Abstract
Introduction
E
Peroxiredoxin-2 (Prx2) is a typical 2-cysteine peroxiredoxin that is able to efficiently scavenge a low concentration of H2O2 without inactivation due to over-oxidation (9a, 21, 29, 33). Although progress has been made in functional characterization of Prx2 as an antioxidant system in erythrocytes and in various cell models (29, 50), much still remains to be investigated on the role of Prx2 during erythropoiesis. Recently, we reported that Prx2 expression is upregulated during both murine and human β-thalassemic erythropoiesis, suggesting a potential functional role for Prx2 to serve as the stress-response cytoprotective system in pathological erythropoiesis (7, 9a, 13, 21, 33). In other cell models, Prx2 expression has been reported to be under the control of different transcriptional factors such as nuclear factor-erythroid 2 (Nrf2) (45), Foxo3a (37), STAT3 (47), and NF-kB (49).
Prx2−/− mice show abnormal erythropoiesis similar to that seen in β-thalassemia, and the absence of peroxiredoxin-2 (Prx2) worsens ineffective erythropoiesis of β-thalassemia. Prx2−/− and Hbb3th/+ mice show activated nuclear factor-erythroid 2 and upregulation of antioxidant responsive element-containing genes of antioxidant systems to reduce oxidant damage. Fused recombinant Prx2 fused to cell-penetrating carrier cell-penetrating peptides (PEP1) peptide ameliorates Hbb3th/+ hematological phenotype and erythropoiesis.
β-Thalassemias are common inherited red cell disorders characterized by absent or reduced synthesis of β globin chains. Despite extensive knowledge of the molecular defects causing thalassemia(s), less is known about the mechanisms responsible for the associated ineffective erythropoiesis (44). Increased levels of ROS have been reported to contribute to the anemia of β thalassemia; however, the protective mechanisms against oxidative stress in β thalassemia have not been comprehensively addressed (1, 17).
Activation of redox-sensitive transcriptional factors such as Nrf2, which induces transcription of antioxidant genes (18), has been implicated as the physiological cellular response to acute and/or chronic oxidative stress in different cell types. Nrf2 is ubiquitously expressed and binds to the antioxidant responsive element (ARE), sustaining the induction of cytoprotective genes such as heme-oxygenase-1 (HO-1) or NAD(P)H dehydrogenase quinone 1 (nqo1), Prx1, and Prx6 (30, 46). The upregulation of these systems limits the ROS generation, thereby contributing to cellular resistance and survival against cytotoxic events such as oxidative stress-mediated cytotoxic events. It is of interest to note that Nrf2 is also involved in the expression of sulfiredoxin (Srxn), thioredoxin reductase (Txnrd), and sestrin-2 (Sesn2), which are systems involved in reversing Prx2 over-oxidation (22, 30, 41, 48). Mice genetically lacking Nrf2 develop an immune-mediated anemia with the age and show reduction in the expression of ARE-genes in the spleen (26).
In this study, we sought to determine the role of Prx2 as a redox-switch protein during erythroid maturation by focusing on stress erythropoiesis. Our goal was to examine whether the absence of Prx2 in the context of thalassemic background (Prx2−/−Hbbth3/+) will impact pathological erythropoiesis, a physiologically relevant model of stress erythropoiesis. We report that the absence of Prx2 is associated with (i) ineffective erythropoiesis; (ii) increased ROS levels in erythroid precursors; and (iii) activation of Nrf2 with upregulation of Nrf2-activated antioxidant genes such as HO-1. We propose that Prx2 co-operates with Nrf2 and acts as an adaptive mechanism to protect against severe oxidative stress. In fact, the extent of Nrf2 activation was higher in Prx2−/−Hbbth3/+erythroid precursors compared with either normal or Hbbth3/+ erythroid precursors. The administration of the recombinant protein PEP1Prx2, in which Prx2 is fused to cell-penetrating carrier cell-penetrating peptides (PEP1) peptide (5, 20), to Hbbth3/+ mice decreased the extent of ineffective erythropoiesis with a significant reduction in ROS levels and apoptosis. Our data suggest that Prx2 is a key cytoprotective system in stress erythropoiesis and in conjunction with Nrf2 to limit amplification of oxidative damage in β-thalassemia. These findings suggest potential novel therapeutic options for treatment of β-thalassemia through the modulation of endogenous antioxidant systems to overcome the imbalance between oxidative stress and cytoprotective mechanisms.
Results
Prx2−/− mice show ineffective erythropoiesis
Previous studies have shown that the absence of Prx2 results in chronic hemolytic anemia with splenomegaly and increased basophilic erythroblasts in Prx2−/− juvenile mice (25). In this study, we used the recently developed flow cytometry strategy that combines CD44, TER119, and cell size as markers to quantitate terminal erythroid differentiation. We used the same strategy to sort erythroid precursors at each distinct developmental stage (Population I: pro-erythroblasts; population II: basophilic erytroblasts; population III: polychromatic erythroblasts; population IV: orthochromatic erythroblasts) (13, 28). As shown in Figure 1A, Prx2−/− erythroid cells showed an irregular nuclear shape and decreased nuclear condensation, nuclear fragmentation, and multinuclear erythroblasts, cellular features characteristic of dyserythropoiesis. This was associated with extramedullary erythropoiesis as evidenced by increased erythropoietic activity in Prx2−/− spleen (Supplementary Fig. S1A; Supplementary Data are available online at
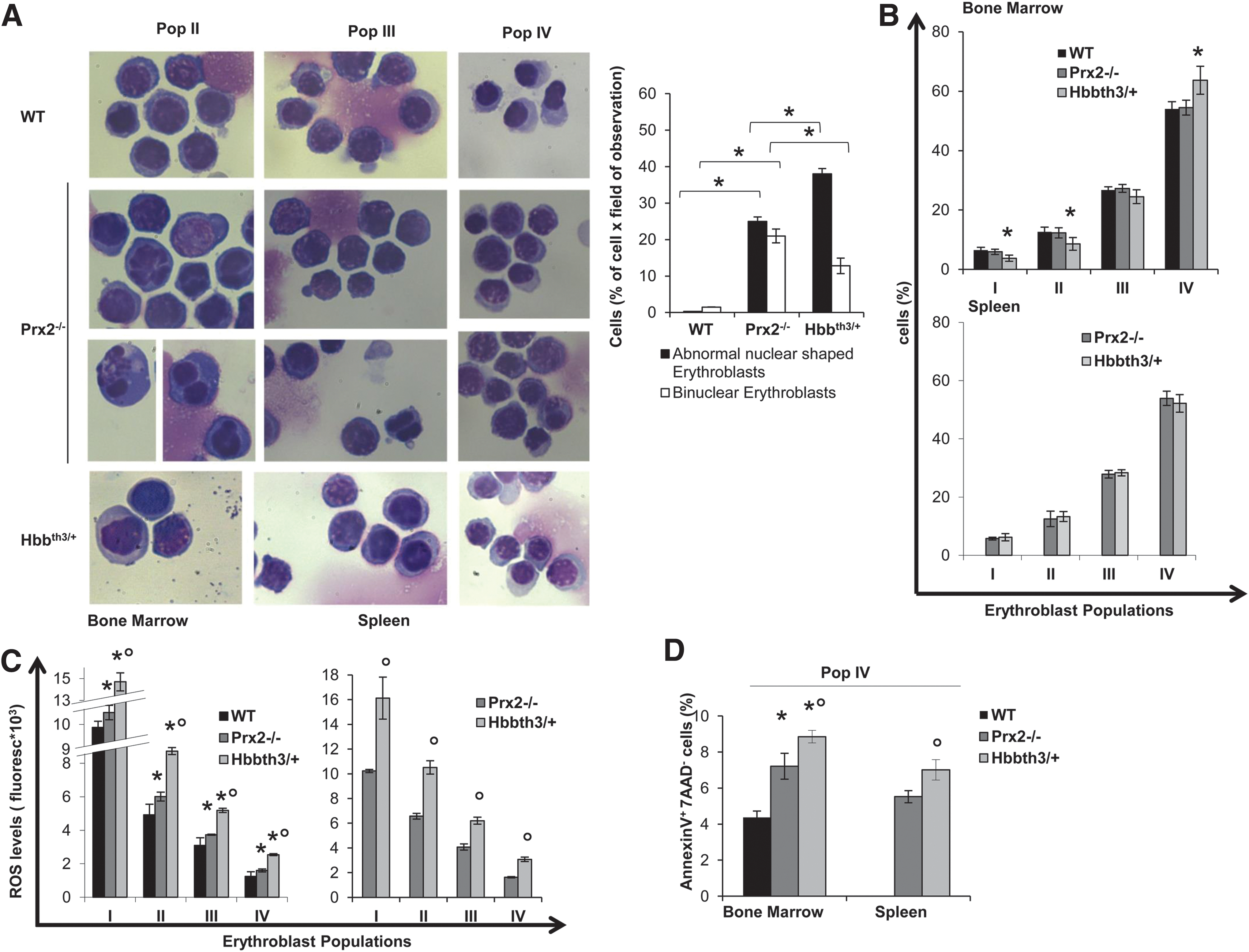
β-Thalassemia is a paradigmatic model of ineffective erythropoiesis with dyserythropoiesis mainly related to oxidative stress (7, 43). Hbbth3/+ mice showed dyserythropoiesis and ineffective erythropoiesis with expansion of erythroid precursors with extramedullary erythropoiesis (Fig. 1A, B). In bone marrow of Hbbth3/+ mice, we found a significant reduction in basophilic erythroblasts (pop II) and polychromatic erythroblasts (pop III) and increased orthochromatic erythroblasts compared with wild-type (WT) mice (Fig. 1B). The maturation profile of erythroid populations from the spleen of Prx2−/− and Hbbth3/+ mice was similar (Fig. 1B). These data suggest that the absence of Prx2 promotes ineffective erythropoiesis with expansion of erythroid precursors similar to β-thalassemia. To test this hypothesis, we evaluated ROS levels during erythropoiesis in Prx2−/− and Hbbth3/+ mice compared with WT.
ROS levels and apoptotic cells are increased in erythroid precursors from Prx2−/− mice
As shown in Figure 1C, ROS levels were significantly increased in bone marrow basophilc and polychromatic erythroblasts from Prx2−/− compared with WT mice. Such an increase in ROS levels was a feature of erythroid populations at all different stages of terminal erythroid differentiation from Prx2−/− mouse spleens (Fig. 1C). The finding of increased ROS levels only in basophilic and polychromatic erythroblasts from bone marrow of Prx2−/− mice may be related to different mechanisms involved in heme biosynthesis in the bone marrow and spleen involving macrophages. This is in agreement with our recent observation that Prx2 binds heme in erythroid precursors (6). In Hbbth3/+ mice, we found significant increased ROS levels in all erythroid populations from bone marrow and spleen compared with either WT or Prx2−/− mice (Fig. 1C). This might be related to the combination of different factors contributing to ROS accumulation in Hbbth3/+ erythroblasts such as iron import, heme biosynthesis, and the imbalance in globin-chain synthesis. To evaluate whether apoptosis might contribute to ineffective erythropoiesis in Prx2−/− similar to that observed in Hbbth3/+ mice, we evaluated the amount of Annexin-V+ 7-aminoactinomycin D (7-AAD)-negative cells used to define early apoptotic cells. As shown in Figure 1D, we found a significant increase in apoptotic orthochromatic erythroblasts in both Prx2−/− and Hbbth3/+ mice compared with WT. Similar changes were also noted in the orthochromatic erythroblasts from Prx2−/− and Hbbth3/+ mouse spleens. The amount of apoptotic orthochromatic erythroblasts was higher in Hbbth3/+ than in Prx2−/− mice (Fig. 1D).
These data suggest that the absence of Prx2 sustains ineffective erythropoiesis with increased ROS production and apoptosis compared with WT mice and partially mimicking Hbbth3/+ mice.
In order to test the link between the ineffective erythropoiesis of Prx2−/− mice and increased oxidative stress, Prx2−/− mice were treated with N-acetyl-cysteine (NAC) as an exogenous antioxidant, which has been previously shown to ameliorate Hbbth3/+ erythropoiesis (31). In Prx2−/− mice, NAC significantly increased hematocrit (Hct) and hemoglobin (Hb) levels and reduced splenic erythropoiesis (Supplementary Fig. S1B, C).
These data indicate that the ineffective erythropoiesis of Prx2−/− mice is related to increased ROS production similar to that observed in Hbbth3/+mice. However, the preservation of erythropoietic activity of Prx2−/− mice in the absence of Prx2 suggests that the activation of additional adaptive mechanism(s) against the chronic oxidative stress is likely to ensure erythroblast survival and maturation.
Prx2−/− mouse erythroid precursors show activation of Nrf2 and upregulation of cytoprotective genes in response to oxidative stress
Recent studies using other cellular models have pointed out a critical role of Nrf2, a redox-related transcriptional factor that induces the upregulation of antioxidant genes in response to oxidative stresses (18, 36). In this context, we found increased nuclear localization of Nrf2 in polychromatic and orthochromatic Prx2−/− and Hbbth3/+ erythroblasts compared with WT (Fig. 2A). This was confirmed by immunoblot analysis with specific antibodies, showing increased active Nrf2 (phosphor-Nrf2-S40; P-Nrf2) (39) and total Nrf2 in Prx2−/− compared with WT (Fig. 2B). To validate both the immunostaining and immunoblot data on Nrf2 activation and nuclear localization, we analyzed Nrf2 in cytosolic and nuclear fractions from flow sorted erythroid precursors. As shown in Supplementary Figure S1D, Nrf2 nuclear localization was increased in Prx2−/− compared with WT mice, further validating Nrf2 activation in Prx2−/− mice. As it has been previously reported that Nrf2 is activated only in response to oxidative stress (18, 19), it is not surprising that WT erythoblasts express low levels of active Nrf2 (Fig. 2B). In order to test the inducability of Nrf2 in WT mice in response to oxidation, we treated WT mice with phenylhydrazine (PHZ) to induce a severe oxidation damage, mimicking the β-thalassemic hematological phenotype (31). As shown in Supplementary Figure S1C, PHZ (40 mg/kg ip) induced a dramatic drop in Hct levels at day 2 after PHZ injection followed by the gradual recovery, reaching baseline values 13 days after PHZ treatment. At day 2 after PHZ injection, we found increased nuclear localization of Nrf2 in both polychromatic and orthochromatic erythroblasts from PHZ-treated WT compared with vehicle-treated WT mice (Supplementary Fig. S2B), supporting the transient activation of Nrf2 in response to oxidative stress.
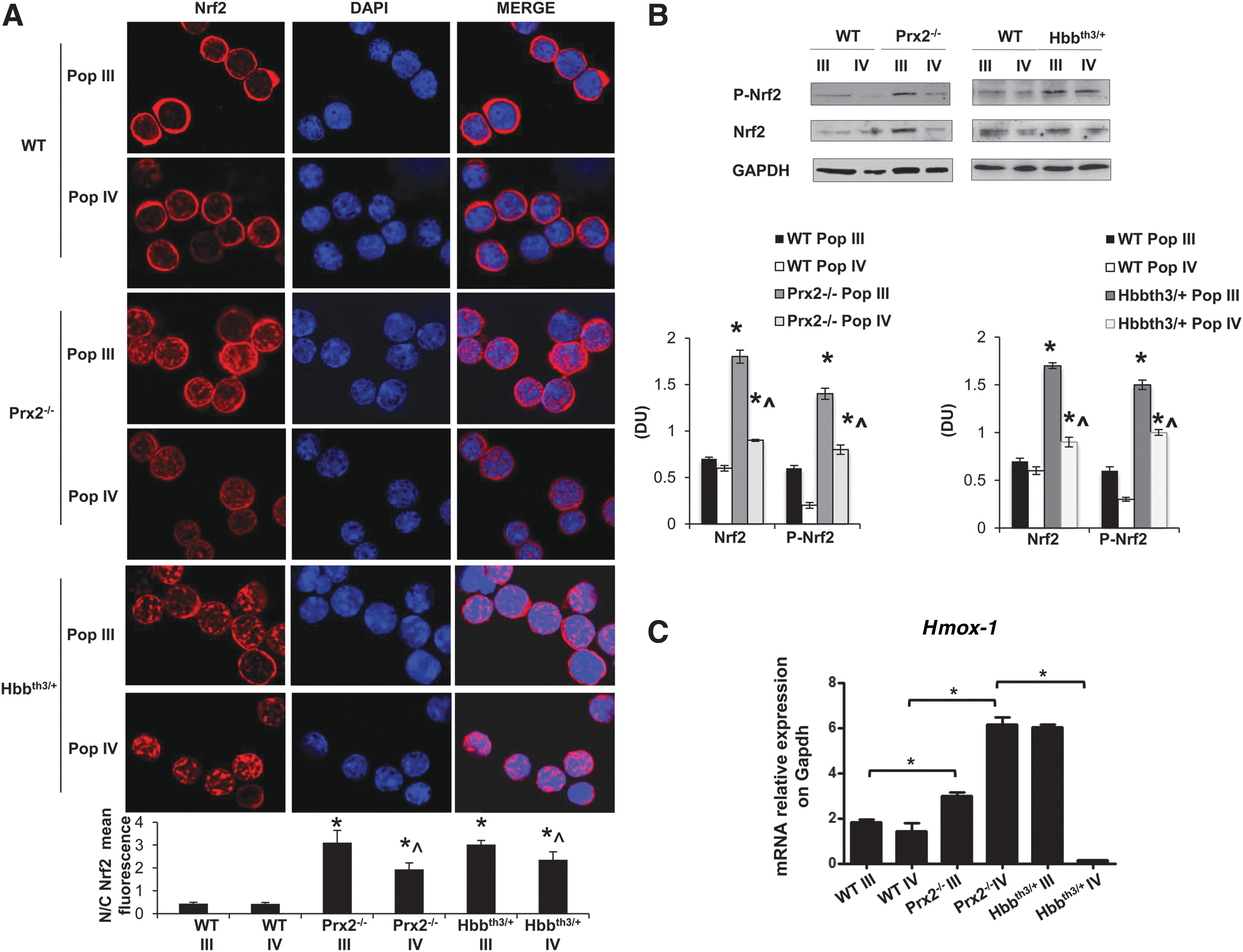
The increased activation of Nrf2 in Prx2−/− and Hbbth3/+ erythroid precursors was associated with upregulation of HO-1 gene expression, an ARE-redox-sensitive gene (Fig. 2C). Prx2−/− mice showed increased expression of HO-1 in both polychromatic and orthochromatic erythroblasts compared with WT (Fig. 2C), whereas Hbbth3/+ mice showed increased Hmox-1 mRNA levels only in polychromatic erythroblasts (Fig. 2C). These data indicate that Prx2−/− and Hbbth3/+ mice are characterized by increased oxidative stress and constitutive activation of Nrf2 during erythropoiesis. In contrast, in WT erythroid precursors Nrf2 is only transiently activated in response to exogenous oxidative damage.
We finally evaluated the expression of Srxn, Txnrd, and Sesn2, which are important systems in preventing Prx2-overoxidation (22, 30, 41, 48). In Hbbth3/+mouse polychromatic erythroblasts, Txnrd and Sesn2 expression was similar to WT mice and upregulated compared with Prx2−/− mice, whereas Srxn expression was downregulated compared with WT. In Hbbth3/+mouse orthochromatic erythroblasts, Srxn, Txnrd and Sesn2 were markedly upregulated compared with either WT or Prx2−/− mice (Supplementary Fig. S3), indicating Sesn2 and Txnrd as main Prx2 repairing systems in polychromatic Hbbth3/+ mouse erythroblasts and all three, Srxn, Txnrd, and Sesn2, in Hbbth3/+mouse orthochromatic erythroblasts.
Our data suggest that the activation of Nrf2 may act as a back-up mechanism providing an interface between oxidative stress and erythropoiesis but in the absence of Prx2 such as in Prx2−/− or when upregulation of Prx2 expression is insufficient to face oxidative stress such as in Hbbth3/+ mice it makes a contribution to limit damage. To explore whether the absence of Prx2 might worsen in vivo Hbbth3/+ erythroid phenotype resulting in further activation of Nrf2, we generated a β-thalassemic mice knockout for Prx2 (Prx2−/−Hbbth3/+).
The absence of Prx2 worsens β-thalassemic hematologic phenotype, ineffective erythropoiesis, increased ROS levels, and apoptosis
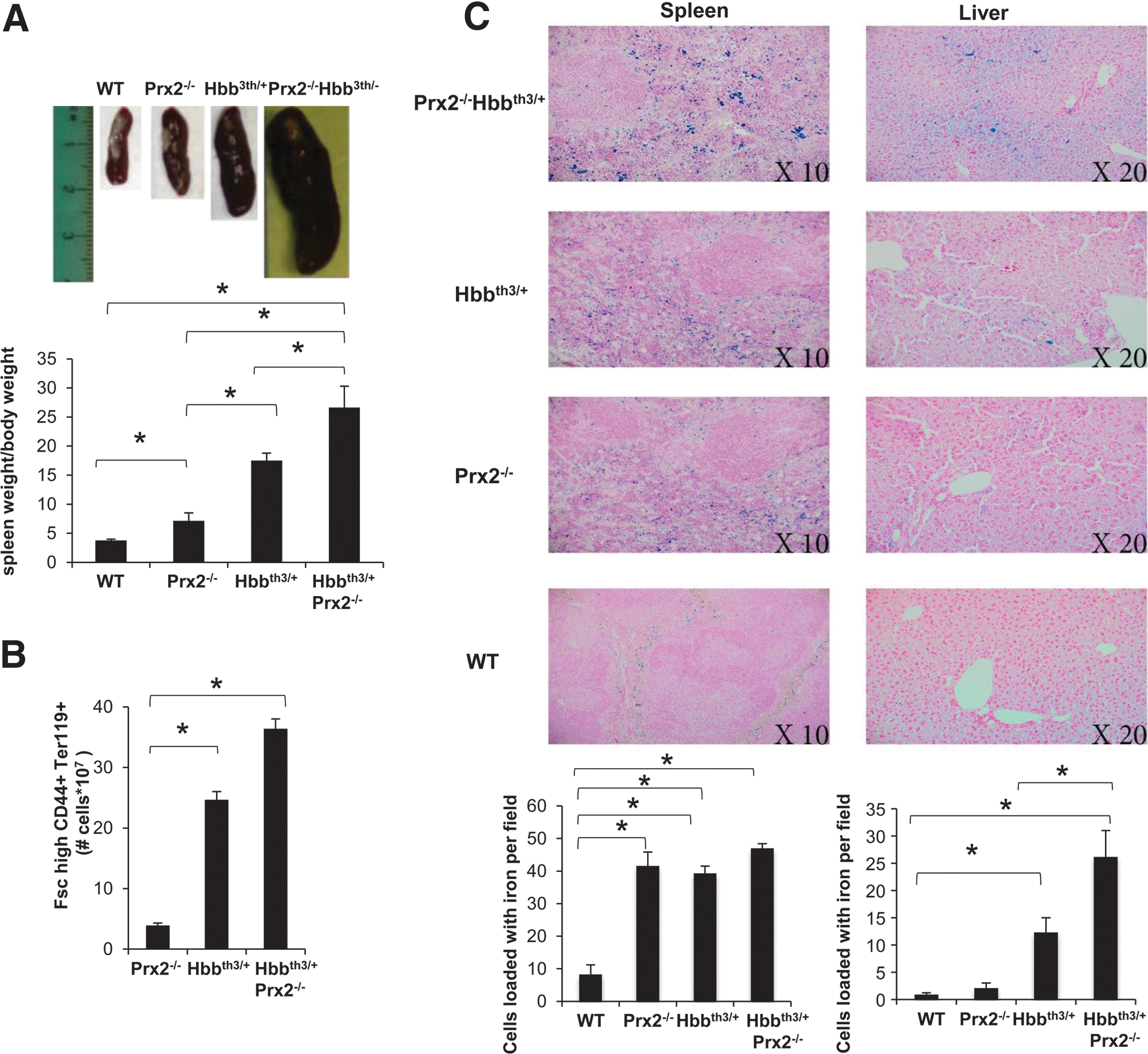
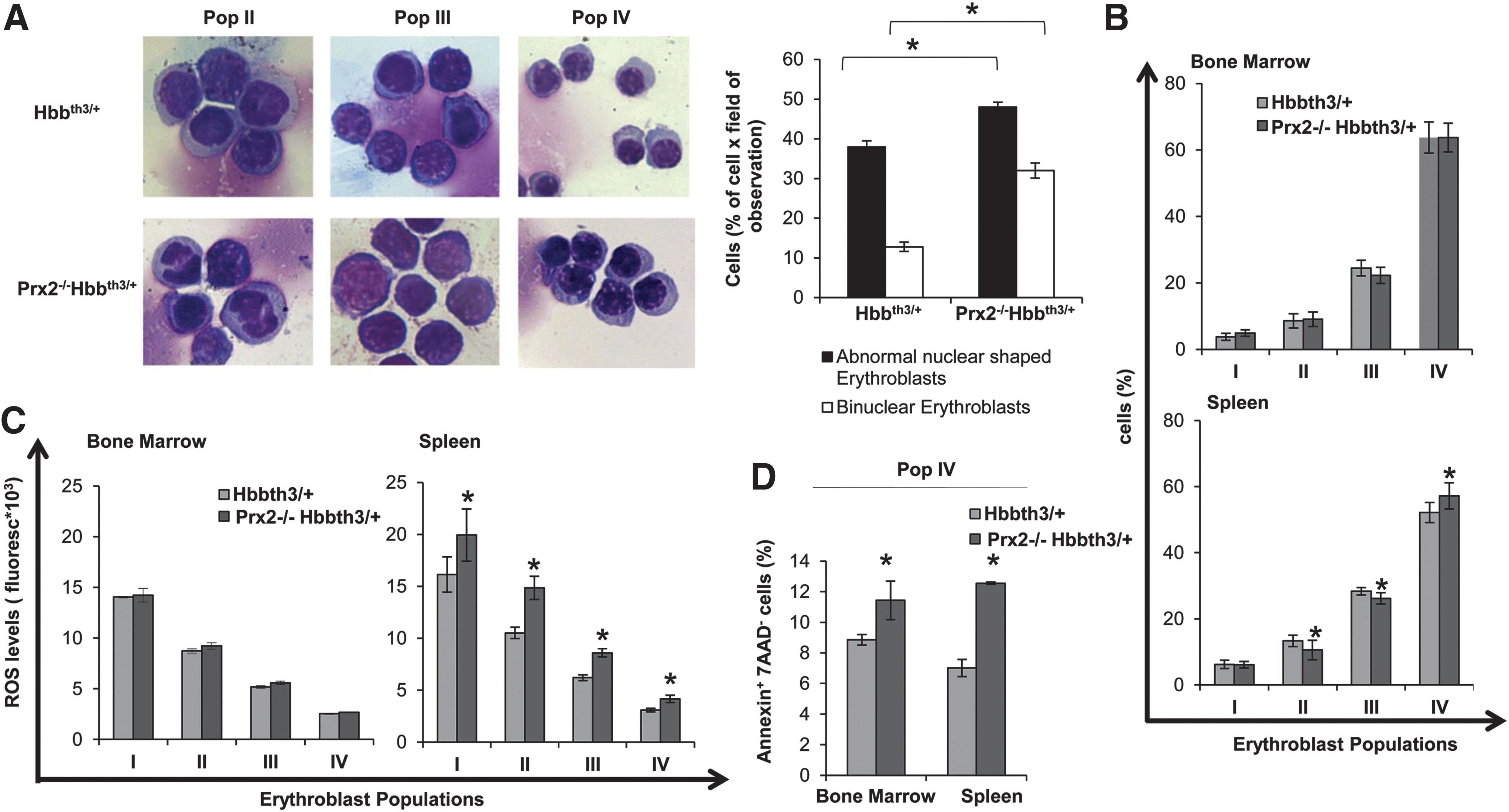
In Prx2−/−Hbbth3/+ mice, the absence of Prx2 worsened the hematological phenotype of β-thalassemic mice (Table 1; Figs. 3 and 4; and Supplementary Table S2). This is reflected by (i) the increased splenomegaly (Fig. 3A); (ii) increased amount of Fschigh CD44+ Ter119+ cells (Fig. 3B); and (iii) increased iron staining in the spleen of Prx2−/−Hbbth3/+ compared with either Prx2−/− or Hbbth3/+ mice (Fig. 3C). Prx2−/−Hbbth3/+ also showed increased iron levels in the liver, which was mainly sequestered in Kupffer cells without signs of liver fibrosis (Fig. 3C). This was also associated with more severe signs of dyserythropiesis in Prx2−/−Hbbth3/+ compared with Hbbth3/+ mice (Fig. 4A). Although the maturation profile of erythroid precursors from Prx2−/−Hbbth3/+ bone marrow was similar to that of Hbbth3/+ mice, we found a significant reduction in basophilic and polychromatic erythroblasts with an increase in orthochromatic erythroblasts from spleens of Prx2−/−Hbbth3/+ compared with Hbbth3/+ mice (Fig. 4A, B). These differences might be related to the two different compartments of erythropoiesis: the bone marrow and the spleen, the latter of which, we suggest, represents a highly pro-oxidant environment compared with the bone marrow. In fact, the ROS levels were similar in erythroid precursors from bone marrow of Prx2−/−Hbbth3/+ and Hbbth3/+ mice, but we found a significant increase of ROS production in all erythroid precursors from the spleen of Prx2−/−Hbbth3/+ compared with Hbbth3/+ (Fig. 4C). The amount of early apoptotic orthochromatic erythroblasts was markedly increased in both the bone marrow and spleen from Prx2−/−Hbbth3/+ compared with Hbbth3/+ mice (Fig. 4D).
p<0.05 compared with wild-type mice; °p<0.05 compared with b- thal3th/− mice.
Hb, hemoglobin; Hct, hematocrit; MCH, mean corpuscular hemoglobin; MCV, mean corpuscular volume; RDW, red cell distribution width; Retics, reticulocytes.
These findings indicate that the absence of Prx2 markedly affects β-thalassemic erythropoiesis by amplifying oxidative stress and ineffective erythropoiesis.
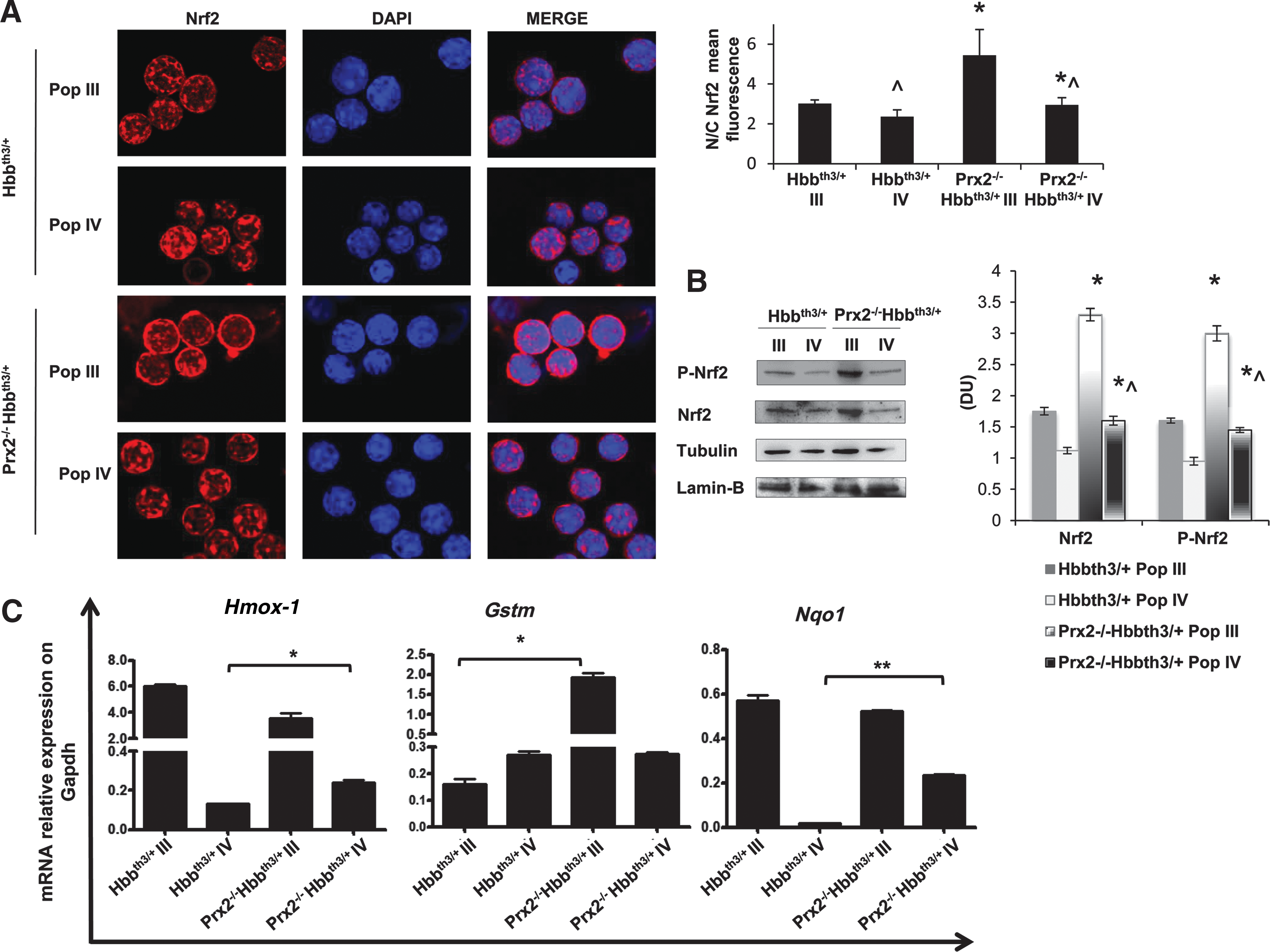
In Prx2−/−Hbbth3/ +, the absence of Prx2 is associated with activation of Nrf2 in orthochromatic erythroblasts
Since our data indicate the central role of the interplay between Prx2 and Nrf2 to limit oxidative stress in β-thalassemic erythropoiesis, we evaluated Nrf2 activation in polychromatic and orthochromatic erythroblasts from Prx2−/−Hbbth3/+ mice. As shown in Figure 5A, Nrf2 was similarly activated in polychromatic erythroblasts from Prx2−/−Hbbth3/+ and Hbbth3/+ mice, but it was significantly more activated in orthochromatic erythroblasts from Prx2−/−Hbbth3/+ mice compared with those from Hbbth3/+ mice. This finding implies a critical role for Nrf2 at late stages of terminal erythroid differentiation as an adaptive mechanism in response to amplified oxidative stress due to the absence of Prx2 on β-thalassemic erythropoiesis. The expression of a set of ARE responsive anti-oxidant genes such as HO-1 (Hmox1), glutathione S-transferase (Gstm), and nqo1 was upregulated in polychromatic from Prx2-/-Hbbth3/+ compared with Hbbth3/+ mice (Fig. 5B). However, Hmox-1 and Nqo1 were the only ARE responsive antioxidant genes upregulated in orthochromatic erythroblasts from Prx2-/-Hbbth3/+ compared with Hbbth3/+ mice.
Recombinant PEP1 Prx2 fusion protein ameliorates β-thalassemic erythropoiesis
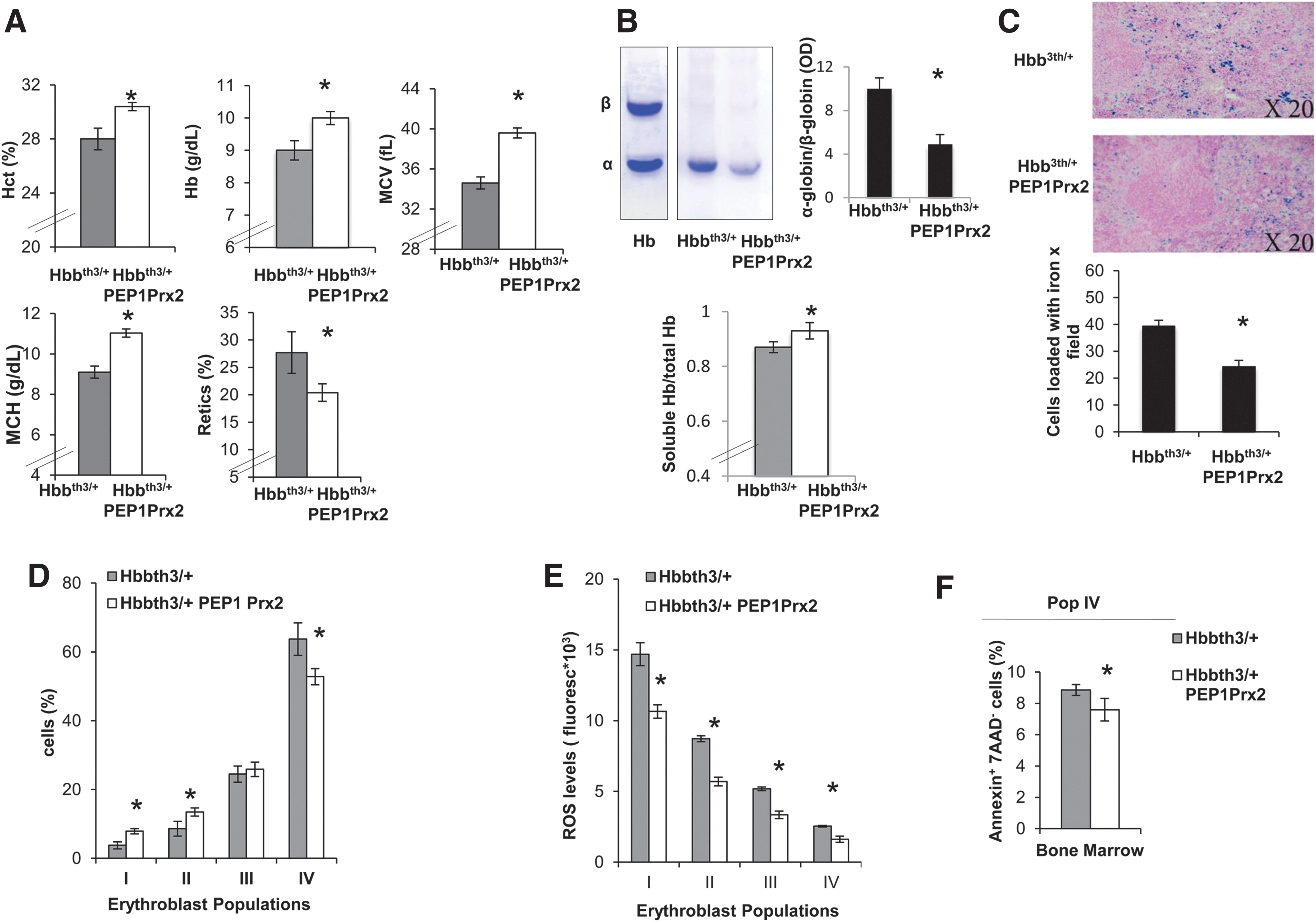
Finally, we tested the ability of recombinant PEP-1 Prx2 (PEP1 Prx2) fusion protein to ameliorate ROS levels and decrease ineffective erythropoiesis of Hbbth3/+ mice. PEPPrx2 has been previously shown to be able to penetrate cells and to efficiently counteract oxidative stress in two models of neurodegenerative diseases (5, 20). Thus, we carried out a preliminary study to determine the PEP1 Prx2 dosage required to impact stress erythropoiesis. We found that PEP1 Prx2, when administrated at 1 mg/kg/d ip for 4 weeks, significantly increased Hct, Hb levels and MCV and MCH and significantly reduced reticulocyte count in Hbbth3/+ mice (Fig. 6A). Hbbth3/+ PEP1 Prx2-treated mice showed a reduction in red cell membrane-associated α-globin precipitates, suggesting an amelioration of Hbbth3/+ erythropoiesis (Fig. 6B). This was also associated with a significant reduction in circulating erythroblasts compared with Hbbth3/+ vehicle-treated mice (Hbbth3/+ vs. Hbbth3/+ PEP1 Prx2 mice 38.1±6.4 vs. 8.0±3.3%). We observed a reduction in iron staining in the spleens from PEP1 Prx2 treated Hbbth3/+ mice compared with vehicle-treated mice (Fig. 6C). The profile of Hbbth3/+ erythroid precursors in the bone marrow showed a significant increase in basophilic erythroblasts (pop II) and a reduction in Hbbth3/+ orthochromatic erythroblasts (Fig. 6D). The ROS levels were significantly decreased in all erythroid populations from both bone marrow and spleens of PEP1 Prx2-treated Hbbth3/+ mice compared with vehicle-treated mice (Fig. 6E). This finding was also paralleled by a significant reduction in apoptosis of Hbbth3/+ orthochromatic erythroblasts from bone marrow of PEP1 Prx2-treated Hbbth3/+ mice (Fig. 6F). It is of interest to note that PEP1 Prx2 treatment reduced Nrf2 activation in Hbbth3/+ orthochromatic erythroblasts compared with vehicle-treated Hbbth3/+ mice (Supplementary Fig. S4A). Similar results were also observed in PEP1 Prx2-treated Prx2-/- mice (Supplementary Fig. S4B, C). No changes in hematological parameters, reticulocyte count (data not shown), and erythropoiesis were evident in PEP Prx2-treated WT mice compared with vehicle-treated WT (Supplementary Fig. S4D).
Discussion
In this study, we documented a novel interplay between Prx2 and Nrf2 to limit oxidative stress in β-thalassemic erythropoiesis. The abnormalities in erythropoiesis of Prx2−/− mice suggest that Prx2 is required to interface between the physiological oxidative stress and maturation events involved in erythropoiesis (Fig. 1). This is also supported by the amelioration of Prx2−/− mouse erythropoiesis in response to NAC antioxidant treatment (Supplementary Fig. S1B). β-Thalassemia is a paradigmatic example of stress erythropoiesis characterized by severe oxidative stress with upregulation of Prx2 expression (13, 33, 43). The similarities between Prx2−/− and β-thalassemic erythropoiesis further support the key role of Prx2 in stress erythropoiesis.
At the crossroad of the redox response is the transcriptional factor Nrf2 (18), which might co-operate with endogenous antioxidant system(s) such as Prx2 against severe oxidative stress during erythropoiesis. Indeed, Prx2−/− and Hbbth3/+ erythroblasts showed increased activation of Nrf2 compared with WT cells. The transient activation of Nrf2 in WT erythroblasts in response to exogenous oxidative stress such as PHZ indicates that Nrf2 is generally inactive under physiological condition and can be transiently activated in response to acute severe oxidative damage. On the other hand, in β-thalassemia or in Prx2−/− mice characterized by chronic oxidative stress, Nrf2 is constitutively activated. This working model for Nrf2 is also supported by previous reports on Nrf2−/− mice, which show only mild hematological phenotype only in aged mice with reduced antioxidant protection (38). In Px2−/− and Hbbth3/+ mouse erythroblasts, the activation of Nrf2 was associated with upregulation of ARE-response genes such as HO-1 (Fig. 2C). The differences in HO-1 gene expression between Px2−/−and Hbbth3/+ orthochromatic erythroblasts might be related to the relative heme deficiency previously reported by us and others in β-thalassemic erythroid precursors in the late stages of erythropoiesis (6, 12).
The absence of Prx2 worsened the β-thalassemic hematologic phenotype in Prx2−/−Hbbth3/+mice, indeed supporting the key role of Prx2 in stress erythropoiesis. This was also associated with further activation of Nrf2 in Prx2−/−Hbbth3/+mice compared with Hbbth3/+ mice and the upregulation of various ARE-related genes such as Gstm, Hmox-1, and nqo1 (Fig. 5C). We were not surprised to find differences in timing of upregulation of Nrf2-related ARE-genes, since it has been shown that the induction and modulation of the battery of ARE-genes is more efficient in cellular protection against toxic events than the induction of only a few of them (10). These findings suggest that Prx2−/−Hbbth3/+ mice require a resetting of the balance between oxidative stress and antioxidant systems in favor of antioxidant systems to sustain β-thalassemic erythropoiesis. As a proof of concept of the role of Prx2 in pathological erythropoiesis, we administrated fused recombinant PEP1-Prx2 (PEP1Prx2) to Hbbth3/+ mice. In PEP1Prx2-treated Hbbth3/+ mice, we observed decreased ineffective erythropoiesis in β-thalassemic mice, indicating that a potentiation of endogenous antioxidant systems may limit the oxidation damage and ensure cell survival during β-thalassemic erythropoiesis.
Thus, we propose the interplay between Prx2, a key anti-oxidant system, and Nrf2 as an oxidative response transcriptional factor, in cellular adaptation to oxidative stress, resulting in upregulation of antioxidant (ARE)-genes required to ensure cell survival. It is of interest to note that a similar interplay between Nrf2 and seleno-proteins has been previously reported to impact ROS production in erythrocytes from seleno-protein-deficient mice (24).
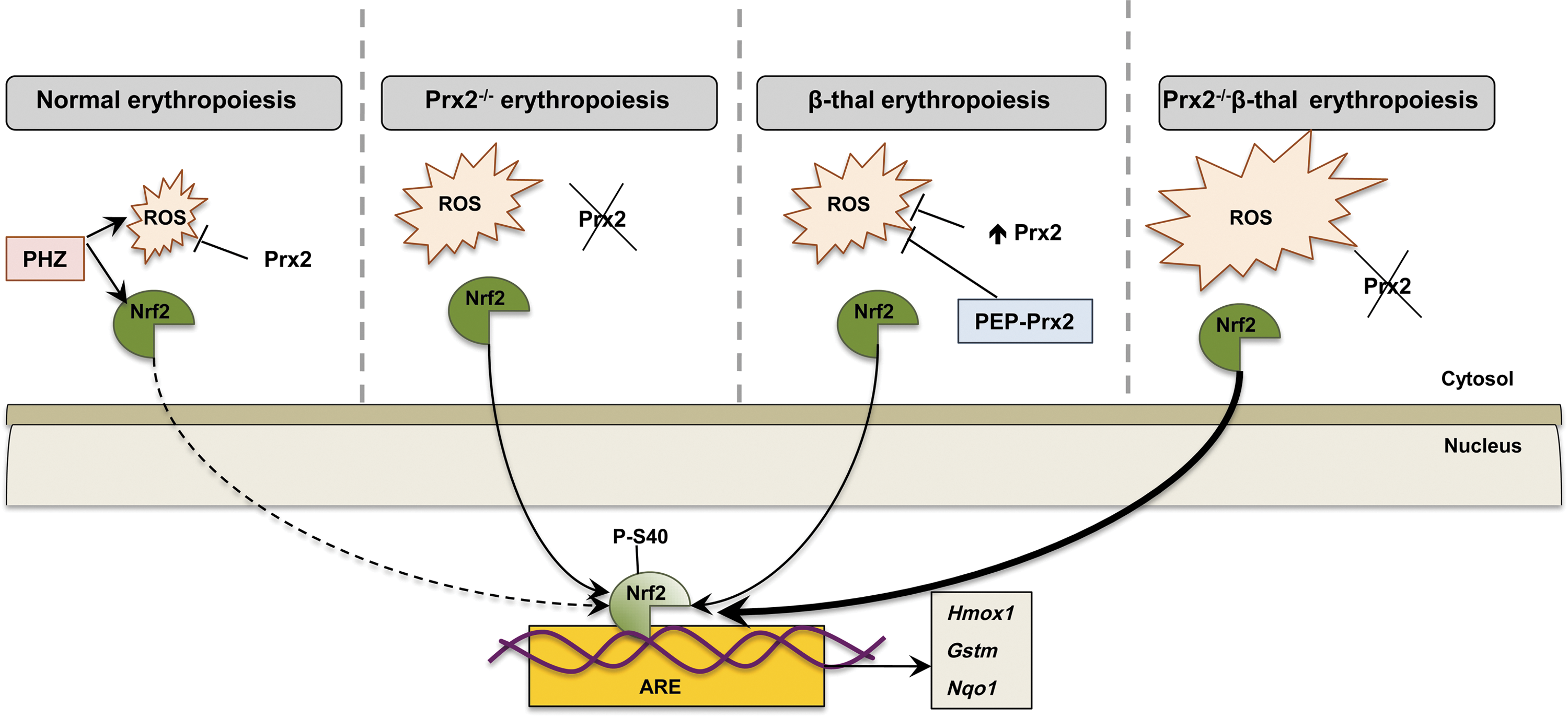
Our working model of how Prx2 and Nrf2 may co-operate in stress erythropoiesis is shown in Figure 7. In WT mice, Prx2 controls the intrinsic oxidative stress present during cellular maturation events; but in the presence of acute exogenous oxidative stress (PHZ), Prx2 interplays with Nrf2 to contain the toxic effects of high ROS levels. The absence of Prx2 promotes ineffective erythropoiesis and activation of Nrf2, which is similarly induced in β-thalassemic erythroid precursors. When Prx2 is knocked out in Hbbth3/+ mice, in the face of increased ineffective erythropoiesis, the ROS levels and the activation of Nrf2 are further increased compared with Hbbth3/+ mice. Treatment with PEP1-Prx2 markedly reduced ROS levels and cell apoptosis in Hbbth3/+ mice with an amelioration of β-thalassemic ineffective erythropoiesis and reduction in Nrf2 activation. Our data shed new light on adaptive mechanisms against oxidative stress mediated through Prx2 and Nrf2 during erythropoiesis to minimize oxidative damage and also suggest new possible therapeutic options in modulation of endogenous antioxidant systems to downregulate pathological erythropoiesis.
Materials and Methods
Drugs and chemicals
NaCl, Na3VO4, phosphate-buffered saline (PBS) phosphate buffer, bicine, benzamidine, β-mercaptoethanol, bromophenol blue, sodium dodecyl sulphate, NaF, ethylenediaminetetraaceticacid, May-Grunwald stain, Giemsa stain, PHZ hydrochloride, albumin from bovine serum (bovine serum albumin [BSA]), and glycerol were obtained from Sigma/Aldrich (St. Louis, MO); protease inhibitor cocktail tablets were from Roche (Basel, Switzerland); dithiotreithol was from Fluka (Buchs, Switzerland); Triton X-100 was from GE Healthcare Life Sciences (Little Chalfont, United Kingdom); 40% Acrylamide/Bis Solution, 37.5:1 was from BIO-RAD (Hercules, CA); Dulbecco's Phosphate-Buffered Saline was from Lonza (Verviers, Belgium); and Luminata Forte and Luminata Classico Hrp solutions were from Merck Millipore (Armstadt, Germany).
Mouse strains and design of the study
C57B6/2J mice as WT controls, Prx2−/− mice and Hbbth3/+, as a mouse model of β-thalassemia intermedia were used in this study (13, 15, 16, 35). Hbbth3/+ and Prx2−/− had the same C57BL6J background as the WT mice and were backcrossed for 17 generations to generate Prx2−/−Hbbth3/+ mice. The genotype of Prx2−/−Hbbth3/+ mice was validated at each generation. The Institutional Animal Experimental Committee, University of Verona (CIRSAL) and the Italian Ministry of Health approved the experimental protocols. Based on our preliminary data and previous reports, we used 3–4 month-old female mice from the four mouse strains (16).
Where indicated, Prx2−/− mice were treated with NAC at the dosage of 100 mg/kg/day (in NaCl 0.9%, NaOH 36 mM, pH 9.4; ip) or vehicle only for 3 weeks (9a, 31). Hbbth3/+ and Prx2−/− mice were treated with PEP1Prx2 (in PBS) at the dosage of 1.25 mg/kg/day ip or vehicle for 4 weeks (5, 20).
Hematological parameters, red cell indices, and reticulocyte count were determined as previously reported (4, 8, 9, 13). Blood was collected with retro-orbital venipuncture in anesthetized mice using heparined microcapillary tubes. Hematological parameters were evaluated on a Bayer Technicon Analyser ADVIA. Hct and Hb were manually determined (8, 13).
Determination of α-globin and β-globin ration was carried out as previously reported by Dussiot et al. (11).
Flow cytometric analysis of mouse erythroid precursors and cell sorting of murine bone marrow erythroblasts
Flow cytometric analysis of erythroid precursors from the bone marrow and spleen of mice from the four strains was carried out as previously described using the CD44-TER-119 strategy (28). Population II, III, and IV corresponding to basophilic erythroblasts, polychromatic erythroblasts, and orthochromatic erythroblasts were sorted from bone marrow of mice from the four strains as previously reported (28). Sorted cells were used for (i) morphological analysis of erythroid precursors on cytospin preparations stained with May Grunwald-Giemsa; (ii) immunofluorescence assay for Nrf2; (iii) immune-blot analysis for Nrf2 and P-Nrf2; and (iv) real time-polymerase chain reaction analysis (13, 28). Details on primers used are reported in Supplementary Table S1. In immunofluorescence assay, Nrf2 quantification was performed using the ImageJ software (
For immunoblot-analysis, the following specific antibodies were used: anti Nrf2-phospho-S40, anti Nrf2 (Abcam, Cambridge, United Kingdom), anti glyceraldehyde 3-phosphate dehydrogenase (Sigma/Aldrich), anti beta-Tubulin (clone E7; DSHB, Iowa, IA), and anti Lamin B (Santa Cruz Biotechnology, Santa Cruz, CA), used as loading controls. Images were acquired using Image Quant Las Mini 4000 Digital Imaging System (GE Healthcare Life Sciences), and densitometric analysis was performed using the ImageQuant TL software (GE Healthcare Life Sciences) (4, 13, 23). Analysis of apoptotic orthochromatic erythroblasts was carried out on the CD44-Ter-119 gated cells using the Annexin-V PE Apoptosis detection kit (eBioscience, San Diego, CA) following the manufacturer's instructions (15). Flow cytometric analysis was carried out with the FACSCanto™ flow cytometer (Becton Dickinson, San Jose, CA).
Analysis of ROS levels on different populations of erythroid precursors
The ROS levels from erythroid precursors were determined using the General Oxidative Stress Indicator CM-H2DCFDA (LifeTechnologies, Carlsbad, CA) on erythroid precursors as previously described, (31) with minor changes. Briefly, CD44-PE (Miltenyi Biotec, Bergisch Gladbach, Germany), TER-119 APC, CD45 APC-eFluor 780, GR1 APC-Cy7, and CD11b APC-Cy7 stained cells (28) from mouse bone marrow or spleen were incubated with CM-H2DCFDA (10 μM in PBS, BSA 1% at 37°C for 20 min). Cells were then washed once with PBS BSA 1%, stained in ice with 7-AAD, and analyzed by an FACS Canto II flow cytometer (Becton Dickinson).
Generation of recombinant-PEP-1 Prx2 fusion protein
The fusion protein PEP1Prx2 was generated as previously reported (5, 20). PEP1Prx2 was intraperitonealy administered once a day at the dosage of 1.25 mg/kg for 4 weeks based on previous reports (5, 20).
Histological analysis of spleen and liver
Spleen and liver were, immediately after mice dissection, formalin fixed and paraffin embedded. For each paraffin block, after 3 μm-thick sections were cut, they were stained with hematoxylin eosin, Masson's trichrome, and May-Grunwald-Giemsa. Tissue iron was stained using Perls prussian blue stain after treatment with diluted hydrochloric acid to prevent ferric ions from binding proteins. The analysis of iron staining was performed on four different fields at magnification×200 by two pathologists (A.J., C.L.) blinded to the experimental groups. Results were expressed as the mean number of cells loaded with iron on Perls' stain.
Statistical analysis
Data were analyzed using either t-test or the two-way analysis of variance for repeated measures between the mice of various genotypes. A difference with a p value<0.05 was considered significant.
Footnotes
Acknowledgments
This work was supported by PRIN (L.D.F. and A.I.: 201228PNX83) and FUR_UNIVR (L.D.F.).
Authorship and Contributions
A.M., L.D.F., N.M., and A.I. designed the experiments, analyzed data, and wrote this article; X.A. analyzed the data and wrote this article; A.J., C.L. carried out the histological analysis; A.M., A.S., and L.D.F. carried out the experiments; L.D.F. performed the molecular experiments and analyzed the data; and S.Y.C. and D.W.K. generated the PEPPrx2.
Author Disclosure Statement
No competing financial interests exist.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
