Abstract
Introduction
I
Paradoxically, pathological iron-mediated oxidative reactions may disrupt critical physiological signaling pathways and damage normal cells, especially during ischemia (reduced blood supply due to vascular obstruction that impairs tissue oxygenation) and reperfusion (restoration of blood flow to ischemic tissues) (1, 2, 34, 36). By selectively inhibiting these processes, appropriate iron chelators could potentially prevent abnormal cell proliferation in cancer or other settings of abnormal cell growth and reduce oxidative cell injury in ischemia and reperfusion.
In this review, we discuss novel iron chelators called “exochelins” that are unique and highly potent siderophores of Mycobacterium tuberculosis and other pathogenic mycobacteria (26 –28, 56, 62). Exochelins are lipid and water soluble and can be utilized as probes to decipher the role of iron in normal and pathological states. In addition, their ability to chelate intracellular iron with little or no toxicity renders them potentially useful for the treatment of certain iron-dependent diseases.
The Exochelins of M. tuberculosis and Other Pathogenic Mycobacteria
The pathogenic mycobacteria, which include M. tuberculosis, the primary agent of tuberculosis in humans; Mycobacterium bovis, the primary agent of tuberculosis in cattle and other domesticated animals; and Mycobacterim avium, one of the most prominent opportunistic pathogens in patients with AIDS, are unusual in having two types of siderophores: the water-insoluble cell wall-associated mycobactins and the secreted water- and lipid-soluble exochelins. Structural analysis has revealed that exochelins and mycobactins share a common core structure and differ only in the composition of a single side chain (27, 28, 39, 56, 62) (Fig. 1). In contrast to the mycobactins, where the R1 side chain exists as a saturated or unsaturated alkyl group, the side chain of the exochelins exists as a saturated or unsaturated alkyl methyl ester or carboxylate (27, 28, 62). Moreover, the side chains of the exochelins are much shorter than the side chains of the mycobactins. As a result of these two differences, the exochelins are more polar than the mycobactins, and hence water soluble; whereas the highly nonpolar mycobactins are water insoluble and confined to the lipid-rich cell wall of the mycobacteria.
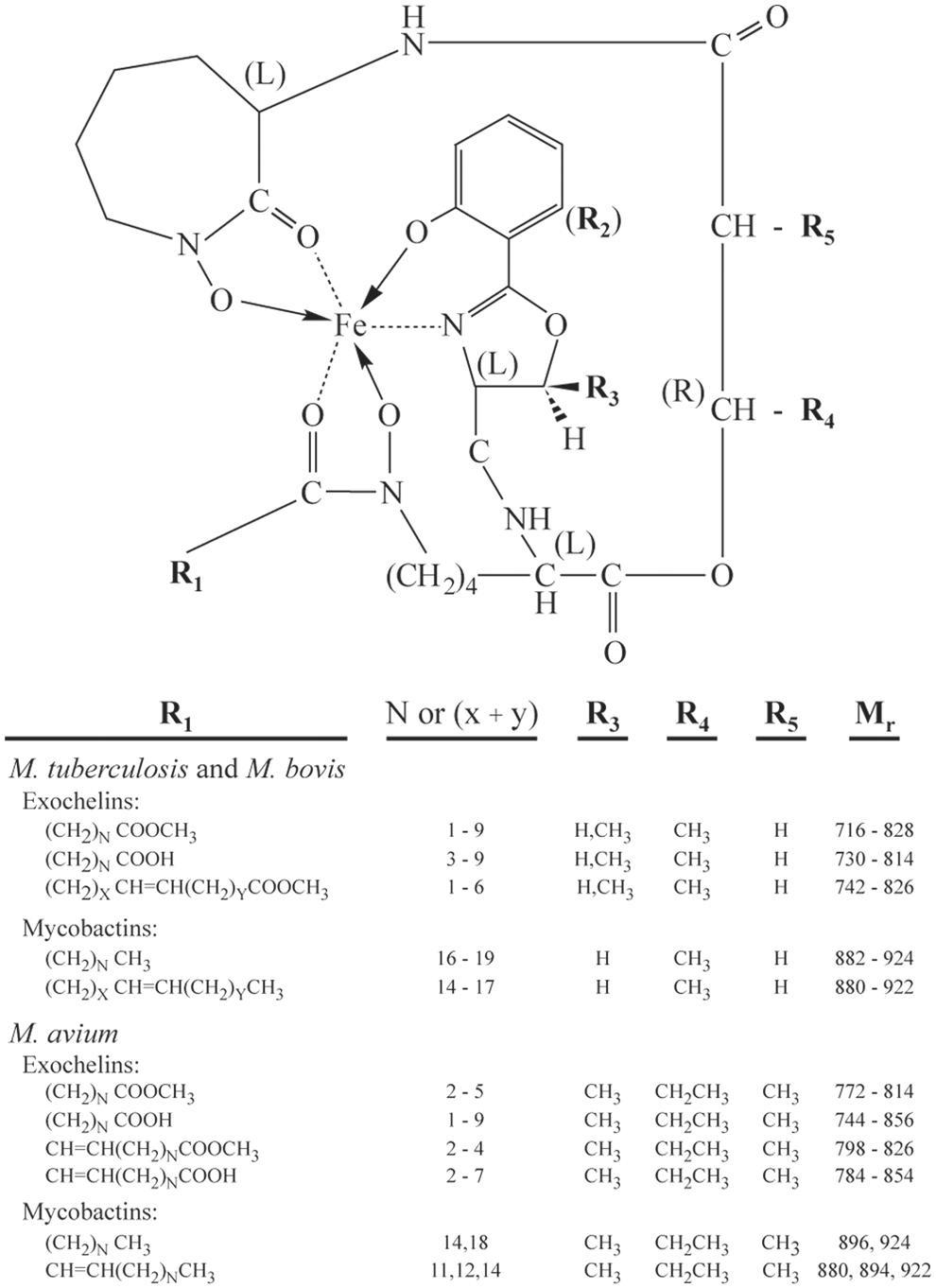
In view of the similarity of their core structure to mycobactins, the exochelins of pathogenic mycobacteria might reasonably be termed “exomycobactins.” The term “carboxymycobactins” also has been used to describe these exochelins based on an earlier study that identified only the carboxylic acid forms at R1 (39), but this term is somewhat misleading, because it excludes the most abundant species of exochelins in which the R1 side chain terminates in a methyl ester rather than in a carboxylic acid moiety (27, 33, 62). In addition, it is not certain that the carboxylic acid forms exist in vivo. Exochelins terminating in a methyl ester convert to carboxylic acids on prolonged storage or brief treatment with esterases (62) (M.A. Horwitz, unpublished studies). Whether the carboxylic acid forms actually exist in M. tuberculosis infection or are a phenomenon of in vitro culture in artificial medium or purification from such cultures is not known.
M. tuberculosis and M. bovis produce essentially the same set of saturated and unsaturated exochelins, except for a few species thus far unique to M. bovis (27, 28) (Fig. 1). M. avium exochelins show the same variation at R1 as M. tuberculosis and M. bovis exochelins; however, R3 is always CH3 in M. avium exochelins, whereas this side chain can be either H or CH3 in the exochelins of M. tuberculosis and M. bovis (4, 62). Moreover, the M. avium exochelins differ at R4, which is a CH2CH3 group instead of a CH3 group, and at R5, which is a CH3 group instead of an H.
The polarity of exochelins is reflected by their elution pattern from a reverse-phase high-performance liquid chromatography (HPLC) column (Fig. 2), with the relatively polar exochelins eluting early and the relatively nonpolar exochelins eluting late in an acetonitrile gradient (27, 28, 62). All else being equal, nonpolarity increases with an increasing length of the R1 side chain, retention of the methyl ester group, or presence of a threonine (CH3) versus a serine (H) at R3. The high affinity of exochelins for iron is independent of their polarity, as in mixing experiments more polar ferric exochelins readily donate iron and reach equivalence in iron saturation with less polar desferri-exochelins (exochelins absent an iron molecule) and vice versa (26).
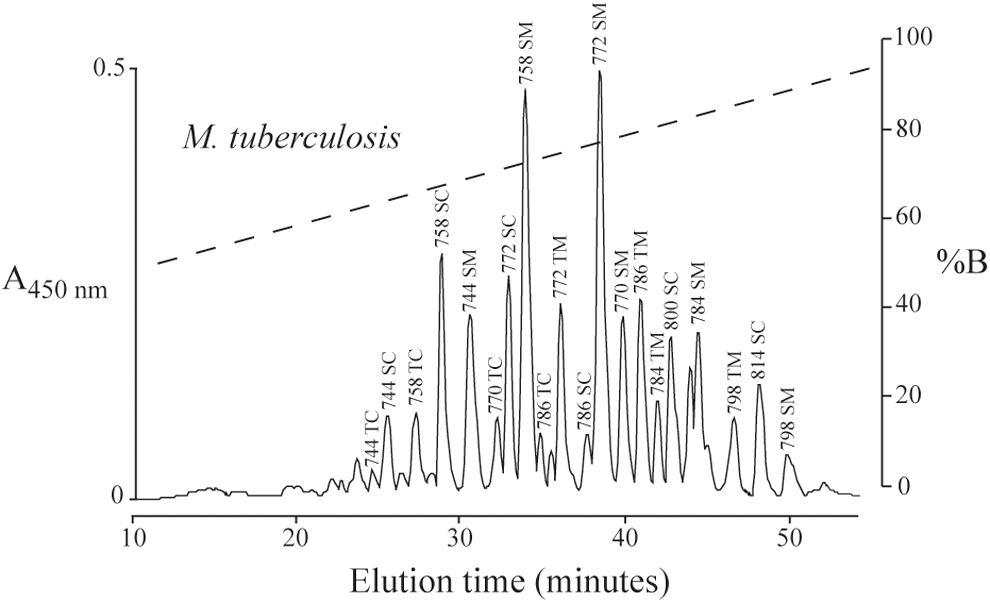
The exochelins of M. tuberculosis are named according to their mass in Daltons in the iron-loaded state; whether R3 is H (derived from Serine; S) or CH3 (derived from Threonine; T) moiety; and whether R1 terminates in a methyl ester (M) or carboxylate (C) moiety (Fig. 2) (27). Thus, for example, Exochelin 772SM, one of the most abundant exochelins produced by M. tuberculosis and an exochelin that has been synthesized and extensively studied (see sections on physiologic and preclinical studies with exochelins below), has a mass of 772 in the iron-loaded form, an H (derived from Serine) at R3, and a Methyl ester at the terminus of the R1 group, with R1=(CH2)5COOH3; as with all M. tuberculosis exochelin species, R4=CH3 and R5=H (27). The saturated (Ferri-) and unsaturated (Desferri-) exochelins each form a 14 Da incremental series. Between these series, exochelins of the same form and side chain length differ from each other by 2 Da (27).
The exochelins have four to six asymmetric carbons; four are common to all exochelins and are denoted in Figure 1 by the “(L)” or “(R),” and two additional ones are dependent on the composition of side groups R3 and R5 (56). Exochelin 772SM, synthesized with the (R) form at R4, is a highly active iron chelator; whereas the identical Exochelin 772SM with the (S) form at R4 is inactive (M.A. Horwitz, unpublished studies).
The exochelins and mycobactins are synthesized at the mbtA-J locus of the M. tuberculosis genome (15, 41, 48). Several of these genes (mbtA, mbtB, and mbtI) contain IdeR boxes for transcriptional regulation by IdeR-Fe2+, a dual-functional regulator of genes involved in iron acquisition and storage (29). Synthesis of exochelins and mycobactins by mycobacteria is increased under iron-deficient conditions and decreased under iron-replete conditions.
Physiological Role of Exochelins in Pathogenic Mycobacteria
Desferri-exochelins are synthesized in the bacterial cytoplasm and then exported across the cytoplasmic membrane by the membrane-associated proteins MmpS4 and MmpS5 and their cognate inner membrane transporter proteins MmpL4 and MmpL5 (Fig. 3) (61). After binding iron in the extracellular milieu of the mycobacterium, the ferric-exochelins transfer iron into the bacterial cell for utilization via one of several routes (Fig. 3). First, the ferri-exochelins can transfer the iron to desferri-mycobactins that are anchored in the cell wall (26). The iron is subsequently internalized, most likely after it is reduced by a ferric reductase, which lowers its affinity for mycobactin (8, 50). However, mycobactins are not required for iron utilization from exochelins, because an M. tuberculosis mbtB − mutant strain completely lacking mycobactins and exochelins can, nevertheless, utilize iron from exochelins (M.V. Tullius and M.A. Horwitz, unpublished data).
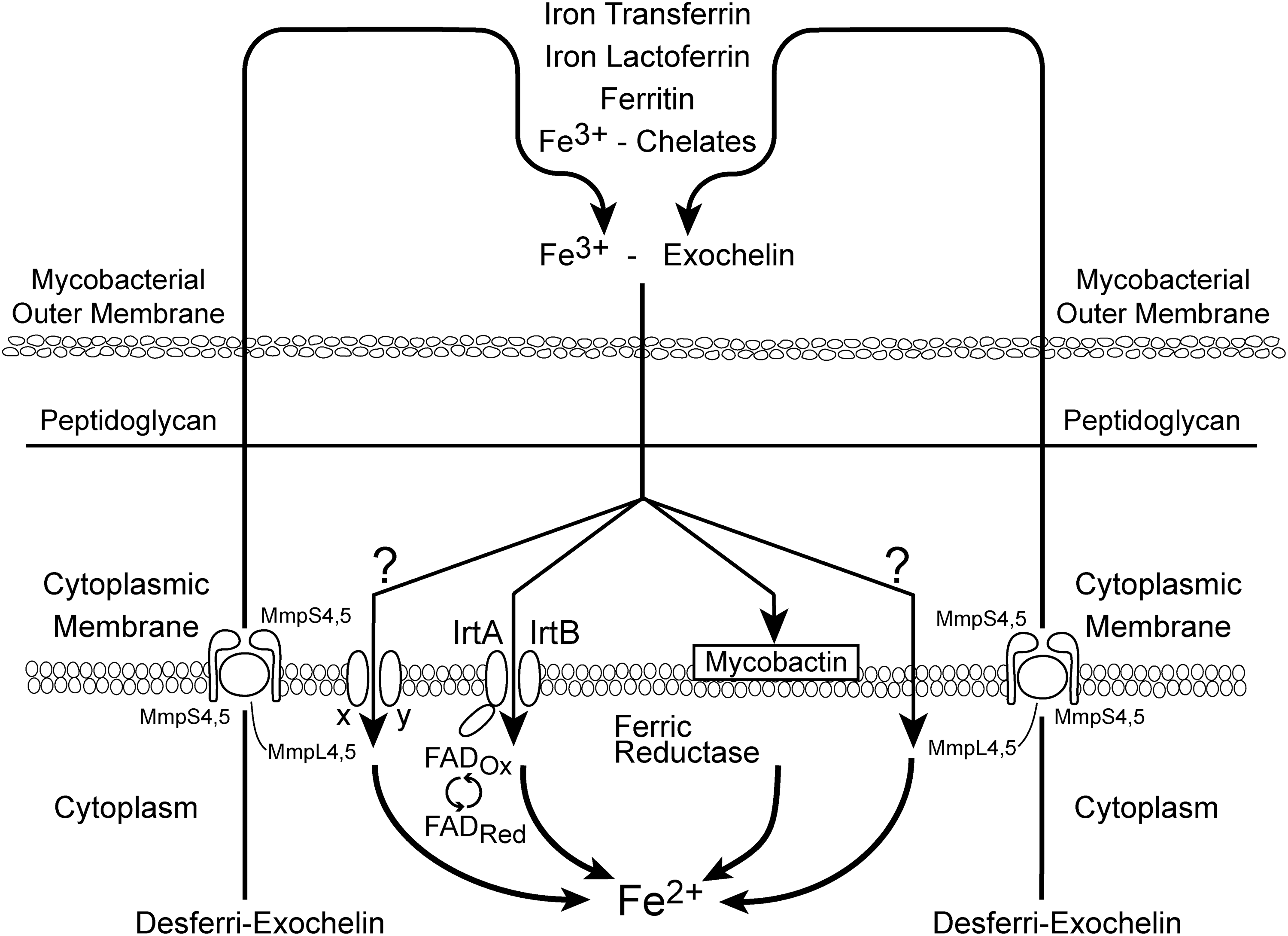
A second pathway for uptake of iron into the bacterial cell involves the ABC Transporter, IrtAB (51). IrtA contains an FAD-binding domain at its cytoplasmic N terminus that is necessary for its function (53) (Fig. 3). Inactivation of the irtAB system reduces, but does not completely eliminate, the growth of M. tuberculosis under iron-deficient conditions, suggesting that other transporters may be involved. In this regard, one potential candidate is the iron deficiency-induced protein Irp10 (Rv3269) and the metal-transporting ATPase Mta72 (Rv3270): Both are encoded by tandem genes and bear similarity to two-component metal transporting systems (11). Whether this system actually transports iron has not been determined. Finally, the lipid- and water-soluble ferri-exochelins may be capable of traversing back across the cell membrane and into the cytoplasm without the aid of transporters; however, this has not been established.
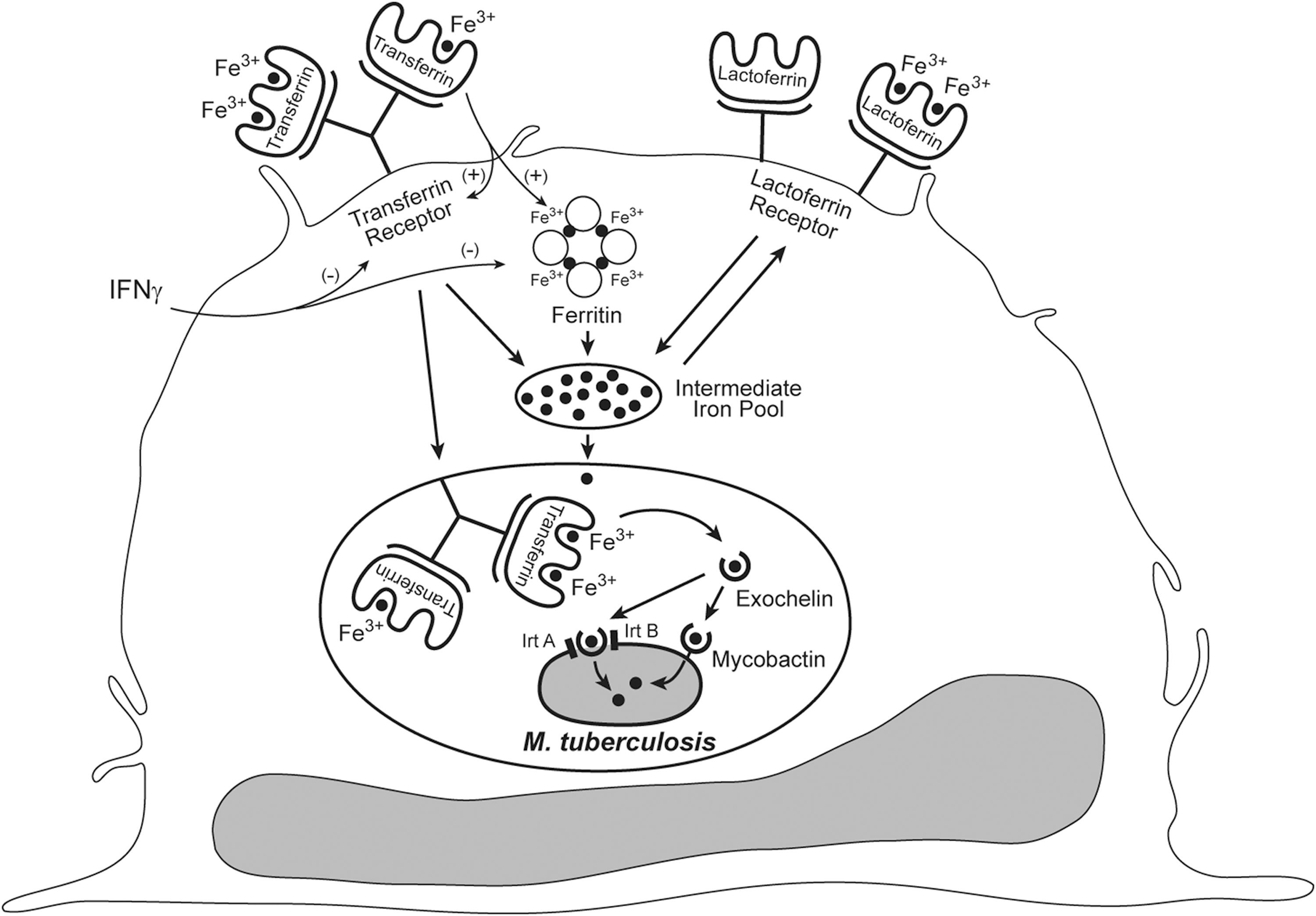
In the human host, M. tuberculosis multiplies both intracellularly in mononuclear phagocytes, especially lung macrophages, and extracellularly within lung cavities. At extracellular sites of M. tuberculosis infection, the organism likely encounters transferrin and lactoferrin, which play key roles in sequestering iron in tissues. Ferritin, which is found at low levels in the blood, may also be present at extracellular sites. One potential source is bleeding, which may occur in lung cavities, a pathological consequence of M. tuberculosis infection. In vitro, desferri-exochelins rapidly (minutes) remove iron from iron-transferrin (both 95% and 40% iron saturated, with the latter being the saturation level in serum) and iron-lactoferrin, and more slowly (hours) from ferritin (26). Ferric exochelins, but not iron transferrin, can donate iron to desferri-mycobactins in the mycobacterial cell wall, underscoring the importance of exochelins for iron acquisition (26). Thus, in vitro, the water-soluble exochelins, but not the cell wall-associated water-insoluble mycobactins, are capable of accessing iron bound to the transferrin molecule.
In mononuclear phagocytes (Fig. 4), M. tuberculosis resides in a mildly acidified phagosome (16) that displays arrested maturation (13); that is, the phagosome fuses with early and late endosomes but does not readily fuse with lysosomes. At this site in the host, the organism encounters several sources of iron. One source is iron-transferrin, which is targeted directly to the phagosome by transferrin receptors (14). Indeed, M. tuberculosis within macrophages has been demonstrated to acquire radiolabeled iron from exogenously delivered iron transferrin (42). Desferri-exochelins may acquire iron directly from the iron-transferrin molecule, from the pH-dependent release of an iron atom from the transferrin molecule within the mildly acidified (∼pH 6) phagosome, or from the intermediate iron pool of the cell, which is replenished with iron from iron lactoferrin and ferritin, among other iron sources. In this regard, M. tuberculosis within macrophages has been demonstrated to acquire radiolabeled iron from exogenously administered iron-loaded lactoferrin and iron chelates (42). The importance of siderophores in the acquisition of iron intracellularly in macrophages has been underscored by the finding that siderophore-deficient strains of M. tuberculosis show retarded growth in macrophages in vitro (18, 58). However, since M. tuberculosis contains an alternative heme iron acquisition system that is independent of the siderophore-mediated iron acquisition system, siderophore-deficient M. tuberculosis can acquire iron and grow normally within macrophages cultured in the presence of exogenously administered hemin or hemoglobin (58).
Isolation of Exochelins and Chemical Synthesis
The exochelins were initially isolated from cultures of M. tuberculosis by saturating the culture supernatant with iron, extracting the ferri-exochelins into chloroform, and purifying by reverse-phase HPLC on a C18 column with an acetonitrile gradient (Fig. 2); individual exochelins were subsequently purified on an alkyl phenyl column (27). Ferriexochelins were converted to their desferri forms by incubation in 50 mM EDTA, pH 4, and re-extraction into chloroform; this removed 60%–90% of the iron without altering their structure, as confirmed by mass spectrometry (26).
Subsequently, two members of the exochelin family were chemically synthesized by a multistep process—desferri-Exochelin 772SM and desferri-Exochelin 786SM (22 –24). Preclinical studies were performed primarily with desferri-Exochelin 772SM, the most abundant exochelin species secreted by M. tuberculosis into broth culture (27, 28) and in the mid-range of exochelins in terms of polarity (Fig. 2). Synthesized desferri-Exochelin 772SM was shown to have identical biological activity to the natural product (M.A. Horwitz, unpublished studies).
Chelating Properties of Exochelins Versus Other Clinically Available Iron Chelators
Desferri-exochelins strongly bind trivalent cations (Fe3+, Ga3+, and Al3+), but only weakly bind divalent cations (Ca2+, Mg2+, and Mn2+), and they do not appreciably bind monovalent cations (Na+ and K+) (M.A. Horwitz, unpublished data). Dissociation of iron bound to exochelins occurs only at a pH below 1.0 (M.A. Horwitz, unpublished data). Desferri-exochelins do not remove iron from hemoglobin or iron-containing enzymes.
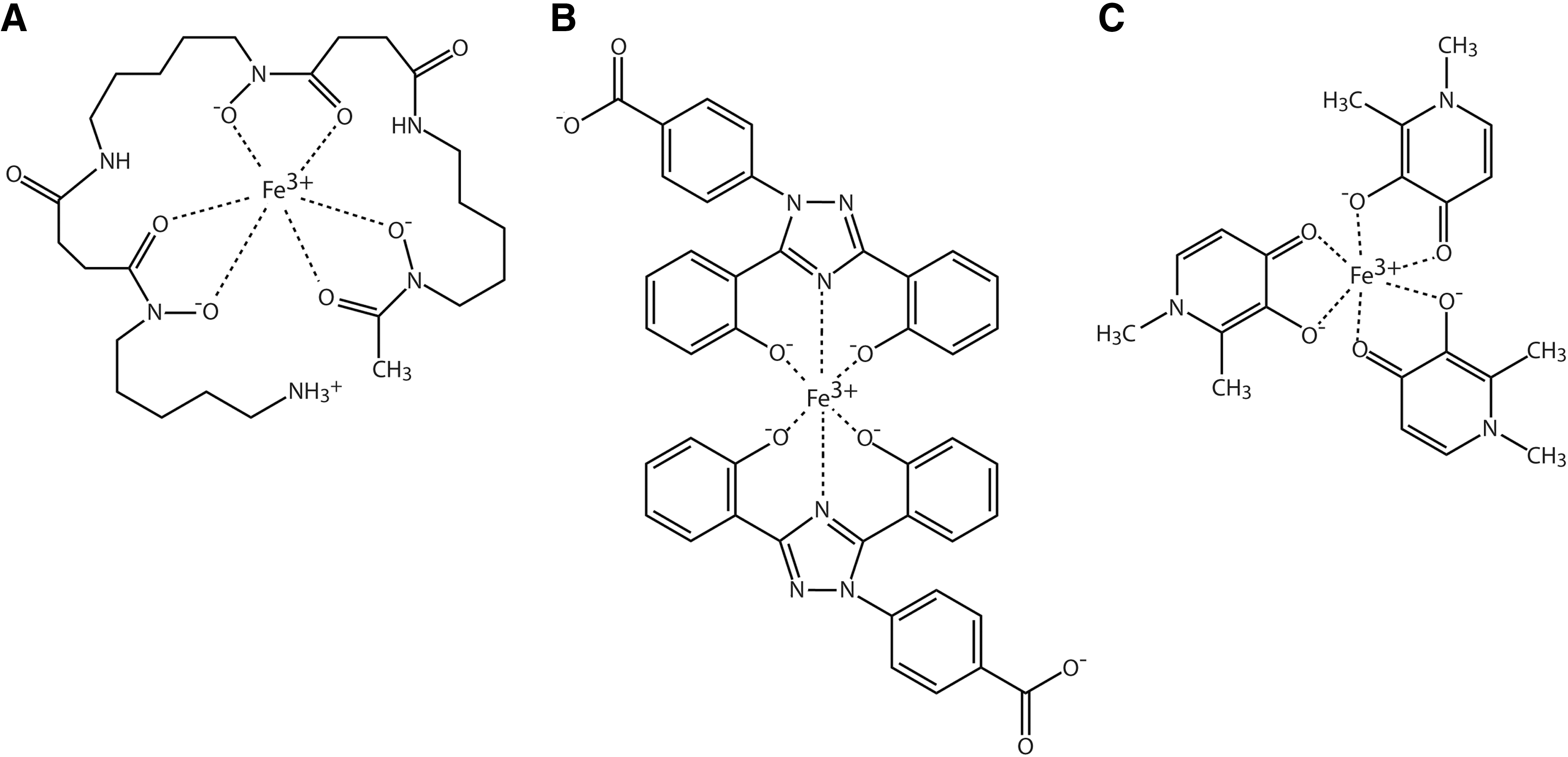
When complexed with iron, most iron chelators do not prevent the production of •OH or other reactive oxygen species (30). There are six coordination sites on the iron molecule that are either open or occupied by readily dissociable ligands, such as water, and each are capable of catalyzing free radical reactions (30). Exochelins and deferoxamine (also known as desferrioxamine) are hexadentate molecules that are capable of binding to all six iron coordination sites, thereby forming a one-to-one molecule binding relationship with iron and preventing free radical reactions involving the iron chelator complexes (30) (Figs. 1 and 5A). Other available iron chelators for clinical application are divalent or trivalent molecules: Although they can remove iron, their circulating iron complexes can only prevent free radical generation if there is a substantial surplus of chelator to the iron molecule such that two or more chelator molecules bind to each iron molecule (Fig. 5B, C). Deferoxamine does not readily enter cells, and toxicity limits its ability to remove substantial amounts of iron in clinical use (7, 38). Desferri-exochelins, unlike deferoxamine, are soluble in lipids (26, 27), a property that enhances their ability to enter cells rapidly, and consequently, they can remove intracellular free iron. In addition, probably since these siderophores have evolved over thousands of years to be nontoxic to human hosts of M. tuberculosis, exochelins have had little or no toxicity in pharmacological studies performed in vitro or in animals.
Exochelins as Probes for Iron-Mediated Reactions
Due largely to their unique ability to rapidly enter cells, exochelins have proved to be of exceptional experimental value as probes to understand the timing and extent of iron effects on cell cycle progression during normal and abnormal cell growth. In addition, their efficient blockade of catalytic sites has confirmed the importance and timing of iron-mediated oxidative toxicity during ischemia and reperfusion in acute cardiac events or organ transplantation.
Effect of Exochelins on Cell Cycle Progression in Vascular Smooth Muscle and Endothelial Cells
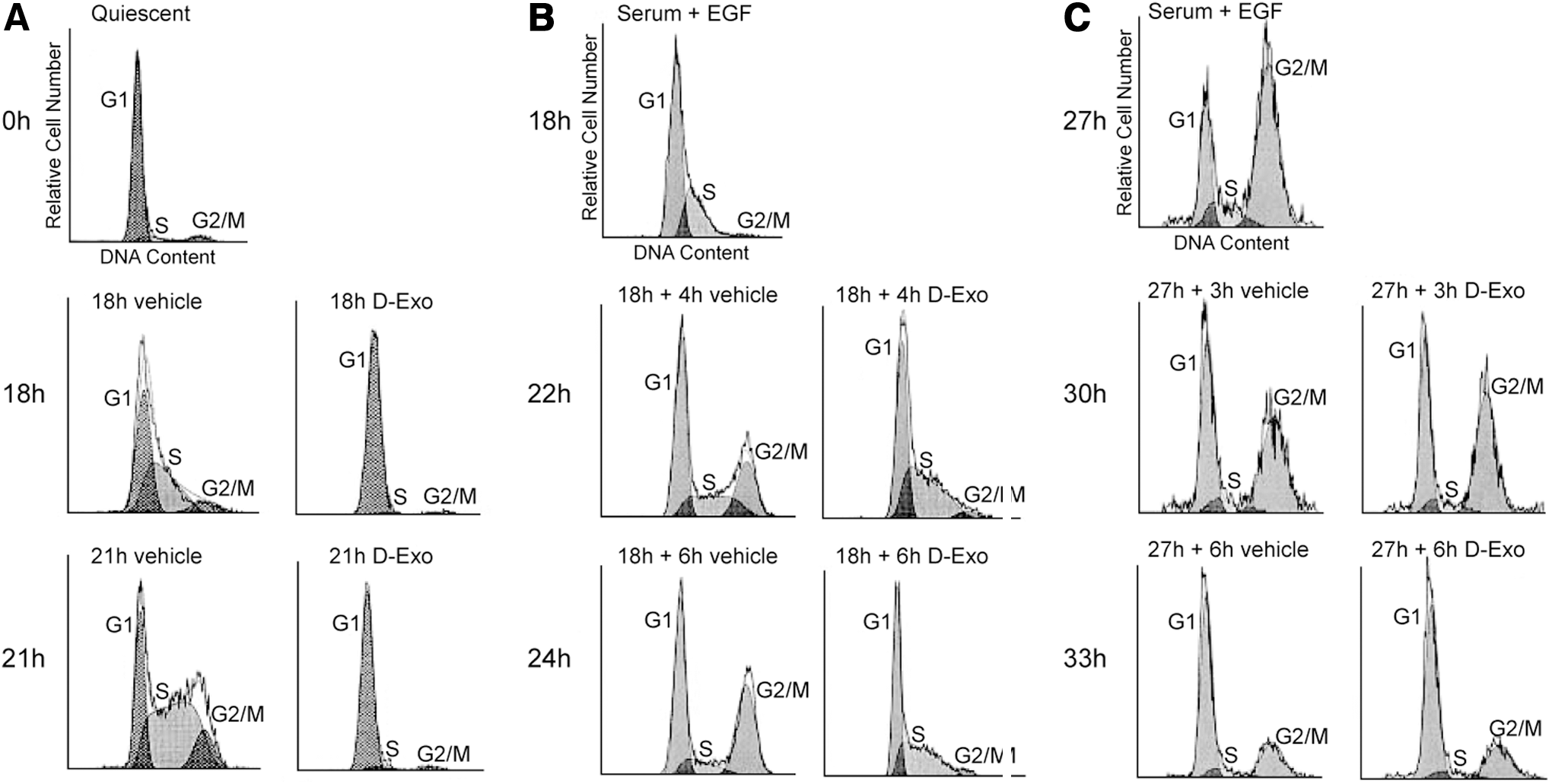
Vascular smooth muscle cells are usually quiescent, but when exposed to serum growth factors in cell culture they grow actively. In vivo, if the vascular endothelium that usually separates vascular smooth muscle from the blood is injured, the smooth muscle cells will proliferate due to exposure to circulating growth factors. In studies of cultured human vascular smooth muscle cells, growth factor-induced proliferation was blocked by desferri-Exochelin 772SM (46). Administration of serum growth factors alone induced progression of normal human vascular smooth muscle cells from the quiescent G0/G1 phase into S phase, initiating DNA synthesis; whereas co-administration of low concentrations of desferri-Exochelin 772SM with the serum growth factors prevented exit from G0/G1 (Fig. 6). When these cells were growth synchronized by the addition of growth factors after three days of quiescence and desferri-Exochelin 772SM added only during the S phase, the cells were growth arrested in S phase and did not enter the G2/M phase of cell division (46). However, administration of desferri-Exochelin 772SM to growth synchronized cells during the G2/M phase did not prevent progression to G0/G1. The blocks in the G0/G1 and S phases were reversible when the cultured cells were washed after 24 h of desferri-Exochelin 772SM exposure and incubated in fresh medium containing a growth factor (46). Studies of uptake of radioactive thymidine, uridine, and leucine during synchronized cell growth demonstrated that the normal high uptake of radioactive thymidine during S phase and the normal uptake of radioactive uridine in G1 and S phase are blocked by desferri-Exochelin 772SM, but uptake of radioactive leucine is unaffected (46). Therefore, with normal human vascular smooth muscle cells, chelation of iron with a desferri-exochelin reversibly inhibits DNA replication in S phase and RNA transcription in both the G0/G1 and S phases, but has no effect on protein synthesis.
In growth factor-stimulated cultured human vascular smooth muscle cells, administration of desferri-Exochelin 772SM down-regulated the cyclins and cyclin-dependent kinases (CDKs) that mediated progression from the quiescent G0/G1 phase into the S phase, and from the S phase into the G2/M phase (46). Specifically, there was down-regulation of cyclins E and A and CDK 2 activity. This was due to iron chelation, as iron-loaded desferri-Exochelin 772SM had no effect on cell cycle progression.
Desferri-Exochelin 772SM had similar effects on cell cycle progression due to growth factor stimulation in cultured human umbilical vein endothelial cells (HUVECs) to those observed in human vascular smooth muscle cells, with block of cell cycle progression in G0/G1 and S phases (32). In both HUVECs and human vascular smooth muscle cells, the desferri form of Exochelin 772SM induced hypoxia-inducible factor-1α (HIF-1α). The induction of HIF-1α was much greater and more sustained in the smooth muscle cells than in the HUVECs (33). In the smooth muscle cells, but not in the HUVECs, there was downstream activation of vascular endothelial growth factor (VEGF), a potent stimulus of endothelial cell growth. It would appear that iron chelation with a desferri-exochelin can directly inhibit endothelial cell growth but paradoxically offers a means of enhancing endothelial cell growth by inducing release of VEGF from adjacent vascular smooth muscle cells through the HIF-1α pathway.
Desferri-Exochelins Reversibly Inhibit the Growth of Normal Breast Epithelial Cells but Kill Breast Cancer Cells Via Apoptosis
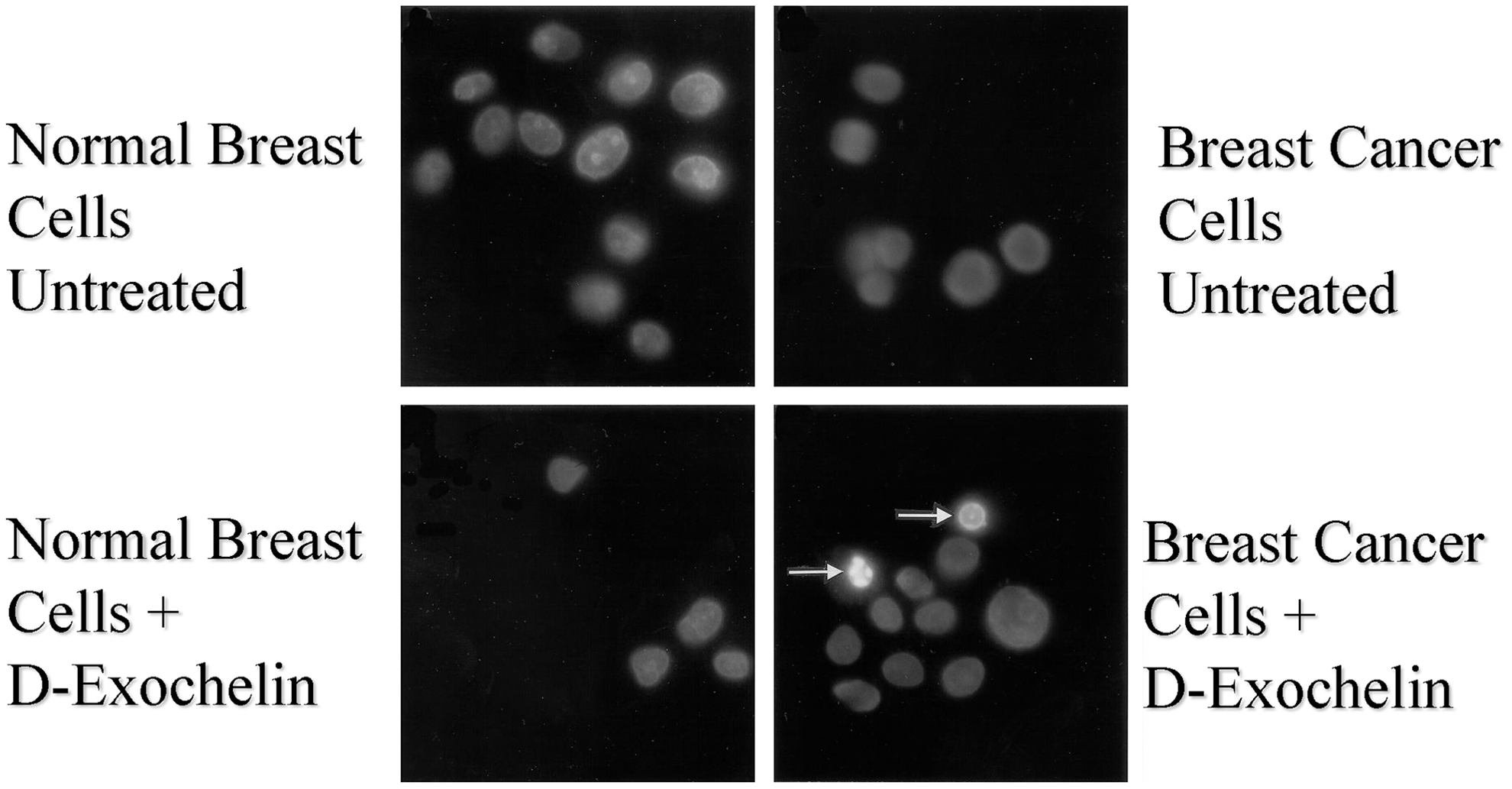
When desferri-Exochelin 772SM was added to cultures of human breast cancer cells and normal human breast epithelial cells (44), it reversibly inhibited cell growth in the normal epithelial cells but killed two strains of human breast cancer cells (MCF-7 and T47D) by apoptosis (Fig. 7). Since iron-loaded Exochelin 772SM had no growth inhibiting or toxic effects on any of the cell types, iron chelation was the mechanism of action with the desferri-Exochelin. When the T47D breast cancer cells, which have functional progesterone receptors, were growth arrested with the progesterone agonist R5020, exposure to desferri-Exochelin 772SM did not cause apoptosis. When R5020 was removed and cell growth resumed, the T47D cells died on exposure to desferri-Exochelin 772SM. Similarly, transient growth arrest of the MCF-7 cells with an anti-estrogen protected them from desferri-Exochelin 772SM until the anti-estrogen mediated growth arrest waned, at which point exposure to desferri-Exochelin 772SM caused cell death. Therefore, iron chelation with desferri-Exochelin 772SM selectively kills proliferating breast cancer cells without damaging either normal cells or quiescent cancer cells.
Studies of MDA-MB-468 cells derived from a metastatic breast adenocarcinoma revealed that desferri-Exochelin 772SM induces increased expression of HIF-1α and HIF-2α, with downstream activation of the proapoptotic protein NIP3 (nineteen kDa interacting protein-3, also known as BNIP3) and VEGF (12). Hypoxia, which is present in many tumors, as well as iron chelation, increases expression of HIF-1α, which, in turn, up-regulates NIP3, which damages mitochondrial permeability (54). The increased expression of NIP3 is a plausible explanation for the apoptosis in this in vitro preparation. However, the situation is more complex in vivo where tumor characteristics vary widely. In some cases, concomitant up-regulation of the angiogenic protein VEGF may enhance tumor growth, because the resultant increased vascularity decreases intratumor hypoxia (55). Therefore, further studies of individual tumors in vivo would be necessary to assess whether the chelator-induced NIP3 activation is an important mechanism potentially applicable to clinical use.
In the experiments described earlier, in both normal breast cells and breast cancer cells, the growth inhibitory effect of iron chelation with lipid-soluble desferri-Exochelin 772SM, which readily enters cells, was ∼10-fold greater and considerably more rapid than the effect of iron chelation with the lipid-insoluble, poorly diffusible, iron chelator deferoxamine. In a separate study (31), inhibition of the iron-containing enzyme ribonucleotide reductase in cultured MCF-7 cells was 10-fold greater with desferri-Exochelin 772SM than with deferoxamine.
Effects of iron chelation with desferri-Exochelin 772SM on cyclin and CDK binding were studied in MCF-7 and normal human breast epithelial cells (45). Binding of specific cyclins to CDKs enables cell cycle progression. In co-immunoprecipitation experiments in actively growing normal breast cells, binding of both cyclin A and cyclin E to CDK 2 was inhibited by desferri-Exochelin 772SM. However, in the MCF-7 cells, binding of cyclins A and E to CDK 2 was markedly enhanced. As a result, CDK 2 activity was suppressed in the normal cells but increased in the cancer cells by exposure to desferri-Exochelin 772SM. In the cancer cells, there were marked and sustained increases in HIF-1α and p53. The p53 cell tumor suppressor protein regulates multiple cell cycle checkpoints and can induce either cell cycle arrest or apoptosis. A hypothesis compatible with these data is that in breast cancer cells, iron chelation stabilizes HIF-1α, which activates p53 (45). The overexpression of p53, which in these cells is defective, induces apoptosis rather than cell cycle arrest. However, studies of iron depletion with other chelators in cancer cells have invoked other targets not necessarily involving p53 (46). These include altered expression of the iron regulatory proteins IRP1 and IRP2 (59), altered post-transcriptional regulation of the CDK inhibitor p21CIP1,WAF1 leading to cell cycle arrest (20), and inhibition of topoisomerase IIα causing DNA strand breaks (49). More information is needed to determine whether these or other targets are crucial mechanisms of iron deprivation-mediated apoptotic cell death in cancer cells.
Desferri-Exochelins Prevent Oxidative Injury from Ischemia and Reperfusion
A well-established mechanism of injury after reoxygenation is the generation of reactive oxygen species. Although hydrogen peroxide and superoxide radical are generated during ischemia and reperfusion, they are relatively weak oxidants that are readily neutralized by endogenous scavenger enzymes. However, hydrogen peroxide (H2O2) and superoxide radical (•O2) can participate in iron-catalyzed reactions that generate hydroxyl radical (•OH), a highly reactive molecule for which there is no effective endogenous defense, and possibly other deleterious oxygen-derived radicals as well (43). The ability of lipophilic desferri-exochelins to rapidly enter cells and block all the catalytic sites of the iron molecule has offered a unique opportunity to assess the role of •OH and other iron-dependent free radicals in ischemia and reperfusion.
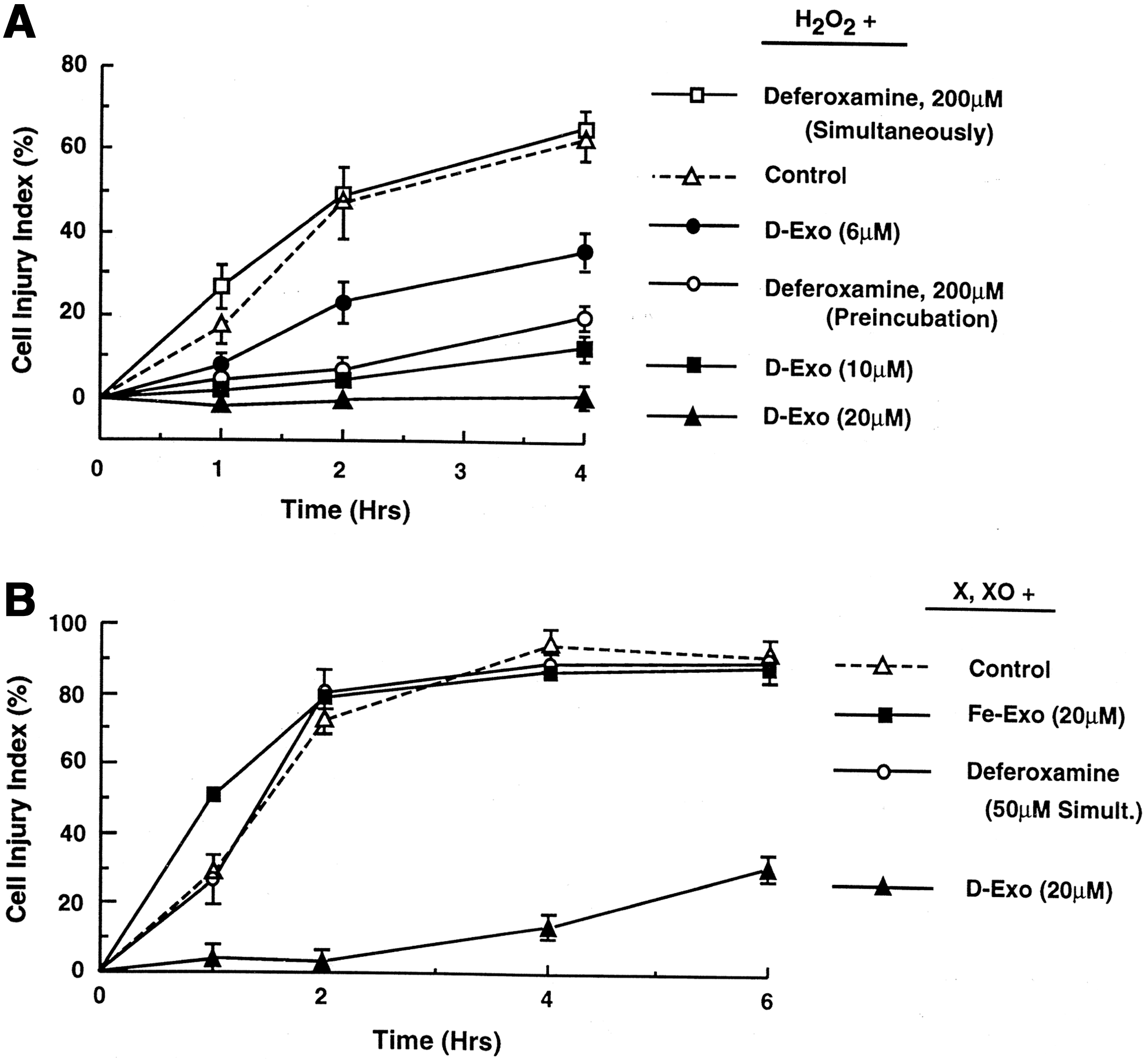
Desferri-exochelins block the generation of •OH in a cell-free •OH-generating system consisting of xanthine and xanthine oxidase, which produce H2O2 and •O2 if iron is available (34) (Fig. 8). The lipid-soluble desferri-exochelins also prevent oxidative injury in cardiac myocytes exposed to H2O2 and •O2, in the presence of iron; whereas under the same conditions, the water-soluble but lipid-insoluble iron chelator deferoxamine had little or no effect (34). The rapidity with which desferri-exochelins block oxidative injury in cultured cardiac myocytes is a function of their lipid solubility, which, in turn, is dependent on their polarity: Highly polar desferri-exochelins act slowly whereas relatively nonpolar desferri-exochelins, such as desferri-Exochelin 772SM, act rapidly. The lipid-insoluble iron chelator deferoxamine can decrease myocardial infarct size in an animal model of ischemia-reperfusion if given before ischemia begins, but is ineffective if given during reperfusion (40). Desferri-Exochelin 772SM reduces ischemia-reperfusion injury in a rabbit model of acute myocardial infarction (35) and in a rat model of liver transplantation (1). In these models of oxidative injury, desferri-exochelins prevent iron-mediated production of •OH, and possibly other highly toxic oxidative products, and preserve levels of the endogenous antioxidant glutathione. Therefore, these studies establish that the binding of iron to exochelins prevents or reduces oxidative injury.
Potential Clinical Utility of Desferri-Exochelins
The unique properties of desferri-exochelins, in particular their water and lipid solubility, hexadentate structure, and low toxicity, offer potential for the treatment of various disease states where iron plays a potentially harmful role. Our animal models support potential value in several areas, including organ damage due to iron overload, excessive smooth muscle cell proliferation associated with restenosis after coronary artery angioplasty, cancer, and oxidative injury associated with ischemia and reperfusion (Fig. 9).
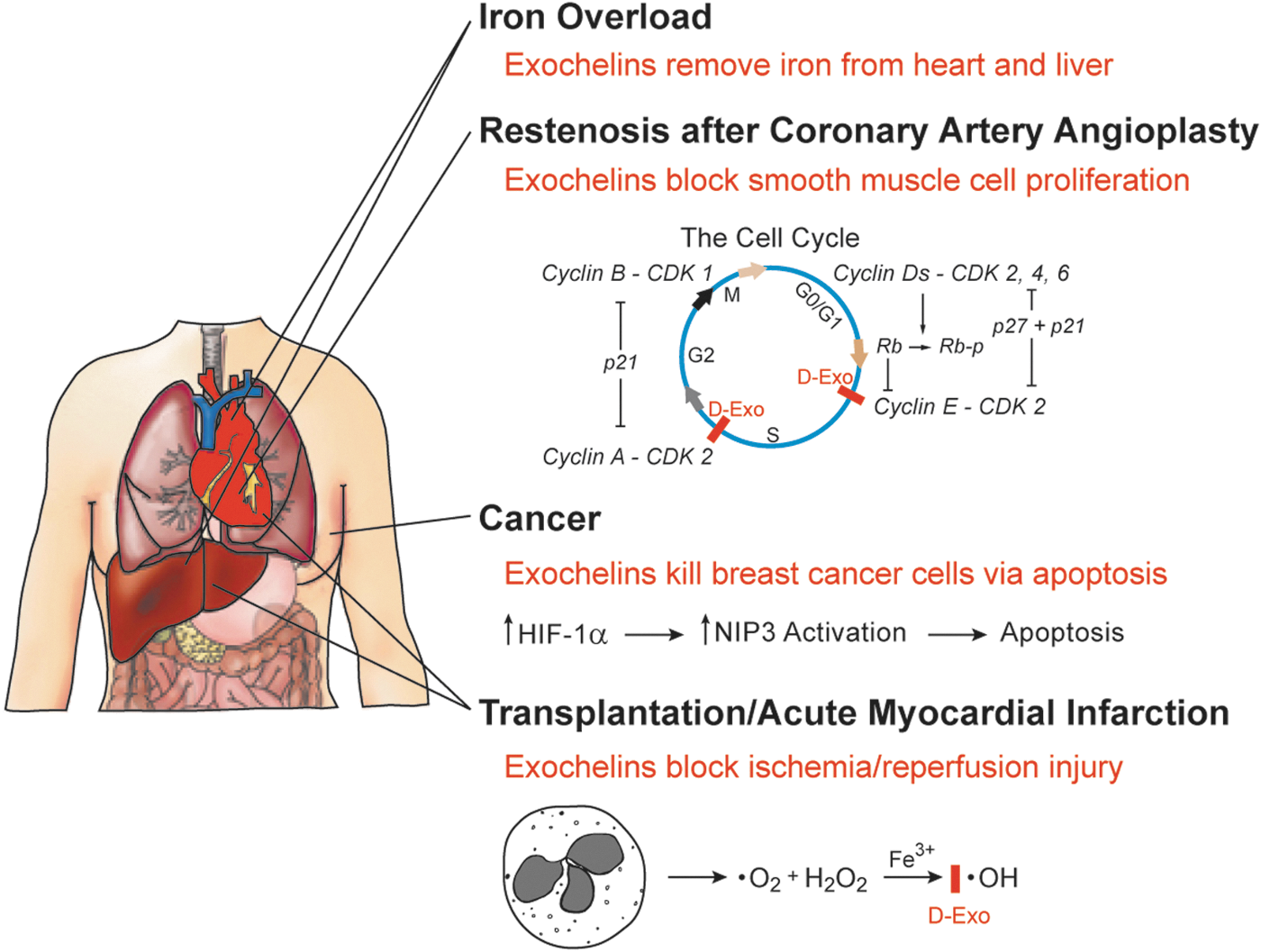
Iron overload
Currently, the most promising potential clinical utility of desferri-exochelins is for the treatment of iron overload. Anemias that require frequent transfusions cause progressive accumulation of iron released by hemoglobin over a period of years until toxic levels are reached. Patients with β-thalassemia, one of the most common of these anemias, will die by age 30 of heart failure, liver failure, or other complications if they are not treated with an iron chelator (6, 7, 38). Iron-induced cardiomyopathy and liver disease are major complications in patients with sickle cell disease who require frequent transfusions.
For many years, the major treatment available for iron overload due to transfusions has been deferoxamine, a siderophore derived from cultures of Streptomyces pilosus. The hexadentate structure of deferoxamine binds all sites that catalyze potentially toxic iron-mediated oxidative reactions (Fig. 5A). There is evidence that deferoxamine lengthens life span and reduces complications in patients with thalassemia (17). However, deferoxamine is poorly absorbed orally, and is usually administered in low doses by slow 12 h subcutaneous or intravenous infusions administered 4 to 6 days a week. Hypotension precludes larger, more rapid infusions. Other serious side effects include pulmonary, neural, or renal toxicity and growth retardation (6, 7, 38). Due to the onerous nature of the infusions, patient compliance has been poor.
Two iron chelators suitable for oral administration have been recently approved for clinical use in the United States. Deferiprone, a bidentate molecule (Fig. 5C), has serious side effects, including agranulocytosis, neutropenia (below normal number of neutrophils in the circulating blood), arthropathy, and gastrointestinal disorders. In addition, whether it is capable of removing adequate amounts of excess iron to be clinically effective has been questioned (37). Deferiprone has a very short half life and requires dosing thrice a day.
Desferasirox (Exjade) is a tridentate molecule (Fig. 5B). The evidence in support of desferasirox for iron chelation is that, in patients with thalassemia, the amount of iron in target organs did not increase over the time it was studied. Whether desferasirox results in clinically effective removal of iron from tissue is not clearly established. Serious drug-induced side effects, including abnormalities in liver and kidney function and gastrointestinal disorders, have been reported (47, 60). With both desferasirox and deferiprone, a theoretical concern is that circulating iron-chelator complexes may catalyze oxidant injury.
As already noted, exochelins are both lipid and water soluble. We have preliminary information that oral administration is feasible. However, unlike compounds that are soluble in lipids but not in water, exochelins can be readily administered parenterally in solutions. Exochelins are remarkably nontoxic, perhaps reflecting the fact that M. tuberculosis has evolved over millennia so as to be capable of quietly persisting for years and even decades in a latent state within humans. By evolving to be nontoxic, exochelins avoid triggering reactions that could cause rejection of the tuberculosis bacillus by its human host. As a result, when administered to animals either orally or parenterally, desferri-exochelins are tolerated in high doses and to date have caused no apparent toxicity. As described next, after administration, the desferri-exochelins are widely distributed in organs where they remove iron that is subsequently excreted into the urine and feces. Similar to deferoxamine, exochelins are hexadentate iron chelators so that circulating iron-chelator complexes cannot catalyze adverse oxidative reactions.
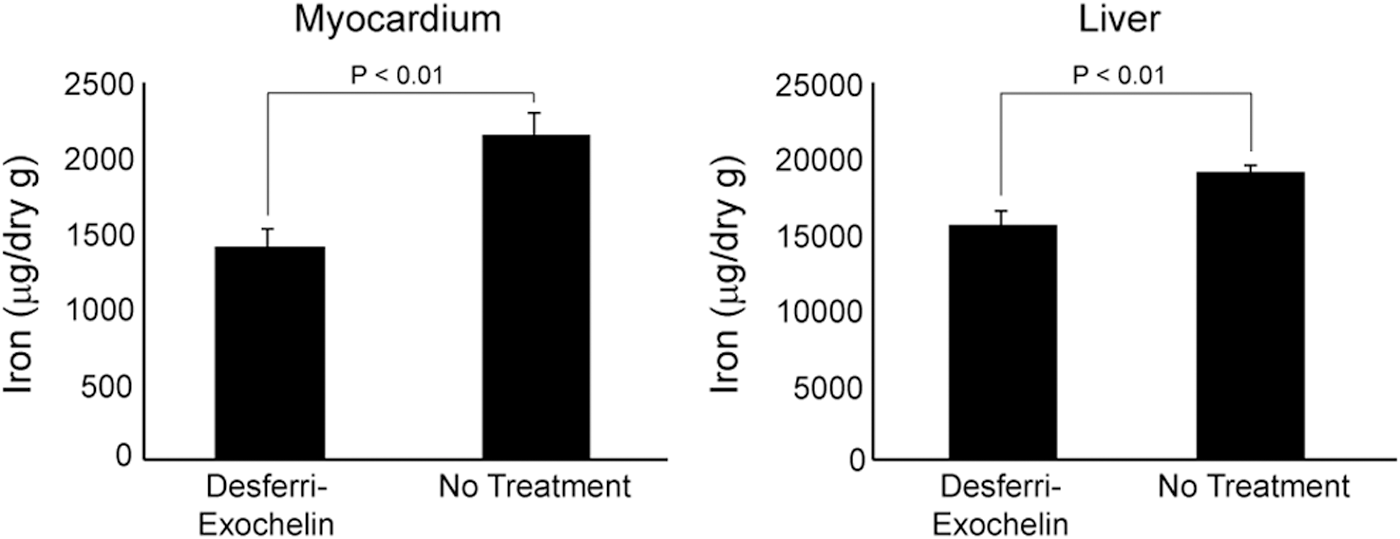
In iron-overloaded mice, desferri-exochelins remove iron from the heart and liver, the major targets of excess iron (Fig. 10). When iron-overloaded mice were treated with desferri-Exochelin 772SM intraperitoneally, four times weekly for 8 weeks, iron levels in the heart and liver, measured by atomic absorption spectrophotometry, were significantly reduced by 25% and 20%, respectively, compared with vehicle-treated or -untreated iron-overloaded mice (33). In iron-overloaded mice treated with desferri-Exochelin 772SM, early excretion of iron into the urine was observed after an injection. In these mice, exposed to high doses of iron for only 5 days before treatment, there was no evidence of left ventricular dysfunction on echocardiographic studies and no histological abnormalities other than interstitial iron accumulation in the heart, liver, and spleen of iron-overloaded mice. The relatively short period of iron exposure may explain the lack of iron-mediated dysfunction in the iron-loaded mice, as patients with transfusion-dependent thalassemia usually do not develop cardiac problems until they are exposed to iron overload from transfusions for many years. Patients with β-thalassemia typically receive blood transfusions every 4–6 weeks. Since infusion of even large quantities of desferri-Exochelin 772SM appears to be nontoxic, it may be feasible to effectively treat patients with thalassemia using intravenous infusions of a desferri-exochelin at the same time that they receive blood transfusions; that is, it is theoretically feasible to treat them with sufficient desferri-Exochelin 772SM to remove a quantity of iron greater than that received with the blood transfusion. This would ensure compliance, and this intermittent use is possibly less likely to interfere with normal iron-mediated effects than the daily exposure required for oral administration.
The recent clinical approval of the oral iron chelators deferiprone and desferasirox, whether administered individually or in combination, has increased options for treatment of chronic iron overload. However, all treatment regimens using these drugs have serious side effects (7, 36 –38). In addition, convincing proof of the long-term efficacy of these drugs in improving outcomes is lacking (37). Whether the new oral agents will result in substantially better patient compliance than has been the case with deferoxamine is also not established. Oral administration of drugs does not necessarily achieve better compliance than parenteral administration, particularly when parenteral drugs require only infrequent administration. For example, in the case of secondary rheumatic fever prophylaxis in age groups similar to those in thalassemia, daily oral dosing with penicillin had poorer compliance than monthly intramuscular injections (25). Therefore, the development of better agents for long-term treatment of chronic iron-overload conditions continues to be a matter of considerable clinical importance.
Distribution of desferri-exochelins after parenteral or oral administration
When administered intravenously or orally, desferri-exochelins rapidly enter host organs throughout the body (33). In rats administered tritium-labeled desferri-Exochelin 772SM intravenously, counts were obtained within 15 min in the heart, lung, thymus, liver, spleen, stomach, small and large intestines, kidney, bladder, testes, brain, skull bone, skeletal muscle, and abdominal fat. Peak counts occurred in all organs between 15 and 60 min, and the counts gradually subsided thereafter. In the heart and liver, two major target organs in iron-overload conditions, 35% and 20%, respectively, of the peak levels were present at 4 h, and 8% and 9%, respectively, of the peak levels were present at 24 h. Relatively high counts were measured in the small intestine, a finding compatible with biliary excretion of iron-chelator complexes. Counts in blood rapidly disappeared during the first hour post injection. Substantial counts were measured in the urine within the first 4 h after an injection of desferri-Exochelin 772SM, an observation consistent with urinary clearance.
In rats that received tritium-labeled desferri-Exochelin 772SM administered by gavage, radioactivity accumulated within 4 h in the same organs where this occurred after parenteral administration (33). Counts subsequently declined in all organs, except the large intestine, by 24 h. The intestinal iron levels, as already noted, probably reflect hepatic excretion of iron-chelator complexes through the bile. Substantial counts were detected in the urine and feces collected over the 24 h after the administration of desferri-Exochelin 772SM. Therefore, with parenteral or oral administration, the lipid-soluble desferri-exochelins are distributed among a wide spectrum of organs and remove iron, which is then excreted in the urine and feces.
Restenosis
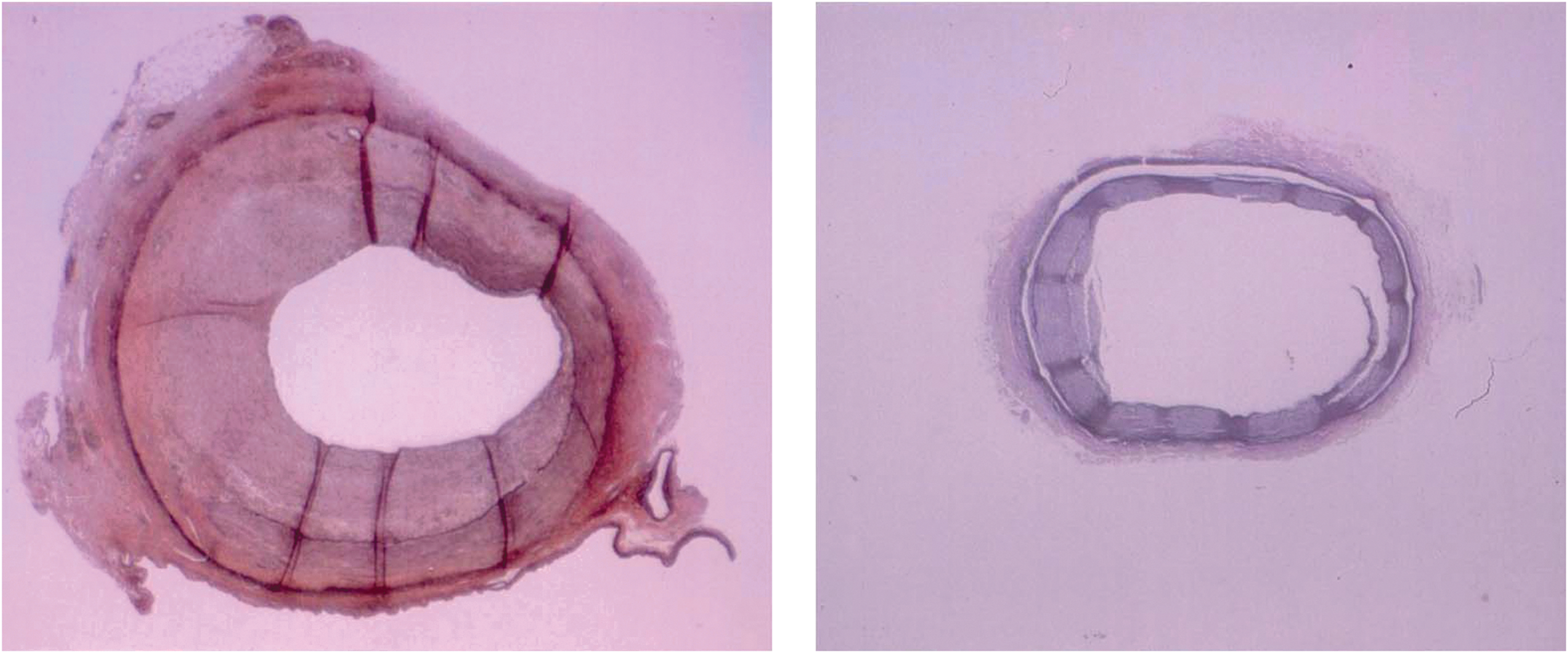
Since desferri-exochelins inhibit the proliferation of normal cells without killing them, that is, the inhibitory effect is reversible, they may have potential therapeutic use in disease states which are characterized by abnormal cell growth. One such disease entity is restenosis, a renarrowing of the coronary artery after coronary artery angioplasty, a widely used treatment for obstructive atherosclerotic coronary lesions. The performance of a balloon angioplasty involves inserting a catheter into an obstruction due to atherosclerosis and then inflating a balloon attached to the catheter, thereby relieving a partial obstruction and re-establishing blood flow to ischemic regions in the heart. Restenosis, characterized by abnormal growth of vascular smooth muscle cells into the vessel lumen to the point where there is recurrence of vascular obstruction, is a common complication of balloon angioplasty. The potential usefulness of desferri-exochelins for prevention of restenosis that is associated with coronary artery angioplasty was tested in a pig model (52). Anesthetized pigs subjected to overstretch coronary artery injury with coronary angioplasty balloons develop stenosis within a month. Thirty-two juvenile domestic swine underwent overstretch injury, accomplished using a balloon attached to a catheter; the balloon was inflated to a size that exceeded the size of the coronary artery. The pigs were randomized to receive either 0.5 mg of desferri-Exochelin 772SM or an equal volume of vehicle (0.09% saline), administered via a specialized catheter for a local intramural coronary artery injection at the injured site (50). Twenty-four pigs, 15 treated with desferri-Exochelin 772SM and 9 treated with placebo, survived for one month and met technical criteria for catheter-induced disruption of the internal elastic lamina of the arterial wall. Such disruption is necessary to induce abnormal vascular cell growth and local restenosis in animal models (50). Histological analysis using an injury score by two investigators blinded as to the treatment group concluded that desferri-Exochelin 772SM injected at the angioplasty site immediately after balloon inflation caused an absolute reduction in the degree of vascular injury present one month later (52). Typical examples of a desferri-Exochelin 772SM-treated and a vehicle-treated coronary artery are shown in Figure 11.
Cancer
The finding in cell cultures that desferri-exochelins selectively kill cancer cells via apoptosis but only reversibly inhibit the growth of normal cells prompted a study of their potential role in the treatment of cancer (44). However, when desferri-Exochelin 772SM was administered systemically in rodent models of cancer, the results were disappointing (L.D. Horwitz, unpublished studies). Cancers in humans and animals have extraordinary ability to mobilize iron, especially through release of transferrin and induction of increased transferrin receptor number in target cells, and this may maintain iron availability despite administration of an exogenous chelator. Possibly, local administration of desferri-exochelins in selected settings or co-administration of a desferri-exochelin with other anti-neoplastic agents could have greater efficacy. Alternatively, as discussed earlier, increased expression of VEGF in response to iron chelation may actually enhance growth of some tumors in vivo.
Oxidative injury: acute myocardial infarction and transplantation
Since desferri-exochelins have been shown to block oxidative injury occurring in association with ischemia and reperfusion, they have potential utility for the treatment of disease states where oxidative injury plays a prominent role. These include acute myocardial infarction and organ transplantation.
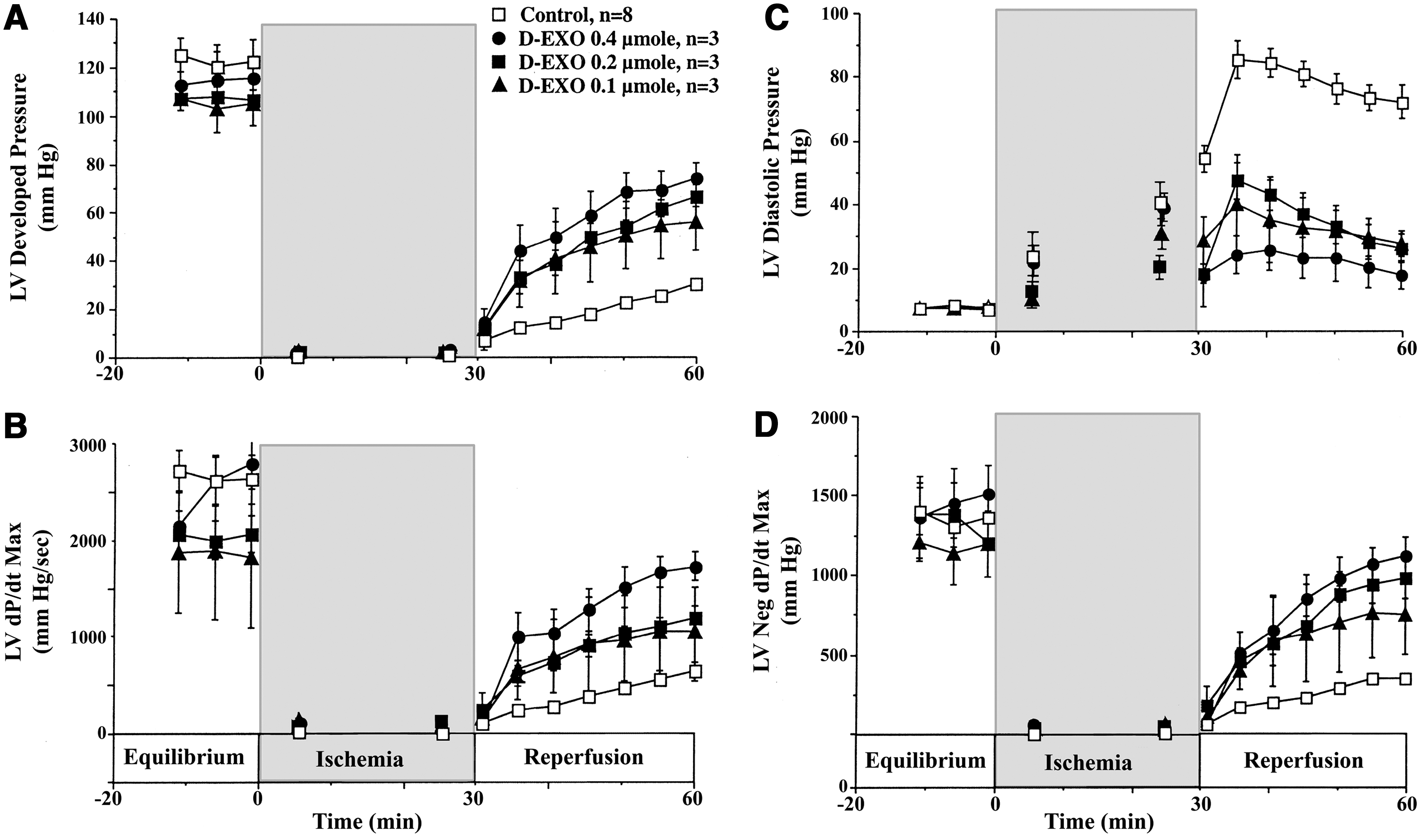
The emergency treatment of acute myocardial infarction involves opening blocked coronary arteries either by angioplasty or by dissolving an obstructive clot with infusion of a thrombolytic agent (“clot buster”). Paradoxically, while this corrects the ischemic phase, it initiates a cascade of molecular and cellular events that result in additional damage to myocardial tissue primarily from reactive oxygen species. In a study of isolated rabbit hearts subjected to a period of ischemia, desferri-exochelins infused during reperfusion at the root of the aorta, where the chelator entered the coronary arteries, dramatically improved systolic and diastolic left ventricular function and preserved coronary blood flow (35) (Fig. 12). Desferri-exochelin treatment also reduced the release of a cardiac enzyme, lactic dehydrogenase, a measure of cardiac cell death, and reduced the concentration of •OH metabolites in the myocardium. Therefore, iron-mediated reactions are an important component of myocardial injury due to ischemia and reperfusion, raising the possibility that in clinical settings the extent of the injury could be limited by iron chelation with an exochelin. There is considerable clinical and experimental evidence that the greater the size of a myocardial infarct, the worse the outcome.
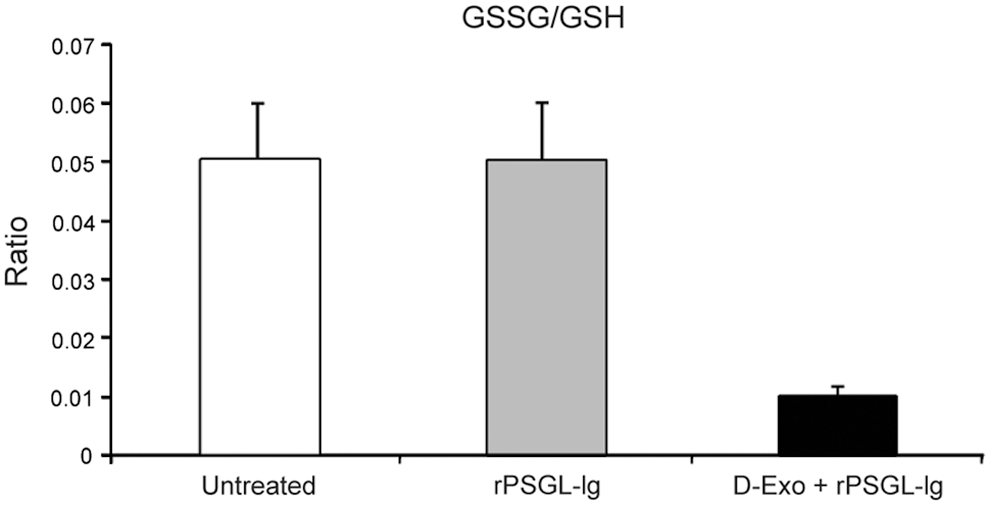
Oxidative injury also accompanies reperfusion of transplanted organs. In liver transplantation, ischemia—reperfusion injury causes early and late failure of the donor liver. Desferri-exochelins have been studied for their capacity to protect against ischemia-reperfusion injury in an ex vivo rat model in conjunction with recombinant P-selectin glycoprotein ligand immunoglobulin (rPSGL-Ig), an inhibitor of neutrophil adhesion (1). Livers treated with desferri-Exochelin 772SM and rPSGL-Ig had significantly higher blood flow than livers treated with rPSGL-Ig alone, and both treatment groups had higher blood flow than untreated controls. Organs treated with both desferri-Exochelin 772SM and rPSGL-Ig, but not with rPSGL-Ig alone, had significantly reduced levels of carbonyl proteins, a protein oxidation product, significantly elevated levels of reduced glutathione (GSH) compared with untreated controls, and a significantly lower ratio of oxidized glutathione (GSSG) to GSH (Fig. 13). Finally, livers treated with desferri-Exochelin 772SM and rPSGL-Ig had essentially normal architecture and no evidence of necrosis, whereas untreated control livers had marked disruption of normal liver architecture and extreme hepatic necrosis. Therefore, co-administration of a desferri-exochelin with another suitable agent may be of value in improving outcomes in liver, or other organ, transplantation.
Conclusions
Better iron chelators are needed to treat a variety of iron-dependent pathological disorders. Current clinically available iron chelators suffer from being either lipid insoluble (deferoxamine), and therefore not able to rapidly cross cell membranes and chelate iron at key intracellular sites, or toxic (deferiprone and desferasirox), inducing serious side effects. The bidentate or tridentate structure of deferiprone and desferasirox may contribute to their toxicity, as such molecules cannot bind iron at all six sites that are capable of catalyzing free radical reactions unless two or more chelator molecules bind to a single iron molecule. Exochelins are uniquely lipid-soluble and hexadentate iron chelators with the capacity both to enter cells rapidly and to chlelate intracellular iron and prevent iron from participating in reactions generating reactive oxygen species. Consequently, the exochelins have considerable potential for the treatment of a number of disease entities in which iron plays a central role in pathogenesis. In addition, the exochelins can be exploited as probes to unravel the role of iron in normal physiologic processes and in iron-dependent pathological states. In intact animal or isolated heart models, exochelins have been demonstrated to have promise for the treatment of transfusion-related iron overload, restenosis after coronary artery angioplasty, cancer, and oxidative injury associated with acute myocardial infarction and transplantation.
Footnotes
Acknowledgments
The work described in this review was supported by NIH grants AI33790 (M.A.H.), AI35725 (M.A.H.), HL077000 (M.A.H.), and HL55291 (L.D.H.). The authors thank Barbara Jane Dillon and Juyao Dong for assistance with graphics.
Author Disclosure Statement
Both authors are major stockholders of Cardioceutics, Inc., which includes exochelins in its product portfolio.
