lipid peroxidation can be terminated by tocopherols, such as α-tocopherol (α-TOH), via reaction with the chain-propagating lipid peroxyl radical (LOO•) (Reaction 4a). The resulting α-tocopherol radical (α-TO•) can be reduced back to α-TOH by certain reducing agents (e.g., ascorbate) (not shown) or react with a second molecule of LOO• to form a nonradical product (NRP) (Reaction 4b).
In the presence of transition metals, lipid hydroperoxides (LOOHs) give rise to LOO• (Reaction 5) or lipid alkoxyl radicals (LO•) (Reaction 6) that are further cyclized and/or degraded into several different reactive aldehydes (Reaction 7) that participate in various biological processes and signal transduction pathways (200). Depending on the type of PUFAs undergoing lipid oxidation, these include trans-4-hydroxy-2-nonenal (4-HNE), malondialdehyde (MDA), and others. Aldehydes derived from lipids are very reactive and therefore readily react with proteins to form Michael adducts, often denoted as advanced lipoxidation end products (49).
Isoprostanes (IsoPs) are formed during free radical-catalyzed oxidation of AA (119). Radical-mediated oxidation of AA occurs independently of whether it is esterified (as in phospholipids, triacylglycerols, or cholesterylesters). The types of IsoPs that will be formed and in what ratio depend on oxygen tension and glutathione concentration (120).
4-HNE and MDA
Different methods are available for the detection of both MDA and 4-HNE. Antibodies against MDA and 4-HNE bound to different amino acids have been developed (179, 186) and used in qualitative and semiquantitative immunocyto- and immunohistochemistry, where their presence and relative abundance are detected in cells and tissues in parallel with evaluation of morphological changes (126). Since Spiteller (163) reviewed the involvement of lipid peroxidation in a variety of chronic diseases, lipid oxidation end products emerged as oxidative stress markers, with 4-HNE and MDA being among the most investigated (126, 200) (Table 3).
References are provided as supplementary material.
4-HNE, trans-4-hydroxy-2-nonenal; AT1R, angiotensin receptor 1; CHD, coronary heart disease; CSF, cerebrospinal fluid; GC, gas chromatography; F2-IsoPs, F2-isoprostanes; HCC, hepatocellular carcinoma; HPLC, high-performance liquid chromatography; IHC, immunohistochemistry; IS, ischemic stroke; MDA, malondialdehyde; PDGF, platelet-derived growth factor; PSE, post-stroke epilepsy; RBC, red blood cell.
Despite their widespread use, all methods that detect both MDA and 4-HNE have their pitfalls (162). In the thiobarbituric acid reactive substances (TBARS) assay, up to 98% of the measured MDA can be formed by the high-temperature conditions during the procedure itself (117). When combined with HPLC, MDA-TBA adducts in in vitro oxidized human plasma can be determined reproducibly and reliably, although this method requires individual sample processing and its validity as a marker of in vivo oxidative stress remains uncertain, making it less applicable for routine clinical use (19). The numerous commercial easy-to-use kits lack specificity and their significance for clinical research is questionable.
In general, direct MDA and 4-HNE measurement is insensitive as the vast majority of these reactive products are bound to proteins and other biomolecules and remain undetected unless released before the assay (49). To measure the presence of 4-HNE in biological samples, including protein-bound aldehydes, protein immunodetection is preferred, either applied as immunohistochemistry or as HNE-His ELISA (187). The specificity of the monoclonal antibodies against HNE-His adducts allows their use in human and animal tissues, tissue homogenates, and in plasma and serum samples (63, 126, 162).
F2-isoprostanes
Several thorough reviews of the biochemistry and utility of F2-isoprostanes (F2-IsoPs) as biomarkers have been recently published (39, 113, 114), so only the most seminal points will be summarized here. Oxidation of AA forms a family of 64 bicyclic endoperoxide regio- and stereoisomers collectively termed H2-isoprostanes (140). Nonenzymatic rearrangement of these H2-isoprostanes forms both stable F2-IsoPs and highly reactive γ-ketoaldehydes termed isolevuglandins (IsoLGs, also known as isoketals) (115). Because of their chemical stability and sensitivity to changes in oxidative stress, F2-IsoPs are often considered the most reliable markers for monitoring oxidative stress in vivo (89). Elevated concentrations of F2-IsoPs are found in CVD, correlate with extent of disease, and predict the outcome (39). Elevated F2-IsoPs are also found in a wide range of human clinical conditions (113).
Despite strong evidence for their utility as biomarkers (Table 3), one challenge to widespread adaptation of F2-IsoPs in clinical trials is that the most reliable methods for their quantitation, gas chromatography–mass spectrometry (GC-MS) and LC-MS/MS, are labor-intensive and require specialized and expensive instrumentation (7, 114). While commercial immunoassays have been developed as a cheaper and easier alternative to mass spectrometry (MS), the results obtained with these immunoassays often do not correlate well with those obtained with GC-MS (78, 136). Thus, the results from immunoassays, particularly for individual patients, must be used with extreme caution, only with appropriate sample cleanup, and validated by MS whenever possible.
Isolevuglandins
Similar to F2-IsoPs, IsoLGs are products derived from the oxidation of AA and are sensitive to changes in oxidative stress. While F2-IsoPs are stable products of lipid oxidation, IsoLGs (Fig. 4) react rapidly and irreversibly with primary amines (e.g., protein lysyl residues and phosphatidylethanolamine) in the cell to form pyrrole (lactam) and oxidized pyrrole (hydroxylactam) adducts (18, 115, 146). Thus, only IsoLG adducts, and not unreacted IsoLGs, are detected in cells and tissues.
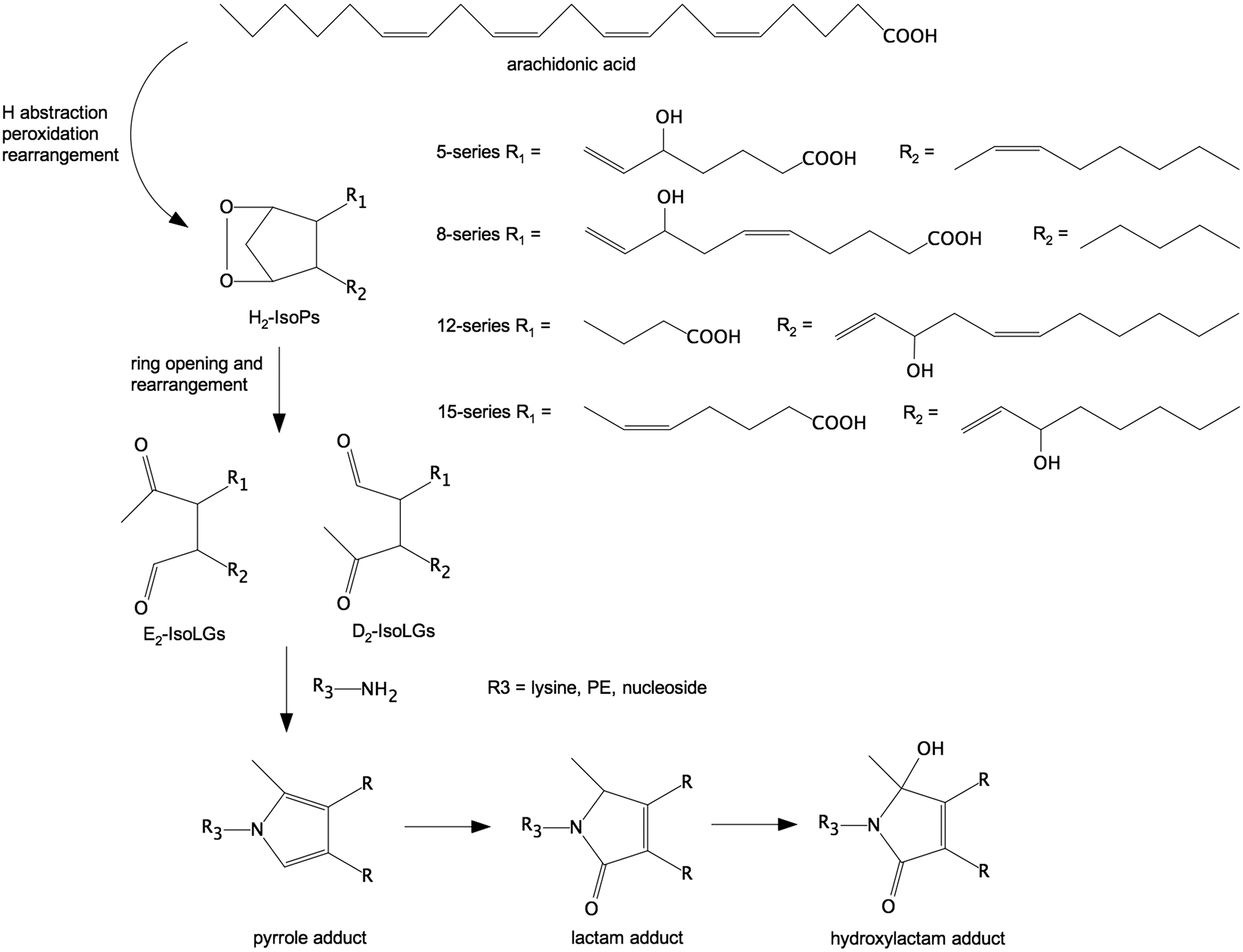
IsoLG adducts may eventually prove to have greater utility as disease biomarkers than more generalized measures of oxidative stress status because they appear to directly participate in pathological processes. The biological effects of exogenous IsoLGs on cultured cells include induction of inflammatory pathways, immune responses, and cell death, as well as inhibiting ion channel function (17, 36, 56, 65, 95). These results, along with the therapeutic effects of administering small-molecule IsoLG scavengers in animal models, suggest that IsoLGs could contribute to disease processes, including inflammation, hypertension, arrhythmia, atherosclerosis, and neurodegeneration (38, 41, 95, 146).
Quantitative immunoassays using polyclonal antibodies against IsoLG-protein adducts detected increased IsoLG-protein adduct formation in plasma from patients with atherosclerosis, in plasma from patients with end-stage renal disease, and in the glaucomatous trabecular meshwork (146). Immunohistochemical staining with the single-chain antibody D11ScFv that selectively recognized IsoLG-protein adducts showed increased adducts in the epicardial border zone of myocardial infarcts (56), in the hippocampus of Alzheimer's disease patients (38), and in heart, aorta, and dendritic cells during hypertension (64).
Mass spectrometric methods have demonstrated increased IsoLG-protein adducts compared with controls in the epicardial border zone of myocardial infarcts (56), in the hippocampus of Alzheimer's patients (38), and dendritic cells during hypertension (64). Using MS, IsoLG-phosphatidylethanolamine adducts have been found to be increased in plasma from patients with macular degeneration (102).
Currently, there are no published studies demonstrating that increased levels of IsoLG adducts predict onset or severity of subsequent disease. Therefore, the utility of measuring IsoLG adducts in urine or plasma as clinical biomarkers remains to be established. Nevertheless, current findings provide strong rationale for further investigation of the potential use of IsoLG adducts as clinical biomarkers, both to identify persons at risk and to determine the efficacy of treatments targeting IsoLGs such as dicarbonyl scavengers.
3-Nitrotyrosine
Nitrotyrosine (Tyr-NO2) is often described as a stable marker of oxidative/nitrative stress in inflammatory diseases (71). Tyrosine nitration involves the replacement of C3 hydrogen atom of the tyrosine aromatic ring with a nitro group (R-NO2) (11) (Fig. 5). This modification can occur within a polypeptide sequence (protein-associated Tyr-NO2) or to free tyrosine amino acids (free Tyr-NO2). Nitration can occur by several pathways in vivo, but always involves RNS and is usually a two-step process (161), in which (i) tyrosine is oxidized resulting in a tyrosine radical and (ii) a radical–radical reaction occurs between the tyrosine radical and nitrogen dioxide (
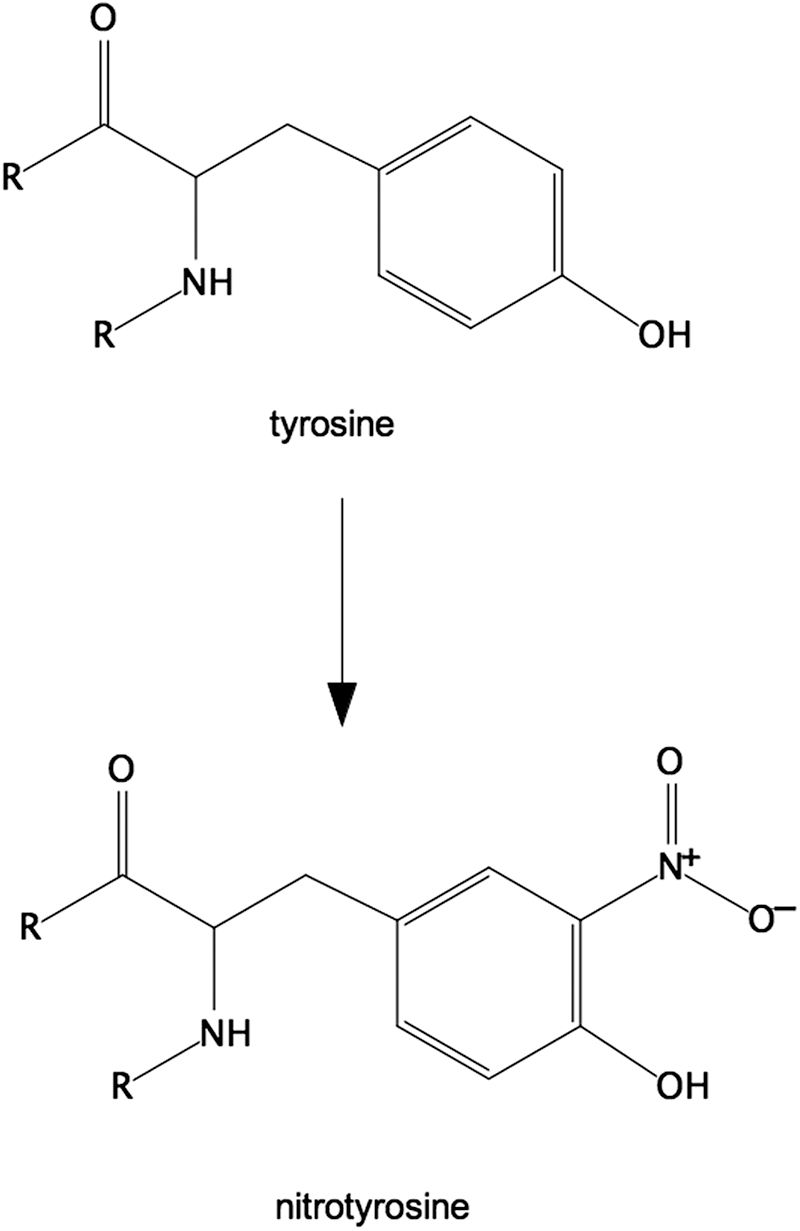
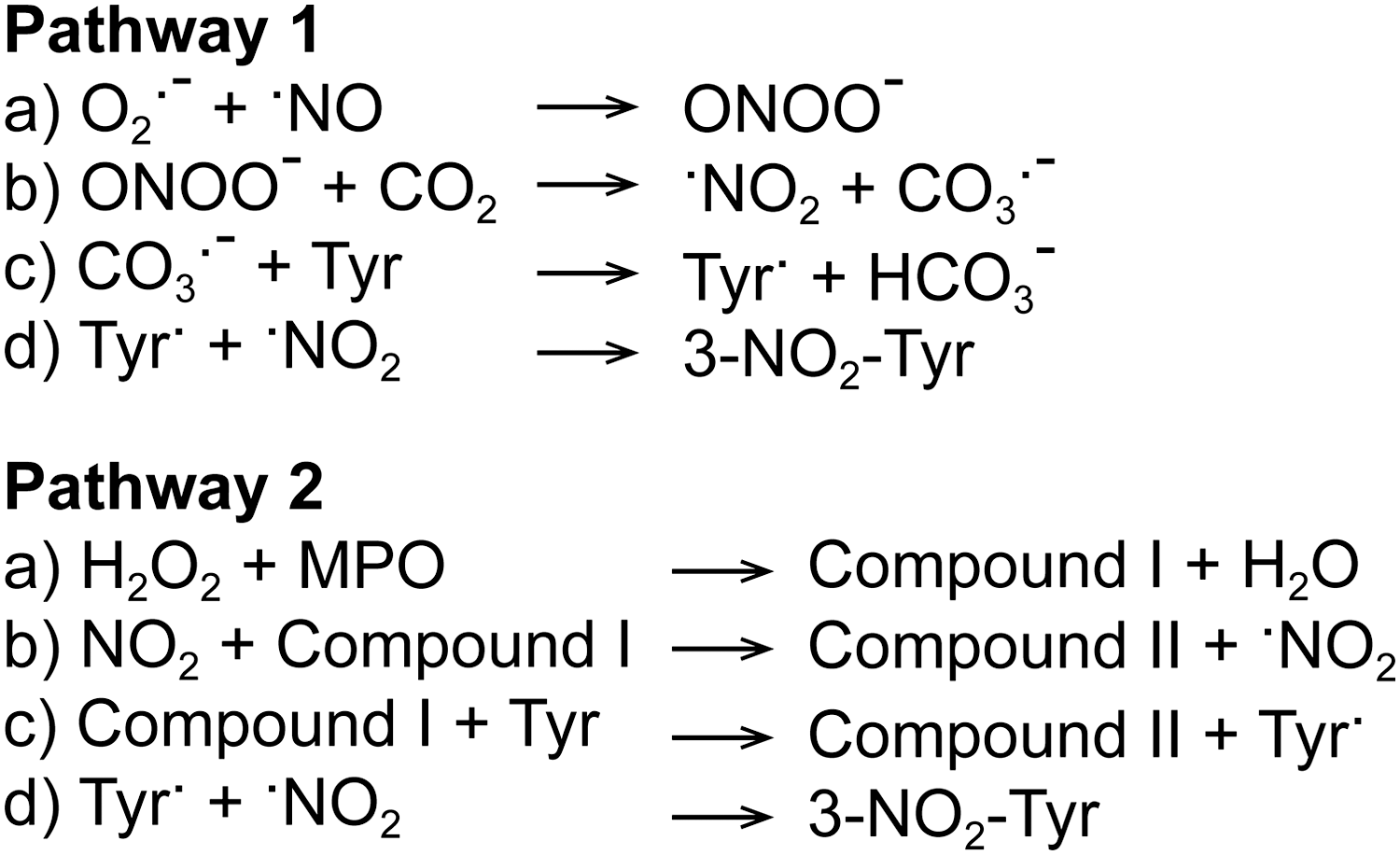
One widely studied pathway for nitration is the production of the RNS peroxynitrite (ONOO−) (Fig. 6, pathway 1) (132). Initially, Tyr-NO2 was believed to be a specific marker of peroxynitrite-mediated damage, but this has since been disproved, with the most-cited alternate pathway involving myeloperoxidase (MPO), as proposed in 1997 (183) (Fig. 6, pathway 2). When solely measuring Tyr-NO2 in complex biological samples, it is not possible to tell which of the mechanisms is responsible for any nitration detected in vivo. The effect of nitration on protein activity varies with the protein being studied and can be a gain of function (180) or loss of function (195).
The issue of which method to employ for the determination of Tyr-NO2 in the high-throughput analysis of clinical samples needs be addressed as MS, while considered the gold standard (176), is not yet feasible for high-throughput analysis and other methods suffer from methodological flaws or cannot be properly assessed due to a lack of detailed methodological information (45, 197). In addition, some authors fail to clearly state whether free Tyr-NO2, protein-associated Tyr-NO2, or total Tyr-NO2 is measured, the concentrations of which may be different. While evidence does suggest that nitration of certain proteins enhances proteolytic degradation (160), a fall in protein-associated Tyr-NO2 concentration will only be measurable if the nitrated protein is degraded in parallel with a decrease in disease activity.
There is still much work to do in assessing the utility of Tyr-NO2 as a clinical biomarker, but findings so far are encouraging, with some studies showing that plasma Tyr-NO2 levels correlate with disease activity and decrease following successful therapeutic interventions. Yet, it is still unclear whether Tyr-NO2 is any more informative, in clinical terms, than other already available markers, for example, C-reactive protein (CRP). CRP is an acute phase protein synthesized by the liver in response to signaling by upregulated inflammatory cytokines (e.g., IL-6). Serum CRP is widely used clinically as a marker of acute inflammation, but of course an increase in serum CRP concentration is delayed until some hours after the initial inflammatory insult because liver synthesis of CRP protein is required. In contrast, chemical modifications to pre-existing cellular or extracellular proteins may occur more swiftly within an inflammatory environment. One application where the measurement of Tyr-NO2 might provide a significant enhancement is in the monitoring of clinical responses to therapy in the assessment of the current pipeline of novel drugs targeted at enzymes involved in ROS generation, for example, NADPH oxidase and MPO inhibitors.
Tyr-NO2 can be detected in tissues by various methods. Among the quantitative approaches, both ELISAs and LC/GC have been described; however, it is important to note that quantification by ELISAs is limited by several factors: (i) antibodies employed in ELISAs will have different affinities for different nitrated proteins (94), although this is addressed by methods that measure a single nitrated protein (188); (ii) some Tyr-NO2 ELISAs may be prone to interference; and (iii) the sensitivity of some Tyr-NO2 ELISAs may be too low to detect Tyr-NO2 in healthy human plasma. LC/GC has been coupled to various detection systems, for example, UV detection, thermal energy analysis, electrochemical detection, and MS. One explanation for the lack of agreement between reported plasma ELISA- and MS-based Tyr-NO2 plasma concentrations is that the reported ELISA results are based on a nitrated bovine serum albumin standard, in which the actual concentration of nitrated tyrosine residues is unknown.
The National Institute of Environmental Health Biomarkers of Oxidative Stress Study concluded that oxidized proteins (of which Tyr-NO2 was one of the measured biomarkers) are not suitable biomarkers of free radical damage caused by carbon tetrachloride treatment in rats (89). Nevertheless, many studies have investigated Tyr-NO2 under inflammatory conditions as a marker of oxidative stress and have found plasma Tyr-NO2 concentrations to be higher in patients in a variety of diseases (summarized in Table 4). Some of the clinically relevant findings include the association of higher Tyr-NO2 levels with higher mortality in sepsis and a fall in plasma Tyr-NO2 concentration, as well as a correlation with other markers following treatment with various anti-inflammatory drugs. In addition, two studies using different methodologies reported a lowering of plasma protein-associated Tyr-NO2 concentrations upon drug treatment (statins in coronary artery disease and rosuvastatin in hypercholesterolemia). Tyr-NO2 may not only be a potential biomarker of oxidative stress in diabetes patients (28, 29) but may also be a biomarker of disease progression/risk for CVD (111a). In asthmatic patients, Tyr-NO2 staining in bronchial biopsies and measurement of Tyr-NO2 by ELISA in exhaled breath condensates both showed a decrease of Tyr-NO2 upon corticosteroid treatment. Conflicting results were obtained in neurodegenerative disorders; although Tyr-NO2 has been reported at increased levels within brain tissue from Parkinson's (60) and Alzheimer's disease patients (167), free Tyr-NO2 is not increased in the cerebrospinal fluid of Alzheimer's disease patients (143). Likewise, protein-bound Tyr-NO2 is not increased in plasma of patients with dementia (44). In contrast, nitrated α-synuclein was detected in peripheral blood mononuclear cells from Parkinson's disease patients, the concentrations of which were inversely correlated with the daily dosage of levodopa (Table 4).
References are provided as supplementary material.
CAD, coronary artery disease; CRP, C-reactive protein; OA, osteoarthritis; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; TNF, tumor necrosis factor.
Thiols
The main thiol compound in the body is the amino acid cysteine, which is susceptible to oxidation. Oxidation of a cysteine residue can change a protein's function. Thus, measuring the thiol status may represent a mechanism-based biomarker. Biologically occurring thiols include low-molecular-weight thiols (cysteine, GSH) and protein thiols.
The use of thiols as biomarkers is affected by a number of factors. In addition to specific limitations for those thiols that are only present intracellularly, a general problem is that oxidation products of protein cysteine residues are unstable. They can be easily reduced by other thiols and thus require immediate treatment with an alkylating agent to prevent further redox changes. A change in GSH levels might not be due to oxidative stress, but might reflect a nutritional/metabolic imbalance. Plasma GSH levels may also be affected by GSH transporters, while cellular mechanisms, such as nuclear factor (erythroid-derived 2)-like 2 (NRF2), counteract oxidative stress by increasing GSH synthesis. Finally, oxidized glutathione (GSSG) concentrations are very low and difficult to measure unless sensitive HPLC methods are used.
Glutathione
The main nonprotein thiol is the tripeptide GSH. Since intracellular concentrations of GSH are high, in the millimolar range, it is an important component of antioxidant defense systems to scavenge ROS, which leads to GSSG. Oxidation of GSH is reversible as GSSG reductase and NADPH reduce GSSG back to two molecules of GSH.
In general, any condition associated with excessive ROS will decrease GSH levels or decrease the GSH/GSSG ratio. Within cells, GSH is present at millimolar concentrations, resulting in high GSH/GSSG ratios (>30) (76). The GSH/GSSG ratio in serum is substantially lower (∼3). Whether this meaningfully reflects a cellular redox state is questionable (90) and it may not be a good indicator of oxidative stress (86). Thus, most studies measure erythrocyte GSH where GSH concentrations are high, but not necessarily a good indicator of oxidative stress across tissues. Furthermore, lower GSH levels may not necessarily be due to oxidation, but rather due to a consequence of lower cysteine levels (cysteine is the rate-limiting GSH precursor) due to nutritional deficiency. Nevertheless, many studies have measured plasma GSH/GSSG. Three meta-analyses confirmed a decrease in plasma GSH and an increase in plasma GSSG in patients with autism spectrum disorders (54) and lower plasma GSH levels in polycystic ovary syndrome (121), two conditions in which oxidative stress has been implicated (127, 144). It should be noted that these meta-analyses are based on studies where GSH/GSSG are measured by a variety of techniques, including enzymatic methods, HPLC with fluorometric, UV, or electrochemical detectors, and LC-MS/MS.
Several pathological conditions are associated with decreased GSH levels (6). In particular, studies on GSH in acquired immunodeficiency syndrome (AIDS) and other conditions have shown very clearly that rather than in plasma, GSH should be measured within cells by fluorescence activated cell sorting (72). GSH measurement is important to identify patients who may benefit from GSH repletion by GSH derivatives or precursors, for example, in clinical trials (6).
Cysteine
Free cysteine is the main nonprotein thiol in plasma (86, 118). Studies have measured plasma cysteine (∼10 μM) and its disulfide, cystine (∼40–50 μM), in CVDs with varying results (43, 112). Cysteine is a semiessential amino acid and its requirement may increase following oxidative stress due to the consumption of GSH (6). Historically, one important condition associated with lower plasma cysteine is AIDS, originally reported by the group of Droge (46).
Protein thiols and mixed disulfides
Protein cysteine residues can exist in many oxidation states (Fig. 7). Protein glutathionylation (mixed disulfides with GSH) received particular attention. Significant amounts of glutathionylated proteins are detected under normal conditions or following exposure to oxidants (58, 156). Most of the glutathionylated proteins are intracellular because GSH is predominantly present in the cytoplasm and intracellular proteins have cysteine residues predominantly in the chemically reduced state and thus are available to form mixed disulfides, in contrast to extracellular proteins where most cysteine residues are engaged in disulfide bridges.
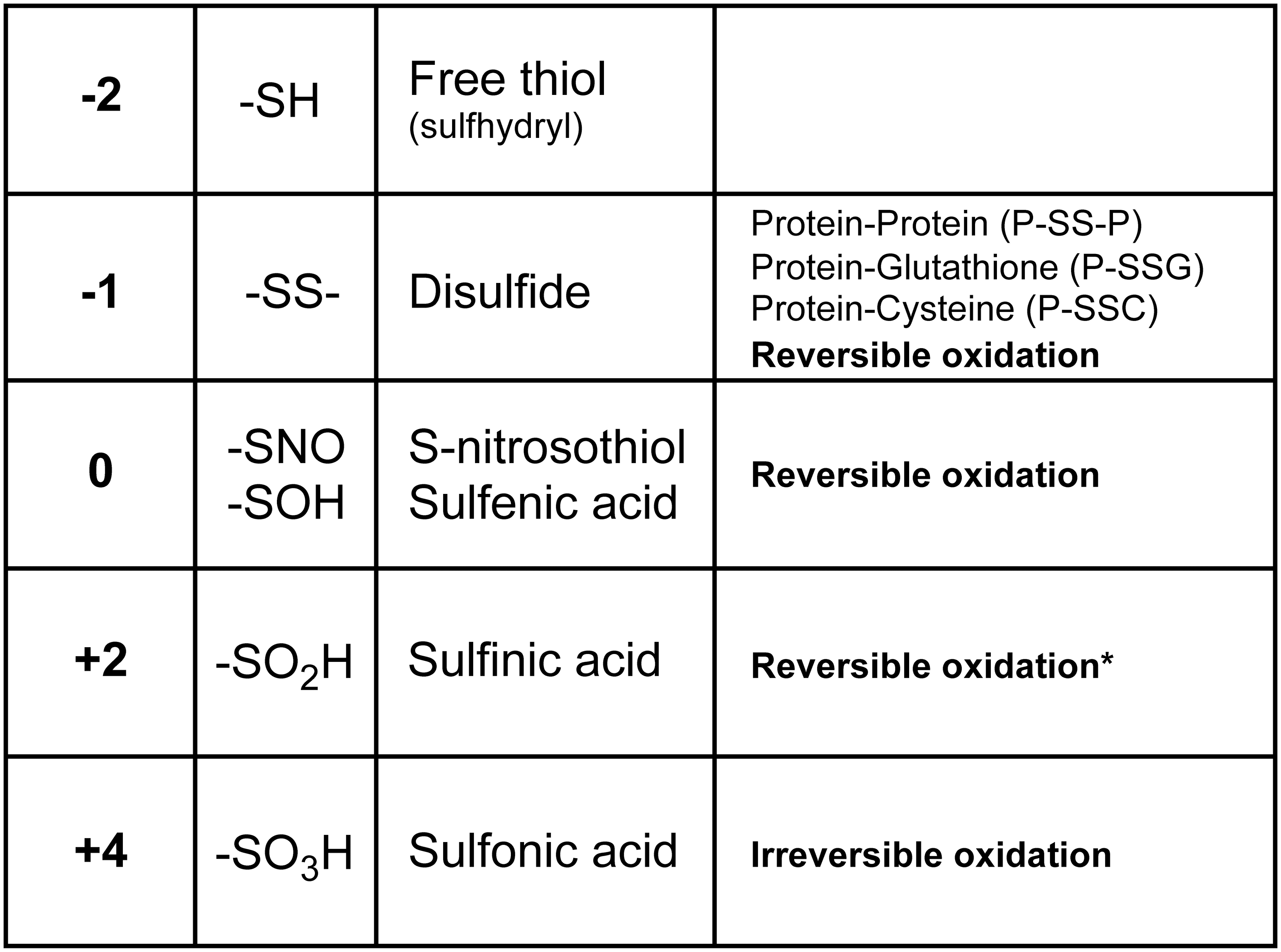
The only plasma protein identified as glutathionylated is transthyretin (58). Many studies propose glutathionylated hemoglobin, measured in red blood cells by MS, as a biomarker of oxidative stress in diabetes, hyperlipidemia, hemodialysis, and chronic renal failure (31, 58). An increase in plasma cysteinylated albumin, measured by MS, has also been reported in chronic liver and kidney diseases and diabetes (124).
Surface thiols
The plasma membrane is the interface between the reducing intracellular and the oxidizing extracellular environments. While one might expect the extracellular (exofacial) membrane thiols to be oxidized, they are in fact not, and active mechanisms maintain specific surface thiols (98), with surface thiols lower in rheumatoid arthritis (RA) (131). Their measurement may provide additional information on the redox state of a patient (145).
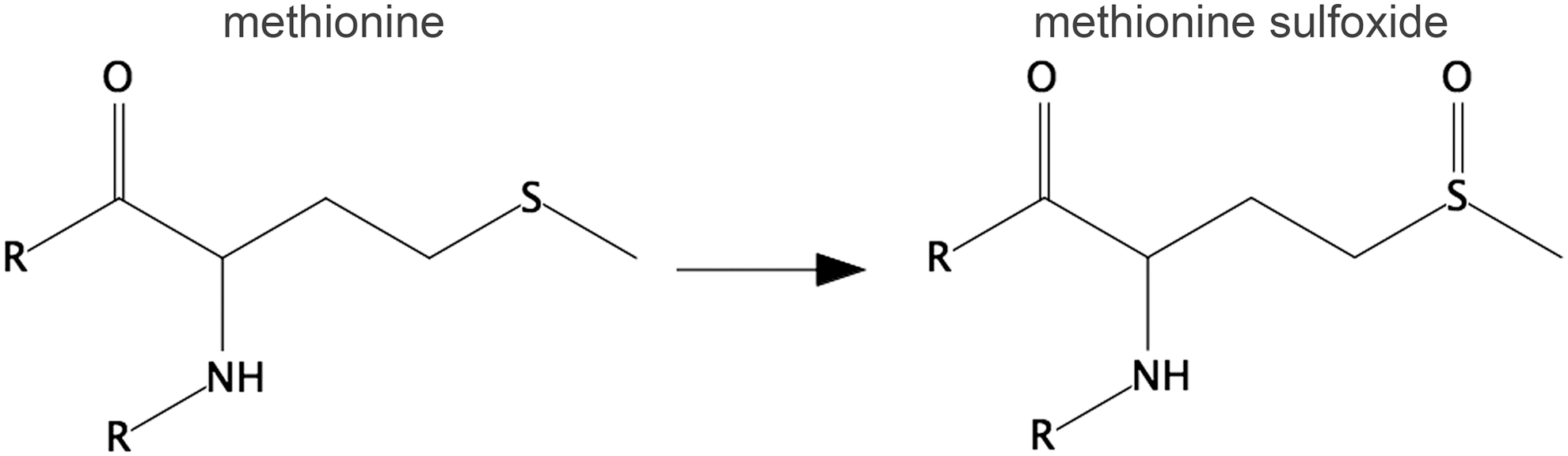
Methionine sulfoxide
Methionine is the other sulfur-containing amino acid beside cysteine (Fig. 8). Sulfur in methionine can be reversibly oxidized by ROS to a sulfoxide. Oxidation of an essential methionine in the abundant serum protein α-1-proteinase inhibitor leads to its inactivation (85). Elevated levels of this sulfoxidized form have been detected in the bronchoalveolar lavage of smokers, contributing to the pathogenesis of emphysema (27). The presence of methionine sulfoxide in plasma proteins and in HDL is increased in sepsis and diabetes (5, 21). Although not as widely studied as a form of thiol oxidation, methionine sulfoxide has potential advantages as a biomarker: it is easily measured with a conventional amino acid analyzer and is more stable than thiol oxidation products.
DNA/RNA oxidation
Oxidative stress induces oxidation of DNA and RNA (Fig. 9), particularly in the guanine moiety. The oxidized nucleosides are excreted into the urine and their measurement can be interpreted as the cumulative total body oxidative stress, that is, number of hits to the nucleic acids in a defined time period, meaning the global rate of DNA and RNA oxidation. Therefore, as urinary biomarkers, they are most relevant to conditions where oxidative stress occurs in all tissues in the body and less to high oxidative stress in minor organs without assumed systemic oxidative stress. Several commercial assays are available to measure 7,8-dihydro-8-oxo-2′-deoxyguanosine (8oxodG) with ELISA. However, the clinical significance of these methods has been questioned (10). While chromatography coupled to MS may not be readily available clinically, biomarkers for oxidative stress measured by oxidation of nucleic acids are among—if not the best—biomarkers that have been examined. Nucleic acid oxidation products have also been demonstrated to be predictive of the development of disease (22, 23).
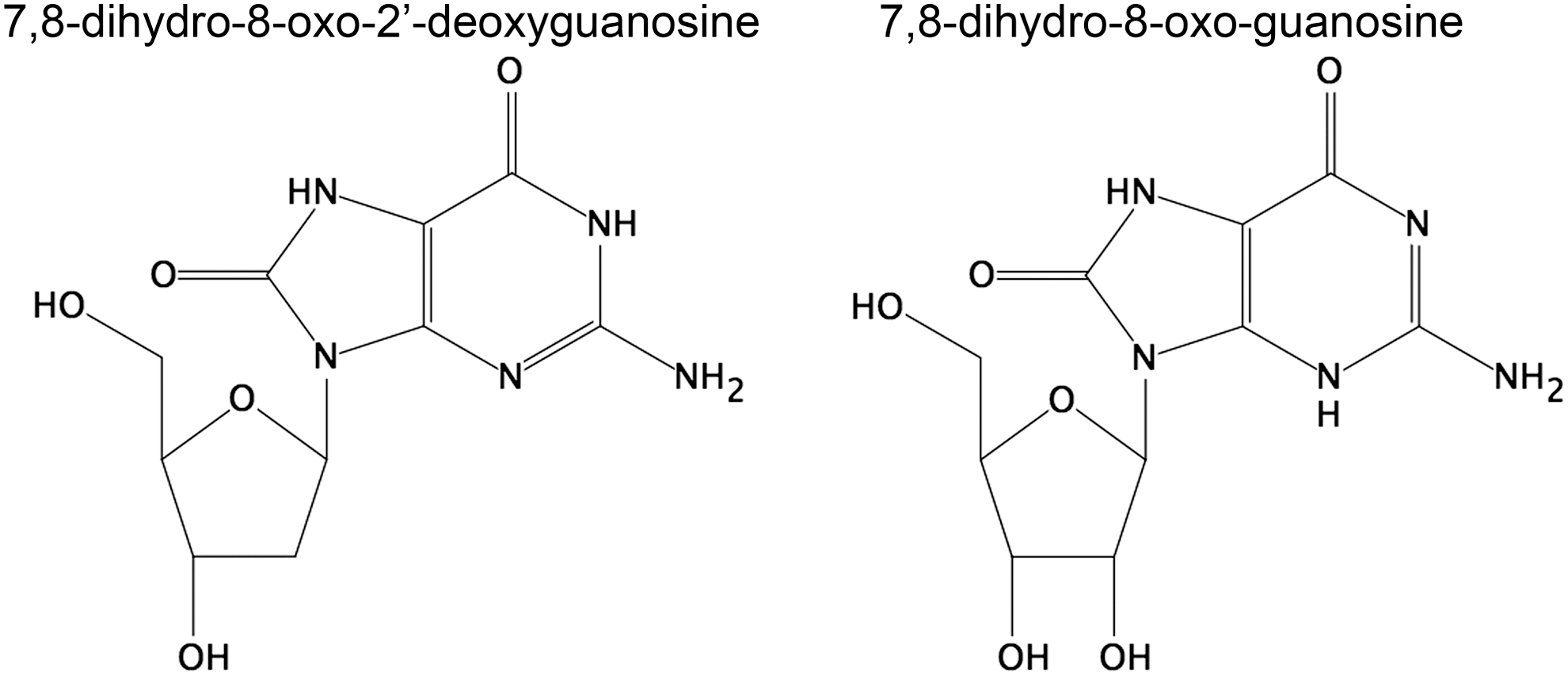
The oxidative modification in DNA can cause mispair and thereby lead to mutations, particularly GC-TA transversion mutations, and therefore relates to cancer (104, 134). Oxidative lesions in DNA are recognized by repair enzymes; the nucleotide pool can be oxidized, but is sanitized by other enzyme systems (133). There is some debate as to whether the lesions in DNA relate to incorporation from the nucleotide pool or direct oxidation in DNA (70). Chronically high oxidation of DNA, measured as urinary excretion of the nucleoside 8oxodG, is associated with risk of lung and breast cancer (103, 105).
Recently, RNA oxidation, measured as 7,8-dihydro-8-oxo-guanosine (8oxoGuo), has been introduced as a marker in relation to diseases, particularly neurodegenerative diseases and diabetes (22, 23, 88). Because of the single strand nature of RNA, repair is not possible. Remarkably, relatively little is known about how RNA integrity is maintained, but it is assumed to rely on quality control and degradation (133). The cellular effects of RNA oxidation also remain largely obscure, although formation of truncated or mutated proteins has been suggested (133, 135). There are indications of formation of mutated proteins (170) and of microsomal stalling induced by oxidized RNAs (159). Very recently, advanced methodology has demonstrated that the effects of RNA lesions fall into two categories, one that includes ribosomal stalling and one that leads to a mixture of full length and truncated translational products (26). It thus appears that nucleic acid oxidation/modification has much more diverse and multifaceted biological effects, exemplified both with different effects on translation stalling and also in the target molecule, for example, in diabetes where RNA oxidation is not only more pronounced than DNA oxidation but also has a very different prognostic value.
Extensive DNA oxidation is predictive for the risk of breast and lung cancer (103, 105). Elevated RNA oxidation is predictive for development of complications and death in type 2 diabetes, and there are indications that high RNA oxidation is associated with breast cancer development in type 2 diabetic females (22). Thus, screening for urinary DNA/RNA oxidation could help to identify such people and patients at risk and help to implement a treatment plan to minimize it.
For measurement of 8oxodG and 8oxoGuo in urine, the most reliable methodology is chromatography coupled with MS (189 –191). 8oxodG can also be measured by HPLC-electrochemical detection, which is rarely used presently.
Markers of ROS Generation
Some ROS-forming enzymes that are normally present intracellularly can also be found in the circulation, independently of the mechanism responsible for their release. For this reason, we will only describe xanthine oxidase (XO) and MPO. Higher circulating levels of XO and MPO could potentially result in increased ROS production, although this depends on other factors such as availability of the substrate (xanthine for XO and H2O2 for MPO) and whether ROS produced by these enzymes overcome the antioxidant defense. In some cases, a better indicator of the enzyme activity in vivo is the formation of the metabolite or reaction product.
Xanthine oxidase
XO catalyzes the oxidation of xanthine to uric acid. While the product is a known antioxidant (4), the enzyme is also a well-known source of O2
While we list XO among the ROS-generating enzymes, it could also be an indicator of oxidative stress. In fact, the protein exists in two forms, an oxidase (that oxidizes xanthine to uric acid using oxygen as the electron acceptor and produces H2O2) and a dehydrogenase (that carries out the same reaction, but uses NAD+ and generates NADH). The dehydrogenase form can be converted into XO by, among other things, thiol oxidation (48). Thus, oxidative stress will increase XO activity by increasing dehydrogenase-to-oxidase conversion.
Myeloperoxidase
MPO is a heme peroxidase that catalyzes the reaction between H2O2 and chloride ions to produce HOCl as the primary oxidant. These are not only important in the innate immune system's antimicrobial activities but also contribute to inflammatory diseases, such as atherosclerosis and related vascular diseases (35) and Parkinson's and Alzheimer's disease (32, 123, 139), via multiple mechanisms (166).
The suitability of MPO as a potential independent prognostic biomarker of inflammation is summarized in Table 5. A limitation of MPO as a biomarker is that current methods are not standardized between laboratories and do not provide direct information on MPO activity. The methods used to directly measure MPO activity as well as biomarkers specific for MPO have been reviewed recently (92). MPO-derived oxidants generate a footprint of specific and nonspecific oxidation products. 3-Chlorotyrosine (3-Cl-Tyr) and chlorinated lipids, as well as glutathione sulfonamide, are specific products for MPO/HOCl (92). Nonspecific oxidation products include protein carbonyls and 3-nitrotyrosine modifications. Of the specific biomarkers, 3-Cl-Tyr has received the most attention and has been detected in a wide variety of diseases (Table 6).
References are provided as supplementary information.
CBS, cardiac biomarker score; CVD, cardiovascular disease; IL-6, interleukin-6; MACE, major adverse cardiac event; MI, myocardial infarct; MPO, myeloperoxidase; PAD, peripheral artery disease; PCR, polymerase chain reaction; TnT, troponin T.
References are provided as supplementary material.
AMI, acute myocardial infarction; ACS, acute coronary syndrome; HDL, high-density lipoprotein.
Chlorinated lipids have been detected in human atherosclerotic plaque (173). 2-Chloradipic acid (185) is excreted in urine and hence offers the potential as a biomarker. Glutathione sulfonamide is present in the airways of preterm infants with respiratory disease as well as in children suffering from cystic fibrosis (93).
A general limitation of the specific biomarkers of MPO activity is the requirement for expensive equipment and time-consuming sample workup and analysis. Often, concentration of these biomarkers in biological samples is low, which complicates accurate measurement. As a result, investigators have fractionated plasma and observed that HDL can be the major carrier of 3-Cl-Tyr in CVD (15). However, the extensive preparation procedures for HDL analysis limit its clinical use. Glutathione sulfonamide is a relatively minor oxidation product derived from the reaction of reduced glutathione (GSH) with HOCl. This limits its application to biological samples that contain significant amounts of GSH. Plasma, which has very little GSH, is therefore not a suitable source to analyze glutathione sulfonamide.
Within these limitations, the determination of MPO protein is a reasonable approach to at least initially assess a potential contribution of MPO-mediated oxidative damage to a disease, and in most studies, MPO and specific MPO activity biomarkers with different specificities provide similar results (Tables 5 and 6).
Markers of Antioxidant Defense
In principle, oxidative stress can also derive from an impaired antioxidant defense. We focus here not only on protein thiol-disulfide oxidoreductases that can be measured in serum or plasma but also the transcription factor NRF2 that drives the transcription of several antioxidant genes. NRF2 is activated in response to oxidative stress and its activation could therefore be used as an indicator of ROS generation that exceeded the existing antioxidant defense systems.
Protein thiol-disulfide oxidoreductases
These enzymes include, among others, thioredoxin (Trx) and peroxiredoxins (Prxs; Fig. 10). Trx main function is to keep protein thiols in the reduced state. For this reason, Trx is often regarded as an antioxidant enzyme. Secretion of Trx was originally discovered in leukemia cells by virologist Junji Yodoi, who initially thought he had identified interleukin-1-γ (199). While it cannot be excluded that Trx secretion may be induced by other factors, it is thought that its secretion is induced by oxidative stress. Its secretion is inhibited by antioxidants in vitro (96) and its plasma levels are elevated in cancer patients (8) and AIDS patients, where it negatively correlates with survival (125). Various reports have suggested that plasma/serum Trx concentrations are diagnostically relevant in a range of diseases. For instance, Qi et al. found that serum Trx concentrations reflected disease severity in acute ischemic stroke (137), and the addition of Trx to an established disease severity score provided a significant improvement in diagnostic capacity.
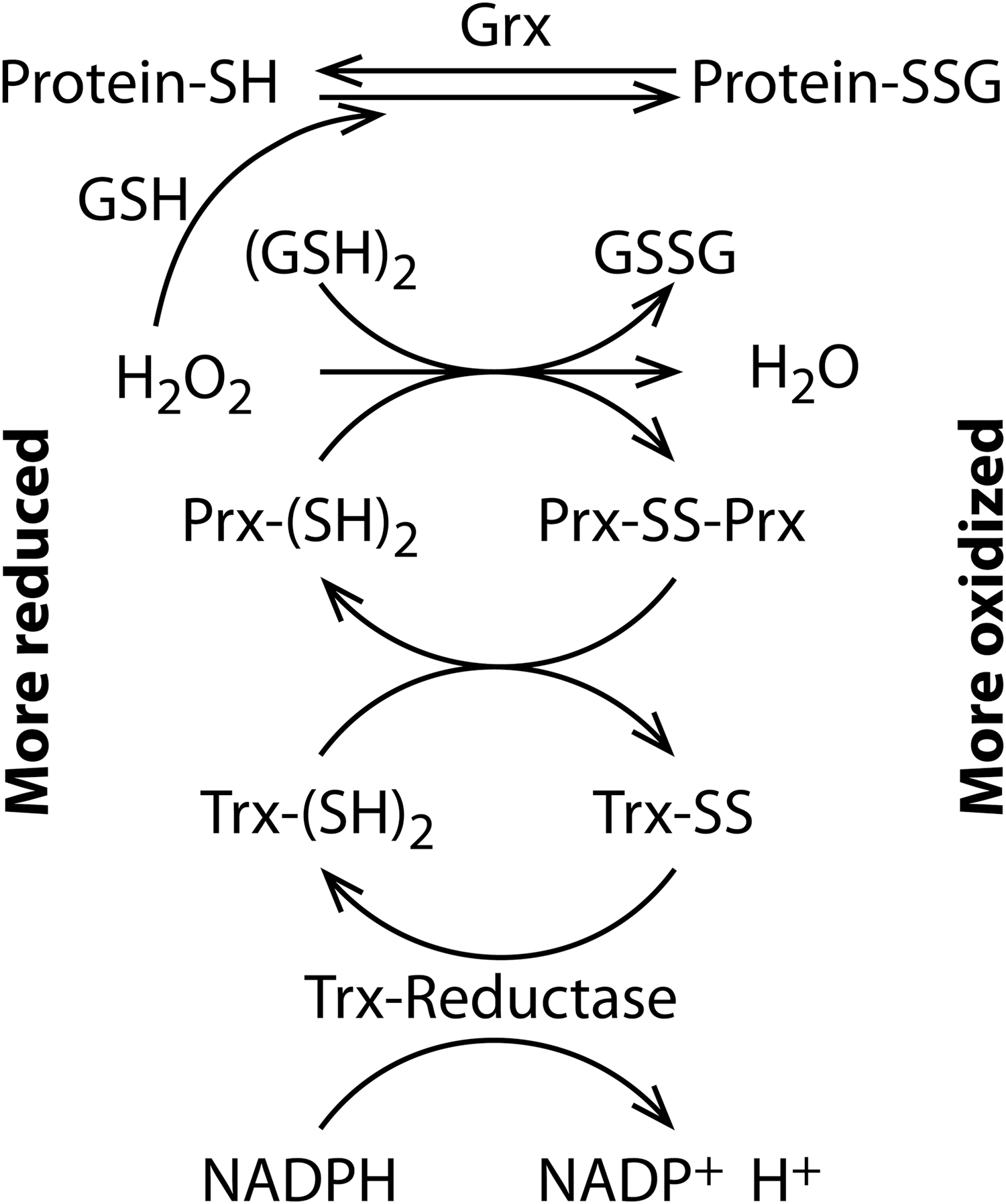
The Prx family has a key role in the elimination of H2O2 because it is highly abundant and reacts with H2O2 at high rates. Prxs are often identified in proteomics studies and several studies identified Prxs in the secretome under various disease conditions (80). In particular, oxidized Prx6 in the cerebrospinal fluid has been proposed as a biomarker of oxidative stress in brain injury, where it is a good predictor of the outcome (106). The importance of the redox state of Prxs as biomarkers is also demonstrated by studies showing that Prx2 is secreted via a nonclassical pathway by inflammatory cells (151) independently of cell death (30) and is present in the serum and synovial fluid of RA patients (168). Bayer et al. (13) have shown in vivo that erythrocyte Prx2 undergoes a transient overoxidation following lipopolysaccharide exposure, and released Prx1/2 is present in the oxidized form (30, 151), suggesting that its redox state, not just its levels, can be an early marker of oxidative stress.
Human serum Prx4 has also been proposed as a diagnostic marker of oxidative stress in diabetes (1).
Nuclear factor (erythroid-derived 2)-like 2
The transcription factor NRF2 is considered a prognostic biomarker in cancer. It is found constitutively and at very high levels in biopsies of malignant tumors with high proliferation rates and poor response to anticancer drugs. NRF2 is a master regulator of the antioxidant response that controls the expression of about 250 genes. Some of these genes encode antioxidant enzymes, including those involved in synthesis and use of GSH and Trx, or participate in metabolic reactions that generate reductive power in the form of NADPH. Others participate in phase II biotransformation reactions and phase III external transport of toxins. Thus, high and constitutive activity of NRF2 provides transformed cells with a defense against the ROS that are generated during active proliferation, while at the same time reducing the effective dose of certain anticancer drugs. To date, a prognostic value of NRF2 has been reported for adenocarcinoma (79), squamous cell carcinoma (75), colon (84), lung (196), and breast cancer (129), among many others. NRF2 does not initiate carcinogenesis, but potentiates tumor promotion or metastasis. Somatic mutations in the NRF2 gene and in its repressor, KEAP1, have been found in a large percentage of human tumors, leading to constitutively increased NRF2 levels (68). Moreover, malignant transformation elicited by some oncogenes such as KRAS (42) or loss of tumor suppressor PTEN (142) leads to abnormally high levels of NRF2 protein and activity. Thus, analysis of NRF2 levels by either immunoblot or qRT-PCR in tumor biopsies is becoming a tool to clinically assess tumor malignancy.
NRF2 activity declines with age as well as in degenerative disorders. Considering that NRF2 is being validated as an antioxidant and immunomodulator target for diseases with an autoimmune component or with low-grade chronic inflammation, it will be necessary to develop reliable predictive markers that help to monitor effective targeting. The most significant advancement in development of these prospective biomarkers has been in patients with chronic obstructive pulmonary disorder. In a small cohort of these patients, it was reported that dietary administration of sulforaphane-rich broccoli sprout extract for two weeks enhanced the mRNA levels of several NRF2-regulated genes in peripheral blood mononuclear cells (67).
Downstream Functional Markers of ROS-Induced Damage
The biomarkers described above are indicative of increased ROS levels, either by increased formation or decreased removal. An alternative would be markers that reflect oxidative stress downstream of the ROS-induced damage. Ideally, this marker would be a direct risk factor so that its modulation by therapeutic interventions would predict a positive outcome. Two markers appear to qualify for this, asymmetric dimethyl L-arginine (ADMA) and phosphorylated vasodilator-stimulated phosphoprotein (P-VASP).
Asymmetric dimethyl L-arginine
ADMA is a ubiquitous metabolite derived from protein modification and degradation. Upon accumulation, it can interfere with arginine metabolism and
Phosphorylated vasodilator-stimulated phosphoprotein
The best-established marker for physiological cyclic guanosine monophosphate (cGMP) signaling is probably P-VASP. It is phosphorylated mainly by cGMP-dependent protein kinases (25), and lowered P-VASP levels are indicative of pathological signaling (77, 110). This pathological signaling can be brought about by oxidative stress, for example, through scavenging of
Conclusion
The markers discussed here have been studied in different disease settings and with different rigor, ranging from meta-analyses of several clinical studies to promising evidence in preclinical studies (Table 7). However, even when the highest evidence level is available, their specificity as a biomarker of oxidative stress could be questionable, as in the case of oxLDL. Oxidative stress likely plays a role in several diseases, yet very few oxidative stress markers have made it into routine clinical use, which may have several reasons. The properties of the oxidative modifications, such as the labile nature of cysteine modifications, or their low abundance poses significant challenges to translate them into a high-throughput, cost-effective clinical diagnostic. Stable oxidative modifications, such as protein carbonyls, certain lipid oxidation products, DNA/RNA oxidation, and 3-nitrotyrosine, certainly circumvent the first issue, which likely contributes to some of their positive clinical findings. Another limitation is methodology. While MS provides sensitivity and specificity and has become more accessible, antibody-based methods remain, for now, the clinical standard. However, as we have seen, some of these methods fall short on specificity, such as antibodies specific for oxLDL, and any new antibody-based marker requires rigorous testing for specificity and sensitivity. Other antibody-based methods, such as immunohistochemistry, require tissues that are not commonly accessible. Circulating cell harvesting methods may provide a future solution to this. For a new biomarker to be established for clinical use, it would also require additional benefit over established clinical markers. Paradoxically, this additional value of oxidative stress biomarkers may come from being indicators of a disease mechanism common to several pathologies rather than diagnostic for a specific disease. Oxidative stress biomarkers may help in identifying patient populations that benefit from certain treatments, allowing patient stratification based on pathogenic mechanisms rather than just disease severity, thus responding to a specific request from regulatory agencies (47). On the other hand, protein-specific modifications such as nitrotyrosine could be disease-specific biomarkers of oxidative stress (Table 4).
References are provided as supplementary information.
Categories are based on the following evidence levels: (A) Meta-analysis or large prospective studies, (B) Prospective studies, (C) Retrospective studies, and (D) Case reports or preclinical studies.
AD, Alzheimer's disease; ADMA, asymmetric dimethyl L-arginine; AIDS, acquired immunodeficiency syndrome; ASD, autism spectrum disorders; CKD, chronic kidney disease; CPB, cardiopulmonary bypass; CRF, chronic renal failure; FC, flow cytometry; IsoLG, isolevuglandins; MCI, myocardial infarction; ox-LDL, oxidized LDL; PCI, percutaneous coronary intervention; qRT-PCR, quantitative real time polymerase chain reaction; UPLC, ultra performance LC.
Outlook
One way forward may be the analysis of oxidative stress markers for specific proteins. Such markers might better represent an underlying specific disease mechanism and a means for therapeutic monitoring and outcome prediction. In addition, as many of the markers have been measured in similar diseases, a combination of them in large-scale panels and pattern analysis could provide an additional approach to measure disease progression or therapeutic outcome (Fig. 3). This will help overcome the problem of the fragmentation of the literature in the field as different markers of oxidative stress are measured in different diseases. Measurement of larger panels of biomarkers in key conditions will help give a more comprehensive picture of their significance. In parallel with the exciting developments on ROS-validated targets and clinical indications, those markers and patterns that correlate best with treatment efficacy or mortality will eventually advance the field of ROS biomarkers, for example, in the form of theranostic couples of a new drug comarketed with a diagnostic marker.
Footnotes
Acknowledgments
The authors acknowledge financial support from the European Cooperation in Science and Technology (COST Action BM1203/EU-ROS) (P.G.W., N.Z., T.R., A.C., H.E.P., H.H.H.W.S., P.G.), the Peptide Research Network of Excellence, European Cross-border Cooperation Programme INTERREG IV A France (Channel)-England, ERDF (P.G.), the Toyota foundation, Denmark (H.E.P.), a Senior Principal Research Fellowship from the National Health and Medical Research Council and National Health and Medical Research Council Project Grants 1020776 and 1060804 (R.S.), and National Institutes of Health Grant HL116263 (S.S.D.). A.K. is supported by a grant from the Defence Science and Technology Laboratory, United Kingdom, to P.G.W. and E.L.T.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
